Abstract
Patterns of care following topical testosterone agent (TTA) initiation are poorly understood. This study aimed to characterize care following TTA initiation and compare results between patients with and without a serum testosterone (T) assay within 30 days before and including TTA initiation. Adult men (N = 4,146) initiating TTAs from January 1, 2011, to March 31, 2012, were identified from a commercially insured database. Patients were included if they initiated at recommended starting dose (RSD) and had ≥12 and ≥6 months of continuous eligibility preinitiation (baseline) and postinitiation (study period), respectively. Patients were stratified by preinitiation T assay. Maintenance dose attainment month was determined using unadjusted generalized estimating equations regression to compare dose relative to RSD month by month. Outcomes included maintenance dose attainment month, time to stopping of index TTA refills or a claim for nonindex testosterone replacement therapy (TRT), and proportion of patients with study period T assay or diagnosis of hypogonadism (HG) or another low testosterone condition, and were compared using chi-square and Wilcoxon rank-sum tests for categorical and continuous variables, respectively. Maintenance dose was attained in Month 4 postinitiation, at 115.2% of RSD. Approximately 46% of patients had a preinitiation T assay; these men were more likely to receive a diagnosis of HG or another low testosterone condition, to have a follow-up T assay, to continue treatment by filling a nonindex TRT, and less likely to stop refilling treatment with their index TTA. Differences in care following TTA initiation suggest that preinitiation T assays (i.e., guideline-based care) may be helpful in ensuring treatment benefits.
Keywords
Introduction
Hypogonadism (HG) in men, a condition characterized by reduced production of testosterone and spermatozoa, is caused by malfunctioning of the pituitary gland, hypothalamus, or testes (Bhasin et al., 2010). HG is a common condition; a population-based study observed that 24% of men ages 30 to 79 were hypogonadal (defined as total testosterone <300 ng/dL; Araujo et al., 2007). Symptoms of HG include reduced energy and libido, sleep disturbance, decreased bone/muscle mass, and diminished work or physical performance (Bhasin et al., 2010). HG is frequently comorbid with varied conditions, including hypertension, hyperlipidemia, diabetes, back/neck pain, sexual dysfunction, and psychiatric disorders (Buvat, Maggi, Guay, & Torres, 2013; Hyde et al., 2012; Kaltenboeck, et al., 2012; Shores, Smith, Forsberg, Anawalt, & Matsumoto, 2012).
The Endocrine Society Clinical Practice Guidelines recommend that symptomatic patients with hypogonadism be treated with testosterone replacement therapy (TRT) and that serum testosterone assays (T assays) be measured prior to initiation of TRT to confirm the diagnosis (Bhasin et al., 2010). The treatment goal is to restore serum testosterone levels to the middle of the normal range of 400 to 700 ng/dL. Following TRT initiation, men whose testosterone levels remain below the desired range should have their doses adjusted. Dose adjustment should be guided by serum testosterone levels, which can be assessed with T assays after TRT initiation, with the timing dependent on the specific TRT formulation (Bhasin et al., 2010). Furthermore, the Clinical Practice Guidelines recommend that patients should be monitored between 3 and 6 months after TRT initiation to assess response to therapy, compliance, and adverse events (Bhasin et al., 2010). Thus, serum testosterone assays should serve a pivotal role in the diagnosis and follow-up of men receiving testosterone replacement therapy for conditions of low testosterone.
Topical testosterone agents (TTAs), formulated as gels or solutions, are commonly used to deliver TRT in the United States. Their benefits include easy application with generally good tolerability and dosing flexibility, and T assays to check response to therapy can be administered as early as 1 week after initiation (Bhasin et al., 2010). Results of various TTA clinical trials indicate that many patients require doses higher than the labeled recommended starting dose (RSD) to attain and maintain normal testosterone levels (Dobs et al., 2012; Kaufman et al., 2011; Steidle et al., 2003; Wang et al., 2000; Wang et al., 2011). A recent retrospective administrative claims data study reported that after initiation of therapy, the dose utilized by patients increased until maintenance dose was achieved in Month 4 (Kaltenboeck et al., 2013). The same study also reported that only about one third of patients (3,470 out of 9,995 patients) received their index TTA during Month 4 after initiation, suggesting that some patients do not remain on therapy long enough to reach stable doses. In addition, TTA treatment patterns have been the subject of recent media articles. However, to date, no published research has evaluated real-world diagnostic and monitoring patterns with T assays among patients initiating TTA therapy.
Aims
The objective of the study was to describe real-world care following TTA initiation, overall and stratified by whether patients received serum testosterone determination, during the diagnostic process. Preinitiation T assays in the month prior to TTA initiation were used as a proxy for clinical confirmation of low testosterone levels.
Method
Data Source
This study utilized a de-identified administrative claims database (OptumHealth Reporting and Insights, formerly Ingenix Employer Solutions) that covers approximately 16 million privately insured lives from 60 U.S.-based companies in a broad array of industries with numerous insurer plans.
Sample Selection
The study included patients with at least one prescription drug claim for a TTA (Axiron®, AndroGel® 1%, AndroGel® 1.62%, Fortesta®, or Testim®) between January 1, 2011, and March 31, 2012. Patients were required to have no other TRT claims (any route of administration) in the 12 months preceding and including the index date and only one TTA initiated on the index date. The index date was defined as the first observed TTA claim meeting the washout criteria on or after January 1, 2011. The sample was restricted to male patients aged 18 years or older on the index date, with at least 12 months of continuous health plan eligibility prior to and including the index date. The index claim was required to be for the recommended starting dose (RSD) of the index TTA (Axiron, 60 mg; AndroGel 1%, 50 mg; AndroGel 1.62%, 40.5 mg; Fortesta, 40 mg; and Testim, 50 mg). Furthermore, only patients continuously eligible in their health plans for the 6 months following the index date were included. Finally, as a proxy of a clinical confirmation of low testosterone levels, patients were stratified based on whether they had a serum T assay (CPT codes 84270, 84402, or 84403) in the 30 days prior to and including the index date (hereafter, preinitiation; see Figure 1).
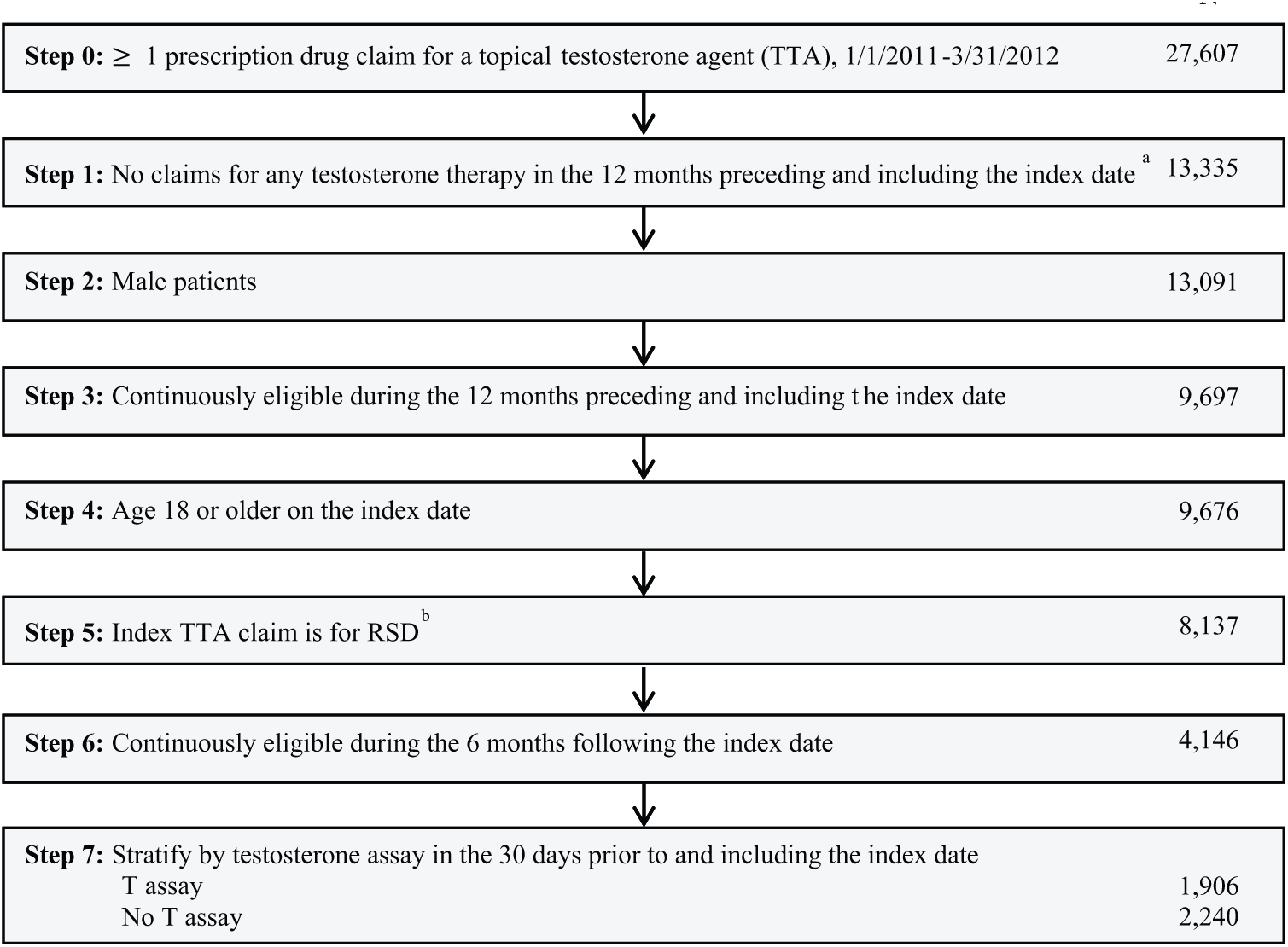
Sample selection of patients with topical testosterone utilization.
Main Outcome Measures
The main outcome measures included proportion of patients with T assay or diagnosis of HG during the 6 months after TTA initiation, maintenance dose attainment month, and time to stopping of index TTA refills or a claim for nonindex TRT. The measures were assessed over the 6 months following treatment initiation.
Definitions
A recorded diagnosis of hypogonadism or other conditions associated with low testosterone (hereafter, low testosterone conditions) was defined by ICD-9-CM codes 257.1-257.9, 253.2, 253.4, 253.7, 259.0, 608.3, 758.7, 752.89, E932.1. Attainment of maintenance dose was defined as the first month without a statistically significant change in the average daily dose compared with the following month at the population level.
Time to stopping or changing therapy was defined as the earlier of when a patient (1) stopped refilling the index TTA (defined as the first day without any index TTA refills for the remainder of the study period) or (2) started using TRT other than the index TTA (defined as observation of any medical or prescription drug claim for a nonindex TTA or TRT with another route of administration; e.g., injection).
Statistical Analysis
Patient characteristics, including age, insurance type, index TTA, concomitant medications, the Charlson Comorbidity Index (Romano, Roos, & Jollis, 1993) and its components, and other selected comorbidities were compared between patients with and without a preinitiation T assay.
Maintenance Dose Attainment
The methodology utilized to determine maintenance dose attainment has been described elsewhere in detail (Kaltenboeck et al., 2013). Because the available topical testosterone products vary greatly in their RSD (i.e., 40 mg to 60 mg of testosterone), dose as a proportion of RSD was calculated to standardize the amount of testosterone across the products. Briefly, dose as a proportion of RSD was calculated as monthly average of dose (the total number of milligrams of testosterone prescribed divided by days of prescription supply) per patient per day divided by the RSD. In this approach, scripts with a refill before the end of days of supply on the claim were assumed to have been used by the date of the refill. Patients were permitted to have gaps in index TTA use (i.e., gaps between the end of one index TTA script and the day the next index TTA script was filled) and were included in the analysis until their doses could not be calculated because of (1) lack of any further refills of the index TTA through the end of the study period or (2) observation of a claim for TRT other than the index TTA.
The mean dose as a proportion of RSD was compared sequentially from Month 1 to Month 6 postinitiation for patients, stratified by preinitiation T assay. The month of maintenance dose attainment was considered to be the first month without a statistically significant difference in dose compared with the following month, using generalized estimating equations as previously described (Kaltenboeck et al., 2013). The maintenance dose analyses were conducted separately for patients with and without a preinitiation T assay; no statistical comparisons were made between patients with and without a preinitiation T assay.
Time to Stopping Index TTA Refills or Filling Nonindex TRT Claim
The total number of patients removed from the sample was compared during each month of the study period and at any point during the study period between patients with and without a preinitiation T assay using chi-square tests. In addition, the reason for patient exclusion from dose calculations (i.e., refill stopping or another form of TRT) was compared between patients with and without a preinitiation T assay using chi-square tests, for all patients excluded at any point in the study period. Among patients with refill stopping or another form of TRT, the mean time on index TTA was compared using Wilcoxon rank-sum tests between patients with and without a preinitiation T assay.
Monitoring Patterns
Finally, to assess patient monitoring after TTA initiation, the proportion of patients receiving a diagnosis of hypogonadism or another condition associated with low testosterone and the proportion of patients receiving a T assay following the index date were compared between patients with and without a preinitiation T assay using a chi-square test. Diagnoses and T assays were assessed for as long as the patient’s daily dose of TTA could be calculated, as described above.
To determine whether monitoring of patients on TTAs differs across physician specialty, patients were stratified by a relevant visit with particular physician specialties (endocrinology and urology) during the month prior to and including the index date, with priority given in the following order: endocrinologist, urologist, and primary care provider (PCP). Among patients remaining on index TTA therapy in each month of the study period, the proportion of patients with a T assay were compared between those seen by a PCP and those seen by an endocrinologist or urologist.
SAS version 9.3 (Cary, North Carolina) was used for all analyses. Statistical significance was defined as a two-sided p value ≤ .05.
Results
Baseline Characteristics
A total of 4,146 patients were selected into the sample, including 1,906 (46.0%) patients with a preinitiation T assay and 2,240 (54.0%) patients without such an assay. Patients with a preinitiation T assay were generally younger (51.8 vs. 56.7 years, p < .001) and were more likely to be insured by a preferred provider organization (53.6% vs. 49.3%, p = .006) or point of service plan (23.3% vs. 16.5%, p < .001; see Table 1). They were more likely to have initiated on a recently approved TTA (AndroGel 1.62%: 11.0% vs. 7.5%, p < .001; Axiron: 5.5% vs. 3.9%, p = .020), and less likely to have used phosphodiesterase-5 inhibitors in the 12 months prior to TTA initiation (12.3% vs. 15.2%, p = .007).
Baseline a Characteristics of Patients With Topical Testosterone Utilization.
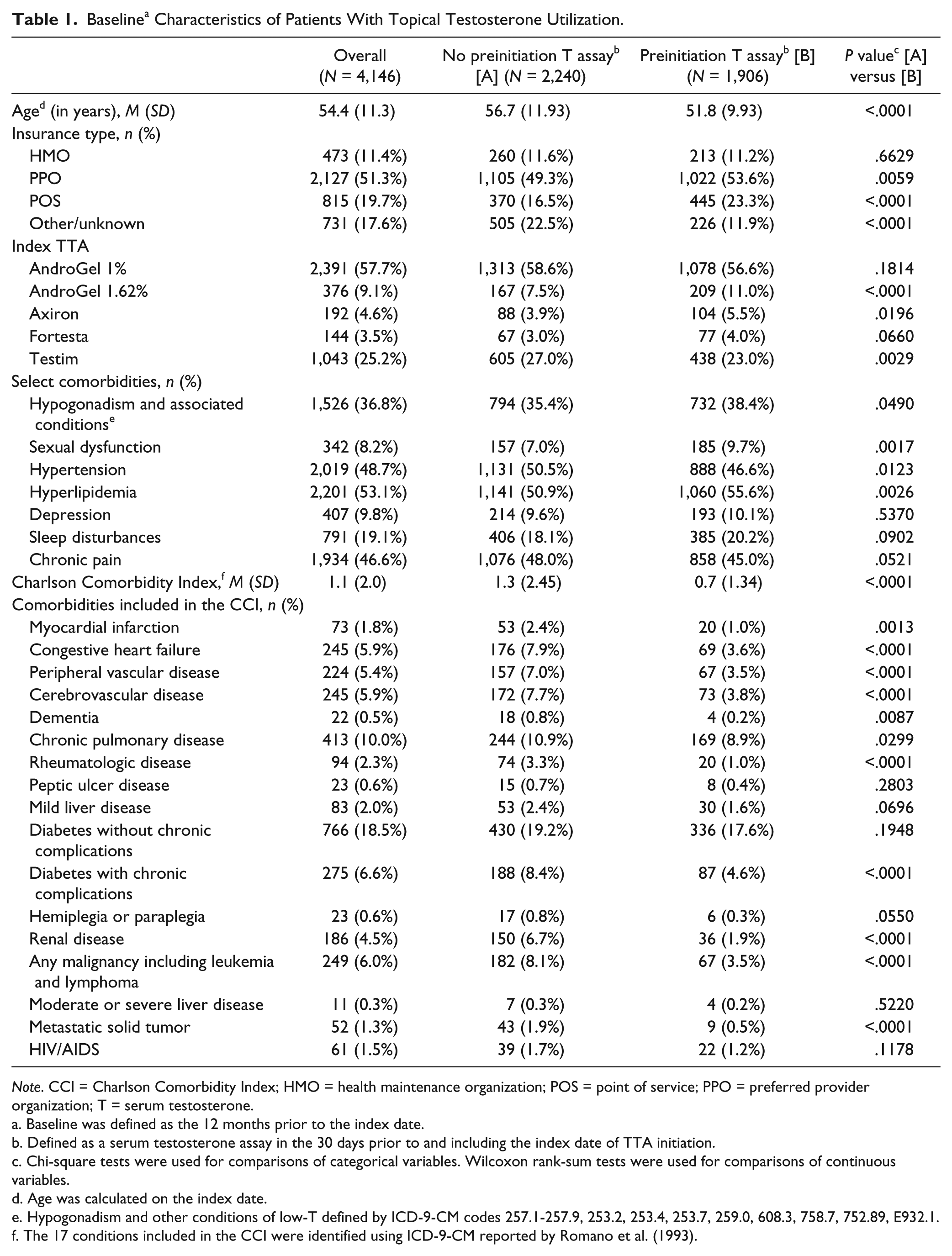
Note. CCI = Charlson Comorbidity Index; HMO = health maintenance organization; POS = point of service; PPO = preferred provider organization; T = serum testosterone.
Baseline was defined as the 12 months prior to the index date.
Defined as a serum testosterone assay in the 30 days prior to and including the index date of TTA initiation.
Chi-square tests were used for comparisons of categorical variables. Wilcoxon rank-sum tests were used for comparisons of continuous variables.
Age was calculated on the index date.
Hypogonadism and other conditions of low-T defined by ICD-9-CM codes 257.1-257.9, 253.2, 253.4, 253.7, 259.0, 608.3, 758.7, 752.89, E932.1.
The 17 conditions included in the CCI were identified using ICD-9-CM reported by Romano et al. (1993).
A higher proportion of patients with a preinitiation T assay had a diagnosis of hypogonadism or other low T conditions (38.4% vs. 35.4%, p = .049), sexual dysfunction (9.7% vs. 7.0%, p = .002), and hyperlipidemia (55.6% vs. 50.9%, p = .003). However, other comorbidities were generally more prevalent in patients without a preinitiation T assay, as reflected in lower mean CCI for patients with a preinitiation T assay (0.7 vs. 1.3, p < .001).
Maintenance Dose Attainment
Among all patients, maintenance dose was attained in the fourth month after TTA initiation, at which time patients were taking on average 115.2% of RSD (Figure 2). After stratification, trends in dose titration were qualitatively similar in patients with and without a preinitiation T assay, although patients with a preinitiation T assay had slightly higher doses in each month after initiation (Figure 2). Patients in both groups were required to have filled the index TTA prescription at the RSD, calculated based on quantity in milligram and days of supply on the claim, but refill patterns indicated that some used the prescription at a higher dose than originally indicated by the index claim; in the first month postinitiation patients used on average over 105% of the RSD. In both groups, maintenance dose was attained in the fourth month after initiation, and dose was approximately 115% of RSD. Following maintenance dose attainment, small dose increases were observed in both groups, but were not statistically significant.

Average dose PPPD as a proportion of recommended starting dose following TTA initiation.
Time to Stopping Index TTA Refills or Filling Nonindex TRT Claim
A total of 2,620 patients (63.2%) stopped refilling the index TTA or filled a nonindex TRT claim at some point during the 6 months after TTA initiation (Table 2), and 1,858 patients (44.8%) stopped refilling or filled a nonindex TRT claim by the end of the third month after TTA initiation. There were no significant differences in time to refill stopping or nonindex TRT claims between patients with and without a preinitiation T assay. A total of 1,229 (64.5%) and 1,391 (62.1%) of patients with and without a preinitiation T assay either stopped refilling their index TTA or filled a prescription for TRT other than the index TTA during the 6-month study period, respectively (p = .113; see Table 2). The mean time to stopping refills or filling a nonindex TRT script was 72.7 versus 70.9 days (p = .477) in patients with and without a preinitiation T assay, respectively.
Monitoring Patterns Following the Index Date Prior to Refill Stopping or Nonindex TRT Claim.

Note. T = serum testosterone; TTA = topical testosterone agent.
Defined as a serum testosterone assay in the 30 days prior to and including the index date of TTA initiation.
Chi-square tests were used for comparisons of categorical variables.
HG and other conditions of low-T defined by ICD-9-CM codes 257.1-257.9, 253.2, 253.4, 253.7, 259.0, 608.3, 758.7, 752.89, E932.1.
Serum testosterone assays defined by CPT codes 84270, 84402, and 84403.
Over the study period, nearly half (43.4%) of all patients stopped refilling the index TTA, but patients with a preinitiation T assay were significantly less likely to stop refilling the index TTA (41.3% vs. 45.2%, p < .001; see Figure 3). They were also more likely to continue TRT by filling a prescription for nonindex TTA (12.4% vs. 9.2%, p = .003) or a non-TTA TRT (10.7% vs. 7.7%, p = .002).
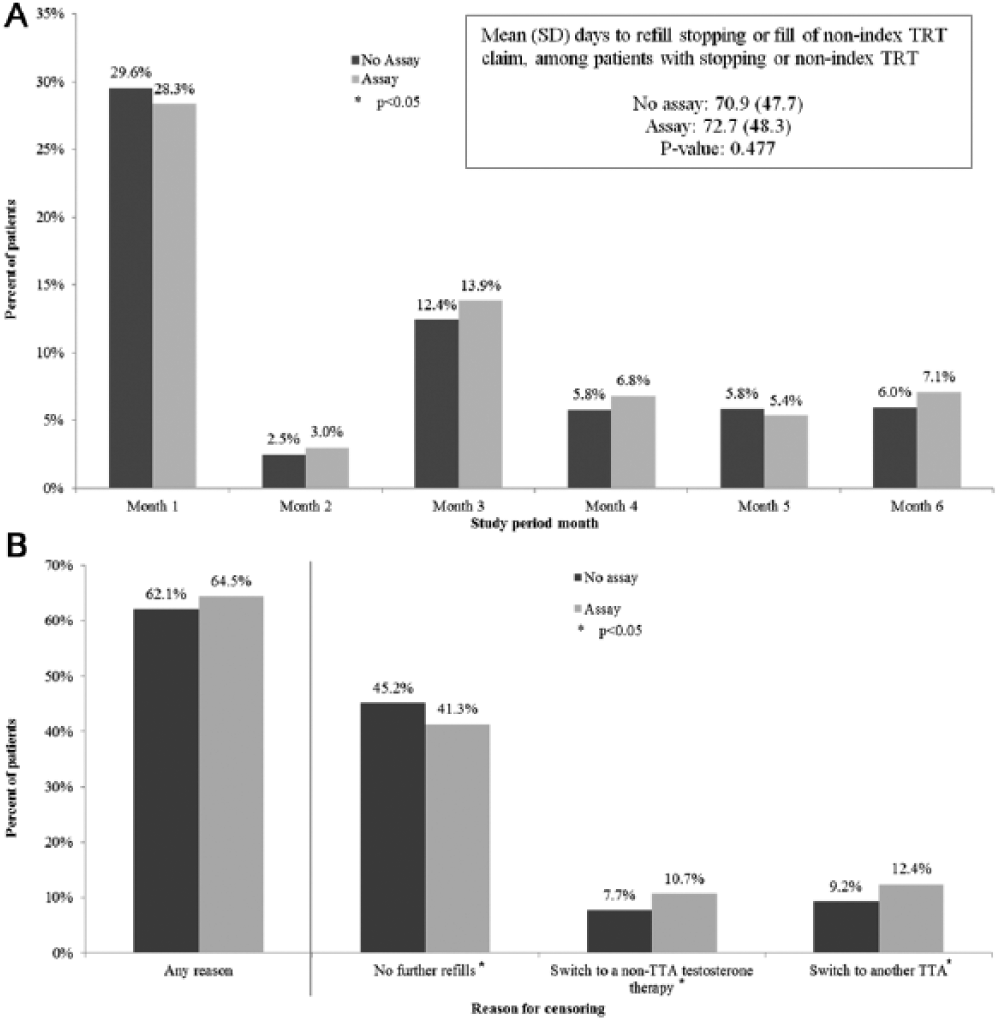
Index TTA refill stopping and nonindex TRT claims during the study period stratified by assay during the 30 days prior to and including the index date. (A) Month of first index TTA refill stopping or nonindex TRT. (B) First reason for inability to calculate daily dose.
Monitoring Patterns
During the 6 months following the index date, patients with a preinitiation T assay were more likely to receive a diagnosis of hypogonadism or another low T condition (44.7% vs. 35.8%, p < .001; see Table 2). More than half (50.6%) of patients with a preinitiation T assay had at least one additional T assay in the 6 months following the index date, while only 28.3% (p < .001) of patients without a preinitiation T assay had a T assay in the 6 months after TTA initiation. Among patients remaining on index therapy, patients most frequently received a T assay during the third month after initiation: 15.6% of patients on index therapy in the third month received a T assay (Figure 4). In general, when stratified by visit to particular physician specialties in the 30 days prior to and including the index date (regardless of receipt of a preinitiation T assay), patients seen by endocrinologists (10.2% to 25.2%) or urologists (10.7% to 23.8%) in the 30 days prior to TTA initiation were more likely to receive a T assay than those seen by PCPs (7.7% to 15.7%) in the 30 days prior to initiation.

Serum testosterone assays during the study period,a stratified by physician specialtyb visited in the month prior to TTA initiation.
Discussion
This study aimed to describe real-world care after initiation of TTAs in men who received a T assay in the 30 days prior to or on the initiation date, compared with men who had no T assay in this timeframe. In this study, 46.0% of patients received a T assay in the 30 days prior to TTA initiation. This represents real-world clinical care, which may deviate from guideline-based care, under which all patients would have received a preinitiation T assay (Bhasin et al., 2010). Patients with a preinitiation T assay were younger and had fewer comorbidities at baseline, although they were more likely to receive a diagnosis of HG or another low T condition. In both groups, maintenance dose was attained in Month 4 after initiation, at approximately 115% of RSD, which is consistent with a previous report (Kaltenboeck et al., 2013).
Index TTA refill stopping or nonindex TRT claims were observed in 44.8% of patients during the first 3 months after TTA initiation, including as early as the first month (29.0%). Similarly, a recent publication by Baillargeon, Urban, Ottenbacher, Pierson, and Goodwin (2013) observed that almost 20% of androgen replacement therapy users received therapy for less than 1 month. In our study, treatment interruptions occurred well before maintenance dose was attained in the overall sample, suggesting that those patients may not have used the medication for a sufficient length of time to experience symptom improvement and therefore may not be receiving the maximum benefits of TTA therapy. Previous research suggests that many patients discontinue prescribed therapy because of lack of efficacy, inconvenience, side effects, inadequate patient education, or unrealistic patient expectation for symptom alleviation (Jin, Sklar, Min Sen Oh, & Chuen Li, 2008). Closer monitoring and education on reasonable expectations of time usually needed to achieve therapeutic dosing levels may help avoid some of these causes of TTA therapy interruption.
This study observed that 46.0% of patients initiating TTA therapy received a T assay in the 30 days prior to initiation. This is lower than a previously reported proportion of 74.7%, but that study assessed T assays in the year prior to androgen replacement therapy initiation (Baillargeon et al., 2013). However, T assays in the 30 days prior to TTA initiation may be the most clinically relevant. Doubling the period during which preinitiation T assays were assessed to 60 days yielded a only small number of additional patients with T assays (from 46% to 57%). Patients who received a T assay prior to TTA initiation were both less likely to stop refilling their index TTA and more likely to fill a script for another TRT or nonindex TTA. As with any long-term therapy, patient compliance with treatment recommendations can be quite poor (as low as 30% to 40%; Martin, Williams, Haskard, & DiMatteo, 2005) and is a crucial factor in treatment success. There are multiple reasons for nonadherence, including a lack of patient understanding of what recommendations they are to follow. It is important for clinicians to engage with and counsel their patients to explain the condition, discuss treatment options, and set treatment expectations (Dobs, 2008; Gooren & Bunck, 2004). Establishing a diagnosis through T assays may help physicians guide the therapy and allow patients to maintain realistic expectations about their response to therapy. In our study, patients with preinitiation T assays were less likely to stop refilling their index TTA and more likely to continue TRT by filling a script for another nonindex TRT, which may indicate that patients without a response to their index TTA switched to another TRT rather than discontinuing. A larger proportion of patients who received a T assay prior to TTA initiation received a study period T assay, compared with those without a preinitiation assay, suggesting they may have also benefited from closer monitoring of treatment than patients without a preinitiation T assay.
Finally, T assay testing patterns differed according to whether patients had visited a particular physician specialty in the 30 days prior to initiating TTA therapy. This suggests that patients with a history of treatment by a particular physician specialty (i.e., endocrinology or urology) versus a PCP within the 30 days prior to and including TTA initiation (regardless of preinitiation T assays) may have been more closely monitored during the study period.
Limitations
This study was limited by typical issues associated with claims data analyses, including reliance on accuracy of diagnoses and services recorded in the claims, and lack of clinical measures, including serum testosterone levels, symptoms, and compliance with a therapy. However, measuring pharmacy patterns of utilization (i.e., refills) over time is a common way to assess persistence in the absence of clinical data. Variations in the structures of the included health plan (e.g., plan tiers, formulary coverage, refill limits, etc.) could not be assessed but may affect cost and coverage of TRT, and therefore utilization of TTAs and services (Gibson, Ozminkowski, & Goetzel, 2005; Piette, Heisler, & Wagner, 2004). Furthermore, the methods used to assess stopping of refills or claims for other TRT in this study were developed to facilitate dose calculations and are not comparable with claims algorithms often used to define discontinuation or switching. Rather, the study identified the time until no further claims of index TTA were filled (refill stopping) within the study period, or the time until a claim for another form of TRT than the index TTA.
The study is descriptive, as no multivariate analysis was conducted during the study, and potential unmeasured confounders were not controlled. The stratification in this study was based on a record of T assays in the 30 days prior to TTA initiation because these are generally the most clinically relevant T levels to guide titration; however, it is possible that physicians also use results of previous T assays completed during the previous 6 months to guide TTA initiation. Testosterone levels measured at earlier time points, such as 2 months before initiation, may not vary significantly from levels within the month prior to initiation. To the extent that patients with T assays within 2 months prior to initiation are similar to patients with T assay within 30 days prior to initiation, but are included in the group without preinitiation T assay, the study findings may underestimate differences between patients with and without preinitiation T assay.
Finally, the database analyzed covers a commercially insured population that may not be geographically representative of the United States. Patients older than 65 years who are dually eligible for Medicare may not be representative of all patients older than 65 years. Because of the requirement of continuous insurance coverage for at least 1 year prior to TTA initiation, patients who changed insurance frequently were unlikely to be represented in the sample. Generalizability to populations with other (i.e., noncommercial) insurance coverage may be limited.
Conclusions
TTAs represent an important form of TRT for testosterone-deficient men, who may be susceptible to burdensome comorbidities. Patients who understand their medical conditions and the benefits and goals of treatment may be more likely to adhere to their treatment recommendations (Martin et al., 2005). In this study, patients who initiated a TTA with a preinitiation T assay were less likely to stop refilling their index TTA and more likely to receive a T assay to monitor TRT during the study period. As suggested by guidelines, T assays prior to and after initiation of a TTA help set expectations about treatment response and allow physicians to monitor dose titration to appropriate serum testosterone levels.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by Eli Lilly and Company, Indianapolis, IN, USA.
