Abstract
Background
Unilateral hemispheric stroke can impair the ipsilesional motor performance, which is crucial for attaining optimal functional outcomes poststroke. However, the specific brain structures contributing to ipsilesional motor performance impairment remain unclear.
Objective
To explore the link between ipsilesional motor performance and the microstructural integrity of relevant neural pathways.
Methods
This study enrolled 60 consecutive patients in the early subacute phase of stroke recovery. Ipsilesional motor performance was assessed using the Box and Block Test. Multiple linear regression was used to evaluate the associations between ipsilesional motor performance and the microstructural integrity of relevant white matter tracts (Biomarker models) and cognitive function test scores (Cognition models).
Results
Biomarker models, including the genu of the corpus callosum, ipsilesional cingulum, fornix, uncinate fasciculus, superior longitudinal fasciculus, and contralesional inferior longitudinal fasciculus, showed a significant association with ipsilesional motor performance. Cognition models, including Mini-Mental State Examination and Trail Making Test-B, were significantly associated with ipsilesional motor performance. Final regression models (combined Cognition and Biomarker models) revealed that the performance time of Trail Making Test-B, in combination with biomarkers, including the genu of the corpus callosum, ipsilesional superior longitudinal fasciculus, and ipsilesional cingulum, predicted ipsilesional motor performance with high explanatory power (adjusted R2 = .721, .709, and .696, respectively).
Conclusions
This study demonstrated that executive function is associated with poststroke ipsilesional motor performance, as evidenced by the microstructural biomarkers involved in executive function. Our findings highlight that the comprehensive role of cognitive functioning rather than the motor system is closely linked to poststroke ipsilesional motor performance.
Keywords
Introduction
Unilateral hemispheric stroke not only causes contralesional hemiparesis but also impairs ipsilesional motor performance.1,2 Ipsilesional upper limb performance poststroke can exhibit slow movement speed, impaired motor control, and difficulty in sequencing complex movements, which can remain impaired in the chronic phase of stroke.1-4 Patients with stroke use their ipsilesional upper limb 2 to 6 times more frequently than their contralesional upper limb.5,6 The impairment of ipsilesional upper limb performance has an adverse effect on functional outcomes and the level of independence in activities of daily living.3,7,8
The underlying mechanisms of impairment of ipsilesional upper limb performance have been suggested from 2 aspects: poststroke cognitive impairment and changes in the motor system.1,4,9-11 Poststroke cognitive impairments affecting the planning and execution of movement and visuospatial perception have been identified as the possible causes of reduced ipsilesional upper limb performance.1,4,11,12 Earlier studies have indicated that motor-related neurological changes in the sensorimotor and motor control systems may be associated with impaired ipsilesional upper limb performance.9,10 Some evidence also suggests that interhemispheric connections or bihemispheric motor networks may have an impact on ipsilesional motor performance.13,14
However, convincing explanations based on the neural basis of microstructural organization or functional connectivity still need to be explored. Only a few studies have investigated the association between the impairment of ipsilesional motor performance and brain changes using functional neuroimaging techniques.13,15 In particular, there is no clear evidence to explain this phenomenon by evaluating microstructural changes in both ipsilesional and contralesional hemispheres poststroke. Identifying neuroimaging biomarkers related to ipsilesional upper limb performance is essential for setting target domains for conventional rehabilitative therapy, planning personalized neuromodulation therapy, and assessing the efficacies of diverse therapeutic strategies.
This study aimed to elucidate the association between the ipsilesional upper limb performance and the microstructural integrity of neural pathways of bilateral hemispheres. We explored specific white matter tracts responsible for the motor system, interhemispheric connections, and cognitive functions, including executive function and visuospatial perception, which may be potentially related to ipsilesional upper limb performance. We hypothesized that (1) unilateral hemispheric stroke impairs ipsilesional upper limb performance by affecting the microstructural integrity of both ipsilesional and contralesional hemispheres and their neural pathways, and (2) microstructural changes in specific white matter tracts would be associated with the degree of impairment of the ipsilesional upper limb performance.
Methods
Participants
This cross-sectional study enrolled a cohort from the inpatient rehabilitation facility of the Yeouido St. Mary’s Hospital, the Catholic University of Korea, in Seoul, Republic of Korea (Figure 1). The study population comprised 60 consecutive patients with stroke who underwent brain diffusion tensor imaging (DTI) upon transfer from the acute stroke unit to the inpatient rehabilitation facility between September 2018 and February 2023. The patients who met the following criteria were included: (1) aged >18 years, (2) first-ever ischemic or hemorrhagic stroke, (3) underwent comprehensive cognitive function tests, and (4) underwent functional assessments and DTI in the early subacute phase of stroke recovery (7 days-3 months poststroke). 16 Patients who—(1) had a bi-hemispheric stroke, (2) a history of prior ischemic or hemorrhagic stroke, and (3) a history of additional neurological disorders other than stroke were excluded. All patients underwent comprehensive rehabilitation at the hospital. The 60 age-matched controls were retrospectively enrolled from a cohort of healthy individuals from our institution who underwent DTI between September 2020 and February 2023. Controls were excluded if they had (1) a history of psychopathology or neurological disorders or (2) structural abnormalities on their scan. The Institutional Review Board of our institution approved the protocols for this study, which were in accordance with the Declaration of Helsinki. Because this study was retrospective and involved existing data, and did not breach subject confidentiality, the requirement for informed consent was waived.
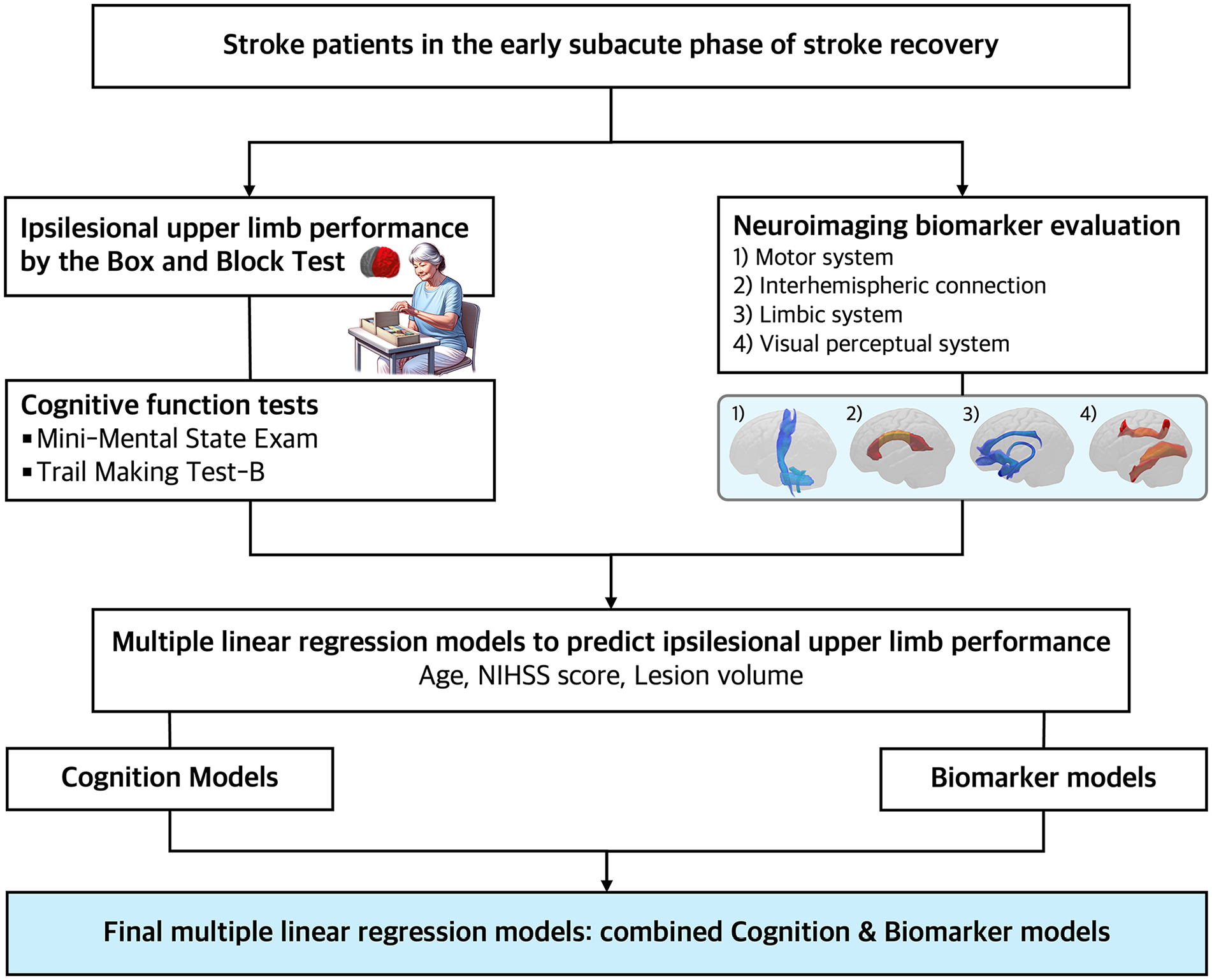
Study scheme.
Functional Assessments
Ipsilesional Upper Limb Performance
Each patient underwent the Box and Block Test to assess the ipsilesional upper limb performance. The Box and Block Test measures unilateral gross upper limb dexterity, which has excellent reliability and validity in patients with stroke. 17 Patients are instructed to move 2.5 × 2.5 × 2.5 cm3-sized wooden blocks as many as possible, 1 by 1, from 1 square compartment to another compartment of equal dimension for 60 seconds. The Box and Block Test is scored by counting the number of blocks carried over, and higher scores indicate better gross upper limb dexterity.
Cognitive Function Tests
The Mini-Mental State Examination (MMSE), ranging from 0 to 30, evaluated global cognitive functioning. 18 The Trail Making Test Part B, adapted for the Korean elderly, was administered to assess executive functioning.19,20 Patients are instructed to connect a set of 15 circles as quickly as possible while maintaining a consecutive sequence within 300 seconds. The test requires alternating between numbers from 1 to 8 and days of the week and is also randomly arranged. The examiner measures the time (in seconds) taken to complete the test, with shorter times indicating better performance.
DTI Acquisition
DTI was performed using a 3.0-T magnetic resonance imager (MAGNETOM® Skyra; Siemens, Erlangen, Germany) equipped with a 6-channel head coil. The data were acquired in the form of single-shot spin-echo echo-planar images, with axial slices covering the whole brain across 65 interleaved slices with a thickness of 2.0 mm (no gap; repetition time/echo time = 8900/41 ms; field of view = 256 × 256 mm2; matrix = 128 × 128; voxel size = 2 × 2 × 2 mm3 (isotropic), number of excitations = 1). Diffusion-sensitizing gradients were applied in 30 noncollinear directions with a b-value of 1000 ms/mm2. The images with b = 0 were scanned before acquiring diffusion-weighted images, and 31 volumes were acquired.
Image Processing
The images were processed using the FMRIB software library (FSL; version 5.0.9; http://www.fmrib.ox.ac.uk/fsl). Source data were corrected for eddy currents and head motion by registering first b = 0 images using affine transformation. Fractional anisotropy (FA) maps were prepared using DTIFIT software. We applied nonlinear registration of all FA images into a 1 mm3 MNI152 standard space.
DTI Analysis
Region of interest analysis was performed to evaluate the microstructural integrity of the white matter tracts. We classified neural functional systems related to ipsilesional motor performance into 4 categories for selecting specific white matter tracts. These categories included the motor system, interhemispheric connections, and limbic and visual perceptual systems. Subsequently, we selected representative white matter tracts for each system. The motor system included the corticospinal tract and the superior, middle, and inferior cerebellar peduncles. The genu, body, and splenium of the corpus callosum were selected for interhemispheric connections. The cingulum, fornix, and uncinate fasciculus represented the limbic system. The visual perceptual system included the optic radiation and the superior and inferior longitudinal fasciculi. We extracted the FA values of each white matter tract by applying standardized templates from the Johns Hopkins University (JHU) DTI White Matter Atlas (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/Atlases). When the template of the JHU atlas did not reflect entire tract integrity, we adopted the template obtained from the XTRACT Atlas derived from the Human Connectome Project (Supplemental Figure 1). 21
Statistical Analysis
The FA values of the white matter tracts between the patient and control groups were compared using independent t-tests. We employed simple and multiple linear regression models to explore the association between the ipsilesional upper limb performance and independent variables, including age, initial stroke severity (National Institutes of Health Stroke Scale [NIHSS]), degree of leukoaraiosis, lesion volume, cognitive function test scores, and the FA values of the specific white matter tracts. Three distinct regression models were constructed: Biomarker models, Cognition models, and combined models including both biomarkers and cognitive function test scores. Each model evaluated the impact of the structural integrity of specific white matter tracts, cognitive functioning, or both on ipsilesional upper limb performance. The multiple regression models that integrated both the structural integrity of biomarkers and cognitive function test scores were used to determine the comprehensive impact of these factors on ipsilesional upper limb performance. Multiple linear regression models were constructed using the stepwise selection method, and the criteria for the stepwise procedure were set with a probability of F = 0.05 for entry and F = 0.10 for removal. The multicollinearity of the independent variables was tested using the variance inflation factor to avoid overfitting the regression models. Adjusted R2 was adopted as a measure of explanatory power to prevent overfitting due to the number of independent variables and to compare the relative explanatory power of models. All tests were 2-sided, and statistical significance was set at P < .05. The corrected P-value threshold was applied to determine the significance of multiple correlations between the ipsilesional upper limb performance and FA values of 13 white matter tracts in the simple linear regression analyses (corrected Pthreshold = .05/13). All statistical analyses were conducted using IBM SPSS Statistics for Macintosh, Version 29.0.1.0, IBM Corp., Armonk, NY, USA.
Data Availability
The clinical and imaging data supporting the findings of this study are available from the corresponding author upon reasonable request.
Results
Demographic and Clinical Characteristics
Table 1 presents the demographic and clinical characteristics. The mean NIHSS score at stroke onset was 11.25 ± 5.42, indicating moderate to severe stroke. The mean modified Barthel index score was 29.07 ± 27.50, indicating that most patients required moderate assistance or more to perform basic activities of daily living. The strokes in this study were primarily ischemic, with lesions limited to the MCA territory, and hemorrhagic, with lesions in the basal ganglia, thalamus, and frontal lobe (Figure 2).
Demographic and Clinical Characteristics of the Patients.

Abbreviations: NIHSS, National Institutes of Health Stroke Scale; FMA-U, Fugl -Meyer Assessment of the upper extremity; MMSE, Mini-Mental State Exam; MBI, Modified Barthel Index.
Median value (interquartile range).
The Modified Barthel Index scores represent post-stroke functional and disability status by measuring the ability to perform activities of daily living. A lower score indicates more severe functional impairment.
Days since stroke onset.
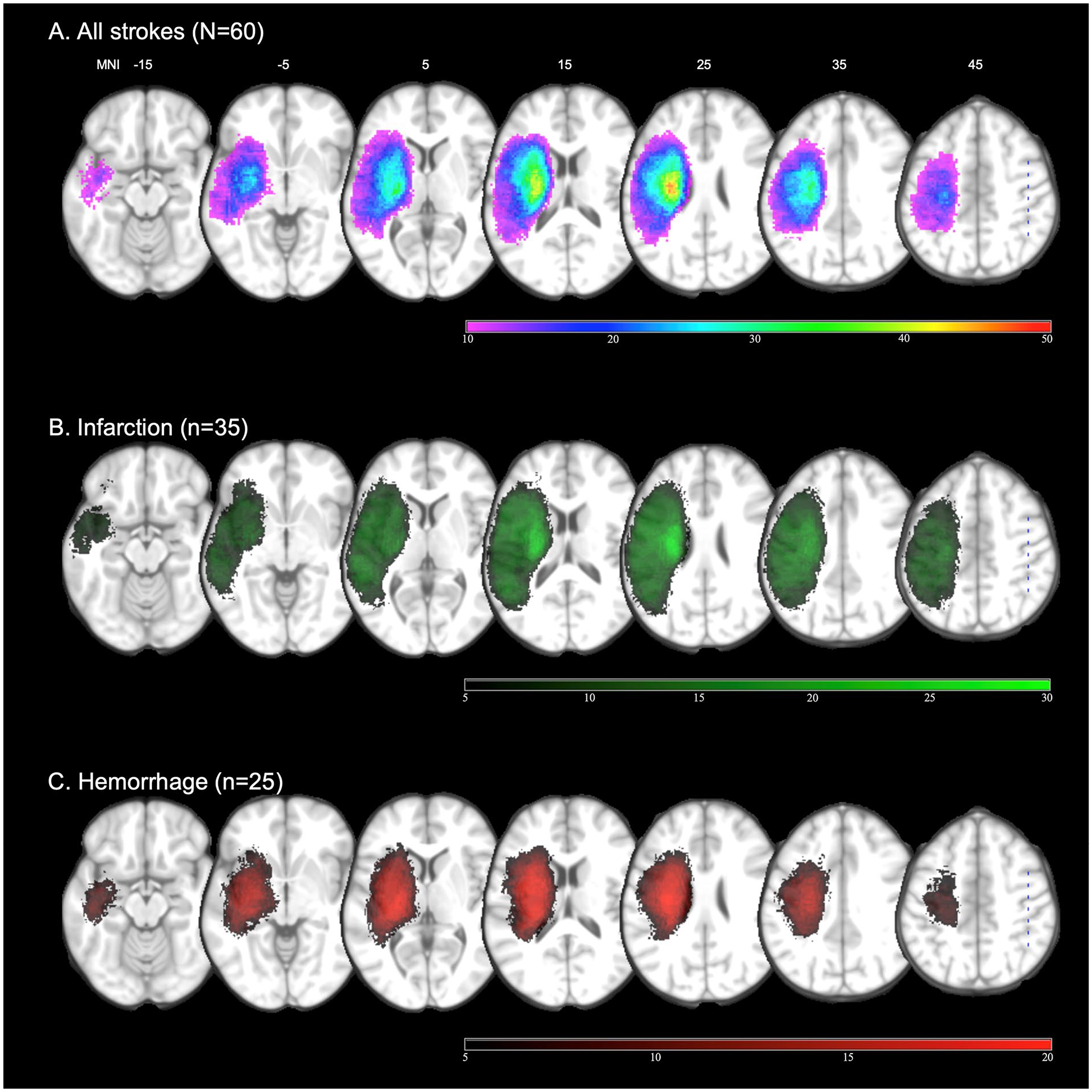
Lesion density map of the study population (z coordinates from −15 to +45 in the Montreal Neurological Institute space). (A) Lesion overlap from all patients (N = 60). (B) Infarction (n = 35, green). (C) Hemorrhage (n = 25, red). Left hemispheric lesions were flipped to the right side.
Comparison of Structural Integrity Between Patients and Healthy Controls
The FA values of the white matter tracts, representing the motor system, interhemispheric connections, and limbic and visual perceptual systems, were compared between the patient and control groups (Supplemental Tables 1 and 2). The patient group had a higher prevalence of vascular risk factors, including diabetes mellitus, hypertension, and atrial fibrillation, compared to the control group (P = .013, <.001, and .008, respectively). The FA values of all white matter tracts of the ipsilesional and contralesional hemispheres in the patient group were lower than in the control group (P < .001 for all white matter tracts).
Clinical Factors Affecting Ipsilesional Upper Limb Performance
Correlation between the ipsilesional upper limb performance and clinical variables, including age, NIHSS, degree of leukoaraiosis, and lesion volume, were evaluated through simple linear regression analyses (Supplemental Table 3). Age and NIHSS were negatively correlated with the ipsilesional upper limb performance (R = .504, P < .001; R = .412, P = .001, respectively). The degree of leukoaraiosis and lesion volume did not show a significant correlation (P = .123 and .070, respectively). The MMSE score as a general indicator for cognitive impairment revealed a positive correlation with the ipsilesional upper limb performance (R = .667, P < .001). The performance time of Trail Making Test-B showed a higher correlation with ipsilesional upper limb performance (R = .820, P < .001) than the MMSE score.
Multiple Linear Regression Models to Predict Ipsilesional Upper Limb Performance
White Matter Biomarker or Cognition Models
All neuroimaging biomarkers representing structural integrity of the motor system, interhemispheric connections, limbic system, and visuospatial perception were associated with the ipsilesional upper limb performance using simple linear regression analyses (Table 2, Supplemental Figure 2). Table 3 presents the results of the Biomarker and Cognition models by multiple linear regression. Among the biomarkers representing the motor system, only the middle cerebellar peduncle showed a significant association (adjusted R2 = .507), and bilateral corticospinal tracts and bilateral superior and inferior cerebellar peduncles were not significant. The Biomarker models, including the genu of the corpus callosum, ipsilesional cingulum, ipsilesional fornix, ipsilesional uncinate fasciculus, ipsilesional superior longitudinal fasciculus, and contralesional inferior longitudinal fasciculus, revealed a significant association with ipsilesional upper limb performance (adjusted R2 = .532, .519, .495, .511, .511, and .521, respectively). The Cognition models, including MMSE or Trail Making Test-B, revealed significant associations with ipsilesional upper limb performance (adjusted R2 = .582 and .661, respectively).
Association Between the Ipsilesional Upper Limb Performance and Neuroimaging Biomarkers using Simple Linear Regression Analyses.
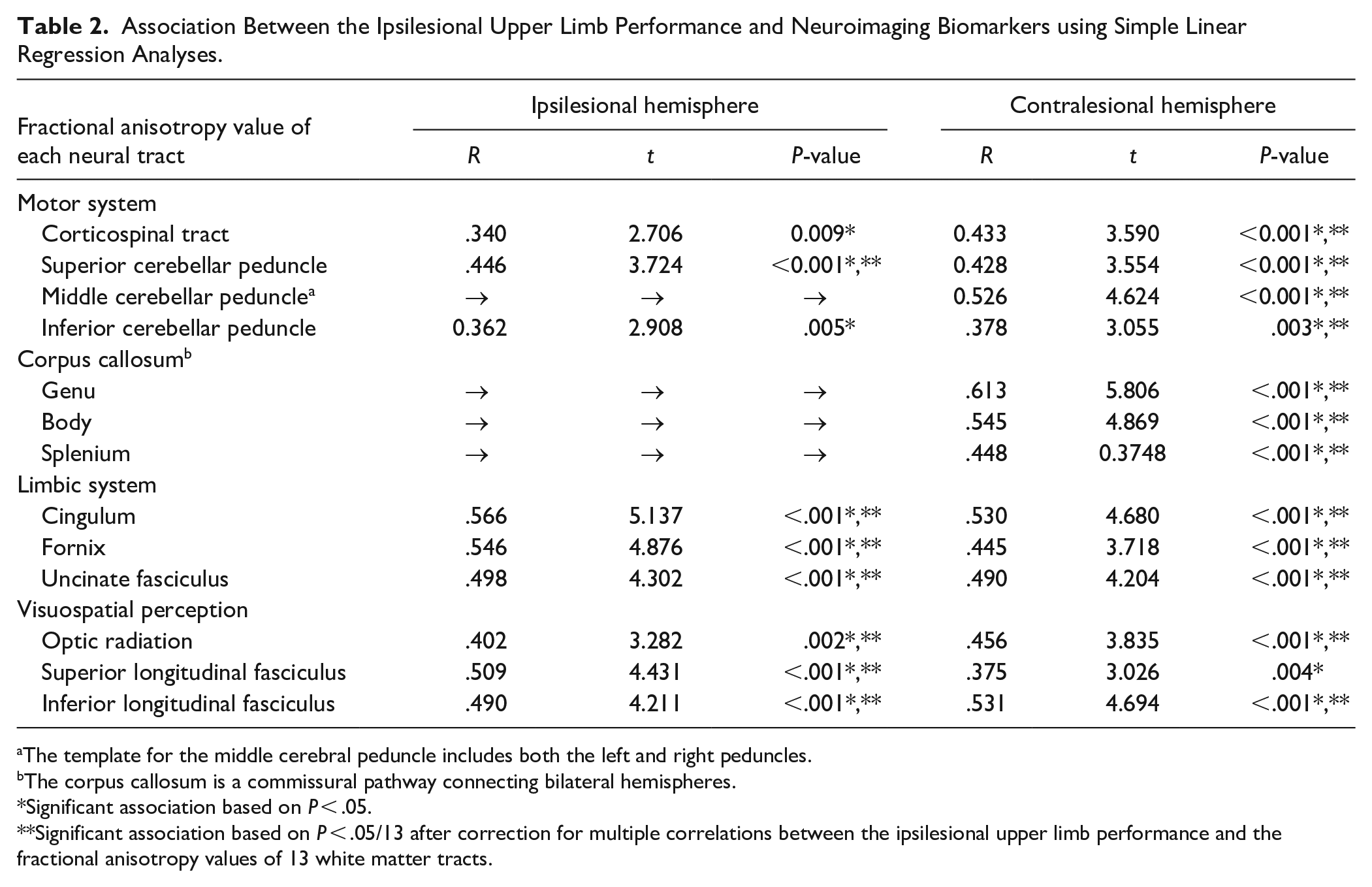
The template for the middle cerebral peduncle includes both the left and right peduncles.
The corpus callosum is a commissural pathway connecting bilateral hemispheres.
Significant association based on P < .05.
Significant association based on P < .05/13 after correction for multiple correlations between the ipsilesional upper limb performance and the fractional anisotropy values of 13 white matter tracts.
Multiple Linear Regression Models to Predict the Ipsilesional Upper Limb Performance Using Neuroimaging Biomarkers or Cognitive Function Test Scores.
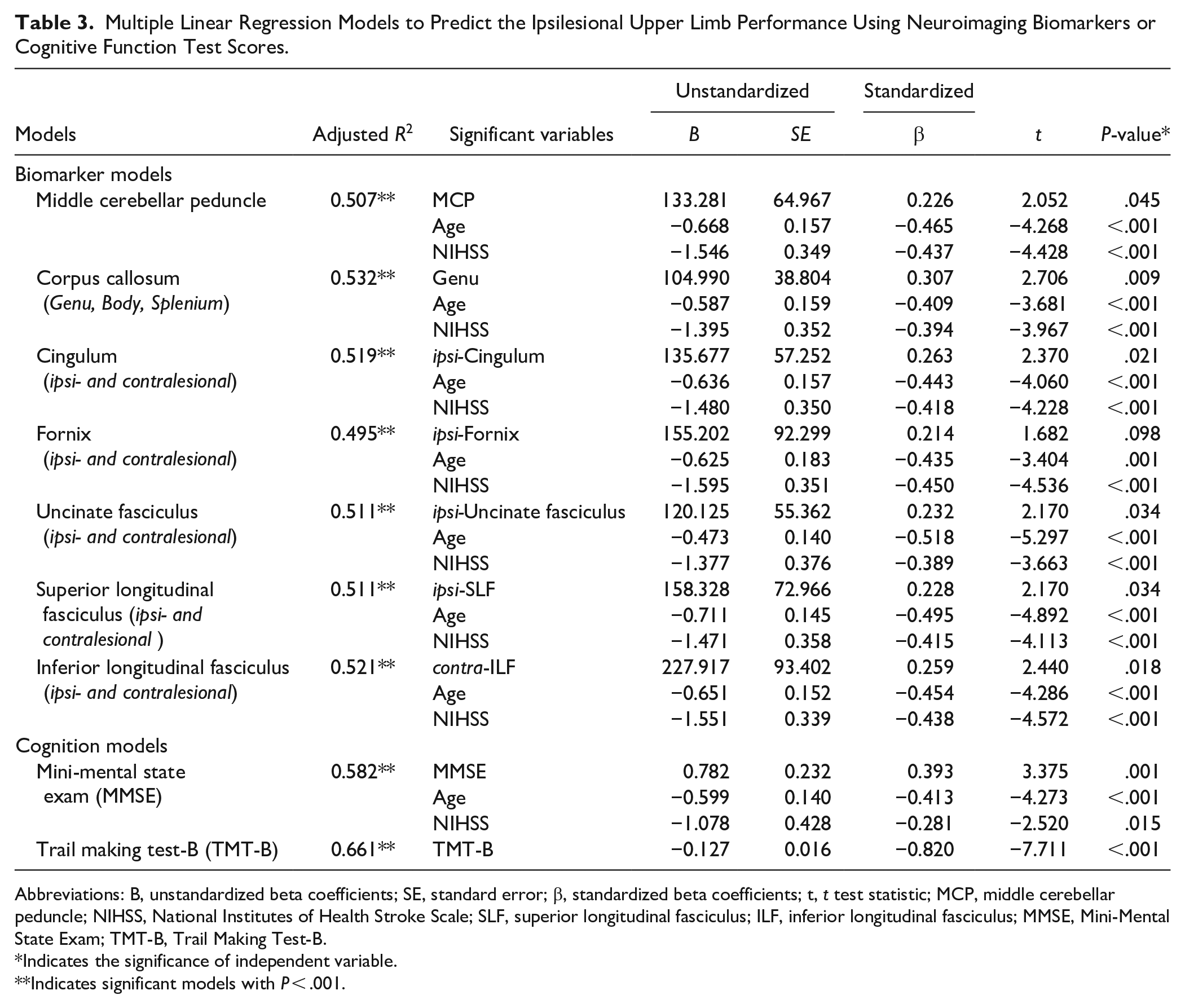
Abbreviations: B, unstandardized beta coefficients; SE, standard error; β, standardized beta coefficients; t, t test statistic; MCP, middle cerebellar peduncle; NIHSS, National Institutes of Health Stroke Scale; SLF, superior longitudinal fasciculus; ILF, inferior longitudinal fasciculus; MMSE, Mini-Mental State Exam; TMT-B, Trail Making Test-B.
Indicates the significance of independent variable.
Indicates significant models with P < .001.
Combined White Matter Biomarker and Cognition Models
Table 4 presents the significant multiple linear regression models to predict ipsilesional upper limb performance by combining the Biomarker and Cognition models. The model, including the performance time of Trail Making Test-B and the FA values of the genu of the corpus callosum, revealed the highest explanatory power for the ipsilesional upper limb performance (adjusted R2 = .721). The second strong explanatory power was shown by the model, including the performance time of Trail Making Test-B and the FA value of the ipsilesional superior longitudinal fasciculus (the adjusted R2 = .709), followed by the ipsilesional cingulum (adjusted R2 = .696). The model, including the MMSE score and the FA value of the contralesional inferior longitudinal fasciculus, also showed a good association with ipsilesional upper limb performance (the adjusted R2 = .605). Other models, by a combination of the cognitive function and white matter structural integrity, did not exhibit significant results.
Multiple Linear Regression Models to Predict the Ipsilesional Upper Lim Performance by Combined Neuroimaging Biomarkers and Cognitive Function Test Scores.
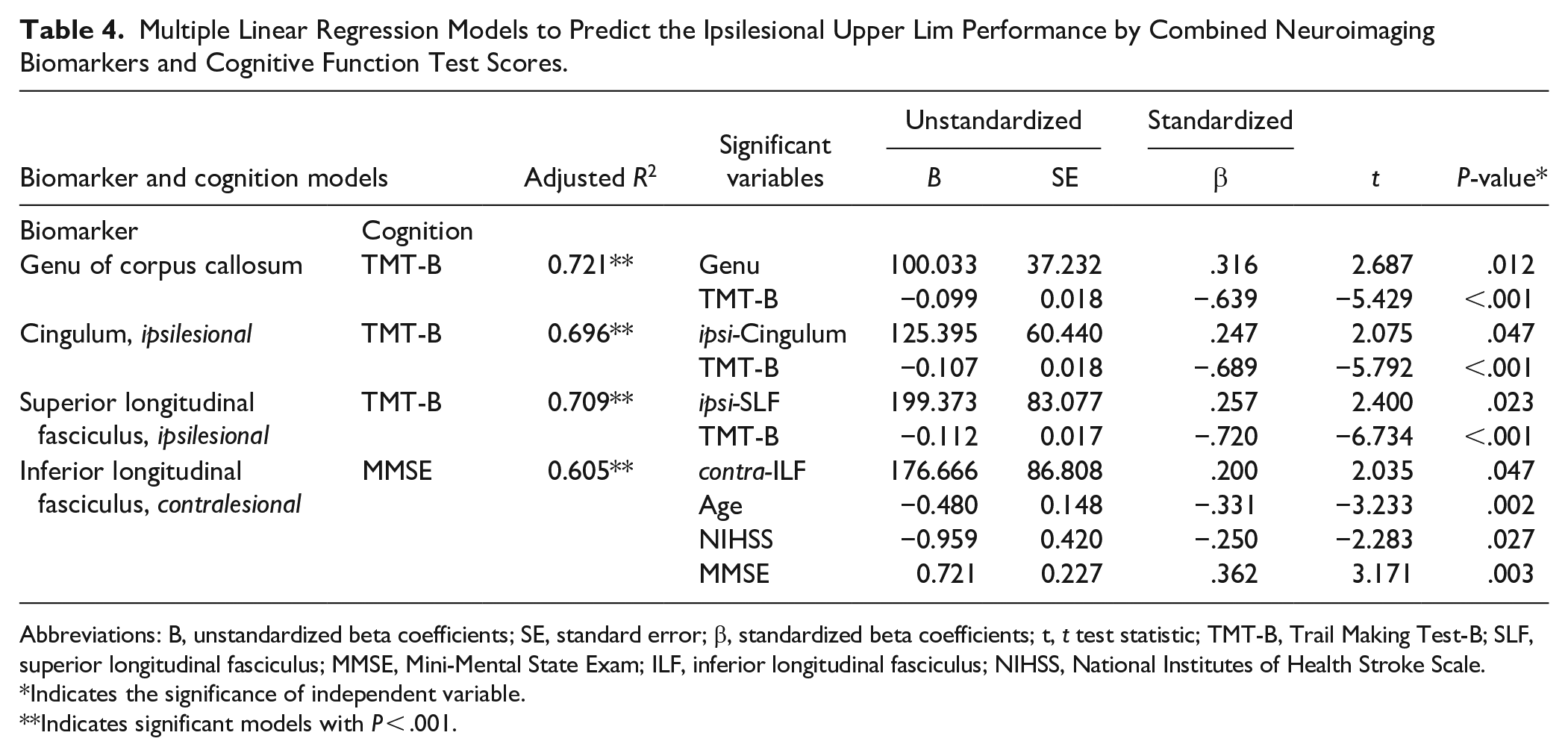
Abbreviations: B, unstandardized beta coefficients; SE, standard error; β, standardized beta coefficients; t, t test statistic; TMT-B, Trail Making Test-B; SLF, superior longitudinal fasciculus; MMSE, Mini-Mental State Exam; ILF, inferior longitudinal fasciculus; NIHSS, National Institutes of Health Stroke Scale.
Indicates the significance of independent variable.
Indicates significant models with P < .001.
Discussion
This study demonstrates that poststroke cognitive impairment, especially executive function, is associated with poststroke ipsilesional upper limb performance, as evidenced by the biomarkers of specific white matter tracts involved in executive function. The final multiple linear regression models revealed that the performance time of Trail Making Test-B, in combination with the white matter biomarkers, including the microstructural integrity of the genu of the corpus callosum or ipsilesional cingulum, predicted ipsilesional upper limb performance with high explanatory power (adjusted R2 of 0.721 and 0.696, respectively). Another notable finding was that the ipsilesional superior longitudinal fasciculus and contralesional inferior longitudinal fasciculus, which are tracts involved in visuospatial perception, were also associated with the ipsilateral upper limb upper limb performance. These visual perceptual pathways may have contributed to the utilization of visual information for motor execution.1,22,23 Conversely, motor-related white matter tracts revealed less association with the ipsilesional upper limb performance. In conclusion, our findings highlight that the comprehensive role of cognitive functioning rather than the motor system is closely linked to poststroke ipsilesional upper limb performance. Ipsilesional motor performance is important for performing activities of daily living after stroke, especially in patients with severe stroke. The study results imply that assessing poststroke cognitive impairments and planning rehabilitative strategies for specific cognitive functions will be crucial in enhancing the ipsilesional upper limb performance.
Impact of Executive Function on Ipsilesional Motor Performance
One of the important findings of this study is that cognitive functioning, especially executive function, significantly impacts ipsilesional upper limb performance. The Cognition models, including the MMSE or Trail Making Test B, showed high explanatory power for ipsilesional upper limb performance. Particularly, the performance time of Trail Making Test B alone exhibited a strong explanatory power with an adjusted R2 of 0.661. Several studies showed that the decline in ipsilesional upper limb performance is attributed to cognitive functioning, particularly in motor planning and execution.1,4,12 Poststroke patients with executive function impairment may have difficulty in planning, initiating, and sequencing a series of purposeful movements and demonstrate a reduced ability to follow complex treatment regimens during rehabilitation. 24 Those patients may also have difficulties in performing activities of daily living and significant restrictions in community participation, even if they have minimal physical restrictions due to stroke. 25 Furthermore, in patients with more severe poststroke motor impairment, the ability to perform activities of daily living using the ipsilesional upper limb is emphasized. 8 Enhancing executive function may improve poststroke outcomes by adopting various compensatory strategies in challenging daily environments.7,26
Evident Association Between Specific White Matter Tracts Engaging the Executive Function and Ipsilesional Motor Performance
A novel finding of this study was that the structural integrity of white matter tracts related to the cognitive-behavioral system could predict the ipsilesional upper limb performance more accurately than those of the motor-related white matter tracts. The genu of the corpus callosum and ipsilesional cingulum, which were not involved by the stroke, showed a high explanatory power for predicting ipsilesional upper limb performance. The genu of the corpus callosum and the cingulum are responsible for executive functioning in healthy and stroke populations.20,27-30 As expected from the structure-function relationship in healthy populations, relevant studies that found the genu of the corpus callosum and cingulum associated with executive functioning in patients with cerebrovascular diseases also support our results.31,32 Furthermore, our findings of the genu of the corpus callosum indicate that the contribution of interhemispheric prefrontal connection from both damaged and nondamaged hemispheres may play an important role in executive function and performing ipsilesional motor tasks. 30 The good explanatory role of the ipsilesional fornix and uncinate fasciculus comparable with the adjusted R2 value of ipsilesional cingulum also supports the engagement of the limb system, located outside of the stroke lesion, in eliciting the ipsilesional upper limb performance.22,28
The contralesional inferior longitudinal fasciculus and the ipsilesional superior longitudinal fasciculus also showed convincing explanatory power for ipsilesional upper limb performance in the biomarker regression models (adjusted R2 = 0.521 and .511, respectively). These 2 white matter tracts are responsible for visuospatial perception; more specifically, the inferior longitudinal fasciculus is associated with object perception and visual memory, and the superior longitudinal fasciculus is related to spatial perception and visuomotor function.30,33 These results are consistent with the previous research findings and suggest that the visual perceptual pathway affected by stroke may engage in ipsilesional upper limb performance or executive functioning.22,23,28 The fact that Trail Making Test-B is a test requiring visuospatial function has a common background with the previous research results showing that visual perceptual pathways are involved in global executive functioning. 30
Weak Relevance of the Motor Pathways to Ipsilesional Motor Performance
The corticospinal tract, a representative motor pathway, 34 and the superior, middle, and inferior cerebellar peduncles, which are core structures of the cerebro-cerebellar motor control loop, 35 were presumed to have a possible association with ipsilesional upper limb performance in bilateral hemispheres using simple regression analyses. However, these pathways, except the middle cerebellar peduncle, demonstrated weaker associations than age and NIHSS score in the multiple regression models and did not serve as significant predictors in the combined Biomarker and Cognition models. These results support our conclusion that the degree of impairment of ipsilesional upper limb performance is mainly determined by poststroke cognitive impairment rather than poststroke modification in the motor-related pathways.
Exclusive Perspectives Differing From Previous Studies on Ipsilesional Motor Performance
The impairment of ipsilesional upper limb performance can remain impaired in the chronic phase of stroke,3,8,36 although most patients show good recovery in the first few months.1,3,7 The impairment may persist according to the stroke severity.7,8 In line with previous studies,7,8 our results demonstrate that ipsilateral hemispheric stroke impairs the ipsilesional upper limb performance, and the degree of impairment was associated with the stroke severity. To clarify the role of specific white matter tracts on the ipsilesional upper limb performance, we applied the inclusion criterion of a fixed-time period poststroke: the early subacute phase of stroke recovery, 16 and included the NIHSS score, representing stroke severity, as a variable in the multiple regression models. By controlling these factors, including the time since stroke onset and stroke severity, we could more selectively identify the neuroimaging biomarkers contributing to the ipsilesional upper limb performance.
Another strength of this study is that most enrolled patients suffered moderate to severe strokes, requiring inpatient rehabilitation (mean NIHSS = 11.25). Their contralesional upper limb motor function, that is, hemiparesis, often remains impaired during the subacute phase of stroke recovery or disabled even after a sufficient recovery period. Hence, ipsilesional upper limb performance for these patients is crucial for conducting daily activities and poststroke functional outcomes. More pronounced impairment in ipsilesional upper limb performance in patients with more severe hemiplegic motor impairment implies that these patients will be more disadvantaged in terms of performing daily activities and returning to daily life. Because our results were derived from a group of patients for whom ipsilesional upper limb performance significantly influences long-term functional outcomes, we expect that our findings will help develop rehabilitative strategies to enhance ipsilesional upper limb performance for these patients. Future investigations into rehabilitation approaches, such as cognitive rehabilitation focused on executive function or noninvasive stimulation targeting specific white matter tracts with reduced structural integrity, as identified in this study, will be warranted to determine whether these approaches can improve ipsilesional upper limb performance and poststroke outcomes. For example, the genu of corpus callosum contains the forceps minor, which connects the bilateral dorsolateral prefrontal cortices responsible for executive function. Neuromodulatory interventions targeting this pathway could be a potential direction for future research to enhance ipsilesional motor performance.
Study Limitations
Some study limitations need to be addressed. This study analyzed both ipsilesional and contralesional white matter tracts to assess their role in ipsilesional motor performance. Compared to the healthy control group, the patient group exhibited reduced structural integrity in all white matter tracts, both ipsilesional and contralesional. These findings suggest that unilateral hemispheric stroke may have a comprehensive poststroke influence on the contralesional hemisphere. However, these results should be interpreted with caution, as the prevalence of vascular risk factors affecting white matter structural integrity was higher in the patient group than in the control group.
Furthermore, we did not evaluate the influence of hemispheric differences of white matter tracts. Several studies have reported the lateralized hemispheric contribution as the underlying mechanism of ipsilesional movement control poststroke.1,6,37 Left hemispheric stroke can lead to impaired coordination of multiple joint movements, while patients with right hemispheric stroke can show errors in the final positioning of movement.1,37 However, there is controversy regarding whether hemispheric specialization exists in ipsilesional movement control.2,8,36,38 Future larger-scale studies are warranted to determine whether the significant biomarkers we present in this study have hemisphere-dependent roles.
All enrolled patients were indicated for comprehensive cognitive assessments, but some patients with severe poststroke cognitive impairment could not perform or complete some tests requiring higher cognitive function. Therefore, the regression models included only the MMSE and Trail Making Test-B results. Furthermore, discriminating executive function from other cognitive domains is often challenging. The Trail Making Test-B is a representative test that assesses executive functioning. However, performing this test also requires other cognitive domains, such as attention, visuospatial perception, and working memory. Despite these challenges, the fact that the genu of the corpus callosum, cingulum, and superior longitudinal fasciculus are white matter tracts that engage executive function and that the results of the Trail Making Test-B explain ipsilesional upper limb performance through regression models supports the conclusion of our study.
Conclusions
Ipsilesional upper limb performance is a crucial factor in attaining maximal functional outcomes poststroke. Performing activities of daily living using the ipsilesional upper limb or functional movements that require bilateral coordination of the upper limbs is integral to poststroke rehabilitation, especially as the stroke severity increases. This study provides the evident associations between the ipsilesional upper limb performance and specific white matter tracts, such as the genu of the corpus callosum, cingulum, and superior longitudinal fasciculus, which microstructural biomarkers are responsible for executive functioning. Moreover, the multiple regression models, which include cognitive assessment results and microstructural biomarkers of specific white matter tracts, could predict the ipsilesional upper limb performance with high explanatory power. This study’s results emphasize that poststroke ipsilesional upper limb performance may be primarily determined by poststroke cognitive functioning, especially executive function, rather than by the motor system, and help elucidate the underlying neural mechanisms unclear in previous studies. We expect that the biomarkers presented in this study will be helpful in future studies aimed at developing personalized neurorehabilitation strategies and evaluating their therapeutic efficacies.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241309580 – Supplemental material for Poststroke Ipsilesional Motor Performance: Microstructural Biomarkers and Their Associations With Executive Function
Supplemental material, sj-docx-1-nnr-10.1177_15459683241309580 for Poststroke Ipsilesional Motor Performance: Microstructural Biomarkers and Their Associations With Executive Function by Youngkook Kim, So Yeon Jun, Jeehae Oh, Jaeun Koo and Eunji Lee in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
None.
Author Contributions
Youngkook Kim: Conceptualization; Data curation; Formal analysis; Methodology; Visualization; Writing—original draft; Writing—review & editing. So Yeon Jun: Data curation; Formal analysis; Methodology; Writing—review & editing. Jeehae Oh: Conceptualization; Visualization; Writing—review & editing. Jaeun Koo: Data curation; Formal analysis; Writing—review & editing. Eunji Lee: Methodology; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
