Abstract
Testosterone replacement improves quality of life and is aromatized in men in adipose tissues to estrogen. Hyperestrogenism is believed to be harmful to male sexuality. This is a description of our experience of screening 34,016 men in the Low T Centers, of which approximately 50% were converted to treatment. Men were treated with injectable testosterone, and we have available data from 2009 to 2014. The data were extracted from our electronic health record (AdvancedMD) of 35 Low T Centers across the United States. In all, 7,215 (20.2%) out of the 34,016 patients had high estradiol levels defined as ≥42.6 pg/ml. Estradiol was measured using electro-chemiluminescence immunoassay. Of the patients who had high estradiol levels, the age distribution was as follows: 132/989 (13.3%) were older than 65 years, 3,753/16,955 (22.1%) were between 45 and 65 years; 2,968/15,857 (18.7%) were between 25 and 44 years, 7/215 (3.3%) were younger than 25 years. The difference between extreme age groups (<25 and ≥65) was statistically significant using a chi-square test (p = .013). The correlation coefficient of serum estradiol to age was .53, SD = 8.21. It was observed that practitioners used aromatase inhibitor and selective estrogen receptor modulator to treat symptoms of hyperestrogenism, irrespective of blood estradiol levels. Gynecomastia was rarely documented as a reason for the prescription. Our finding was that high estradiol levels were not associated with higher rates of low libido but established higher rates of documented low libido with those with normal or lower estradiol levels. The difference was statistically significant (p < .05).
Introduction
Testosterone is a hormone that is synthesized from cholesterol and is broken down to various metabolites, including 17 beta-estradiol (E2) and 5 alpha-dihydrotestosterone (DHT). E2 is derived from aromatization of testosterone in adipose tissues, whereas dihydrotestosterone is derived from 5 alpha reduction of testosterone. In our practices, it was observed that patients develop tender nipples from gynecomastia after injectable testosterone therapy. This could be the result of increased testosterone levels after treatment, which in turn aromatizes into E2. The development of breast tissue in men appears to be directly linked to rises in testosterone levels, E2 levels, and obesity. The Low T Centers have had 4 years experience treating large populations of hypogonadal patients and thus sought to understand if there were predictive factors of patients who were more likely to develop high estradiol levels after treatment with testosterone. We wanted to examine specifically the influence of age and whether there was an impact on the aromatization of testosterone to E2. The aging process is often associated with weight gain, particularly in the middle age groups (Dahl, Reynolds, Fall, Magnusson, & Pedersen, 2013). A physiological change also occurs with aging, as the body exchanges lean body mass for fat. This has a direct influence on drug pharmacokinetics and pharmacodynamics. For example, with aging the higher fat content can influence fat soluble drugs by increasing their half lives and hence their duration of action. Our hypothesis is that age is directly correlated to E2 conversion as a result of accumulating body fat with time, and we sought a population-based database to answer those questions. The role of estrogens in men is clouded in controversy, with some advocating that excessive estrogens could be harmful to sexuality and yet others claiming a beneficial role to the general well-being of men (Kacker, Traish, & Morgentaler, 2012). Currently, there is a lack of guidelines as to the critical level whereby intervention is needed to treat high estradiol levels or hyperestregenism. Most of the decisions are practitioner based and individualized. In some cases, treatment was given to relieve discomfort rather than lower levels.
Research Design
This retrospective multicenter, medical chart review was conducted at 35 geographically diverse sites in the United States, with a concentration in Texas. Prior to including the sites, the principal investigator underwent training with the information technology specialist at the Low T Centers. He was instructed on how to access the electronic health record (EHR) called AdvancedMD and also how to mine data from the EHR. Researchers also underwent research training with Good Clinical Practices with the Federal Drug Administration. We applied for approval for expedited review from a Central Institutional Review Board, to include waiver of consent for a chart review. Each research site de-identified data on case report forms using numeric codes to assure patient confidentiality.
The medical records of all men evaluated in the 35 Low T Centers from October 1, 2009, to January 15th, 2014, were queried for a diagnosis of hypogonadism by the International Center for Diseases-9 (ICD-9) code 257.2, yielding 8,096 men initially. To be diagnosed with hypogonadism, the patient must have reported symptoms in addition to having had a serum testosterone level below 350 ng/dL. Records for patients initiating testosterone replacement therapy (TRT) and having blood testing for hormone assays were reviewed. For inclusion in the study, the patients’ full demographic information must have been available, they were males ≥20 years of age, and they had visited one of the 35 Low T Centers at least once, whether they received treatment or not. Each patient must have had an initial total testosterone and E2 level. There were no exclusion criteria, as the study design was a retrospective chart review of patient records. Demographic data (age, gender) as well as E2 levels were recorded in an Excel spreadsheet. For baseline and all follow-up visits, the visit date and ICD-9 were documented. Adverse events with TRT causing gynecomastia were recorded from the ICD-9 diagnoses available in AdvancedMD. Searches were performed on our database to determine the frequency of our patients’ gynecomastia, relying on ICD-9. Of the patients having gynecomastia, the prescription patterns of our practitioners was evaluated.
Total testosterone and estradiol levels were drawn during a morning clinic, generally between 9 a.m. and 12 p.m., although the exact time varied and was not uniformly recorded. The testosterone was performed primarily by the Qualigen FastPack IP Testo Immunoassay, which is a chemiluminiscent immunoassay for the in vitro quantitative determination of total testosterone in human serum. E2 levels were measured using electro-chemiluminescence immunoassay through Lab Corp. The free testosterone was calculated with a direct radioimmunoassay. The statistical significance of between-cohort differences in categorical variables was tested using the chi-square test and in continuous variables using the two-sample t test. All tests were two-tailed with a significance level of p < .05.
Results
During the analysis of this study, using the AdvancedMD EHR’s capture of 35,711 patients, it was established that 9,017 (25.3%) had symptoms pertaining to andropause (Tan & Philip, 1999), such as low libido (ICD-9: 799.81), erectile dysfunction (ICD-9: 302.72), and fatigue (ICD-9: 780.79) based on review of ICD-9 in their problem list. Of the 34,016 patients, we found the following distribution of age-groups: <25 years (1%), 25 to 44 years (47%), 45 to 65 years (49%), and >65 years (3%). The definition of high estradiol varies by laboratory. In our Centers, estradiol measurement is outsourced to Lab Corp, and high estradiol is defined equal or above 42.6 pg/ml and is based on the electro-chemiluminescence immunoassay methodology. In contrast, Quest diagnostics used as cutoff of 29 pg/ml, and their test is based on liquid chromatography but is not used here. The specific hormonal parameter pre-TRT included having a baseline of 350 ng/dl on two separate occasions before therapy. There was no specific pre-TRT estradiol parameter. Post TRT, levels were titrated not only to a range between 350 and 1000 ng/dl but also according to the symptoms and individualized by the practitioner.
The trend of estradiol levels after treatment with injectable testosterone was looked into. As seen in Figure 1, estradiol levels were generally low in the <25 year age-group, with a rise in the middle ages from 25 to 44 years and a gradual tapering in the later years. Of the patients who had high estradiol levels and with regard to age distribution, 132/989 (13.4%) were >65 years, 3,753/16,955 (22.1%) were between 45 and 65 years, 2,968/15,857 (18.7%) were between 25 and 44 years, 7/215 (3.3%) were <25 years. The difference between extreme age-groups (<25 and ≥65) was statistically significant using a chi-square test (p = .013) with a chi statistic of 6.105. The correlation coefficient of serum estradiol to age was .53, SD = 8.21. The relation of the increase of testosterone levels to E2 was also explored. Of the patients who had high total testosterone (defined as >1,000 ng/dl) and with regard to age distribution, 3.3% were older than 65 years, 54.2% were between 45 and 65 years, 42.5% were between 25 and 44 years, but 0% were younger than 25 years.
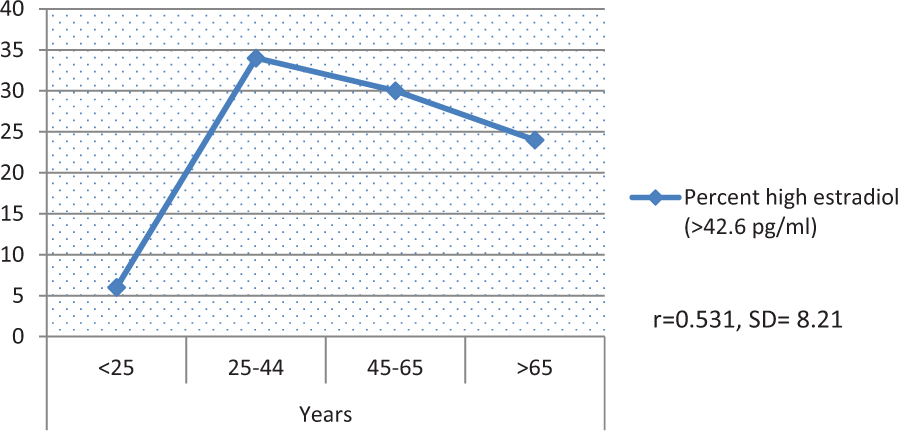
Percentage of high estradiol levels with age.
In our study, it was discovered that practitioners used anti-estrogens such as aromatase inhibitor (AI) and selective estrogen receptor modulator (SERM) to treat symptoms of hyperestrogenism, irrespective of blood estradiol levels. Although the frequency of use of AI remains high, it was rare that practitioners document gynecomastia (611.1) in the problem list (<1%). However, our group did not individually examine each of the charts to see if loss of libido was in other parts of the progress notes as the EHR did not have this mining capability. The results of this study suggest AI and SERM use in 30% of the treated population. As demonstrated in Figure 2, the preferred AI was anastrozole (Arimidex) in 97% of cases, followed by tamoxifen (Nolvadex; 3%). Although approximately 1,200 patients received AI and SERMs, side effects occurred in less than 1%. A few patients reported hot flashes, mood changes, and nausea.

Percentage distribution of aromatase inhibitor/selective estrogen receptor modulator use.
In this study, we also wanted to see if there was a correlation of the differential prevalence of hyperestrogenism by age to the use of AI and SERM by age. Arimidex was the most popular AI used in this practice and established that the use of this AI parallels the prevalence of hyperestrogenism in the different age-groups. AI and SERM were mainly used in the 25 to 44 years group (46%) and the 45 to 65 years group (50%). Those younger 25 years accounted for less than 1%, whereas those older than 65 years were at 3%.
Our study did not link high estradiol levels with diminished sexual performance. Paradoxically, patients with low estradiol below 42.6 pg/ml had more patients complaining of low libido as defined by ICD-9 of 799.81 in the problem list. Patients with higher estradiol levels above 42.6 pg/ml had less sexual dysfunction problems identified by their providers. Of those that providers had identified as having low libido, 2,726 of 7,332 patients (31%) had higher estradiol levels and 4,606 patients (69%) had normal estradiol levels. In contrast to those who did not complain of low libido, 4,811 patients (28%) had high estradiol and 12,360 patients (72%) had normal estradiol levels. When the chi-square test was applied, the difference was statistically significant, p < .05; in other words, there is a real difference with those who complained of low libido and had high estradiol and those who did not complain of low libido and had normal or low estradiol. The result parallels the results of some studies (Finkelstein et al., 2013) but contradicts studies of some others (Bancroft, Tennent, Loucas, & Cass, 1974). A summary is given in Table 1.
Number of Patients With Libido Issues With Respect to Estradiol Levels.
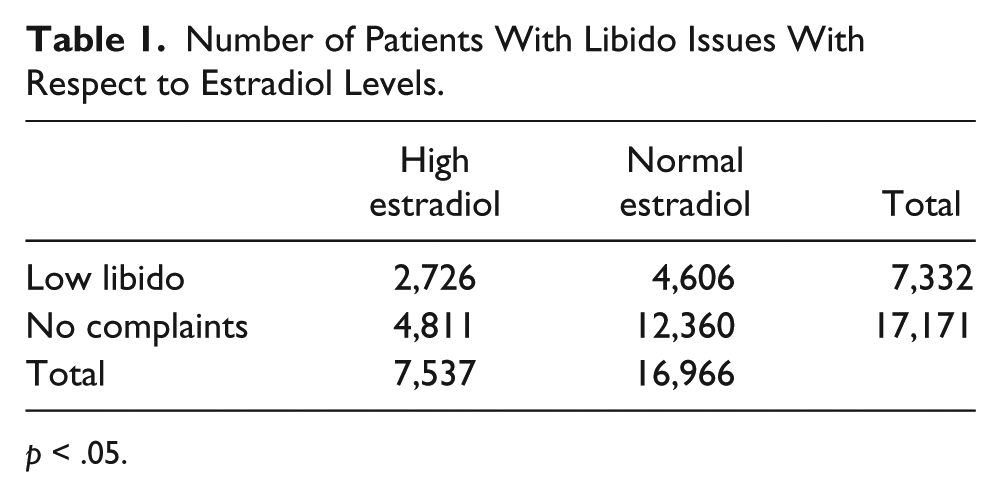
p < .05.
Discussion
Role of Estrogens in Male Sexuality and Aging
Estrogens have been known to be present in men in previous studies and have a well-established and known physiological role in maintaining bone health (Chin & Ima-Nirwana, 2012). However, the role of estrogens in male human sexuality is clouded in controversy. It has been reported that estrogen treatment in eugonadal men diminishes libido, whereas libido is preserved by estrogens in orchidectomized transsexuals as well as in cases of aromatase deficiency (Although the role of endogenous estrogens in sexuality is unclear, the negative effect of exogenous estrogens is better studied. E2 appears to play a key role in the negative feedback control of testosterone. In castrated monkeys, testosterone replacement does not reduce luteinizing hormone, unless estradiol is also given, which suggests the regulatory role estradiol (Resko, Quadri, & Spies, 1977). Men with aromatase deficiency or estrogen resistance also show raised gonadotrophins (luteinizing hormone, follicle-stimulating hormone). Some of our knowledge of the role of estrogens comes from studies that involve estrogens as a treatment of prostate carcinoma in the past. Many of these patients on estrogens complained of sexual dysfunction, including loss of libido (Stege, 1995). Estrogens have also been used to treat male sexual offenders in the past and demonstrated efficacy (Bancroft et al., 1974). Some researchers have purported a direct role of estrogens in improving sexuality in men (Gooren, 1985). In a recent article by Finkelstein et al. (2013) on 198 healthy men, it was reported that estrogen deficiency primarily accounted for increases in body fat and that both contributed to the decline in sexual function. This suggested estrogen as being essential for sexual functioning in men.
Our group looked retrospectively at our database of 34,016 patients who were screened. Of this population approximately 50% or 17,000 patients met the criteria for treatment and were followed. The characteristics of men who had undergone TRT and their relationship to E2 levels were examined. Specifically, we wanted to investigate if age was a determinant to the rise in estradiol levels. A general rise in estradiol levels with age, albeit there appears to be a leveling of this in later years. Our results mirror the results of a recent smaller study on 103 men, where they discovered the rate of aromatization to be higher in older men (Lakshman et al., 2010). It is suggested that the leveling of estradiol levels after 45 years could be due to the lower therapeutic level of testosterone achieved in older patients because of safety concerns. It was not possible to control for weight or body mass index as the data were not uniformly entered into the EHR software by the respective centers. For the same reason, multivariate analysis was not possible. Testosterone is aromatized to estradiol, and thus lower amounts of estradiol was seen in later years as there were lower levels of testosterone in later years. Although longitudinal data on endogenous E2 are not available, the Massachusetts Male Aging Study reports a decrease of 3.6% E1 per year (Feldman et al., 2002), and ours is the first report of a rise in E2 with age with exogenous testosterone as described in a sizeable population.
Anti-Estrogens: Aromatase Inhibitors/SERM
Currently, there is a controversy on whether to treat patients on exogenous testosterone with high estrogens. One of our goals was to find out if treatment was given, what the preferred drug might be. In our population, anastrazole (Arimidex) was the most prescribed AI. It does not appear that the preference is based on pricing alone. Of the AIs, the most expensive is Femara (letrozole), followed by Arimidex (anastrozole) and then Novaldex (tamoxifen), which is a SERM. A separate study would be needed to investigate further provider and patient preference.
Do We Need to Treat High E2 After TRT? Anastrozole
This study was not designed specifically to answer this question. A prospective, randomized controlled study would be needed over several years to understand if indeed high E2 after TRT may be beneficial or harmful. Currently, there are no national guidelines that are evidence based for treatment of high E2 after TRT. However, there was a high treatment rate for our group of patients at 30%. Based on interviews with select practitioners, we found that the reasons for the high rates of prescribing AI and SERM are partly patient pressure, practitioner confusion, and fear of the harmful effects of high E2 in long term. Some studies have indicated an association of high estrogens to higher rates of heart attacks, strokes, and prostate cancer (Basaria et al., 2013; Kristal et al., 2012; Lerchbaum et al., 2011). In one such study, higher estradiol levels in men were significantly associated with prevalent strokes, peripheral vascular disease, and carotid artery stenosis compared to lower estradiol levels. High levels of estradiol were also associated with all-cause and noncardiovascular mortality in a large cohort of older men referred to coronary angiography (Lerchbaum et al., 2011). These studies of association do not infer causality and as such should not be used for the basis for treatment of high estrogen. AI and SERM use may be justified for breast tenderness or gynecomastia. However, gynecomastia is rarely documented in problem list; this is an area of improvement for the practice.
In our study, high estradiol levels did not correlate with higher rates of low libido. On the other hand, low levels of E2 were associated with higher rates of low libido. The explanation could be that low E2 usually results from low testosterone levels, as E2 is aromatized from testosterone. Our study results parallel that of others (Finkelstein et al., 2013) suggesting a positive role of estradiol in human male libido functioning. There are limitations to our present study and we do declare that our results cannot be generalized. This is based on our methodology of assigning low libido as captured in the problem list. Providers may have not uniformly entered the ICD-9 diagnoses, and hence it may be possible that results may be overstated. The other limitation is that although 34,016 patients who presented to the Centers were screened, only 50% were eligible for treatment, based on inclusion and exclusion factors. The analysis was done on patients who presented to the Centers and were screened and not limited to those on treatment. The correlation of libido is definitely an area for further study.
The effects of estradiol on the human brain are unclear. Currently, there are no hormone markers within the brain to determine the association of levels of sex hormones with libido. In this study a presumption was made that serum estradiol reflects brain estradiol levels or activity. It may be possible that if serum estradiol level is low more estradiol is available for the brain, as it is shifted from the rest of the body to the brain. This hypothesis is given to support the results of our study, of not associating low libido to higher estradiol levels. In a previous study, positive emission tomography was used in an attempt to map areas of the brain involved in glucose metabolism after administration of testosterone. The brain stem and parietal lobes were highly metabolic, suggesting that these areas are involved in sexual processing (Tan, 2013). There are current arguments for an optimal T:E2 ratio for sexuality (Shabsigh et al., 2005), rather than the actual amounts, but studies are weakly powered.
Conclusion
Not much work has been done to understand better the role of estrogens in men. There has also been a distinction between work done on endogenous estrogens and also exogenous estrogens, be it given as estrogen itself or converted from testosterone. Our work in a large database of 34,016 patients represents one of the first attempts to understand the characteristics of exogenous estrogens, which in this case are aromatized from exogenous testosterone given to treat hypogonadism. From our study, it appears that age may be a determinant of the conversion of testosterone to estrogens, except for later years in life after 65 years. The clinical importance of high estrogen after TRT continues to be debated. In our study, high estrogen after TRT does not necessarily associate with low libido. However, AI and SERM were prescribed frequently (30% of cases). There are challenges in setting up a guideline for the threshold beyond which AI and SERM are to be used, as there are no evidence-based studies at this time to guide the practice. Normality based on standard deviation can be used, but our study reports that age in itself may cause variations in normal values. Although our study did not associate low libido with high E2 levels; there may be foreseeable dangers to exposure to high estrogen over a longer period of time (Lerchbaum et al., 2011). The use of AI and SERM should be individualized and carefully monitored. The common side effects of AIs include constipation, diarrhea, nausea, vomiting, upset stomach, loss of appetite, body aches and pains, breast swelling/tenderness/pain, headache, dry mouth, scratchy throat, increased cough, dizziness, trouble sleeping, tiredness/weakness, flushing and sweating (hot flashes/hot flushes), hair thinning, and weight change and should be communicated to the patent. Changes in diet such as eating several small meals may help lessen the chance of nausea and vomiting. More work such as a longitudinal, controlled study is needed to assess the role of exogenous estrogens from TRT and the need to treat this condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Opal Medical Clinic is not related to Low T Centers. Mr. Cook and Dr. Reilly see patients at the Low T Centers but are neither employees nor shareholders and are independent contractors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
