Abstract
Because various criteria are used to define metabolic syndrome (MetS), this study examines the most relevant definition for patients with benign prostatic enlargement (BPE). Most studies regarding the link between MetS and BPE/lower urinary tract symptoms (LUTS) have used the National Cholesterol Education Program Adult Treatment Panel III criteria for diagnosis, while a few have used criteria from the International Diabetes Federation and/or American Heart Association. Patients with LUTS due to BPE are classified as having MetS or not by the aforementioned three definitions. Prostate volume, International Prostate Symptom Score, storage and voiding subscores, maximum urinary flow rate, and the postvoid urine of patients with and without MetS were compared separately in the three different groups. Surgical and medical treatment prevalence was also compared between three groups. No matter which definition was used, the International Prostate Symptom Score, the storage and voiding symptom scores, prostate volume, prostate-specific antigen, and postvoid urine were significantly higher in the patients with MetS. The maximum urinary flow rate was similar between patients with and without MetS, according to all three different definitions. There was no significant difference in the aforementioned parameter between patients with MetS diagnosed with the three different definitions. Irrespective of which definition was used, the surgical treatment rate was not significantly different in patients diagnosed with than without MetS, or between the patients with MetS diagnosed with the three different definitions. The authors suggest that it does not matter which of the aforementioned three definitions is used during the evaluation of MetS in men with BPE/LUTS.
Introduction
Metabolic syndrome (MetS) is frequently encountered in countries with Western lifestyles, affecting around one third of the adult population (Golden, Robinson, Saldanha, Anton, & Ladenson, 2009). The definition of MetS varies according to different authorities but commonly comprises a combination of metabolic abnormalities: abdominal obesity, dyslipidemia, raised blood pressure (BP), or insulin resistance (with or without glucose intolerance; Golden et al., 2009).
Today, symptomatic benign prostatic enlargement (BPE) is the most prevalent urologic disease in the male population older than years, with prevalence in one third of men in their 60s and half of men in their 80s (De Nunzio, Aronson, Freedland, Giovannucci, & Parsons, 2012). BPE begins with micronodular hyperplasia and grows into a macroscopic nodular enlargement, which may lead to bladder outlet obstruction, resulting in lower urinary tract symptoms (LUTS; De Nunzio et al., 2012).
The exact molecular and cellular mechanisms resulting in BPE remain unclear. However, molecular and histopathologic studies have uncovered the possible role of MetS in the pathogenesis of BPE (De Nunzio et al., 2012; Gorbachinsky, Akpinar, & Assimos, 2010). Several epidemiologic studies have revealed a relationship between MetS and the occurrence of LUTS in men with BPE (Gacci et al., 2015; Hammarsten & Peeker, 2011).
The pathogenic pathways potentially linking BPE/LUTS and MetS have not been fully elucidated. There are alternative hypothesis trying to explain the mechanism. The proposed mechanisms are insulin resistance, hormone changes, pelvic atherosclerosis, autonomic–sympathetic overactivity, and inflammation (De Nunzio et al., 2012; Vignozzi et al., 2014).
Various criteria are used to define MetS. International organizations like the World Health Organization, European Group for the study of Insulin Resistance, and International Diabetes Federation (IDF), plus expert groups such as the National Cholesterol Education Program Adult Treatment Panel III (NCEP: ATPIII), American Association of Clinical Endocrinology, and American Heart Association/National Heart, Lung and Blood Institute (AHA/NHLBI) have tried to combine the different parameters used to define MetS.
Most studies on the link between MetS and BPE/LUTS have been based on the NCEP: ATPIII definition, while limited studies have used IDF or IDF/AHA definitions (Choi et al., 2011; De Nunzio et al., 2014; Dong et al., 2015; Jeong, Hwang, Kim, Ahn, & Kim, 2010; Kwon, Kang, & Lee, 2013; Park et al., 2013; Park, Min, & Lee, 2012; Ryl et al., 2015). To the best of our knowledge, no study has investigated the relationship between BPE/LUTS and MetS with different MetS definitions separately.
In this study, the NCEP: ATPIII, IDF, and IDF/AHA definitions are used separately in evaluating the link between MetS and BPE/LUTS in an attempt to reveal the most effective MetS definition that can be used in patients with BPE/LUTS.
Material and Method
Each patient with LUTS due to BPE admitted to the authors’ outpatient clinic was retrospectively reviewed. The overall study population consisted of 807 men, ranging in age from 46 to 89 years. The patients’ medical histories and physical examination records were obtained, including information on digital rectal examinations that had been performed. The biochemical analyses included serum glucose, triglycerides, high-density lipoprotein (HDL) cholesterol and prostate-specific antigen (PSA). Other laboratory assessments included a complete blood cell count, liver function test routine, creatinine, and urinalysis. The following methodology was used:
Transrectal ultrasound-guided biopsy was performed if hard nodules were identified on the patient during the digital rectal examination or if the patient had a PSA higher than 3 ng/dL.
Two BP measurements were obtained 10 minutes apart and the results are averaged.
Waist circumference (WC) was measured while the patients were standing and breathing normally, midway between the lowest rib margin and the iliac crest.
LUTS were assessed using the International Prostate Symptom Score (IPSS) and symptoms were further categorized as voiding and storage. The IPSS voiding and storage subscore was calculated by the sum of Questions 1, 3, 5, 6 and Questions 2, 4, 7, respectively.
Patients’ height, weight, and WC measurements were recorded and body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared (kg/m2).
The inclusion criteria were a prostate volume (PV) larger than 30 mL and IPSS >7 (Chute et al., 1993). The maximum urinary flow rate (Qm) was determined with uroflowmetry. Only flows with at least 150 mL of voided volume were included. Measurements of three dimensions of the prostate and postvoid residual (PVR) urine volume were made using a Toshiba, Xario (Toshiba, Tokyo, Japan) 4.0 MHz transabdominal ultrasound probe positioned suprapubically in the transverse and sagittal planes. The volume of the prostate was calculated with the following formula: PV (mL) = width (cm) × height (cm) × length (cm) × 0.523. PVR was calculated with the following formula: PVR (mL) = width (cm) × height (cm) × length (cm) × 0.625.
Exclusion criteria included any of the following:
Medical or surgical intervention for BPE or prostate cancer
Anticholinergic, cholinergic, sympathomimetic, sympatholytic, 5-alpha reductase inhibitor, orphosphodiesterase-5 inhibitor medication
Treatment with any medication affecting testosterone or estrogen levels
The presence of any renal or hepatic impairment
Neurological diseases known to affect bladder function
Active urinary tract infection.
Three different MetS definitions were used in the study: (a) NCEP: ATPIII (“Executive Summary of the Third Report of the National Cholesterol Education Program [NCEP] Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults [Adult Treatment Panel III],” 2001); (b) IDF (Alberti, Zimmet, & Shaw, 2005), and (c) consensus definition (incorporating the IDF and AHA/NHLBI definitions; Alberti et al., 2009).
1. NCEP: ATPIII definition included any three or more of the following:
WC >102 cm
Triglyceride ≥150 mg/dL
HDL cholesterol <40 mg/dL
BP ≥130/85 mmHg
Fasting glucose ≥110 mg/dL (In 2003, the American Diabetes Association changed the criteria for impaired fasting glucose tolerance from 110 mg/dL to 100 mg/dL)
2. IDF’s definition included central obesity (defined as WC >94 cm or BMI >30 kg/m2) with ethnicity-specific values, plus two of the following:
Triglyceride ≥150 mg/dL
HDL cholesterol <40 mg/dL
BP ≥130/85 mmHg
Fasting glucose ≥100 mg/dL
3. IDF and AHA/NHLBI definition included any three of the following:
WC >94 cm (Adjusted for the European population [original >102 cm])
Triglyceride ≥150 mg/dL
HDL cholesterol <40 mg/dL
BP ≥130/85 mmHg
Fasting glucose ≥100 mg/dL
Statistical analysis was performed using the Predictive Analytics Software (PASW) Statistics 18.0 for Windows (SPSS Inc., Chicago, IL, USA). Evaluation of the data distribution identified a normal distribution of the study data set. Differences between groups of patients in the means for the quantitative variables and differences in the distributions of the categorical variables were tested with the independent
Results
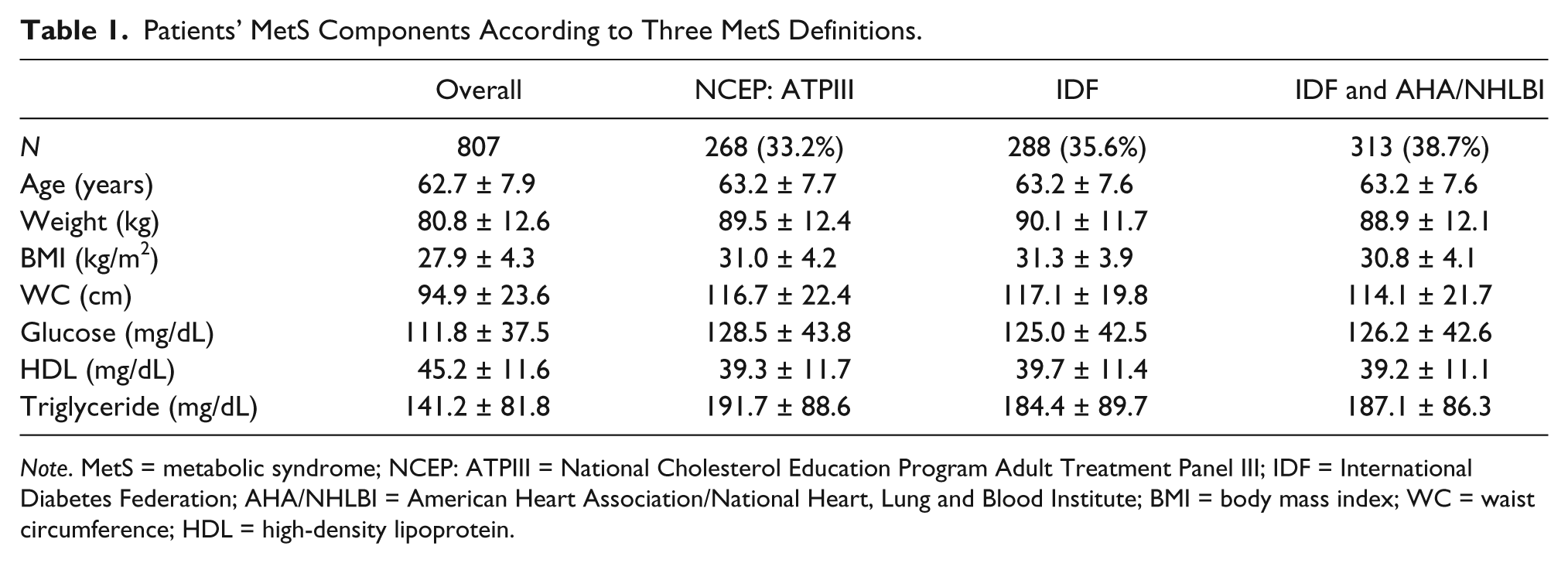
According to this report, the highest MetS prevalence was reached with the IDF and AHA/NHLBI definition (Table 1). In all the groups, the triglyceride level was higher than 150 mg/dL, glucose level was higher than 100 mg/dL, and HDL level was lower than 40mg/dL. There was no significant difference in MetS parameters between the three groups (Table 1). In the patients with MetS according to each of the three definitions, the IPSS, the storage and voiding symptom scores, PV, PSA, and PVR were significantly higher. The Qm, and age were similar between patients with and without MetS according to all three definitions (Table 2). All of the parameters related to LUTS and BPE investigated in this study were similar between patients diagnosed by different MetS definitions (
Patients’ MetS Components According to Three MetS Definitions.
Patients’ BPE/LUTS Parameters According to Three MetS Definitions.

Treatment Distribution According to MetS Definitions.

Discussion
There are various MetS definitions. The three generally accepted definitions used to diagnose the MetS have been presented by the NCEP, IDF, and AHA/NHLBI. The majority of previous studies on MetS and BPE have used the NCEP: ATP III definition (Choi et al., 2011; De Nunzio et al., 2014; Kwon et al., 2013; Pan, Liu, & Zhou, 2014; Park et al., 2012; Park et al., 2013). However, some of the studies also included IDF or IDF and AHA/NHLBI definitions (Dong et al., 2015; Jeong et al., 2010; Ryl et al., 2015). These definitions cannot be considered the gold standard, because they underline different sights of the MetS. Due to the limitations of the classic definitions, it is plausible to research the most relevant definition for association between MetS and BPE/LUTS. This study attempted to examine which of the aforementioned MetS definitions was more related to LUTS and BPE parameters.
In the present study, the highest MetS rate (38.7%) was identified with the IDF and AHA/NHLBI definition. The lowest MetS rate (33.2%) was identified with NCEP/ATPIII definition. MetS prevalence in men with LUTS was compatible with the literature (De Nunzio et al., 2014; Dong et al., 2015; Kwon et al., 2013; Pan et al., 2014; Park et al., 2012; Park et al., 2013). The prevalence of MetS according to different definitions in the same population was also similar to the literature (Nilsson, Engstrom, & Hedblad, 2007; Qiao, 2006).
In various studies, MetS criterion such as BMI, WC, and HDL were reported to be associated with LUTS and BPE (Jeong et al., 2010; Pan et al., 2014). In the present study, the entire set of MetS parameters was similar between the different MetS definitions. The authors believe that this is an advantage for this work because we aimed to study not the effect of individual MetS parameters but MetS definitions on BPE and LUTS. A discrepancy between MetS definitions in terms of MetS criteria could overshadow the purpose of the study.
In the present study, no matter which definition was used patients with MetS had higher PV, PSA and PVR, lower Qm, and were more symptomatic than patients without MetS. However, a statistically significant difference was not identified in the Qm. Several studies have suggested a relationship between LUTS and the presence of MetS. Men with MetS are more likely to have higher IPSS and voiding and storage symptom scores (De Nunzio et al., 2014; Hammarsten & Peeker, 2011; Kirby et al., 2010; Pan et al., 2014; Yang et al., 2012). As in the literature, the men diagnosed with MetS in the current study had higher IPSS, voiding and storage symptoms than participants without MetS, no matter which definition was used. The authors identified no significant difference in IPSS or storage and voiding symptom scores between men diagnosed with different MetS definitions.
PV and PSA level in men with MetS are controversial in the literature. Some studies reported significantly higher values in patients with MetS (Dong et al., 2015; Pan et al., 2014), while others reported similar values (Choi et al., 2011; De Nunzio et al., 2014; Dong et al., 2015). In the present study, the PV and PSA were significantly higher in patients with MetS. There was no difference in PSA and PV between patients diagnosed with the three different MetS definitions.
In a comprehensive study about BPH progression, Roehrborn (2008) defined increased PVR as a predictor of the clinical progression of BPE. In the present study, PVR was significantly higher in the patients with MetS. This difference may be the result of possible high incidence of diabetic cystopathy in patients with MetS. These results are consistent with the reports (Dong et al., 2015; Kwon et al., 2013; Pan et al., 2014; Park et al., 2013). In terms of PVR, there was no difference between the MetS definitions in this study.
Almost all of the studies investigating Qm in MetS reported no difference between MetS versus non-MetS patients (Kwon et al., 2013; Park et al., 2013; Yang et al., 2012). The authors identified only one study that reported a significant difference in Qm between MetS and non-MetS patients (Pan et al., 2014). As in the literature, no matter which definition was used, there was no difference in Qm between the MetS and non-MetS patients in the current study. In terms of Qm, there was no difference between the MetS definitions in the current study.
There are reports regarding the effect of MetS on response to the medical and surgical treatment of BPE (Cyrus et al., 2014; Sener et al., 2015). To the best of our knowledge, no study has investigated the prevalence of surgical therapy on BPE in patients with MetS. Patients with MetS diagnosed using the NCEP/ATPIII definition had higher surgery prevalence than the two other MetS definitions, but the difference was not statistically significant. The authors believe that studies regarding the effect of MetS on therapy alternatives in BPE will be valuable.
Conclusions
Among the patients with BPE, patients with MetS are more symptomatic than patients without. Storage and voiding symptoms were also significantly higher in patients with MetS, but the total score and the storage and voiding symptom scores were not significantly different between the three separate MetS definitions. The high surgical treatment rate for patients with MetS diagnosed using the NCEP/ATPII definition was striking but statistically insignificant. The authors suggest it does not matter which of the aforementioned three MetS definitions is used during the evaluation of MetS in men with BPE/LUTS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
