Abstract
Background:
Spinal cord injury (SCI) can impair motor, sensory, and autonomic function. The formation of the glial scar comprises protective as well as inhibitory neurite outgrowth properties operated by the deposition of chondroitin sulfate proteoglycans (CSPG). Chondroitinase ABC (ChABC) can degrade CSPG and foster neuroaxonal plasticity as a therapeutic approach to restore locomotor function after SCI.
Objectives:
To systematically review experimental ChABC treatments after SCI and assess their efficacy for locomotor function a comprehensive literature search was conducted following pre-registered Prospero Study protocol, selecting animal studies evaluating neurobehavioral outcomes after traumatic SCI followed by the calculation of normalized effect sizes applying meta-analysis and meta-regression methodology. Additional analyses were performed to investigate the impact of animal type, strain, sex, sample size, injury models, level of injury, and treatment duration.
Results:
Within the overall analysis of 1066 animals, a considerable amount of heterogeneity was observed. A subgroup analysis comprising experiments applying the same neurobehavioral measurement (blood–brain barrier/Basso-Mouse-Scale [BMS]-subgroup) demonstrated a 15.9% (95% CI = 11.3%-20.6%) improvement in locomotor outcomes. Different experimental characteristics influenced neurological recovery, including sex, level of injury, used anesthetic, reported dosage of ChABC treatment, the timepoint of assessment and perioperative temperature control. Sensitivity analysis applying Trim and Fill identified 19 hypothetical missing experiments suggestive of reporting bias.
Conclusion:
Reporting bias in experimental SCI research is prevalent and not limited to a specific intervention. ChABC treatment can exert beneficial effects on locomotor recovery after SCI.
Introduction
Spinal cord injury (SCI) often leads to dramatic loss of motor function and functional independency. SCI not only causes life-challenging physiological, mental, and social suffering for the patients but also leads to a significant socioeconomic burden for society and implications for healthcare. 1 After SCI, injured axons fail to regenerate in substantial amounts beyond the lesion site in the adult mammalian central nervous system.2,3 While the scar formation after SCI is essential to restrict primary and secondary damage2,3 it also comprises deposited growth inhibitory molecules, including but not limited to chondroitin sulfate proteoglycans (CSPG).4-8
As a result of an injury to the central nervous system (CNS), proliferating and migrating glial cells such as astrocytes, microglia, and oligodendrocyte precursors accumulate at the lesion site in response to the release of cytokines such as insulin-like growth factor 1. 9 Serum components can be found at the lesion site for up to 14 days after SCI indicative of a disturbed, permeable blood–spinal cord barrier. 10 The mechanical injury to the spinal cord induces the formation of a glial and fibrotic scar,11-13 while the degree of scaring can vary relative to the extent of damage to the meninges or the blood–brain barrier (BBB). 10 The scar demarcating the lesion border contains extracellular matrix molecules including proteoglycans.9,14,15 The most abundant subgroup of the proteoglycan family in the CNS is the cCSPG. CSPG contains a core protein that is covalently attached to glycosaminoglycan sidechains made of chondroitin sulfate disaccharide units. 16 The release of CSPG can persist for up to 2 months post-injury 17 and interfere with neuroaxonal plasticity. 18
Chondroitinase ABC (ChABC) is an enzyme obtained from the Gram-negative bacterium
This systematic review aims to investigate the efficiency of ChABC treatment in animal models of SCI for locomotor recovery.
Materials and Methods
Literature Search
A systematic review of the literature was performed following the guidelines outlined by Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 27 The protocol for this systematic review is registered (ID: CRD42019099001) and can be accessed using the following link: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=99001.
Search Strategy
In this study, we conducted a systematic review of publications from PubMed, ISI Web of Knowledge, and EMBASE. The following search query was used:
(“Chondroitin ABC lyase” OR “Chondroitinase” OR “Chondroitin ABC eliminase” OR “Chondroitinase ABC” OR “ChABC” OR “ChaseABC” OR “Chondroitin sulphate” OR “ChS ABC”) AND (“spinal cord injury” OR “hemisection” OR “contusion injury” OR “dorsal column injury” OR “complete transection” OR “corticospinal tract injury”)
The initial search was performed on 16th November 2017 and updated on 18th May 2022 based on the study protocol. To ensure the inclusivity of animal experiments, we integrated a large animal filter in the PubMed search. 28 Additionally, we manually examined the reference lists of existing reviews on ChABC in animal studies for potential studies. During the literature search and selection process, 2 independent reviewers worked separately (AK and RA) and resolved any disagreements through assessment by a third reviewer (RW).
Selection Process
We applied predefined inclusion and exclusion criteria and generated a flow chart following the guidelines of the PRISMA. Criteria for inclusion involved animal models of SCI, ChABC treatment as the intervention, a control group for comparison, reported values of neurobehavioral recovery, their variance, and the number of animals per study group. Only mammalian models were included in this review. Studies only assessing the efficacy of ChABC treatments in combination with other therapeutics were excluded. Articles without available full text were also excluded.
Data Extraction
We collected primary data that included general information about the studies, such as the study title, first author, publication year, and country. Information related to the animals involved in the studies, including group size, species, gender, age, and weight. The type and severity of SCI was also extracted. Details concerning the therapy regimen, such as dosing, application time, and method of delivery, were documented. Additionally, data on outcome assessment were collected, including the neurobehavioral score utilized, the timing of assessments post-surgery, and the mean behavioral outcome.
For locomotor function, we focused only on the final measurement, along with its standard error or standard deviation. The literature was screened, data was extracted, and quality was subsequently assessed.
Quality Assessment
Study quality was evaluated using a quality score adapted from the CAMARADES quality checklist, 29 which incorporates the following 9 points: (i) publication in a peer-reviewed journal, (ii) control of animals’ temperature, (iii) use of anesthetics without marked intrinsic neuroprotective activity (anesthetics other than ketamine), (iv) randomized treatment allocation, (v) concealment of treatment allocation, (vi) blinded assessment of outcome, (vii) reporting of sample size calculation, (viii) statement of compliance with regulatory requirements, and (ix) statement of potential conflicts of interest.
Statistical Analysis
A normalized effect size (ES) was calculated for each comparison, representing the percentage of improvement in the intervention group compared to the control group on a neurobehavioral scale adjusted for the sham animal value. Where sham animal values were not reported in the experiments, the difference between the intervention group and the control group were calculated as absolute mean differences. For each ES, a standard error and 95% confidence interval (CI) were calculated.
The proportions were combined using a random effects meta-analysis model (DerSimonian–Laird) with logit transformation to determine weighting. The analysis was stratified based on the study details. The statistical analysis was performed using the metafor package in R software (version 4.1.2). 30
The significant difference between n groups was assessed by partitioning heterogeneity and by using the χ2 distribution with n − 1 degrees of freedom. The
Results
Literature Search
We observed a substantial overlap in the search results obtained from different web databases used in this literature research, resulting in a total of 2512 references. After de-duplication based on titles and abstracts, 545 references remained to investigate the effect of ChABC as a treatment approach for SCI in animals. However, after conducting a thorough double screening (AK and RA), this was reduced to 102 studies and after carefully evaluating the abstracts and reading the full texts of these publications, we identified 58 unique studies meeting the eligibility criteria. The literature search process and screening for eligibility are presented in the PRISMA diagram below (Figure 1).
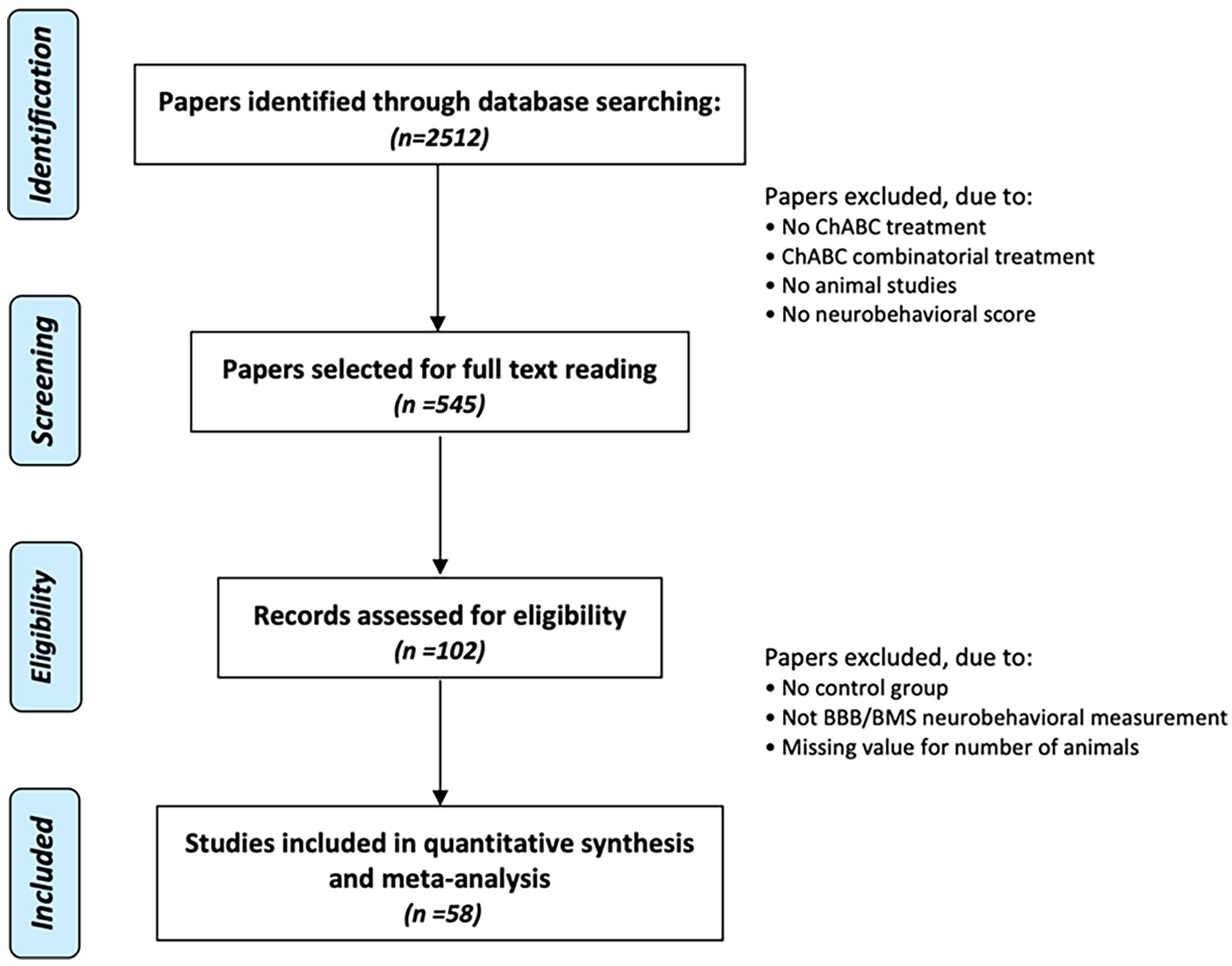
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow chart illustrating the literature search process and screening of papers included in this systematic review.
Characteristics of Studies
The 58 studies we identified encompassed a total of 124 experiments, including 50 studies with rats, 4 with mice, 2 with cats, and 2 with dogs. Within these 124 experiments, 76 involved female animals, 30 involved males, and 3 incorporated both genders, while 15 studies did not specify the genders of the animals. For neurobehavioral assessments, 42 experiments utilized the BBB score, and 2 employed the BMS score. Additionally, 15 experiments featured the ladder walking test, and 65 used alternative neurobehavioral assessment methods, such as the grip strength test. The injury models in these experiments encompassed 41 contusion injuries, 27 compression injuries, 30 hemisection injuries, 19 transection injuries, and 4 experiments reported dorsal column lesions and severe chronic injuries. One study included chronic severe SCI as an injury type and 1 study did not report the injury type.
Neurobehavioral Outcome
In the overall analysis, the administration of ChABC showed an improvement in neurobehavioral outcomes by 69.62% (95% CI = 43.28-95.96; see S1 and S2 in the Supplemental Materials). However, due to missing information about sham animals and given the observed heterogeneity among various neurobehavioral outcome scores within the overall analysis, we conducted a subgroup analysis focusing on the most commonly used neurobehavioral outcome score. Effectively, 58 studies were excluded, containing 4 studies in cats and dogs. The BBB/BMS score emerged as the most prevalent neurobehavioral assessment tool, utilized in 56.9% of all studies. The BBB/BMS-score is an open-field assessment tool for hindlimb function developed for rats (BBB = 21 points item score) and mice (9 points item score).35,36
The Es for this BBB/BMS-subgroup analysis was 15.92% (95% CI = 11.28-20.56) when comparing control and verum (treatment) animals. Furthermore, stratified meta-regression analysis revealed 5 key study characteristics that played a substantial role in accounting for the substantial heterogeneity observed across studies (Figure 2).
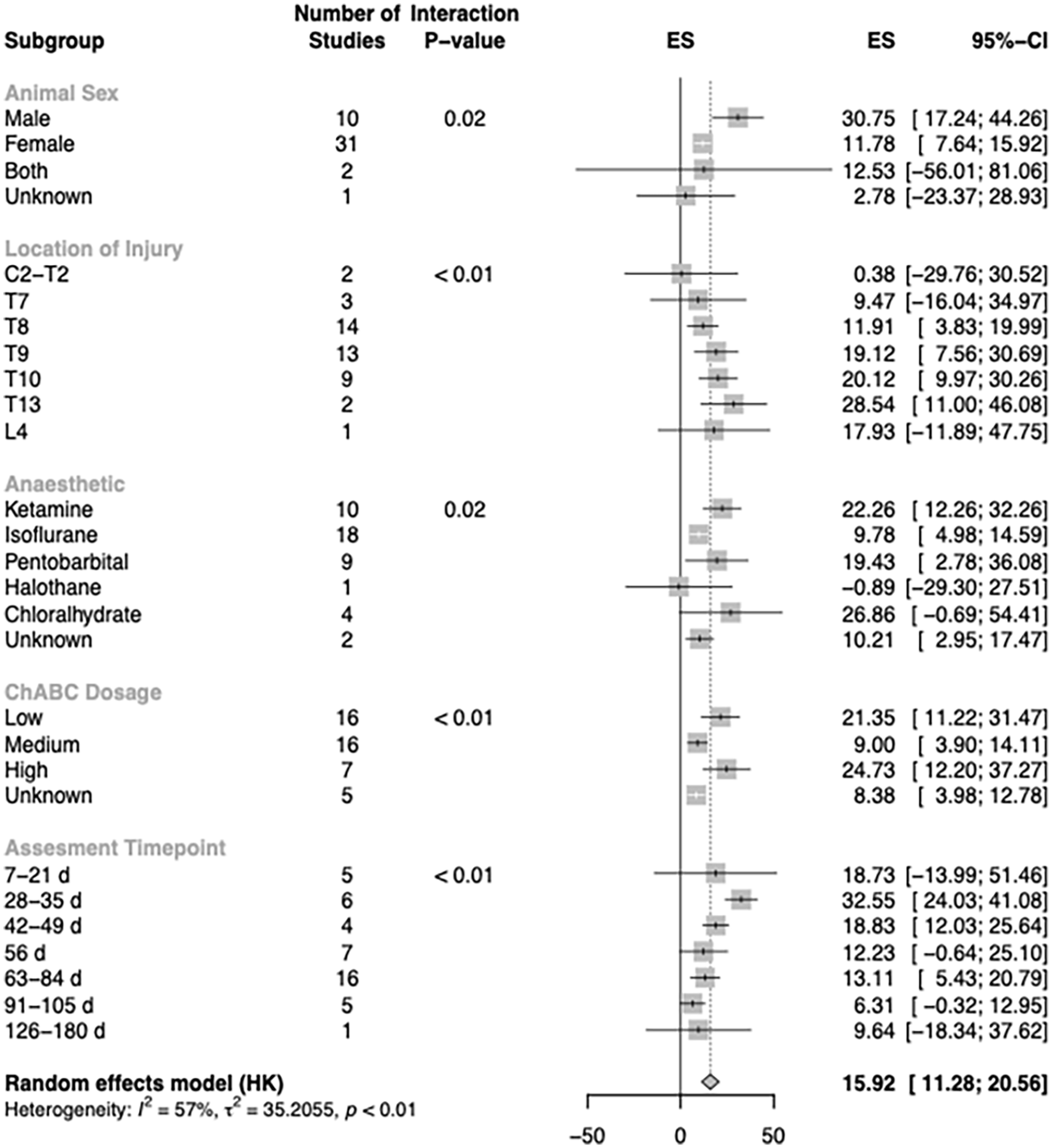
Subgroup-analysis of 44 experiments for the effect of ChABC treatment on the improvement of BBB/BMS-scores after spinal cord injury. Only subgroups accounting for a significant amount of heterogeneity are shown in this graph. Normalized effect sizes (ES) are displayed with their 95%-confidence interval.
Sex
Females were the predominant gender and indicated an ES reduced by two-thirds compared to the male animal models (ES male = 30.8% and ES female = 11.8%).
Injury Level
The most common location for lesions was found at spinal segment T8, followed by T9 and Th10. The level of injury significantly influenced the ES. More caudal lesions were associated with increased efficacy which confirmed the biological plausibility of the underlying data.
Anesthetic
Ketamin and Isoflurance were the most commonly used anesthetic in experiments. Cholorahlhydrate has the most impact on the ES but shows higher variability and less consistency. Ketamine, on the other hand, tended to have larger ESs resulting in decreased variability.
Dosage
Among different ChABC dosage, low and medium were the most commonly applied after SCI but high dosage resulted in slightly better ESs, followed by low dosage.
Time Points of Assessment
Notably, as the assessment period extended after SCI, the ES of the studied interventions exhibited a declining trend.
Analysis of Quality Assessment
Further analyses were conducted to assess the quality of the studies (Figure 3). About 75% of the studies indicated that they were executed with blinding. There appeared to be a trend for studies not reporting the blinded assessment of outcome to show worse neurobehavioral outcome, however statistical significance has not been reached.
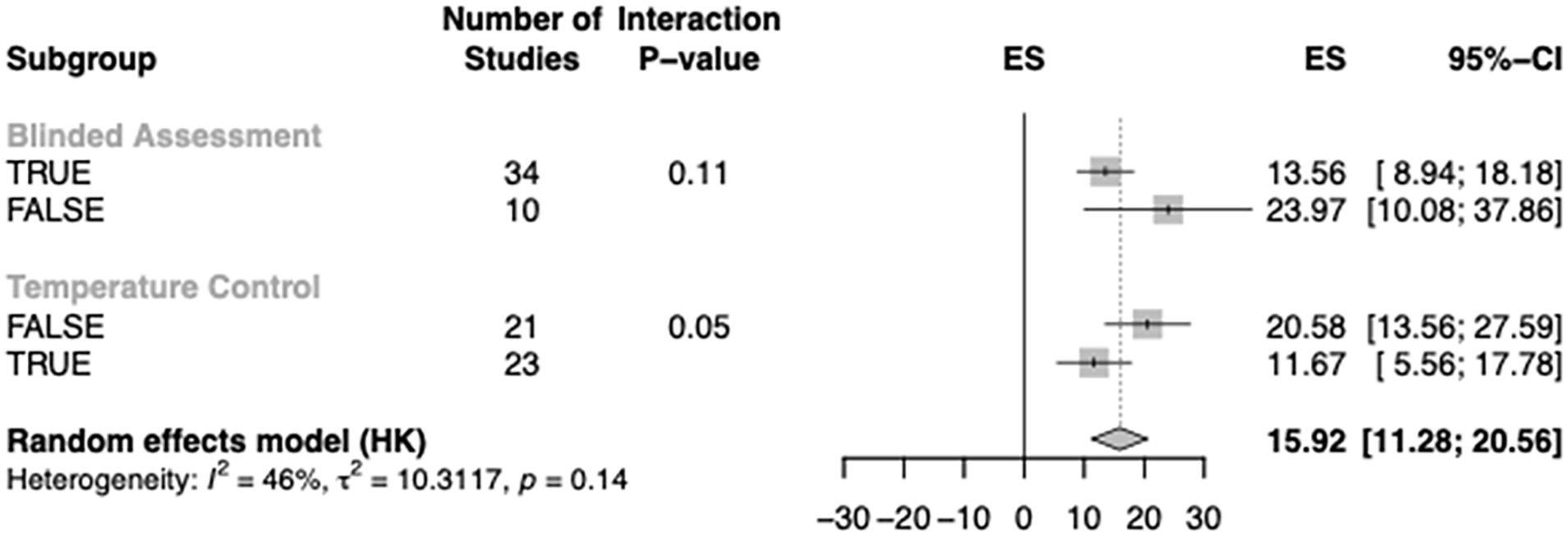
Subgroup meta-analysis of quality assessment of the studies. Normalized effect sizes (ES) are displayed with their 95%-confidence interval.
In addition, the reporting of temperature control during the experiments accounted for a significant amount of heterogeneity within the subgroup analysis. Studies reporting temperature control indicated reduced ESs compared to studies not reporting control of temperature.
Evaluation of Publication Bias
Funnel plotting and Egger regression analysis were used to identify small study effects and the potential impact of publication bias. Publication bias occurs when studies have been performed but their results have not been reported. To address potentially missing data, Trim and Fill analysis was conducted (Figure 4). A substantial amount of missing data was suggested. The observed ES (15.9% (95% CI = 11.3-20.6)) was reduced to 3.5% (95% CI = −2.4-9.5) by the imputation of 19 missing studies. However, subsequent Egger regression analysis yielded a non-significant
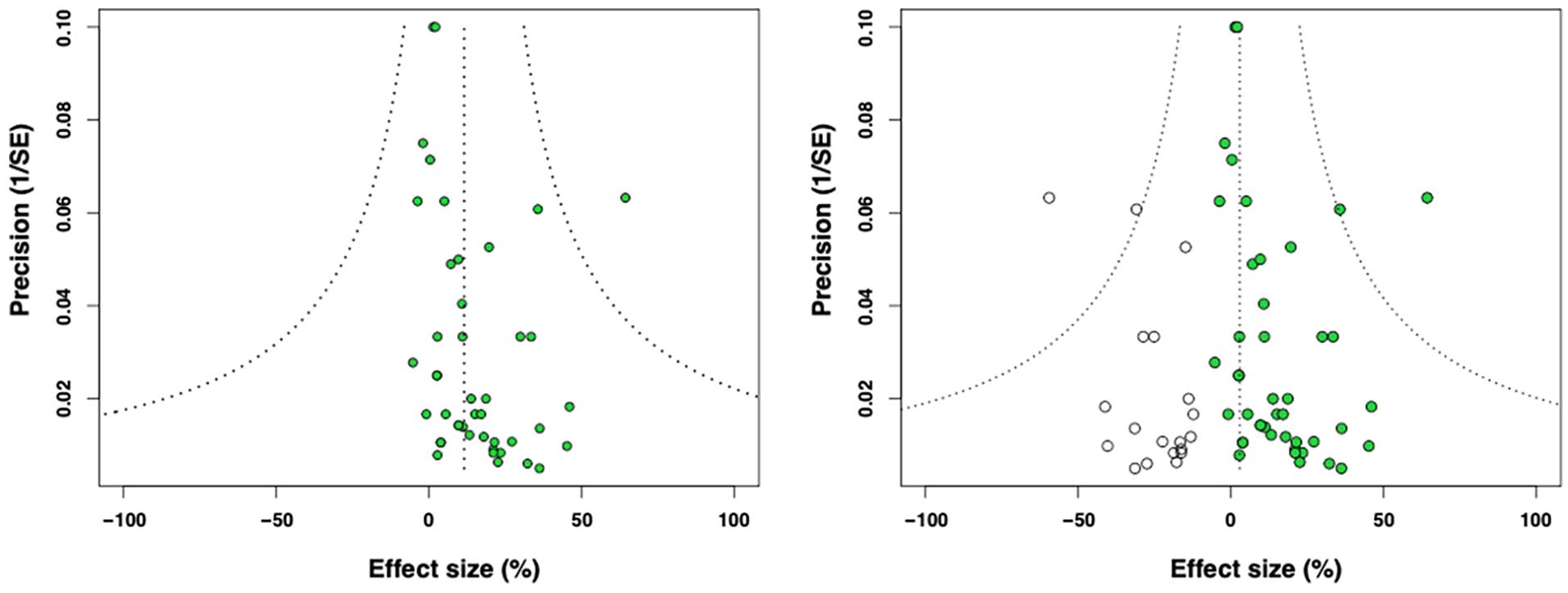
Funnel plots illustrating the precision (1 divided by the SEM,
Discussion
Overall Study Analysis
This preclinical systematic review and meta-analysis provide evidence of an improvement in locomotor behavior among animals with spinal cord injuries following ChABC treatment. The overall analysis of 58 studies including all animal species was followed by a subgroup analysis focusing on the most frequently used neurobehavioral score including 33 studies. We discerned empirical indicators suggestive of reporting/publication bias (missing data) and calculated the potential absence of 19 theoretical experiments, suggesting an absolute overestimation of efficacy by 12.4%.
The overall analysis included data from 4 different species with a total amount of 1066 animals. Due to partially missing data on sham animals (usually animals without lesions to the spinal cord but with bony decompression of the spine) the ESs within the overall analysis were not adjusted to the sham values and remained comparably high. In addition, the utilization of multiple neurobehavioral outcome scores for evaluating recovery after SCI complicated inter-study comparisons. We observed a substantial amount of underlying heterogeneity within the overall dataset and proceeded with a subgroup analysis of the most frequently used neurobehavioral score to increase homogeneity and draw more robust conclusions from the meta-analysis.
Subgroup Analysis to Increase Homogeneity
Within the BBB/BMS-subgroup analysis, the animals’ gender indicated increased neurobehavioral outcome for male animals compared to females. This has been described in previous work. 37 Increased neuropathic pain in female animal models 38 and sex differences due to disrupted neuroinflammation 39 have been reported to contribute to altered locomotor function. However, the absolute number of male animals used in our work was clearly lower compared to female animals.
Ketamine and Isoflurane were the most frequently used anesthetics, and ketamine revealed more than twice the ES compared to Isoflurane. Although the potential neuroprotective properties of ketamine as a non-competitive antagonist of
Differences in the outcome were dependent on the level of SCI confirming the biological plausibility of the meta-analysis. Cervical and high thoracic injuries showed lower ESs compared to injuries located more caudally. Thoracic levels T8 to T10 were the most prevalently used (33 of 58 studies, 57%). During the initial 7 weeks after SCI, there was a peak in the observed effects which subsequently declined, potentially indicating the gradual accumulation of adverse modifiers, such as acquired infections. In preclinical and clinical studies on SCI, acquired infections have previously been identified as an independent risk factor for impaired neurological outcome.41-43
Different ESs for the applied dosage of ChABC have been observed in the subgroup analysis. Studies monitoring the animals’ temperature during or after the experiment revealed a decreased efficacy. Temperature monitoring during SCI induction improves animals’ perioperative safety and represents 1 point on the quality checklist. This is in line with previous work where increased study quality has been demonstrated to be associated with decreased ESs.37,44
Within the sensitivity analysis of the subgroup dataset, Trim and Fill imputed a significant amount of publication bias overestimating the overall ES. A main source of missing data is publication bias and mostly attributed to unpublished studies that have been performed but not reported within the literature.45-47 The imputation of 19 potentially missing experiments led to a reduction of the overall efficacy from 15.9% to 3.5%, although this was not confirmed by Egger regression. Despite the discrepancy between the different methods of bias detection and given the limited number of studies included, underlying publication bias has to be taken into account. The translation of a relatively small ES of 15.9% which is potentially further reduced by underlying publication bias might decrease the probability of a successful translation into the clinics.
Limitations
The statistical methods employed in this study are comprehensive and are commonly utilized in meta-analysis/metascience research. 48 Nevertheless, they are subject to intense scrutiny and debate within the statistical community. We stress the cautious interpretation of conclusions drawn from individual statistical methods, such as funnel plot asymmetry alone. In funnel plots, the distribution of ESs is influenced by study variance but may not fully capture potential variations in true effects due to factors like diverse methods of SCI induction or varying animal models. 49
The robustness of the used neurobehavioral scale within the subgroup analysis has been discussed before. The standard BBB score comprises 21 items, and minor differences of a point or 2 may register as statistically significant but may lack functional significance in certain segments of the scale. Alternative transformations of the BBB score have been proposed 50 based on underlying raw data, which are not available for this meta-analysis. Nevertheless, these outcome measurement scales are widely utilized and acknowledged as the most informative and commonly employed neurobehavioral tools for assessing outcomes in SCI models.
Within this meta-analysis treatment effects of ChABC have been investigated affecting the extracellular matrix and potentially increasing neurite outgrowth in scar tissue. However, the multitude of potential extracellular and intraneuronal factors inhibiting axon regeneration and sprouting may be so extensive that targeting any single factor might offer only limited therapeutic benefit. Furthermore, any single inhibitory factor may produce effects by more than 1 molecular mechanism and several inhibitory factors may converge on the same intracellular signaling pathway. This might underscore the importance of focusing on combinatorial therapeutic approaches to achieve more effective outcomes.
Conclusion
This systematic review and meta-analysis evaluated the efficacy of ChABC therapy in experimental SCI and suggests a potential beneficial effect although substantial publication bias has been detected. Factoring in inter-individual variations, heterogeneity, reporting/publication bias (missing data), can reduce ES and should be taken into consideration when calculating/designing protocols for clinical translation. Researchers should carefully plan, execute, and report their studies to enhance translational robustness, following established guidelines such as ARRIVE 2.0 (www.arriveguidelines.org) and PREPARE (https://norecopa.no/prepare/). Furthermore, the regular use of the Open Data Commons for SCI repository 51 for sharing all data should be standard practice in spinal cord injury research to promote transparency and data accessibility.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241311337 – Supplemental material for Targeting Nerve Fiber Outgrowth Inhibition After Experimental Spinal Cord Injury: A Systematic Review and Meta-analysis of Chondroitinase ABC
Supplemental material, sj-docx-1-nnr-10.1177_15459683241311337 for Targeting Nerve Fiber Outgrowth Inhibition After Experimental Spinal Cord Injury: A Systematic Review and Meta-analysis of Chondroitinase ABC by Alireza Khanteymoori, Clayton Peterson, Roza Atamny, Marc Hohenhaus, Jürgen Beck, David W. Howells, Jan M. Schwab and Ralf Watzlawick in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to express our sincere appreciation to Carmen Fricke for her assistance in extracting data from papers, which contributed to the completion of this scientific study.
Author Contributions
Alireza Khanteymoori: Conceptualization; Formal analysis; Investigation; Methodology; Software; Visualization; Writing—original draft; and Writing—review & editing. Clayton Peterson: Investigation and Writing—review & editing. Roza Atamny: Investigation and Writing—review & editing. Marc Hohenhaus: Supervision; Visualization; and Writing—review & editing. Jürgen Beck: Supervision and Writing—review & editing. David W. Howells: Supervision and Writing—review & editing. Jan M. Schwab: Conceptualization; Supervision; and Writing—review & editing. Ralf Watzlawick: Conceptualization; Formal analysis; Funding acquisition; Investigation; Methodology; Supervision; Writing—original draft; and Writing—review & editing.
Data Availability
A study protocol was finalized in advance of any data collection. The study protocol is registered at the systematic review facility. The analysis plan is included in the study protocol. The analysis was conducted according to the plan. Study selection accounting for all experimental subjects is included within the figures according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for flow diagrams. Data from this study will be made available (as allowable according to institutional regulatory standards) by e-mailing the corresponding author. The analytic code used to conduct the analyses presented in this study are not available in a public repository. It can be made available upon request to the corresponding author. The authors agree or have agreed to publish the manuscript using the “Open Access” option under the appropriate license.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RW and AK receive funding from the Ministeriums für Wissenschaft, Forschung und Kunst, Baden-Württemberg, Germany.
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
