Abstract
To access the possibility that key markers of bacterial vaginosis (KM-BV) could affect seminal parameters and thus fertility a prospective cohort study was designed (a) to develop rapid and sensitive multiplex polymerase chain reaction (M-PCR) assays to screen 13 key markers of bacterial vaginosis (KM-BV) in semen specimens, (b) to determine the prevalence of KM-BV in semen from randomized male partners of couples seeking fertility evaluation. A total of 229 semen samples were included in the study from males who visited the Sperm Analysis Section of Brazil between October 2015 and March 2016. Eligible men were 18 years or older and had a semen analysis due fertility evaluation (after failing to conceive with their partner after 1 year of unprotected intercourse). Basic seminal parameters were analyzed, and KM-BV was detected by M-PCR assays. M-PCR assays clearly distinguished 13 KM-BV in 146 semen samples (63.8%), mainly Gardnerella vaginalis (50.7%). Some important associations occurred between the presence of KM-BV in semen and changes in seminal parameters. KM-BV is commonly present in the semen of males seeking fertility evaluation and could potentially play significant roles in male subfertility and/or infertility.
Infertility is defined as the inability for couples to conceive even after ⩾12 consecutive months of regular unprotected sexual intercourse (Boivin, Bunting, Collins, & Nygren, 2007; Chandra, Coppen, & Stephen, 2013). Infertility is an increasingly common condition, and male factors (exclusively or in combination with female factors) are now estimated to play a significant role in approximately 40%–50% of infertility cases (Carr, 2013; Rittenberg & El-Toukhy, 2010). Despite contemporary therapies, which have undoubtedly increased the likelihood of conception among couples suffering from male infertility, these solutions often overlook the absence of a defined etiological or pathophysiological diagnosis. Unfortunately, male infertility is still considered “idiopathic” in a large proportion of cases (Carr, 2013; Rittenberg & El-Toukhy, 2010; Weng et al., 2014).
Since 2000, the World Health Organization (WHO) recognized the role of genital tract infections in human infertility (Gimenes et al., 2014; Rowe & Comhaire, 2000). Most Sexually transmitted diseases, such as Chlamydia trachomatis, Neisseria gonorrhoeae and human papillomavirus can target different tissues along the male genital tract, causing functional alterations that can result in reduced fertility or even infertility. In particular, pathological bacterial strains that are present in semen can directly affect sperm quality by reducing motility (Rybar et al., 2012), inducing apoptosis or necrosis (Villegas, Schulz, Soto, & Sanchez, 2005), and altering sperm morphology (Isaiah, Nche, Nwagu, & Nnanna, 2011). Although in the past 6 years it has become accepted that bacterial infections, such as Chlamydia trachomatis and Neisseria gonorrhoeae can lead to changes in male fertility (Abusarah, Awwad, Charvalos, & Shehabi, 2013; Isaiah et al., 2011; Pajovic, Radojevic, Vucovik, & Stjepcevic, 2013), little is known about the impact of most bacterial infections on seminal parameters.
Since the beginning of 2000s, it is becoming more evident that countless microorganisms coexist in the human body as part of a complex community known as the microbiome. The microbiome has multiple implications in diverse multifactorial diseases, such as obesity, Crohn’s disease, asthma, and infertility (Althani et al., 2016; Franasiak & Scott, 2015; Morgan, Segata, & Huttenhower, 2013). In this scope, the female reproductive tract has been more intensively studied in healthy and diseased individuals. It has been established that Lactobacillus spp. have an important role in controlling the overgrowth of other bacteria (Althani et al., 2016; Braundmeier et al., 2001; Franasiak & Scott, 2015). An altered microbiota is associated with bacterial vaginosis (BV), the most common genital tract syndrome in women. BV is associated with an increased risk of perinatal complications (Foxman et al., 2014), pelvic inflammatory disease (Peipert et al., 2001), and HIV acquisition and transmission (Hay, 2014). The etiology of BV is not completely understood, and the syndrome is considered to be an ecological disorder of the vaginal microbiota. BV is characterized by a reduction in lactic acid-producing bacteria (mainly Lactobacillus) and an increase in the number and diversity of facultative and strictly anaerobic bacteria, mainly Gardnerella vaginalis (Bautista et al., 2016; Hay, 2014; Ravel et al., 2011). Over the past 6 years studies using modern molecular techniques on vaginal specimens have dramatically increased the number of bacterial species known to inhabit the vaginal environment. Many of these bacteria, mainly the anaerobic, have been identified to be key markers for BV (KM-BV) and/or highly associated with BV, including Atopobium vaginae; Bacteroides fragilis; Clostridium-like bacterial vaginosis-associated bacteria (BVAB) 1, 2, and 3; Sneathia spp.; Megasphaera type 1; Mycoplasma hominis; Mycoplasma genitalium; Mobiluncus spp.; and Ureaplasma urealyticum (Fethers et al., 2012; Foxman et al., 2014; Malaguti, Bahls, Uchimura, Gimenes, & Consolaro, 2015; Shipitsyna et al., 2013; Srinivasan et al., 2012).
Much less attention has been given to the male reproductive system, particularly the characterization of bacterial communities in the seminal plasma (Franasiak & Scott, 2015; Monteiro et al., 2018). It is reasonable to suspect that bacteria involved in female reproductive tract diseases, such as KM-BV, may contribute to male genital microbiota and affect reproductive outcomes. Despite its importance for men and their partners, little is known about the prevalence and role of KM-BV in the male reproductive tract. Furthermore, the significance of bacteriospermia in asymptomatic men remains unclear. Therefore, this work presents a prospective cohort study (a) to develop rapid and sensitive multiplex PCR assays to screen 13 key markers of BV (KM-BV) in semen specimens, (b) to determine the prevalence of KM-BV in the semen of randomized male partners of couples seeking fertility evaluation, and (c) to access the possibility that KM-BV could affect seminal parameters and thus fertility.
Materials and Methods
Study Population
A total of 285 men attended the Sperm Analysis Section of Brazil, between October 2015 and March 2016. As part of a work-up for conjugal infertility investigations (after failing to conceive with their partner after 1 year of unprotected intercourse), all participants had requested semen analysis for medical action. Men who presented with any symptoms of genitourinary infections, had received antibiotics within the previous 3 months, had reproductive system abnormalities (e.g., varicocele), or had undergone infertility therapy in the preceding year were excluded from the study. In addition, subjects who tested positive for C. trachomatis, N. gonorrheae, and HPV were excluded from the study.
All procedures performed in the study were in accordance with the ethical standards of the Committee for Ethics in Research Involving Humans at Brazil (report no. 1163409/2015) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All subjects were aged at least 18 years. Participation was voluntary, and patients were properly informed about the aim of the study. At recruitment, written informed consent was obtained from all study subjects.
Semen Collection and Analysis
Prior to semen analysis, all subjects were asked to abstain from sexual intercourse or masturbation for 3–5 days before presenting for semen collection in the laboratory. Before semen collection, subjects were instructed to wash their hands with soap. Subjects urinated and washed the glans penis and coronal sulcus with soap and water before samples were collected. Semen was collected via masturbation and ejaculated directly into sterile standard containers previously ascertained to have no cytotoxic effects on human spermatozoa. To prevent sample contamination, contact was avoided with the interior of the sterile wall of the container. Freshly collected semen was immediately incubated at 37°C for 15–60 min for liquefaction. Samples were then homogenized, and 300 µL of the sample was transferred to tubes containing 1.0 ml of sterile 0.9% NaCl solution. Samples were immediately stored at −80°C until DNA extraction.
Detection of basic semen parameters was performed according to the WHO (2010) criteria to determine the following variables: seminal volume, pH, viscosity, sperm concentration, vitality, total progressive motility (category [a + b]), rapid progressive motility (category [a]) and morphology (normal forms), leukocytospermia, and hematospermia. Semen was considered to be acidic when pH was < and alkaline when pH was > 8.2. Hypospermia was defined as seminal volume < 1.5 mL, oligozoospermia was defined as sperm concentration < 15 × 106/mL, asthenozoospermia was defined as sperm motility < 50% (category [a + b]), necrozoospermia was defined as sperm vitality < 58%, teratozoospermia was defined as normal morphology < 30%, leukocytospermia was defined as leukocyte concentration > 1 × 106 ml, and hematospermia was defined as erythrocyte concentration > 1 × 106 ml (WHO, 2010).
Genomic DNA Extraction and Purification
To remove any polymerase chain reaction (PCR) inhibitors from the semen, samples were incubated for 15 min with proteinase K in phosphate-buffered saline and then centrifuged. Extraction of genomic DNA was performed using a Purelink® Viral RNA/DNA Mini Kit (Invitrogen, CA, USA), according to manufacturer’s instructions. Measurement of the quantity and quality of purified DNA was performed by spectrophotometry (NanoDrop 2000 Spectrophotometer, Thermo Scientific, Wilmington, USA).
Multiplex PCR (M-PCR) Assays
M-PCR assays proposed here were previously developed for use on vaginal specimens (Malaguti, Bahls, Uchimura, Gimenes, & Consolaro, 2015). To adapt and validate these assays for semen specimens, 13 primers were selected based on published papers (Ferris, Mastzal, & Martin, 2004; Fredricks, Fiedler, Thomas, Mitchel, & Marrazzo, 2009; Ingianni, Petruzezlli, Morandotti, & Pompei, 1997; McIver et al., 2009; Muvunyi et al., 2011; Nassar, Abu-Elamreen, Shubair, & Sharif, 2008; Obata-Yasuoka, Ba-Thein, Hamada, & Haiashi, 2002; Riley, Samadopour, & Krieger, 1991; Tiveljung, Forsum, & Monstein, 1996). Specificity was checked against all sequences in the GenBank using SeqSearch, and primers were aligned using the Clustal X program (v. 1.81, NCBI, Bethesda, MD). Subsequently, all primers were evaluated by performing a BLAST analysis against the sequences in NCBI database. The primers were approved when no mismatches within the critical regions of the primers (e.g., no mismatch at the 3′ end of a primer) were identified. All selected primers were designed to have similar physical characteristics to allow for simultaneous amplification in a multiplex reaction without the loss of sensitivity and easy separation using gel electrophoresis as follows: melting temperature −55°C–65°C; length −18–26 base pairs (bp), and amplicon size −80–842 bp (Supplementary Table 1).
To assess the specificity of these primers, they were tested in a single-target PCR (sPCR) or an M-PCR assay with different samples. Upon amplification of clinical samples that tested positive for any of the 13 bacteria, no cross-reactivity was observed among the primers.
Different parameters (MgCl2 and primers concentrations, annealing and extension temperatures, and number of cycles) were tested in different combinations. Different annealing temperatures for the primers were identified, and the analysis was splited into three M-PCR assays. Assay 1 was standardized to detect six KM-BV: G. vaginalis, M. curtisii, B. fragilis, Mycoplasma hominis, U. urealyticum and Megasphaera type 1. Assay 2 was standardized to detect three KM-BV: A. vaginae, BVAB1, and BVAB2. Assay 3 was standardized to detect four KM-BV: M. mulieris, BVAB3, S. sanguinegens, and Mycoplasma genitalium.
The optimized protocol for each M-PCR assay was a mixture of 25 µL containing 2.5 mM aliquots of each dNTP, 0.6 mM MgCl2, 25 mM aliquots of each primer, 5 µL of extracted DNA (50 ng of total sample), and 1 U of platinum Taq DNA polymerase (Invitrogen, CA, USA). PCR conditions comprised 35 amplification cycles of denaturation for 10 min at 94°C, annealing for 1 min with variable temperature depending on the assay (55°C, 62°C, and 63°C for assays 1, 2, and 3, respectively), and extension for 1 min at 72°C and a final extension for 10 min at 72°C (Thermal cycler, Biosystem, CA, USA). Resulting M-PCR products were electrophoresed in 8% polyacrylamide gel using 1X TBE buffer (45 mM Tris-borate, 1 mM EDTA, pH 8.0). All gels were stained after running with 1 mg/mL ethidium bromide and photographed under UV light (UVP BioImaging Systems, Upland, CA, USA).
Single-Target PCR (sPCR) Assays
For standardization and validation of the detection by M-PCR, sPCR was also performed for the 13 bacteria in all samples studied and in positive controls using the same primers used for the M-PCR. sPCR (the gold standard) is generally more sensitive than M-PCR, and it prevents cross-reactions, which can occur during M-PCR (McIver et al., 2009). The sPCR assay comprised 15 µL containing 2.5 mM aliquots of each dNTP, 0.6 mM of MgCl2, 25 mM aliquots of each primer, 5 µL of extracted DNA (50 ng), and 1 U of Taq DNA polymerase (Invitrogen, CA, USA). sPCR conditions were kept identical to those in M-PCR assays 1, 2, and 3. Amplification fragments were electrophoresed in a 1.5% agarose gel stained with 1 mg/mL ethidium bromide.
Co-amplification of the human β-globin gene using specific primers GH20 (5′-GAAGAGCCAAGGACAGGTAC-3′) and PC04 (5′-CAACTTCATCCACGTTCACC-3′) was performed by sPCR and M-PCR for all semen and control samples. Co-amplification of the β-globin gene was performed for assessing inhibition, sample adequacy, and integrity. In particular, it was used as an internal control for amplification to ensure that amplifiable DNA was successfully extracted from the samples and controls. Finally, it was used to check for PCR inhibitors (Fredricks & Relman, 1999) under the same conditions as M-PCR and sPCRs.
Two types of controls, “no-DNA” (negative control) and “positive DNA” (positive control), were also included in each reaction series (M-PCR, sPCR, and β-globin PCR). These controls were run for all studied KM-BV that were derived from positive clinical samples previously detected and identified using reference methods, including culture and/or sPCR.
Statistical Analysis
For validation of M-PCR assays, was calculated the sensitivity [(true positive/(true positive + false negative))*100], specificity {[true negative/(true negative + false positive)]*100}, positive predictive value {[true positive/(true positive + false positive)]*100}, and negative predictive value {[true negative/(true negative + false negative)]*100}. Then, the results from M-PCR were compared with those from sPCR (gold standard).
Seminal parameters were compared between total KM-BV-positive and -negative groups using the χ2 test. Mean values for age and seminal parameters were compared in the total study population and in strata of KM-BV presence. KM-BV-negative and mutually exclusive KM-BV-positive subgroups were compared by one-way analysis of variance (ANOVA). Two-sided p ⩽ .05 was considered statistically significant. Statistical analysis was performed using GraphPad Prism 6.0 (San Diego, CA, USA).
Results
After application of the inclusion and exclusion criteria of patients for the study, which have been described previously, a total of 229 men were included. The patient age range was 18–52 years, with a mean age and standard deviation of 32.87 ± 6.6 years.
For validation of M-PCR assays, 81 semen samples were used. The M-PCR assay clearly distinguished and correctly identified KM-BV analyzed in semen samples, whether single agent (1 KM-BV) or coinfection (⩾ 2 KM-BV). In addition, false positive results were not detected. BVAB3 and S. sanguinegens were not detected in semen samples analyzed in the study, but their positive controls and β-globin were correctly amplified in M-PCR and sPCR assays (Figure 1). Thus, parameters of the M-PCR assay were established considering positive controls for both bacteria. Figure 1 also shows amplification of the positive control and B-globin of M. genitalium, which was detected in a small number of samples in the present study. The overall agreement of M-PCR results with sPCR was 99.3%; validation parameters were as follows: 96.79% sensitivity, 100.00% specificity, 100.00% PPV, 99.78% NPV, and 99.80% ACC (Supplementary Table 2).

Electrophoretic analysis of amplified fragments using M-PCR in 2% agarose gel.
When the agents were individually analyzed, M-PCR reported to have 100.0% specificity and PPV for all bacterial agents. For M. curtisii, M. hominis, G. vaginalis, B. fragilis, A. vaginae, BVAB1, BVAB2, BVAB3, Mycoplasma genitalium, M. mulieris, and S. sanguinegens, M-PCR values of 100% were observed for all other parameters. For both U. urealyticum and M. type 1, M-PCR showed 98.60% NPV and 98.70% ACC. These bacteria differed in sensitivity; 83.30% for U. urealyticum and 75.0% for M. type 1. In four semen samples (9.5%), coinfections were detected in both M-PCR and sPCR assays (100% agreement with sPCR) (Supplementary Table 2). Figure 2A–C shows M-PCR amplification fragments of positive semen samples for different KM-BV in an 8% polyacrylamide gel.

Electrophoretic analysis of amplified fragments using multiplex polymerase chain reaction (M-PCR) in 8% polyacrilamide gels.
KM-BVs were detected in 146 of the 229 semen samples (63.8%), as a single agent or in coinfections. The most prevalent KM-BV was G. vaginalis (n = 116 samples, 50.7%) as a single agent (n = 10, 43.6%) and in coinfections (n = 14, 6.11%). Exclusive infection by KM-BV other than G. vaginalis occurred (n = 25, 10.92%) (Table 1) as follows: M. curtisii (n = 12, 5.24%); U. urealyticum (n = 9, 3.93%); M. type 1 and M. hominis (n = 7, 3.10% each); M. mulieris (n = 4, 1.80%); BVAB2, A. vaginae, and B. fragilis (n = 3, 1.31% each); and M. genitalium and BVAB1 (n = 2, 0.90% each). As previously stated, BVAB3 and S. sanguinegens were not detected (Table 2).
Presence of KM-BV in Total Semen Samples (n = 229).

Note. KM-BV = key markers of bacterial vaginosis. aPercentage not sum to total due to rounding.
Total Individual or Simultaneous KM-BV Detected by M-PCR Assays.

Note. M-PCR = multiplex polymerase chain reaction; KM-BV = key markers of bacterial vaginosis; BVABs 1, 2 and 3 = bacterial vaginosis-associated bacteria 1, 2, and 3.
KM-BVs were detected as a single agent in 127 semen samples (55.5% of total samples and 87% of total KM-BV-positive samples). KM-BV coinfections were detected in 19 semen samples (8.6% of total semen samples and 13.01% of KM-BV-positive samples; Table 2).
Table 3 displays the correlation between KM-BV-positive semen samples and adverse effects on seminal parameters. It also presents results of bacteria that individually showed association with semen alterations. Total KM-BV-positive samples showed a tendency to be correlated with teratozoospermia (p = .08). Individual KM-BV bacteria that showed significant association with some abnormal semen parameters were as follows: G. vaginalis was associated with increased seminal viscosity (p = .03), B. fragilis was associated with reduced seminal viscosity (p = .04) and with increased seminal coagulation time (p = .003), U. urealyticum was associated with teratozoospermia (p = .022), and M. mulieris was associated with decreased seminal viscosity (p = .01) (χ2 test, Table 3).
Correlation Between Testing KM-BV Positivity and Changes to Seminal Parameters.
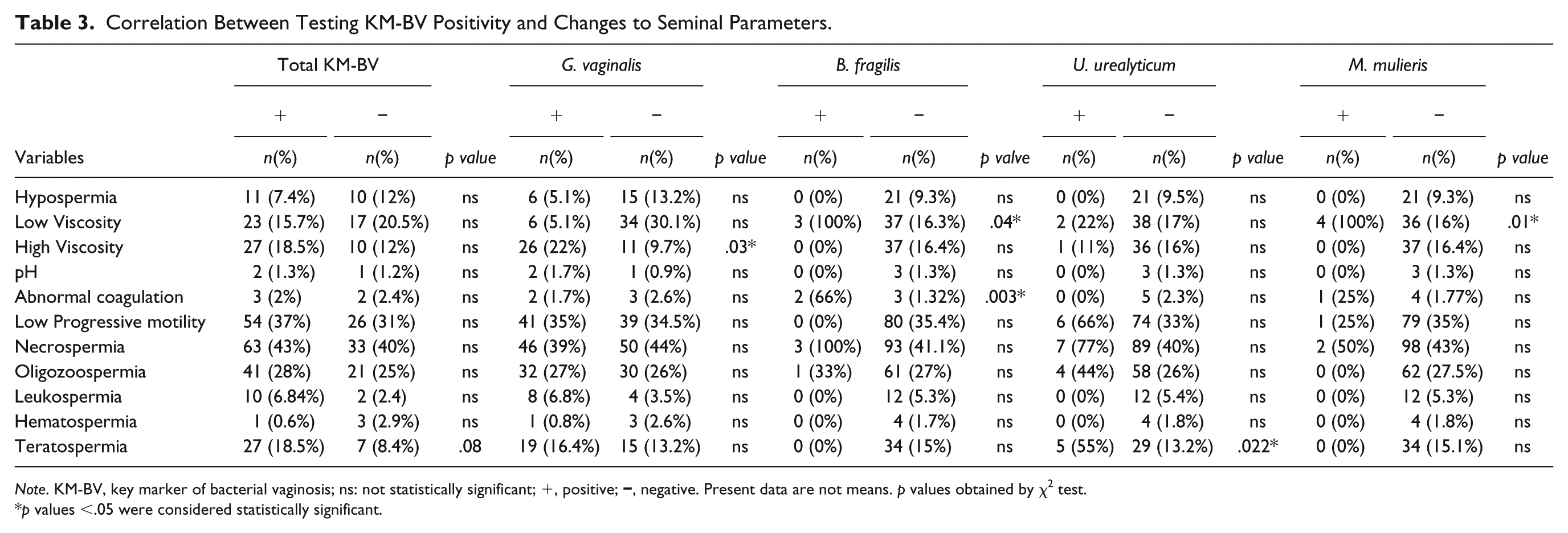
Note. KM-BV, key marker of bacterial vaginosis; ns: not statistically significant; +, positive;
p values <.05 were considered statistically significant.
Table 4 presents mean participant ages and seminal parameters in the total study population and in strata by KM-BV positivity (total KM-BV-negative samples and mutually exclusive KM-BV-positive subgroups). The presence of KM-BV was not related to the mean age of the participants. The following significant associations were observed: B. fragilis was associated with increased coagulation time (p = .0001); A. vaginae was associated with decreased coagulation time (p = .0001); and BVAB1 was associated with increased mean semen pH (p = .006), increased coagulation time (p = .0001), and decreased mean seminal volume (p = .058). M. mulieris was associated with increased mean semen pH (p = .006). M. genitalium was associated with increased mean semen pH (p = .006) and increased coagulation time (p = .0001) (one-way ANOVA, Table 4).
Mean Participant Age Seminal Parameters in the Study Population (n = 229) and in Strata by KM-BV Positivity.

Note. KM-BV, key markers of bacterial vaginosis. Presented data are means (95% CI); p value was obtained by one-way ANOVA, comparing the negative group (control) with the positive mutually excluded groups.
p values <.05 were considered statistically significant.
Discussion
To the best of the knowledge, this study was the first to use M-PCR assays for determining the prevalence of several KM-BV in the semen of randomized male partners of couples seeking fertility evaluation. It was also the first to examine the possibility that KM-BV could affect seminal parameters and thus fertility. The results presented high prevalence of KM-BV, particularly G. vaginalis, in semen. Furthermore, were demonstrated some important associations between the presence of KM-BV in semen and changes in seminal parameters. These changes potentially play significant roles in male subfertility and/or infertility.
In total, a high prevalence of KM-BV was detected in semen samples from male partners of infertile couples (63.8%). In addition, individual analysis of each KM-BV revealed that 50.6% of the total samples were positive for G. vaginalis, as a single infection or via coinfections. This prevalence was much higher than that described previously by De Francesco, Negrini, Ravizzola, Galli, and Manca (2011) wich reported that G. vaginalis was also the most prevalent bacterium in their semen culture; however, it was recorded in only 6.32% samples. Andrade-Rocha (2009) also detected G. vaginalis at a lower prevalence (22.2%) than this study. However, contrary to the results presented here, De Francesco et al. (2011) reported significant association with the presence of leukocytes only in patients with seminal infection by G. vaginalis. Additionally, these authors reported reduction in sperm concentration, motility, and morphology compared with the controls with G. vaginalis infection. Similarly, this study identified an association between total KM-BV and teratozoospermia and an association between U. urealyticum infection and teratozoospermia. However, an association of teratozoospermia specifically with G. vaginalis infection was not observed.
Furthermore, substantial research has associated U. urealyticum and Mycoplasma hominis infections with deterioration of semen parameters (De Francesco et al., 2011; Potts et al., 2000; Reichart, Kahane, & Bartoov, 2000). In a report by Gdoura et al. (2007) the prevalence of mixed species of mycoplasmas and ureaplasmas in semen was 6.7% comparable to that demonstrated in this study. However, when comparing infected and uninfected samples, Al-Sweih, Al-fadli, Omu, and Rotimi (2012) showed an association between Mycoplasma hominis infection and higher leukocyte count, lower sperm vitality, and reduced progressive motility, respectively. These associations were not demonstrated in this study.
Two other studies support the hypothesis that G. vaginalis may negatively influence seminal fertility parameters. Virecoulon et al. (2005) identified that G. vaginalis colonizes semen from men attending infertility clinics in high proportions (44%). Mändar, (2013) reported that the predominance of G. vaginalis in the female partner was significantly related to inflammation in the male genital tract. On the other hand, the results presented here indicated that G. vaginalis semen positivity may be associated with increased seminal viscosity, suggesting that the infection affects prostate function and alters coagulation–liquefaction of semen.
The second most frequent KM-BV was M. mulieris (5.26%). M. curtisii was detected at a much lower prevalence (1.4%). To the best of the knowledge, this was the first molecular analysis of M. curtisii and M. mulieris in semen samples. The results indicated that M. mulieris semen infection is correlated with decreased seminal viscosity and increased mean pH. This suggests that M. mulieris disrupts function of seminal vesicles and is responsible for alterations in the coagulation–liquefaction of semen, as observed in G. vaginalis infection.
U. urealyticum was detected in 4.56% samples, M. type 1 in 3.85% samples, Mycoplasma hominis in 2.8%, BVAB2 and M. mulieris in 1.4% each, B. fragilis and A. vaginae in 1.05% each, and BVAB1 in 0.7% of all samples. Furthermore, other KM-BV that showed an association with impairment of seminal parameters were as follow: B. fragilis with reduced seminal viscosity and increased coagulation time; BVAB1 with increased mean semen pH, increased coagulation time, and reduced mean seminal volume; M. genitalium with increased mean semen pH and increased coagulation time; and A. vaginae with decreased coagulation time.
A few important points deserve further discussion. Increased viscosity and abnormal coagulation times observed in B. fragilis infections may be due to prostatic gland infection. Benway and Moon (2008) previously reported isolation of B. fragilis in patients with prostatic abscesses. Therefore, among KM-BV, it may be important to study B. fragilis in isolation. Atopobium spp. was not associated with urethritis but was detected at a higher frequency (8.1%) by Manhart et al. (2013) than in this study (1.31%). Megasphaera spp. was only detected in men with urethritis, but at a lower frequency (0.4%) than presented in this study (3.10%).
Although total prevalence of KM-BVs in semen was high, the low presence of non-G. vaginalis bacteria may have influenced the results of this study. Furthermore, even though positive controls and the β-globin gene were correctly amplified in M-PCR and sPCR assays, BVAB3 and S. sanguinegens were not detected in the analyzed semen samples. This suggests that negative results for BVAB3 and S. sanguinegens were true negatives. Contrary, Manhart et al. (2013) detected Sneathia spp. and BVAB3 in semen samples. In their study, infection with Sneathia spp. was significantly associated with urethritis and BVAB3 was only detected in men with urethritis. To establish the effects of low prevalence non-G. vaginalis KM-BVs in male subfertility or infertility, it is necessary that future studies have a greater sample size.
Overall, the results presented here indicate most KM-BVs, as single infections or via coinfections, seem to affect the sperm and seminal fluid, that is, two main components related to male fertility. In particular, male fertility depends on two major components of semen: healthy spermatozoa (as measured by vitality, motility, and morphology) and composition of the seminal fluid, which is important for sperm function. Secretory products from seminal vesicles and the prostate are crucial for sperm motility, viability, and chromatin stability. They are very important for semen coagulation and liquefaction. Seminal vesicles secrete the major fraction of the ejaculate (≈60%) (WHO, 2010) and produce compounds, such as fructose, semenogelin-I (the predominant component of the coagulum), and sperm motility stimulators, that play significant roles in seminal physiology (Bukovsky, Thaler, & McIntyre, 1991; Gonzales, 2001; Robert & Gagnon, 1995). Seminal hyper-viscosity suggests deficient secretory activity in the seminal vesicles. The prostate secretes 30%–35% of the ejaculate and produces several compounds that are available for analysis in the seminal plasma, including enzymes for semen liquefaction. Reduced levels of prostate markers are indicative of glandular dysfunction, often associated with abnormalities in pH (⩾7.8), volume (decreased or increased), liquefaction, and/or viscosity (WHO, 2010).
Considering the findings of this study and the current evidence in literature, it can be reasonably hypothesized the mechanisms underlying the effects of KM-BV on the spermatozoa and seminal fluid: (a) bacterial gametotoxicity hampering spermatogenesis, increasing the number of apoptotic cells, and altering the sperm morphology; (b) inflammatory response to KM-BV causing pathologic lesions, leading to seminal vesicle and/or prostate dysfunction and changes in seminal fluid composition; (c) bacterial colonization and/or infection inducing an immune response against KM-BV that may cross-react with the host’s tissues affecting male genital tract, spermatogenesis, and seminal fluid; and (d) bacterial activity resulting in epigenetic and DNA damage during spermatogenesis, among many other reasons.
Limitations of this study include absence of data on mixed antiglobulin reaction testing, which was not conducted during each primary semen analysis. This test was performed upon the physician’s request only. Second, as this study did not include an analysis of medical records, it does not include data on the female partner’s characteristics, such as age, hormone levels, tubes, and pelvic status. In addition, other male partner characteristics, such as clinical analysis of the prostate and seminal vesicles, are not included. Furthermore, semen results were taken from only one semen analysis per subject and did not include a control group of normally fertile subjects. Finally, although the detection of KM-BV was associated with changes in seminal parameters, this association happened with a small number of patients in each subgroup. However, these results may encourage further studies to evaluate the influence of these 13 KM-BV on semen and changes in seminal parameters.
Conclusions
This study identified that KM-BV is commonly present in the semen of males seeking fertility evaluation. Furthermore, were demonstrated some important associations between the presence of KM-BV in semen and changes in seminal parameters. Therefore, screening for KM-BV in the semen should be considered as a diagnostic tool in cases of unexplained infertility, assisted reproduction, and for semen donation to prevent unnecessary bacterial transmission to the female genital tract.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Male Partners of Infertile Couples With Seminal Positivity for Markers of Bacterial Vaginosis Have Impaired Fertility
Supplemental material, Supplementary_Table_1 for Male Partners of Infertile Couples With Seminal Positivity for Markers of Bacterial Vaginosis Have Impaired Fertility by Edilson Damke, Fábio A. Kurscheidt, Mary M.T. Irie, Fabrícia Gimenes and Marcia E. L. Consolaro in American Journal of Men’s Health
Supplemental Material
Supplementary_Table_2 – Supplemental material for Male Partners of Infertile Couples With Seminal Positivity for Markers of Bacterial Vaginosis Have Impaired Fertility
Supplemental material, Supplementary_Table_2 for Male Partners of Infertile Couples With Seminal Positivity for Markers of Bacterial Vaginosis Have Impaired Fertility by Edilson Damke, Fábio A. Kurscheidt, Mary M.T. Irie, Fabrícia Gimenes and Marcia E. L. Consolaro in American Journal of Men’s Health
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazilian Government.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
