Abstract
Guijiajiao-Lujiaojiao (GL) is a combination of Traditional Chinese Medicine (TCM) that can be used to treat oligoasthenozoospermia (OAS). However, its mechanistic role in OAS needs to be better understood and necessitates more studies. This study was planned to investigate GL’s therapeutic effects and its mechanistic role in the tripterygium wilfordii polyglycoside (GTW)-induced OAS rat model. In total, 60 Sprague–Dawley (SD) rats at 8 weeks of age were assigned to six groups: blank (NC), model (GTW), GL low-dose (GL-L, 0.3 g/kg/day), GL medium-dose (GL-M, 0.6 g/kg/day), GL high-dose (GL-H, 1.2 g/kg/day), and GL high-dose + PI3K inhibitor LY294002 (GL-H 1.2 g/kg/day + LY 1.2 mg/kg/day) groups. The model was characterized after 8 weeks to examine sperm concentration and viability, serum hormone levels, testes histopathology, and specific protein markers. The treatment efficacy was evaluated by mRNA and protein expression levels, among other parameters. Compared with the GTW group, the viability and concentration of rat spermatozoa were significantly increased after GL intervention (p < .01). Meanwhile, the serum levels of luteinizing hormone (LH), follicle-stimulating hormone (FSH), and T hormones in rats in the GL-M and GL-H groups were significantly higher than those in the GTW group (p < .05). Furthermore, GL enhanced the proliferation of spermatogenic cells by modulating the PI3K/AKT signaling pathway, increasing and decreasing the levels of Bcl-2 and Bax proteins, respectively. It is concluded that the mechanism by which GL effectively enhanced the spermatogenic function of the GTW-induced OAS model may be attributed to the PI3K/AKT signaling pathway activation and the elevation of serum LH, FSH, and T hormone levels.
Keywords
Introduction
The most prevalent form of semen abnormality among infertile male patients is oligoasthenozoospermia (OAS), which is defined as the inability of a spouse to conceive primarily due to the male partner’s factors after ≥ 1 year of regular sexual intercourse without contraception (Alahmar, 2022). It has been observed that the quality of the semen of young Chinese men decreased annually from 2001 to 2015, particularly in both sperm concentration and viability (Huang et al., 2017). Its etiology is highly intricate, comprising multiple factors, including genetic mutations, reproductive tract infections, varicocele, poor lifestyle habits, endocrine abnormalities, and environmental pollution (Majzoub & Agarwal, 2017). Currently, there are no specific drugs available in the clinic for the treatment of OAS. Most of these drugs are empirical, such as coenzyme Q10, carnitine, and vitamin E, which are commonly used to enhance the quality of semen. There is still a lack of documentation-based medical evidence (Kallinikas et al., 2024). However, assisted reproductive technology remains controversial due to its ethical, genetic, economic, and safety implications, despite its ability to resolve the majority of fertility problems (S. Zhang et al., 2024).
In male infertility, Traditional Chinese Medicine (TCM) is a recognized safety measure and a significant component of alternative and complementary medicine. It has the potential to considerably improve the quality of semen (Ma et al., 2022). Guijiajiao (Colla Carapacis et Plastri) and Lujiaojiao (Cervus elaphus Linnaeus [Cervidae]) are the core pairs of the traditional Chinese formula “Guilu-erxian-Glue” (Ding et al., 2023), which have been widely used clinically for the treatment of OAS, but their mechanistic role has not been specifically discussed. A compound, Tripterygium wilfordii polyglycoside (GTW) derived from Tripterygium Wilfordii Hook F, is therapeutically used to treat rheumatoid arthritis. However, its prolonged use can compromise the reproductive function of the organism (Dai et al., 2022). In the previous study (Guan et al., 2020), the OAS rat model was developed using GTW. Therefore, in this study, its possible effect was also evaluated via the analysis of semen quality, serum sex hormone levels, testicular histology, and ultrastructure after developing an OAS rat model.
Spermatogenesis is a multifaceted physiological process that is dependent on the synchronized propagation and apoptosis of spermatogonia. When there is a high level of spermatogonial apoptosis, it can result in abnormalities in sperm (Q. Fang et al., 2024). Hence, the inhibition of excessive spermatogonia apoptosis and restoration of the balance between spermatogonia proliferation and apoptosis could potentially serve as a viable approach for addressing OAS.
There has been a recent discovery about the significance of this pathway in the process of spermatogonia proliferation and differentiation (Xin et al., 2024). In previous research (J. Wang et al., 2021), this pathway was modulated by the Lycium barbarum–Cuscuta medicinal combination, which has the potential to treat OAS. Thus, this study was planned to evaluate the possible mechanistic role of Guijiajiao-Lujiaojiao (GL) in the treatment of OAS and to provide empirical evidence for its clinical application. It was also focused on determining whether GL could enhance spermatogenesis in the GTW-induced OAS rat model via modulating the PI3K/AKT pathway in line with the relevant literature (W. Y. Fang et al., 2023).
Materials and Methods
Drug Formulation
In this study, GTW (10 mg, 2212111B) was prepared by Zhejiang Deinde Pharmaceutical Co. The desired suspension was composed by mixing 800 mg of tretinoin tablets in 200 mL of deionized water before administration. Lujiaojiao (5 g, 6203220901) and Guijiajiao (5 g, 6305221001) were acquired from Tortoise and Deer Pharmaceutical Group Limited (Henan, China); mixed the turtle shell and deer antler glues in equal proportions (1:1 ratio), dissolved in DI water, and prepared GL solutions with 0.15, 0.3, and 0.6 g/mL concentrations for further experiments. PI3K inhibitor LY294002 (CAS No. 154447-36-6, HY-10108, MCE, NJ, USA).
Analysis of the Chemical Composition of GL
High-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) coupled in positive (+ve) and negative (−ve) ion modes were used to characterize the compounds in the GL pure extract (Supplemental Table 1). Chromatography was performed on a C18 column (1.8 μm × 2.1 × 100 mm; Zorbax Eclipse C18) under the following parameters: injection volume of 2 μL, autosampler temperature of 4 °C, column temperature of 30 °C, flow rate of 0.3 mL/min, and the mobile phase comprised A solution (0.1% formic acid aqueous solution) and B solution (pure acetonitrile). Positive ionization mode: 325 °C was the heater temperature; auxiliary gas flow rate: 15 arb; sheath gas flow rate: 45 arb; purge gas flow rate: 1 arb; capillary temperature: 330 °C; electrospray voltage: 3.5 KV. Negative ionization mode parameters were the same as those of +ve ionization mode. The parameters of −ve ion mode were the same as those of +ve ion mode. Scanning modes: one-level full scan (Full Scan, m/z 100–1,500) and data-dependent two-level mass spectrometry scan (dd-MS2, TopN = 5). Data were acquired via the LC-MS (Thermo, Vanquish, Q Exactive HF) platform.
Animal Models
The SD male rats (8 weeks; 260 ± 10 g) were procured from Beijing Viton Lever (Beijing, China). All rats were kept in a laboratory under a regulated environment (22 °C and 60% humidity) and maintained a light/dark cycle for 12 hr. Unlimited food and water were provided to all rats. The animal-related protocols were approved by the Medical and Laboratory Animal Ethics Committee of Beijing University of Chinese Medicine (approval no. #BUCM-2023052503-222). The same protocol was followed as mentioned in the National Institutes of Health (NIH) Laboratory Animal Care and Use.
Experimental Design and Drug Treatments
After 1 week of acclimatization feeding, all rats were weighed. Approximately, 60 rats were randomly assigned to six groups (the blank [NC], GTW, GL low-dose [GL-L], GL medium-dose [GL-M], GL high-dose [GL-H], and GL high-dose + PI3K inhibitor LY294002 [GL-H+LY] groups) (n = 10). The NC group of rats was gavaged with DI water (10 mL/kg) for 4 weeks, and the rats in the remaining five groups were administered GTW solution (10 mL/kg/day) (1 mL of 4 mg GTW; 40 times the quantity employed in humans) for 4 weeks (Bao et al., 2022). Thus, four rats from each group were randomly selected for epididymis removal semen analysis to characterize and validate the OAS model (J. Wang et al., 2022). During experiment weeks (4–8 weeks), rats in NC and GTW groups were provided DI water (10 mL /kg) by gavage, whereas rats in GL-L, GL-M, and GL-H groups were administered 0.3, 0.6, and 1.2 g/kg/day GL solution by gavage. The clinical dosage of GL in the Chinese Pharmacopeia is 6–8 g (Xu et al., 2021), which is converted based on the body weight (BW) of rats and humans (70 kg), so 0.6 g/kg is designed as the middle-dose group (equivalent to six times the human dosage), 0.3 g/kg as the low-dose group, and 1.2 g/kg as the high-dose group. Simultaneously, an intraperitoneal injection of LY294002 (1.2 mg/kg/day) was administered to the GL-H+LY group (Md Mokhtar et al., 2019). After 12 hr of last administration, the BW of all rats was measured and anesthetized with sodium pentobarbital (50 mg/kg). The testes and epididymis were removed and weighed. The left epididymis was dissected to assess the quality of the sperm, and the remaining testes and epididymis were either fixed in 4% paraformaldehyde (PFA) or preserved in liquid nitrogen for further examinations.
Evaluation of Testicular and Epididymal Organ Indicators
The testes and epididymis were weighed by an electronic balance, and then the testicular and epididymal organ indices were calculated. The organ index was measured as follows: organ factor = organ weight/BW × 100%.
Sperm Concentration and Viability Analysis
The epididymis was chopped in Phosphate Buffered Saline (PBS) and kept in a 33 °C water bath for 15 min. A drop of sperm suspension was added to a slide and analyzed via a computer-assisted semen analyzer (Hamilton Thorne, MA, US).
Measurement of Serum LH, FSH, and T Sex Hormone Levels by ELISA
Blood was collected from all rat models (abdominal aorta) and serum was separated from the blood by centrifugation using a centrifuge at 4,000 x g for 20 min. In serum, luteinizing hormone (LH, CEA441Ra), follicle-stimulating hormone (FSH, CEA830Ra), and testosterone (T, CEA458Ge) levels were quantified via ELISA kits as per the provided protocols.
Hematoxylin and Eosin Staining of Testicular Tissue
To evaluate the structural integrity of spermatogonial, supporting cells, and spermatogonial tubules, testicular tissues were kept in 4% PFA for 48 hr, dehydrated by adding ethanol (various gradients), and water, preserved in paraffin wax, sectioned into 3 mm in thickness, and stained with Hematoxylin and Eosin (H&E) stains. These sections were analyzed under a light microscope (BK-FL4, Chongqing, China). (To avoid subjective bias in the selection of sections, this study used randomization to select tissue sections. Also, to reduce the assessor’s subjective factor, blinding was performed when the sections were observed and analyzed under the microscope, and the assessor did not know to which group the samples belonged.)
Ultrastructural Observation of Testicular Tissue
Hardened testicular sections (1 × 1 × 1 mm3) were pre-fixed with glutaraldehyde for 2 h at 4 °C. The sections were rinsed thrice with phosphate buffer, dehydrated with various gradients of ethanol and acetone solutions, and embedded with epoxy resin. The sections were observed via transmission electron microscopy (TEM) (Hitachi) after staining with lead citrate and uranyl acetate.
Protein Blotting Analysis
Testicular tissues preserved in liquid nitrogen were thawed and homogenized by adding RIPA buffer (Cell Signaling, USA) for 30 min on ice. The homogenized mixture was centrifuged for 15 min at 4 °C to obtain total protein content. Total protein was quantified by BCA kit (Solarbio). These protein samples were mixed with protein electrophoresis-loaded buffer. An equal amount of denatured protein from each sample was then separated and transferred to PVDF membranes which were kept with PI3K (AF6241), p-AKT (GB150002), AKT (GB15689), Bcl-2 (GB113375), Bax (GB114122), ACTIN (GB11001) primary antibodies (1/2,000) at 4 °C. The protein bands were detected and quantified via an enhanced chemiluminescence (ECL) system. The ImageJ software (NIH) was employed to estimate the relative signal intensity of the protein bands.
Reverse Transcription-Quantitative PCR
The total content of RNA was isolated from rat testicular tissues via TRIpure Reagent Total RNA Extraction Reagent (G3013, Wuhan Xavier Biotechnology Co., Ltd.) as per the provided recommendations. The isolated RNA was employed as a template for cDNA synthesis via reverse transcription with a 5X SweScript All-in-One SuperMix for qPCR (G3337, Wuhan Xavier Biotechnology Co., Ltd.). The transcribed cDNA was amplified and detected using a 96 real-time fluorescent qPCR system (Bio-Rad CFX CT, USA). The PCR protocol comprised a 15-s step at 95 °C, followed by 40 cycles of 15 s at 95 °C and 30 s at 60 °C. This study employed β-actin as an endogenous (reference) control. The results were displayed as a fold change in comparison to the control, which was adjusted at 100%. A list of used primers is depicted in Table 1.
Sequence of Primers Used for Gene Expression Profiling

Statistical Analysis
Data from all experiments were represented as the mean ±SD. Data were statistically examined via SPSS v26.0 software and compared to evaluate the differences between groups via an unpaired t-test (two groups) and a one-way ANOVA (ANOVA) (≥ 2 groups). The SNK test was then conducted for further analysis. The significant criteria were adjusted when p < .05.
Results
Compounds of GL
The compounds in GL were analyzed using the LC-MS system, and the chromatogram is depicted in Figure 1. After analysis, a total of 102 compounds in positive (+ve) and 67 compounds in negative (−ve) ion modes were detected in GL.
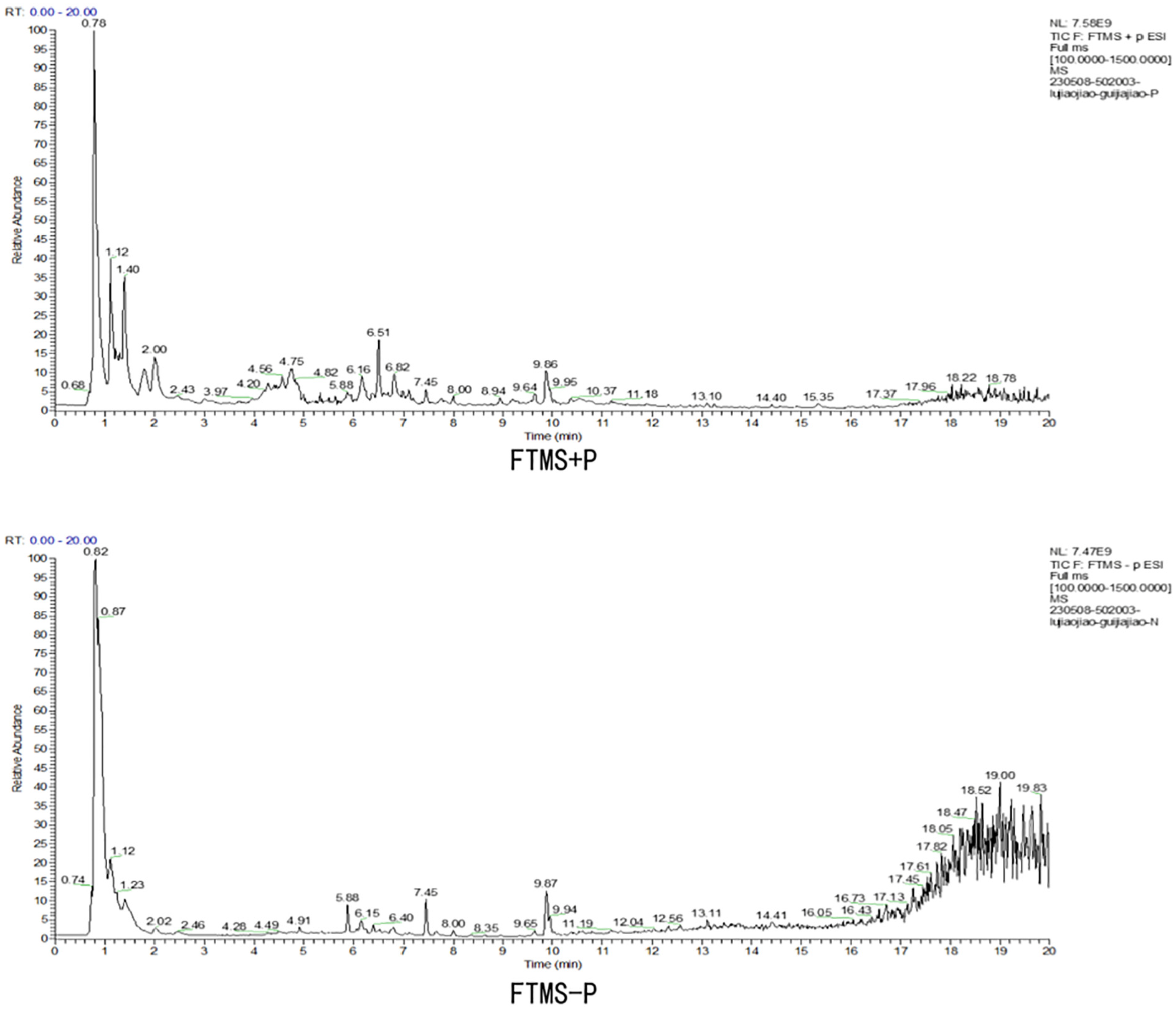
Total Ion Chromatographic Analysis of GL (With +ve and −ve Ion Modes) and its Chemical Composition
Body Weight and Organ Coefficients
Before the experiments, there were no observed variations in BW between the groups (Figure 2A). After the experiment, it was revealed that the rats in the GTW group displayed a substantial weight reduction relative to those in the NC group (p < .01; Figure 2B). In comparison to the GTW group, the rats in the GL-L, GL-M, and GL-H groups revealed markedly higher weights (p < .01; Figure 2B). Furthermore, the BW in the GL-H+LY group of rats was substantially reduced (p < .01; Figure 2B) relative to the GL-H group.

BW and Testicular and Epididymal Coefficients of Rats in all Groups. (A) The Pre-Experiment BW of the Rats in all Groups. (B) BW of Rats in all Groups After the Experiment. (C) Testis Coefficient. (D) Epididymal Coefficient
The testicular and epididymal organ coefficients of rats in the GTW group were considerably lower (p < .01; Figure 2C and D) relative to the NC group. The GL-H group revealed a substantial increase in testicular and epididymal coefficients as opposed to the GTW group (p < .01; Figure 2C and D). Relative to the GL-H group, testicular and epididymal coefficients were substantially decreased in the GL-H+LY group (p < .01; Figure 2C and D).
GL can Improve Semen Quality in GTW-Induced OAS Rat Model
In comparison to the NC group, the sperm viability and concentration in the GTW group of rats were substantially reduced (p < .01;Figure 3A and B). After GL intervention, the sperm concentration and viability of rats in the GL-L, GL-M, and GL-H groups were substantially better than those in the GTW group (p < .01; Figure 3A and B). This suggested that the highest effective dose of the drug was administered. In contrast to the GL-H group, the sperm viability and concentration in the GL-H+LY group of rats were substantially reduced (p < .01;Figure 3A and B).

Effect of GL on Semen Quality in Rats. (A) Sperm Viability. (B) Sperm Concentration
GL can Increase LH, FSH, and T Hormone Levels in GTW-Induced OAS Rat Model
As shown in Figure 4, compared to the NC group, the GTW group demonstrated substantially decreased levels of LH, FSH, and T serum sex hormones (p <.01; Figure 4A–C). The levels of these hormones were considerably higher in the GL-M and GL-H groups (p < .01 or p <.05; Figure 4A–C), however, there was no statistical significance in the GL-L group relative to the GTW group. As opposed to the GL-H group, the GL-H+LY group demonstrated substantially decreased levels of LH, FSH, and T hormones (p < .01 or p < .05; Figure 4A–C).

Effect of GL on Serum LH, FSH, and T hormone Levels in Rats. (A) LH. (B) FSH. (C) T
GL Attenuates Histopathological Sections of Testis in GTW-Induced OAS Rat Model
In the histologic analysis of testicular (Figure 5), the seminiferous tubules in the testes of rats in the NC group were structurally normal, well-orientated, and had intact lumens, and distinct and uniform boundaries between the basement membrane and the stroma. Conversely, the GTW group of rats revealed a significant number of structurally irregular seminiferous tubules, including expanded interstitial spaces, atrophied, attenuated, and disordered lumens, and vacuolization of the cytoplasm of the supporting cells. After GL treatment, the morphology of spermatogonial cells improved, the arrangement and structure of the seminiferous tubules were enhanced, and the number of supporting cells was raised. In contrast to the GL-H group, the GL-H+LY group revealed evidence of damage in the spermatogenic tubules of their rats.

Testicular Morphology. Rat Testicular Tissues Were Stained With H&E, and Pathological Alterations were Identified via Scanning Electron Microscopy (SEM). Black Arrows Denote Magnified Regions. The Upper Image was Captured at 10x Magnification (100 μm), the Middle Image at 20x Magnification (50 μm), and the Lower Image at 40x Magnification (20 μm)
GL can Repair the Ultrastructure of Testis in GTW-Induced OAS Rat Model
Major modifications in the mitochondria of spermatogonia were demonstrated after GTW treatment via TEM. The number of mitochondria decreased, and several showed swelling, dissolution of cristae, fragmentation, or even disappearance. Furthermore, the mitochondria displayed irregular shapes in contrast to the NC group. There was an observed increase in the number of mitochondria in spermatogonia after GL treatment. Furthermore, mitochondria with tubular ridge structures became visible, while the number of vesicular mitochondria decreased. In the GL-H+LY group, the quality of mitochondria in spermatogonia was found to be lower relative to the GL-H group. The specific results are shown in Figure 6.

Ultrastructure Examination of Testicular Tissue. The Number and Structure of Mitochondria in Spermatogonia of Testicular Tissue were Detected via TEM. The Upper Image was Captured at 3,000x Magnification (5.0 μm), the Middle Image at 8,000x Magnification (2.0 μm), and the Lower Image at 20,000x Magnification (500 nm)
Expression of PI3K/AKT Signaling Pathway-Related Proteins
As shown in Figure 7, in the GTW group, PI3K, p-AKT, and Bcl-2 protein levels were considerably downregulated (p < .01; Figure 7B–F) and Bax protein was substantially higher (p < .01; Figure 7E) as opposed to the NC group. Similarly, in the GL-L, GL-M, and GL-H groups, PI3K, p-AKT, and Bcl-2 protein expressions were substantially higher (p <.01; Figure 7B–F), while Bax protein was significantly downregulated (p <.01; Figure 7E) relative to the GTW group. PI3K, p-AKT, and Bcl-2 protein levels were considerably reduced (p < .01; Figure 7B–F) and Bax protein was enhanced (p < .01; Figure 7E) in the GL-H+LY group relative to the GL-H group.

Effect of GL on the Expression of Proteins Correlated with PI3K/AKT Signaling Pathway. (A) PI3K, p-AKT, AKT, Bax, Bcl-2 Protein Bands. (B) PI3K.(C) p-AKT.(D) AKT.(E) Bax.(F) Bcl-2
Expression of mRNA Related to PI3K/AKT Signaling Pathway
As shown in Figure 8, the analysis of RT-qPCR demonstrated a considerable reduction in the mRNA levels of PI3K, AKT, and Bcl-2 in the GTW group as opposed to the NC group (p <.01; Figure 8A–D). Conversely, the mRNA level of Bax was substantially higher in the NC group (p < .01; Figure 8C). As opposed to the GTW group, the GL-L, GL-M, and GL-H groups showed a considerable upregulation (p < .01; Figure 8A–D) of PI3K, AKT, and Bcl-2 mRNA. However, the level of Bax mRNA was considerably decreased (p < .01; Figure 8C) in these groups. In contrast to the GL-H group, the GL-H+LY group depicted a substantial reduction (p < .01; Figure 8A–D) in the levels of PI3K, AKT, and Bcl-2 mRNA, while Bax mRNA was substantially elevated (p < .01; Figure 8C).
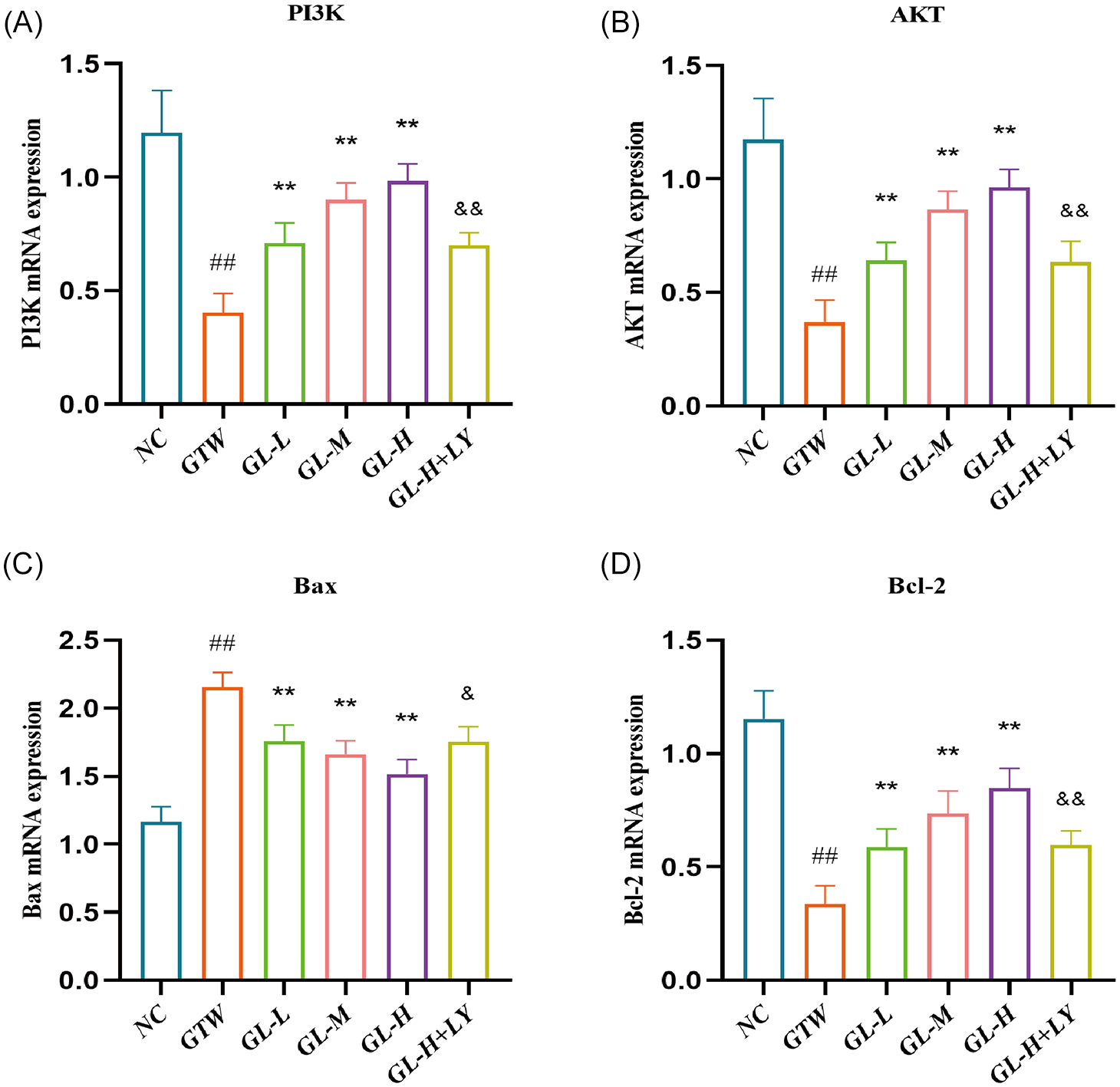
Impact of GL on mRNA Level Associated with the PI3K/AKT Signaling Pathway. (A) PI3K. (B) AKT. (C) Bax. (D) Bcl-2
Discussion
Globally, the quality of semen in men has been decreasing annually due to changes in the natural environment and human lifestyle patterns (Rodprasert et al., 2023). There are currently no effective measures to counteract this trend. The therapeutic strategies for OAS are still restricted in their clinical application (Colpi et al., 2018). Therefore, finding other suitable drugs to improve semen quality in men has an important social value. One of the effective methods of treating OAS clinically is TCM, which is a significant component of alternative and complementary. It is also known for its high level of safety concerns (M. Wang et al., 2020). In a rat model of GTW-induced oligozoospermia, this study discovered that GL (a) substantially improved sperm concentration and viability; (b) elevated serum LH, FSH, and T hormone levels; (c) decreased pathological damage to testicular tissues; (d) elevated the number of mitochondria in spermatogonial cells and attenuated impeded mitochondrial morphology; and (e) triggered the PI3K/AKT signaling pathway and stimulated Bcl-2 protein expression, promoting spermatogonia proliferation.
In this study, the compounds of GL were examined by LC-MS system, in which various amino acid components, such as L-arginine, L-proline, threonine, and hydroxyproline (metabolite of proline) were detected. The synthesis of creatine, polyamines, and guanidinamides in mammals is significantly influenced by L-arginine (C6H14N4O2), one of the amino acids obtained from GL. This compound effectively reduces the reproductive toxicity of the heavy metal aluminum, thereby protecting male fertility (Ozcan Yildirim et al., 2020). L-arginine was also found to reduce apoptosis and increase mitochondrial activity by promoting Bcl-2 and inhibiting the Bax and caspase-3 gene levels (Y. F. Zhang et al., 2020). Among the 20 common amino acids, L-Proline (C5H9NO2) is the only imino-amino acid that is antioxidant in nature. It can reduce sperm damage induced by oxidative stress, thereby protecting sperm quality and preserving chromatin integrity (Moradi et al., 2022). Threonine and hydroxyproline are two important sources of glycine biosynthesis (Wang et al., 2013). Glycine has been shown to promote cell proliferation by activating PI3K/AKT signaling (Liu et al., 2016; Sun et al., 2016).
Testicular tissue primarily consists of supporting cells, which play a crucial role in providing nutritional support to germ cells, and mesenchymal cells, which serve as the primary source of androgens (Lv et al., 2024). They collectively create a favorable microenvironment for the development of germ cells into mature spermatozoa (Ning et al., 2022). Consequently, any damage to the testicular structure will have a severe impact on spermatozoa. Herein, it was observed that GTW caused substantial damage to rat testicular tissues, including fragmentation of spermatogenic tubules, interrenal tubular edema, and cytoplasmic vacuolization of supporting cells (Figure 5). GL treatment attenuated the lesions above the testicular tissues, which led to an increase in spermatozoa viability and concentration in the OAS rat model (Figure 3A–B). The serum levels of LH, FSH, and T hormones in all groups of rats were also measured. In normal conditions, the hypothalamus secretes gonadotropin-releasing hormone (GnRH), which induces the pituitary gland to secrete FSH and LH. This LH acts on mesenchymal stromal cells, while FSH affects supportive cells to promote the production of testosterone (T) and inhibin B, respectively. These hormones are crucial for spermatogenesis (Omolaoye et al., 2023; R. S. Wang et al., 2009). The data revealed that the levels of serum LH, FSH, and T hormones displayed an increase in GL-treated rats as opposed to the GTW group. Particularly, the high-dose group presented the most substantial improvement (Figure 4A–C). This suggests that GL has a protective effect on testicular spermatogenesis by elevating the levels of serum LH, FSH, and T hormones.
Studies have demonstrated that the proliferation and differentiation of spermatogonia in the testis can be influenced by the PI3K/AKT pathway (Xin et al., 2024). PI3K is a lipid kinase that can be triggered by receptor tyrosine kinases (RTKs) and consists of p110 (catalytic) and p85 (regulatory) subunits (Chaudhuri et al., 2023). AKT is a protein downstream of PI3K. It is activated by the recruitment of phosphatidylinositol 3,4,5 trisphosphate (PIP3) to the cell membrane after PI3K activation. This activation leads to the phosphorylation of AKT’s serine 473 (Ser473) and threonine 308 (Thr308) sites. Once activated, AKT enters the cytoplasm and phosphorylates various downstream substrates, thereby regulating different cellular functions (Y. Zhang et al., 2023). For example, the PI3K/AKT signaling pathway can regulate apoptosis by modifying Bcl-2 family proteins (Roopashree et al., 2024). These proteins have distinct functions in apoptosis, which can be classified into two distinct groups. Bax and Bak proteins are examples of one type that can induce apoptosis, while Bcl-2 and Bcl-XL proteins are examples of the other type that can inhibit apoptosis (Gu et al., 2021). One group of proteins (Bax and Bak) can stimulate apoptosis, while another group (Bcl-2 and Bcl-XL), can hinder apoptosis (Saha et al., 2024). Studies have depicted that the Bcl-2 proteins are crucial mediators of mitochondrial inner membrane permeability and are strongly associated with the mitochondrial apoptotic pathway (Czabotar et al., 2023). When stress-induced apoptosis initiates, Bax and Bak become attracted to the outer mitochondrial membrane, thereby increasing the permeability of the mitochondrial outer membrane (Sovilj et al., 2024). Thus, cytochrome C is secreted from the mitochondria and forms a multimeric complex with Apaf-1 in the cytoplasm. This complex triggers the cysteine asparaginase cascade reaction, which leads to apoptosis via self-shearing (Jemmerson et al., 2021).
In this study, the levels of PI3K, p-AKT, and Bcl-2 proteins were substantially downregulated after GTW induction, while the Bax protein level was considerably increased. Furthermore, the number of mitochondria in spermatogonia was lower and structurally abnormal. The levels of PI3K, p-AKT, and Bcl-2 proteins were substantially upregulated (Figure 7B, C, and F) and the Bax protein level was downregulated (Figure 7E) after treatment with GL. Moreover, GL treatment also increased the number of mitochondria and improved its structure and tubular ridges (Figure 6). Thus, it was suggested that GL may regulate the PI3K/AKT signaling pathway to enhance the Bcl-2 protein level, block Bax protein expression, and suppress apoptosis in the mitochondrial pathway of spermatogonia. To further analyze the association between GL and the PI3K/AKT pathway, the PI3K inhibitor LY294002 was administered to the GL-H group. The GL-H+LY group showed substantially reduced levels of PI3K, p-AKT, and Bcl-2 proteins, while the level of Bax protein was substantially elevated as compared to the GL-H group. These findings suggested that LY294002 reversed the effect of GL on GTW-induced spermatogenesis in the OAS model, indicating that the primary mechanism of GL is to stimulate the PI3K/AKT pathway.
Conclusion
In conclusion, GL has the potential to enhance spermatogenesis in the GTW-induced OAS rat model. Its mechanism may be associated with the stimulation of the PI3K/AKT pathway, high levels of FSH, LH, and T sex hormones, and Bcl-2 protein, thus hindering spermatogonia apoptosis. Therefore, this study presents preliminary results supporting the use of GL in the clinical management of OAS.
Supplemental Material
sj-xls-1-jmh-10.1177_15579883241293941 – Supplemental material for Guijiajiao-Lujiaojiao Synergistically Promote Spermatogenesis in Tripterygium Wilfordii Polyglycoside–Induced Oligoasthenozoospermia Rats via PI3K/AKT Signaling Pathway
Supplemental material, sj-xls-1-jmh-10.1177_15579883241293941 for Guijiajiao-Lujiaojiao Synergistically Promote Spermatogenesis in Tripterygium Wilfordii Polyglycoside–Induced Oligoasthenozoospermia Rats via PI3K/AKT Signaling Pathway by Nianwen Huang, Haisong Li, Longji Sun, Junlong Feng, Zixiang Gao, Zhechao Lin, Yong Yang, Bin Wang and Jisheng Wang in American Journal of Men's Health
Footnotes
Acknowledgements
All the authors of the manuscript thank the foundations for their valuable support.
Author Contributions
All authors contributed to the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Beijing Municipal Natural Science Foundation Program (grant no. 7244488); National Natural Science Foundation of China (grant no. 82174385); Horizontal cooperation project between Dongzhimen Hospital of Beijing University of Chinese Medicine and Guolu Pharmaceutical Group Co. Ltd. (HX-DZM-202302); the Major Engineering Special Project of the State Administration of Traditional Chinese Medicine (grant No. GHC-2023-WTBS-014); and the Clinical Research Operating Expenses of Centralized High-level Chinese Medicine Hospitals.
Consent for Publication
Not applicable.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
