Abstract
Testosterone levels in men appear to be prognostic of a number of disease outcomes, including severe COVID-19 disease. Testosterone levels naturally decline with age and are lower in individuals with a number of comorbidities and chronic conditions. Low testosterone may therefore be both a cause and a consequence of illness, including COVID-19 disease. The present project examines whether preexisting conditions for severe COVID-19 disease were themselves related to serum-free testosterone levels in men who had not been infected with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus. A clinical risk score for severe COVID-19 disease was computed based on the results of previously published meta-analyses and cohort studies, and relationships between this score and testosterone levels were tested in 142 men ages 19 to 82 years. Greater burden of preexisting conditions for severe COVID-19 disease was related to lower testosterone levels among men younger than 40 years of age. In older men, the decrease in testosterone that accompanies aging attenuated the effect of the clinical risk score on free testosterone levels. Given that older age itself is a predictor of COVID-19 disease severity, these results together suggest that the presence of preexisting conditions may confound the relationship between testosterone levels and COVID-19 disease outcomes in men. Future research examining relationships among testosterone and outcomes related to infectious and chronic diseases should consider potential confounds, such as the role of preexisting conditions.
A growing body of research finds that in men, testosterone levels may be prognostic of clinical outcomes related to COVID-19 disease. While severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection itself is associated with a decrease in serum testosterone relative to healthy controls, testosterone levels tend to be even lower in men who progress to severe or fatal disease (Cinislioglu et al., 2022; Lanser et al., 2021; Salonia et al., 2021). Given that much of this research is cross-sectional and relationships between testosterone and infectious disease are known to be bidirectional (Muehlenbein, 2008; Muehlenbein & Bribiescas, 2005; Muehlenbein et al., 2017), it is difficult to determine whether low testosterone is a cause, in addition to a consequence, of severe COVID-19 disease.
The presence of preexisting chronic conditions in many patients with COVID-19 disease further complicates the relationship among testosterone and severe outcomes. That is, older age and conditions such as cardiovascular disease, diabetes, and cancer both increase one’s risk for severe COVID-19 disease (Geng et al., 2021) and are often accompanied by lower testosterone levels (Araujo et al., 2011; Kelsey et al., 2014; Morris & Channer, 2012; Svartberg et al., 2003; Yao et al., 2017). In other words, it is possible that the link between low testosterone and severe COVID-19 disease is confounded by the presence of chronic comorbid conditions that existed prior to SARS-CoV-2 infection. This possibility is important to consider, as androgen manipulation therapies have been proposed, and tested, as treatments for COVID-19 disease (Nickols et al., 2022; Ory et al., 2020; Salciccia et al., 2021; Stárka & Dušková, 2021). If testosterone levels are, at least in part, incidentally related to COVID-19 disease severity, such treatments would likely have limited utility. Consistent with this hypothesis, several studies reported no effect of androgen deprivation therapy on COVID-19 disease outcomes (Gedeborg et al., 2021; Jiménez-Alcaide et al., 2021; Karimi et al., 2021; Nickols et al., 2022). Although data on the topic are very limited, testosterone replacement therapy does not appear to increase men’s risk for severe or fatal COVID-19 disease (Rambhatla et al., 2021).
The current research examines whether preexisting conditions for severe COVID-19 disease were themselves related to serum-free testosterone levels in men who had not been infected with the SARS-CoV-2 virus. A clinical risk score for severe COVID-19 disease was calculated based on the presence of several chronic conditions (Gassen et al., 2021), and relationships between this score and testosterone levels were tested across a range of ages (19–82 years old). It was hypothesized that greater risk for severe COVID-19 disease would be associated with lower testosterone levels and that this effect may vary by age, which is itself a predictor of disease severity (Chidambaram et al., 2020; Petrilli et al., 2020; Romero Starke et al., 2020).
Method
Study Participants
Data used in the current study were collected from men (n = 142, Mage = 44.66, SDage = 14.04) who participated in the Waco COVID Survey, a longitudinal project surveying anti-SARS-CoV-2 IgG in the sera of asymptomatic residents of McLennan county, Texas from July to November 2020. Additional details of the study, as well as information about the community of McLennan County, are available in a previous publication (Gassen et al., 2021). All data included in the current research were collected as part of the initial intake survey (completed online using Qualtrics software) and first laboratory appointment. Men included in the current study were without symptoms of COVID-19 disease between March 13, 2020, and their date of participation, and all tested negative for anti-SARS-CoV-2 IgG at their first laboratory visit.
Research Ethics
Given that this project functioned as a public health surveillance activity during the SARS-CoV-2 pandemic, it was approved and endorsed by the Waco-McLennan County Public Health District. Therefore, the project met the criteria for exclusion for institutional review board approval at 45 CFR 46.102(e) and (l) for all Baylor University researchers, staff, and volunteers. The institutional review board at Ascension Providence Hospital and Medical Center of Waco, Texas approved participation of Waco Family Medicine researchers, staff, and volunteers. All research subjects completed a written informed consent prior to participation.
Clinical Risk Score for Severe COVID-19 Disease
As part of the comprehensive initial intake questionnaire, participants provided information about several preexisting conditions reported in previous research to increase one’s odds for experiencing severe cases of COVID-19 disease (Centers for Disease Control and Prevention [CDC,] 2020; Chidambaram et al., 2020; Petrilli et al., 2020). Conditions included in the survey were high body mass index (BMI), smoking, and diagnosis of diabetes, cardiovascular disease, chronic obstructive pulmonary disease, kidney disease, liver disease, or cancer.
For a prior study that used these data (Gassen et al., 2021), a clinical risk score for severe COVID-19 disease was computed based on the results of previously published meta-analyses and cohort studies (Chidambaram et al., 2020; Petrilli et al., 2020). The frequency of each condition is available in this previous publication. The presence of each risk factor was weighted based on adjusted odds ratios identified for the association between that factor and severe COVID-19 disease. Reference groups for each factor were assigned 1 (e.g., absence of cardiovascular disease), with groups experiencing higher and lower risk than the reference group assigned a value higher or lower than 1, respectively. Odds ratios for BMI were obtained from Petrilli et al. (2020), whereas odds ratios for all other factors were taken from Chidambaram et al. (2020).
To form the clinical risk score, weights for all factors were averaged (M = 1.13, SD = 0.08). For the original study using this score (Gassen et al., 2021), age was also included as a risk factor. However, age was excluded from the score in the present study so that age-specific relationships between risk and testosterone levels could be examined.
Testosterone Levels
Venous blood was obtained using sterile technique and serum was extracted, aliquoted, and frozen at −80°C until being assayed for free testosterone using an enzyme-linked immunosorbent assay (11-TESHU-E01; ALPCO, Salem, NH, USA). The Laboratory for Evolutionary Medicine at Baylor University is a BSL2+ facility equipped for most clinical biomarker analyses, and the equipment and personnel are certified and monitored by the Department of Environmental Health and Safety. All samples were assayed in duplicate according to manufacturer’s instructions. The average intra-assay (within-person) coefficient of variation (CV) was 7.86% and the inter-assay (between-plate) CV was 12.53%.
Analyses
Four men were excluded from analysis for reporting that they were undergoing testosterone replacement therapy. Generalized linear modeling (R Core Team, 2019) was used to examine age-specific relationships between clinical risk for severe COVID-19 and free testosterone levels (R code available upon request). In the primary regression model, testosterone levels were regressed on age, risk scores, and the interaction between these two variables fit with a Gamma error distribution and log link to account for the positively skewed distribution of the dependent measure. Interactions using simple slopes analysis were unpacked using Johnson-Neyman regions of significance.
Results
Results revealed a significant main effect of age (b = −0.01, SE = 0.004, t = −3.20, p = .002), but not clinical risk scores (b = −1.20, SE = 0.72, t = −1.66, p = .10) on testosterone. However, these results were qualified by a significant interaction between age and risk score (b = 0.11, SE = 0.05, t = 2.16, p = .03). See Figure 1 for interaction effect.
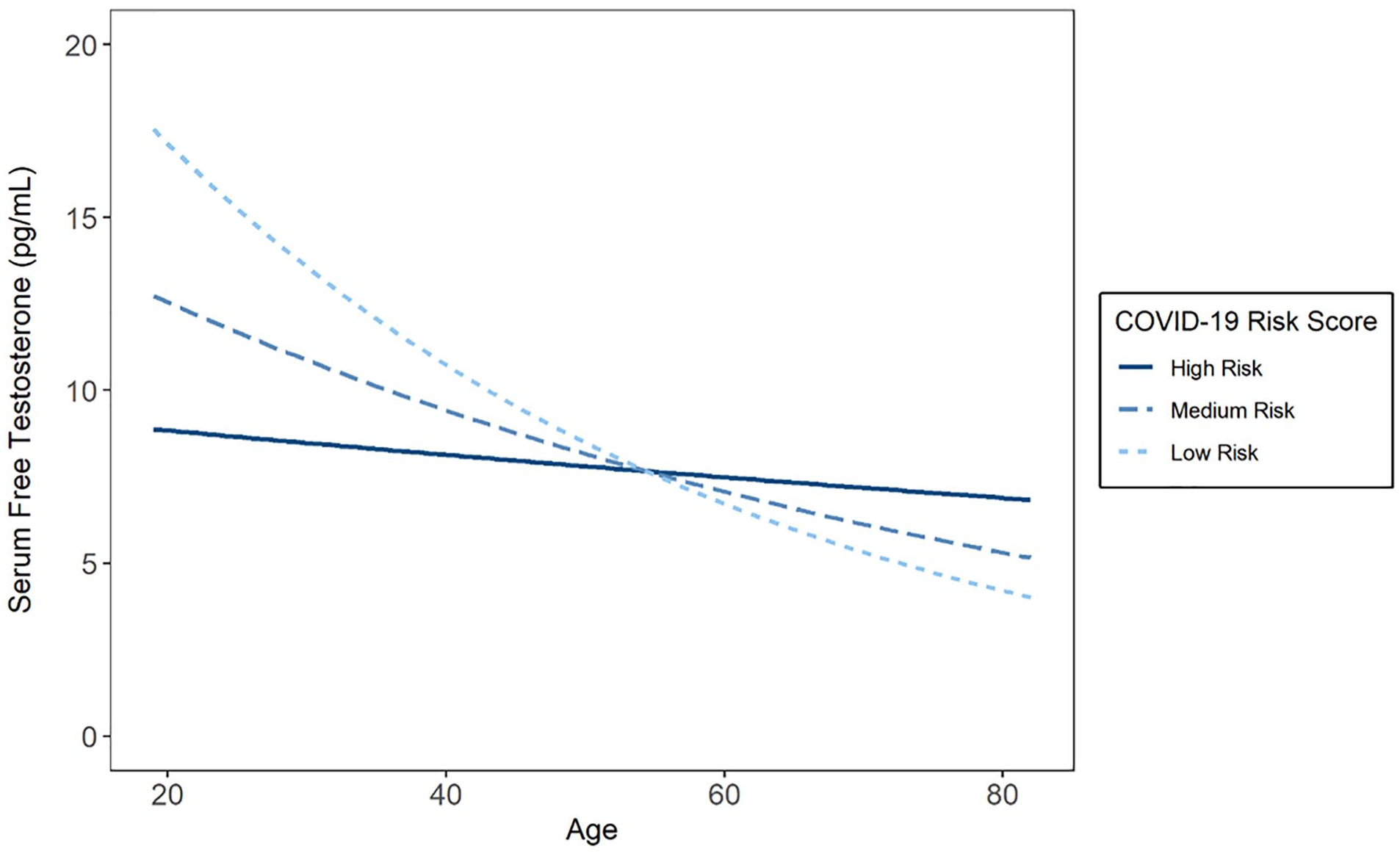
Relationships Between Severe COVID-19 Risk Score and Serum-Free Testosterone Levels Across Levels of Age.
Higher clinical risk scores were associated with lower testosterone levels at younger ages (i.e., 1 SD below mean age; b = −2.72, SE = 1.12, t = −2.43, p = .02), but not older ages (i.e., 1 SD above mean age; b = 0.43, SE = 0.92, t = 0.47, p = .64). Specifically, the effects of clinical risk scores on testosterone levels were significant (p < .05) below 40.91 years of age. The pattern and significance of these results were unchanged when adjusting for race and treating smoking as a covariate, rather than risk factor (interaction: b = 0.12, SE = 0.05, t = 2.18, p = .03).
Discussion
The current research revealed that serum-free testosterone levels decreased as a function of age, as predicted. Greater burden of preexisting conditions for severe COVID-19 disease (i.e., higher clinical risk score) was also related to lower testosterone levels among men younger than 40 years of age. In older men, the decrease in testosterone that accompanies aging attenuated the effect of the clinical risk score on free testosterone levels. Given that older age itself is a predictor of COVID-19 disease severity (Chidambaram et al., 2020; Petrilli et al., 2020; Romero Starke et al., 2020), these results together suggest that the presence of preexisting conditions may confound the relationship between testosterone levels and COVID-19 disease outcomes in men.
The current research of course has limitations that should be considered when interpreting the results. First, the sample size is relatively small and the characteristics of the sample are not representative of the local population as a whole (Gassen et al., 2021). Accordingly, the current findings need to be replicated in a larger, more diverse sample. Furthermore, the clinical risk score used in the present study was additive; there may be interactive or nonlinear relationships between risk factors that should be analyzed and validated in larger samples. It is possible, and perhaps even likely, that certain comorbidities have stronger relationships with testosterone levels than others. This could not be explored here given the limited sample size. Finally, testosterone levels were only measured at a single time point. Although this is not an uncommon practice, single measurements may not reflect an individual’s aggregate exposure to testosterone across time.
Despite these limitations, the current research adds important insights into the complex role of androgens in chronic and infectious diseases and contributes to the growing body of literature on relationships between chronic disease and men’s testosterone levels (Araujo et al., 2011; Kelsey et al., 2014; Morris & Channer, 2012; Svartberg et al., 2003; Yao et al., 2017). Future research examining relationships among testosterone and outcomes related to these diseases should consider potential confounds, such as the role of preexisting conditions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the Waco COVID Survey, from which the current data are derived, was generously provided by the Cooper Foundation of Waco, the Bernard & Aubre Rapoport Foundation of Waco, Waco Family Medicine, and Baylor University.
