Abstract
Background
Spatial neglect (SN) is a common visual attention deficit affecting stroke patients due to large-scale disruptions within brain networks. Most studies have focused only on resting-state, but effective rehabilitation requires a clearer understanding of how brain networks change during visuospatial tasks.
Objective
This study aims to identify network disruptions associated with neglect by comparing resting-state and task-based electroencephalography (EEG) connectivity patterns in stroke patients with and without neglect.
Methods
We recorded EEG data from 28 stroke patients using the augmented reality (AR)-based EEG-guided neglect detection system (AREEN) during resting-state and a visuospatial task. Connectivity was measured using coherence in delta, theta, alpha, and beta bands for both conditions, with gamma-band coherence assessed only during the task. Graph-based metrics were applied to model network-level disruptions. Classification models evaluated the significance of connectivity features to find patterns predictive of neglect.
Results
The neglect group showed reduced connectivity in frontal and right parieto-occipital (ParOcc) regions, primarily in beta and theta bands, during both conditions, with additional gamma-band connectivity differences in the task condition, compared to the non-neglect group. Conversely, connectivity was greater in central and midline regions, which may indicate a maladaptive shift in network organization. Classification models accurately classified patients into neglect and non-neglect groups (resting-state: 87.0% ± 0.7%; task: 80.9% ± 16.0%). Feature importance analysis identified eigenvector and closeness centrality within frontal, right ParOcc, and central regions as key predictors.
Conclusions
Network disruptions can effectively identify SN and provide potential targets for connectivity-based rehabilitation. Future studies should investigate whether these interventions improve attention and recovery in stroke patients.
This study was registered at ClinicalTrials.gov under ID NCT04187131.
Introduction
Spatial neglect (SN) is a common neurological impairment following stroke, characterized by deficits in visual perception and spatial awareness on the contralesional side of the environment. 1 About 30% of stroke survivors experience SN, particularly after right-hemispheric lesions, due to the dominant role of the right hemisphere in spatial attention. 2 Although symptoms often improve, persistent deficits interfere with essential daily activities such as dressing, eating, and navigating.3,4
SN mainly disrupts the dorsal attention network (DAN) and ventral attention network (VAN), which coordinate goal-directed attention and responses to salient stimuli. 5 Damage to critical cortical regions, including the temporoparietal junction, superior parietal lobule, frontal eye fields, and inferior frontal gyrus, as well as key white matter pathways like the superior longitudinal fasciculus and inferior fronto-occipital fasciculus, is associated with interhemispheric connectivity disruption and neglect severity.6 -9 These findings indicate that SN arises from large-scale network dysfunction rather than focal lesions alone.
Functional connectivity studies, primarily using resting-state functional magnetic resonance imaging (fMRI), have further identified disruptions within DAN and VAN associated with neglect, particularly in right-lateralized regions.10,11 Alterations are also evident in other networks, including the salience, sensorimotor, and default mode networks.12,13
Electroencephalography (EEG) complements fMRI by providing high temporal resolution and enabling frequency-specific analyses, alongside advantages such as portability and usability in clinical settings. Previous EEG studies have reported increased delta and decreased alpha activity, which correlate with visuospatial impairments.14,15 In particular, reduced alpha-band connectivity within the VAN consistently relates to neglect severity. 16 Despite these insights, EEG research on SN has largely focused on power spectral changes during resting conditions, with limited attention to frequency-specific connectivity and task-related network dynamics that are important for targeted rehabilitation.
To quantitatively characterize these connectivity disruptions, recent studies have increasingly applied graph metrics such as eigenvector centrality and modularity.17,18 These studies frequently report reduced network efficiency in key “neglect hubs” and disruptions in the balance between network integration and segregation. 19 Additionally, frequency-specific analyses have shown that alpha- and beta-band connectivity disruptions are closely correlated with neglect. 20 However, many existing studies still focus primarily on resting-state conditions, use a limited set of graph metrics, rarely directly compare neglect and non-neglect groups, and provide limited explanations of the underlying connectivity disruptions.
In the current study, we address these gaps by conducting a comprehensive EEG connectivity analysis of stroke patients with and without SN. Specifically, we (1) investigate connectivity during resting-state and task-based conditions, (2) assess alterations across multiple frequency bands, (3) apply an expanded set of graph metrics, and (4) use machine learning and feature importance evaluations to interpret clinical significance. We hypothesize that patients with neglect will show larger disruptions in higher-frequency connectivity and more prominent local and global network inefficiencies than those without neglect, particularly during contralesional tasks. By directly comparing patient groups across resting-state and task conditions, our findings aim to clarify distinct connectivity patterns underlying visuospatial neglect to guide targeted, connectivity-based rehabilitation methods.
Methods
In this study, we used the augmented reality (AR)-based EEG-guided neglect detection system (AREEN), previously developed by our team as a promising tool for detecting neglect in stroke patients. 21 The system was designed to overcome the limitations of traditional pen-and-paper tests, which, though widely used, lack the precision and spatial resolution needed to evaluate dynamic attention patterns or map deficits across the visual field in detail. 22 AREEN addresses these challenges by combining AR and EEG to deliver controlled stimuli and simultaneously record synchronized neural activity. Previous studies have shown the system’s effectiveness in detecting neglect and quantifying visuospatial deficits.21,23,24 Building on this foundation, we used the recorded EEG data for connectivity analysis to study network-level disruptions associated with neglect.
Participants
A total of 28 individuals with a history of stroke were recruited from the community and the University of Pittsburgh Medical Center hospital network. Recruitment was conducted under the University of Pittsburgh Institutional Review Board protocol (STUDY19060390) and registered at ClinicalTrials.gov (NCT04187131). Participants were divided into 2 groups: 16 individuals with SN and 12 without SN (WSN).
Eligible participants were at least 18 years old, had normal or corrected-to-normal vision, and had experienced a stroke. Exclusion criteria included severe visual field deficits, significant cognitive impairments, a history or high risk of seizures, metallic implants in the head, neurodegenerative diseases, or severe speech impairments. SN was assessed using the Behavioral Inattention Test Conventional subtests (BIT-C). 25 Participants scoring 129 or below on the overall BIT-C or failing at least one BIT-C subtest (Line Crossing, Letter Cancellation, Star Cancellation, Figure Copying, Line Bisection, or Representational Drawing) were classified as SN. In cases where BIT-C results indicated neglect but the field-of-view (FOV) map did not show corresponding deficits, group assignment was reviewed using both assessments. This applied to 1 SN and 6 WSN participants.
Demographic and clinical characteristics, including age, sex, stroke hemisphere, months since stroke, BIT-C scores, and data availability for each analysis, are summarized in Table 1. Only participants who contributed at least 1 valid dataset were included in the analysis. A dataset was considered valid if the participant completed the corresponding recording session and the data met preprocessing quality criteria. Based on these criteria, 16 participants (10 SN and 6 WSN) were included in the resting-state analysis, and 23 participants (12 SN and 11 WSN) were included in the task-based analysis.
Participant Characteristics.
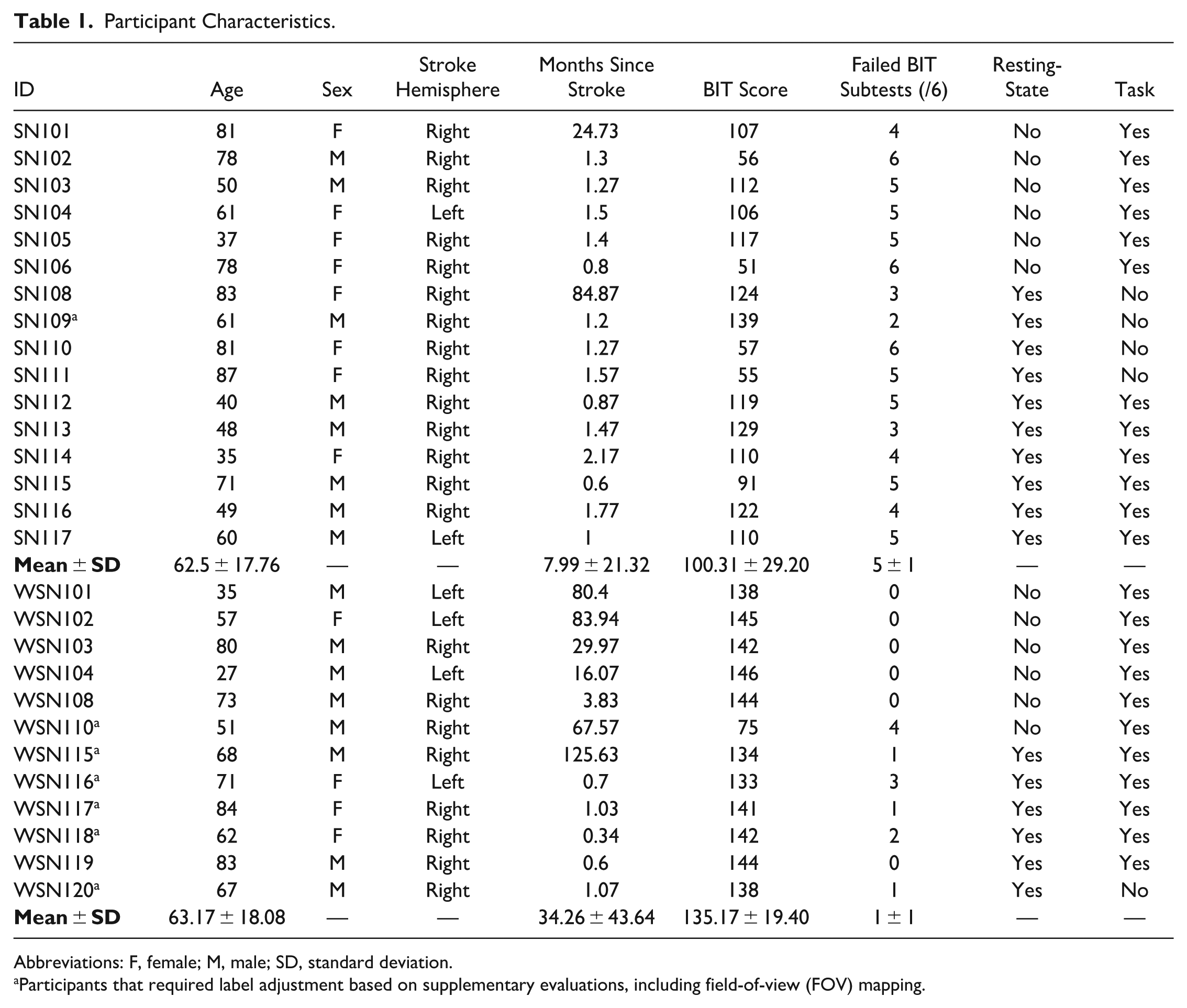
Abbreviations: F, female; M, male; SD, standard deviation.
Participants that required label adjustment based on supplementary evaluations, including field-of-view (FOV) mapping.
Experimental Setup and Data Collection
The AREEN system included a Microsoft HoloLens AR v1 headset for visual stimulus presentation and a g.USBamp amplifier for 16-channel EEG acquisition (Figure 1(A)). Stimuli were presented using a modified version of the Starry Night Test on a virtual 6 × 12 grid, with each cell corresponding to a specific region of the visual field. 26 Red targets (primary stimuli) and green distractors (secondary stimuli) were presented in randomized sequences to minimize anticipatory responses. Each grid cell was tested 3 times for a total of 216 trials per session. Stimulus durations ranged randomly between 0.05 and 0.25 seconds, with inter-stimulus intervals varying from 1.2 to 2.5 seconds (Figure 1(B)). 27
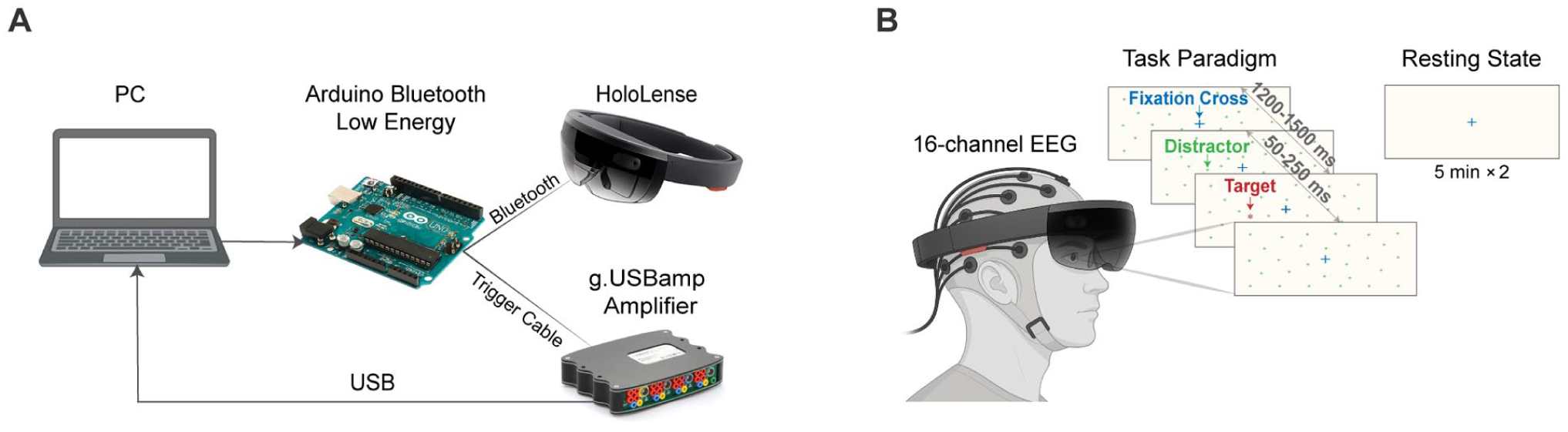
(A) The AREEN system setup for EEG data acquisition and stimulus presentation, including a PC for data recording, an Arduino Bluetooth Low Energy module for trigger synchronization, a Microsoft HoloLens AR headset for visual stimulus presentation, and a g.USBamp amplifier for 16-channel EEG recording. (B) Participant using the AREEN system during resting-state and task-based EEG recordings. Task paradigm shows fixation cross, distractors, and targets from the modified Starry Night Test, with randomized stimulus durations and inter-stimulus intervals. Resting state consisted of two 5-minute recordings per session.
EEG signals were recorded at a sampling frequency of 256 Hz using electrodes positioned according to the international 10-20 system at locations Fp1, Fp2, F3, F4, Fz, FC1, FC2, Cz, P1, P2, C1, C2, CP3, CP4, O1, and O2. The ground electrode was located at Fpz, and the reference electrode was placed over the left mastoid. A Bluetooth-enabled Arduino trigger system synchronized stimulus presentation with EEG recordings to ensure precise temporal alignment between visual stimuli and neural activity. 21
Data were collected across 2 experimental sessions for each participant. Each session lasted approximately 1 hour and included resting-state recordings, a clicker-based calibration phase, and a task-based EEG recording.
Participants sat comfortably in a quiet room wearing the EEG cap and Microsoft HoloLens AR headset. During resting-state recordings, participants were instructed to keep their eyes open and fixate on a central fixation cross displayed through the AR interface. Each resting-state recording lasted 5 minutes and was performed twice per session: once before the clicker-based calibration and once after task-based EEG recording to ensure data availability in case of quality issues with the first recording. After the first resting-state recording, participants completed a calibration phase using the modified Starry Night Test. In this phase, participants used a Bluetooth clicker to respond to the red targets. Targets remained visible until clicked or for a maximum of 3 seconds. Responses from this phase provided precise labels of detected and neglected targets, which were then used to generate FOV maps for group classification. In the subsequent task-based EEG recording phase, participants viewed the previously described visual stimuli without providing behavioral/clicker responses. EEG signals recorded during this phase were used for connectivity analyses.
Preprocessing
Data preprocessing was performed using the EEGLAB toolbox. 28 Separate pipelines were applied to resting-state and task-based data, with several initial steps common to both. During data acquisition, signals were filtered using an eighth-order Butterworth bandpass filter (2-62 Hz) and a fourth-order notch filter (58-62 Hz). Resting-state data were segmented into 2-second epochs and further bandpass-filtered (1-30 Hz) to reduce contamination from muscle artifacts and non-neural high-frequency noise, which disproportionately affect higher frequencies in resting-state EEG. This consideration is especially relevant in stroke patients, as muscle-related artifacts can be more pronounced in EEG recordings.29,30 Frequencies above 30 Hz were therefore not included in the resting-state analyses. Task-based data were bandpass-filtered (1-40 Hz), time-locked to event markers to correct transmission delays and segmented into epochs from 200 ms before to 800 ms after stimulus onset. Epochs were baseline-corrected using the mean amplitude of the pre-stimulus interval.
Channels were marked as bad if they showed flat signal (<1 μV standard deviation over extended segments) or excessive noise, defined as clearly abnormal high-amplitude or unstable signal relative to the other electrodes, with peak-to-peak amplitudes exceeding 150 μV in more than 20% of epochs. Marked channels, up to a maximum of 3 of 16 per recording, were interpolated using spherical spline estimates from the 3 nearest electrodes. The remaining channels were re-referenced to the average of all electrodes. Independent Component Analysis (ICA) was then used to identify and remove ocular, muscular, and other non-neural artifacts. 31 Epochs with substantial residual artifacts in more than 3 channels were discarded. Task-based trials were labeled as contralesional or ipsilesional based on the lesion side and stimulus location. Data from participants with left-hemisphere lesions were mirrored along the midline by mapping left-sided electrodes to corresponding right-sided positions to ensure consistent representation of the lesioned hemisphere on the right across analyses.
For participants with 2 recording sessions, either the higher-quality session was retained, or both sessions were merged if quality was comparable. Session quality was evaluated based on the proportion of ICA artifact components, number of interpolated channels, percentage of usable epochs, and presence of residual noise after preprocessing. Sessions compromised by issues such as EEG cap misalignment, pervasive noise, or more than 3 bad channels were excluded from analyses. Across retained task-based and resting-state sessions, approximately 92% of epochs were kept and about 0.3 of 16 electrodes (range: 0-3, ~2% of channels) were interpolated per session.
Connectivity Analysis
Functional connectivity was calculated using coherence, which quantifies the consistency of the relationship between 2 signals at specific frequencies. Coherence values range from 0, indicating no consistent interaction, to 1, representing perfect synchronization.
32
Coherence between 2 signals,
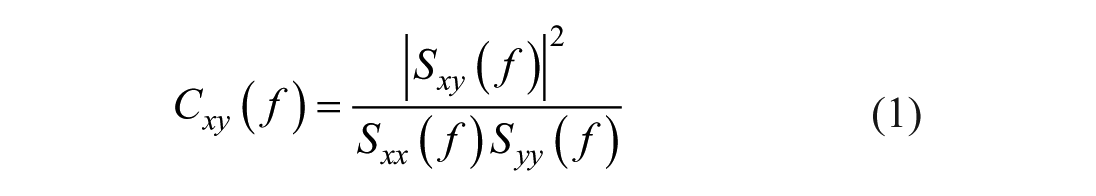
where
Pearson correlation and phase-locking value (PLV) were also calculated as alternative connectivity measures. Pearson correlation assesses linear relationships between signals in the time domain but lacks sensitivity to frequency-specific differences. PLV quantifies the consistency of phase differences between signals regardless of amplitude and showed similar patterns to coherence across participants.33,34 Coherence was selected as the primary connectivity metric because it best distinguished between neglect and non-neglect groups and is well-suited to capturing frequency-specific connectivity, which was central to our analysis (the performance of PLV and Pearson is reported in Supplemental Table 1, and the evaluation procedure is described in Classification and Feature Evaluation).
Connectivity measures were computed separately for resting-state and task-based data using MATLAB 2022a (MathWorks Inc., Natick, Massachusetts, USA). Resting-state coherence was calculated from non-overlapping 2-second epochs, segmented from 5 minutes of continuous EEG data in 4 frequency bands: delta (1-4 Hz), theta (4-7 Hz), alpha (8-12 Hz), and beta (13-30 Hz). For task-based data, coherence was also calculated in the gamma band (30-40 Hz). 35 Task-based analyses were divided into contralesional and ipsilesional conditions according to stimulus location relative to each participant’s lesion side. Each connectivity matrix had dimensions of 16 × 16 × N, where 16 corresponds to EEG channels, and N represents the number of epochs. Coherence values were computed for all possible channel pairs.
For subsequent graph analysis, coherence matrices were binarized into adjacency matrices representing significant connections. Coherence values were averaged across epochs to produce 1 connectivity matrix per participant for each frequency band. An adaptive threshold for each band was calculated using Otsu’s method. 36 This threshold was then uniformly applied across participants at the epoch level. Coherence values above the threshold were set to 1 (significant connection), and values below it were set to 0.
Graph Construction and Feature Extraction
Adjacency matrices derived from coherence calculations for each epoch, frequency band, and condition were used to create undirected, unweighted graphs for feature extraction using the NetworkX library in Python. 37 A comprehensive set of 8 graph metrics, categorized into global and node-level measures, was computed to model network topology and function. Global metrics included efficiency, density, modularity, and transitivity, and node-level metrics included betweenness centrality, clustering coefficient, eigenvector centrality, and closeness centrality. These metrics describe different aspects of the network, including the effectiveness of information transmission, the strength of overall and local connectivity, the organization into communities, and the influence of individual nodes. Full definitions and formulas are provided in Supplemental Table 2.38 -43
Node-level metrics were averaged across anatomical regions defined as follows: left frontal (Fp1 and F3), right frontal (Fp2 and F4), left central (FC1 and C1), right central (FC2 and C2), left parieto-occipital (ParOcc; P1, O1, and CP3), right ParOcc (P2, O2, and CP4), and midline (Fz and Cz). This regional grouping was guided by cortical regions underlying standard EEG electrode positions to balance dimensionality reduction and accurate representation of functional areas. 44
Classification and Feature Evaluation
To discriminate between neglect and non-neglect groups, classification analyses were performed separately for resting-state and task-based data using the scikit-learn library in Python. 45 Multiple classifiers were selected, including Random Forest, Support Vector Machines (SVM) with radial basis function kernels, Gradient Boosting, Linear Discriminant Analysis (LDA), and Gaussian Naive Bayes.
Model performance was evaluated using nested Leave-One-Participant-Out cross-validation to reduce subject-specific bias and assess generalization to unseen individuals. 46 In each fold, all epochs from 1 participant were held out as an independent test set, and data from the remaining participants formed the training set used for model development.
Within each fold, any preprocessing or feature extraction steps that required estimating thresholds (such as binarizing connectivity matrices described above) were performed using only these training participants, and the resulting thresholds were then applied to both the training set and test data in that fold. To account for variability in the number of epochs across participants, epochs from each participant were weighted inversely proportional to their epoch count. True labels were assigned at the participant level, with neglect participants defined as the positive class (SN = 1) and non-neglect participants as the negative class (WSN = 0). Within the training set, hyperparameters were tuned using group k-fold cross-validation (k = 5). In each inner split, epochs from a subset of participants were used for model fitting and epochs from the remaining participants were used for validation. After tuning, the model was refit on all available participants within the set and evaluated on the held-out participant. Hyperparameters for Random Forest and Gradient Boosting classifiers were optimized using Randomized Grid Search. Tuned parameters included number of estimators, maximum depth, minimum samples per split, subsample ratio, learning rate (Gradient Boosting only), and the number of features per split. A full list of tuned parameters and their search ranges is provided in Supplemental Table 3. Additionally, Recursive Feature Elimination with a Random Forest estimator was fit only on the training data within each fold, and the learned selector was then applied to the test data in that fold to retain the 30 most predictive features and improve interpretability by highlighting the most informative metrics.
Classification performance was summarized using sensitivity, specificity, and accuracy. Sensitivity assessed the proportion of neglect participants correctly classified; specificity measured the proportion of non-neglect participants correctly classified; and accuracy represented the overall proportion of correctly classified participants. For each participant, a confusion matrix was constructed from the corresponding epochs to compute accuracy, sensitivity, and specificity, and these metrics were then averaged across folds. Additionally, we computed the area under the receiver operating characteristic curve (AUC) with 95% confidence intervals (CIs) and reported positive and negative predictive values (PPV and NPV), with neglect as the positive class for clinical interpretability. To assess the significance of classifier performance, we implemented a permutation test for the classifier with the highest AUC in each dataset. For this analysis, the model’s predictions were first summarized for each participant by averaging the predicted probability of neglect across all epochs. Group labels (SN vs WSN) were then randomly permuted 1000 times relative to these scores, and the AUC was recomputed for each permuted sample to form a null distribution under the hypothesis of no association between labels and features. The permutation P-value was then computed from this null distribution.
Participant group imbalance in the resting-state dataset (10 neglect vs 6 non-neglect participants) was addressed by incorporating class-weight adjustments directly within the Random Forest algorithm. For task-based data, analyses were initially performed separately for contralesional and ipsilesional conditions. However, given the similar classification performance across conditions, datasets were combined into a single analysis to increase statistical power and simplify interpretation.
Finally, SHapley Additive exPlanations (SHAP) analysis was applied to the trained model with the best performance to interpret feature importance. 47 SHAP values clarified the contribution of each selected metric in distinguishing between groups and identified network patterns most strongly associated with neglect.
Statistical Analysis
Statistical analyses included comparisons of all node-level metrics between neglect and non-neglect groups. Special attention was given to metrics identified by SHAP analysis as most influential, particularly eigenvector centrality, closeness centrality, and betweenness centrality. For task-based data, within-subject differences between ipsilesional and contralesional conditions across frequency bands were compared using Wilcoxon signed-rank tests due to the non-parametric nature of the data. Since no significant differences were observed, these conditions were merged to increase statistical power. Group-level differences were then evaluated separately for the merged task-based and resting-state data using Wilcoxon rank-sum tests. False Discovery Rate (FDR) correction was applied to control for multiple comparisons across EEG channels and metrics, with statistical significance set at an FDR-corrected threshold of P < .05.
Results
Of the 28 stroke participants recruited, 16 were included in the resting-state analyses (10 SN and 6 WSN), and 23 were included in the task-based analyses (12 SN and 11 WSN). Demographic characteristics were comparable between groups, with no significant age difference (SN: 62.5 ± 17.8 years, WSN: 63.2 ± 18.1 years; P > .05). BIT scores were significantly lower in the SN group (100.3 ± 29.2) compared to the WSN (135.2 ± 19.4; P < .01). Although time since stroke was shorter for SN participants (8.0 ± 21.3 months) compared to WSN (34.3 ± 43.6 months), this difference was not statistically significant (P > .05). Lesion distribution was predominantly right-hemispheric in the SN group (14 right and 2 left), whereas the WSN group had a less right-lateralized distribution (8 right and 4 left).
Connectivity Biomarkers of Neglect at Rest
Resting-state data effectively discriminated between SN and WSN participants (2491 epochs, SN prevalence: 63%). Gradient Boosting achieved the highest overall accuracy (87.0% ± 0.7%), with strong specificity (73.5% ± 1.5%) and sensitivity (94.9% ± 0.6%). This model yielded an AUC of 0.90 (95% CI: 0.89-0.92), with PPV of 0.86 and NPV of 0.89 at a probability threshold of 0.5. Random Forest also performed well with slightly lower accuracy (86.9% ± 0.7%) and specificity (70.8% ± 1.5%) but achieved the highest sensitivity (96.3% ± 0.5%), and a slightly higher AUC of 0.93 (95% CI: 0.91-0.94; PPV: 0.85, NPV: 0.92). SVM, LDA, and Naive Bayes showed comparatively weaker performance (Figure 2(A)). Full classification metrics, including AUC with 95% CIs, PPV, and NPV, are provided in Supplemental Table 4. Gradient Boosting was selected for the SHAP analysis since it provided a good balance between sensitivity and specificity, high AUC with well-calibrated probabilities (Supplemental Figure 1A), and its performance remained significant under permutation testing (null accuracy 0.50 ± 0.16; P < .001).
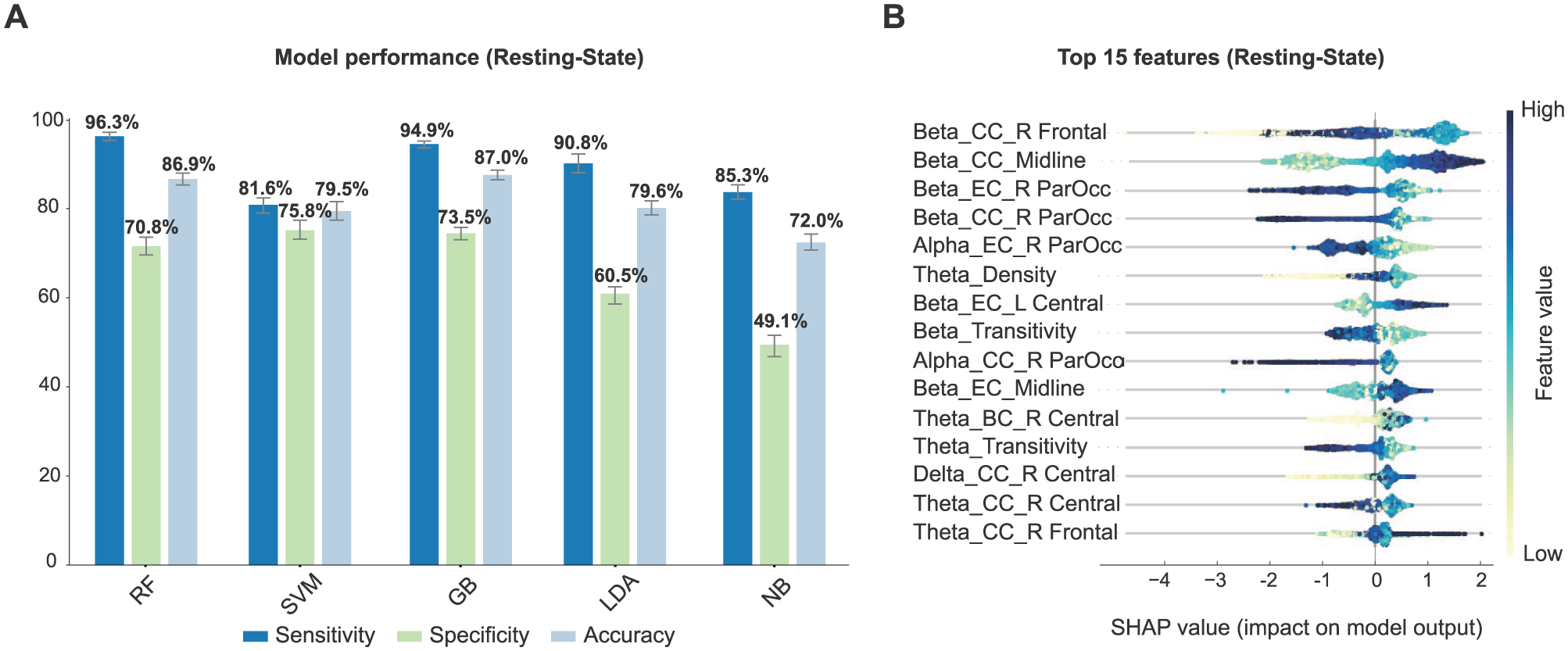
(A) Classification performance (mean ± SD) of resting-state data using leave-one-participant-out cross-validation across 5 classifiers. Bars indicate sensitivity (dark blue), specificity (green), and overall accuracy (light blue) for each model. (B) Top 15 EEG features contributing to classification, ranked by SHAP values. Each row corresponds to 1 feature, and each dot represents an individual epoch. Dot placement shows the strength and direction of each feature’s influence on predictions toward SN (positive values, right) or WSN (negative values, left). Color reflects feature value magnitude, ranging from low (yellow) to high (blue).
Figure 2(B) highlights the top features contributing to the classification. The SN group showed decreased beta-band closeness centrality in right frontal and right ParOcc electrodes. In contrast, SN participants showed higher beta-band closeness and eigenvector centrality along midline sites, as well as higher beta-band eigenvector centrality in the left central region. Additionally, lower beta-band transitivity was associated with neglect. Other important predictors included increased theta-band betweenness centrality in the right central region, larger delta-band closeness centrality in the right central area, and higher theta-band closeness centrality in right frontal electrodes. Lower alpha-band eigenvector centrality in right ParOcc electrodes and decreased theta-band density were also significant indicators differentiating SN from WSN participants.
Statistical analyses showed significant group differences primarily involving centrality metrics in distinct regions, consistent with SHAP feature importance results (Figure 3). Eigenvector centrality was significantly higher for SN participants at electrode C1 within the beta frequency band (Cohen’s
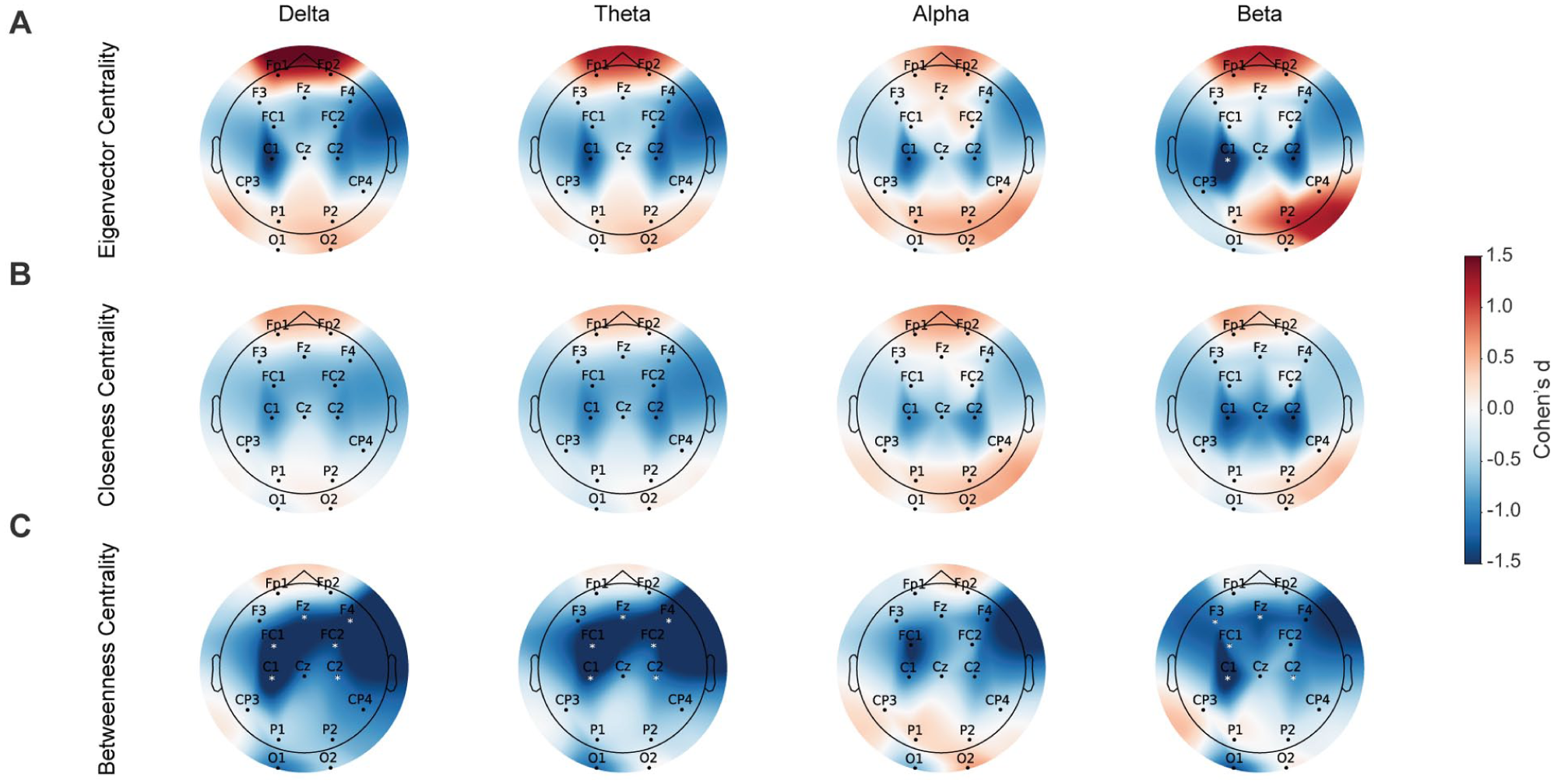
Topographic maps representing resting-state differences between WSN and SN groups across 4 frequency bands in: (A) eigenvector centrality, (B) closeness centrality, and (C) betweenness centrality. Colors illustrate Cohen’s
Connectivity Patterns of Neglect During a Visuospatial Task
As previously mentioned, there were no statistically significant differences between contralesional and ipsilesional conditions, and classification performance was also similar, with only slightly higher accuracy for contralesional targets. Therefore, data from both conditions were combined (5977 epochs, SN prevalence: 54%) to simplify interpretation and increase statistical power. Among classifiers, LDA achieved the highest overall accuracy (80.9% ± 16.0%), along with robust sensitivity (85.5% ± 11.6%) and specificity (75.8% ± 18.9%). Random Forest had the highest sensitivity with the lowest variation (85.6% ± 7.9%) and comparable accuracy (79.9% ± 12.8%). LDA and Random Forest yielded similar discrimination performance, with AUCs of 0.89 (95% CI: 0.88-0.90; PPV: 0.81, NPV: 0.81) and 0.88 (95% CI: 0.87-0.89; PPV: 0.79, NPV: 0.81), respectively. Gradient Boosting performed similarly (accuracy: 79.7% ± 12.0%), with an AUC of 0.88 (95% CI: 0.87-0.89; PPV: 0.81, NPV: 0.79), but had slightly lower sensitivity. SVM and Naive Bayes classifiers showed relatively weaker performance (accuracy: 67.9% ± 22.3% and 77.1% ± 19.6%, respectively; Figure 4(A)). Full classification metrics, including AUC with 95% CIs, PPV, and NPV, are provided in Supplemental Table 4. Given Random Forest’s high sensitivity, consistent specificity, lower variance in accuracy, reliable calibration of predicted probabilities (Supplemental Figure 1B), and robust performance under permutation testing (null accuracy 0.50 ± 0.12; P < .001), it was selected for feature interpretation.
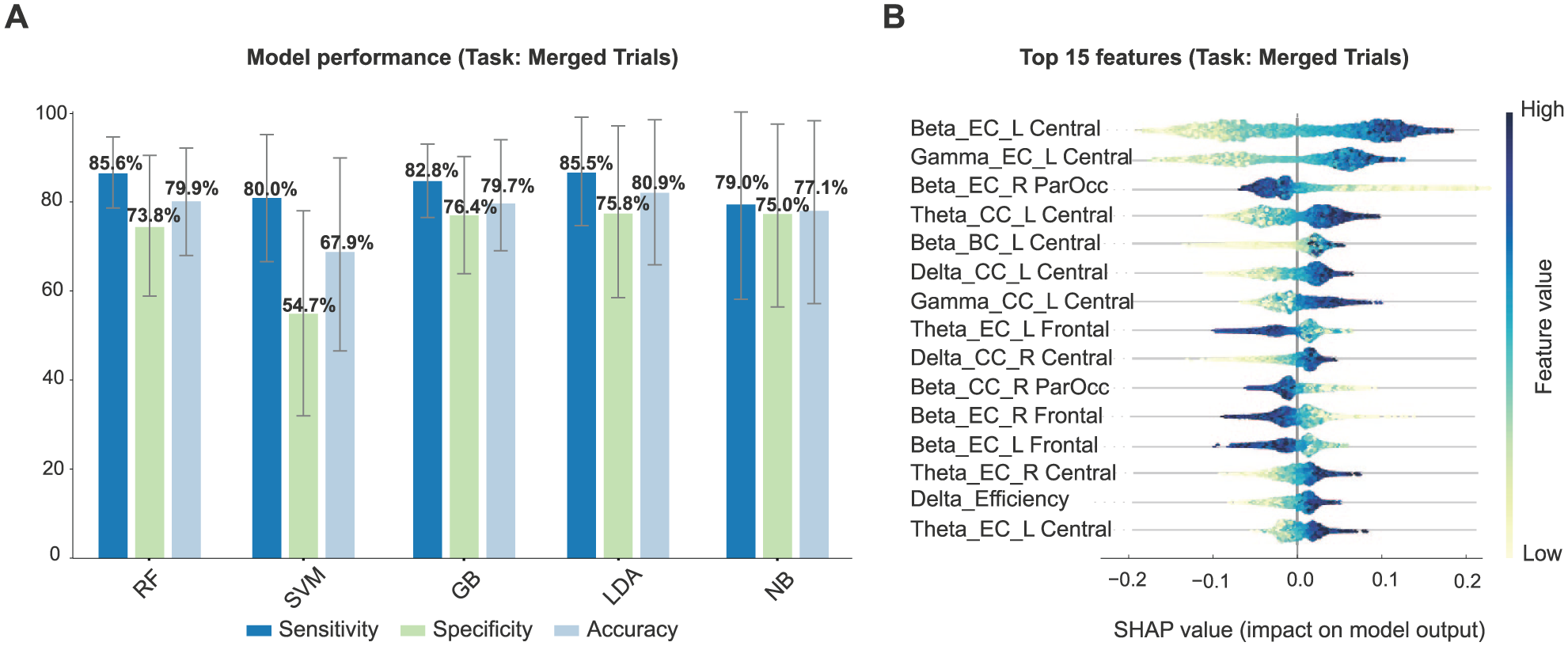
(A) Task-based classification accuracy (mean ± SD) using leave-one-participant-out cross-validation across 5 classifiers. Results are based on merged ipsilesional and contralesional trials. Bars indicate sensitivity (dark blue), specificity (green), and overall accuracy (light blue) for each model. (B) Top 15 EEG features influencing classification, ranked by SHAP values. Each row represents a single feature, with dots corresponding to individual epochs. Dot positions indicate how strongly each feature influences predictions toward SN (right) or WSN (left), with colors representing feature values from low (yellow) to high (blue).
Figure 4(B) summarizes features that best distinguished SN from WSN participants during the visuospatial task. The SN group displayed distinct patterns, with higher centrality in central electrodes and lower centrality in frontal and ParOcc sites. Specifically, in the gamma band, SN participants showed increased eigenvector and closeness centrality at left central electrodes. In the beta band, SN participants had higher eigenvector and betweenness centrality at left central electrodes but lower eigenvector and closeness centrality at bilateral frontal and right ParOcc electrodes. Within the theta band, SN was associated with increased closeness centrality at left central electrodes, higher eigenvector centrality at bilateral central electrodes, and lower eigenvector centrality at left frontal sites. In the delta band, SN showed increased closeness centrality at bilateral central electrodes and higher overall network efficiency.
Significant group differences were observed primarily involving centrality metrics across regions. Eigenvector centrality (Figure 5(A)) was significantly increased for SN participants at central electrodes within delta, theta, and beta bands (Cohen’s
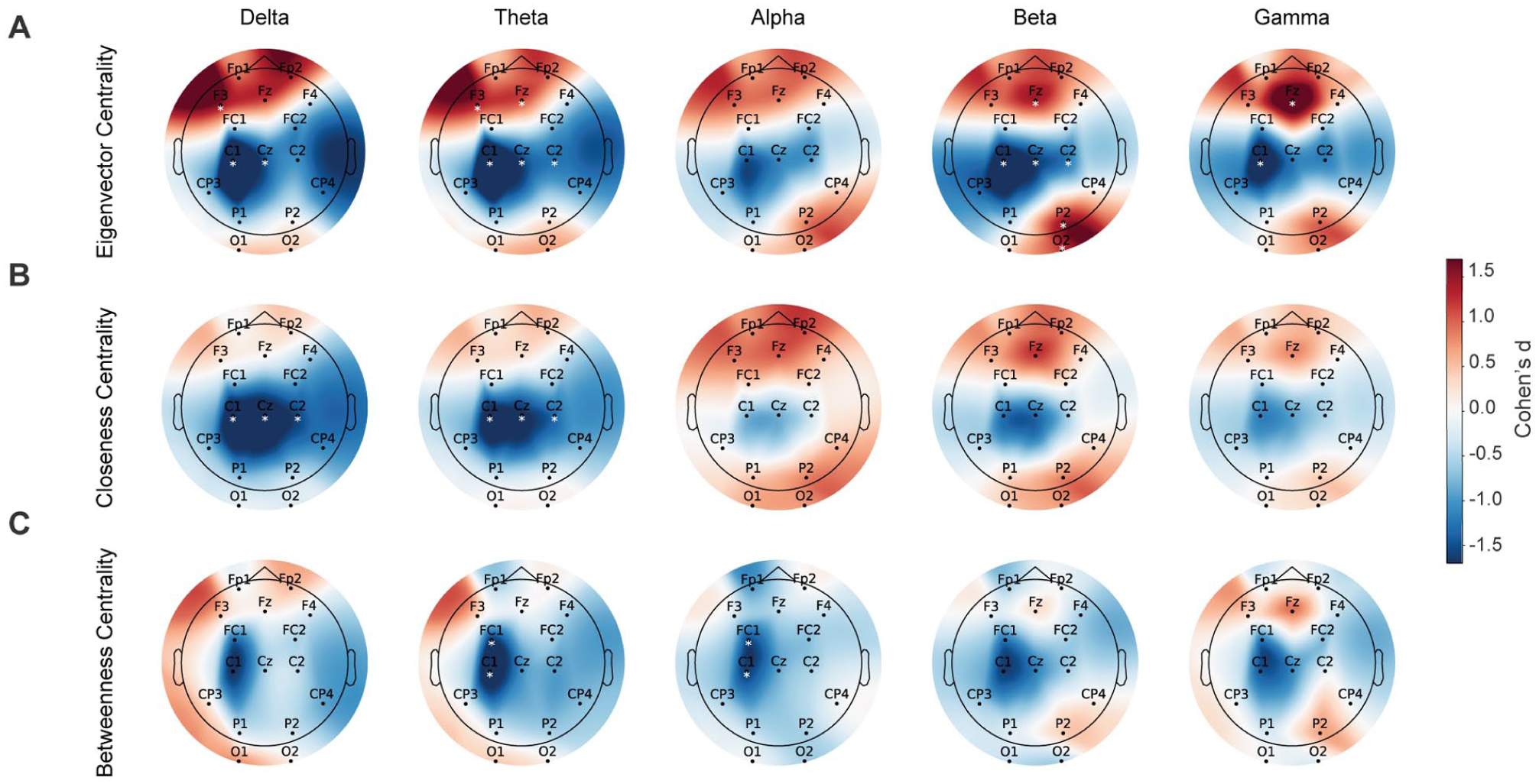
Topographic maps representing task-based (merged ipsilesional and contralesional trials) differences between WSN and SN groups across 5 frequency bands in: (A) eigenvector centrality, (B) closeness centrality, and (C) betweenness centrality. Colors illustrate Cohen’s d effect sizes, with red indicating relatively higher values in the WSN group and blue indicating relatively higher values in the SN group. White asterisks indicate channels with significant group differences (P < .05, FDR corrected).
Discussion
In this study, we compared functional connectivity patterns in stroke patients with and without SN during resting-state and task-based conditions across multiple frequency bands. At rest, stroke patients with neglect showed lower eigenvector and closeness centrality in frontal and right ParOcc electrode sites, particularly within the beta and theta bands. Centrality metrics describe how effectively brain regions communicate, integrate information, and influence the broader network.48,49 Lower centrality values suggest that frontal and ParOcc sites are less effective at supporting information integration within the attention network. Similar disruptions in frontal and parietal connectivity in patients with SN have been widely reported in the literature.50 -52 In contrast, we found increased eigenvector centrality in central and midline electrodes, which suggests that these areas became more dominant hubs in the brain network. This pattern aligns with previous findings showing that, in right-hemisphere lesions, brain organization tends to shift from higher-level associative regions toward lower-level visual and sensorimotor areas. Although this reorganization may help maintain network communication after stroke, it is often considered maladaptive, as it can lead to less efficient spatial processing and contribute to persistent neglect symptoms. 53 We also found higher betweenness centrality at central and fronto-central electrodes in SN patients, which indicates that network communication became more dependent on a limited set of critical regions. A similar pattern has been observed in stroke patients with neglect, where dependence on specific key hubs leads to weaker and less stable network organization. 11 At the global level, we observed disruptions in network density and transitivity within the theta band. These findings are consistent with previous reports of reduced network density and clustering following stroke. 54
During the visuospatial task, we found a similar pattern of lower eigenvector centrality, with some differences across frequency bands. In the delta and theta bands, disruptions were mainly localized to bilateral frontal electrodes, whereas in the beta and gamma bands, lower centrality values extended to both frontal and right ParOcc sites. Eigenvector and closeness centrality remained higher in central and midline electrodes across the delta, theta, and beta bands, as observed at rest. Betweenness centrality also stayed higher at fronto-central electrodes. However, unlike in the resting-state, global network measures did not show significant changes during the task. This is consistent with previous work showing that task demands often lead to more localized network disruptions. 55 We initially hypothesized that when participants processed contralesional targets, the increased task demands would result in clearer group differences and higher classification accuracy compared to ipsilesional targets. This was based on prior studies that reported more substantial attentional deficits on the neglected side under increased cognitive load.26,55 However, contrary to previous findings, we observed only a small and statistically non-significant difference in classification accuracy between contralesional and ipsilesional conditions. Several factors might explain this result, including relatively short trial durations, limited time for target representation, the limited spatial resolution of our 16-channel EEG setup, or the possibility that the task was not demanding enough to detect subtle lateralized differences in connectivity patterns.
Our findings can help guide the design of targeted interventions for neglect. The reduced connectivity we observed in right frontal and ParOcc electrodes, particularly during the task, suggests these areas as potential targets for intervention. Previous studies have shown that excitatory stimulation, such as high-frequency repetitive transcranial magnetic stimulation applied to the right posterior parietal cortex, significantly improves neglect.56,57 Our results align with these findings by identifying similar connectivity disruptions within right-hemisphere sites. In addition, previous EEG neurofeedback methods focusing on alpha-band activity showed modest and temporary improvements. Our findings suggest that targeting beta and gamma bands during tasks might lead to better outcomes. 58 We also found similar connectivity patterns during resting-state and task conditions. The overlap shows that interventions focused on either resting-state or task-related networks could improve both. A recent study further supports this idea by showing that virtual reality-based visual scanning rehabilitation, in which patients repeatedly practiced directing their attention toward the neglected space, led to improved resting-state connectivity within the DAN in chronic neglect patients. 59
Several limitations should be considered in this study. The small sample size and uneven group distribution, particularly in the resting-state data, may have limited the generalizability of our findings to the broader stroke population. There was also high variability in lesion laterality and time since stroke within the non-neglect group. Both factors are known to substantially influence patterns of brain network disruption after stroke, and the cross-sectional design also prevented us from exploring how connectivity and neglect symptoms change over time. This variability may partly explain the lower classification accuracy observed in the non-neglect group. Additionally, the modest sample size and this heterogeneity prevented us from performing stratified analyses by chronicity, lesion side, or neglect severity. Another limitation is the use of a 16-channel EEG system, which limited spatial resolution and the ability to detect more subtle connectivity patterns. Accordingly, we were not able to perform reliable source localization, and interpretations are confined to electrode-level topographies rather than cortical sources. 60 Finally, as discussed, our task design was brief and low in behavioral demands to be feasible for patients in the acute and subacute stages after stroke. However, the lack of trial-by-trial behavioral responses may have reduced sensitivity to subtle differences between ipsilesional and contralesional conditions.
Future studies could address these limitations by designing more cognitively demanding tasks to better differentiate between contralesional and ipsilesional processing, combined with higher-density EEG setups to improve spatial resolution. A larger sample of the stroke population with more balanced groups by neglect status that can be stratified by major potential confounding variables would also allow more direct comparisons of how left and right hemisphere lesions differently affect network connectivity, which can provide a clearer understanding of hemisphere-specific disruptions in neglect. Future work should also extend these network-based approaches to chronic stroke and evaluate whether integrating them with standard clinical assessments provides incremental diagnostic value and improves detection and characterization of subtle or residual neglect. In addition, to move toward rehabilitation studies, future work should assess the behavioral relevance of these connectivity alterations by associating them with functional measures such as the Catherine Bergego Scale and activities of daily living. Finally, longitudinal studies with repeated EEG assessments in additional patient cohorts and with multiple EEG setups are needed to determine whether these measures remain stable over time, reflect clinical change, and generalize across different clinical environments.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683261419423 – Supplemental material for Brain Network Connectivity During Resting-State and a Visuospatial Task as a Biomarker for Spatial Neglect in Stroke Patients
Supplemental material, sj-docx-1-nnr-10.1177_15459683261419423 for Brain Network Connectivity During Resting-State and a Visuospatial Task as a Biomarker for Spatial Neglect in Stroke Patients by Golnaz Haddadshargh, Richard Gall, Emily S. Grattan, Sarah Ostadabbas, George F. Wittenberg and Murat Akcakaya in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to acknowledge Katie Mullen, Brice Hounshel Smith, and Aleia Kramer from the University of Pittsburgh’s Department of Occupational Therapy for their support in organizing and supervising data collections. We also thank Dr. Jennifer Mak and Dr. Deniz Kocanaogullari for their extensive contributions to data collection and development of the Areen system.
Author Contributions
Golnaz Haddadshargh: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Visualization; Writing—original draft; and Writing—review & editing. Richard Gall: Investigation; Software; and Writing—review & editing. Emily S. Grattan: Conceptualization and Writing—review & editing. Sarah Ostadabbas: Conceptualization; Software; and Writing—review & editing. George F. Wittenberg: Conceptualization; Supervision; and Writing—review & editing. Murat Akcakaya: Conceptualization; Methodology; Project administration; Supervision; and Writing—review & editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science Foundation grants #1915083, #1915065, and #2234346.
Ethical Considerations
All experimental procedures were approved under the University of Pittsburgh Institutional Review Board number STUDY 19060390 on October 8, 2019.
Consent to Participate
All participants provided written informed consent prior to participating.
Consent for Publication
In accordance with the consent form, only de-identified participant data may be shared publicly. Consequently, no images, videos, or other potentially identifying information is included in this publication.
Data Availability Statement
Due to the inclusion of sensitive personal information, the datasets of the current study are not publicly available. However, the data supporting this publication are available from the authors upon reasonable request.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
