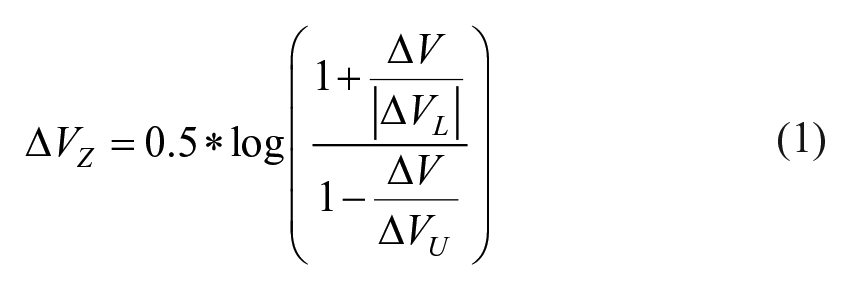Abstract
A major problem in neurorehabilitation is the lack of objective outcomes to measure movement quality. Movement quality features, such as coordination and stability, are essential for everyday motor actions. These features allow reacting to continuously changing environment or to resist external perturbations. Neurological disorders affect movement quality, leading to functionally impaired movements. Recent findings suggest that the central nervous system organizes motor elements (eg, muscles, joints, fingers) into task-specific ensembles to stabilize motor tasks performance. A method to quantify this feature has been previously developed based on the uncontrolled manifold (UCM) hypothesis. UCM quantifies movement quality in a spatial-temporal domain using intertrial analysis of covariation between motor elements. In this point-of-view article, we first describe major obstacles (eg, the need for group analysis) that interfere with UCM application in clinical settings. Then, we propose a process of quantifying movement quality for a single individual with a novel use of bootstrapping simulations and UCM analysis. Finally, we reanalyze previously published data from individuals with neurological disorders performing a wide range of motor tasks, that is, multi-digit pressing and postural balance tasks. Our method allows one to assess motor quality impairments in a single individual and to detect clinically important motor behavior changes. Our solution may be incorporated into a clinical setting to assess sensorimotor impairments, evaluate the effects of specific neurological treatments, or track movement quality recovery over time. We also recommended the proposed solution to be used jointly with a typical statistical analysis of UCM parameters in cohort studies.
Introduction
One of the current problems in the field of neurorehabilitation is uncertainty about the best type of interventions for individuals with neurological conditions, for example, stroke, Parkinson’s disease (PD), and incomplete spinal cord injury.1-4 There have been several randomized clinical trials published in recent years examining the effectiveness of various novel neurological treatments in comparison to standard care physical therapy.1,5 The majority of these studies reported no differences in the primary outcome measures between the experimental and control groups. 5 All participants improved; however, their sensory-motor recovery was incomplete.1,6 While current clinical trials focus on interventions based on principles of neuroplasticity and motor learning (eg, repetition, task difficulty, motivation, etc), 7 they do not consider basic concepts of neural motor control and how control processes are disrupted after a brain injury. 8
Thus, the need for implementing current theories of the neural control of movements into clinical practice has been increasingly recognized in the field of neurorehabilitation as a possible solution to develop theory-based clinical assessments and treatments.9-12 Translating findings from the area of neural motor control into clinical practice can provide tools that accurately measure fundamental properties of movements such as motor performances (eg, endpoint accuracy, velocity, and smoothness) 13 and movement quality (eg, intersegmental and multimuscle coordination).14,15 Motor performance is described as the end-effector’s movement smoothness, speed and precision in the external space and, thus, cannot fully detect the recovery processes since movement might be performed with compensation at different levels (eg, reaching to an object with abnormal trunk displacement).16,17 Movement quality is quantified in body-centered coordinates providing spatial and temporal characteristics of joint angles, segment (ie, trunk) and muscle activation pattern suited to distinguish recovery from compensation strategies.15,18 Recently, movement quality has been operationally described as a comparison between a motor task or action executed by a patient with regard to a reference population of age-matched healthy individuals. 18 Specifically, from a motor control perspective, movement quality is quantified by the extent to which coordination among joints and muscles provides stability at the endpoint level, ensuring accurate and adaptable movements. For example, during a functional task, eg, reaching or walking, individual joints, angles, and segments, as well as forces/moments, are coordinated to stabilize in spatial and temporal domains the resultant final position of the body.15-18 The difference between the quantified movement quality characteristics in healthy and neurological individuals may be considered a proxy of its clinically measure outcomes able to differentiate restitution from compensatory strategies.15,18 Features of movement quality, such as stability and coordination, are essential characteristics of everyday motor actions. Coordinated and stable movements allow healthy individuals to react to the continually changing environment or to resist unpredictable external perturbations. Neurological or musculoskeletal disorders affect stability and coordination, leading to functionally impaired movements (ie, object dropping) or compromised movement safety (ie, increased the risk of falling). Thus, in neurorehabilitation, there has been increasing recognition of movement quality as a core measure for clinical research in individuals with neurological disorders.16,18,19
Recent neurophysiological findings suggest that the central nervous system (CNS) organizes motor elements (ME, eg, muscles, joints, fingers, etc) into task-specific ensembles called synergies.20,21 In neurophysiology and clinical practice, the term synergy has various meanings, which often can be confusing. In movement science, “synergy” can be described as coherent activation of a group of muscles involved in a motor task (MT). 22 In clinics, the term synergy has a negative connotation describing abnormal motor patterns (ie, flexor or extensor synergies) due to brain lesions23,24 (eg, damage of descending motor pathways). To avoid confusion, in the current article, the term synergy refers to task-specific ensembles (ie, multimuscle or multifinger coordination) that ensure stability of salient performance variables related to MT.20,25 The latter definition explicitly highlights the specific task context of motor control as described by Levin et al 26 and endorsed by the Second Stroke Recovery and Rehabilitation Roundtable. 18 Synergies result from specific covariation between ME aimed at stabilizing particular performance variables (eg, moments of force, center of pressure [COP]), related to MT (eg, rotational stability of the held object, or postural stability during quiet standing) 21 . A method to objectively quantify this feature has been developed based on the uncontrolled manifold (UCM) hypothesis.20,27 Briefly, UCM analysis uses intertrial analysis to estimate 2 components of variance that do (VORT) and do not affect (VUCM) a specific performance variable related to MT. The normalized difference between these two components is used to compute the index of stability (ΔV). ΔV reflects the degree of ME coordination (eg, intersegmental and multimuscle coordination) used to stabilize MT performance. In more intuitive terms, carrying a glass of water while walking requires inter-segmental coordination to keep the glass vertical. Strong intersegmental coordination suggests that spontaneous deviations in joint angles or external perturbation applied to the upper limb would lead to changes in joint configuration primarily within the UCM, therefore, not affecting the glass orientation and preventing the water from spilling. Such behavior is expected to lead to relatively high VUCM values and positive ΔV.
Furthermore, a drop in ΔV stabilizing a MT in preparation for a quick action or a perturbation involving that MT has been denoted as anticipatory synergy adjustments (ASAs).28,29 For example, previous studies29,30 showed that, in anticipation of postural adjustments, the intermuscular coordination changes in a feed-forward fashion (ΔV decrease) to modify stability during a steady-state postural action. Therefore, ASAs are considered as an index of agility—the ability to modify intersegmental or intermuscular coordination to attenuate stability of the task performance, in preparation for MT changes. 21
Evidence from several studies suggested the capability of ΔV as a sensitive biomarker to assess movement stability and coordination in various neurological impairments9,31 (eg, basal ganglia impairments,32,33 PD,34,35 large-fiber peripheral neuropathy, 36 and stroke). 37 Moreover, UCM has been used to evaluate the effectiveness of specific neurological treatments (effects of deep brain stimulation, DBS) 38 and dopamine-replacement drugs (eg, levodopa, 35 in individuals with PD). Significant changes in ΔV and delayed or reduced ASAs have been described in neurological populations, even without clinically identifiable motor impairments, like individuals with PD at stage I of Hoehn and Yahr scale 9 or well-recovered poststroke individuals. 37
The application of UCM analysis has so far been limited to experiments that involve multiple subjects to generate a distribution of UCM parameters for group comparisons (within-subject or between-subject study design).14,31 Group comparisons allow researchers to use statistical tools to identify possible significant motor control changes in clinical populations (pre- vs post-treatment or patients vs control group). This approach, however, is impractical to replicate in a clinical setting. Instead of group comparisons, clinicians are usually interested in testing single individuals to estimate the level of neurological impairment, monitor the trajectory of recovery, and to help them develop the best treatment strategies. The purpose of this point-of-view article is to identify problems that hinder the use of UCM in a clinical setting and to suggest possible solutions that would help translate this method into clinical practice. We also propose a novel application of bootstrapping estimation to quantifying movement quality using UCM for a single individual as an objective biomarker for neurorehabilitation in a clinical setting.
The Problem
In the following sections, we will describe 2 significant obstacles that hinder the use of UCM analysis in a typical clinical setting.
The Number of Subjects Required for the UCM Analysis
The UCM analysis quantifies the variability structure of ME across several repetitions of a tested movement. Another UCM-based analysis used to quantify the task-specific organization of ME in a single trial is called motor equivalence analysis.39,40 Motor equivalence analysis measures reorganization of ME following the perturbation of MT. UCM analysis; however, allows the computation of ΔV either for a particular phase of a MT (ie, final hand position during reaching,37,41 steady-state phase of a prehensile task) 42 or at each time point during a movement (COP displacement during whole-body sway, 43 or walking, 44 or multifinger force tracking). 36 In all current applications, UCM analysis provides only one ΔV, VUCM, and VORT estimates (or one trajectory of these estimates) per subject. As stated earlier, UCM analysis partitions variance of the elemental variables into components that affect (VORT) and do not affect (VUCM) the value of a particular performance variable. Experiments that use UCM, typically compute these parameters across many individuals with neurological impairments and their healthy counterparts to generate distributions of estimated parameters for group comparisons. A subsequent problem associated with the nature of one variance estimates per individual is the lack of standard error of measurement and minimal detectable change related to the magnitudes of ΔV, VUCM, or VORT. 14 The theoretical distribution and the confidence intervals of UCM parameters are unknown, and most probably differ between a healthy population and individuals with neurological disorders. Thus, the individual magnitudes of UCM parameters might be ambiguous. This issue is usually solved through statistical inference to detect differences between the probability distribution of UCM parameters, generated from the data collected from several subjects (for within-group or between-group comparisons). Research studies involving many participants allow identifying significant differences in average UCM parameters between individuals or group effects of brain stimulation on movement. However, clinicians and health care professionals are mainly interested in testing individuals to assess levels of neurological impairments, which makes translating the UCM in its current form to a clinical setting impractical.
Magnitudes and Uncertainty of UCM Parameters
Traditionally, UCM values are evaluated based on their magnitude (VUCM, VORT, ΔV) or sign (ΔV). According to the UCM theoretical framework, selected ME should be independent of each other. Therefore, it is assumed that positive ΔV values (VUCM > VORT) indicate the presence of multielemental coordination stabilizing MT performance, with larger ΔV values representing stronger MT stabilization. 21 In contrast, negative ΔV values (VUCM < VORT) indicate MT performance destabilization. Randomly selected ME values should produce an equal amount of 2 components of variance that do (VORT) and do not affect (VUCM) a specific MT, with ΔV value equal to zero (VUCM = VORT). 25 Figure 1 illustrates 3 hypothetical scenarios for UCM results.
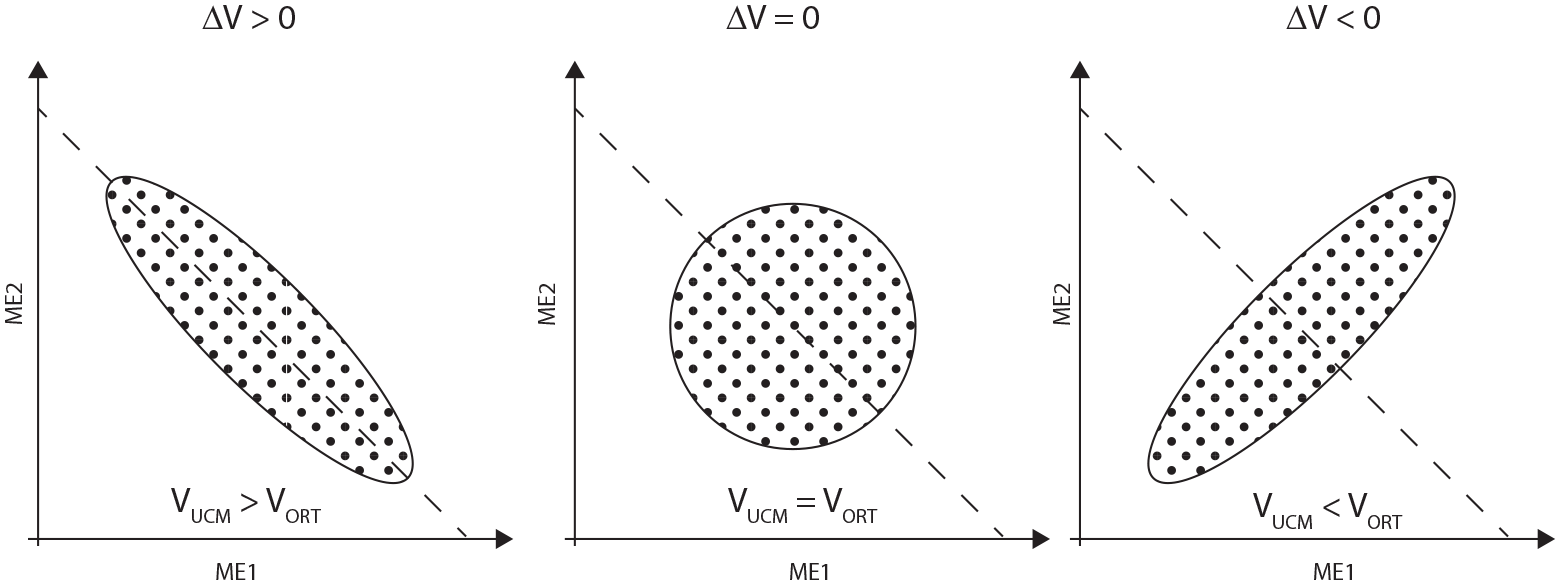
An illustration of 3 theoretical groups of the uncontrolled manifold (UCM) results. A subject produces a required total force (dashed line) with 2 fingers (motor elements ME1 and ME2) across many repetitions. Black dots illustrate data points of individual trials. The left panel shows data distribution observed when a subject stabilizes the motor task (total force), using negative covariation of individual finger forces to minimize the variance of the total force (VUCM > VORT and ΔV > 0). The right panel shows data distribution when the motor task is destabilized, with large variability in the total force due to positive covariation of individual finger forces (VUCM < VORT and ΔV < 0). The central panel shows theoretical data distribution, with no covariation between finger forces.
However, considering only the numerical values of the estimated UCM parameters might be misleading. Variance parameters (VUCM and VORT) estimated from a random distribution of ME have arbitrary values, and their magnitude depends on the number of trials used in the UCM analysis. When a finite number of movement repetitions is used, the random set of ME does not produce ΔV = 0, (VUCM = VORT). Generally, the difference in random magnitudes between VUCM and VORT increases with the decreasing number of trials used. A small number of random trials (ie, 20) may incidentally produce a sizeable difference between VUCM and VORT, and a large positive ΔV. The increasing number of random trials (ie, 1000), makes the estimated variance components closer to each other, with ΔV values asymptotically approaching zero (see Figure 2). Also, there is always a 50:50 chance that VUCM will be larger than VORT (ΔV > 0) when estimated from the finite random ME data set. In a clinical setting, it is impractical and frequently impossible to repeat MT many times. Usually, for UCM purposes, subjects repeat MT between 15 and 30 times. For example, the recently published recommendations 45 advise for 15 to 20 MT repetitions for accurate ΔV approximation in a gait task. Thus, in some situations, ΔV > 0 may not necessarily indicate the presence of underlying ME coordination to stabilize MT performance. Positive ΔV values, even with high magnitude, may be observed merely because of the effect of random noise, especially when the inadequate (ie, not enough) number of trials are used to estimate UCM parameters.
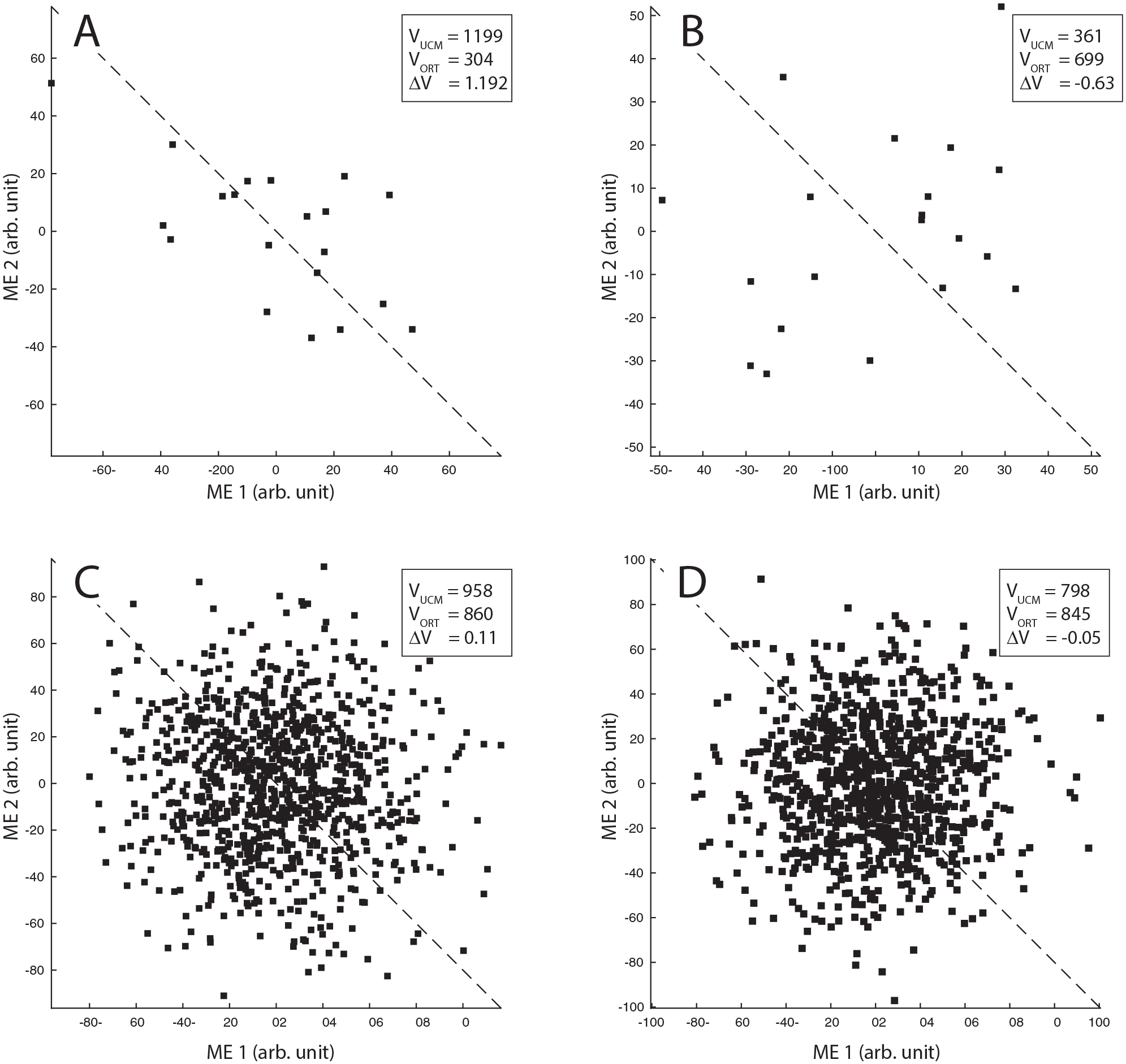
Graphical representations of typical uncontrolled manifold (UCM) results generated from the random data sets of motor elements (MEs). Please note large differences between VUCM and VORT for computations with 20 trials (panels A and B). When a very large number of random trials (ie, 1000) are used, the magnitudes of VUCM and VORT tend to equalize (panels C and D), and ΔV values asymptotically approach zero.
A reliable biomarker for neurorehabilitation must provide clinicians the UCM estimates (VUCM, VORT, ΔV) that describe the real behavior of an individual and with a very low probability that these parameters were obtained just by chance.
The Solution
The use of the UCM method is currently limited to experiments that require many subjects to generate a distribution of variance parameters for group comparisons. Thus, we propose a novel use of bootstrapping estimation that allows quantifying movement quality using UCM parameters for a single individual. First, we will introduce the process of computing and evaluating UCM parameters for a single subject. Then, we reanalyzed previously published data from individuals with PD performing a wide range of MTs, that is, multidigit pressing and postural tasks. Our findings will show that the individually estimated UCM parameters reliably identified motor quality impairments in subjects with PD, consistent with group differences (control vs. PD subjects) reported in previous publications.
Typical Uncontrolled Manifold Analysis
To illustrate the novel application of bootstrapping simulations for UCM parameters, we used previously published data by Solnik et al. 42 In this study, the authors aimed to identify different motor control strategies while maintaining the rotational equilibrium of the object held by either (1) 2 persons or (2) a single person using both hands. The main task of the experiment was to pass the object between 2 persons or between the hands of the same person. The forces applied to the object by hands were measured for both conditions. Then, the moments of force were calculated during the steady-state phase when both hands were holding the object. More specifically, the authors evaluated the multifinger coordination strategies stabilizing the object’s rotational equilibrium (ie, moments of force applied to the object).
From previously published data, we selected a typical data from 1 subject and 1 pair of subjects repeating the prehension task 20 times. The data consisted of 20 pairs of hand forces recorded during the steady-state phase for each condition (ie, single-person and 2-person). For each experimental dataset, we followed the typical UCM analysis procedure (for computational details, see Latash et al, 20 Klous et al, 29 and Solnik et al 42 ). The variance across trials was separated into 2 components VUCM and VORT. Then, we quantified the performance–stabilizing coordination by calculating ΔV for each performance variable, using: ΔV = (VUCM – VORT)/VTOT, where VTOT is total variance. For further statistical analyses, computed ΔV values were log-transformed using a modified Fisher’s z-transformation, using
where ΔVL and ΔVU are lower and upper limits of the ΔV, respectively. The modified Fisher’s transform is adjusted for the actual computational limits of ΔV to ensure that the transformed ΔVZ values are equal to zero when VUCM = VORT (for details see Equation 4 in Solnik et al 41 ).
In a typical UCM study, researchers repeat this procedure for several subjects and subject pairs to identify different motor control strategies between 2 groups. However, our solution, based on bootstrapping simulations, allows us to evaluate UCM variance estimates and identify synergies for a single–subject design.
Bootstrapping Simulations and UCM Confidence Intervals
As outlined in the previous section, UCM parameters are calculated from a set of ME, recorded from several repeated trials, which results in one UCM parameter per patient. However, previously described issues do not allow to draw clinical conclusions based on individual VUCM, VORT, or ΔVZ magnitudes. Each of these values needs to be validated against the sampling distribution computed from a random selection of recorded ME, to evaluate if the observed positive or negative ΔVZ values represent a purposeful organization of ME. We propose the bootstrapping method to quantify the amount of uncertainty of UCM parameters (eg, VUCM, VORT, or ΔVZ) by computing measures of accuracy (eg, confidence intervals) of these estimates. Clinicians should be aware of measurement errors of clinical tests administered to a patient. This information should guide a clinician in deciding if the observed test values are meaningful and not due to the measurement error (eg, goniometry errors in the range of motion estimation). 46 Our solution provides clinicians with an estimated error of UCM-based biomarker of movement quality, using the bootstrapping method.
Specifically, for Solnik et al, 42 the following procedure was performed. For bootstrapping simulations, we randomly sampled the recorded ME values (ie, 20 pairs of hand forces for single-person and 2-person) with replacement. The random sampling with replacements keeps values and the sample size equal to the ME previously used to compute UCM parameters, but destroys any covariation 47 between them (ie, sampling left- and right-hand forces from different trials). We then computed simulated UCM parameters from the randomly selected ME values. Next, we repeated this process 10 000 times to create the distribution of simulated UCM parameters. The simulated VUCM, VORT, or ΔVZ distributions, allow estimating the uncertainty of UCM values from testing of an individual subject or subject pair. The range of values between 95% confidence intervals (CIs) 48 represents UCM parameters obtained without any determined organization of ME. Thus, if UCM parameters fall outside the computed CI, there is a 95% chance that ΔVZ, VUCM, and VORT estimates describe the purposeful motor control to stabilize (ΔVZ > upper CI) or destabilize (ΔVZ < lower CI) MT performance (Figure 3).
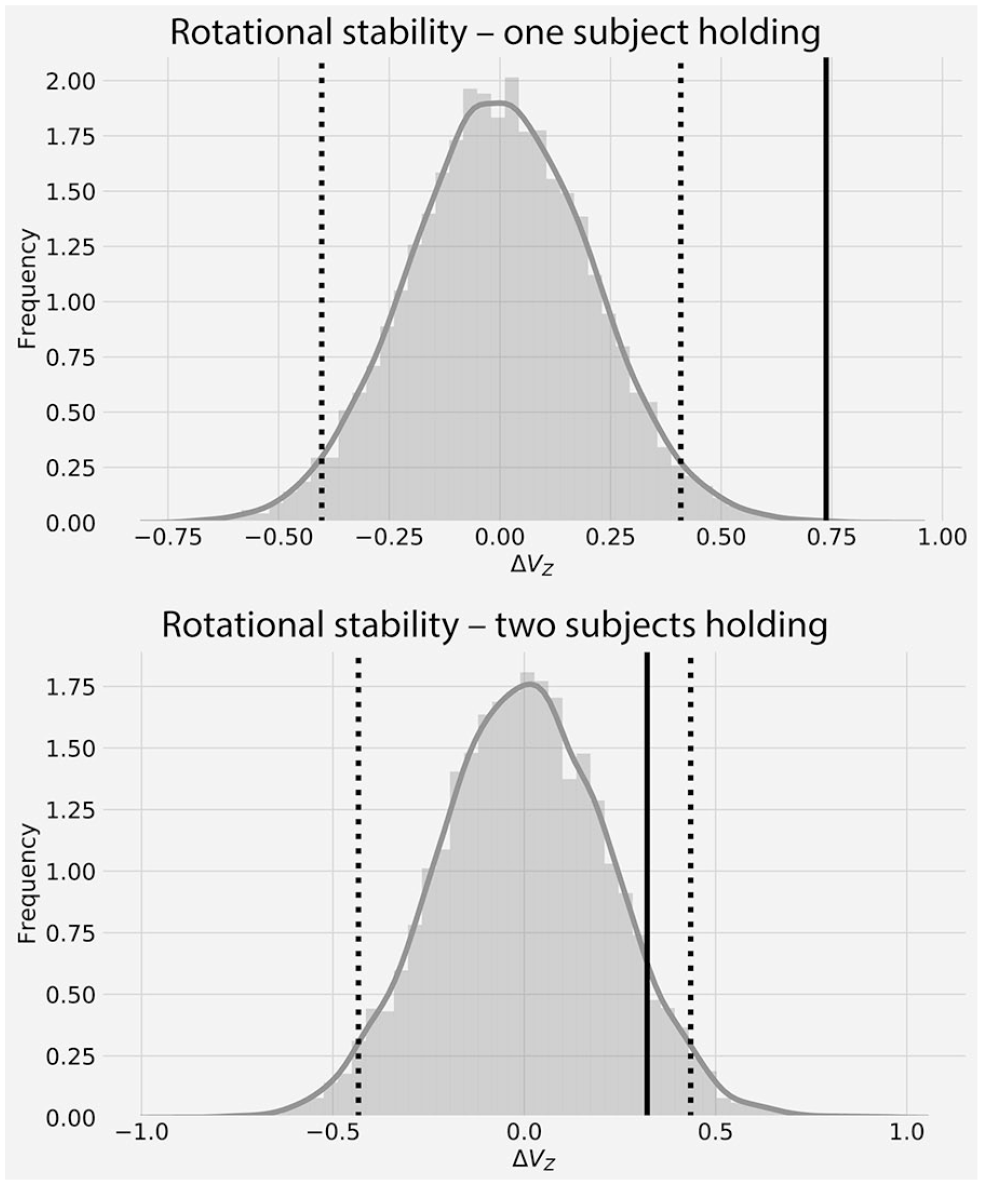
Example of bootstrapping simulations of indices of stability (ΔVZ) from previous experimental data from 1 subject (top panel) and a pair of subjects (bottom panel). Note that the distributions obtained from bootstrapping simulations (shaded areas) represent a sampling distribution of UCM parameters, computed from the observed motor elements (MEs), but with random resampling with substitutions. Dotted vertical lines indicate 95% confidence intervals. The solid vertical line indicates experimentally obtained ΔVZ. Only ΔVZ that lies outside the computed confidence interval (CI) (1 subject, top panel) describes the real behavior of the individual with .05 significance level.
The subject performing the prehension task alone displayed multifinger coordination stabilizing the object’s rotational equilibrium (ie, ΔVZ > upper CI, Figure 3 top panel). In a 2-person condition, ΔVZ were positive, but they did not exceed 95% CIs. It is worth noticing that our approach provides additional information that could not be obtained from the group comparisons, where statistical tests are designed to identify differences in ΔVZ magnitudes. In some situations, due to the small number of repetitions or high variability in the experimental data, the range of CI may be broad, and even high positive values of ΔVZ would not be indicative of MT stabilizing multifinger coordination. For example, in the original study by Solnik et al, 42 the authors did not detect significant differences in multi-finger coordination stabilizing moments of force between one–person and two-person conditions, with the assumption that both conditions showed motor behavior stabilizing MT (both groups had ΔVZ > 0). Our method showed that in a two-person condition, subjects did not utilize multifinger coordination to stabilize moments of force, because positive ΔVZ value fell within the computed 95% CI (Figure 3, bottom panel). Therefore, we suggest comparing ΔVZ values to 95% CI, instead of relying only on the estimated magnitude or sign of the ΔVZ. Only indices of stability that lie outside of their random distribution can be identified as signs of a true task-stabilizing (ΔVZ > upper CI) or task-destabilizing (ΔVZ < lower CI) behavior.
These results support the use of bootstrapping simulations as a valid tool to quantify and validate movement quality using UCM for a single individual. Next, we will discuss the use of the proposed solution as objective biomarkers for neurorehabilitation in a clinical setting.
Effect of Deep Brain Stimulation on Motor Quality of a Multifinger Task of an Individual With Parkinson’s Disease
We tested the ability of our solution to identify the effectiveness of a specific neurological treatment on subjects with PD. For this purpose, we reanalyzed data from a previously published article 38 aimed to investigate the effects of DBS on multifinger coordination and agility in individuals with PD. Participants have been asked to produce a specific force with 4 fingers of one hand, followed by a rapid force pulse at a self-paced time. Authors computed ΔVZ and ASAs to assess multifinger coordination to stabilize the total force during the steady-state, and the ability to attenuate the stability of the motor task in preparation for quick force change, respectively. All subjects repeated the MT in 2 conditions, with and without DBS of nuclei within the basal ganglia. We reanalyzed data from one individual with PD: a 75-year-old man, with PD duration for 14 years, taking oral medication with levodopa equivalent daily dose (LEDD) of 755 mg. The bilateral DBS leads were implanted in the subthalamic nucleus 5 months before the data collection. The subject’s motor scores, estimated from the Unified Parkinson’s Disease Rating Scale–Part III (UPDRS III), were 49 and 47 for DBS-on and DBS-off states, respectively. In brief, the multifinger MT started by pressing on miniature force sensors and maintaining the total force at 8% of maximal voluntary contraction (MVC) level for about 5 seconds. Then, at a self-selected time, the subject performed a quick force pulse into the target set at 25% of MVC. The subject repeated the multifinger motor tasks 17 times for DBS-on and 21 times for DBS-off conditions. Both conditions were performed with the right hand. We selected individual finger forces from 600 ms before to 200 ms after the quick pulse for further analysis. The finger forces were aligned with respect to the onset of the quick force pulse (time = 0). For further analysis, we replicated the typical steps of UCM analysis (see details of data processing in Falaki et al 38 ). We separated the intertrial variance of ME (eg, finger forces) into two components VUCM and VORT. Then, we quantified the multifinger coordination stabilizing total force by calculating ΔV for each time step. ΔVZ was computed using Equation 1.
Similar to the previous study, we estimated the time of ASA initiation as the point in time when ΔVZ decreased more than 2 standard deviations from the average ΔVZ from the steady-state (from 600 ms to 400 ms before time = 0, as performed in the original work). This procedure was repeated for both DBS-on and DBS-off conditions (see ΔVZ trajectories and ASA in Figure 4, and VUCM and VORT trajectories in Figure 5). Next, we estimated 95% CI of the computed ΔVZ, VUCM, and VORT using the bootstrapping simulation, for each time step (shaded areas in Figures 4 and 5).
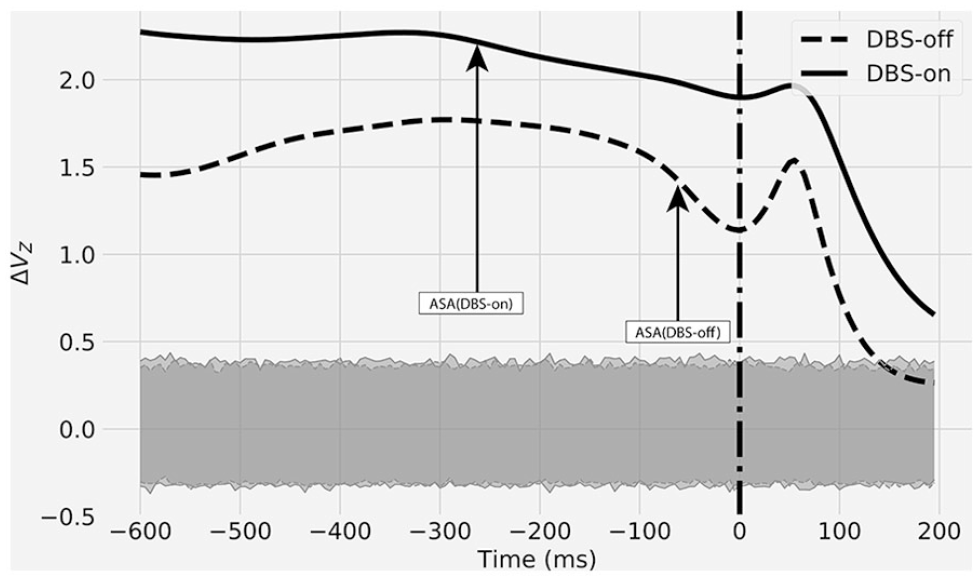
Reanalyzed data 38 from one individual with Parkinson’s disease (PD) with (DBS-on) and without (DBS-off) deep brain stimulation (DBS) performing a multifinger task. Index of stability (ΔVZ) trajectories from 600 ms prior and 200 ms after the onset of motor task change (time = 0). The dotted vertical line indicates the onset of a quick pulse. Note that the anticipatory synergy adjustment (ASA) was observed earlier during DBS-on when compared with the DBS-off condition. Shaded areas indicate 95% confidence intervals (CIs) computed from bootstrapping simulation.
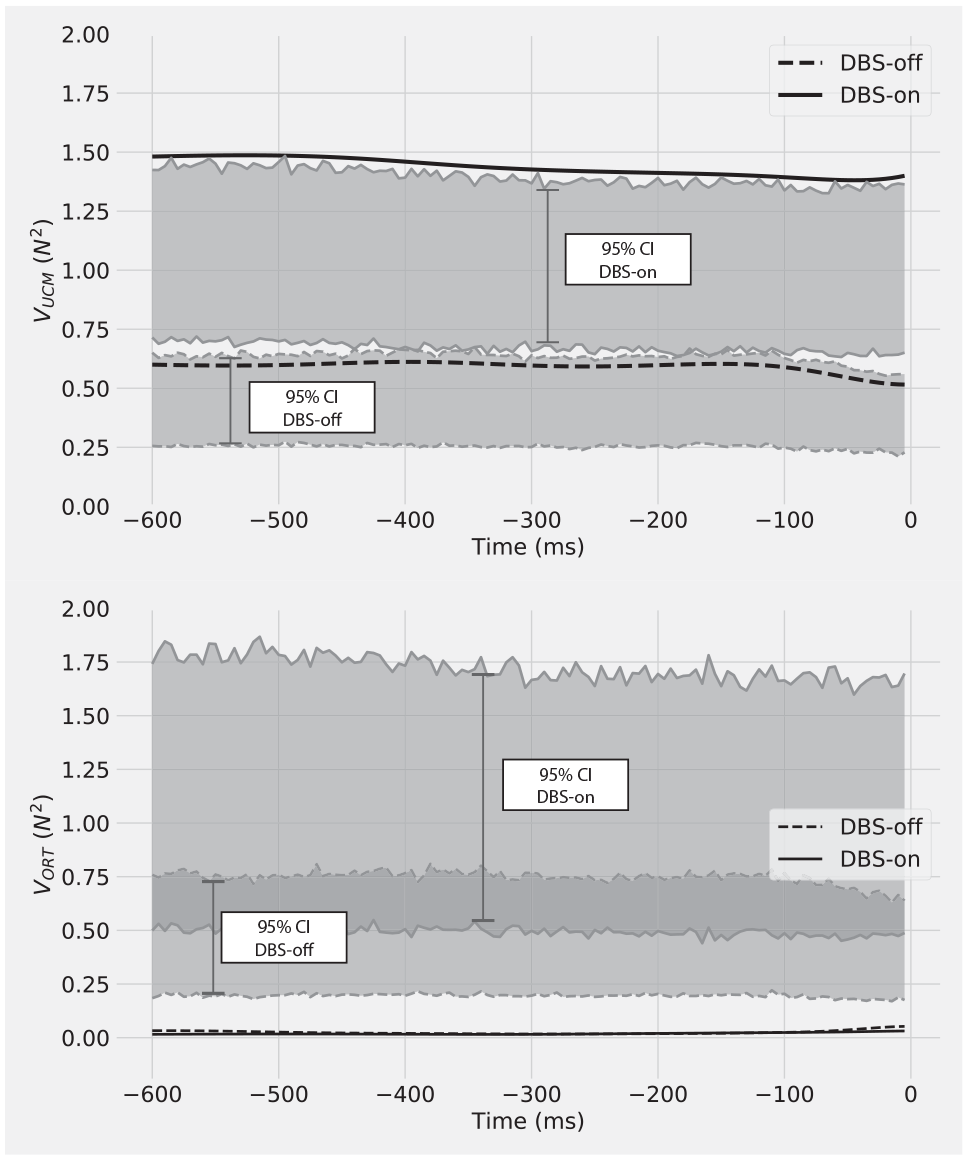
Results of bootstrapping simulations of 2 components of variance that do (VORT) and do not affect (VUCM) a specific motor task revealed a strong effect of deep brain stimulation (DBS) on movement quality. Data obtained from one individual with Parkinson’s disease (PD) performing a multifinger task with (DBS-on) and without (DBS-off) DBS. Shaded areas represent 95% confidence intervals (CIs). Black lines represent experimentally obtained VUCM (top panel) and VORT (bottom panel). Dashed lines indicate conditions with DBS-off and solid lines with DBS-on. In both DBS-off and DBS-on conditions, the individual with PD actively decreased the total force variability (VORT < lower 95% CI). DBS had; however, a strong effect on VUCM. DBS-on caused the emergence of multifinger coordination that increases the motor task stability (VUCM > upper 95% CI).
The bootstrapping simulations showed that the individual with PD displayed multifinger coordination that stabilized the total force, regardless of the stimulation. ΔVZ were above 95% CI, in both DBS-on and DBS-off conditions during the steady-state (see Figure 4). However, ΔVZ magnitudes were higher when DBS was on, indicating stronger stability of the MT. The ASA analysis revealed that DBS improved the ability to decrease stability of the motor task in preparation to sudden action change. The ASA was observed earlier (–260 ms) during DBS-on, when compared with the DBS-off condition (–55 ms). These results align well with the group-averaged results published previously. 38
Notably, the use of bootstrapping on VUCM and VORT variance components revealed DBS effects on movement quality, that were not detected previously. During the MT, the individual with PD actively decreased the total force variability (VORT), regardless of the DBS state. Indeed, VORT trajectories had small magnitudes, with values below the 95% CI in both DBS-on and DBS-off conditions (Figure 6, bottom panel). DBS had a strong effect on the variance of finger forces within the UCM. In DBS-off, VUCM values were within 95% CI (ie, not significant). In other words, with DBS-off, the individual did not utilize multifinger coordination to exploit many possible solutions to perform the MT successfully. Conversely, with DBS-on, the overall variability of the performed movements increased (note increased 95% CI in DBS-on, Figure 5). The variance increase, however, was primarily within the UCM. In the DBS-on condition, VUCM trajectories increased to values above 95% CI, for the whole MT. This demonstrates that DBS caused the emergence of multifinger coordination that increases the stability of MT performance.
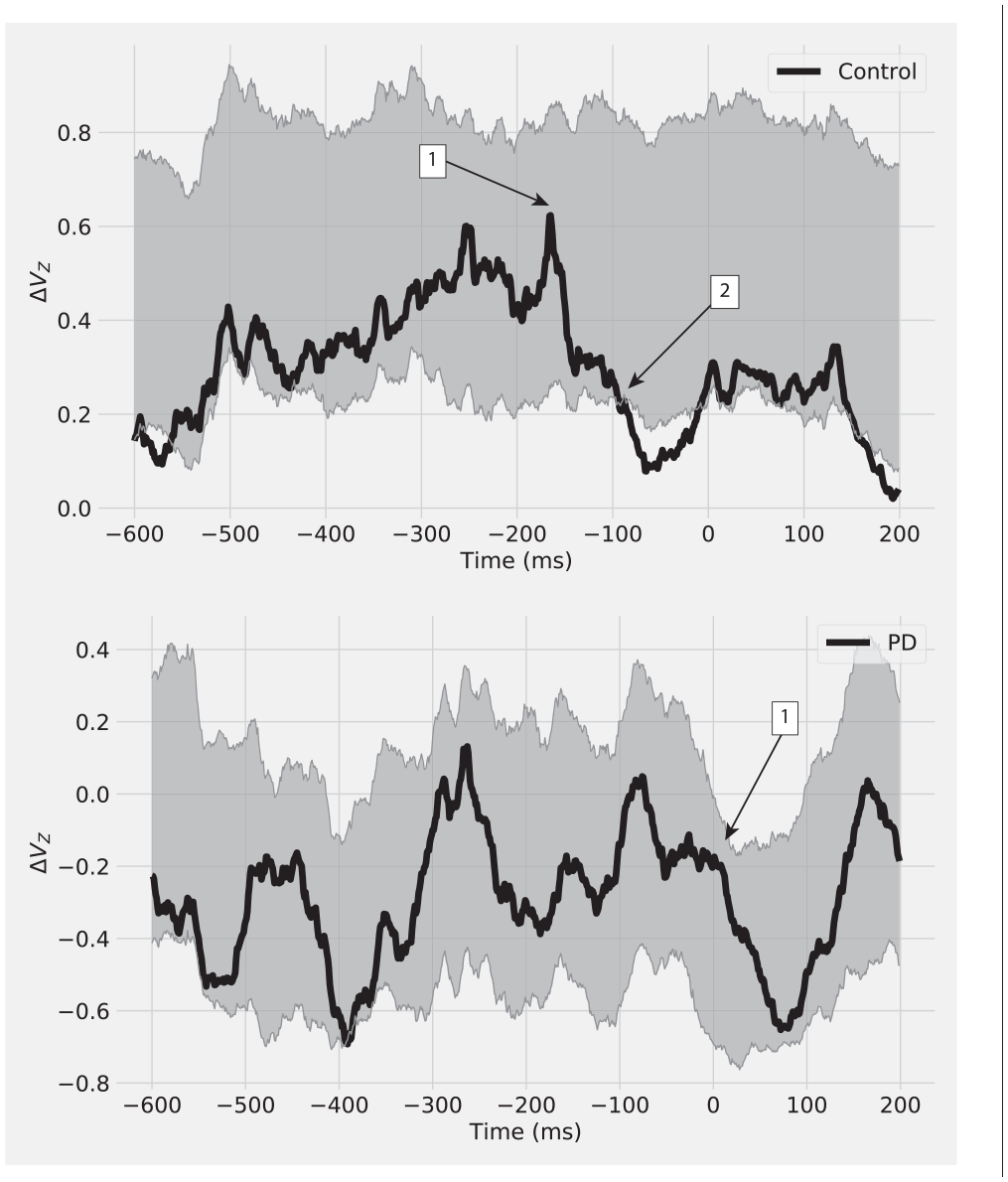
Index of synergy (ΔVz) during the preparation for the motor task change at time = 0 for a control (Control) participant and a patient with Parkinson’s disease (PD). Shaded areas represent 95% confidence intervals (CIs) of the distribution generated with the bootstrapping method. During the steady-state (600 ms to 200 ms before time = 0), the overall ΔV was higher for Control than for PD; however, these values were within 95% CI. The consistent drop of ΔV, related to anticipation of the motor task change, started earlier for Control than for PD (1). The significant attenuation of the motor task stability (ΔVz < lower CI) is only observed in Control (2), at around 100 ms before the movement task change.
In general, the application of bootstrapping simulation to VUCM and VORT parameters revealed new information about the effect of DBS on the individual with PD. Only in DBS-on conditions, the subject exhibited movement quality characteristic, typical to healthy population—actively compensating MT errors (ie, minimized VORT) and utilizing multifinger coordination to stabilize MT performance (ie, increased VUCM).
Postural Synergic Control of an Individual With Parkinson’s Disease
To investigate the use of the proposed solution for a whole-body MT, we reanalyzed data from a previously published article aimed at exploring the changes in postural control in the PD population. 35 More specifically, the authors investigated multimuscle coordination stabilizing the COP trajectory in a postural task performed by PD patients without identified clinical symptoms (≤Hoehn-Yahr stage II). Additionally, the authors quantified ASA, that is, preparatory decrease in ΔVZ that represents MT stability adjustments in anticipation of a self-triggered postural perturbation. The authors recorded electromyographic (EMG) signals from muscles of lower extremities and measured ground reaction forces to calculate COP trajectories. Subjects were asked to hold a weight in a fully extended arm in front of them for about 3 seconds (steady state), and at a self-paced time, to release the weight (self-triggered postural perturbation), while maintaining the upright posture. In the current study, we reanalyzed data from one individual with PD and from one representative of the control group. We used 24 repeated trials to compute the multimuscle coordination that stabilized COP in the anterior-posterior direction, from 600 ms before to 200 ms after the weight drop at time = 0. The computational details are described elsewhere. 35 For this selected time, we estimated 95% CI of the computed ΔVZ using the bootstrapping simulation (see Figure 6).
During the steady state (600 ms to 200 ms before the weight release), the overall ΔVZ was higher for the control subject when compared with the individual with PD; however, these values were within 95% CI. The consistent drop of ΔVZ, indicating ASA of the MT, was observed in the control subject only. The noisy characteristics of EMG signals make the 95% CI to have a wider range; therefore, the initial ΔVZ drop was not significant (point 1, Figure 6 top panel). The significant ASA was observed in control subject at around −100 ms before the MT change (point 2, Figure 6). Notably, the individual with PD did not exhibit such anticipatory behavior, and the onset of ΔVZ drop occurred at the moment of weight release (point 1, Figure 6, bottom panel).
In general, the observed reduction in the time of ASA in the individual with PD supports the findings of the original study. The modification we propose provides additional information that potentially has significant importance in the clinical application. Our approach shows that none of the subjects exhibited significant COP-stabilizing behavior during the steady state. However, only the healthy individual displayed anticipatory attenuation of the MT stability before the self-elicited perturbation.
Recommendations
For the first time, we offer a solution for translating the UCM framework from theoretical neuroscience and experimental findings to clinical practice. Our solution may be incorporated into a clinical setting to assess sensorimotor impairments, evaluate the effects of specific neurological treatments, or track movement quality recovery over time. Measuring movement quality employing UCM may help the development of new treatment strategies based on motor control of movement.
There is growing evidence that low-cost sensors could be used in clinical settings to measure joint kinematics, 49 finger forces, 18 and EMG signals.50,51 A reach-to-grasp task may be performed with inertial sensors to collect kinematics data and estimate joint angles. An instrumented handle could measure finger forces during a prehension tasks, while low-cost EMG sensors50,51 can be used to measure muscle activation in whole-body tasks (eg, body sway). These clinically accessible devices to measure biomechanical variables are promising tools in implementing the use of UCM in clinical practice. In our approach, a patient would need to perform 25 to 30 repetitions of a MT, while selected ME variables are recorded. Then, a computer software would present a clinician the computed UCM parameters with the associated range of 95% CI from bootstrapping simulations. This analysis could be performed for a specific epoch (ie, steady state in prehension tasks) or for the entire movement trajectory (ie, reach-to-grasp or postural tasks).
We recommend the proposed solution as an extension of the current UCM analysis for situations when movement quality assessment is needed in a clinical setting. Our solution provides clinicians with a UCM-based biomarker that can be used as a proxy of movement quality. It provides clinicians with an objective evaluation of movement that overcomes measurement limitations when using ordinal scales. 52 Our bootstrapping simulation is effective in detecting the effects of selected neurological procedures and impairments on motor behavior of single individuals.
The use of our approach is also recommended for research experiments using within or between-group designs. The proposed solution should be used jointly with a typical statistical analysis of UCM parameters. Evaluation of each experimentally obtained UCM parameters against 95% CI, would provide additional information about the motor behavior changes of the individuals included in the tested groups.
Future studies should investigate the minimum number of trials for reliable UCM estimations and the feasibility of employing UCM in clinical practice with low-cost sensors and clinically accessible EMG systems. Further development of open-source data-science friendly programs might increase the application of UCM in neurorehabilitation research and clinical settings.
Footnotes
Acknowledgements
We wish to thank Dr Ali Falaki, Dr Hang Jin Jo, and Prof Mark Latash for sharing data from their previous investigations.
Authors’ Note
Python implementation of our algorithm developed for this study is available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the University of North Georgia Presidential Award.