Abstract
Background.
Pelvic floor dysfunction (PFD) is a frequent yet underrecognized complication of neurological disorders such as multiple sclerosis (MS), Parkinson’s disease (PD), and stroke. Its multifactorial pathophysiology involves complex neural mechanisms affecting bladder, bowel, and sexual function, often resulting in decreased quality of life and psychosocial distress.
Objective.
This focused mini-review aims to synthesize current evidence on the pathophysiology, clinical features, and rehabilitation approaches for neurogenic PFD in major neurological conditions.
Methods.
A targeted literature search was performed in PubMed, Scopus, and Web of Science databases to identify clinical and experimental studies published between 1990 and 2025 addressing urinary and PFD in neurological populations. Special attention was given to rehabilitation-based interventions such as pelvic floor muscle training (PFMT), biofeedback, neuromuscular electrical stimulation (NMES), and percutaneous tibial nerve stimulation.
Findings.
Neurogenic PFD is highly prevalent, with up to 90% of MS patients, 60% of PD patients, and nearly half of stroke survivors experiencing urinary symptoms. Conservative rehabilitation, particularly PFMT combined with biofeedback and NMES, improves muscle function, reduces incontinence frequency, and enhances quality of life. However, standardized rehabilitation protocols are lacking, and adherence remains a major barrier.
Conclusion.
Evidence supports a multidisciplinary rehabilitation approach integrating PFMT and adjunct modalities for neurogenic PFD. Further large-scale randomized studies are required to establish standardized, evidence-based clinical guidelines.
Introduction
Neurogenic Pelvic Floor and Lower Urinary Tract Dysfunction
Disorders of the central nervous system (CNS), through various neurological etiologies, can disrupt normal bladder and pelvic floor function. Such dysfunctions negatively affect daily living activities, social participation, and quality of life and may lead to long-term urological complications.1,2 Normal lower urinary tract function relies on the coordinated activity of parasympathetic, sympathetic, and somatic neural pathways, ensuring low-pressure bladder filling and voluntary, timely emptying. Effective voiding depends on intact communication between supraspinal centers—particularly the pontine micturition center—and the sacral spinal cord. 3
Disruption of this complex neural network results in a spectrum of lower urinary tract symptoms (LUTS), which are classically categorized as storage, voiding, and post-micturition symptoms. Storage symptoms include increased urinary frequency, urgency, nocturia, urinary incontinence, and overactive bladder (OAB), most commonly associated with detrusor overactivity. Voiding symptoms, such as weak stream, hesitancy, and straining, are typically related to impaired bladder emptying or outlet dysfunction, while post-micturition symptoms include incomplete emptying and post-void dribbling.4-6
Although neurogenic lower urinary tract dysfunction has traditionally been conceptualized primarily within urological frameworks, growing evidence highlights the central role of the pelvic floor as an integral component of the neuromuscular continence system. In neurological disorders, disruption of central and peripheral control mechanisms affects not only bladder function but also pelvic floor muscle coordination, sensation, and load transfer capacity. This underscores the growing need for a multidisciplinary rehabilitation-oriented perspective that bridges pathophysiology with targeted physiotherapy and pelvic floor rehabilitation strategies.
Multiple Sclerosis (MS)
In MS, neurogenic pelvic floor dysfunction (PFD) represents a multidimensional rehabilitation challenge, as central demyelination affects not only bladder control but also bowel continence, sexual function, and pelvic floor muscle coordination, necessitating an integrated physiotherapy-oriented approach. Accordingly, MS is a multifactorial demyelinating disease of the CNS, characterized by a broad spectrum of symptoms and clinical findings. One of the most disabling manifestations is sphincter dysfunction, affecting urination, defecation, and sexual function. 7 A large North American survey including 14 268 individuals with MS reported that approximately one-third experienced moderate to severe pelvic floor symptoms, which negatively impacted daily activity performance, mobility, and physical aspects of quality of life; the prevalence of dysfunction was 41% for bladder dysfunction, 30% for bowel dysfunction, and 42% for sexual dysfunction.7,8 Demyelinating plaques in MS frequently localize to the cervical spinal cord, particularly within the lateral corticospinal and reticulospinal tracts, which play a key role in detrusor and external urethral sphincter innervation, thereby contributing to urinary symptom development. 9 Intracranial plaques are reported in 60% to 90% of cases, while suprasacral spinal cord involvement is observed in up to 80%, commonly resulting in detrusor–sphincter dyssynergia and detrusor overactivity. In contrast, sacral involvement, present in approximately 20% of patients, is associated with impaired detrusor contractions and underactive bladder function. 9
During the disease course, nearly two-thirds of individuals with MS experience moderate to severe urinary problems. LUTS are highly prevalent, with reported rates ranging from 32% to 96%, while OAB symptoms are observed in 60% to 80% of patients. Overall, 50% to 90% of people with MS develop urinary complaints, and after 10 years of disease duration, up to 90% present with some form of urinary dysfunction.10,11 Among these, urgency and urge incontinence are the most commonly reported symptoms. OAB, characterized by urgency, frequency, and urge incontinence, has been documented in 37% to 99% of MS cases, with approximately half of patients exhibiting a combination of OAB and voiding dysfunction, reflecting the complex nature of neurogenic bladder involvement in MS.12,13
Although fewer studies have specifically examined stress urinary incontinence (SUI) in MS, reported prevalence rates range from 16% to 55.9%. 14 These studies suggest that women with SUI tend to be older, are more likely to have relapsing–remitting MS, have higher body mass index, and demonstrate higher mean maximum urinary flow compared with those without SUI. Importantly, Ghezzi et al 15 emphasized that even in asymptomatic cases of sphincter dysfunction, patients with pyramidal involvement, longer disease duration, and higher Expanded Disability Status Scale (EDSS) scores remain at increased risk for urinary symptoms and therefore require close monitoring. Despite the high frequency and burden of urinary symptoms in MS, these manifestations remain markedly undertreated. Bladder and bowel symptoms are consistently reported as among the most distressing aspects of the disease, substantially impairing health-related quality of life, yet available evidence indicates a lack of comprehensive data on symptom prevalence and highlights that urinary dysfunction is often insufficiently addressed in routine clinical practice, where treatment strategies primarily focus on disease progression rather than symptomatic management. 16
Parkinson’s Disease (PD)
In PD, PFD reflects impaired neuromuscular regulation and autonomic control, influencing urinary, bowel, and sexual functions and highlighting the importance of rehabilitation strategies targeting pelvic floor muscle timing, coordination, and central motor control. In this context, PD is a progressive neurodegenerative disorder of the extrapyramidal system. Globally, it affects approximately 6.2 million individuals, corresponding to nearly 1% of the population over 60 years of age, and its prevalence is expected to increase to almost 12 million cases by 2040, making it the fastest-growing neurodegenerative condition worldwide. In addition to cardinal motor symptoms such as tremor, rigidity, bradykinesia, akinesia, and postural instability, PD is characterized by a wide range of non-motor manifestations, including sensory, autonomic, neuropsychiatric, and sleep-related dysfunctions. 17 Although less frequently investigated, bowel and sexual dysfunction have also been reported in PD and may further contribute to pelvic floor dyscoordination and reduced quality of life.
Urinary dysfunction represents one of the most common autonomic manifestations of PD, with OAB symptoms such as urgency and frequency being particularly prevalent. These symptoms are associated with older age, reduced quality of life, dysautonomia, cognitive impairment, and an increased risk of falls. In contrast to bowel dysfunction, urinary symptoms in PD typically emerge as the disease progresses rather than preceding the onset of motor symptoms. 17
The predominant pathophysiological mechanism underlying urinary dysfunction in PD is impaired dopaminergic regulation of the micturition reflex. Dopaminergic neurons play a central role in bladder control, whereby D1 receptors exert inhibitory effects on micturition, while D2 receptors facilitate voiding. Degeneration of neurons in the substantia nigra pars compacta results in loss of D1-mediated inhibitory control, leading to detrusor overactivity. 18 In addition, α-synuclein pathology—the neuropathological hallmark of PD—has been identified in the pontine micturition center, sacral spinal cord, pelvic plexus, and genitourinary tract, providing anatomical evidence for widespread involvement of autonomic pathways. 19
Epidemiological studies indicate that 57% to 83% of individuals with PD experience storage symptoms, while 17% to 27% report voiding symptoms. Nocturia is the most frequently reported complaint, affecting up to 60% of patients, followed by urgency in 33% to 54% and urinary frequency in 16% to 36%, most commonly due to detrusor overactivity characterized by early, involuntary bladder contractions. Obstructive voiding symptoms, including hesitancy, weak urinary stream, and intermittency, are reported in approximately 27% of patients. In many cases, detrusor underactivity contributes to impaired urinary flow, often resulting from delayed relaxation of the urethral sphincter and pelvic floor muscles—a phenomenon referred to as sphincter bradykinesia.17,20
Stroke
Following stroke, PFD emerges as a consequence of disrupted suprapontine and subcortical control, frequently coexisting with bowel and sexual dysfunction and requiring rehabilitation approaches that integrate continence training with broader neurorehabilitation goals. Against this background, stroke is defined as a rapidly developing clinical syndrome of focal cerebral dysfunction of presumed vascular origin and remains one of the leading causes of death and long-term disability worldwide, accounting for an estimated 5.54 million deaths annually. Depending on the size and anatomical location of the lesion, stroke survivors may present with a wide range of motor, sensory, and cognitive impairments. As the incidence of stroke increases markedly with advancing age, urinary dysfunction has become an increasingly important clinical concern, particularly in older populations. 21
Urinary dysfunction following stroke is closely related to the location and extent of cerebral injury. Lesions involving the anteromedial frontal lobe, periventricular white matter, and putamen are most commonly associated with bladder dysfunction. Urodynamic studies have demonstrated that the bladder is typically areflexic during the acute post-stroke phase; however, as recovery progresses, detrusor hyperactivity and urge incontinence frequently develop. Nevertheless, detrusor hypoactivity accompanied by urinary retention has also been documented in both ischemic and hemorrhagic stroke subtypes.22,23
During acute hospitalization, the incidence of urinary incontinence ranges between 28% and 79%, and approximately 40% of stroke survivors report urinary incontinence within the first week following the event. In a cohort study of 699 previously continent patients, incontinence was observed in 35% of individuals at day 7 post-stroke. Longitudinal follow-up data further demonstrate that 44% of patients continue to experience urinary incontinence at 3 months and 38% at 12 months, with urge incontinence representing the most common subtype. 23 , 24 Importantly, persistent urinary incontinence during the first year after stroke has been identified as a strong predictor of increased mortality, poorer functional recovery, and higher rates of rehospitalization. 25 The patterns of lower urinary tract dysfunction observed across major neurological disorders reflect distinct pathophysiological mechanisms, as summarized in Table 1. While bowel and sexual dysfunctions are integral components of the broader pelvic floor pathology in neurological conditions, the present focused narrative review primarily centers on lower urinary tract dysfunction and its rehabilitation.
Overview of Pathophysiology and Clinical Characteristics of Lower Urinary Tract Dysfunction in Major Neurological Disorders

Abbreviations: DO, detrusor overactivity; DSD, detrusor–sphincter dyssynergia; DU, detrusor underactivity; UUI, urgency urinary incontinence.
Methods
This manuscript was developed as a focused narrative mini-review rather than a systematic review. A targeted literature search was conducted in PubMed, Scopus, and Web of Science to identify relevant studies published between January 1990 and March 2025 addressing PFD in neurological populations. The search combined Medical Subject Headings terms and free-text keywords related to neurological conditions, PFD, and rehabilitation-based interventions, including physiotherapy, pelvic floor muscle training (PFMT), biofeedback, neuromuscular electrical stimulation (NMES), and neuromodulation approaches. Studies were selected based on clinical relevance and contribution to understanding pathophysiology and rehabilitation principles and were synthesized narratively due to heterogeneity in study designs and outcomes.
Assessment Methods in Urinary Dysfunction
In neurogenic PFD, assessment is essential not only for diagnostic classification but, more importantly, to guide individualized rehabilitation planning and monitor functional outcomes. Given the heterogeneity of neurological conditions and symptom presentations, a multimodal assessment approach is required to capture bladder storage and voiding dysfunction alongside pelvic floor muscle function and patient-reported symptom burden. A comprehensive evaluation of urinary dysfunction in neurological populations should integrate complementary methods to capture storage and voiding abnormalities in a standardized manner.10,26 The process begins with a detailed medical history and a focused physical examination, followed by targeted tests such as the Q-tip test to quantify urethral hypermobility. Patient-reported records, including a voiding diary, elucidate bladder capacity and habitual voiding patterns, while the pad test offers a noninvasive estimate of leakage volume and severity. Assessment of pelvic floor muscles—with explicit attention to spasticity in neurological patients—adds crucial information on neuromuscular contributors. Clinical evaluation of pelvic floor muscle function may include digital palpation-based scales such as the Modified Oxford Scale, which is commonly incorporated within structured assessment frameworks such as the PERFECT scheme. 27 Although the Modified Oxford Scale has demonstrated acceptable inter-rater reliability in women with PFD,28,29 its interpretability in neurological populations may be limited by impaired voluntary motor control, spasticity, altered muscle tone, and reduced sensitivity to functional change. Therefore, it is best applied as an adjunct to instrumental assessments and patient-reported outcome measures rather than as a stand-alone evaluation tool in neurogenic PFD. Validated questionnaires further characterize symptom burden and impact, for example the Actionable Bladder Symptom Screening Tool and the Neurogenic Bladder Symptom Score for MS. Structural and functional insights are provided by lower urinary tract imaging to identify capacity, pelvic pathology, and urethrovesical anomalies, and by urodynamic studies, which directly interrogate detrusor storage and outlet function to refine diagnosis and guide management. Together, these assessment components provide a functional framework to inform rehabilitation strategies rather than serving as isolated diagnostic tools.
Treatment
Based on current rehabilitation principles, a stepwise clinical approach to the assessment and management of neurogenic PFD is illustrated in Figure 1. In neurological disorders, physiotherapy and rehabilitation are inherently multidisciplinary and encompass a broad range of interventions. 30 A rehabilitation program may be devised using pharmacological treatment; catheter-assisted and catheter-free bladder emptying methods; bladder training; lifestyle modifications; pelvic floor muscle exercises; electrical stimulation; and biofeedback—applied either as standalone modalities or in combination.2,30
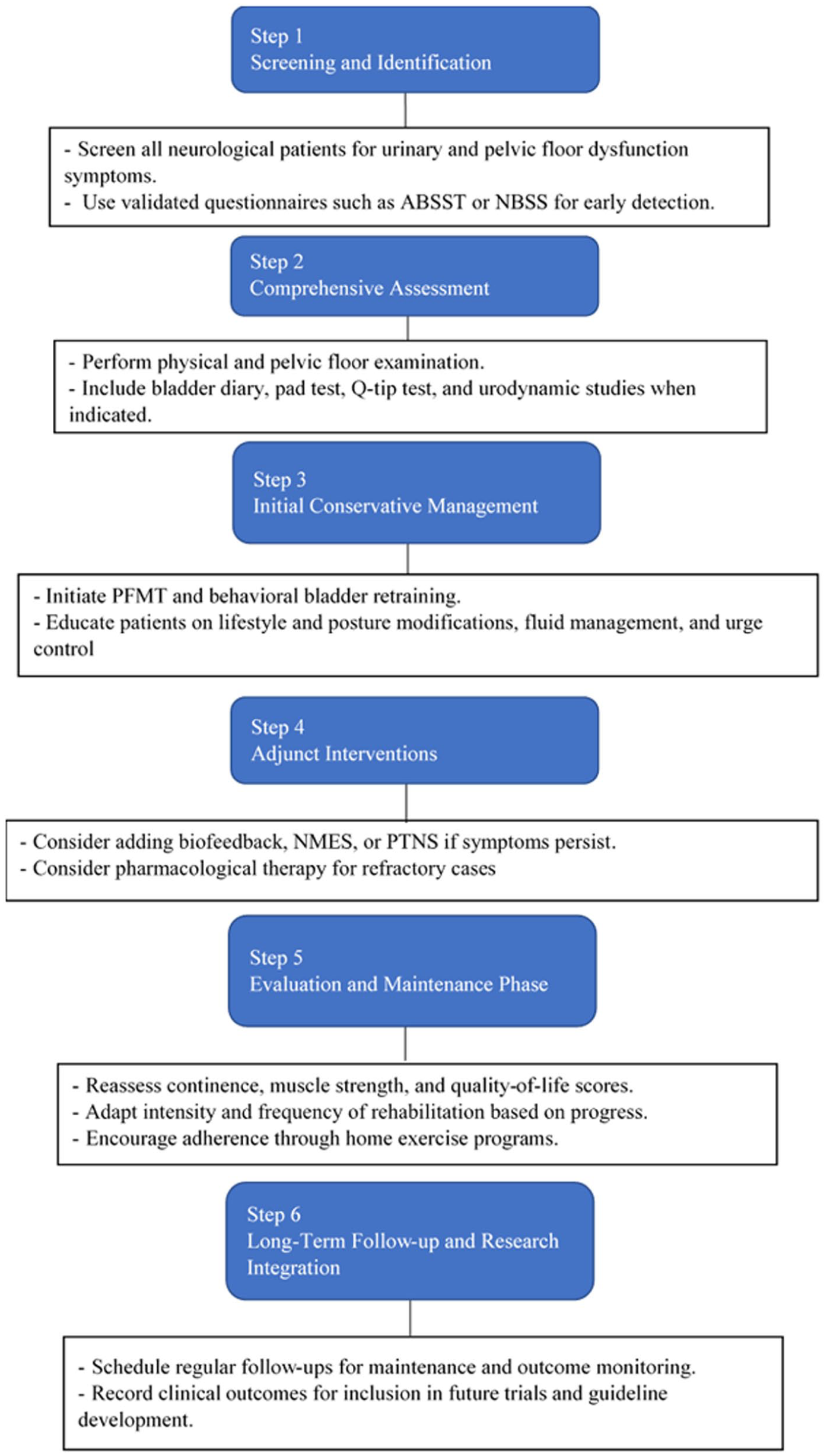
Rehabilitation flow algorithm for neurogenic pelvic floor dysfunction.
Review of the literature indicates that for PFDs associated with neurological disorders, pelvic floor rehabilitation primarily involving PFMT is generally recommended as a first-line conservative approach.31-33
Pelvic Floor Muscle Training
PFMT aims at the functional restoration of perineal muscles and is underpinned by 2 complementary theoretical frameworks: the behavioral theory, which focuses on training patients to consciously and anticipatorily activate the pelvic floor in response to increases in intra-abdominal pressure, and the neuromuscular adaptation theory, which emphasizes progressive strength training-induced hypertrophy and increased muscle stiffness to enhance structural support, with additional contributions from synergistic abdominal muscle activation. 34
Although studies on PFD management in neurological patients have not followed standardized protocols, the majority are based on a conservative approach emphasizing pelvic floor-focused programs involving PFMT, biofeedback, NMES, and posterior tibial nerve stimulation (PTNS). 35 When PFMT is combined with instrumental techniques such as biofeedback and electrical stimulation, this is referred to as Pelvic Floor Muscle Rehabilitation (PFMR). PFMR is most frequently applied in MS but also in other neurological disorders. However, there is currently no standardized protocol regarding duration or treatment approach.
Systematic reviews generally support conservative interventions—particularly PFMT—as first-line therapy for urinary incontinence. In individuals with neurological disease-related urinary symptoms, PFMT has been shown to reduce the frequency and severity of incontinence, improve pelvic floor muscle function, and enhance quality of life. 36 Recent systematic reviews further support these findings, showing that PFMT—either alone or combined with biofeedback or electrostimulation—reduces urinary incontinence severity and improves pelvic floor muscle strength and quality of life in both general and neurological populations.33,37 In people with MS, PFMT-based programs have been shown to moderately decrease urine leakage and OAB symptoms and to improve sexual function and health-related quality of life.33,38,39
Studies reported that PFMT in stroke patients improved pelvic floor muscle contraction and dynamic endurance, while reducing resting activity, daily frequency, and urinary symptoms.40-42 Despite heterogeneity of protocols, PFMR has been associated with improvements in the severity of urinary symptoms, quality of life, anxiety and depression scores, and sexual dysfunction in several clinical studies. 43 In women with MS, PFMR appears to result in greater improvements compared with placebo, including reductions in storage and voiding symptoms, pad weight, urgency frequency, and episodes of urge urinary incontinence. 44 Questionnaire-based evaluations further demonstrated reductions in pad usage and nocturia frequency, along with improvements in muscle strength, endurance, rapid pelvic floor contractions, and reductions in anxiety and depression.
Electrical Stimulation
Therapeutic electrical stimulation can be delivered through superficial, intravaginal, or anal modalities. Its physiological rationale is the activation of pudendal afferent nerve fibers, which in turn elicits contractions of the pelvic floor musculature and the striated urethral sphincter. The resultant increase in urethral closure pressure may enhance outlet resistance during storage, thereby contributing to a reduction in urinary incontinence episodes in appropriately selected patients.45-48 Clinical studies indicate that NMES, when used alongside PFMT and/or biofeedback, can further reduce urinary incontinence episodes, improve pelvic floor muscle strength, and enhance quality of life, with emerging data also suggesting benefits for sexual function and patient-reported satisfaction.48-50
Biofeedback
Biofeedback aims to enhance proprioceptive awareness of the pelvic floor and to train the accurate execution of targeted contraction–relaxation patterns. In neurological populations, it is employed both to teach patients to volitionally contract the pelvic floor in response to urgency—thereby delaying micturition—and to cultivate relaxation awareness that facilitates efficient bladder emptying. 38
Combined Modalities
Across interventions for neurogenic lower urinary tract dysfunction, adjunctive neuromodulation appears to augment the effects of conservative therapy in selected neurological populations. In MS, combining PFMT with NMES has been associated with greater reductions in OAB symptoms, stronger perineal contractions, and greater improvements in quality-of-life outcomes than PFMT alone. 51 When biofeedback is paired with PFMT, the addition of NMES further improves objective continence outcomes, with larger effects on pad-test totals and mean leakage episodes (reported ~85% vs ~47% improvement). Notably, the durability of benefit may be limited without continued therapy: while approximately 85% of MS patients became symptom-free during NMES, only ~18% maintained symptom-free status 3 months after cessation, underscoring the potential need for chronic or maintenance stimulation. 50 Complementarily, PTNS has been associated with reductions in OAB symptoms, enhancements in health-related quality of life, and favorable shifts in urodynamic parameters.45,52 These observations are consistent with a recent systematic review and meta-analysis of bladder rehabilitation in MS, which demonstrated that both PTNS and PFMT significantly improve neurogenic detrusor overactivity and LUTS, supporting their role as key components of conservative management in MS-related neurogenic lower urinary tract dysfunction. 53 A more recent narrative review also highlights PTNS as a promising neuromodulatory option for detrusor overactivity in MS, while emphasizing the need for standardized stimulation protocols and longer-term follow-up data. 39 Recent reviews of neurogenic OAB across multiple neurological conditions similarly conclude that conservative and rehabilitation-based strategies—including PFMT, neuromodulation, and adjunctive physical therapies—play a central role alongside pharmacological treatment.54,55 For clinicians, an overview of commonly applied rehabilitation-based interventions and their reported clinical outcomes across neurological populations is summarized in Table 2.
Rehabilitation-Based Interventions and Reported Outcomes in Neurogenic Lower Urinary Tract Dysfunction.
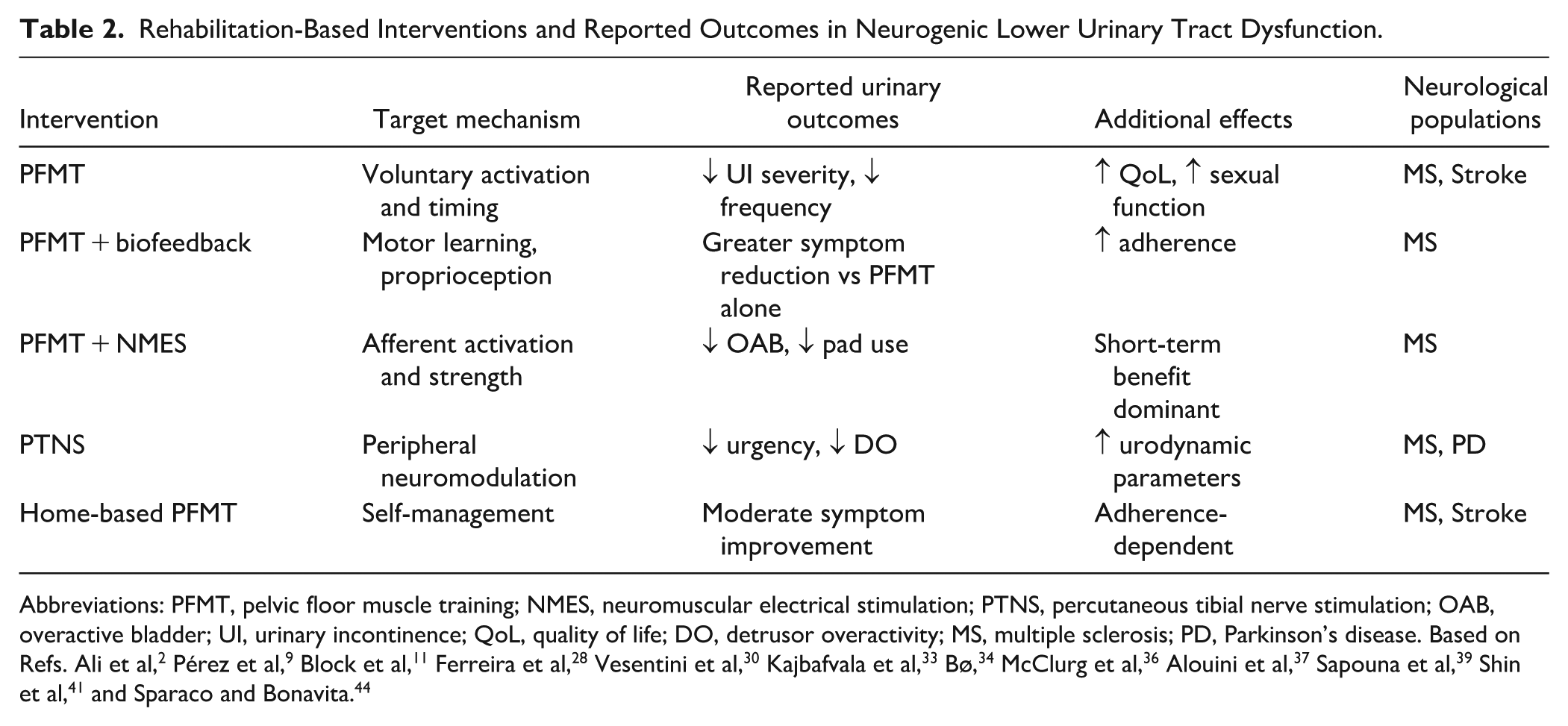
Abbreviations: PFMT, pelvic floor muscle training; NMES, neuromuscular electrical stimulation; PTNS, percutaneous tibial nerve stimulation; OAB, overactive bladder; UI, urinary incontinence; QoL, quality of life; DO, detrusor overactivity; MS, multiple sclerosis; PD, Parkinson’s disease. Based on Refs. Ali et al, 2 Pérez et al, 9 Block et al, 11 Ferreira et al, 28 Vesentini et al, 30 Kajbafvala et al, 33 Bø, 34 McClurg et al, 36 Alouini et al, 37 Sapouna et al, 39 Shin et al, 41 and Sparaco and Bonavita. 44
Clinical Messages
PFD is a frequent yet often underrecognized problem in neurological disorders such as MS, PD, and stroke, significantly affecting quality of life and functional independence.
Early screening and multidisciplinary rehabilitation, including PFMT, behavioral strategies, and bladder training, can substantially reduce symptom burden and improve continence outcomes.
Combined modalities, such as biofeedback, NMES, and PTNS, enhance treatment efficacy and promote neuromuscular re-education, but standardized protocols remain lacking.
Future research should focus on developing evidence-based clinical guidelines, improving adherence and long-term maintenance strategies, and integrating pelvic floor rehabilitation into routine neurorehabilitation practice.
Limitations
This narrative mini-review has several limitations that should be acknowledged. First, due to its narrative design, the review does not follow a systematic methodology, and relevant studies may have been selectively included based on clinical and thematic relevance rather than exhaustive retrieval. Second, the available literature on neurogenic PFD is characterized by substantial heterogeneity in neurological populations, assessment tools, outcome measures, and rehabilitation protocols, limiting direct comparison across studies. Third, many of the studies included involve small sample sizes and short follow-up periods, which restrict the generalizability of findings and preclude firm conclusions regarding long-term effectiveness. Finally, the lack of standardized rehabilitation protocols and the relative underrepresentation of bowel and sexual outcomes in the literature may further constrain the interpretation and applicability of current evidence
Conclusion
PFD is highly prevalent across major neurological disorders, including MS, PD, and stroke, and represents a significant yet often underrecognized contributor to reduced quality of life and functional independence. Current evidence supports conservative, rehabilitation-based interventions—particularly PFMT—as a cornerstone of management, with adjunctive modalities such as biofeedback, NMES, and PTNS offering additional benefits in selected populations.
However, considerable heterogeneity in intervention protocols, outcome measures, and follow-up durations limits direct comparison across studies and precludes definitive conclusions regarding optimal treatment strategies. Future research should prioritize well-designed randomized controlled trials that directly compare combined rehabilitation approaches versus PFMT alone, particularly in populations such as PD and MS. In addition, studies evaluating long-term maintenance strategies, home-based and digitally supported pelvic floor rehabilitation programs, and the integration of bowel and sexual outcomes alongside urinary measures are critically needed. Advancing standardized, multidisciplinary rehabilitation frameworks will be essential to improving continence outcomes and embedding pelvic floor rehabilitation within routine neurorehabilitation practice.
Beyond lower urinary tract outcomes, emerging evidence underscores the necessity of addressing the pelvic floor as an integrated functional system within neurorehabilitation. Although lower urinary tract dysfunction is often the primary focus in neuro-urological rehabilitation, a truly multidisciplinary approach must address the pelvic floor as a functional unit. Neurogenic bowel dysfunction, particularly in conditions such as MS and spinal cord injury (SCI), is intrinsically linked to urinary symptoms and requires condition-specific management strategies.56,57 Furthermore, sexual dysfunction remains a neglected yet vital determinant of quality of life in neurological populations, warranting targeted rehabilitation approaches beyond pharmacological management. 58
While this review primarily emphasized MS, PD, and stroke, the complex rehabilitation needs of individuals with SCI—characterized by distinct pathophysiology—necessitate separate evaluation frameworks and intervention strategies.59,60 Looking forward, the integration of emerging neuromodulatory technologies such as repetitive transcranial magnetic stimulation offers promising avenues for influencing central control of pelvic floor function. 61 Ultimately, alignment with contemporary evidence-based frameworks, including the 2024 European Association of Urology Guidelines on Neuro-Urology, 62 will be essential to standardize rehabilitation pathways and optimize clinical outcomes.
Footnotes
Acknowledgements
The authors gratefully acknowledge the contributions of the research assistants and clinical colleagues from the Department of Physiotherapy and Rehabilitation, Recep Tayyip Erdoğan University, for their valuable insights and support during the development of this manuscript. Editorial assistance and language review were performed to improve clarity and fluency of the text. The authors are solely responsible for the conception, design, data interpretation, and writing of this manuscript. Language editing and academic writing support were utilized to improve clarity and fluency of the text. No artificial intelligence tools were used to generate original data or scientific conclusions.
Author Contributions
Tugba Birben Kurt: Conceptualization; Investigation; Methodology; Project administration; Resources; Software; Validation; Visualization; Writing—original draft; and Writing—review & editing. Derya Caglar: Investigation; Resources; and Writing—review & editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
As this paper is a narrative mini-review and does not involve human or animal subjects, ethical approval was not required. All data discussed were derived from previously published studies that complied with ethical standards.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
