Abstract
Objective:
Up to 15% of lung cancer patients have multiple suspicious nodules. While some of these nodules may represent metastatic lung cancer, others represent synchronous multiple primary lung cancer (SMPLC). The incidence of SMPLC ranges from 0.8% to 8.4% and appears to be increasing. Inconsistent identification of SMPLC can be detrimental for patients who are misdiagnosed as having intrapulmonary metastasis and not offered stage-based treatment. We sought to identify the contemporary incidence of SMPLC at a tertiary institution.
Methods:
From January 2018 to September 2019, patients who underwent lung cancer resection were retrospectively reviewed. Patients with SMPLC were identified using the modified Martini-Melamed criteria.
Results:
During the 21-month period, 227 patients underwent lung cancer resection. There were 47 patients (20.7%) who had 119 pathologically confirmed SMPLC. Most patients had ipsilateral tumors (n = 24, 51.1%) with at least 1 adenocarcinoma (n = 40, 85.1%). Considering histologic subtyping, 38 (80.9%) had histologically distinct tumors. Overall and cancer-specific survival at 4 years was 86% and 90%, respectively. Only patients with 3 or more SMPLC had poor 4-year overall (P = 0.002) and cancer-specific survival (P = 0.043) compared with those with 2 SMPLC. Patient demographics, histology, tumor location, and highest pathologic staging did not affect survival outcomes.
Conclusions:
Using a strict inclusion criterion, the incidence of SMPLC is higher than previously reported. SMPLC patients have favorable survival outcomes, suggesting that they behave like primary lung cancer, not intrapulmonary metastasis. Awareness of SMPLC by thoracic surgeons is critical in optimizing outcomes in this patient population.
Keywords
Central Message
Patients often present with multiple suspicious lung nodules, some of which represent SMPLC. Enhanced imaging and screening have improved identification and survival of SMPLC patients. Adoption of standardized criteria and awareness among the thoracic community are vital to ensure optimal management of patients with SMPLC.
Introduction
Synchronous multiple primary lung cancers (SMPLC) consist of multiple unrelated primary lung cancers that occur simultaneously. Recent evidence suggests that the incidence of SMPLC is rising.1–3 This rise can be attributed to improved accessibility to lung cancer screening and advanced imaging techniques, particularly high-resolution computed tomography (CT) and positron emission tomography (PET) scans, which often detbibrect multiple suspicious nodules in lung cancer patients. 3 Patients with multiple lung cancers may fall into 2 categories. In some cases, they may indicate the presence of metastatic disease originating from a single primary lung cancer. In this case, patients are staged with T3, T4, or M1a disease. The second clinical scenario involves unrelated SMPLC.
Diagnostic criteria for SMPLC were initially introduced by Martini and Melamed in 1975 4 and have since been adjusted to accurately distinguish SMPLC from intrapulmonary metastases (IPM). 5 The most recent modifications to the Martini–Melamed criteria involve the use of histologic subtyping in patients with multiple pulmonary nodules to differentiate SMPLC from T3 tumors. 6 However, diagnosing SMPLC remains challenging and can result in misdiagnosis of patients with SMPLC as T3 (same lobe), T4 (ipsilateral different lobe), or M1a (contralateral lung) disease. 1 Incorrect staging of these tumors can lead to inadequate treatment and have an adverse impact on patient survival. We sought to identify the contemporary incidence of SMPLC in patients referred for surgical resection of lung cancer at a single high-volume academic institution.
Methods
This study received formal approval from the institutional review board. From January 2018 to September 2019, consecutive patients who underwent lung cancer resection at a single academic center were reviewed. Modified Martini–Melamed (Table 1) diagnostic criteria were used to identify patients with SMPLC. Patients were excluded if they presented with only 1 tumor or extrapulmonary metastatic cancer. Data regarding patient demographics, preoperative assessments, surgical treatments, pathology, mortality, and follow-up information were collected from medical records, radiographic studies, and hospital computer information systems.
Modified Martini-Melamed Criteria.

Adapted from Smith et al. 7
Statistical Analysis
Survival was calculated from the date of the initial surgery to the date of last follow-up. Death from any cause determined overall survival (OS). Patients who died of noncancer-related reasons were censored in cancer-specific survival (CSS). Continuous variables are reported as means ± standard deviation, and categorical variables are presented as percentages. OS and CSS were determined using the Kaplan–Meier method, with P values reported by log-rank analysis. Analyses were performed using R Version 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria). A P value <0.05 was considered statistically significant.
Results
During the 21-month study period, a total of 227 patients who underwent resection for 297 lung cancers were retrospectively reviewed to identify patients with SMPLC. Of those, 47 patients (20.7%) had 119 distinct tumors that were pathologically confirmed SMPLC and met our inclusion criteria. Among patients with SMPLC, 19 SMPLC (40.4%) were diagnosed with cancer as a result of lung cancer screening. All patients included in the study underwent high-resolution CT and PET scans, with brain imaging performed in patients who met our indication criteria, which includes the presence of neurologic symptoms and/or a headache, N1 disease, or tumor(s) greater than 4 cm in size. Patients with SMPLC were more commonly female patients (n = 27, 57.4%), reported present/prior tobacco use (n = 41, 87.2%), and more often had a history of hypertension (n = 31, 66.0%) and hypercholesterolemia (n = 32, 68.1%). A significant proportion (n = 18, 38.3%) had a history of previous extrapulmonary cancer. Patient demographics are detailed in Table 2.
Characteristics of Patients With Synchronous Multiple Primary Lung Cancers.
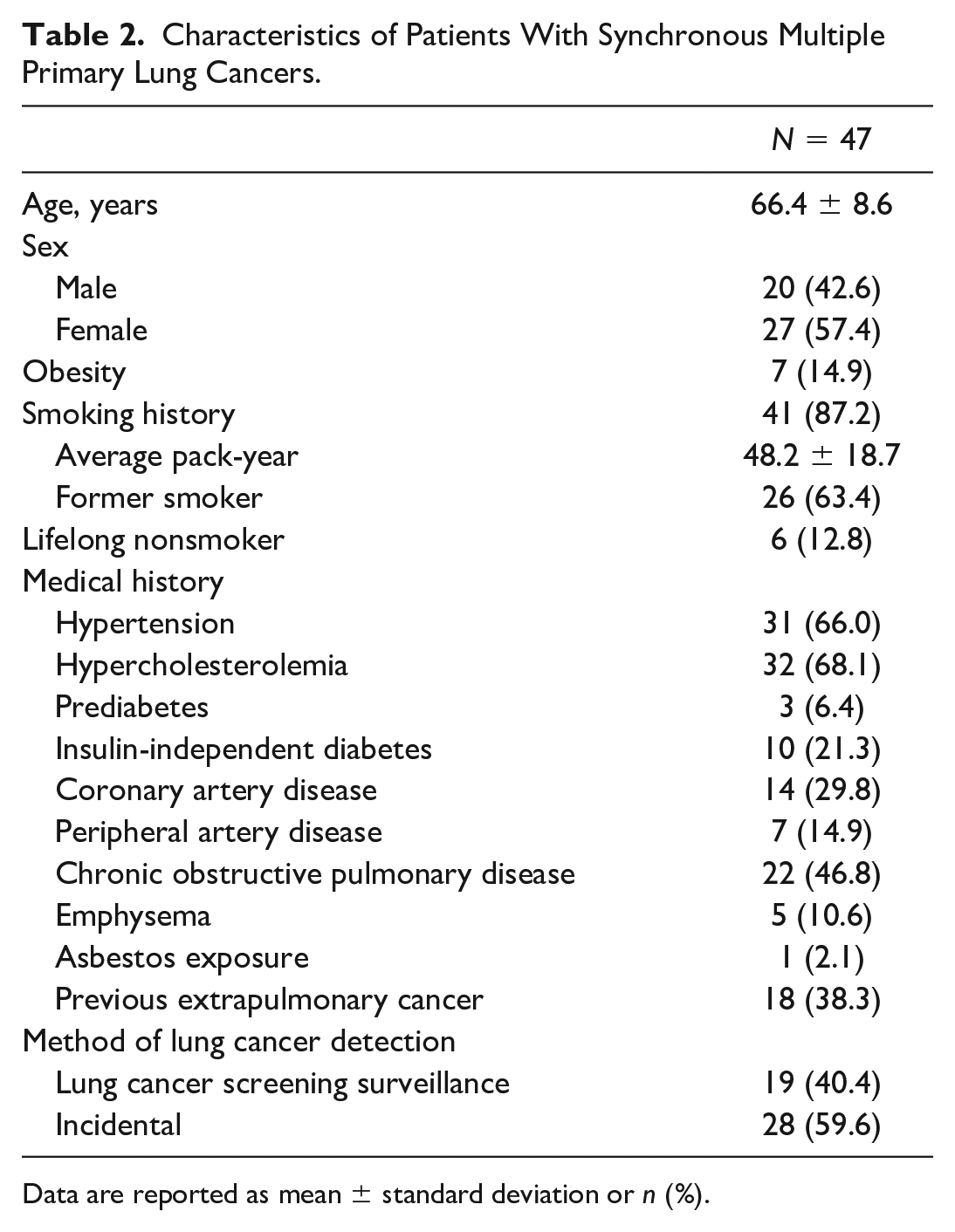
Data are reported as mean ± standard deviation or n (%).
Histologically distinct tumors were found in 38 patients (80.9%), while the same histological subtype was found in 9 (19.1%). Adenocarcinoma was present in at least 1 tumor in 40 of the 47 patients (85.1%), squamous cell carcinoma was present in 10 (21.3%), carcinoid in 7 (14.9%), and large cell carcinoma in 2 (4.3%).
Ipsilateral tumors were found in 24 of 47 patients (51.1%) while bilateral tumors were found in 23 patients (49.4%). The distribution of SMPLC in patients with ipsilateral and bilateral tumors is depicted in Figure 1. In patients with ipsilateral SMPLC, 14 patients (58.3%) had synchronous tumors located in the same lobe, and 10 patients (41.7%) had at least 1 tumor in a different lobe. Patients were staged based on the highest pathologically staged malignant tumor. The most common pathologic stage was stage IA (n = 34, 72.3%). Three or more SMPLC were seen in 13 patients (27.7%).
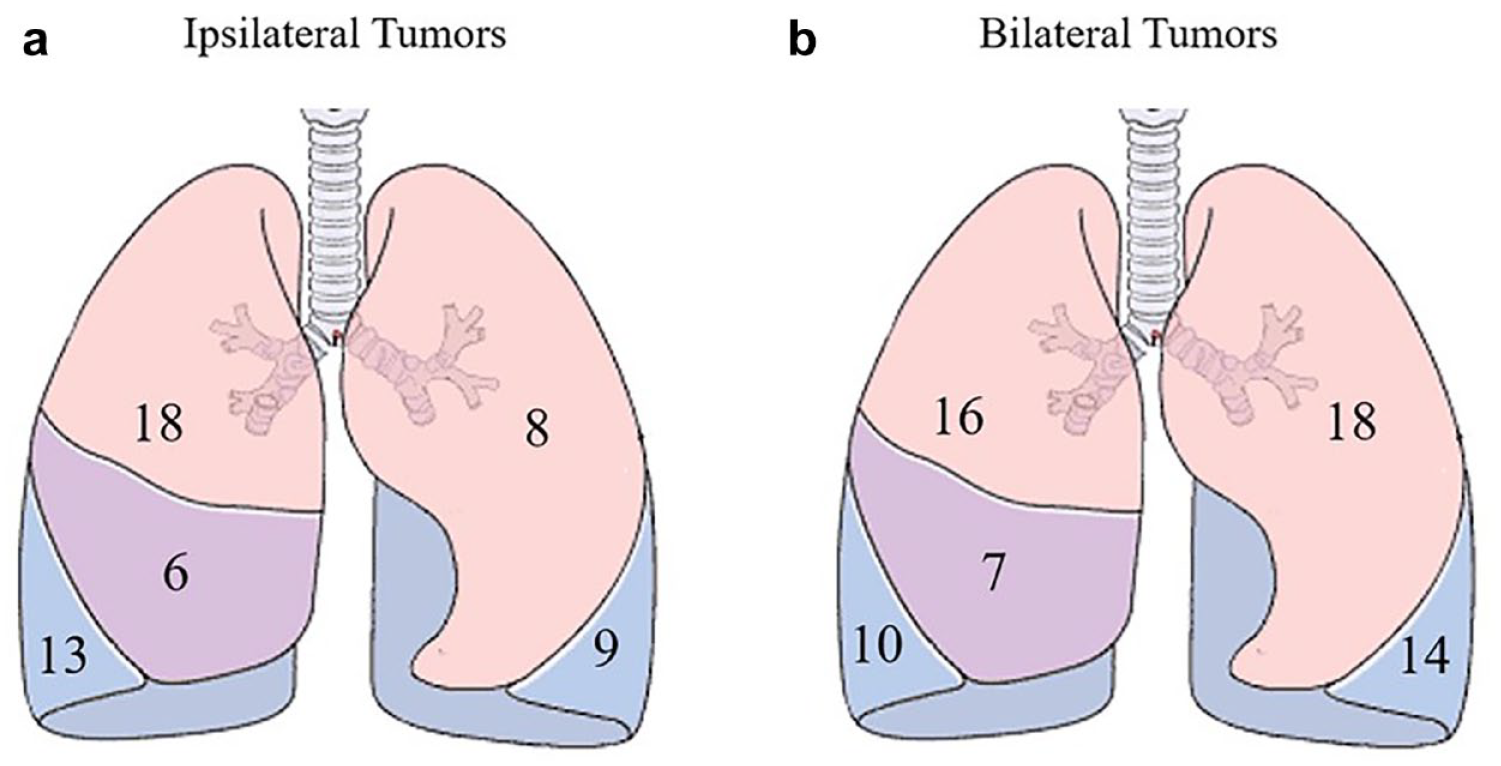
Tumor distribution in patients with (a) ipsilateral and (b) bilateral tumors.
Among the 24 patients with ipsilateral tumors, the most common tumor combinations were 2 adenocarcinomas (n = 16, 66.7%) followed by adenocarcinoma and squamous cell carcinoma (n = 3, 12.5%), 2 carcinoid tumors (n = 3, 12.5%), and adenocarcinoma and other tumor (n = 2, 8.3%). Among the 23 patients with bilateral tumors, the most common tumors combinations were 2 adenocarcinomas (n = 12, 52.2%) followed by adenocarcinoma and squamous cell carcinoma (n = 5, 21.7%), 2 squamous cell carcinomas (n = 2, 8.7%), 2 carcinoid tumors (n = 2, 8.7%), and adenocarcinoma and other tumor (n = 2, 8.7%).
Among the 47 patients, 36 (76.6%) had no nodal involvement, whereas 5 patients (10.6%) had N1 disease at the time of resection. In 6 patients (12.8%), regional lymph nodes could not be ascertained. Tumor details and pathologic stage are shown in Table 3.
Tumor Characteristics.
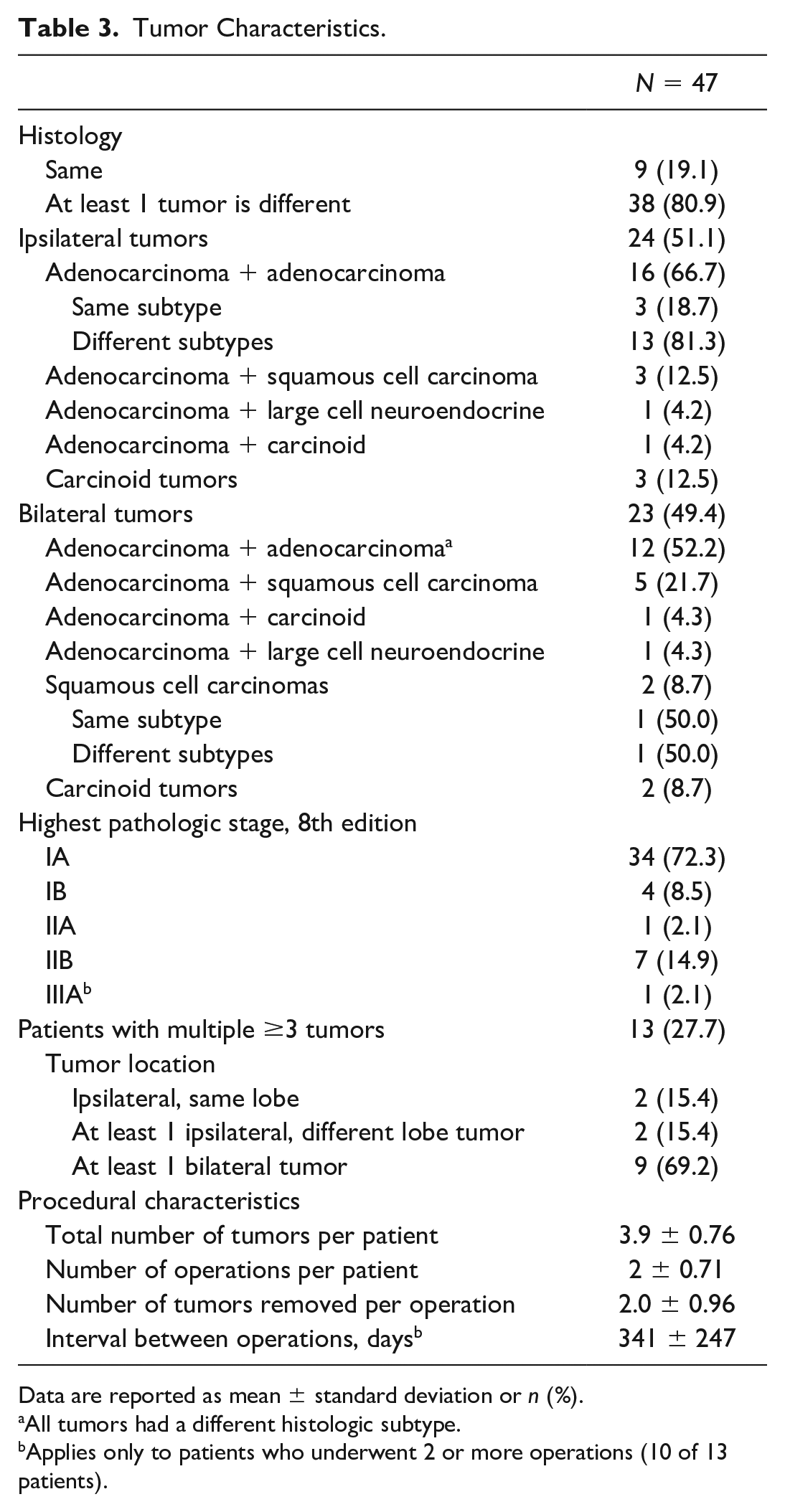
Data are reported as mean ± standard deviation or n (%).
All tumors had a different histologic subtype.
Applies only to patients who underwent 2 or more operations (10 of 13 patients).
There were 47 patients who underwent 105 surgical procedures to resect 119 tumors, which included 56 nonanatomic resections (53.3%) and 49 anatomic resections (46.7%). Of the anatomic resections, 30 (61.3%) underwent lobectomy, 18 (36.7%) underwent segmentectomy, and 1 patient (2.0%) underwent pneumonectomy. There were no surgical mortalities. Major postoperative complications, including hemothorax and prolonged air leak requiring decortication, were seen in 2 patients (4.3%). Operative details are shown in Table 4.
Operative Details.
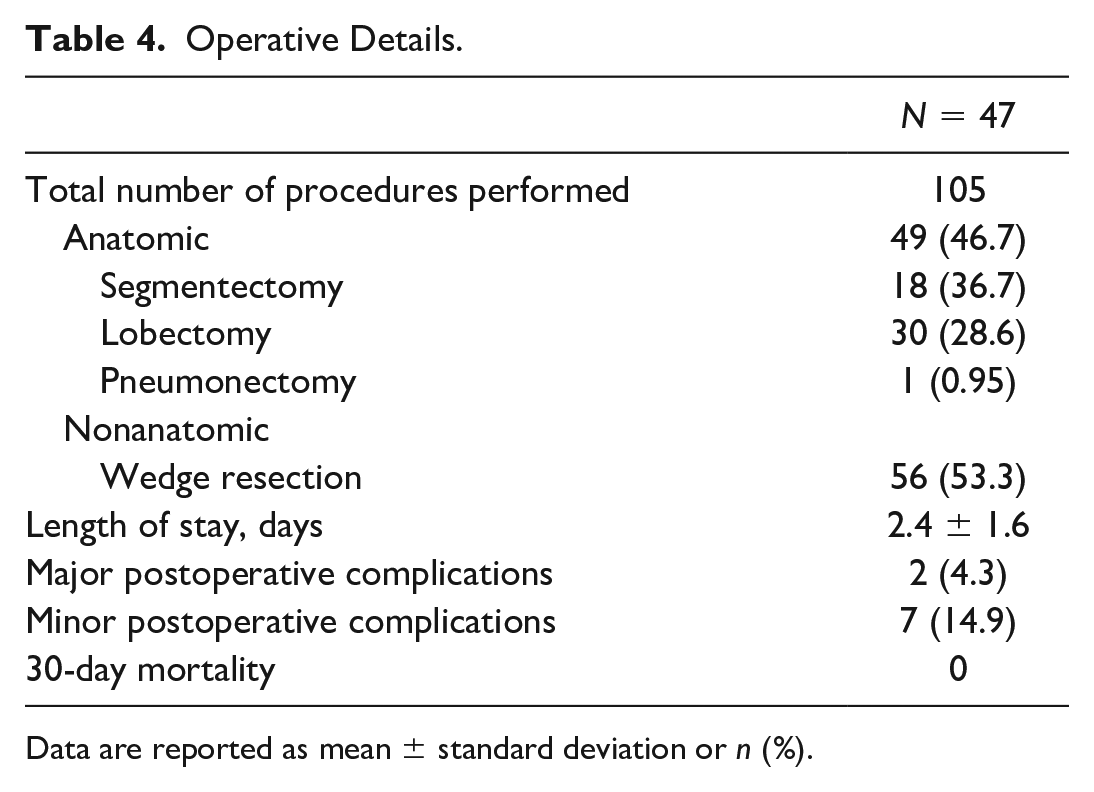
Data are reported as mean ± standard deviation or n (%).
Among the 47 patients diagnosed with SMPLCs, 3 patients (6.4%) did not undergo the planned surgical resection. One patient experienced hypertensive crisis during the scheduled operation for the second SMPLC, and as a result, the operation was aborted. The remaining 2 patients declined surgical treatment for their second primary tumor and instead opted for chemotherapy. Although only 1 of these patients was deceased at the time of the study, we excluded them from the survival analysis due to failure to undergo resection with intention to treat.
OS at 1, 2, 3, and 4 years was 98%, 96%, 88%, and 86%, respectively. CSS at 1, 2, 3, and 4 years was 100%, 98%, 93%, and 90%, respectively (Fig. 2). Patients with 3 or more SMPLC had significantly worse 4-year OS (57% vs 95% vs 100%, P = 0.002) and 4-year CSS (70% vs 95% vs 100%, P = 0.043) compared with those with 2 ipsilateral and 2 bilateral SMPLC, respectively. Among patients with 2 SMPLC, those with ipsilaterally located tumors had a 4-year CSS rate of 95%, whereas those with bilaterally located tumors had a 4-year CSS rate of 100%, which was not statistically significant. Univariable analysis of patient demographics, histology (same vs different), tumor location (ipsilateral, bilateral), and highest pathologic staging revealed no significant differences in survival. The results of univariable analysis on OS and CSS are shown in Table 5.
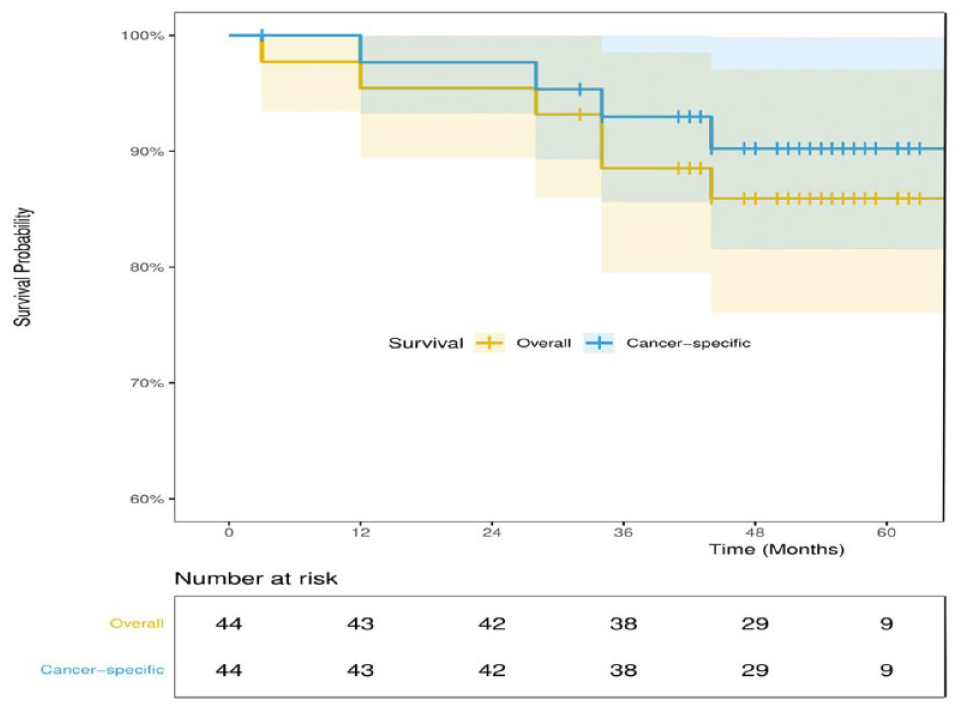
Kaplan–Meier survival curve depicting overall and cancer-specific survival. Shaded lines indicate 95% confidence intervals.
Kaplan–Meier Overall and Cancer-Specific Survival (N = 44).
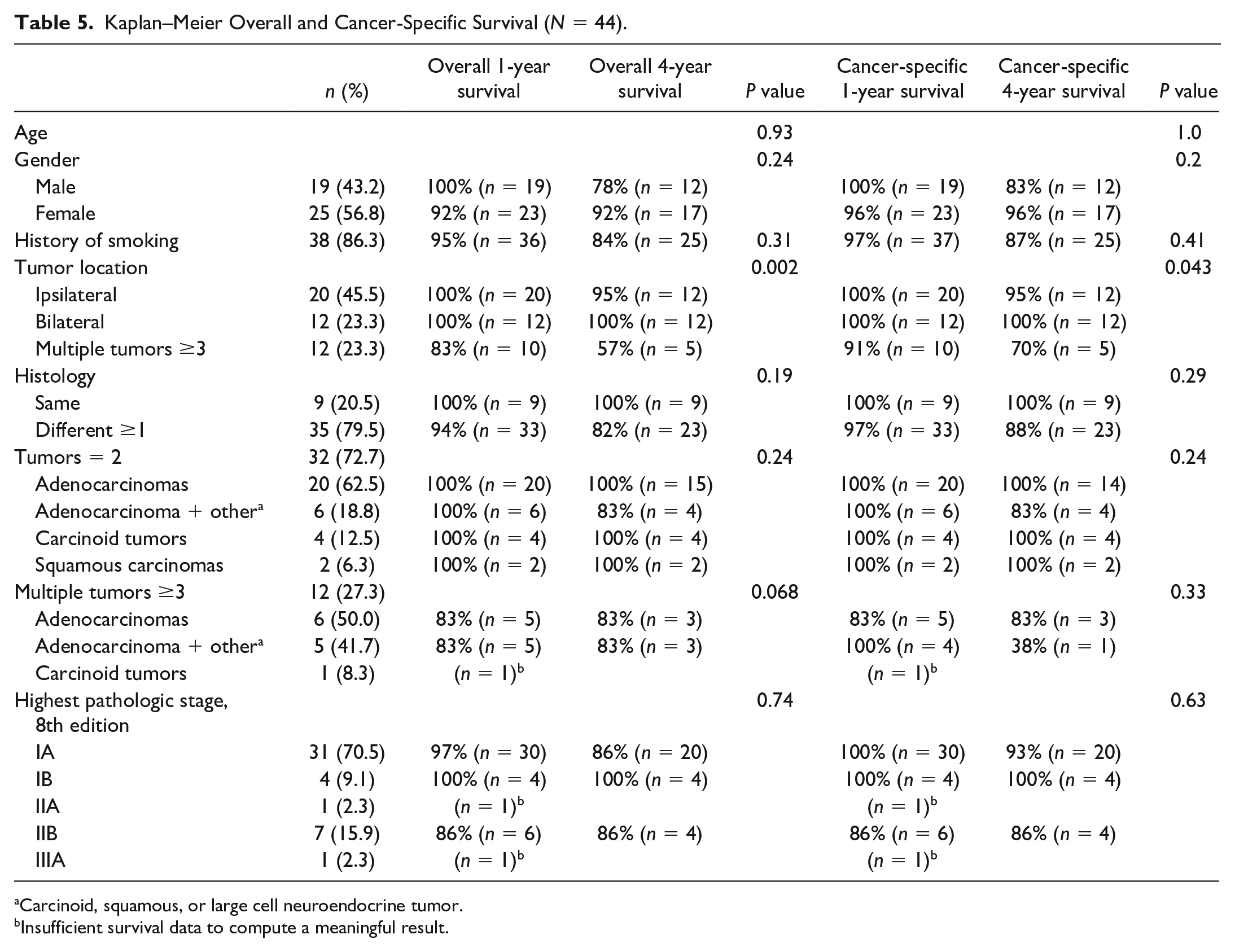
Carcinoid, squamous, or large cell neuroendocrine tumor.
Insufficient survival data to compute a meaningful result.
Discussion
Patients commonly present with multiple suspicious lung nodules. 8 While some of these nodules may represent metastatic lung cancer from a single original tumor and are appropriately staged as T3 (same lobe), T4 (ipsilateral different lobe), and M1a (contralateral lobe) disease, many others represent SMPLC. The historical incidence of SMPLC ranges from 0.8% to 8.4%,9,10 and there is evidence suggesting that its frequency is on the rise.1,2 According to the International Association for the Study of Lung Cancer (IASLC) database, the incidence of patients presenting with a separate tumor nodule has increased from 3.0% (1999 to 2005) to 6% (2007 to 2010), 11 reaching up to 15% of lung cancer patients in a large retrospective study by Girard et al. 8 Clinicians may face various clinical scenarios when managing patients with multiple lung nodules. While the presence of tumors with different histology can confirm the presence of SMPLC, having the same histology does not guarantee IPM. To address this, Martini and Melamed published guidelines in 1975 4 that are still mostly applicable today. 5 The largest modification allows for tumors in the same lobe (T3) to be defined as SMPLC based on histologic subtyping. 12
The challenge for clinicians is deciding which nodules to biopsy: one, both, or none. In patients with multiple suspicious lung nodules, biopsies can often be misleading or insufficient to establish a diagnosis, let alone differentiate between the subtypes of non-small cell lung cancers. Radiographic patterns can provide clues suggestive of SMPLC. For instance, the presence of a solid spiculated nodule with a second nonsolid ground-glass opacity (GGO) may raise suspicion for SMPLC. 13 However, the predominant pattern observed is a solid lung cancer tumor accompanied by a second solid tumor of similar histology. 14
The IASLC Lung Cancer Staging Project 14 has categorized 3 distinct patterns of patient presentation with multiple pulmonary tumors. Type A represents patients with a second primary lung cancer based on different histology, type B represents patients with a solid primary lung cancer and a satellite nodule of the same histology, and type C represents multiple lung cancer nodules with prominent ground glass or lepidic features. Types A and C clearly represent SMPLC patients, whereas type B can represent SMPLC or IPM. Attempts to further classify type B tumors of the same histology are mired in complexity. Even genetic comparisons based on mutation testing fail to clarify the issue as even metastatic tumors are frequently discordant. 3
In this study, 35 patients (74.5%) had tumors with similar histology. After subtyping, only 9 patients (19.1%) had histologic concordance. These 9 patients were included in the study based on either having tumors originating from separate foci within the same lobe or having tumors in a different lobe. Based on the modified Martini–Melamed criteria outlined in Table 1, tumors of the same histology in different lobes should be classified as SMPLC provided they do not share positive intervening lymph nodes. 4 These complex scenarios necessitate evaluation by a multidisciplinary team consisting of thoracic surgeons, radiologists, medical oncologists, and pathologists. In this study, all patients underwent thorough evaluation by our multidisciplinary tumor board to ensure adherence to the modified Martini–Melamed criteria. In previous publications, 15 we excluded patients with similar histology in the same lobe to avoid diluting survivorship. Nevertheless, the current literature permits the inclusion of cases that some might categorize as T3 or T4, which is critical for accurately determining the incidence of SMPLC in a contemporary patient population. In this series, 14 patients (29.8%) had tumors in the same lobe, which were deemed histologically different based on subtyping and/or multidisciplinary tumor board reconciliation. Ten patients (21.3%) with tumors in the ipsilateral different lobe were included based on the absence of involvement in shared lymph node basins.
The true incidence of SMPLC is likely still underestimated, as historical staging practices may have incorrectly overstaged patients as T3 or T4. In a large series of SMPLC patients, Finley et al. demonstrated the importance of histologic subtyping in distinguishing SMPLC from what would be conventionally labeled as T3. 12 In that study, 27 of 34 patients (79.4%) were reclassified as SMPLC, whereas only 7 (20.6%) were considered true T3 tumors. It stands to reason that prior to this modification and the subtyping capabilities of contemporary pathology departments, prior studies, including our own, 16 may have incorrectly staged tumors as IPM. Our study reveals a high incidence of SMPLC at 20.7%. It is important to emphasize that these patients represent a more contemporary population than those enrolled in the National Lung Cancer Screening Trial in 2011. 17 In our series, 40.4% of patients with SMPLC were identified through a lung cancer screening program. Notably, such screening programs tend to detect tumors at an earlier stage and reveal fewer cases of metastatic disease than is traditionally seen. Improved availability of high-resolution CT scans with finer cuts is another contributing factor in the identification of a greater number of small nodules. Prior to the introduction of lung screening programs and advanced imaging techniques, it is plausible that some patients with advanced metastatic disease may have harbored a second unrelated primary tumor that was masked by the presence of metastasis from the primary malignancy. Should this hypothesis hold true, it would result in a lower identification of SMPLC cases, thus falsely reducing the incidence rate even further.
While T4 and M1a cases are typically not treated with curative intent, SMPLC patients demonstrate favorable outcomes with surgical resection. In our study, the 4-year OS and CSS rates were notably high at 86% and 90%, respectively. Other studies have reported varying survival rates, ranging from 10% 18 to 76% 19 at 5 years.6,20,21 A meta-analysis of 42 studies by Tie et al. 21 reported OS rates of 87%, 72%, and 63% at 1, 2, and 3 years, respectively but did not provide data on CSS as we have done in Figure 2. In another meta-analysis, Nie et al. revealed notable advancements in 5-year OS rates for studies published after 2010 compared with earlier studies (55% vs 34%). 6 These improvements likely reflect advancements in the diagnosis and management of SMPLC patients, potentially attributed to increased awareness of SMPLC. There are 2 potential factors that may contribute to our study’s superior OS and CSS rates. The first factor is our decision to include carcinoids in our analysis, and the second is the presence of lead-time bias. Carcinoid tumors are classified as lung cancer by the World Health Organization, and as such, they were included in this study. The authors acknowledge the favorable outcomes associated with these tumors. When patients with carcinoid tumors were excluded from the analysis, the 4-year OS and CSS rates were 82% and 88%, respectively. Given our interest in SMPLC, it is likely that we identify and treat tumors at earlier stages. Based on this study’s inclusion criteria, patients were classified as having SMPLC if they had additional nodule(s) at the time of their initial tumor resection, which were later confirmed to be cancerous.
Among the patients included, 13 of 47 patients (27.7%) had 3 or more lung cancers. The average interval between their first and last surgery was 341 ± 247 days, suggesting that many of these patients had early lesions that were monitored until they progressed. These were predominantly nonsolid GGOs. Malignant GGOs with nonsolid components are typically associated with higher CSS rates. 22 This raises concerns about potential lead-time bias, which may have influenced our survivorship data. In 2011, we previously reported 3-year and 5-year CSS rates of 73% and 69%, respectively. 20 That study had notable differences. First, we excluded all potential T3 tumors, irrespective of histologic subtyping, and only included same lobe tumors of different histologic type. We also excluded tumors that were previously categorized as bronchoalveolar carcinoma. However, both groups were included in our present study, which likely contributed to our improved survival outcomes.
Prognostic factors for survival in patients with SMPLC remain largely unknown, partially attributed to the low reported incidence of SMPLC in lung cancer patients. In this study, only patients with 3 or more tumors had a statistically inferior CSS (P = 0.043). Patient demographics, laterality, histology, and pathologic staging did not significantly influence survival. Other meta-analyses also failed to find an association between laterality, histology, and survival. However, larger tumor size, advanced T stage, lymph node involvement, adjuvant chemotherapy, tumors composed of only adenocarcinomas, subtype other than lepidic dominant, symptomatic disease, and pneumonectomy have been associated with poor survival in patients with SMPLC.5,6,21
The optimal surgical approach for patients with SMPLC has been controversial, with some studies recommending lobectomy as the primary method of treatment while others prefer sublobar resection.6,23 In addition, there is no consensus regarding the choice between staged or simultaneous resection for SMPLC patients. In our study, approximately 47% of the procedures performed involved anatomic resections such as segmentectomy, lobectomy, and 1 pneumonectomy. When managing patients with SMPLC, it is crucial to consider parenchymal-sparing approaches, which are supported by our OS and CSS. This approach can be especially beneficial in younger patients who are at risk of developing metachronous second primaries. Preserving lung parenchyma may allow for additional treatment options if these patients develop additional tumors in the future.
In the present study, we are limited to reporting the incidence of SMPLC among patients who were referred to a thoracic surgery department and lack information regarding the number of patients who may have been misdiagnosed and referred elsewhere despite having SMPLC. Our findings indicate that a substantial number of patients were identified as having a second primary lung cancer by the thoracic surgery department, and many secondary nodules were overlooked by the referring physician and radiologists. The authors hope that this publication may provoke additional research to address these questions and improve physician awareness of patients with SMPLC.
Conclusions
Patients frequently present with multiple suspicious lung nodules, some of which represent SMPLC. Our data suggest that the incidence of SMPLC is increasing, likely due to advancements in imaging, surveillance, diagnostic criteria, screening, and epidemiologic patterns, particularly the rise of adenocarcinomas, along with heightened awareness among thoracic oncology teams. Thoracic surgeons, oncologists, and radiologists would benefit from a thorough knowledge of SMPLC. Accurate staging requires a comprehensive approach, encompassing meticulous radiologic imaging review, histologic assessment, and genomic analysis. 3 However, distinguishing between SMPLC and IPM remains challenging, even with histopathologic subtyping and application of genomic analyses, as the same histology does not always indicate IPM, and genomic analysis can be unreliable due to discordance between primary tumors and metastatic sites. 3 Implementing a standardized criteria, such as the modified Martini–Melamed criteria described in this study, can lead to favorable survival outcomes in patient with SMPLC. In addition, referring to these cases as “simultaneous multiple primary lung cancers” may be more intuitive when discussing these patients. By optimizing recognition of this disease, health care professionals can provide patients with stage-based treatment and improve survival outcomes.
Footnotes
* This study was presented at the ISMICS 2023 Annual Meeting in Boston, MA, USA on June 1, 2023.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
