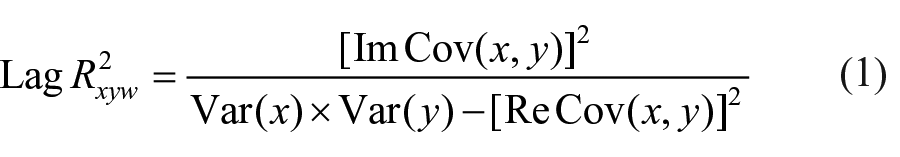Abstract
People with multiple sclerosis (MS) frequently complain of excessive fatigue, which is the most disabling symptom for half of them. While the few drugs used to treat MS fatigue are of limited utility, we recently observed the efficacy of a personalized neuromodulation treatment. Here, we aim at strengthening knowledge of the brain network changes that occur when MS fatigue increases, using graph theory. We collected electroencephalographic (EEG; 23 or 64 channels) data in resting state with eyes open in 27 relapsing-remitting (RR) patients with mild MS (EDSS ≤2), suffering a wide range of fatigue as scored by the modified Fatigue Impact Scale (mFIS) (2-69, within a total range 0-84). To estimate graph theory small-world index (SW), we calculated the lagged linear coherence between EEG cortical eLORETA sources, in the standard frequency bands delta (2-4 Hz), theta (4-8 Hz), alpha1 (8-10.5 Hz), alpha2 (10.5-13 Hz), beta1 (13-20 Hz), beta2 (20-30 Hz), and gamma (30-45 Hz). We calculated the SW of these undirected and weighted networks separately in the four left and right frontal (motor) and parieto-occipito-temporal (sensory) brain networks. A correlative analysis demonstrated increased fatigue symptoms along with the SW specifically in the Sensory network of the left dominant hemisphere in the beta1 band (Pearson’s r = 0.404, P = .020). Our study indicates a specific involvement of the dominant-hemisphere sensory network in MS fatigue. It suggests that compensatory neuromodulation interventions could enhance efficacy in relieving this debilitating symptom by targeting this area.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system (CNS) characterized by multiple lesions affecting the neuronal myelin and accumulative clinical signs due to demyelination and progressive axonal damage. 1 Meanwhile, it seems that MS disease progression predominantly depends on accumulated axon degeneration. MS axonal loss occurs early in the course of MS, and may be more significant in generating neurological function impairment than the loss of myelin. 2 Thus, in this phase, the MS impairment arises predominantly from impaired nervous impulse propagation,3,4 which can disrupt functional interaction between widely distributed brain regions.5,6 In fact, the disease-related tissue damage delays and disrupts neural signal transmission along cortico-cortical,3,7 cortico-spinal, and cortico-subcortical connections, causing altered information transfer.
Fatigue is a bothersome symptom caused by a variety of neurological diseases; in MS, fatigue is the single symptom that patients identify as interfering most with their daily activities. 8 With MS, up to 90% of patients complain of fatigue even early in the disease course. Fatigue can affect the social relationships as well as the daily cognitive and physical activities of MS sufferers. Clinically, MS patients report fatigue as exhaustion, lack of energy, increased somnolence, worsening of MS symptoms with augmented weakness exacerbated by activity and endogenous body temperature increase. 9 Although several findings confirm the existence of peripheral nervous system components of fatigue in MS, there are clear indications that “central” mechanisms 10 play a main role. Crucial elements of this phenomenon involve sensorimotor system alterations.11,12 In particular, previous findings show that functional connectivity alterations appear in MS-related fatigue before any local functional or structural damage becomes evident.13,14 Here, we tested the working hypothesis that in MS patients the connectivity and network topology patterns are altered in tandem with the level of fatigue symptoms. Among cortical districts, sings of a parietal lobe pivotal role in MS fatigue come from functional and structural studies. In fact, fatigued MS patients show signs of reduced primary somatosensory area (S1) excitability3,7 and higher excitability of their primary motor area (M1) than nonfatigued patients and healthy subjects. The reduction of S1 excitability may be at the origin of enhanced M1 excitability, although this phenomenon is generally attributed to a failure of local intracortical inhibition (for a review, see Yusuf and Koski 15 ). Nevertheless, the opposite direction of neuronal excitability (too low in S1 and too high in M1) speaks in favor of a parietal sufferance originating from the neuronal alterations in MS fatigue. Furthermore, other magnetic resonance imaging (MRI) and functional MRI (fMRI) data strengthen the notion of structural and functional parietal involvement in MS fatigue symptoms.16-18 A specific question of our working hypothesis is whether or not cortical network connectivity is impaired in parietal sensory regions when fatigue symptoms increase. To also test possible interhemispheric asymmetries in network topologies, as we observed an increase of hemispheric imbalances with increasing fatigue, 14 we investigated connectivity properties separately in the 2 hemispheres. In pursuit of this aim, we recruited patients in the early phase and at low clinical severity to minimize the confounding effects on the fatigue-related brain signs associated with severe clinical conditions.
In investigating the networking nature of the brain, connectivity-based studies, recently termed connectomics,” 9 have shown several clinically relevant findings regarding neurological disorders, including MS.6,20 Resting state functional connectivity studies are becoming progressively more popular in the assessment of brain connectivity in neurological disease.21-24 Graph theory is a mathematical approach that measures the clustering coefficient, a measure of local connectivity, and path length, a measure of overall network efficiency, to describe the topology of the brain network architecture. 19 Assessed using these measures, functional changes in the brain are related to the amount of structural damage present and to clinical severity.25-27 Furthermore, the ratio between the individual normalized clustering coefficient and the normalized path length, termed small-world (SW), 28 describes the balance between the local connectedness and the global integration of a network. To test our working hypotheses, we measured graph analytical parameters in individual patients in parallel with levels of fatigue symptoms. In accordance with the expected specific parietal sensory involvement, we assessed SW separately in motor (precentral) and sensory (postcentral) cortical regions. In the expectation that differences would be found between the two hemispheres in their relationship with fatigue symptoms, we assessed the left and right hemispheres separately.
Methods
Study Design
We investigated the connectivity properties of left and right motor and sensory cortical districts by utilizing graph theory measures of dependence on fatigue levels. In order to be sensitive to fatigue dependence, we enrolled patients suffering a wide range of fatigue, from minimal to high levels. Prior to conducting the correlative analysis, we selected those brain connectivity traits that differed between two groups, defined as suffering from higher or lower levels of fatigue. That is to say, we subdivided the recruited patients in 2 groups; with modified Fatigue Impact Scale (mFIS) cutoff equal half of the mFIS range. After selecting the divergent parameters between these 2 groups, we evaluated them through correlative analysis of the entire group of patients, to avoid any dependence on mFIS cutoff.
Participants
We recruited patients at the MS center of “San Giovanni Calibita” Fatebenefratelli Hospital (Rome, Italy) following the criteria of McDonald et al (2001) and Lublin and Reingold (1996).29,30 Specifically, we applied the following inclusion criteria: (a) minimal clinical severity (Extended Disability Status Scale, EDSS ≤2) and (b) absence of clinical relapse or radiological evidence of disease activity over the past 3 months. The exclusion criteria were the following: (a) assumption of symptomatic drugs that may affect the level of fatigue, depression, and anxiety within the past 3 months; (b) symptomatic drugs for fatigue treatment (suspended at least 2 months before inclusion in the study); (c) epilepsy or other central/peripheral nervous system comorbidities; and (d) systemic conditions that may cause fatigue—assessed by clinical examination and history collection: anemia, pregnancy, infectious diseases, hypo- or hyperthyroidism, cardiovascular disease, pulmonary disease, renal disease, and hepatic disease. No physical therapy was administered and patients suspended use of drugs intended to counter fatigue before inclusion in the study.
For the control group, 11 healthy volunteers (9 females; age range 28-49 years, mean = 36 years, independent samples 2-tailed t test P = .233) were recruited.
All patients and control subjects were right-handed (Edinburgh Inventory >85).
Patient’s Assessment
During clinical examination, we obtained information on ongoing medical therapy, disease duration and annual relapse rate, Beck Depression Inventory (BDI) and EDSS. The modified Fatigue Impact Scale (mFIS) was used to score the severity of fatigue symptoms.
Electrophysiological Recordings
In the first 18 patients recruited, we recorded EEG from a precabled cap (model CUEE60M, Sei EMG s.r.l., Cittadella, Italy, Figure 1A) with 23 Ag/AgCl electrodes (the standard 19 channels of the 10-20 international system plus FC4, FC3, CP4, CP3 to enhance the study of primary sensorimotor networks), with a mid-frontal reference and an occipital ground. To control eye blinking and cardiac interferences, we used other Ag/AgCl cup electrodes’ pairs for recording electrooculogram (EOG) and electrocardiogram (ECG). We sampled all data at 1024 Hz (presampling analogical bandpass filtering 0.48-256 Hz) and we stored data on a computer for off-line processing (Micromed System Plus equipped with SAM32 headbox, Micromed s.p.a., Mogliano Veneto, Italy). In the 9 people with MS recruited later, we acquired EEG using a 64-channel actiCHamp System (Brain Products GmbH, Munich, Germany) with electrodes positioned in the sites of 10-10 EEG International System. We used 5 kHz of sampling frequency (after bandpass filter 0.1-2000 Hz). Signals were down-sampled at 1024 Hz for homogeneity with the 23-channel recordings. Electrode impedances were maintained at less than 5 kohm in both cases.
Experimental setup and connectivity measures. (A) Electroencephalography (EEG) cups for 64- and 23-channel recordings. (B) Functional connectivity estimated as LagR among the Brodmann area activities as obtained by eLORETA in 4 brain regions: left and right frontal, left and right parieto-occipito-temporal lobes (MotorL, MotorR, SensoryL, SensoryR). (C) Small-world (SW) organization (middle graph, SW about 1) is intermediate between that of regular networks or lattices (left graph, SW higher than 1, high local clustering and short path length), and that of random networks (right graph, SW lower than 1, low local clustering and long path length).
All subjects sat comfortably on a chair in front of a screen displaying a fixation point. For each subject, we collected 5 minutes’ EEG in resting state with open eyes.
EEG Data Preprocessing
A semiautomatic independent component analysis (ICA)–based procedure was used in order to identify and remove cardiac and ocular artifacts without rejecting the contaminated epochs (Figure 2). 31
Artifact identification. First column, time evolution (Time) of the independent components (ICs) identified as follows (from the first row on top to bottom): ocular (Blinking, and Horizontal [H] movement), cardiac, power line, electrode and noise artifacts. For blinking and cardiac artifact, the reference electrodes’ dynamics (electro-oculogram [EOG], green and electrocardiogram [ECG], red) are shown to emphasize the timing of the artefactual occurrence in the selected ICs. We show proper time scale for each IC. From the wsecond column, for each ICs, the power spectral density (PSD). For blinking and cardiac artifact ICs, the EOG/ECG PSD (green/red line) are superimposed and the spectral correlation values (R) indicated. The probability density function (pdf) of each IC with the grey line indicating the corresponding normal probability density, with the values of kurtosis (K), and skewness (S). Spatial distribution of each artifact IC (Topography) obtained via the IC weights on recording channel positions. As independent component analysis (ICA) procedure performs whitened preprocessing, each quantity is in arbitrary unit.
Graph Theory Estimates
Functional Connectivity Analysis
EEG connectivity analysis has been performed using the exact low-resolution electromagnetic tomography (eLORETA) software, as provided by Roberto Pascual-Marqui (http://www.uzh.ch/keyinst/NewLORETA/LORETA01.htm). The eLORETA algorithm is a well-established linear inverse solution for EEG signals. Via individual analysis, brain connectivity was computed by sLORETA/eLORETA software on 4 cerebral regions, divided in frontal and parieto-occipito-temporal lobes, in the left and right hemispheres. The frontal (MotorL, MotorR) and partieto-occipito-temporal networks (SensoryL, SensoryR) included the precentral and postcentral Brodmann areas (BAs) in each hemisphere: MotorL/R: BA4, 6, 8, 9, 10, 11, 24, 25, 32, 33, 44, 45, 46, 47 and SensoryL/R: BA1, 2, 3, 5, 7, 17, 18, 19, 20, 21, 22, 27, 28, 29, 30, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43 (Figure 1B).
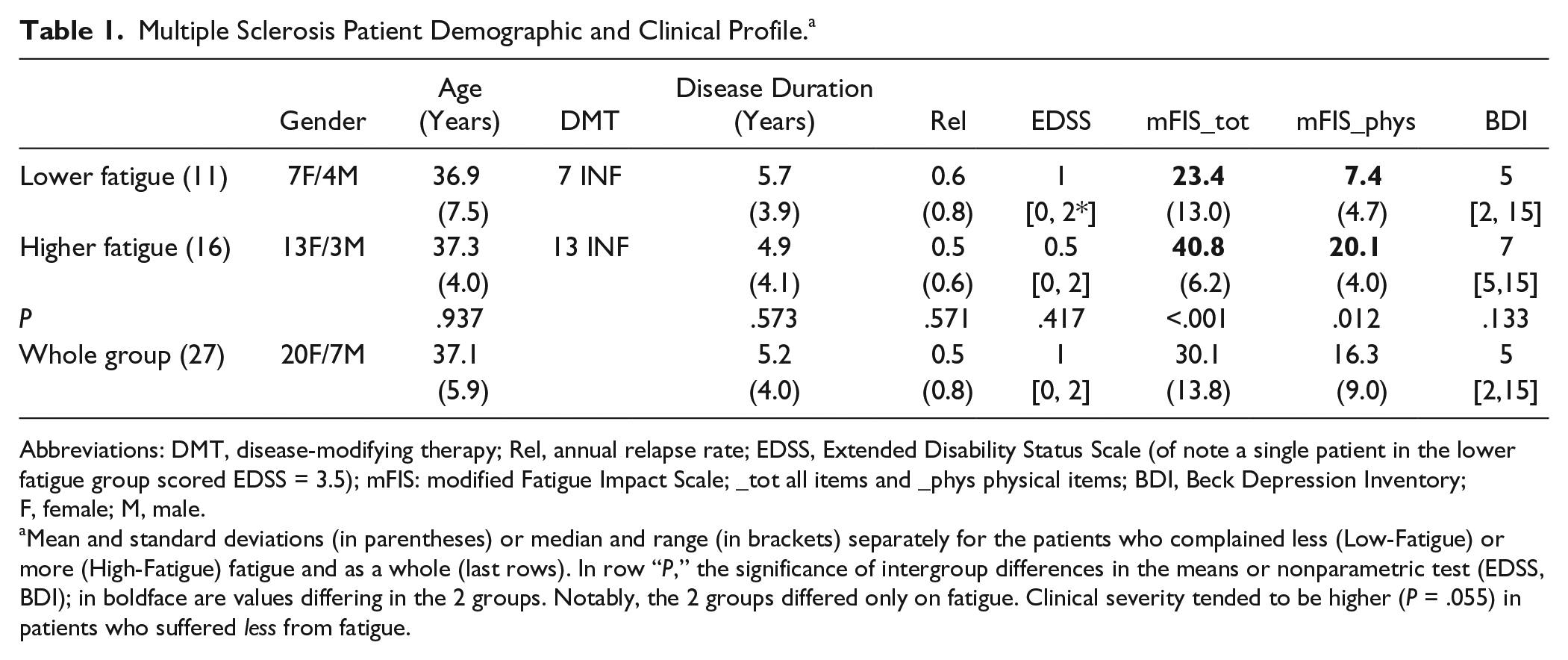
We used the eLORETA current density time series of all BAs within each of the networks (MotorL/R and SensoryL/R), to estimate the functional connectivity of the four nets. We used the Lagged Linear Coherence (LagR) algorithm, implemented in eLORETA, as a measure of functional physiological connectivity not affected by volume conduction and low spatial resolution. 32 The LagR in the frequency band ω is defined by the following equation 32 :
where x and y are the time series of 2 BAs, Im and Re are the imaginary and real part, respectively and Var and Cov are variances and covariance, respectively, of the signals. Lag R was computed between all possible pairs of BAs in each of the 4 networks for the seven independent EEG frequency bands delta (2-4 Hz), theta (4-8 Hz), alpha1 (8-10.5 Hz), alpha2 (10.5-13 Hz), beta1 (13-20 Hz), beta2 (20-30 Hz) and gamma (30-45 Hz).
Graph Analysis
We computed the core measures of graph theory that summarize the aspects of integration and segregation of a network, adopting the estimates presented at http://www.brain-connectivity-toolbox.net, adapted by our own Matlab scripts. 33 Segregation (or specialization) refers to the degree to which a network’s elements form separate clusters (clustering coefficient, C). Integration refers to the capacity of the network as a whole to become interconnected and exchange information (defined by the characteristic path length coefficient, L). Here, we evaluated the weighted clustering coefficient (Cw) and characteristic path length (Lw). These 2 parameters were used in order to compute the small-world coefficient (SW, Figure 1C), considered a summary of the brain network organization, as it measures the balance between the local connectedness and the global integration of a network. The measure of network small-worldness is defined as 34 the ratio of the normalized Cw, and the normalized Lw. To obtain individual normalized measures, we divided the values of characteristic path length and of clustering coefficients by the mean values obtained by the average in all bands of each subject. When SW is about 1, a network is said to have “small-world properties” meaning a good combination of high levels of local clustering among nodes (to form families or cliques) and proper paths that globally link all network nodes. This means that all nodes of a large system are linked through relatively few intermediate steps, despite the fact that most nodes maintain only a few direct connections each—mostly within a clique of neighbors. Small-world values higher than 1 indicate high levels of local clustering among nodes of a network and many short paths that globally link all nodes of the network. Small-world values smaller than 1 indicate poor local connectivity and stunted connections to the network at large.
Statistical Evaluation
After fitting the variable distribution to a Gaussian (checked by the Shapiro-Wilk test), an analysis of variance (ANOVA) was chosen since it is known to be robust with respect to the departure of normality and homoscedasticity of the data being treated. 35 We used the Greenhouse and Geisser correction for protection against violation of the sphericity assumption in the repeated-measure ANOVA. Additionally, we performed post hoc analysis with Duncan’s test. We performed the statistical analysis with the software Statistica v.7 (StatSoft Inc, http://www.statsoft.com).
We treated the frontal and parieto-occipito-temporal regions separately since these networks presented different numbers of edges. Then, Motor and Sensory SW values were submitted to two 3-way ANOVA models with Hemisphere (left, right) and Frequency band (delta, theta, alpha1, alpha2, beta1, beta2, and gamma) as within-subject factors and Group (Higher Fatigue, Lower Fatigue) as the between-subject factor. We checked preliminarily whether the SW value differed when estimated through the 23-channel versus the 64-channel EEG investigation by adding the EEG resolution (23 channels, 64 channels) between-subjects factor to this model. Once the absence of a main or interaction factor with EEG resolution had been confirmed, we omitted it from the following steps.
We tested Pearson’s linear correlations between the mFIS scores and the variables that differed in the two fatigue-dependent groups in the previous ANOVAs (Bonferroni corrected). To check for the selectivity of the effects with fatigue, we also calculated the correlations with depression levels as assessed by BDI.
In all cases, the significance level was set at P < .05 and only significant effects were reported as results.
Results
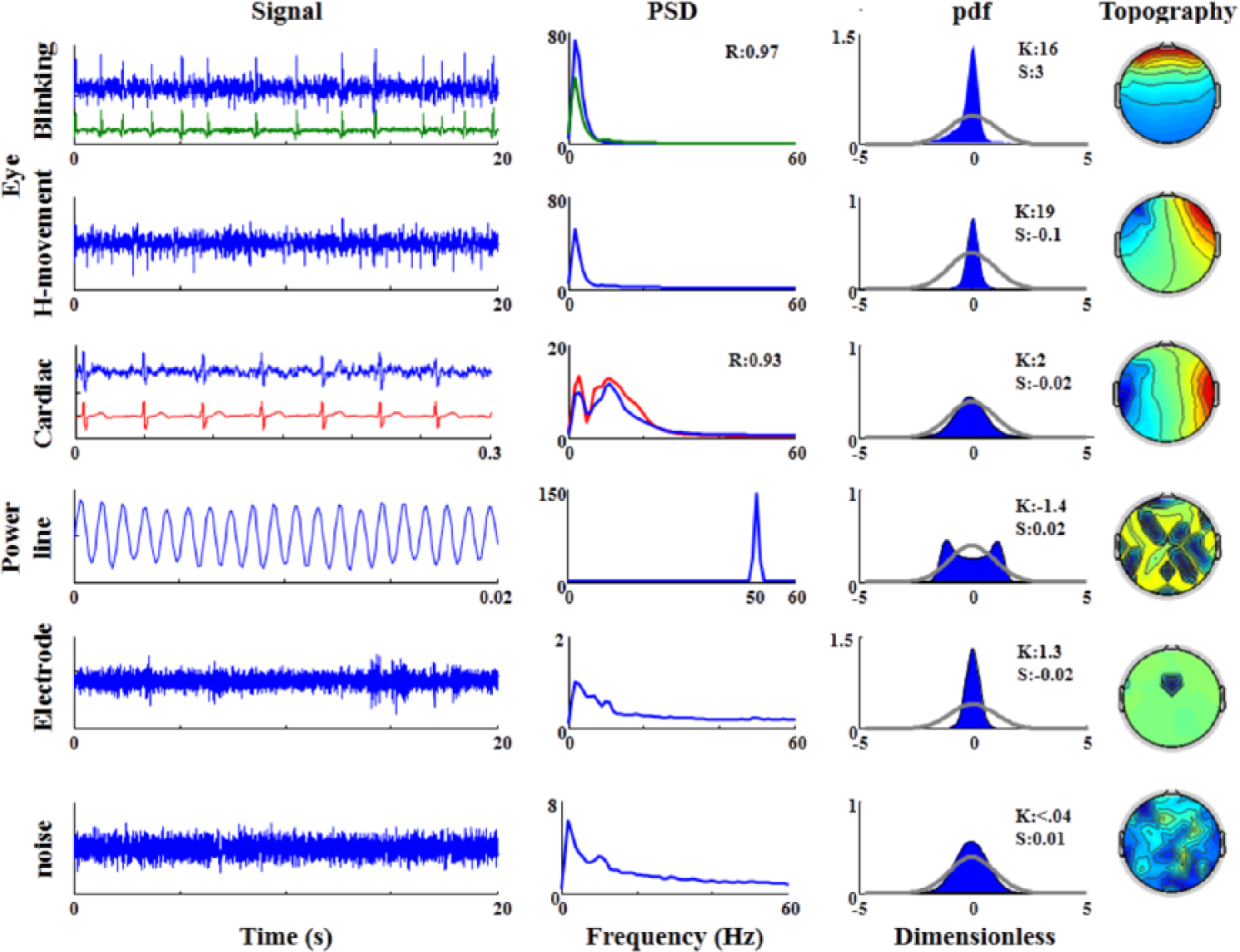
We recruited 27 MS patients (20 females; age range 22-44 years, mean = 37). Overall, they suffered from heterogeneous levels of fatigue, as total mFIS ranged between 2 and 69 (total scale range 0-84). Applying the mFIS cutoff of 35 (approximately half of our mFIS range), the 2 subgroups consisted of 17 patients who suffered from a higher level (mFIS>35) and 11 who experienced a lower level of fatigue (mFIS ≤35, Table 1).
Multiple Sclerosis Patient Demographic and Clinical Profile. a
Abbreviations: DMT, disease-modifying therapy; Rel, annual relapse rate; EDSS, Extended Disability Status Scale (of note a single patient in the lower fatigue group scored EDSS = 3.5); mFIS: modified Fatigue Impact Scale; _tot all items and _phys physical items; BDI, Beck Depression Inventory; F, female; M, male.
Mean and standard deviations (in parentheses) or median and range (in brackets) separately for the patients who complained less (Low-Fatigue) or more (High-Fatigue) fatigue and as a whole (last rows). In row “P,” the significance of intergroup differences in the means or nonparametric test (EDSS, BDI); in boldface are values differing in the 2 groups. Notably, the 2 groups differed only on fatigue. Clinical severity tended to be higher (P = .055) in patients who suffered less from fatigue.
SW values did not depend on the number of channels used in the EEG investigation, as indicated by absence of main or interaction effects with EEG resolution (23 channels, 64 channels) between-subjects factor (P > .200 consistently in the 4 regions).
The difference of Sensory networks’ SW between patients suffering from higher and lower fatigue levels depended on the hemisphere and frequency band of the oscillatory EEG activity, as indicated by the 3-way Hemisphere * Frequency Band * Group interaction effect: F(6, 96) = 1.90; P = .012; Figure 3. The Duncan post hoc testing showed that patients in the high-fatigue group presented higher values of small-worldness in comparison with the low-fatigue group in alpha2 and beta1 (P < .05) bands of the left hemisphere. No effect emerged in the right sensory network. No difference was detected between the left and right motor networks of the 2 fatigue-dependent groups.
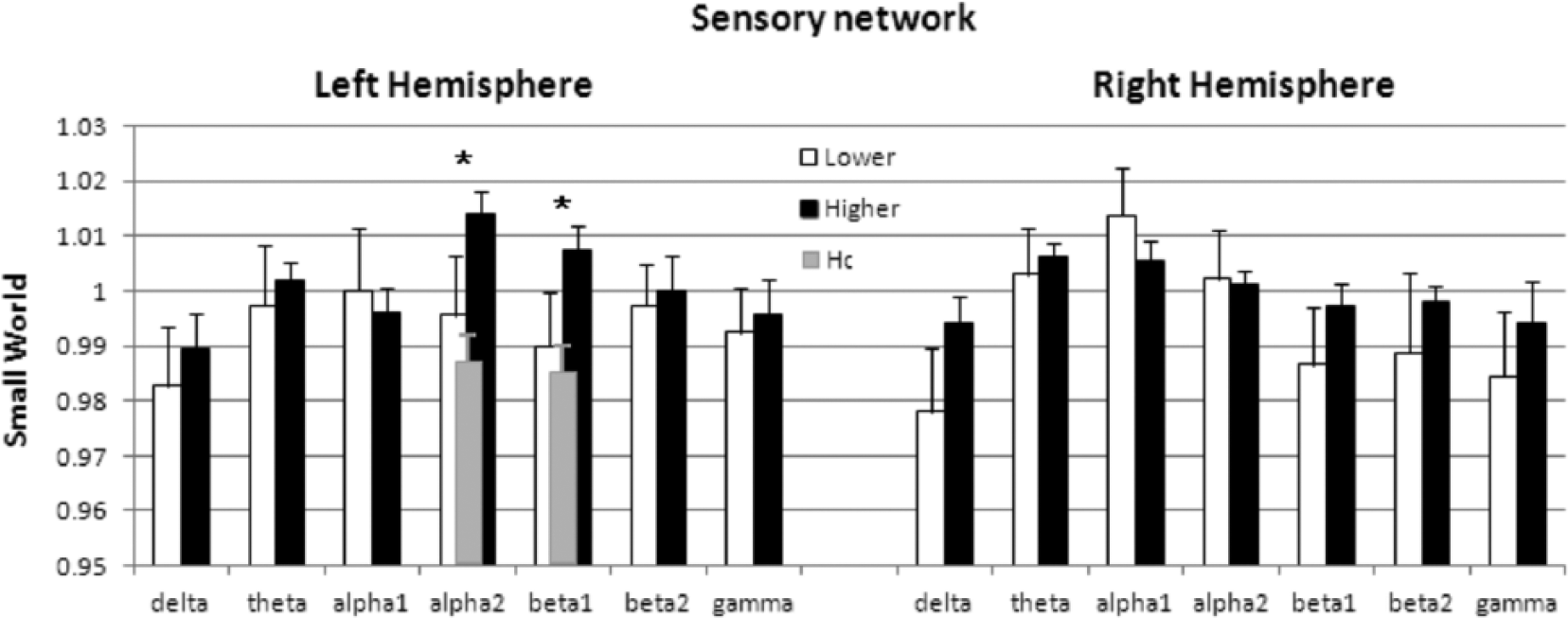
Small-world (SW) in Sensory regions. Mean and standard deviation (vertical segments) of SW in the Sensory network of the 2 hemispheres in dependence on the EEG frequency band and the level of fatigue (Low: white; High: black). The asterisks indicate fatigue-related groups’ difference. We present the values in healthy controls (HC) for SW in frequency bands differing between High- and Low-fatigue groups.
Correlation analyses for SW in alpha2 and beta1 bands showed mFIS an increase along with the small-world of beta1 oscillatory activity of the left Sensory network (r = 0.41 P = .030; Figure 4). No correlation with BDI was observed for SW in either alpha2 or beta1 bands (P > .200 consistently).
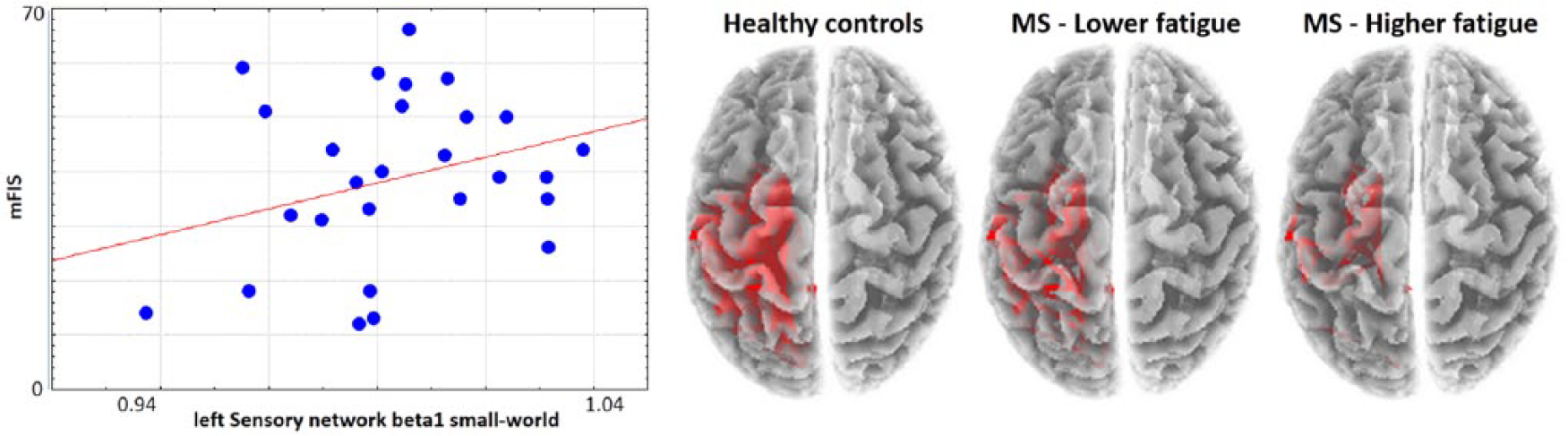
Relationship between left sensory network beta1 small-world (SW) and fatigue. (Left) Scatterplot of modified Fatigue Impact Scale (mFIS) and SW in beta1 EEG oscillatory activity of left Sensory network for all people with multiple sclerosis (MS). (Right) Schematic representation of the left Sensory network with LagR connectors in beta1 (arbitrary threshold 0.28) among the Broadmann areas within the left parieto-temporo-occipital region, distinct for healthy controls, Lower and Higher Fatigue groups.
To develop a deeper understanding of the direction of changes due to fatigue, SW values of the left Sensory network in alpha2 and beta1 bands of MS patients were compared with those of the control group. The corresponding ANOVA included Frequency band (alpha2, beta1) as within-subject factors and Group (Higher Fatigue, Lower Fatigue, Healthy Controls) as between-subjects factor. The results of the low-fatigue group were intermediate between the healthy and high-fatigue groups, as indicated by post hoc comparisons generating the Group effect: F(2, 36) = 4.80; P = .0146. In fact, in alpha2 band SW was HC: 0.989 ± 0.005, Lower Fatigue: 0.995 ± 0.011, Higher Fatigue: 1.014 ± 0.004; and in beta1 band: HC: 0.985± 0.005, Lower Fatigue: 0.989 ± 0.009, Higher Fatigue: 1.007 ± 0.004; Figure 3.
Discussion
The main result of the present EEG study is that functional connectivity changes of the left sensory cortical network at rest occur in MS fatigue, mediated by beta band oscillatory activity.
The human brain is a complex and interconnected dynamical system, as demonstrated by recent studies on the intrinsic topological properties underlying network organization. The possibility of adopting an appropriate mathematical approach notably improves the potential for detecting relevant features in real complex networks. In this sense, graph theory can aid the analysis of connectivity patterns estimated from EEG recordings. Here, MS fatigue increased in association with an increase of small world values of the brain parieto-occipito-temporal network of the dominant hemisphere. This probably reflects an impaired local and global connectivity balance of the area. Previous literature has shown that rather than occurring in the whole-brain, primary MS-fatigue-related alterations emerge in specific brain regions.36,37 Indeed, a higher lesion load in fatigued patients affected the network involving the internal capsule, the peritrigonal white matter, the posterior parietal cortex, and the right parieto-temporal and left frontal lobes. 38 We observed that MS fatigue increases in association with functional, more than structural, alterations, both as functional connectivity between the brain and muscles and as interhemispheric asymmetries of sensorimotor homologous areas.15,16 Consistently, in 44 MS patients, fatigue severity did not correlate with grey matter density, white matter integrity or basal ganglia volumes. In contrast, fatigue increased with the reduction of functional connectivity of basal ganglia nuclei with multiple cortical and subcortical structures. 39 In our study, the functional connectivity at rest within the left post-central sensory network altered in parallel with fatigue symptoms, strengthening the idea that region-specific and homologous region asymmetric functional dysfunctions occur in MS fatigue. Notably, in another fMRI study, 19 the only brain activation positively correlated to experienced fatigue was the left posterior parietal cortex. In yet another study, 16 it was found that fatigue increased along with the functional interhemispheric imbalance of sensorimotor homologous areas EEG activities, both at rest and during movement. The left dominant sensorimotor areas generated higher power than the right nondominant ones, conceivably corresponding to an impoverished local organization of the dominant hand representation. This is supported by previous data in healthy control groups, where we have proven that the dominant hand representation (in the left hemisphere) has a lower EEG power at rest than the nondominant one. 40 In MS patients, there is a distortion 3 in the intracortical connectivity index specific of the left dominant hemisphere coding for sensorimotor dexterity. 14 Moreover, the increase of power and interhemispheric imbalances are related to an increase in aging healthy people in the same direction as was observed in these MS patients. This may be a sign of reduced functional organization in the region of the hand’s sensorimotor control. 41 The present finding of the impaired local and global connectivity balance in parallel with fatigue of the left sensory network supports the notion of a local impairment that is more evident in the dominant sensorimotor control regions; clear indication of a parietal role in the underlying mechanism. These convergent indications of parietal implication and interhemispheric asymmetries in MS fatigue inject further impetus for the continued investigation of neuromodulation intervention as a method for alleviating this debilitating symptom. 42
We observed that functional connectivity in the resting state with eyes open, changes in tandem with fatigue symptoms in the beta band. This was revealed through cortical EEG sources network graph-theory analysis. As this range of oscillations is typical of cortico-spinal communication, this finding strengthens the notion of sensorimotor network involvement in MS fatigue. In particular, we found that with increasing fatigue, the left parietal SW value increases. The literature indicates that SW increases in brain networks are typically due to a higher reduction of the denominator (path length) than of the numerator (clustering). The more evident impairment of integration than segregation, reflected in the SW increase, suggests the impairment of beta-mediated sensorimotor communication.
Functional brain connectivity simultaneously reconciles the opposing demands of functional integration and segregation. The small-world index reflects the balance of functionally specialized (segregated) modules with a robust number of intermodular (integrating) links. 43 We observed that fatigue increases with the progressive loss of small-world organization in sensory areas. This is in line with a previous observation, in which the intracortical connectivity index specific to the left dominant hemisphere coding for sensorimotor dexterity 44 in MS patients appeared to be distorted. 3 The network alterations found in the present study spreads throughout the primary sensory areas. It includes in particular the posterior parietal cortex, which receives visual information from primary visual areas, somatosensory information from the primary somatosensory area, and motor information from primary motor, supplementary motor, and premotor areas. Thus, posterior parietal cortex is a crucial node of the cortical neural network subservient to integration of information regarding the intra- and extrapersonal space for coordinated limb movements. 45
The increasing parietal prevalence of the connectivity alteration in parallel with increasing fatigue, strengthens the notion that perception plays a pivotal role in fatigue generation. In particular, this finding supports the hypothesis posed in chronic fatigue syndrome that prolonged perception of fatigue reduces motivation, and opposes the competing hypothesis in which decreasing motivational drive increases the perception of fatigue. 46
A limitation of our study relates to the small size of the population investigated. We were able to observe the reported phenomena in the subgroup of the first 17 patients recruited, and the confirmation observed in the larger population allows for acceptable confidence in the reported results. Nevertheless, study of the bigger population will strengthen understanding about the connectivity-related alterations occurring with increases in MS fatigue. A second limitation is that fatigue symptoms were assessed using the mFIS self-report questionnaire. All our patients completed the questionnaire by themselves, scoring how much fatigue affects their lives in terms of physical, cognitive, and psychosocial functioning. We also collected mFIS in stable conditions for 10 patients twice and observed a small mFIS divergence in the 2 scorings (mean difference 0.1 ± 1.9, intraclass correlation 0.96). 42 Nevertheless, all studies investigating these symptoms are subject to a lack of the ability to quantify patients’ suffering via entirely objective measures. A third limitation of our study is the separate assessment of connectivity parameters in the 2 hemispheres. Although it would be very interesting to assess the functional connectivity between the homologous regions of the 2 hemispheres, this was not possible via the graph-theory approach, which aims to quantify the balance between local clustering and global links among all network nodes. Another limitation is that we were only able to evaluate our patients via EEG and morphological MRI (data under study), when a study by concurrent EEG and fMRI would allow a much better assessment of involved neuronal pools.
Conclusion
MS fatigue increased along with regional-specific alterations of the sensorimotor system resting state functional connectivity as explored with EEG–graph theory analysis. Specifically, the left dominant sensory regions suffered from a reduced networking organization. This finding suggests that neuromodulation may be able to modify the networking of left postcentral sensory regions and, in doing so, provide a treatment for relieving fatigue in patients with MS.
Footnotes
Acknowledgements
The authors wish to thank Maria Filippi, Anna Ghazaryan, and Giancarlo Zito for clinical and scientific collaboration, and Matilde Ercolani who assisted with electrophysiological acquisition. We are sincerely grateful to all patients for the time and cooperation required to participate (devoted to participation) in the study. Dr Francesca Miraglia participated in this study as part of the completion of her PhD program at the Doctoral School in Neuroscience, Department of Neuroscience, Catholic University of Rome, Italy.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by: (1) FISM–Fondazione Italiana Sclerosi Multipla–Cod.2014/R [FaReMuS CuNeH], (2) MIUR Prot. 2010SH7H3F “Functional connectivity and neuroplasticity in physiological and pathological aging [ConnAge],” (3) PNR-CNR Aging Program 2012-2018, and (4) Italian Ministry of Health for Institutional Research (ricerca corrente).