Abstract
Background:
Chest computed tomography (CT) may provide evidence to forecast unexpected recurrence and metastasis following radical surgery for stage I synchronous multiple primary lung cancer (SMPLC).
Objective:
This study aims to develop and validate a novel CT-based multi-parametric decision tree algorithm (CT-DTA) model capable of accurate risk assessment.
Design:
A multicenter retrospective cohort study.
Methods:
There were 209 patients with pathological stage I SMPLC from three tertiary centers included. We initially screened all of the CT-derived imaging parameters in the training cohort (130 patients from Center A) and then selected those showing statistical significance to construct a DTA model. The discriminative strength of the CT-DTA model for postoperative recurrence and metastasis was then validated in the validation cohort (79 patients from Centers B and C). Moreover, the performance of the CT-DTA model was further evaluated across different subgroups of the entire cohort.
Results:
Five key imaging parameters measured on chest thin-section CT, including consolidation tumor ratio (CTR), long-axis diameter of the lesion, number of pure solid nodules, presence of spiculation and pleural indentation, constituted a CT-DTA model with nine leaf nodes, and CTR was the leading risk contributor of them. The CT-DTA model achieved a satisfactory predictive accuracy indicated by an area under the curve of more than 0.80 in both the training cohort and validation cohort. Meanwhile, this CT-DTA model was also exhaustively demonstrated to play as the only independent risk factor for postoperative recurrence and metastasis. Its promising predictive performance still remained stable across nearly all of the subgroups stratified by clinicopathological characteristics.
Conclusion:
This CT-DTA model could serve as a noninvasive, user-friendly, and practicable risk prediction tool to aid treatment decision-making in operable stage I SMPLC.
Keywords
Introduction
Rationale
Lung cancer is almost the most prevalent solid malignancy and occupies the world’s leading cause of cancer-related deaths. 1 With rapid advances in the multi-slice spiral computed tomography (CT) and other medical imaging techniques, multiple primary lung cancer (MPLC), which refers to primary lung cancer in which ⩾2 lesions occur simultaneously or successively in different locations of the lung within the same individual, has been increasingly diagnosed in the latest decades. 2 In addition, thoracic surgeons have paid growing attention not only to the benefits of radical surgery for synchronous MPLC (SMPLC) but also to potential risk factors resulting in treatment failure and unfavorable prognosis. Despite the current 5-year survival rate of pathological stage I non-small-cell lung cancer (NSCLC) can be stably over 90%, early or late recurrence and metastasis, with an incidence hovering around 10%, still emerges as the most principal reason for the failure of surgical treatment.3,4
An increasing number of SMPLC has been initially diagnosed at an early stage of the disease with widespread popularization of chest CT screening. 5 As the most common imaging test in daily practice, chest CT has also been widely demonstrated to offer a series of qualitative and quantitative features that are of clinical significance for risk prediction of poor outcomes in NSCLC. 6 However, to our knowledge, there was no investigation yet on the key contributing factors derived from chest CT to recurrence and metastasis that surprisingly occurred after surgical resection for stage I SMPLC. 7 Besides, with the aim to help inform personalized surveillance protocols in the patients with stage I SMPLC who are considered at high risk of unexpected recurrence and metastasis, a practicable and effective CT-based multi-parametric scoring system will be particularly valuable for accurate risk assessment preoperatively.
Objectives
Decision tree algorithm (DTA) serves as a non-parametric supervised learning method with the goal to create a model that predicts the value of a target endpoint by learning simple decision rules inferred from the data features. 5 It has been recently reported that a newly proposed grading system by integrating pathological characteristics through a DTA approach can provide compelling and user-friendly information to aid treatment decision-making for stage I invasive lung adenocarcinoma. 8 Given the above concerns, the purpose of this multicenter study was to develop a novel DTA model based on chest CT imaging features of pulmonary nodules for precise risk evaluation of recurrence and metastasis in patients with pathological stage I SMPLC following surgery.
Methods
Study design, study protocol, and settings
This multicenter retrospective cohort study was performed on the independent datasets of surgical patients with SMPLC prospectively collected from three Grade A tertiary hospitals in the People’s Republic of China between January 2012 and June 2020.
We received formal approval from the Institutional Review Board (Protocol No. B2022-293-01) and then accomplished all the processes in conformity with the Declaration of Helsinki. The requirement of informed consent was waived. Finally, we produced this report according to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 9
Participants
Recruitment and study groups
We initially reviewed the clinicopathological characteristics and CT imaging records of 315 consecutive patients who were pathologically diagnosed as SMPLCs following radical lung resections at three tertiary centers between January 2012 and June 2020. As shown in Figure 1, after excluding 106 of these patients from the modeling analysis, we finally determined to include the remaining 209 patients (65.3%) confirmed with pathological stage I SMPLC postoperatively (130 cases in Center A, 46 cases in Center B, and 33 cases in Center C).
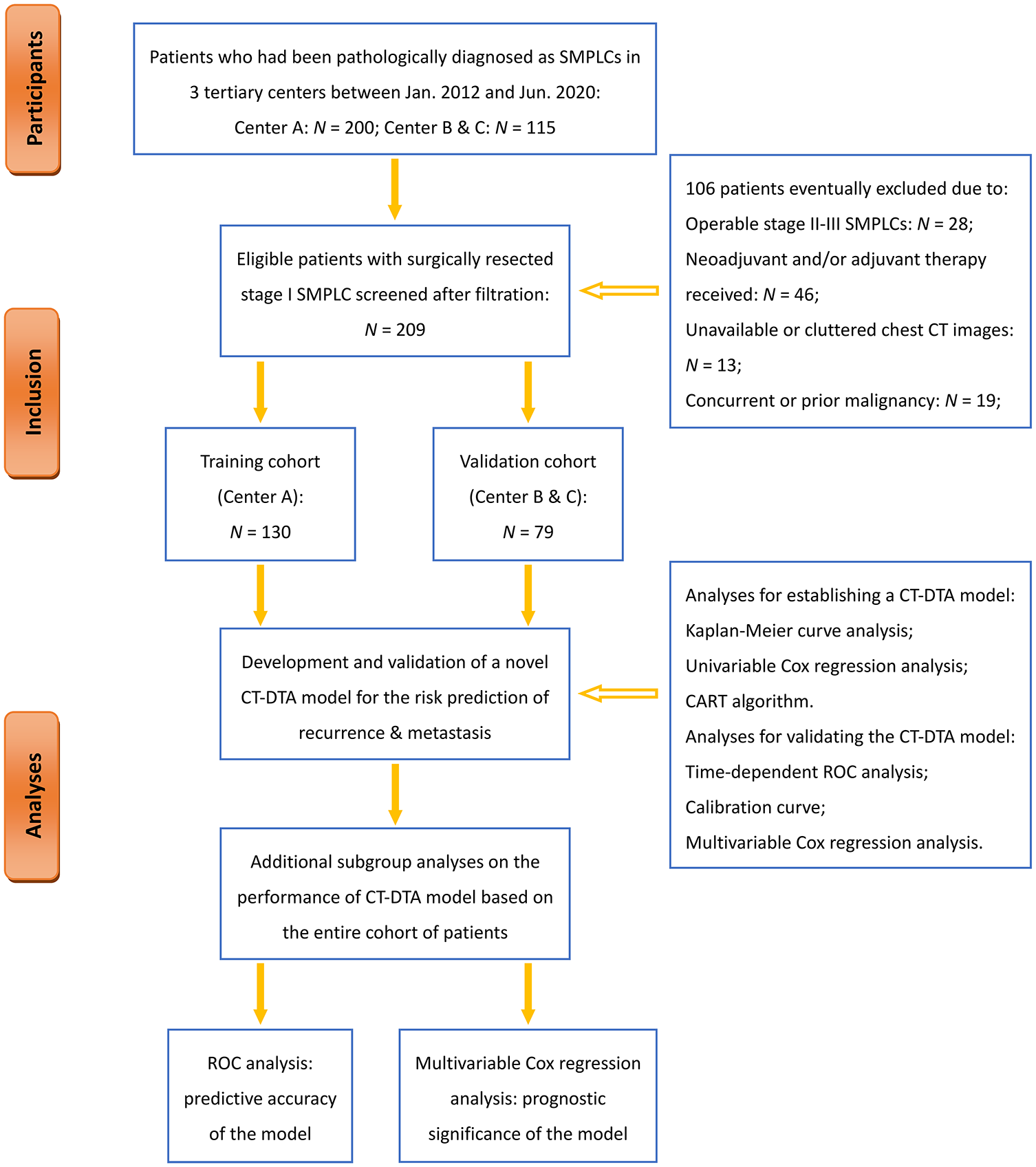
Flowchart of the study design and patient enrollment.
We divided the 130 patients from Center A into the training cohort and the remaining 79 patients from Center B and C into the validation cohort, respectively. A CT-based multi-parametric DTA model (CT-DTA) was built up based on the training cohort, and its predictive capacity for risk of recurrence and metastasis was further validated in the external validation cohort.
Inclusion and exclusion criteria
The following criteria were utilized to judge the candidate’s suitability for inclusion or exclusion:
(i) Patients who were postoperatively confirmed with pathological stage I SMPLC (T1N0M0 or T2aN0M0 for each nodule) in compliance with the 2013 American College of Chest Physicians criteria and the AJCC/UICC TNM classification system (8th Edition) were included.10,11 Patients with operable stage II-III SMPLCs and intrapulmonary metastasis would be excluded;
(ii) Curative-intent anatomical/wedge resections of the lung with systemic mediastinal lymph node dissection were eligible;
(iii) The clinicopathological and chest thin-section CT (TS-CT, slice thickness ⩽1 mm) imaging details must be obtained within 3 months prior to surgical procedure. To ensure the objectivity and accuracy of the data analyzed, the scarcity of complete medical records, chest TS-CT images, and follow-up evaluation would be excluded;
(iv) Patients who had received neoadjuvant/adjuvant anticancer therapy until any cancerous recurrence and/or metastasis detected would be excluded to minimize their potential confounding effects on postoperative prognosis;
(v) Patients with concurrent or prior malignancies would be excluded to avoid potential selection bias caused by metastasis originated from additional primary tumors out of the lungs.
Follow-up evaluation
A regular follow-up program began on postoperative day 1. A series of exhaustive assessment tools, including peripheral blood tests, multi-slice spiral CT scans on the head, neck, chest, and abdomen, and necessary biopsy of suspicious neoplasms, were performed every 3 months for the first 2 years, every 6 months for the next 3 years, and then annually until the terminal follow-up date of January 2022. Importantly, systemic surveillance for possible recurrence and metastasis was always prepared when patients had unscheduled visits due to any symptom or suspicious metastatic image.
Measurement and definitions of outcome data
Clinicopathological characteristics
(i) Baseline information estimated: age (years), gender (male/female), and smoking status (never/current/former);
(ii) Preoperative comorbidities estimated: respiratory comorbidities (chronic obstructive pulmonary disease, emphysema, tuberculosis, pneumonia, asthma, bronchiectasis, and interstitial lung diseases), cardiovascular comorbidities (hypertension, coronary artery disease, valvular heart disease, cerebrovascular disease, cardiac arrhythmias, chronic heart failure, and peripheral artery disease) and diabetes mellitus;
(iii) Pathological features concerning each lesion: grade of differentiation (well/moderate/poor), pleural invasion (present/absent), lymphovascular invasion (present/absent), and T stage (T1/T2a). All of the above results were estimated by our experienced pathologists according to the 2013 American College of Chest Physicians criteria and the AJCC/UICC TNM classification system (8th Edition).10,11
Chest TS-CT imaging parameters
Radiological assessment based on available chest TS-CT images for each included patient, with both lung window (width: 1500HU; level: −450HU) and mediastinal window (width: 350HU; level: 40HU) settings, was independently executed by two radiologists who have work experience exceeding 10 years in a blind manner. Besides, in case of disagreement between two primary radiologists, another radiologist with a 20-year of experience would then be invited to adjudicate a final decision.
The following TS-CT imaging features regarding each nodule of SMPLC were cautiously measured and recorded, including the type of nodule (pure solid nodule/ground glass nodule/solid-predominant nodule/ground glass opacity-predominant nodule), 12 long-axis diameter of the lesion, long-axis diameter of the solid portion, consolidation tumor ratio (CTR), spiculation (present/absent), lobulation (present/absent), pleural indentation (present/absent), bubble-like vacuole (present/absent), and air bronchogram (present/absent). Then, we combined the results associated with the same kind of TS-CT sign of all existing nodules as one covariable incorporated into the CT-DTA model (Table S1). Qualitative and quantitative evaluation criteria of all the above parameters have been detailed in Table S1 and described in our previously published studies.5,13
Outcomes of interest
The primary endpoint was the existence of cancerous recurrence and metastasis following surgery. Recurrence was defined as the intrapulmonary neoplasms that exhibited different histological subtypes and/or genetic mutations from the primary lesions. Metastasis was defined as the neoplasms outside of the lung that shared the same histological subtypes and/or genetic mutations as the primary lesions. The diagnosis of recurrence and metastasis would be judged by an online multidisciplinary meeting if necessary. In addition, our secondary goal was overall survival (OS), which was defined as the time interval between pulmonary resections and the date of death from any cause or censor at the end of follow-up.
Cutoffs of imaging parameters
The “Maxstat” package of R was employed to identify the optimal cutoff points for continuous variables of CTR, long-axis diameter of the maximal lesion, and long-axis diameter of solid portion in the maximal lesion respectively for the risk prediction of postoperative recurrence and metastasis in the training cohort of patients. 14 The threshold values of these imaging parameters were further determined to be the grouping criteria regarding each lesion of SMPLC.
Statistical analysis
Statistical differences between groups
We used Pearson’s chi-square test, Yates’ correction test, or Fisher exact test to compare categorical variables and Mann–Whitney U test to compare continuous data (mean ± standard deviation (SD); median and interquartile range (IQR)), respectively. In addition, a Kaplan–Meier curve analysis with a Log-rank test was applied to distinguish differences in the cumulative risk of recurrence and metastasis, as well as in the time interval and rate of OS between groups stratified by every evaluated TS-CT sign. Statistical significance was suggested by p < 0.050 in a two-sided test.
Prognostic factor analysis
The correlations between all of the evaluated characteristics and time-to-event results of endpoints were initially investigated through univariable Cox proportional hazards regression analysis. Thereafter, all the clinicopathological and imaging covariables with p < 0.15 would be included in multivariable Cox regression models, and then, hazard ratios (HRs) with 95% confidence intervals (CIs) were generated to determine which factors could play significantly predictive roles for the risk of unfavorable outcomes. Ultimately, we computed Harrell’s concordance statistic (C-statistic) to measure the goodness-of-fit of each multivariable Cox regression model.
All the above statistical methods were accomplished using the IBM SPSS 27.0 software (IBM SPSS Statistics, Version 22.0., IBM Corp, Armonk, NY, USA).
Establishment and validation of DTA model
On the basis of the training cohort, a DTA model was comprised of both qualitative and quantitative parameters of chest TS-CT signs, all of which were found to show statistical significance in the Kaplan–Meier curves and the Cox regression analyses. The eligible imaging parameters characterized as categorical or optimal cutoff values were input to establish a DTA model whose tree-growing methodology was set as follows: classification and regression tree algorithm; “Gini” impurity criterion; the minimum number of samples required to split an internal node as 5; the minimum number of samples required to locate at a leaf node as 2; the maximum depth of the tree as 30. This decision-making process would be terminated when there was no substantial contribution in the next cycle of splitting.5,8
The predictive performance of this CT-DTA model with respect to the risk of recurrence and metastasis was further externally validated based on the validation cohort. We conducted the time-dependent receiver operating characteristic (t-ROC) analysis, a tailor-made ROC analysis for time-to-event variables, to estimate the capacity of our CT-DTA model and main clinicopathological characteristics to discriminate between the incidences of recurrence and metastasis throughout the follow-up period. In addition, their areas under curve (AUCs) would be inferred and further compared using Spiegelhalter’s Z-test. A risk prediction model with AUC > 0.80 would be considered to be clinically useful. 8 Finally, we plotted calibration curves with bootstrap repetitions to reveal the consistency between the predicted probability and the real-world probability of postoperative recurrence and metastasis.
All the above statistical methods were accomplished using the JMP Pro 16.0 software (SAS Institute, Cary, NC, USA) and R Studio 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria).
Subgroup analyses
In addition, the efficiency of the CT-DTA model was also assessed across all of the subgroups stratified by clinicopathological characteristics. An ROC analysis based on each subgroup of the entire cohort was employed to measure the predictive accuracy of the CT-DTA model for the emergence of postoperative recurrence and metastasis. The prognostic independence of the CT-DTA model in each subgroup of the entire cohort was further confirmed by a multivariable Cox regression analysis.
Results
Demographics and outcomes
Clinicopathological and imaging characteristics
The demographic data were tabulated in Table 1. Females and nonsmokers represented 61.2% and 78.5% of the 209 patients receiving surgical resections for stage I SMPLC in three tertiary centers, respectively. The mean age of all the enrolled patients was 62.0 ± 10.5 years, and meanwhile, 59 (28.2%) of the patients were suffering from health burdens due to at least one kind of comorbidity preoperatively. The majority of SMPLC contained two nodules (85.2%) and developed within the ipsilateral lobes (65.6%), thus, lobectomy with additional wedge resection was chosen to be operated on 54.1% of the entire cohort of patients. Adenocarcinoma was the most predominant histological subtype of all the lesions in SMPLC, and there were 153 (73.2%) of the patients diagnosed with T1N0M0 SMPLCs. Pleural and lymphovascular invasion was postoperatively confirmed in the specimens of 39 (18.7%) cases and 11 (5.3%) cases, respectively. Notably, there was no significant difference in any clinicopathological characteristic between the training cohort and validation cohort, as detailed in Table 1.
Estimated characteristics between training cohort and validation cohort.

AC, adenocarcinoma; GGN, ground glass nodule; GGO, ground glass opacity; IQR, interquartile range; SCC, squamous cell carcinoma; SD, standard deviation; TS-CT, thin-section computed tomography.
The details of imaging parameters are listed in Table 1. On the one hand, the majority of the lesions in SMPLC were found to be without any CT-based sign of spiculation (67.0%), bubble-like vacuole (66.0%), and abnormal air bronchogram (79.9%). On the other hand, CT-based signs about pleural indentation (69.9%) and lobulation (90.9%) were both more frequently present in one or more lesions of SMPLC. The mean long-axis diameters of the maximal lesion and solid portion in the maximal lesion were 22.8 ± 12.6 mm and 15.9 ± 14.9 mm, respectively. We found no significant difference in either quantitative or qualitative features obtained by chest TS-CT between the training cohort and validation cohort (Table 1).
Surgical outcomes after follow-up
The median follow-up time of multicenter cohorts was 30 months. As shown in Table 1, the overall incidence of recurrence and metastasis until the final follow-up date was 12.9%, including 9 cases developed locally intrapulmonary recurrence and 19 cases occurred distant metastasis (9 pulmonary metastasis, 7 brain metastasis, 6 bone metastasis, and 2 pleural metastasis). Besides, 12 of the patients died from early or late recurrence/metastasis during the follow-up, with an OS rate of 94.3%. Finally, no significant difference was observed in the incidence of recurrence, metastasis, or death between the training cohort and the validation cohort (Table 1).
Derivation of CT-DTA model
Prediction of recurrence and metastasis by TS-CT imaging parameters
With respect to the occurrence of postoperative recurrence and metastasis, the optimal cutoff points of CTR and long-axis diameters of the maximal lesion with its solid portion suggested by the Maxstat analysis were 0.80, 32and 28 mm, respectively. As exhibited in Table S2 and Figure 2, the potential effects of each clinicopathological and imaging parameter on the risk of recurrence and metastasis in the training cohort were initially assessed by both Kaplan–Meier curves and univariable Cox regression analysis. We found that the number of pure solid nodules (PSNs; Log-rank p < 0.001; Cox p = 0.001), CTR (Log-rank p < 0.001; Cox p < 0.001), presence of spiculation (Log-rank p = 0.002; Cox p = 0.004) and pleural indentation (Log-rank p = 0.027; Cox p = 0.030), and long-axis diameter of the lesion (Log-rank p < 0.001; Cox p = 0.002) and the solid portions (Log-rank p = 0.001; Cox p = 0.003) were significantly associated with an increased risk of recurrence and metastasis. There was no significant relationship of the other imaging parameters with postoperative recurrence and metastasis (Table S2 and Figure 2(g)–(i)).

Kaplan–Meier analyses on the risk of recurrence and metastasis between patients of the training cohort stratified by the following imaging parameters obtained on chest thin-section computed tomography: (a) long-axis diameter of the lesion, (b) long-axis diameter of the solid portion, (c) consolidation tumor ratio, (d) number of pure solid nodules, and the presence of (e) spiculation, (f) pleural indentation, (g) lobulation, (h) air bronchogram, and (i) bubble-like vacuole.
After adjustment by all the covariable estimates holding p < 0.15, a multivariable Cox regression analysis demonstrated that none of the above six imaging parameters and other clinicopathological features could be independently predictive of postoperative recurrence and metastasis in the training cohort of patients, as detailed in Table 2 (Model A).
Multivariable analysis on the independent risk factors for postoperative recurrence and metastasis in patients of the training cohort.

This multivariable Cox regression model was established on clinicopathological and imaging parameters with p < 0.15 in the univariable analysis for risk of recurrence and metastasis in the training cohort (−2 Log likelihood value: 126.34; Omnibus test p < 0.001).
This multivariable Cox regression model was established on the DTA model and other clinicopathological characteristics with p < 0.15 in the univariable analysis for risk of recurrence and metastasis in the training cohort (−2 Log likelihood value: 118.69; Omnibus test p < 0.001).
AC, adenocarcinoma; CI, confidence interval; CTR, consolidation tumor ratio; DTA, decision tree algorithm; HR, hazards ratio; SCC, squamous cell carcinoma.
Construction of CT-DTA model
We incorporated all the categorical data of the above imaging parameters showing statistical significance in both Kaplan–Meier curves and univariable Cox regression analyses to train a DTA model. As illustrated in Figure 3, a DTA model consisting of CTR (⩾1 lesion > 0.80; all lesions ⩽0.80), long-axis diameter of the lesion (⩾1 lesion > 32 mm; all lesions ⩽32 mm), number of PSNs (0, 1, ⩾2), presence of spiculation (⩾1 lesion present; absent) and pleural indentation (⩾1 lesion present; absent), which had been named as the CT-DTA model, was finally generated from the training cohort of patients.

A CT-based multi-parametric decision tree algorithm model by combining consolidation tumor ratio, long-axis diameter of the lesion, number of pure solid nodules, presence of spiculation, and pleural indentation together according to classification and regression tree algorithm.
This CT-DTA model contains nine leaf nodes with a predicted probability of postoperative recurrence and metastasis ranging from 0.5% to 74.1%. In addition, the importance of CT-based covariables contributing to the risk of recurrence and metastasis as estimated by the Bayesian network was visualized in Figure S1. We found that CTR occupied the most predominant risk factor accounting for 46.5% of the occurrence of postoperative recurrence and metastasis, followed by the presence of spiculation (22.0%), long-axis diameter of the lesion (16.1%), PSNs (10.6%), and the presence of pleural indentation (4.8%).
Validation of CT-DTA model
Predictive performance of CT-DTA model
Figure 4 shows the dynamic AUC of the CT-DTA model and main clinicopathological features from t-ROC analysis for the risk prediction of recurrence and metastasis in both the training cohort and validation cohort.

Time-dependent ROC analyses on the predictive accuracy of the CT-DTA model indicated by the dynamic values of area under the curve for the risk of cancerous recurrence and metastasis during the follow-up in both (a) training cohort and (b) validation cohort.
This CT-DTA model was found to have an excellent predictive accuracy to distinguish the patients developed with recurrent and/or metastatic tumors postoperatively in the training cohort, with clinically meaningful AUC values of 0.836 (95% CI: 0.676–0.996; p = 0.011), 0.864 (95% CI: 0.773–0.956; p < 0.001) and 0.862 (95% CI: 0.782–0.941; p < 0.001) at 1-year, 3-year and 5-year follow-up, respectively (Figure 4(a)). The discriminative power of our CT-DTA model was further externally validated among patients of the validation cohort. We found that this CT-DTA model still played substantially predictive roles for the risk of recurrence and metastasis, as revealed by AUC values got at 1 year (0.846; p = 0.042), 3 years (0.814; p = 0.011) and 5 years (0.835; p = 0.002) following surgery, respectively (Figure 4(b)). There was no significant distinction in the predictive accuracy of the CT-DTA model between the training cohort and validation cohort (Z-test p = 0.76).
Moreover, the dynamic AUC of the CT-DTA model throughout the whole course of follow-up was found to be always higher than those of T stage of the lesion, histology, differentiation grade, pleural and lymphovascular invasion in both the training cohort and validation cohort, showing a better discriminatory ability when compared to conventional pathological characteristics (Figure 4).
By plotting the calibration curves with bootstrap repetitions in both the training cohort and validation cohort, we also confirmed a significant agreement between the incidences predicted by the CT-DTA model and the real-world incidences of recurrence and metastasis during different follow-up periods, as shown in Figure 5.

Calibration curves revealed a good consistency between the incidence predicted by CT-DTA model and the real-world incidence of recurrence and metastasis during the follow-up in both training cohort and validation cohort.
Prognostic significance of CT-DTA model
In the training cohort, we run a multivariable Cox regression analysis again by replacing the raw data of CT-derived features with our CT-DTA model (Model B in Table 2). Finally, we verified that the CT-DTA model was considered the only independent predictive factor for postoperative recurrence and metastasis (HR: 2.04; 95% CI: 1.37–3.03; P < 0.001). Such evident predictive value of the CT-DTA model was further externally validated. As detailed in Table 3 (Model A), we found that neither clinicopathological nor imaging characteristics involved in a multivariable Cox regression model could be independently associated with the development of recurrence and metastasis in patients of the validation cohort. Accordingly, after incorporating our CT-DTA model, a new multivariable Cox regression analysis (Model B in Table 3) based on the validation cohort of patients demonstrated that only the CT-DTA model could independently predict the risk of postoperative recurrence and metastasis (HR: 2.0; 95% CI: 1.09–3.68; p = 0.026).
Univariable and multivariable analyses on the independent risk factors for postoperative recurrence and metastasis in patients of the validation cohort.

This multivariable Cox regression model was established on clinicopathological and imaging parameters with p < 0.15 in the univariable analysis for risk of recurrence and metastasis in the validation cohort (−2 Log likelihood value: 42.54; Omnibus test p = 0.002).
This multivariable Cox regression model was established on the DTA model and other clinicopathological characteristics with p < 0.15 in the univariable analysis for risk of recurrence and metastasis in the validation cohort (−2 Log likelihood value: 44.32; Omnibus test p < 0.001).
AC, adenocarcinoma; CI, confidence interval; CTR, consolidation tumor ratio; DTA, decision tree algorithm; HR, hazards ratio; SCC, squamous cell carcinoma.
In addition, we further carried out univariable and multivariable Cox regression analyses to determine the potential prognostic significance of the CT-DTA model for postoperative OS. In the training cohort, we recognized potential candidate variables with univariable p < 0.15, including the CT-DTA model, long-axis diameter of solid portion in the lesion, gender, smoking status, cardio-cerebrovascular comorbidity, location of lesions and histology, and then integrated them into a multivariable Cox regression model. The CT-DTA model (HR: 5.05; 95% CI: 1.31–19.48; p = 0.019) was eventually found to be the only independent prognostic indicator for postoperative OS of patients in the training cohort. Moreover, as for external validation, a multivariable Cox regression analysis based on the CT-DTA model and other clinicopathological characteristics with univariable p < 0.15 was conducted in the validation cohort. Finally, none of them was validated to be predictive of postoperative OS, although the CT-DTA model (HR: 3.69; 95% CI: 0.92–14.74; p = 0.065) almost reached statistical significance to perform prognostic independence for unfavorable OS.
Risk stratification according to CT-DTA model
With the aim to help therapeutic decision-making based on accurate risk stratification, we tried to classify the two independent cohorts into low-risk (predictive probability 0.45%–4.05%), moderate-risk (predictive probability 9.63%–21.49%) and high-risk (predictive probability 24.31%–74.11%) populations according to every three consecutive leaf nodes of the CT-DTA model. Given such criteria, there were 62 (47.7%) low-risk, 44 (33.8%) moderate-risk, and 24 (18.5%) high-risk patients in the training cohort, and 41 (51.9%) low-risk, 26 (32.9%) moderate-risk, and 12 (15.2%) high-risk patients in the validation cohort, respectively.
In the training cohort, Kaplan–Meier analysis showed that the time interval until the date of cancerous recurrence and metastasis was significantly shortened in a step-wise fashion with an increasing risk stratified by the CT-DTA model (Log-rank p < 0.001; Figure 6(a)). Besides, a significant difference was also observed in the incidence of recurrence and metastasis across the low-risk (1.6%), moderate-risk (15.9%), and high-risk (45.8%) groups (Figure 6(a)). Subsequently, a multivariable Cox regression analysis determined that the CT-DTA model (HR: 4.51; 95% CI: 1.54–13.18; p = 0.006), when analyzed as a three-category grading system, could be regarded as a significantly effective risk prediction tool for postoperative recurrence and metastasis (Figure 6(b)).
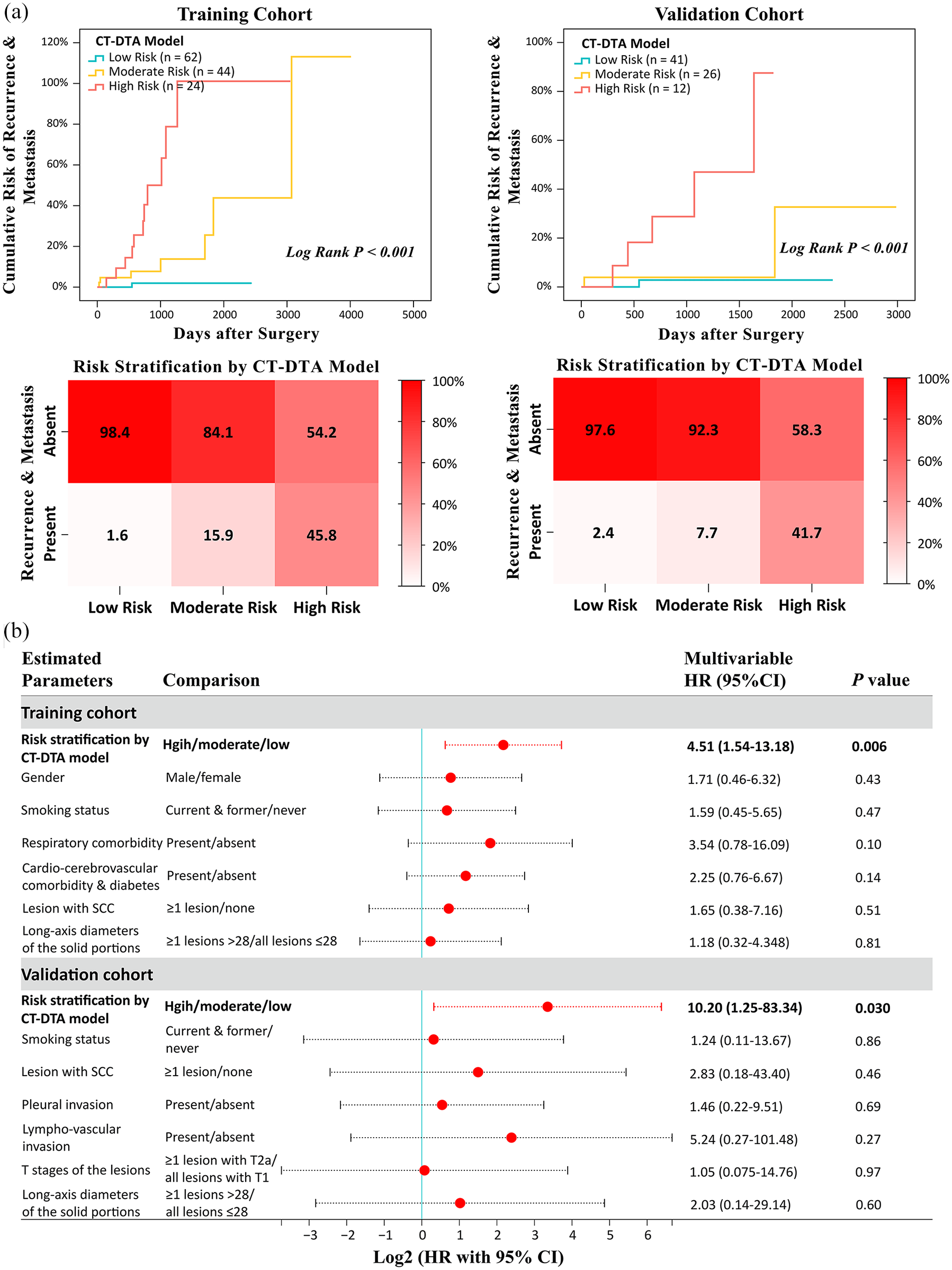
Risk stratification according to the CT-DTA model. (a) Kaplan–Meier analyses assessing potential differences in the risk of recurrence and metastasis between the low-risk, moderate-risk, and high-risk patients stratified by CT-DTA model in both training cohort and validation cohort. (b) Multivariable Cox regression analyses demonstrated an independent prognostic role of CT-DTA model when analyzed as a three-category assessment system for the risk of recurrence and metastasis in both training cohort and validation cohort.
Effects of the CT-DTA model on risk stratification were further externally validated. In the validation cohort, Kaplan–Meier analysis suggested that the time interval of recurrent and/or metastatic tumors that occurred postoperatively in high-risk patients was significantly shortened than that in low-risk and moderate-risk patients (Log-rank p < 0.001; Figure 6(a)). Furthermore, the high-risk (41.7%) patients had a significantly elevated incidence of postoperative recurrence and metastasis when compared to the low-risk (2.4%) and moderate-risk (7.7%) patients. Finally, when estimating the CT-DTA model in terms of a three-category grading system, its potent prognostic value for the risk of recurrence and metastasis was still stable in the validation cohort as demonstrated by a multivariable Cox regression analysis (HR: 10.20; 95% CI: 1.25–83.34; p = 0.030; Figure 6(b)).
Subgroup analyses on the entire cohort
As AUC values generated from subgroup ROC analyses indicated, the predictive accuracy of the CT-DTA model for the risk of recurrence and metastasis remained significantly reliable across all the subgroups stratified by age, gender, smoking status, preoperative comorbidity, number of lesions, location of lesions, surgical procedure, histology, grade of differentiation, pleural invasion, and T-stages of the lesions (Figure 7). Besides, the CT-DTA model also held a powerful distinguishing ability for postoperative recurrence and metastasis among patients without any lymphovascular invasion by tumor cells.

Subgroup ROC analyses regarding the predictive accuracy of CT-DTA model for postoperative recurrence and metastasis based on the entire cohort of patients with stage I SMPLC.
Another forest plot depicting HR statistics of the CT-DTA model from subgroup multivariable Cox regression analyses is shown in Figure 8. After controlling confounding effects from other clinicopathological covariables, we found that the prognostic significance of the CT-DTA model as an independent risk factor for postoperative recurrence and metastasis continued to stay robust across all the subgroups stratified by age, gender, smoking status, number of lesions, surgical procedure, histology, grade of differentiation, pleural invasion, lymphovascular invasion and T-stages of the lesions. Furthermore, we could also independently predict the risk of recurrence and metastasis by applying the CT-DTA model among patients without any preoperative comorbidity and with lesions distributed on the ipsilateral side of the lung (Figure 8).

Subgroup multivariable Cox regression analyses regarding the prognostic significance of CT-DTA model for postoperative recurrence and metastasis based on the entire cohort of patients with stage I SMPLC.
Discussion
Key results and interpretations
To the best of our knowledge, this is the first time to employ a DTA modeling technique in a multi-parametric risk assessment based on the relevant imaging features measured on chest TS-CT, particularly for surgically resected pathological stage I SMPLC. In this multicenter study, we established a novel and noninvasive CT-DTA model by efficiently integrating five key determinants from chest TS-CT preoperatively, including CTR, long-axis diameter of the lesion, number of PSNs, presence of spiculation and pleural indentation, to precisely predict the incidence of cancerous recurrence and metastasis following surgery in patients with pathological stage I SMPLC, who were traditionally considered as the low-risk population. After external validation, the excellent performance of the CT-DTA model to discriminate between the patients with different risks of recurrent and/or metastatic tumors surprisingly developed following surgery and might have the potential to warn thoracic surgeons of poor prognosis in advance.
One of our focuses was to insert a novel DTA model into conventional radiological evaluation in the current clinical practice of surgically resected stage I SMPLC. For the first time, the present study offered multivariable analytical results showing possible CT-derived features related to the risk of recurrence and metastasis in surgical patients with pathological stage I SMPLC, but none of them was observed with any predictive independence. Given such concerns, we developed a DTA model by incorporating five critical imaging parameters with statistical significance in the univariable analyses and finally validated it as a clinically useful risk assessment tool by t-ROC analysis, calibration curve, and multivariable Cox regression analysis in discerning which patients could easily suffer from treatment failure caused by recurrent and/or metastatic tumors following surgery. The nine leaf nodes generated in the CT-DTA model were from binary splits of five pivotal CT-based imaging features of pulmonary nodules, including CTR, long-axis diameter of the lesion, the number of PSNs, the presence of spiculation, and pleural indentation. The following possible evidence from the most recent investigations might help to elucidate its predictive strength.
Firstly, it had been well demonstrated by a meta-analysis that the percentage of solid components in a nodule of more than 80% on chest CT was an independent risk factor related to worse OS of NSCLC. 15 CTR > 0.80, especially appearing in all the existing nodules, had also been proven as the leading contributor in our CT-DTA model to the occurrence of recurrence and metastasis. Moreover, as previously reported, we had clarified the substantially prognostic roles of long-axis diameter of the lesion and PSNs for spread through air spaces and poor OS in single or multiple lung adenocarcinomas.13,16 Choi et al. 17 also emphasized the importance of lymph node evaluation in pure solid NSCLC with ⩾2 cm in its long-axis diameter since such a PSN almost always had a strong linkage to invasive features in pathology. Second, a margin spiculation sign apparent on chest CT could be indicative of fibrotic stroma or desmoplasia, which was characterized by tumor microenvironmental remodeling due to desmoplastic reaction, proliferation of fibroblasts, and dense deposition of extracellular matrix, all of which participated in modulating normal stroma into tumor stroma and then enhancing the growth and viability of cancer cells. 18 During the process of oncogenesis, cancer-associated fibroblasts produced a variety of tumor growth factors, cytokines, chemokines, and immune modulators and played an essential role in tissue fibrosis and desmoplasia. 18 Therefore, the presence of spiculation was generally associated with distant metastasis from primary NSCLC, even though at an early stage when the disease was newly diagnosed. 19 Third, pleural indentation revealed by chest CT was usually predictive of visceral pleural invasion, which had been widely accepted as a T2 descriptor in the pathological staging of NSCLC, although the CT definitions of pleural indentation still remained controversial. 20 As one key imaging parameter in our CT-DTA model, the presence of pleural indentation contributed much less to the risk of recurrence and metastasis.
Another highlight of our multicenter study was to perform both ROC analysis and multivariable Cox regression analysis on the clinical significance of the CT-DTA model in each specific set of patient subgroups. Finally, our CT-DTA model was found to be well validated for predicting the risk of recurrence and metastasis across nearly all the subgroups classified according to clinicopathological characteristics, especially in those subgroups of patients who were traditionally regarded as the low-risk populations, such as the non-elderly, nonsmokers, and the patients with well-differentiated and T1N0M0 lesions, and without any comorbidity, pleural or lymphovascular invasion. As regards the subgroups that failed to generate significant AUC or HR statistics, we speculated that the restricted sample size had the potential to attenuate analytical performance when employing risk evaluations.
Generalizability
Our findings in the present multicenter study provide solid evidence to support the involvement of a noninvasive, easy-to-use, and well-validated CT-DTA model in accurate risk stratification prior to curative-intent resections for stage I SMPLC to distinguish the patients who have a higher risk of postoperative recurrence and metastasis. We judiciously advocated that clinical contributions by applying this CT-DTA model might extend toward three key dimensions: (i) optimizing the pathways to monitor stage I SMPLC patients according to their recurrence/metastasis risks; (ii) allocating the patients to non-surgical procedures (such as stereotactic body radiation therapy) if they were considered at substantial operative mortality but at low recurrence/metastasis risk; (iii) tailoring the clinical trials to address the need of adjuvant treatments following surgery for the high-risk patients with stage I SMPLC. Therefore, this CT-DTA model has been proposed to aid thoracic surgeons in more accurate risk evaluation during the decision-making process and then inform personalized surveillance protocols to limit potential adverse events.
Limitations
Despite the above insightful findings, the following several limitations in this study should be sufficiently acknowledged. First of all, it was designed as a multicenter retrospective cohort study based on three prospectively maintained datasets with external validation and internal subgroup analyses. Due to the intrinsic limitations of its retrospective nature, potential selection bias, such as variations in clinical pathways and treatment options across research centers, might have still weakened the demonstrative power of the CT-DTA model as a reliable risk prediction tool. Second, due to the SMPLC itself belonging to a rare subtype of NSCLC, the relatively small cohort size, while enrolled from three high-volume tertiary centers, might have brought negative effects on statistical strength. Thus, a prospective study covering much more research centers is urgently warranted, and that is also a future project led by our research team on the basis of the present study. Third, this tailor-made CT-DTA model could not be enforceable in more common single-nodule stage I NSCLCs. Fourth, all of the enrolled patients were exclusively Chinese, which might restrict the generalizability of our CT-DTA model due to potential ethnic differences in genetic mutations (like EGFR, KRAS, etc.). Finally, qualitative evaluation of radiological features sometimes depends on the expertise of radiologists, and thus, such a subjective factor might result in confounding influence.
Conclusion
In conclusion, this study proposed a novel user-friendly and noninvasive DTA model based on multiple radiological features easily obtained on chest TS-CT for accurate risk prediction of recurrence and metastasis following surgical resections for pathological stage I SMPLC. The CT-DTA model can serve as a practically useful tool to improve the predictive performance of traditional risk assessment and aid thoracic surgeons in the decision-making of treatment options for potential high-risk patients with stage I SMPLC. Larger-scale multicenter prospective studies are warranted to further validate the CT-DTA model for clinical utility.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251325443 – Supplemental material for A novel decision tree algorithm model based on chest CT parameters to predict the risk of recurrence and metastasis in surgically resected stage I synchronous multiple primary lung cancer
Supplemental material, sj-docx-1-tar-10.1177_17534666251325443 for A novel decision tree algorithm model based on chest CT parameters to predict the risk of recurrence and metastasis in surgically resected stage I synchronous multiple primary lung cancer by Shuangjiang Li, Guona Chen, Wenbiao Zhang, Huiyun Ma, Baocong Liu, Li Xu and Qiong Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251325443 – Supplemental material for A novel decision tree algorithm model based on chest CT parameters to predict the risk of recurrence and metastasis in surgically resected stage I synchronous multiple primary lung cancer
Supplemental material, sj-docx-2-tar-10.1177_17534666251325443 for A novel decision tree algorithm model based on chest CT parameters to predict the risk of recurrence and metastasis in surgically resected stage I synchronous multiple primary lung cancer by Shuangjiang Li, Guona Chen, Wenbiao Zhang, Huiyun Ma, Baocong Liu, Li Xu and Qiong Li in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We give special thanks to Dr. Ying Zhu, from the Department of Radiology, The First Affiliated Hospital of Sun Yat-sen University, and Dr. Wenjing Gou, from the Department of Radiology, Sichuan Guangdong Provincial People’s Hospital, for their great assistance in radiological evaluation in this study. We give special thanks to Mrs. Hong Xie and Mrs. Peng Wang, from the Department of Medical English, West China School of Medicine, Sichuan University, for their English language editing of this manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
