Abstract
This study aimed to detect the impact of bacteriospermia on semen parameters and sperm DNA fragmentation. This prospective case–control study was conducted over a period of 9 months. Samples were collected from andrology outpatient clinic attendants of Cairo University Hospitals. We enrolled 68 semen samples divided into two groups: a study group (34 semen samples with bacteriospermia) and a control group (34 semen samples without bacteriospermia). The characteristics of the semen, including morphology, motility, count, liquefaction, viscosity, pH, volume, and appearance, were evaluated according to the standard protocols. Patients with and without bacteriospermia had a comparable liquefaction time (
Introduction
Infertility affects 10–15% of couples in the reproductive age group (Deka & Sarma, 2010). Approximately 15% of male factor infertility is caused by infectious etiologies such as protozoa, fungi, viruses, and bacteria (Diemer et al., 2003; Keck et al., 1998; Weng et al., 2014). Genital tract obstruction due to fibrosis and inflammation, antisperm antibody formation due to blood–testes barrier breach, reactive oxygen species (ROS) formation leading to increased DNA fragmentation index, altered sperm morphology, altered acrosome reaction, decreased sperm motility, and diminished spermatogenesis are all methods in which bacteriospermia can interfere with normal fertility (Jarow et al., 1990; Keck et al., 1998; Köhn et al., 1998; Moustafa et al., 2004).
DNA damage in sperm is most often caused by ROS while the sperm passes through the male reproductive system (Agarwal et al., 2014; Schulte et al., 2010). The majority of these genitourinary tract infections are asymptomatic, which raises the question of whether or not to treat the affected individuals. There is growing evidence that decreased semen quality and asymptomatic bacteriospermia are linked. Since a large proportion of fragmented DNA was reported in infertile individuals, sperm DNA integrity has been deemed a predictor of male fertility (Kumaresan et al., 2020; López et al., 2013). Condensation of sperm DNA seems to be an inevitable factor in male fertility, and sperm DNA integrity appears to influence early embryonic development (Schulte et al., 2010).
The effects of bacteriospermia on subfertile and infertile men’s semen parameters, sperm DNA integrity, and ROS generation are not well understood. Bacterial infection has been reported to be detrimental to the male urogenital system, according to a few studies; nevertheless, according to other investigations, a bacterial infection could not alter the characteristics of sperm. Therefore, in this study, we aimed to detect samples with bacteriospermia and its effect on semen parameters and sperm DNA fragmentation (SDF).
Methods
Study Design
This prospective case–control study was conducted over a period of 9 months, from June 2021 to February 2022. Samples were collected from andrology outpatient clinic attendants of Cairo University Hospitals, and further processing was done in the Medical Microbiology and Immunology Department, Faculty of Medicine, Cairo University. Our study included a total of 68 semen samples divided into two groups: a study group (34 semen samples with bacteriospermia) and a control group (34 semen samples without bacteriospermia). The research was approved by the Ethics Committee of the Institutional Review Board, Faculty of Medicine, Cairo University (code: MS-428-2021).
Participants
This study included men undergoing fertility investigations at the andrology outpatient clinic at Cairo University Hospitals. The age of the included patients ranged from 20 to 50 years, with female partners younger than 30 years, with regular menses, and normal hormonal profiles. Patients with varicocele, genetic abnormalities, endocrine disturbances, testicular failure, erectile problems, or smokers and those who were on antibiotics at the time of sample collection were excluded from the study.
Following the recommendations of the World Health Organization (WHO), the characteristics of the semen, including morphology, motility, count, liquefaction, viscosity, pH, volume, and appearance, were recorded (World Health Organization, 1999, 2021). Within 3 hours of specimen collection, the microbiology laboratory performed a Gram stain and cultured the samples in blood agar and MacConkey agar, as recommended by the WHO (1999, 2021). The incubation temperature for the cultures was set at 37°C. Isolates of organisms with a colony-forming units/mL count of >103 were deemed to be significant (Domes et al., 2012). In accordance with standard protocols, the isolated organisms were tested for antimicrobial susceptibility using the Kirby–Bauer disk diffusion technique (Patel et al., 2015). Clinical and Laboratory Standards Institute (CLSI) standards were used to calculate and evaluate the zone of inhibition (Limbago, 2019).
SDF Analysis
The Halosperm G2 kit from Halotech, based on the sperm chromatin dispersion test, was used to analyze the DNA fragmentation of 20 samples from the study group’s sperm. Float, Solution 4 thiazine staining solution B, Solution 3 eosine staining solution A (SSA), Solution 2 lysis solution (LS), Solution 1 denaturant agent (DA), Eppendorf tubes, super-coated slides, and agarose gel support were all included in the kit’s reagents.
Procedure
After inserting the agarose screw tube into the float, it was melted in a water bath (or a beaker with water on a hot plate) at 95–100°C for 5 minutes or until it was totally melted. Ten Eppendorf tubes, each holding 100 l, were filled with agarose solution that had been melted. The subsequent 5 minutes were spent maintaining a 37°C temperature in the to-be-used Eppendorf to inhibit gelification. Eppendorf tubes not needed at the time were placed in the refrigerator. Both Solution 1 and Solution 2 were maintained at 22°C. The kit’s super-coated slide was all set for usage. A human sperm extender was used to dilute the sperm sample to a concentration of no more than 20 million sperm/mL. The sperm sample was then transferred to an Eppendorf tube and gently combined using a micropipette. Subsequently, 8 l of the cell suspension was deposited into the middle of the sample well (“S”). The whole time, the slide was kept in a horizontal position. A control sample was processed in well “C.” After 5 minutes in the fridge at 4°C, the agarose was firm, and the slide could be removed. The cover slip was slipped off the slide carefully when it was taken out of the refrigerator. All the work was done at a standard room temperature of 22°C.
In a Petri dish, the slide was positioned horizontally and raised. After Solution 1 (DA) was added into the well, it was set aside for incubation for 7 minutes, ensuring it was completely covered by the reactive during the whole procedure. The reactive was then eliminated by tilting the slide until it was entirely dry, at which point it was set horizontally in an elevated position. It was crucial to take the reactive out without shaking. By tilting it and letting it slide, we were able to remove it. The well was treated with Solution 2 (LS), ensuring it was completely submerged, and then incubated for 20 minutes. The reactive was then eliminated by tilting the slide until it was completely dry, after which it was positioned horizontally in an elevated position.
A disposable pipette was used to wash the slide for 5 minutes with plenty of distilled water. The reactive was then removed from the slide by tilting it until it was completely dry, after which the slide was positioned horizontally and lifted. Using a disposable pipette, 70% ethanol was flooded into the sample to induce dehydration. After incubation for 2 minutes, the sample was drained, then 100% ethanol was added for another 2 minutes. The slide was then drained and let to air dry on filter paper in a horizontal position. The slide was put into a Petri dish in an elevated, horizontal position. The wells were submerged in Solution 3 (SSA) for 7 minutes before being analyzed. After the stain had thoroughly dried, the slide was inverted and set at an angle to remove it. Fragmented DNA in sperm was evaluated. All clinical and analytical data pertaining to the sperm sample were taken into account while assessing the results. Evenson and Wixon (2006) have proposed 30% as a cutoff level for the frequency of SDF.
Statistical Analysis
Results were presented as mean ± standard deviation or number and percentage. The chi-square or Fisher exact test was used to compare categorical variables. Data distribution was analyzed using the Kolmogorov–Smirnov test. Therefore, a paired
Results
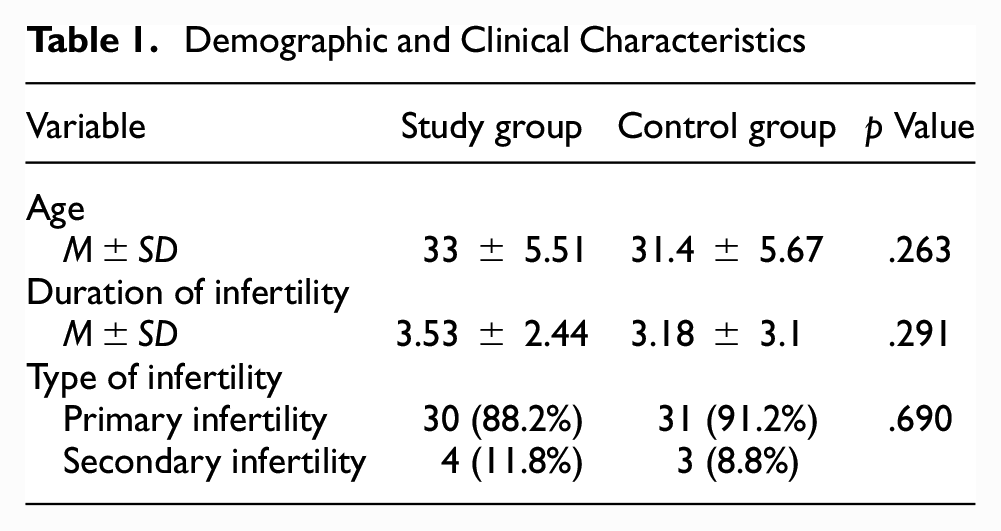
Demographic and Clinical Characteristics
A total of 68 subjects were included; 34 with bacteriospermia and 34 without bacteriospermia. The mean age and duration of infertility of the two groups were comparable (
Demographic and Clinical Characteristics
Semen Analysis
All patients (100%) in both groups had normal pH levels and were homogeneous and grey opalescent. About 88.9% (
Comparison of Semen Parameters Among Study and Control Group Samples

Statistically significant.
Comparison of the Mean Value of Semen Parameters Between the Study and Control Groups

Statistically significant.
Types of Semen Abnormalities in the Study and Control Groups

Microbiological Evaluation
Regarding the microbiological evaluation of the study group, four bacterial species were isolated. These include
Different Organisms Isolated and Their Frequency

Antibiotic Sensitivity
Approximately 78.3% of the
In MRSA samples, the mean of the total sperm count was higher than that of the control group, with no statistically significant difference (
Comparison of the Mean Value of Semen Parameters in the MRSA and Control Groups

SDF Analysis
Supplementary Table S5 presents the semen characteristics of the study group tested for SDF. The SDF analysis was done for 20 samples of the study group with a mean value of 38.25 ± 14.25%. The SDF was <30% in six (30%) samples (group A) and ≥30% in 14 (70%) samples (group B). A significant difference was reported between Groups A and B in terms of total motility percentage (
Comparison of Semen Parameters Among SDF Subgroups

Discussion
This study showed that patients with and without bacteriospermia had a comparable liquefaction time, semen appearance and color, semen pH, velocity, and total sperm count. Patients with bacteriospermia were associated with lower progressive motility, nonprogressive motility, total motility, and normal forms. The prevalence of abnormal semen analysis was 64.71% in the study group compared to 35.29% in the control group. Similar to our findings, Nasrallah et al. (2018) reported that viscosity, color, and pH were normal in patients with or without bacteriospermia. They also demonstrated that the volume of semen was abnormal in 13% of the patients with bacteriospermia and 6.5% of the control group (Nasrallah et al., 2018). Sanocka-Maciejewska et al. (2005) reported that the semen mean volume was not affected by the presence of bacteriospermia. Pagliuca et al. (2021) reported a significant decrease in semen mean volume in samples with bacteriospermia. Low semen volume can result in a low sperm count and low motility, parameters that are very necessary for fertilization. High-volume semen could result in overdilution of the sperm cells, hence low sperm concentration. Domes et al. (2012) reported that sperm count was not significantly affected in samples with bacteriospermia. Other studies reported a significant reduction in sperm count in samples with bacterial infection (Berjis et al., 2018; Pagliuca et al., 2021). Sperm morphology and motility have proved to be remarkable factors in semen parameters (Xu et al., 2020). Sanocka-Maciejewska et al. (2005) and Eini et al. (2021) reported a negative influence of bacteria on sperm motility. However, Domes et al. (2012) and Nasrallah et al. (2018) reported no significant reduction in sperm motility in samples with bacteriospermia.
According to our findings,
Despite the prevalence of positive semen cultures, research on antibiotic resistance is limited. According to Bhatt et al. (2015), both gentamycin (54.5%) and levofloxacin (63%) were reported to be effective against
Seventy percent of the semen samples analyzed in this study had an SDF higher than 30%, indicating significant DNA fragmentation in the sperm samples. Eini et al. (2021) reported that the rate of DNA fragmentation in infected patients’ sperm was substantially greater than that of noninfected individuals. Another study by Zeyad, Hamad, and Hammadeh (2018) reported that bacterial infection hinders DNA folding in sperm. A significant level of SDF was indicated in the semen samples infected with
Limitations
This study has some limitations, including the small sample size and the absence of a control group in the DNA fragmentation analysis. This study did not account for the potential change in semen parameters following treatment of the infection and also excluded certain bacteria, including
In conclusion, our results revealed that bacteriospermia has deteriorative effects on sperm quality parameters, such as semen volume, sperm motility, and sperm normal morphology. Patients’ semen samples most often yielded
Supplemental Material
sj-docx-1-jmh-10.1177_15579883231181861 – Supplemental material for The Impact of Bacteriospermia on Semen Parameters Among Infertile Egyptian Men: A Case–Control Study
Supplemental material, sj-docx-1-jmh-10.1177_15579883231181861 for The Impact of Bacteriospermia on Semen Parameters Among Infertile Egyptian Men: A Case–Control Study by Rania Yahia Mohammad Shash, Gehad Attia Abdallah Mohamed, Salah E. Shebl, Mohamed Shokr and Soliman Aref Soliman in American Journal of Men's Health
Footnotes
Author Contributions
S.A.S. contributed to study concept and development. G.A.A.M. was responsible for data collection, performed the statistical analysis, and wrote the manuscript. S.E.S. and R.Y.M.S. performed data interpretation. M.S. provided critical revision for important intellectual content and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The protocol of the current trial was approved by the local ethics committee of faculty of medicine at Cairo University, Cairo, Egypt (no applicable number). All procedures were in compliance with the standards of the Declaration of Helsinki (JAVA, 2013). Written informed consent was obtained from all patients before enrollment.
Data Availability
The data set used or analyzed during the current study is available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
