Abstract
Objective
To investigate the relationship between glutathione S-transferase enzyme (GSTM1, T1, and P1) genetic variants and semen quality in men with idiopathic infertility.
Methods
Sperm characteristics were measured using computer-assisted sperm analysis. The malondialdehyde (MDA), nitric oxide (NO), and total antioxidant capacity (TAC) activities were detected by spectroscopic analysis, and 8-hydroxy-2′-deoxyguanosine (8-OHdG) was detected by enzyme-linked immunosorbent assay.
Results
This study included 246 idiopathic infertile men and 117 controls. The GSTM1(−), T1(−), and M1/T1(−/−) genotype frequencies significantly differed between the groups. The GSTM1(−) and T1(−) genotypes in idiopathic infertile men negatively correlated with sperm concentration, motility, mitochondrial membrane potential, and other parameters. However, these genotypes positively correlated with the amplitude of the lateral head displacement and NO and 8-OHdG levels. The GSTT1(−) genotype positively correlated with mean angular displacement and MDA activity. GSTM1(−) and T1(−) had a synergistic effect on semen quality. Sperm motility, normal morphology, straightness, and TAC were lower and amplitude of lateral head displacement and MDA were higher in the GSTP1(A/G + G/G) group than in the GSTP1(A/A) group among men with idiopathic infertility.
Conclusions
GSTM1, T1, and P1 genetic variants may be risk factors for infertility by affecting the semen quality men with idiopathic oligoasthenospermia.
Keywords
Introduction
Male infertility is a complex condition observed globally that is defined as the failure of a couple to achieve pregnancy after at least one year of unprotected, regular sexual intercourse. This condition is estimated to affect 10% to 15% of couples.1–3 In these cases, the only abnormality is observed by semen analysis, and this phenomenon occurs in approximately 30% of all male infertility cases.4–6 Moderate reactive oxygen species (ROS) production is essential for sperm–oocyte fusion and normal sperm function, but excessive ROS generation can lead to oxidative stress (OS). This may result in damage to sperm DNA and impair fertilization.7,8
Glutathione S-transferases (GSTs) comprise a superfamily of ubiquitously expressed multifunctional enzymes that play very important roles in phase II cellular detoxification and bioactivation reactions, as well as in protecting cells against OS. Therefore, these enzymes have been considered antioxidant enzymes. The GSTM1, T1, and P1 genes are located on chromosomes 1p13.3, 22q11, and 11q13, respectively.9,10 GSTM1, T1, and P1 variants can affect the binding affinity or cellular activity of these enzymes. The genotype notations for GSTM1/T1 are (−/−) for homozygotes, (+/−) for heterozygotes, and (+/+) for wild-type. The genotype notations for GSTP1 are (G/G) for homozygotes, (A/G) for heterozygotes, and (A/A) for wild-type.11,12 Genetic variants in GST genes may influence the activity of these enzymes and further disturb the balance of the detoxification system, thereby increasing individual host susceptibility to OS damage and possibly lead to male infertility.13–15
Research has shown that men in Iran or India with the GSTM1(−) and/or GSTT1(−) genotypes displayed an increased risk of developing infertility.16,17 The GSTT1(−) genotype was related to a reduced sperm count and concentration in semen in men from the USA. 18 Our past studies found that the GSTT1(−) genotype led to a predisposition to sporadic idiopathic oligospermia or azoospermia, and that the GSTM1(−) and GSTT1(−) genotypes may lead to increased oxidative damage in sperm in male infertility cases associated with varicoceles.19–21
The purpose of this study was to prospectively examine the effect of metabolic enzyme gene polymorphisms on semen quality in patients with idiopathic male infertility.
Materials and methods
Subjects
The present case–control study included male patients diagnosed with primary idiopathic infertility from November 2014 to December 2018 in the First Affiliated Hospital of the Medical College of Xi’an Jiaotong University. These diagnoses were based on the failure to conceive after 1 year of unprotected intercourse and abnormal semen parameters according to the guidelines of the World Health Organization (WHO). 22 The patients were required to provide evidence that their wives were healthy and had no specific characteristics associated with delayed conception. Healthy men whose wives had a history of giving birth in the previous 2 years served as the control group. All individuals provided written informed consent prior to the study. Participants were instructed to complete a brief questionnaire addressing lifestyle variables, any history of disease, and age. According to the medical records and chromosomal examinations, men with varicoceles, sex chromosomal aneuploidy/mosaicism, or Yq microdeletion were excluded. Patients were excluded from this research if they reported drug consumption, excessive alcohol intake (defined as drinking more than 50 mL of alcohol per day for at least 6 months), smoking, urogenital infections, hypogonadism, or leukocytospermia. 20 This research was approved by the Ethics Committee of the First Affiliated Hospital of the Medical College of Xi’an Jiaotong University (Trial registration: Current Controlled Trials ChiCTRIPR14005580, 23 November 2014).
Sperm collection and analysis
Semen samples were collected by masturbation after sexual abstinence for 3 to 5 days. Leukospermic or viscous semen was excluded. Sperm characteristics were measured by computer-assisted semen analysis (CASA) (WLJY-9000, Weili New Century, Beijing, China) for all participants. Spermatozoa were separated by centrifugation at 300 × g for 10 minutes, and seminal plasma was used for biochemical assays. 20
GST gene variants
An AxyPrep TM Genomic DNA Miniprep Kit (Axygen Biosciences, Union City, CA, USA) was used to isolate genomic DNA from blood samples. The GSTM1 and GSTT1 genotypes were identified by multiplex polymerase chain reaction (PCR) using published primer sequences as follows: GSTM1 (forward, 5′-GAA CTC CCT GAA AAG CTA AAG C-3′; reverse, 5′-GTT GGG CTC AAA TAT ACG GTG G-3′) and GSTT1 (forward, 5′-TTC CTT ACT GGT CCT CAC ATC TC-3′; reverse, 5′-TCA CCG GAT CAT GGC CAG CA-3′). The PCR products for GSTM1 and GSTT1 were 215 bp and 480 bp, respectively. Additionally, a 400-bp fragment for the β-actin gene (forward, 5′-ACT CCC CAT CCC AAG ACC-3′; reverse, 5′-CCT TAA TGT CAC GCA CGA T-3′) was used as an internal control for DNA amplification. The GSTP1 genotype was identified by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). The GSTP1 primers were as follows: forward, 5′-ACC CCA GGG CTC TAT GGG AA-3′; reverse, 5′-TGA GGG CAC AAG CCC CT-3′. The PCR product was digested with BsmAI. Homozygous Ile-Ile (A/A) individuals had a single 177-bp fragment, homozygous Val-Val (G/G) individuals had 85- and 92-bp fragments, and heterozygous Ile-Val (A/G) individuals had 85-, 92-, and 177-bp fragments, as described previously. 20 Blank control samples used double distilled water in place of DNA in the reactions.
Measurement of nitric oxide (NO), malondialdehyde (MDA), and total antioxidant capacity (TAC)
MDA and NO activities and the TAC of seminal plasma were measured using commercial assay kits (Jiancheng, Nanjing, China) and by spectroscopic analysis, as described previously.20,23
Detection of 8-hydroxy-2′-deoxyguanosine (8-OHdG) levels
The total sperm DNA was extracted by the Chelex method: 200 μL of sperm suspension from each of the patients was extracted by adding 150 μL 5% Chelex-100 resin (Sigma-Aldrich, St Louis, MO, USA). Samples were vortexed for 10 minutes and spun for 3 minutes at 12,000 rpm. The mixture was incubated at 56°C for 3 to 6 hours, then vortexed for 10 minutes and spun for 3 minutes at 12,000 rpm. Samples were incubated at 100°C for 10 minutes and spun for 3 minutes at 12,000 rpm, then collected and stored at 4°C for a follow-up experiment. Quantitation of total sperm DNA was determined by an ultra-micro microporous plate spectrophotometer (BioTek Epoch Company, Winooski, VT, USA). The levels of 8-OHdG were evaluated using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Highly Sensitive 8-OHdG Check ELISA; Fukuroi, Shizuoka, Japan), as previously described.24–25
Sperm chromatin structure assay (SCSA)
SCSA measures the susceptibility of sperm DNA to acid-induced denaturation in situ by using the metachromatic properties of acridine orange (AO). By quantifying this metachromatic shift of AO from green to red after acid treatment using flow cytometry, the extent of DNA denaturation can be determined. The sperm DNA fragmentation index (DFI) was determined by SCSA as previously described.26,27
Sperm mitochondrial membrane potential (MMP)
MMP regulates the intact functional mitochondria and is directly associated with the motility of spermatozoa. The sperm MMP was detected by flow cytometry using 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethyl-benzimidazolyl-carbocyanine iodide (JC-1) molecular probes. 28
Statistical analysis
All data are expressed as the mean ± standard deviation (SD). Data on general sperm parameters, sperm motility parameters, oxidative stress parameters, the DFI, and the 8-OH-dG levels were analyzed using a t-test. The differences in the frequencies of the GST genotypes between groups were analyzed using the chi-square test and the correlations between the GSTs genotypes and semen parameters were examined using regression analysis. The odds ratios (OR) with 95% confidence intervals (CI) are reported. A two-tailed P-value < 0.05 was considered statistically significant. All analyses were performed using SPSS software version 20.0 (IBM Corp., Armonk, NY, USA).
Results
The present case–control study included 246 male patients diagnosed with primary idiopathic infertility. A total of 117 healthy men were included in the control group. The mean age of the patients was 27.2 ± 3.6 years. The mean age of the control group was 26.7 ± 4.8 years. No distinct differences were found between the patients and the controls concerning age, lifestyle, or environmental exposure. In this study, the levels of testosterone, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) were not noticeably different between the patients and healthy controls. The results show that all genotypes examined were consistent with Hardy–Weinberg equilibrium (HWE). The frequencies of alleles GSTM1(+) and GSTM1(−) were consistent with p and q (=1-p). The GSTT1 and GSTP1 genotypes also followed this rule (Table 1).
The distribution of the glutathione S-transferase (GST) genotypes in patients and controls.

+, wild-type genotype; −, homozygote genotype; a, compared with GSTM1/T1(+/+); OR, odds ratio; 95% CI, 95% confidence interval; GST, glutathione S-transferase.
Genetic variants of GSTM1, T1, and P1
The frequencies of the various GSTM1, T1, and P1 polymorphism genotypes in patients and controls are displayed in Table 1. The frequency of the GSTM1(−) genotype was 41.88% in the control group and 60.57% in the patient group (P = 0.001; OR=2.132; 95% CI = 1.363–3.335). The frequency of the GSTT1(−) genotype was 47.86% in the control group and 62.60% in the patient group (P = 0.008; OR=1.832; 95% CI = 1.168–2.846). The frequency of the GSTM1/T1(−/−) genotype was 14.53% in the control group and 38.62% in the patient group (P < 0.0001; OR=3.701; 95% CI = 2.083–6.575). The frequency of the GSTP1(A/G + G/G) genotype was 27.35% in the control group and 32.11% in the patient group (P = 0.847; OR=1.257; 95% CI = 0.772–2.0445).
Associations between GST variants and general and motility parameters in sperm
The sperm concentration, motility, and viability were lower in the GSTM1(−) and T1(−) genotype groups than in the GSTM1(+) and T1(+) genotype groups (P < 0.01 for both). The percentage of sperm cells with normal morphology was lower in the GSTM1(−) genotype group than in the GSTM1(+) genotype group (P < 0.01). Furthermore, sperm concentration, sperm motility, sperm viability, and percentage of sperm with normal morphology were conspicuously lower in the GSTM1/T1(−/−) group compared with the GSTM1/T1(+/+) genotype group (P < 0.01). Sperm motility was lower in the GSTP1(A/G + G/G) group than in the GSTP1(A/A) group (P = 0.015). Linearity (LIN), curvilinear velocity (VCL), path velocity (VAP), straight-line velocity (VSL), beat cross frequency (BCF), straightness (STR), and wobble (WOB) were significantly lower and the amplitude of lateral head displacement (ALH) was higher in the GSTM1(−), T1(−), and M1/T1(−/−) genotype groups than in the GSTM1(+), T1(+), and M1/T1(+/+) genotype groups (P < 0.01 for all). STR was lower and ALH was higher in the GSTP1(A/G + G/G) genotype group compared with the GSTP1(A/A) genotype group (P < 0.05) (Tables 2 and 3).
The general sperm parameters and motility parameters of each group.

#: P<0.05; *: P<0.01; GST, glutathione S-transferase; VCL, curvilinear velocity; VSL, straight-line velocity; VAP, path velocity; MAD, mean angular displacement; STR, straightness; LIN, linearity; WOB, wobble; ALH, lateral head displacement; BCF, beat cross frequency.
Regression analysis of the GSTM1(−), T1(−), and semen quality in idiopathic oligozoospermic infertility.
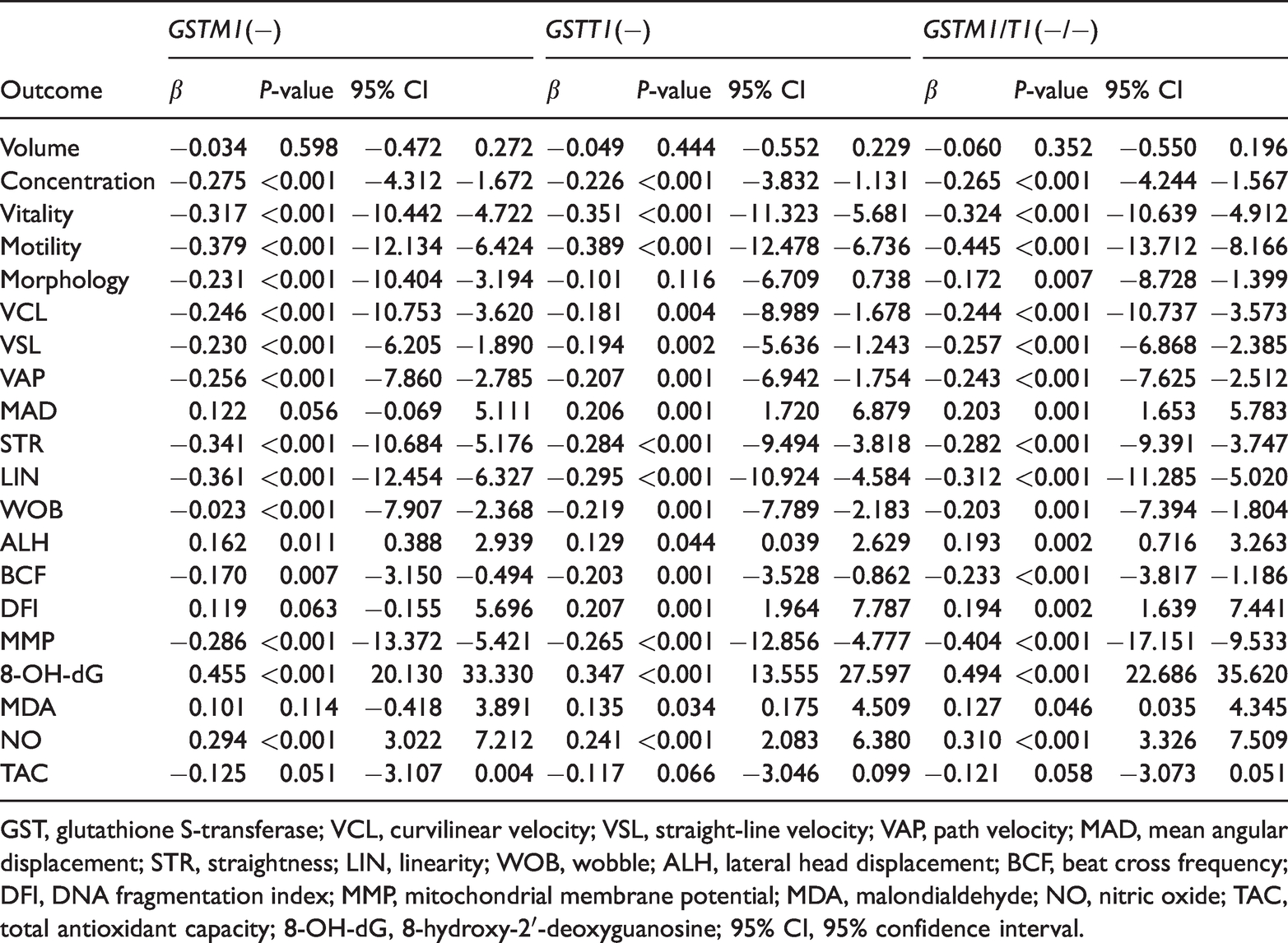
GST, glutathione S-transferase; VCL, curvilinear velocity; VSL, straight-line velocity; VAP, path velocity; MAD, mean angular displacement; STR, straightness; LIN, linearity; WOB, wobble; ALH, lateral head displacement; BCF, beat cross frequency; DFI, DNA fragmentation index; MMP, mitochondrial membrane potential; MDA, malondialdehyde; NO, nitric oxide; TAC, total antioxidant capacity; 8-OH-dG, 8-hydroxy-2′-deoxyguanosine; 95% CI, 95% confidence interval.
Associations between GST variants and OS, MMP, and DFI of sperm
NO and 8-OHdG levels were higher in the GSTM1(−), T1(−), and M1/T1(−/−) genotype groups than those in the GSTM1(+), T1(+), and M1/T1(+/+) genotype groups (P < 0.01 for both). MDA activity was higher in the GSTT1(−) and M1/T1(−/−) genotype groups than in the GSTT1(+) and M1/T1(+/+) genotype groups (P < 0.05, P < 0.01, respectively). TAC was much lower in the GSTM1/T(−/−) genotype group than in the GSTM1/T1(+/+) genotype group (P < 0.01). The NO and 8-OHdG levels were higher and TAC was lower in the GSTP1(A/G + G/G) genotype group than in the GSTP1(A/A) genotype group (P < 0.05) (Tables 3 and 4).
The oxidative stress (OS) level, mitochondrial membrane potential (MMP), and DNA fragmentation index (DFI) of each group.

#P > 0.05 vs. wild-type; *P < 0.05 vs. wild-type; **P < 0.01 vs wild-type; GST, glutathione S-transferase; MDA, malondialdehyde; NO, nitric oxide; TAC, total antioxidant capacity; 8-OH-dG, 8-hydroxy-2′-deoxyguanosine; JC-1, 5,5′,6,6′-Tetrachloro-1,1′,3,3′-tetraethyl-benzimidazolyl-carbocyanine iodide; DFI, DNA fragmentation index.
The sperm MMP was lower in the GSTM1(−), T1(−), and M1/T1(−/−) genotype groups than in the GSTM1(+), T1(+), and M1/T1(+/+) genotype groups (P < 0.01 for all). However, there was no significant difference between the GSTP1(A/G + G/G) genotype group and the GSTP1(A/A) genotype group (P = 0.085) (Tables 2 and 4).
The sperm DFI was significantly higher in the GSTT1(−) and M1/T1(−/−) genotype groups compared with the GSTT1(+) and M1/T1(+/+) genotype groups (P < 0.01 for both). However, there were no obvious differences between the GSTM1(−) and P1(A/G + G/G) genotype groups or between the GSTM1(−) and P1(A/G + G/G) genotype groups (P = 0.063, P = 0.347, respectively) (Tables 3 and 4).
Discussion
The underlying importance of GSTs in male reproductive function has been demonstrated by their presence in testis, seminiferous tubule fluid, and sperm.18,29 Developing and maturing sperm were also shown to be hypersensitive to lipid peroxidative and DNA damage caused by ROS, and this damage has been related to male infertility.30,31 Significant differences in the distribution of GST gene variants have been demonstrated among various populations and in patients with different diseases. 32 According to the current study, the distribution of variants in the GSTM1, T1, and M1/T1 genes were significantly different between the patient and control groups. Previous work suggested that the GSTT1(−) genotype had strong effects on the sperm concentration and count, suggesting the importance of this enzyme. 18 Other studies have shown that participants with the GSTM1(−) genotype had reduced sperm motility and concentration compared with participants with the GSTM1(+) genotype in both the patient and control groups.15,17 However, sperm concentration and motility were significantly lower in patients with the GSTM1-null genotype compared with patients with the GSTM1 wild-type genotype, but this was not observed in controls.16,33 In this research, the sperm concentration, motility, and viability, percentage of sperm with normal morphology, VCL, VSL, VAP, LIN, STR, BCF, and WOB values were lower and the ALH value was higher in patients with the GSTM1(−), T1(−), and M1/T1(−/−) genotypes compared with patients with the GSTM1(+), T1(+), and M1/T1(+/+) genotypes. Patients simultaneously carrying the GSTM1(−) and T1(−) genotypes showed effects on both the general parameters and motility parameters of the sperm. A mutation in GSTP1 may cause sperm motility and STR to decrease and ALH to increase.
Sperm viability could be easily evaluated by determining the inner MMP in sperm cells. The energy status of the mitochondria is indicated by the MMP, which regulates the function of mitochondria and is directly related to the motility of sperm.34,35 In this study, we found that the sperm MMP was lower in the GSTM1(−), T1(−), and M1/T1(−/−) genotype groups, which may indicate a significant negative correlation between the GSTM1(−) and T1(−) genotypes and MMP.
Previous epidemiological studies have suggested that the GSTM1(−) and T1(−) genotypes that give rise to a lack of functional protein were related to an increased susceptibility to diseases associated with OS. They also indicated that sperm were vulnerable to oxidative damage and that excessive ROS generation may result in male subfertility or infertility.36,37 Barati et al. analyzed the GSTM1 and GSTT1 null genotypes that could be considered genetic risk factors for male infertility, interfering with some oxidative stress markers in infertile men. Their findings are consistent with our research results. 38 In this study, the GSTM1(−), T1(−), and P1(A/G + G/G) genotypes were associated with increased NO and 8-OHdG levels, and the GSTT1(−) genotype was associated with increased MDA levels. While patients concurrently carrying the GSTM1 and T1(−) genotypes could show evidence of increased OS, they could also show a reduction in antioxidant substances.
Sperm DNA fragmentation may negatively affect fertilization, embryogenesis, implantation, and pregnancy.39–41 In the present study, the sperm DFI was significantly higher in patients with the GSTT1(−) and M1/T1(−/−) genotypes with idiopathic male infertility than in patients with the GSTT1(+) and M1/T1(+/+) genotypes without idiopathic male infertility. However, the GSTM1(−) and P1(A/G + G/G) genotypes had fewer effects on idiopathic male infertility than other genotypes.
Conclusion
Our results suggest that genetic variants in GSTM1, T1, and P1 may be risk factors for men with idiopathic infertility by affecting semen quality. Significant differences in semen quality resulted from variants in each gene. The patients concurrently carrying the GSTM1(−) and T1(−) genotypes could show increased effects. However, this study is limited by being a single-center study and having a relatively small sample size. Additionally, we did not completely exclude other genetic abnormalities that may also result in semen abnormalities.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: Financial support was received from the National Natural Science Foundation of China (No. 81660263).
