Abstract
To investigate the potential use of cyclooxygenase-2 (COX-2) inhibitors in the treatment of lower urinary tract symptoms (LUTS) in male patients, we conducted a comprehensive meta-analysis. Our study involved the identification and collection of randomized controlled trials (RCTs) from leading databases including PubMed, MEDLINE, EMBASE, and Cochrane Library. The primary objective of this analysis was to evaluate the effectiveness of COX-2 inhibitors for the treatment of LUTS. Our analysis involved six short-term (within 3 months) RCTs involving 707 patients. We found that COX-2 inhibitor treatment significantly improved the International Prostate Symptom Score (IPSS) of patients (mean difference [MD] = −2.99, 95% confidence interval (CI): −3.65 to −2.33, p < .00001), nocturia frequency (MD = −1.90; 95% CI: −3.18 to −0.61, p = .004), and maximum flow rate (Qmax) (MD = 1.02; 95% CI: 0.06 to 1.98, p = .04). However, no significant differences were found between patients in terms of changes in prostate-specific antigen (PSA) (MD = 0.02; 95% CI: −0.39 to 0.43, p = .92) and total prostate volume (TPV) (MD = −2.93; 95% CI: −6.45 to 0.59, p = .10). Therefore COX-2 inhibitors are an effective treatment for LUTS.
Introduction
Benign prostatic hyperplasia (BPH) is a significant contributor to lower urinary tract symptoms (LUTS) in elderly men (Garraway et al., 1991), presenting with a range of symptoms such as urinary hesitancy, urgency, increased daytime frequency, nocturia, incontinence, and dysuria (Chapple et al., 2008; D’Ancona et al., 2019). The main treatment methods recommended by the guidelines are 5-α reductase inhibitors (5-ARI) and α-1 receptor blocker. The 5-ARI blocks the action of 5-arylreductase, the enzyme that converts testosterone to dihydrotestosterone (DHT), which ordinarily stimulates prostate growth, and lowers prostate size to alleviate blockage symptoms (McConnell et al., 1992; Russo et al., 2021). α-1 receptor blockers are thought to lower prostate tone and bladder outlet blockage by blocking the action of endogenously produced norepinephrine on prostate smooth muscle cells (Gorgel et al., 2013). Furthermore, α-1 adrenergic receptor-mediated contraction of the human prostate is dominant (Michel & Vrydag, 2006). Now, the preferred treatment for LUTS remains α receptor blockers.
Cyclooxygenase (COX) is a key enzyme in the inflammatory process, which converts arachidonic acid into prostaglandins and other eicosanoids (Ozdemir et al., 2009). Several clinical trials comparing combination therapy with monotherapy for BPH have been done, with encouraging outcomes for the combination therapy. In one of the clinical trials, combination therapy of tamsulosin and meloxicam, a selective COX-2 inhibitor, produced greater results compared to tamsulosin treatment alone (Gorgel et al., 2013). Although COX-2 inhibitors have been gradually used in combination for the treatment of LUTS; however, there is a lack of evidence-based studies on the feasibility of using COX-2 inhibitors for the treatment of LUTS. Given their superior efficacy, COX-2 inhibitors hold promise as a new option for the treatment of LUTS.
In this systematic review and meta-analysis, we evaluate the short-term efficacy of COX-2 inhibitors in the treatment of LUTS, providing a feasible reference for clinical use while adhering to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist.
Materials and Methods
Search Strategy
Three authors independently employed the Picos strategy, which encompasses population, intervention, comparator, outcome, and study design, to identify randomized controlled trials (RCTs) associated with the treatment of LUTS with COX-2 inhibitors from PubMed, MEDLINE, EMBASE, and Cochrane Library databases. The search strategy has been succinctly summarized in Table 1. Our database search encompassed the use of the following search terms: LUTS, BPH, cyclooxygenase-2 inhibitor, and RCTs. In addition, the references in the retrieved articles were also perused by three authors to identify additional relevant literature. It is pertinent to note that the search terms were not constrained by language or region and duplicate studies were excluded.
Details of the Included Studies
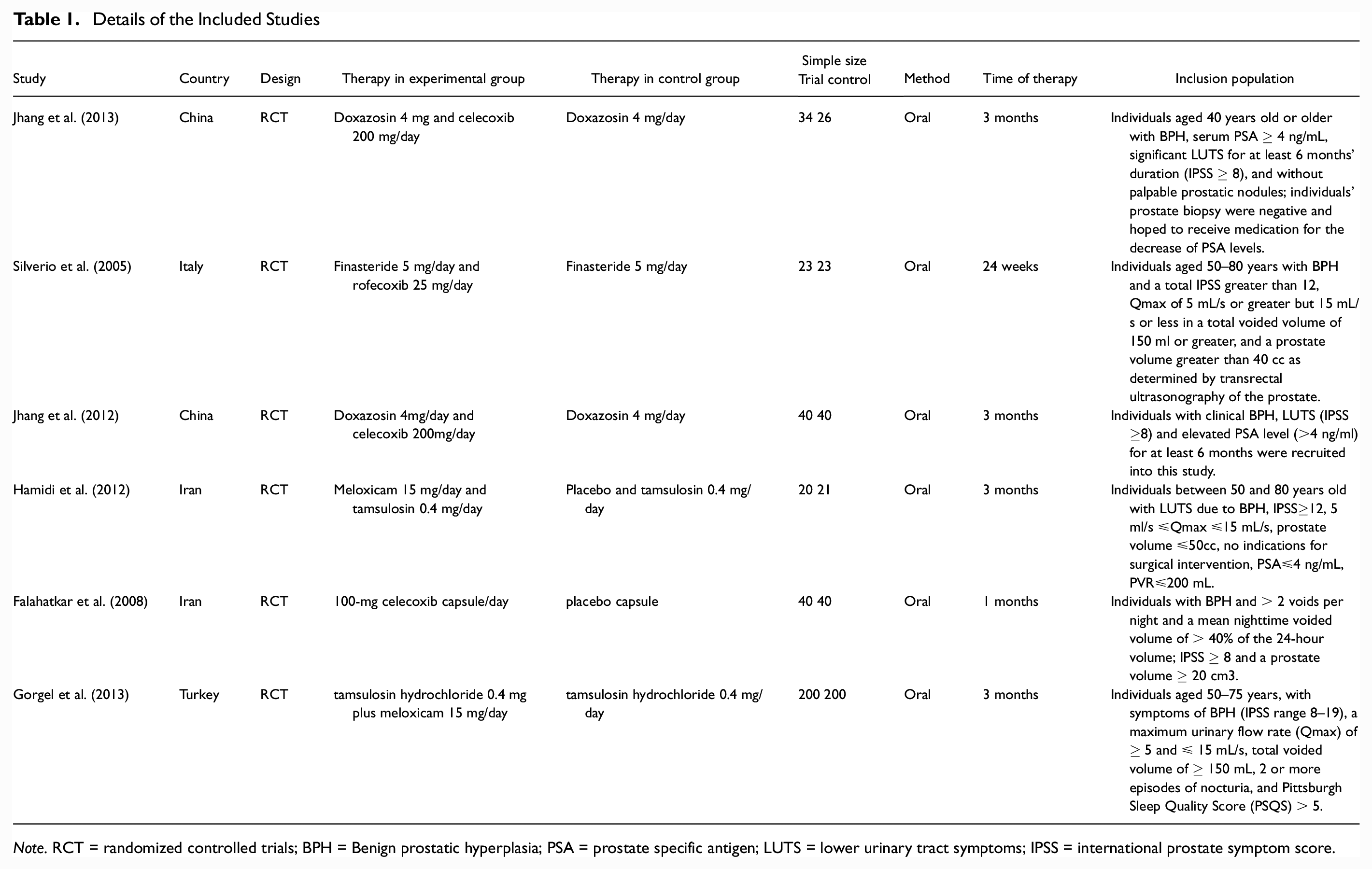
Note. RCT = randomized controlled trials; BPH = Benign prostatic hyperplasia; PSA = prostate specific antigen; LUTS = lower urinary tract symptoms; IPSS = international prostate symptom score.
Inclusion Criteria
To be considered for inclusion in our study, RCTs must meet the following stringent criteria: (a) they must have thoroughly examined the effects of COX-2 inhibitors on LUTS, and (b) they must have provided comprehensive and precise data, including participant sample size and outcomes for each measure. It should be noted that RCTs are subject to more exacting requirements for inclusion and exclusion than their prospective and retrospective counterparts.
Quality Assessment
We used the Jadad scale and the Cochrane risk of bias assessment tool to assess the quality of the selected RCTs (Cumpston et al., 2019). Our team has established a meticulous classification for the excellence of each conducted study, based on the following standards: (a) studies that fully satisfy all quality criteria and show minimal risk of bias; (b) studies that demonstrate the fulfillment of most quality criteria, albeit with a moderate level of bias; and (c) studies that only partially meet a few quality criteria and are therefore highly vulnerable to bias. The entire panel of authors concurred in reaching an elevated consensus regarding the employment of this classification system.
Data Extraction
We extracted a range of valuable information from each RCT: (a) name of the first author, (b) types of studies, (c) sample size of each group, (d) treatment modality, (e) dose and timing of treatment, and (d) study outcomes, including International Prostate Symptom Score (IPSS), maximum flow rate (Qmax), nocturia frequency, prostate-specific antigen (PSA) and total prostate volume (TPV).
Statistical and Meta-Analysis
We performed statistical analysis using Review Manager software (RevMan, version 5.3.0, Cochrane Collaboration Green & Higgins, 2008). The effectiveness of COX-2 inhibitors in treating LUTS was evaluated by IPSS, Qmax, TPV, PSA, and the frequency of nocturia. We employed both fixed and random effects models to assess these indicators, and utilized mean difference (MD) to analyze and interpret continuous data. For dichotomous outcomes, odds ratios (ORs) with 95% confidence intervals (CIs) (DerSimonian & Laird, 2015) were presented. The homogeneity study was deemed unified if p > .05. To investigate homogeneity, a fixed effects model was utilized, while a random effect model is suitable for examining heterogeneity. Furthermore, we employed the I2 statistical method to assess inconsistency. When the I2 value exceeds 50%, the random effect model is adopted. Statistical significance was defined as a p value < .05.
Results
Characteristics of Eligible Studies
After applying the inclusion/exclusion criteria, 234 scholarly works were culled from the database. Our initial filter entailed perusing titles and abstracts, which yielded the impartial removal of 209 publications. Subsequently, we rejected 19 papers from the remainder of the cohort on account of their lacking utility. Finally, in ensuring an unblemished dataset, we expunged 2 papers due to their inadvertent duplication. Finally, our study included 6 high-quality RCTs (Di Silverio et al., 2005; Falahatkar et al., 2008; Gorgel et al., 2013; Hamidi et al., 2012; Jhang et al., 2012; Jhang et al., 2013). Figure 1 presents the flowchart of the selection process. Table 1 embodies the study characteristics and patient characteristics.

Flowchart of the Study Selection Process
The Quality of Eligible Studies
The included studies were all RCTs, one of which was a randomized, double-blind controlled trial with a quality grade of A (Jhang et al., 2013). Five RCTs were rated grade B (Di Silverio et al., 2005; Falahatkar et al., 2008; Gorgel et al., 2013; Hamidi et al., 2012; Jhang et al., 2012). One study did not finish the entire follow-up period, and 46 patients were lost to follow-up. The bias in the articles was mainly caused by the type of COX-2 inhibitor as well as the different follow-up time. The quality assessment of the total studies is presented in Table 2.
Quality Assessment of Individual Study

Note. A = all quality criteria met (adequate): low risk of bias; B = most quality criteria met (adequate): moderate risk of bias; ANCOVA = analysis of covariance.
Efficacy
International Prostate Symptom Score
We analyzed the IPSS in 707 patients from 6 RCTs (Figure 2A). Since p > .05, we used the fixed effects model, which yielded an MD value of −2.99 (95% CI: −3.65 to −2.33, p < .00001). The results of this analysis showed that COX-2 inhibitors significantly reduced the IPSS of the patients.
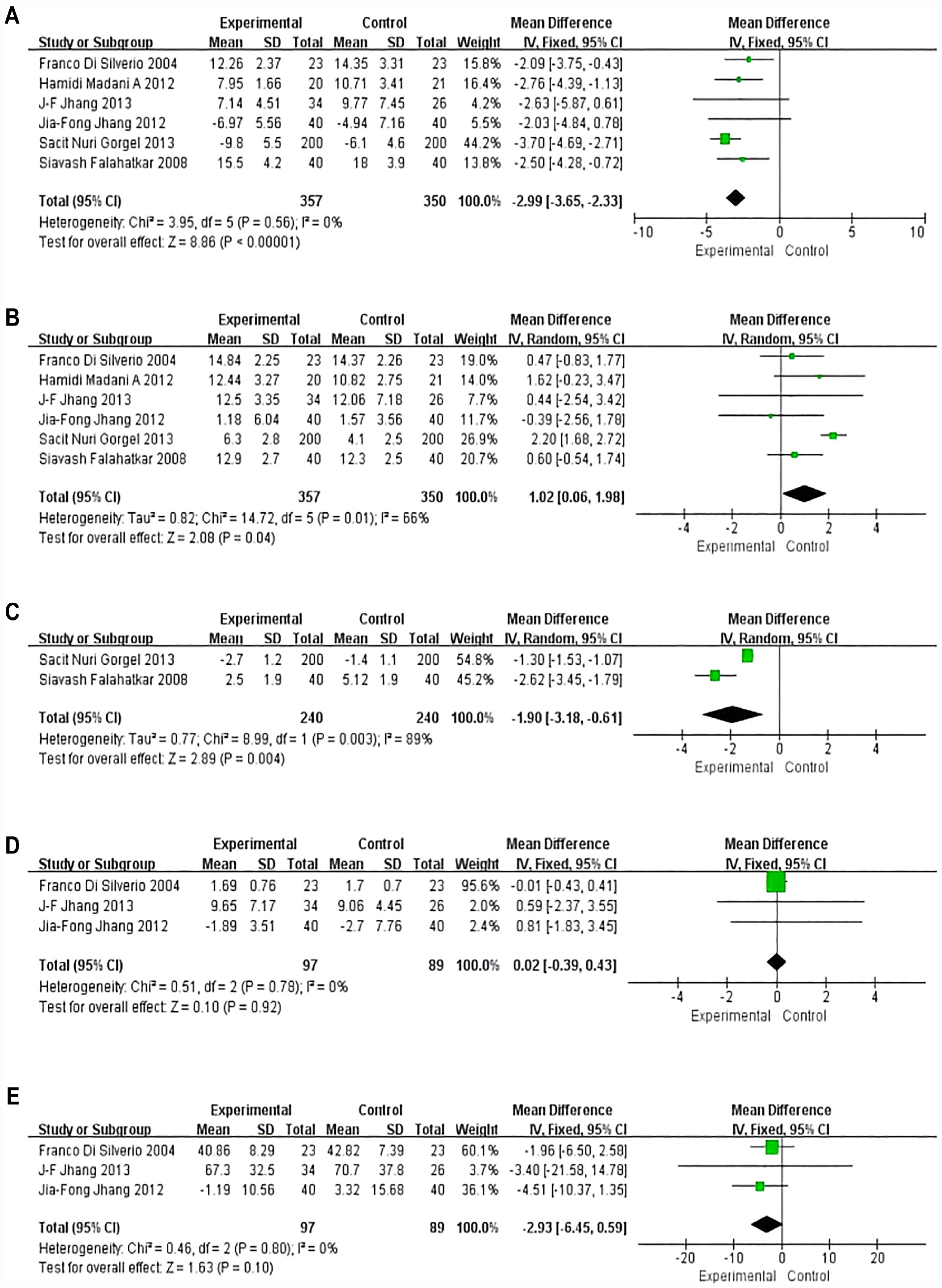
Forest Plots Showing Changes in (A) International Prostate Symptom Score, (B) Maximum Flow Rate, (C) Nocturia Requency, (D) Prostate Specific Antigen, and (E) Total Prostate Volume
Maximum Flow Rate
Six RCTs reported changes in Qmax in 707 patients (Figure 2B). The random effects model showed that patients who received COX-2 inhibitor combination therapy had a significantly better improvement for Qmax than the control group (MD = 1.02; 95% CI: 0.06 to 1.98, p = .04).
Nocturia Requency
Two RCTs investigated differences in nocturia frequency among 480 patients (Figure 2C). A random effects model showed that COX-2 inhibitors significantly improved symptoms in patients with nocturia frequency (MD = −1.90; 95% CI: −3.18 to −0.61, p = .004).
Prostate Specific Antigen
Three RCTs reported differences in PSA change in 186 patients (Figure 2D). The combined results from the fixed effects model showed that, with respect to PSA measures, there was no statistically significant difference between the group with the addition of COX-2 inhibitors and the control group (MD = 0.02; 95% CI: −0.39 to 0.43, p = .92).
Total Prostate Volume
Three RCTs reported changes in TPV in 186 patients (Figure 2E). The fixed effects model assessment was applied to show that there was no significant difference between the experimental and control groups in terms of TPV change (MD = −2.93; 95% CI: −6.45 to 0.59, p = .10).
Discussion
LUTS can be described as abnormal voiding sensations, the frequency or severity of which occur affecting quality of life. Common LUTS include urinary frequency, urgency, nocturia, intermittency, incomplete emptying, and decreased urine flow, with nocturia being the most common presenting symptom (Coyne et al., 2009; Irwin et al., 2006). These symptoms are commonly associated with irritating or obstructive voiding problems in men, such as BPH or bladder overactivity (Gammack, 2010). With increasing age, most symptoms become more frequent and severe (Irwin et al., 2006; Marszalek et al., 2009). In addition to LUTS, male may also experience a plethora of other sexual dysfunctions, including erectile dysfunction (ED), ejaculatory dysfunction, and premature ejaculation. Moreover, the severity of LUTS has been found to be intricately linked with the occurrence and intensity of ED (Barqawi et al., 2005; Wein et al., 2009). Given the overlapping nature of these symptoms with both benign and malignant conditions, it is of paramount importance to undertake a comprehensive evaluation to accurately discern the presence of any underlying pathology.
LUTS can be effectively addressed through a diverse range of pharmaceutical options, encompassing α-blockers, 5α-reductase inhibitors, antimuscular relaxants, phosphodiesterase type 5 inhibitors (PDE5I), COX-2 inhibitors, and botulinum toxin expertly administered via intradetrusor injection (Peyronnet et al., 2018). Notwithstanding, it remains incontrovertible that extant therapeutic modalities do not universally ameliorate LUTS in patients. Hence, the quest for novel treatments persists through experimental and clinical investigations. The COX is crucial in the inflammatory response, and exists in two isoforms, COX-1 and COX-2. The latter is especially implicated in bladder inflammation, with sustained COX-2 production observed in the context of chronic inflammation (Andersson, 2007; Caruso et al., 2009). COX-2 promotes cell growth and alters phenotype in association with its ability to increase prostaglandin synthesis, promote angiogenesis, inhibit apoptosis, and modulate inflammation in the pericellular microenvironment (Di Silverio et al., 2005). In normotensive, fluid homeostatic subjects, the role of prostaglandin in maintaining renal plasma flow is minimal (Dilger et al., 2002); however, the employment of prostaglandin to sustain renal plasma flow is progressively becoming more prevalent among individuals who present with fluid imbalance (renal, hepatic, or cardiac failure), hypertension, obstructive kidney disease, or advanced age (Alsalameh et al., 2003). BPH is primarily a disease of the prostate smooth muscle cells, and prostaglandin play an important role in smooth muscle cell activity and proliferation (Hussain et al., 2003). Despite such a strong association between inflammation, COX-2, and BPH, there are few clinical trials evaluating the efficacy of COX-2 inhibitors in LUTS and BPH (Di Silverio et al., 2005; Gorgel et al., 2013), and this was the primary objective of our study to provide some reference to the population who need to use this medicine.
The role of COX-2 inhibitors in LUTS has attracted increasing attention. For example, Ozdemir et al. conducted a retrospective study to demonstrate that combining α-receptor blockers with COX-2 inhibitors can minimize LUTS-related voiding and urinary storage symptoms, enhance urinary flow rates, and decrease postvoiding leftover urine volume (Ozdemir et al., 2009). Jhang et al. have reported that COX-2 inhibitors exhibit anti-inflammatory properties in the context of LUTS, BPH, and serum PSA elevation resulting from prostatic inflammation, as revealed by another study. (Jhang et al., 2013). Silverio et al. have reported in a recent study that the concurrent administration of COX-2 inhibitors and finasteride exhibits a pro-apoptotic effect on BPH tissues (Di Silverio et al., 2005). Furthermore, Falahatkar et al. demonstrated that the COX-2 Inhibitor Celecoxib is effective for the treatment of nocturia due to BPH (Falahatkar et al., 2008). Gorgel and colleagues have exhibited that the employment of α-receptor antagonists, when used alongside COX-2 inhibitors, could prove to be a secure and efficacious therapeutic alternative for addressing the symptoms of benign prostatic hyperplasia, including its adverse influence on sleep quality and nocturia (Gorgel et al., 2013).
In this analysis, we used IPSS to evaluate the treatment effect of patients (the total IPSS range from 0 to 35). The IPSS as a subjective evaluation of voiding symptom severity in individuals afflicted with BPH, and is frequently utilized to evaluate men experiencing LUTS or BPH. This tool proves particularly advantageous for longitudinal monitoring of shifts in symptom severity, as well as for gauging the effects of various treatments (Trafford Crump et al., 2019; Yuan et al., 2019).
In our meta-analysis, we meticulously scrutinized 6 RCTs encompassing a cohort of 707 patients with LUTS. Our primary objective was to assess the effectiveness of COX-2 inhibitors in managing LUTS. The integrated analysis revealed that the administration of COX-2 inhibitors was significantly more efficacious than the control group in ameliorating IPSS, Qmax, and frequency of nocturia. However, concerning PSA and TPV, no substantial differences were observed between the COX-2 inhibitor group and the control group. A previous study conducted by Ozdemir et al demonstrated that COX-2 inhibitors also exhibited a positive impact on the quality of life (QOL) score and overactive bladder symptom score (OABSS) (Ozdemir et al., 2009). The Pittsburgh sleep quality score was considerably higher in the COX-2 inhibitor group than in the control group in a previous RCT (Gorgel et al., 2013). Based on the findings of this meta-analysis, it is evident that COX-2 inhibitors present a significant improvement in the symptoms of LUTS in comparison to controls. Moreover, all the pertinent studies included in this analysis showed no evidence of serious adverse events. These compelling outcomes affirm the efficacy of COX-2 inhibitors as a successful short-term therapeutic modality for the management of LUTS.
Although no safety studies using COX-2 inhibitors for the treatment of LUTS were directly mentioned in these short-term RCTs, it was found in previous studies on COX-2 inhibitors that gastrointestinal reactions resulting from COX-2 inhibitors in non-steroidal anti-inflammatory drugs (NSAIDs) were mild (García-Rayado et al., 2018; Tacconelli et al., 2017). In addition, similar results have been observed in animal experiments (Langenbach et al., 1995; Sigthorsson et al., 2002). And COX-2 inhibitors do reduce the incidence of hypertension compared with other nonselective NSAIDs in terms of long-term effects (Ruschitzka et al., 2017). Whereas, in terms of gastrointestinal effects, COX-2 inhibitors, similar to conventional NSAIDs, showed no significant relationship with rheumatological diagnosis, age, gender or duration of drug intake (Maiden et al., 2007).
Our meta-analysis boasts several notable advantages. First, every study that we dissected was a RCT, thereby signifying a diminished risk of partiality. Second, to the best of our knowledge, only a handful of prior studies have endeavored to scrutinize the efficacy of COX-2 inhibitors in ameliorating LUTSs. Consequently, this investigation serves as a robust endorsement for the medical implementation of COX-2 inhibitors in the short-term treatment of LUTS.
There exist certain limitations that warrant consideration. Primarily, the limited number of studies encompassed in this analysis may potentially engender publication bias. To tackle this predicament, our forthcoming research shall strive to seek out current RCTs. Furthermore, the diverse COX-2 inhibitors were not classified in our study, and the short-term follow-up time may also introduce partiality in the outcomes. Considering the conspicuous absence of safety evaluations in short-term COX-2 inhibitor studies pertaining to LUTS, we regrettably refrained from conducting a pertinent analysis.
Conclusion
Overall, our study demonstrates that COX-2 inhibitors can alleviate male LUTS related symptoms in short-term and no apparent phenomenon of drug complications.
Footnotes
Authors’ Contribution
G.L.T. and M.L. designed the research, analyzed the data, and wrote the draft manuscript. S.J.L. and G.X.D. performed literature search, extracted and analyzed the data. G.L.T. and Y.L.C. analyzed data and provided critical scientific input. J.T.W. and Y.S.C. was responsible for resolving discrepancies regarding quality of the included studies, reviewing the manuscript, and providing critical scientific input. All authors made contribution to this article, read, and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Nature Science Foundation of China (Nos. 81870525), Taishan Scholars Program of Shandong Province (No. tsqn201909199).
Ethical Statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was based on previously published studies; therefore, ethical approval and patient consent were not required.
