Abstract
Numerous reports point to the beneficial effects of testosterone replacement therapy for patients with late-onset hypogonadism (LOH) syndrome. The aim of this study was to evaluate the effect of intramuscular injection of testosterone enantholactam acid ester on Aging Males’ Symptoms (AMS) scores in hemodialysis patients with LOH. A total of 24 male patients with LOH (total AMS scores ≥27) were randomized into groups receiving intramuscular injections of either placebo or testosterone enantholactam acid ester at the dose of 250 mg for 6 months. In all, 13 and 11 participants from the active treatment and placebo groups, respectively, completed this study. An intramuscular injection of either placebo or testosterone enantholactam acid ester was given every 2 weeks. Self-administered AMS questionnaires were completed at the start, at Week 12 and at Week 24. The total AMS score was significantly more improved in the treatment group than in the placebo group (p = .049) during the 24-week period. The change in the mean of total AMS score was +1% in the placebo group and −13.2% in the treatment group. The mean somato-vegetative domain scores decreased significantly only in the treatment group, and not in the placebo group (−1.21 vs. −2.43, p = .028). Although a large-scale study is needed, testosterone treatment may be effective in male patients with hemodialysis who have poor health-related quality of life resulting from LOH.
Keywords
Age-related decreases in testosterone levels result in a disease known as late-onset hypogonadism (LOH) syndrome. Decreasing testosterone in aging males results in depression, sexual dysfunction, decreased cognitive function, osteoporosis, an increase in visceral fat, a decline in both insulin sensitivity and high-density lipoprotein (HDL) cholesterol, and an increase in both total cholesterol and low-density lipoprotein (LDL) cholesterol, all of which are risk factors for metabolic syndrome, eventually causing cardiovascular disease, diabetes, and respiratory illness (Bhasin et al., 2006; Kaushik, Sontineni, & Hunter, 2010; Nieschlag et al., 2005; Shabsigh, Katz, Yan, & Makhsida, 2005; Shores et al., 2004). Epidemiological studies in humans also report that low testosterone levels are associated with chronic kidney disease (CKD; Khurana et al., 2014). Serum concentrations of total and free testosterone were significantly reduced in hemodialysis patients. Several studies suggested that testosterone concentrations inversely correlate with all-cause and cardiovascular disease related mortality, and low testosterone levels at the time of transplantation are associated with patient and graft loss among male dialysis patients (Carrero et al., 2009; Shoskes, Kerr, Askar, Goldfarb, & Schold, 2014). Although the etiology is likely multifactorial because many hemodialysis patients have comorbidities that can contribute to decreased testosterone including diabetes, vascular disease, atherosclerosis, older age, and obesity, it would appear that testicular dysfunction becomes more severe with increasing age. At minimum, a low serum level of testosterone may be a risk factor for adverse outcomes and poor quality of life in hemodialysis patients. Testosterone deficiency is a major contributor to muscle atrophy in hemodialysis patients, in that it might suppress muscle protein synthesis directly and indirectly through the inflammatory process (Cobo et al., 2017; Sasagawa et al., 1998).
More than five decades of clinical experience has demonstrated that testosterone replacement therapy improves various LOH symptoms. During the past decade, testosterone prescriptions in the United States have increased fourfold, growing 25%–30% annually, with costs reaching 1.6 billion dollars by 2011 (Spitzer, Huang, Basaria, Travison, & Bhasin, 2013). Pharmaceutical preparations of testosterone come in both injectable and gel form. Recently, 859 hypogonadism patients were reported to have participated in replacement therapy using testosterone gel in a multi-institutional study (Testim Registry in the United States: TriUS). The topical gel was prescribed over a 12-month period, elevating testosterone and free testosterone levels, improving sexual function, and alleviating depression (Khera et al., 2011, 2012). Testosterone replacement therapy also improved sexual activity, sexual desire, and erectile function in older men (Cunningham et al., 2016; Snyder et al., 2016).
The data on use of testosterone in chronic renal disease patients or hemodialysis patients are even more limited. One report suggested that testosterone replacement therapy in men with end-stage renal disease improved scores on the International Index of Erectile Function (IIEF) and frequency of weekly sexual intercourse (Canguven et al., 2010).
This pilot study attempts to evaluate if testosterone supplementation therapy improves the symptoms of LOH in hemodialysis patients, because there are few randomized controlled trials that have investigated the outcome of testosterone supplementation therapy in such populations.
Methods
A double-blind, parallel, randomized, placebo-controlled trial was conducted as the pilot study. Participants were recruited in Nagakubo Hospital. All patients gave written informed consent, and approval was obtained from the Research Ethics Board of Teikyo University School of Medicine (Approval number:13-149). The inclusion criteria were: over 40 years of age and total AMS score ≥27. The exclusion criteria were: uncontrolled diabetes mellitus; clinical hypothyroidism or hyperthyroidism; known prostate cancer; other significant medical conditions or psychological conditions. The primary endpoint of the present study was treatment effect on AMS scores. The secondary endpoints were adverse events and changes in laboratory blood results, that is, total testosterone, free testosterone, and hemoglobin level. The study utilized AMS scores, which indicate severity of subjectively perceived complaints in each of the 17 items on a scale of 1 to 5, with a higher score meaning greater symptom severity (Moore et al., 2004). The three domains of the AMS scale are psychological, somato-vegetative, and sexual. AMS scores are common symptom scores used around the world. Horie et al. (2014) compared AMS scores to those from the Japanese Aging Male Questionnaire (JAMQ) and reported that a maximum coefficient of correlation existed with the corresponding AMS subgroup. The severity of the symptoms is defined as: no/low (17–26 points), mild (27–36 points), moderate (37–49 points), and severe (≥50 points). Hemodialysis patients with LOH (total AMS scores ≥27) were randomized into groups receiving intramuscular injections of either placebo (saline) or testosterone enantholactam acid ester 250 mg. Participants, who gave written informed consent to participate, underwent initial screening tests which included an early morning total testosterone test and the completion of the AMS questionnaire. In all, 13 and 11 participants from the active treatment and placebo groups, respectively, completed the study. An intramuscular injection of either placebo or enantholactam acid ester was given at every 2 weeks before the dialysis. Self-administered AMS questionnaires were completed at Weeks 0, 12, and 24. In addition, assessments were made of body mass index, abdominal circumference, body fat, grip strength, serum testosterone (ng/ml), free testosterone (pg/ml), and blood hemoglobin level (g/dl). Blood samples were collected between 7 and 11 a.m. before hemodialysis and all analyses were performed in the same laboratory. Serum testosterone levels were measured using an ARCHITECT Testosterone Assay (Abbott Japan, Tokyo, Japan). Free testosterone levels were measured by radioimmunoassay (Beckman Coulter, Minnesota, USA). Body fat was calculated by the bioelectrical impedance analysis.
The effects of active treatment on AMS scores were estimated using repeated measure analysis of variance (ANOVA) by including the intervention x time interaction terms. T-test was used for continuous data. The two-sided level of significance (p) was set at .05. Data analysis was done using the Statistical Package for the Social Sciences (SPSS Inc., Chicago IL, USA) version 15.
Results
Twenty-four patients were included in the study. In all, 13 and 11 participants from the active treatment and placebo groups, respectively, completed the study. Their mean age ± SD were 60 ± 12.4 and 69 ± 11.6 years, respectively. There were no statistical differences in factors such as age, body mass index, and abdominal circumference (Table 1) between placebo and testosterone supplementation groups before treatment. In the placebo group, 10 out of 13 patients had less than 5 ng/ml of serum testosterone. In the testosterone supplementation group, six of 11 patients had less than 5 ng/ml of serum testosterone. Before testosterone supplementation therapy, patients of the testosterone supplementation group had a mean serum testosterone of 4.71 ± 1.41 (ng/ml) which significantly increased to 10.44 ± 3.05 (ng/ml) and 10.52 ± 4.24 (ng/ml) after 3 months and 6 months, respectively (p < .001; Figure 1A). A statistically significant increase in free testosterone level was detected after testosterone therapy (p < .001) (from 5.40 ± 1.70 [pg/ml] to 20.02 ± 5.13 [pg/ml] and 17.29 ± 7.82 [pg/ml], respectively; Figure 1B). There was no statistically significant increase in either total testosterone or free testosterone level in the placebo group (Figure 1B). No major complications, such as cardiovascular events, were reported. The hemoglobin levels from Months 0 to 6 in the placebo and testosterone supplementation groups were 10.76 ± 0.7, 10.93 ± 0.8, 10.98 ± 0.8 (g/dl), and 10.58 ± 1.3, 10.95 ± 1.6, 10.3 ± 1.5 (g/dl), respectively. No improvements in hemoglobin level between the placebo and testosterone supplementation groups were observed (Figure 1C). Improvement in the total AMS score was significantly greater in the treatment group than in the placebo group (p = .049) over the 24-week period (Figure 2). The mean (SD) total AMS score was 38.92 (11.48) at baseline, 41.58 (11.89) at 12 weeks, and 39.3 (11.25) at 24 weeks for the placebo group, and 36.0 (10.16) at baseline, 31.18 (8.13) at 12 weeks, and 31.25 (7.83) at 24 weeks for the treatment group. The mean change in the total AMS score was +1% in the placebo group and −13.2% in the treatment group (Figure 2). Over the 48-week period, the mean somato-vegetative domain scores only decreased significantly in the treatment group, and not in the placebo group (−1.21 vs. −2.43, p = .028). There were no significant differences in the change in psychological and sexual subscale scores between the placebo and the treatment groups (Table 2).
Baseline Characteristics of the Placebo and Testosterone Replacement Treatment (TRT) Groups.


Comparison of serum testosterone levels (A), serum free testosterone levels (B), and hemoglobin (C) in the testosterone replacement treatment (TRT) treated with 250 mg enantholactam acid ester group versus the placebo group from baseline to 3 months and 6 months using repeated measure analysis of variance.
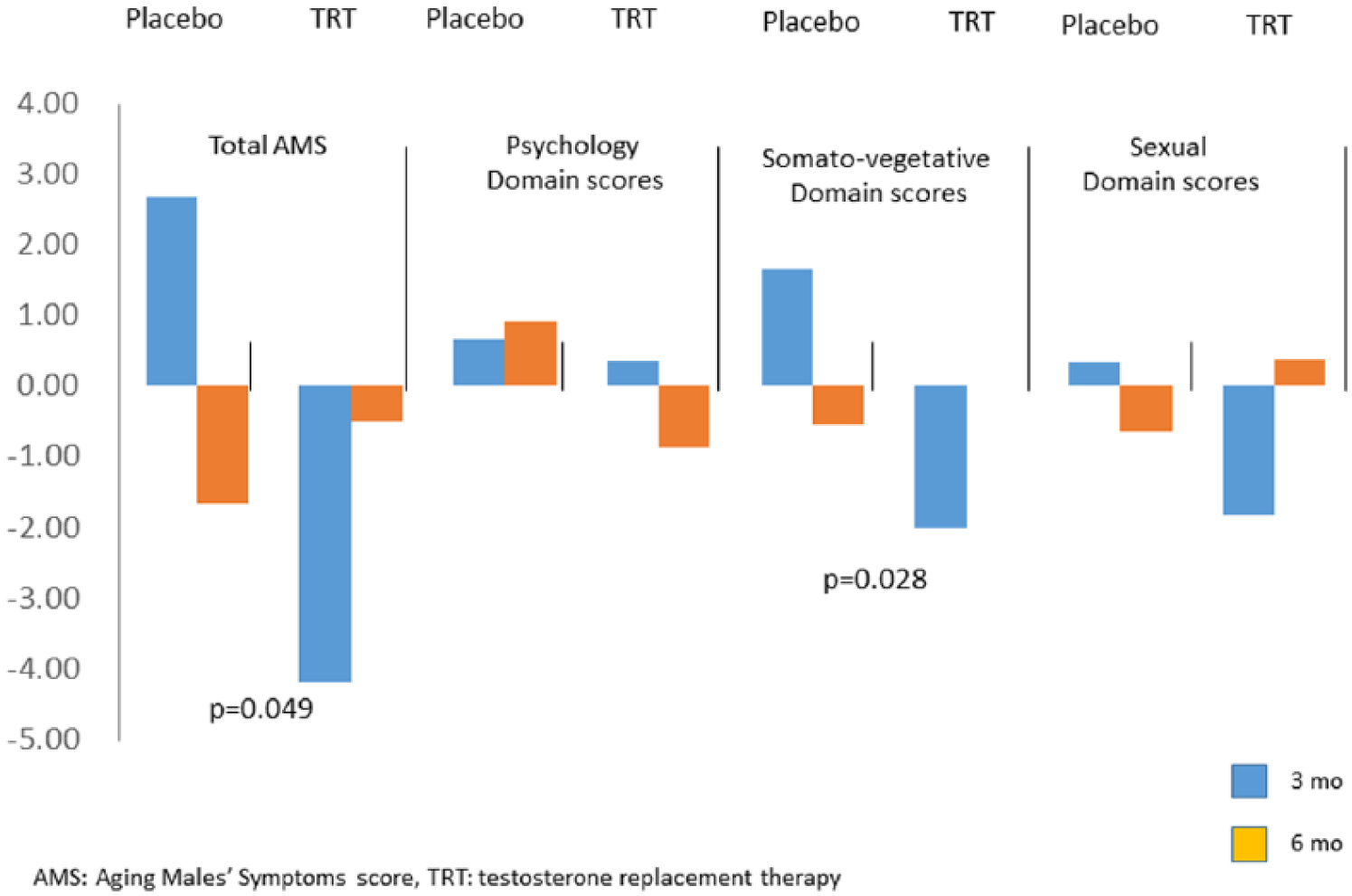
Comparison of changes in the total Aging Males’ Symptoms scores and domain scores in the testosterone supplementation group versus the placebo group.
Comparison of Total Aging Males’ Symptoms (AMS) score and AMS domain scores of the placebo and testosterone replacement treatment (TRT) groups.

Discussion
The population of hemodialysis patients in Japan is increasing, in part, because of longer survival. The most common primary cause of renal failure among new dialysis patients is diabetic nephropathy. Indeed, the mean age of new dialysis patients is 68.7 years and the number of patients with diabetes mellitus (DM) is increasing (Khurana et al., 2014; Masakane et al., 2015). The greater longevity indicates a rapidly growing number of elderly patients among the incident hemodialysis population who have a high prevalence of comorbidities according to decreasing of testosterone. In addition, there are few brain-dead cadaveric donors in Japan and the number of kidney transplants is one-tenth that of the United States. Therefore, the ratio of patients under long-term dialysis has not decreased (Yoshimura et al., 2010). The importance of quality of life for these patients must be highlighted. Indeed, erectile dysfunction (ED) is common in hemodialysis patients (Canguven et al., 2010). In addition, low total testosterone levels have been associated with higher all-cause mortality in these men. A study of men with CKD and low total testosterone indicated significantly increased all-cause mortality (Khurana et al., 2014). Although testosterone deficiency is a prevalent condition in men with hemodialysis (Bello et al., 2014), it has so far received scant medical attention. The effects of testosterone supplementation on sexual dysfunction in end-stage renal disease or transplants are controversial but studies suggest variable improvements (Snyder & Shoskes, 2016). The most important issue is whether testosterone represents a therapeutic target for intervention in men who are hemodialysis patients, and whether there is a window of opportunity for affecting outcomes. Thus, the objective of the current study was to investigate the effects of testosterone supplementation therapy in men receiving hemodialysis.
Previous analysis reports that when total serum testosterone levels drop as a result of LOH, likely symptoms include a decline in morning erections and libido, as well as ED (Wu et al., 2010). This study was conducted based on the hypothesis that exogenous testosterone would associate with muscle strength and physical activity, as well as with sexual activity. The increase in testosterone levels was associated with significantly increased somato-vegetative domain scores, as assessed by the AMS score (p = .028). In this pilot study, there were no effects of testosterone therapy on sexual function. One of the reasons is the low sexual activity of Japanese. In addition, the pathogenesis of ED in hemodialysis patients is manifold and consistent including hormonal disturbances, zinc deficiency, peripheral neuropathy, and peripheral vascular disease. In the corpora cavernosa in patients treated with hemodialysis, there is major damage in the structure of smooth muscle cells, and an increase of interstitial collagen fibers (Bellinghieri et al., 2004).
Previous reports suggest that testosterone deficiency has been associated with anemia and increased resistance to erythropoietin-stimulating agents in dialysis patients (Carrero & Stenvinkel, 2012). The current results suggest no improvements in the hemoglobin level between the placebo and testosterone supplementation groups. All these patients have already been treated with erythropoietin-stimulating agents and a greater testosterone level may be required to show clinical benefit in men receiving hemodialysis (Brockenbrough et al., 2006). Longer administration of testosterone therapy in males who are hemodialysis patients may gradually improve hemoglobin levels.
Limitations
There are limitations to the design of this study that should be considered. The design did not limit testosterone levels and, therefore, the possibility that the effects of the testosterone supplementation therapy were secondary to the placebo effect cannot be excluded. A further limitation of this study is its relatively small sample size and relatively short follow-up period. This is regarded as a pilot study that will hopefully lead to further investigations including larger numbers of participants over a longer duration. The ultimate goals of testosterone supplementation are to maintain or regain the highest quality of life and reduce disability. Hypogonadism may be an additional treatable risk factor for patients receiving hemodialysis.
Conclusions
This pilot study attempted to evaluate if testosterone supplementation therapy improves the symptoms of LOH in hemodialysis patients. The study supports the evidence that testosterone treatment may be beneficial in male hemodialysis patients who have poor health-related quality of life resulting from LOH, and a larger study on the impact of testosterone supplementation therapy is essential.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
