Abstract
Intrauterine insemination (IUI) is a treatment of choice compared with other invasive and expensive techniques of assisted reproduction. Sperm quality is used to predict its outcome and success. Establishing threshold levels for sperm parameters is useful to avoid spending time and money to do other assisted reproductive techniques. This study was carried out to compare various semen parameters in a group of men eligible to participate in an IUI program with those of fertile men whose wives were pregnant at the time of the study. Two hundred and thirty-four semen samples were evaluated from subfertile men whose partners were candidates for IUI and 234 semen samples were evaluated from fertile men whose partners were pregnant less than 12 weeks. To assess the sensitivity and specificity of the main semen parameters, receiver operating characteristic (ROC) curves were used. Normal sperm morphology is more sensitive and specific compared with its progressive motility and concentration. No significant differences in various semen parameters of fertile men and those of the male partners of IUI candidates were observed. ROC analysis identified that sperm normal morphology using strict criteria may be a good indicator of fertility status in men. No significant difference in various semen parameters between the male partners of IUI candidates and the fertile men was seen. However, utilizing ROC curves, sperm morphology using strict criteria could be a good predictor of fertility.
Introduction
Approximately 15% of couples cannot have a child after 1 year of regular unprotected intercourse. Male infertility is solely responsible in about 20% of infertile couples, but it is also a contributor in another 30% to 40% (Thonneau et al., 1991). Semen analysis is one of the most important laboratory tests in the investigation of men’s fertility status (Khalili, Aghaie-Maybodi, Anvari, & Talebi, 2006). Semen analysis, which contains the main criteria for the quality of semen such as sperm count, sperm motility, and the percentage of sperm with normal morphology, is the basis of the laboratory evaluation of infertile men (Badawy, Elnashar, & Eltotongy, 2009; Merviel et al., 2010).
The intrauterine insemination (IUI) method, which is used to increase the quality and quantity of sperms at the site of fertilization, is a noninvasive, less expensive, and more acceptable treatment compared with other complex assisted reproduction techniques (ART; Dadkhah, Nahabidian, & Ahmadi, 2004). IUI has a variety of indications such as unexplained infertility, congenital anomalies of the genital tract, erectile dysfunction, retrograde ejaculation, antisperm antibodies, female sexual dysfunction (vaginismus), cervical factors, and infertility caused by endometriosis (Marchetti & Dewailly, 2006).
Semen quality and several seminal parameters such as sperm motility and their normal morphology might be strong predictors of IUI success (Ombelet, Dhont, Thijssen, Bosmans, & Kruger, 2014; van Weert et al., 2004; Zhang, Tao, Xing, Cai, & Zhang, 2014). Several studies have reported that total motile sperm count, especially its progressive motility, is the best predictor of pregnancy after sperm processing (Miller et al., 2002; Zhao et al., 2004). Farimani and Amiri (2012) have revealed that male factors do not correlate with the treatment outcome. Reference values for semen parameters are not precise guides for fertility in men, while fertility is reported in men who do not have at least these criteria. Infertility can be seen in men with normal semen parameters. Difficulties in establishing thresholds to define fertile and infertile men have made the diagnostic value of a semen analysis controversial (Van der Merwe, Kruger, Oehninger, & Lombard, 2005). Previous studies have demonstrated that different geographical areas have variable values for semen parameters (Fisch & Goluboff, 1996), probably related to environmental factors. However, ethnic or genetic differences cannot be excluded (Jørgensen et al., 2001; Swan et al., 2003) so each laboratory should determine its own reference range for each semen variable (Haugen, Egeland, & Magnus, 2006).
Haugen et al. (2006) suggested that World Health Organization (WHO) cutoff values are not sufficient to label fertility status in men everywhere around the world and these values differ in each region (Fisch & Goluboff, 1996; Jørgensen et al., 2001; Swan et al., 2003). It seems that each geographical area requires certain criteria as reference values to evaluate the fertility rate of the residents. Thus, in different regions of the world, regional studies are being conducted on semen parameters to seek standards for each geographical area separately (Haugen et al., 2006; Jørgensen et al., 2001; Menkveld et al., 2001; Swan et al., 2003).
A review by Ombelet et al. (2014) identified a serious need for more comprehensive and more accurate studies considering the prognostic value of semen parameters on IUI pregnancy rate. Around the world, clinical approaches associated with ART would benefit from determining threshold levels for semen variables. IUI pregnancy rate can increase above these levels and it is unlikely that pregnancy would occur below these levels. Because of the lack of region-specific standards for semen parameters, and different IUI treatment strategies available for each patient (Ombelet et al., 2014), this study investigated such threshold levels in a region of Iran to compare fertile and subfertile populations in order to define normal values for different semen parameters.
Materials and Method
From October 2014 until March 2015, 606 couples who were pregnant less than 12 weeks were consulted. Two hundred and thirty-four male participants agreed to participate in the study and donated semen samples for the research as a control group. All couples in the control group had become pregnant within 1 year of unprotected coitus. During the same period, 234 consecutive couples who were referred to the Fertility and Infertility Center of Beheshti Hospital with a history of subfertility for at least 13 months were chosen as part of the case group. The IUI candidate couples had a history of idiopathic subfertility. The age range was 16 to 35 years for women and 20 to 45 years for men in both fertile and IUI candidate groups. All participants were educated on the study protocol and completed a consent form before participating in the study.
All participants were instructed to abstain from sexual activity for 2 to 7 days before semen collection (Khalili, Adib, Halvaei, & Nabi, 2014). The specimens were collected at the participant’s home or in the Beheshti Hospital’s Andrology Laboratory by masturbation or by intercourse using special semen collection condoms that do not contain substances such as latex rubber that are detrimental to sperm. The semen specimens were kept at room or body temperature and were examined within half an hour of collection. After complete liquefaction, all samples were evaluated in a blinded fashion by a qualified person to prevent the interobserver variation based on WHO (2010) criteria. The concentration, motility, and percentage of spermatozoa was evaluated with normal forms in all samples based on these criteria.
The receiver operating characteristic (ROC) curve was used to evaluate the sensitivity and specificity of each semen parameter from both fertile and subfertile couples using fertility status as the standard (Kruger et al., 1986; Ombelet et al., 1997). The data were analyzed by SPSS software version 2.0. Minimum and mean values were recorded for each semen parameter.
Results
A total of 468 semen samples were evaluated, 234 samples from subfertile couples which were referred for IUI, and 234 samples from couples who were pregnant less than 12 weeks. There was no significant difference between the mean age of men (
Frequencies of Parameters in Natural Fertile Group: Statistics.

Frequencies of Parameters in Intrauterine Insemination Candidate Group: Statistics.

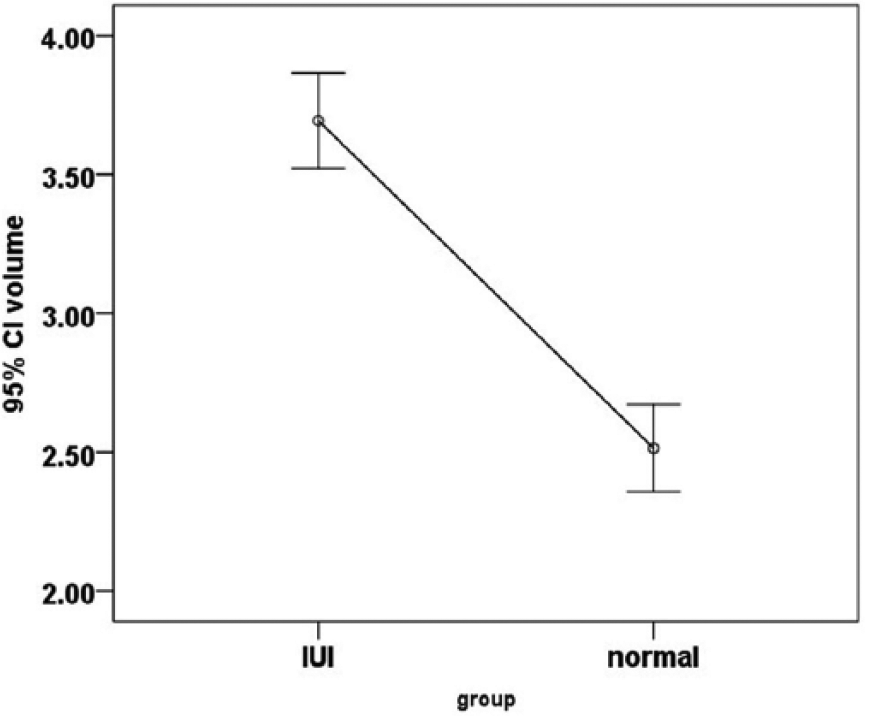
Mean and confidence interval (CI) of the semen volume in fertile (normal) and subfertile (intrauterine insemination [IUI]) groups.
Descriptive Statistics of the Sperm Parameters in Fertile and Subfertile Groups (


Mean and confidence interval (CI) of the sperm concentration in fertile (normal) and subfertile (intrauterine insemination [IUI]) groups.

Correlation between the mean volume of the semen and the mean of the sperm concentration.
The mean percentage of sperms with normal morphology in fertile and subfertile groups was 12.7 ± 5.2 and 11.5 ± 6.5, respectively (
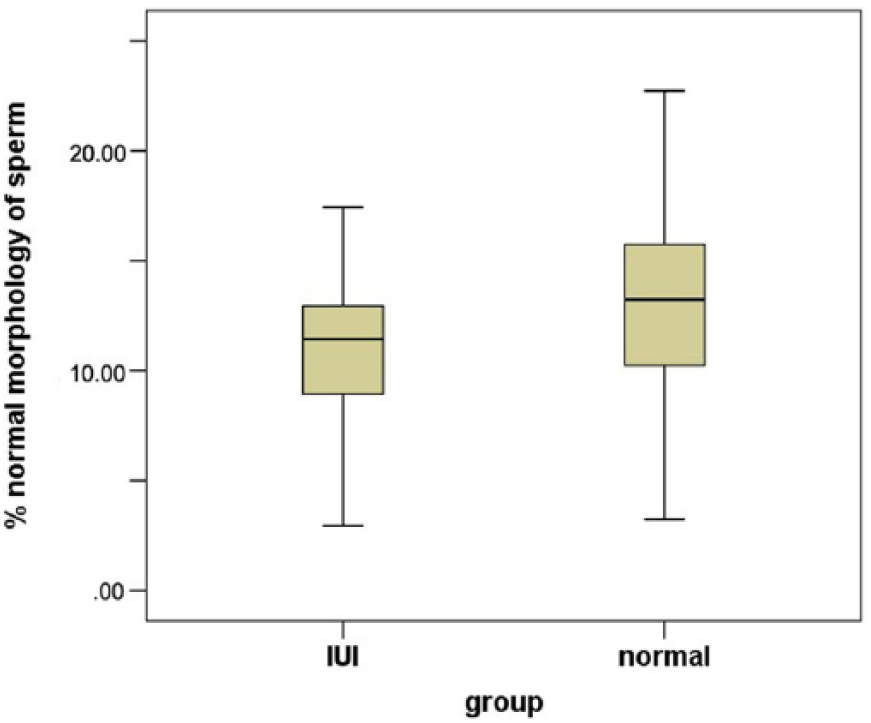
Twenty-fifth and 75th percentiles, plus the median and range of the sperms with fast progressive motility in fertile (normal) and subfertile (intrauterine insemination [IUI]) groups.

Twenty-fifth and 75th percentiles, plus the median and range of the sperms with slow progressive motility in fertile (normal) and subfertile (intrauterine insemination [IUI]) groups.
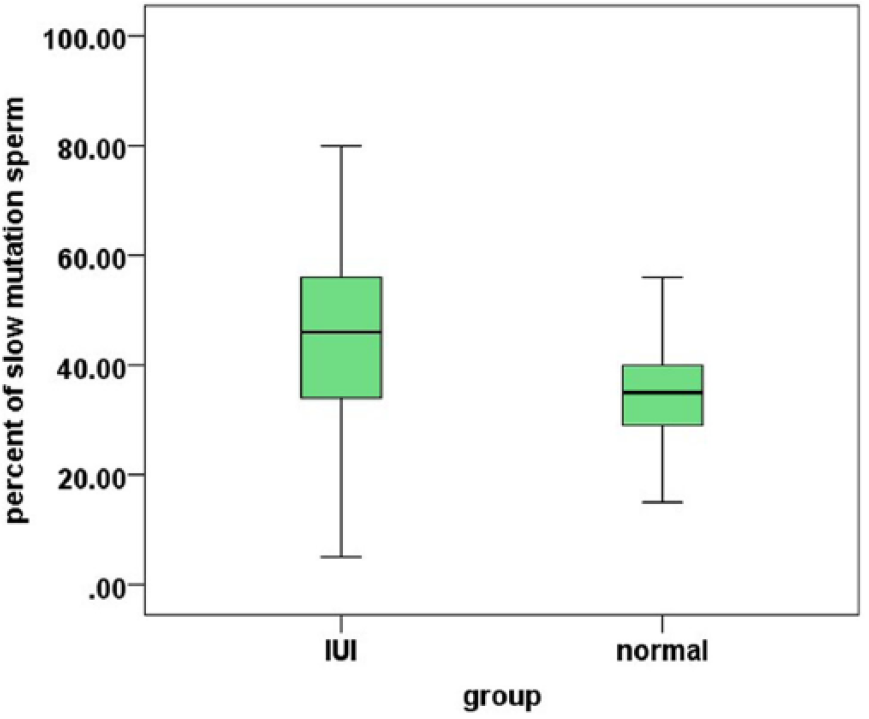
Twenty-fifth and 75th percentiles, plus the median and range of the sperms with fast progressive motility in fertile (normal) and subfertile (intrauterine insemination [IUI]) groups.

Area under the receiver operating characteristic (ROC) curve for semen parameters.
Area Under the ROC Curve for Main Semen Parameters.

Under the nonparametric assumption. bNull hypothesis: True area = 0.5.
Discussion
This study evaluated semen parameters in IUI candidates and fertile couples. Based on the findings, there was no significant difference between the main parameters of semen and sperm, such as semen volume, sperm concentration, motility, and morphology between the two groups. ROC analysis revealed that sperm morphology has the highest diagnostic value for prediction of fertility rate. Semen analysis is the most important diagnostic tool of male fertility. When parameters are unfavorable, no pregnancies occur. Both sperm count and total motile sperm count after processing have a significant relationship with pregnancy rate. The main guide for semen analysis assessment in recent years has been the WHO criteria (Kruger et al., 1986).
IUI is widely used for infertile couples’ treatment. When this method is properly selected with regard to sperm parameters and female factors, there is a great chance of success. Nevertheless, the selection of appropriate ovarian stimulation is very important (Merviel et al., 2010). Success rates of IUI improve linearly with progress in sperm processing methods which increases sperm count and total motile sperm count (Kruger et al., 1986). Semen quality is a main factor to predict IUI success rate (Ombelet et al., 2014) and among the semen parameters, sperm motility is important (Shulman et al., 1998). Studies have confirmed that progressive motility and total motile sperm count after sperm processing are the best predictors of pregnancy (Miller et al., 2002; van Weert et al., 2004; Zhao et al., 2004). In most cases of infertility such as unexplained, cervical, and male factor subfertility, IUI with homologous semen is the first choice of treatment. Other methods of assisted reproduction are more invasive and expensive (Ombelet et al., 2014).
Previous reports have been controversial about the threshold values of the main sperm parameters related to semen characteristics for IUI success (Badawy et al., 2009; Dorjpurev et al., 2011; Merviel et al., 2010; Wainer et al., 2004). Because there is no agreement on the ideal levels of these items, including sperm concentration and motility, it is difficult to guess which couples would benefit from this procedure. In view of this point, gynecologists who specialize in the human reproduction field prefer to indicate three to six or more cycles of IUI before attempting more complex techniques (Merviel et al., 2010). Another important issue is the variations in sperm counts between different geographical regions (Fisch & Goluboff, 1996).
Dorjpurev et al. (2011) have mentioned the motility of ejaculated sperm as an important success factor for natural pregnancy. Shulman et al. (1998) demonstrated that the number of highly motile sperm and their survival are the most important parameters for assessing their fertilizing ability in successful insemination. Brasch, Rawlins, Tarchala, and Radwanska (1994) and Campana et al. (1996) reported that there is a significant association between total motile sperm count and pregnancy rate.
In this regard, Merviel et al. (2010) reported a concentration of more than five million motile sperm and teratospermia less than 70% following semen processing as the most important predictive factors. Success of IUI for male factor subfertility is strongly reduced when a woman’s age is more than 35 years, less than five million motile sperm are present in the semen sample and normal sperm morphology is less than 30% (Badawy et al., 2009). Results of Dorjpurev et al.’s (2011) study were the same as above. Wainer et al. (2004) reported these levels at 30% for normal sperm morphology, but 10 million/ml for sperm motility in male subfertility cases.
Conversely, other studies have demonstrated that sperm morphology alone, before or after preparation, do not help predict IUI outcomes (Brasch et al., 1994; Miller et al., 2002; Zhao et al., 2004). Similar to the current findings, Akl et al. (2011) reported that they reached 65% success rate following IUI with an optimum cutoff point of 2% for the percentage of morphologically normal sperms. In line with Merviel et al. (2010), Zhang et al. (2014) evaluated infertile couples with male factors. Zhang et al. (2014) reported poor outcomes for pregnancy rates after IUI procedures in couples with sperm counts less than five million. After three unsuccessful IUI cycles for each couple, they preferred to work on other methods of ART.
The WHO modified normal morphology cutoff values to more than 4% in the 2010 WHO manual (Shulman et al., 1998). Before this, Karabinus and Gelety (1997) observed no association between sperm morphology and the result of IUI and mentioned that there is still a controversy regarding the definition of a morphologically normal sperm. In support of this statement, another study revealed low probability of pregnancy rate in samples with less than 10% normal morphology of sperms. Stanford, Mikolajczyk, Lynch, and Simonsen (2010) recommended other methods of ART (i.e., in vitro fertilization and/or intracytoplasmic sperm injection). In 1999, the critical percentage of normal morphology was 14% (Natali et al., 2013). In 2014, Li et al. concluded that when normal sperm morphology rate is less than 4%, intracytoplasmic sperm injection method is preferred and when this item becomes equal or more than 4%, in vitro fertilization technique should be considered first.
Knowing the importance of sperm motility, especially after processing, can determine which couples benefit from ART. A meta-analysis by van Weert et al. (2004) confirmed this and demonstrated that sperm motility after processing could be used as a useful factor to choose patients for either IUI or in vitro fertilization. In the current study, the area under the curve for morphology value was near 0.8 so this value can be considered to have a higher sensitivity and specificity than the other parameters.
Conclusion
Many variables may influence success rates after IUI treatment procedures. Scientists from different geographical regions reported variable results on the main effective parameters. Although there was no significant difference between the main semen and sperm parameters, it is obvious that first, normal morphology of the sperms and, second, their progressive motility have higher sensitivity and specificity for prediction of IUI success according to the ROC data. The purpose of this study was to prevent unnecessary cycles and reduce the waiting time to pregnancy and childbirth. The results could have benefits in saving time and costs for infertile couples who are referred to ART laboratories. More research is needed to identify differences in the cutoff values in semen parameters.
Footnotes
Acknowledgements
The authors express their gratitude to Isfahan University of Medical Sciences for budgeting this research (Project number 293281) and the Andrology Laboratory of Isfahan Beheshti Hospital for their collaboration. We would like to thank Mr. Ali Mehrabi Koshki for his cooperation with biostatistics in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Isfahan University of Medical Sciences (Project number 293281).
