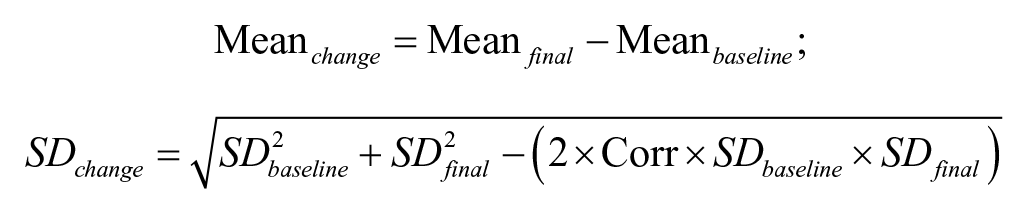Abstract
Background
Transcranial magnetic stimulation (TMS) is a non-invasive neuromodulation technique that has been closely examined as a possible treatment for Parkinson’s disease (PD). Owing to various rTMS protocols and results, the optimal mode and suitable PD symptoms have yet to be established.
Objectives
This study intends to systematically evaluate the efficacy of rTMS intervention and identify optimal stimulation protocol of rTMS for specific motor symptoms.
Methods
PubMed and web of Science databases were searched before January 2022. Eligible studies included sham-controlled and randomized clinical trials of rTMS intervention for motor dysfunction in patients with PD. Standard mean difference (SMD) was calculated with random-effects models. The effects of rTMS on motor symptoms were mainly estimated by the UPDRS-III.
Results
A total of 1172 articles were identified, of which 32 articles met the inclusion criteria for meta-analysis. The pooled evidence suggested that rTMS relieves motor symptoms of patients with PD (SMD 0.64, 95%CI [0.47, 0.80]). High frequency stimulation on M1 is the most effective mode of intervention (SMD 0.79, 95%CI [0.52, 1.07]). HF rTMS has significant therapeutic effects on limbs motor function (SMD 1.93, 95%CI [0.73, 3.12] for upper limb function and SMD 0.88, 95%CI [0.43, 1.33] for lower limb function), akinesia (SMD 1.17, 95%CI [0.43, 1.92), rigidity (SMD 1.02, 95%CI [0.12, 1.92]) and tremor(SMD 0.91, 95%CI [0.15, 1.67]).
Conclusion
rTMS therapy is an effective treatment for motor symptoms of PD and the individualized stimulation protocols for different symptoms would further improve its clinical efficacy.
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disease, characterized by resting tremor, bradykinesia, rigidity, gait disorder, and postural instability.1,2 It is estimated that 6 to 10 million people worldwide have PD, affecting all races and ethnicities. 3 Pharmacologic therapies such as dopaminergic medications can provide relief in some cases. However, most patients develop complications after several years of treatment, including dyskinesia and motor fluctuations. 2 Deep-brain stimulation surgery is approved by Food and Drug Administration for select indications such as medication-refractory tremors whereas less than 5% of the PD population may be eligible for the procedure. 4
During the past two decades, repetitive transcranial magnetic stimulation (rTMS), a painless, non-invasive, well-tolerated technique of brain stimulation, has been closely examined as a possible treatment for PD.5,6 It delivers repeated magnetic pulses to a specific brain area through a stimulation coil placed over the scalp. The repeated magnetic pulses can not only alter excitability at the site of stimulation but also influence brain regions anatomically connected to the stimulation site, which provide profound influence on the characteristics of brain circuitry. 7 The progressive loss of dopaminergic neurons in PD results in functional disruption within the cortico-basal ganglia–thalamocortical motor circuit. 8 In particular, there is an excessive inhibition of thalamocortical projections to various cortical targets, including the primary motor cortex (M1), the supplementary motor cortex (SMA), and the dorsolateral prefrontal cortex (DLPFC). Abnormal neural activities in these cortical areas have also been observed in neuroimaging studies. Based on the neuromodulation effect, TMS has become a potentially effective therapeutic strategy for PD. 9
Several controlled and uncontrolled studies have validated the therapeutic effect of rTMS on PD,10-43 especially on motor symptoms. However, most of them have involved small sample sizes and vary greatly in outcome measures, inclusion/exclusion criteria, methods of sham-TMS, and monitoring safety and tolerability. Despite various motor symptoms occurring in PD, few reviews evaluated the effect for specific symptoms. It is meaningful to figure out the effects of rTMS on different PD symptoms. The objectives of this meta-analysis are to (1) systematically evaluate the efficacy of rTMS on motor dysfunction in PD from randomized clinical trials (RCTs), (2) identify factors of rTMS protocols that may moderate effects, and (3) find out what symptoms can provide better relief after rTMS.
Methods
Search Strategy
We adhered to the recommendations made by the Meta-Analysis of Observational Studies in Epidemiology Group, the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 guidelines, 44 and the Cochrane Collaboration definition for systematic review and meta-analysis. Articles published before January 2022 from PubMed, Cochrane Library, and the Web of Science were systematically searched. The following search strategy was used: ((((transcranial magnetic stimulation) OR TMS) OR noninvasive brain stimulation)) AND ((((((Parkinson disease) OR Parkinson) OR Parkinson’s) OR Parkinson’s disease) OR parkinsonism) OR parkinsonian).
Selection Criteria
Eligible studies were included if they simultaneously fulfilled the predefined criteria. These studies (1) were randomized controlled trials in which the PD participants were given TMS or sham stimulation; (2) involved patients who reported motor symptoms of PD 45 ; and (3) included motor scales and standard deviation or enough data to calculate these figures. Studies were excluded if they did not meet the inclusion criteria. Second-hand unoriginal research (reviews, meta-analyses, commentaries, letters, reports, conference abstracts, and editorials) and duplicated studies were excluded.
Data Extraction and Quality Assessment
The following information was extracted from each study: first author’s name, year of publication, country, patient source, age, gender proportion, duration of disease, H&Y (Hoehn and Yahr Stage), TMS protocol, evaluation method, evaluation results, and standard deviation. Data extraction and quality assessment were conducted by two independent investigators, and any disagreement was resolved by a third investigator. If >1 article was found to have used the same data, we chose the one with a higher quality score, and where the quality score was equal, we chose the study with the larger sample size. Two authors independently evaluated the eligibility of all studies. If there was disagreement regarding whether to include some articles, these articles would be further evaluated by a third author and discussed in detail until an agreement was reached.
If the mean and SD of change scores were shown in the articles, they were extracted. If the mean and SD of the change scores were not clearly described in the article, the change scores were calculated by using the following formula based on the principles of the Cochrane Handbook for Systematic Reviews of Interventions 46
If the studies provided the mean values and the standard errors of the means or reported original materials of each patient, then we calculated the mean values and SDs based on the principles of the Cochrane Handbook for Systematic Reviews of Interventions. If the outcome was reported only as a graph, data were extracted by using the software GetData Graph Digitizer.
We used the Physiotherapy Evidence Database (PEDro) scale to quantify the quality of the studies. 47 The PEDro scale scores 11 items as either present or absent. The final score is the number of positive answers to all questions. We considered a PEDro score of: < 4 are considered “poor”, 4 to 5 are considered “fair”, 6 to 8 are considered “good”, and 9 to 10 are considered “excellent”.
Statistical Analyses
Effect Size Calculation
We used standardized mean difference (SMD [Cohen d]) to express the size of the rTMS effect on motor symptoms mainly measured by the UPDRS-III. A random-effect model was used to calculate pooled effect sizes and test whether the mean effect size was significantly different from zero (P ≤ 0.05, 2-tailed). The mean effect was expressed as SMD with 95%CIs. If a study had multiple effect sizes from the same patient group (eg, short-term and long-term rTMS effects), we obtained one mean effect size across multiple effect sizes within this study.
Heterogeneity Analysis
We used the Q statistic and the I2 to assess the statistical heterogeneity. A probability value of P ≤ 0.05 and an I2 value of greater than 40% is indicative of the heterogeneity of these studies as the values exceed what is expected by chance.
Publication or Selection Bias
Publication or selection bias was evaluated by the Egger test of asymmetry. In the absence of publication or selection bias, effect sizes were symmetrically distributed around the overall mean effect size, since the sampling error was random. The Egger test evaluates whether the amount of asymmetry is significant.
Risk of Bias Assessment in Individual Studies
We assessed the risk of bias using the Cochrane risk of bias assessment tool outlined in chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0. The Cochrane tool classifies studies as having low, high, or unclear risk of bias in the following domains: selection bias, performance bias, detection bias, attrition bias, reporting bias, and carryover effect.
Subgroup Analysis
For subgroup analysis, our prespecified comparisons included rTMS site, rTMS frequency, patients’ status, sessions, the interaction between rTMS site and rTMS frequency.
Results
A total of 823 articles from PubMed, 341 articles from Cochrane Library, and 1551 articles from Web of Science were retrieved from the above keywords. After removing duplicates, patents, book sections, and other language records, 1506 articles were screened by title and abstract for possible inclusion. Out of them, 269 articles were selected for full-text examination. Finally, 32 articles were included according to the pre-determined inclusion criteria (Figure 1) (Table 1 and Table 2).10-43
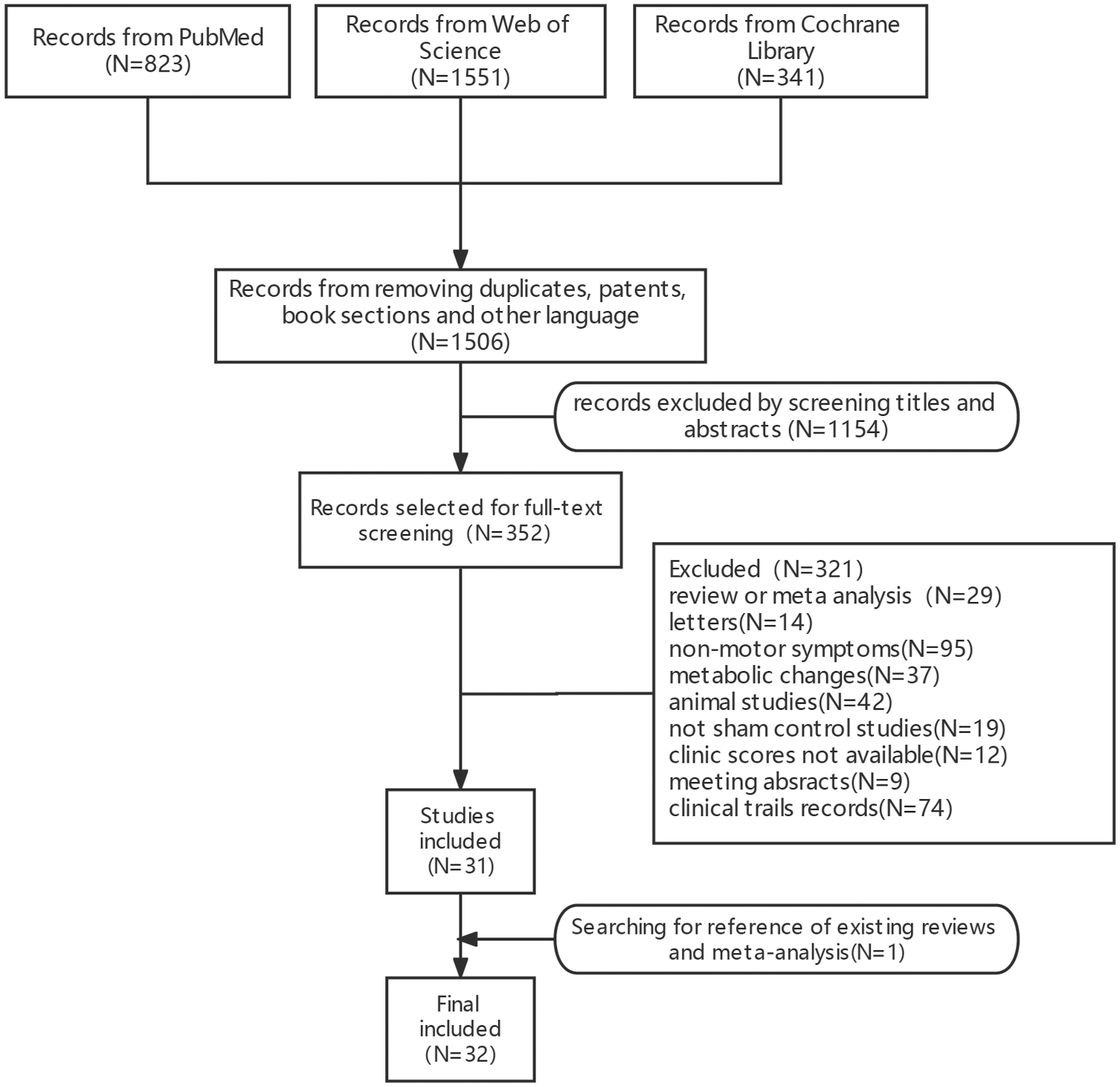
Flowchart of study selection.
Characteristics of included studies.
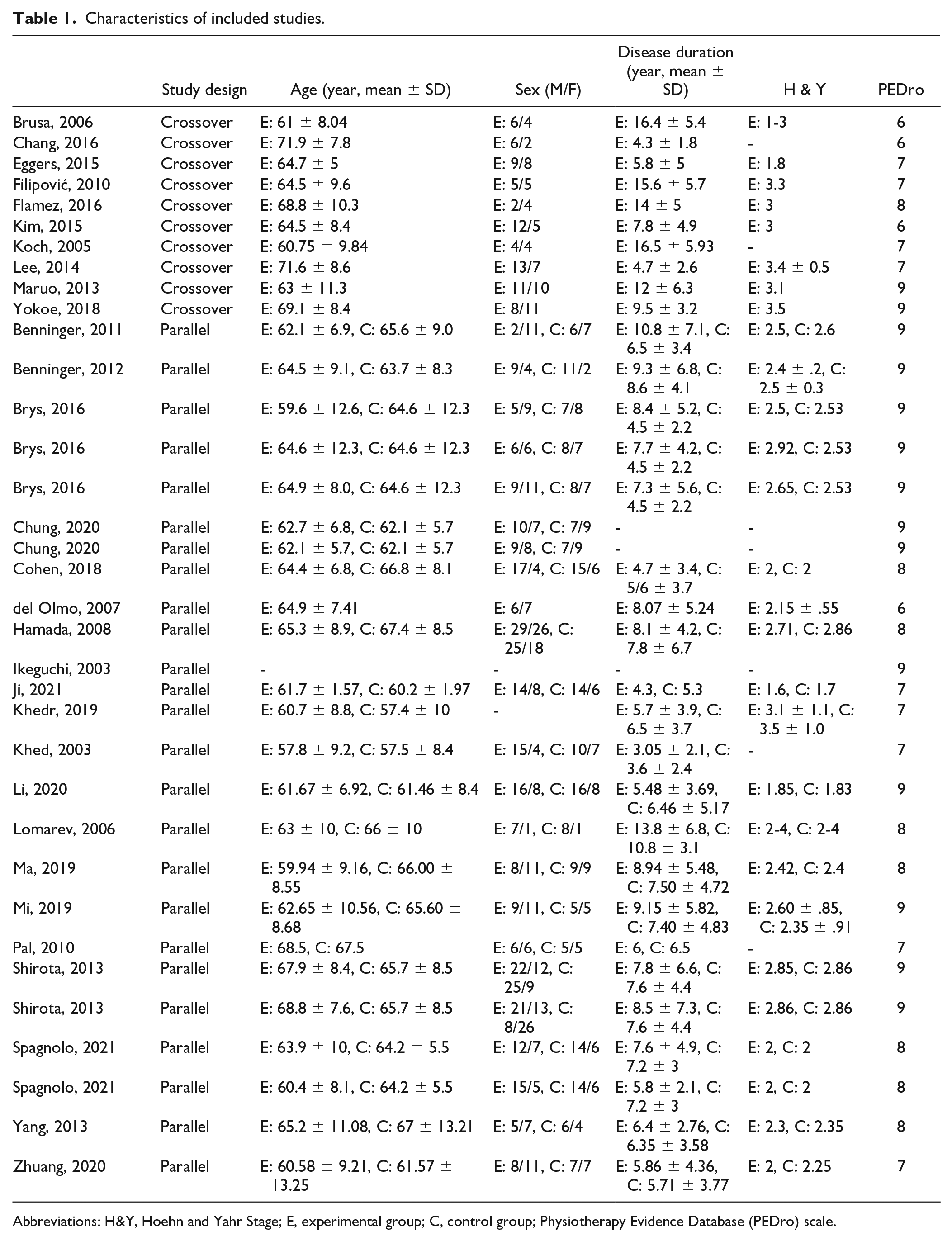
Abbreviations: H&Y, Hoehn and Yahr Stage; E, experimental group; C, control group; Physiotherapy Evidence Database (PEDro) scale.
Characteristics of Included Studies.
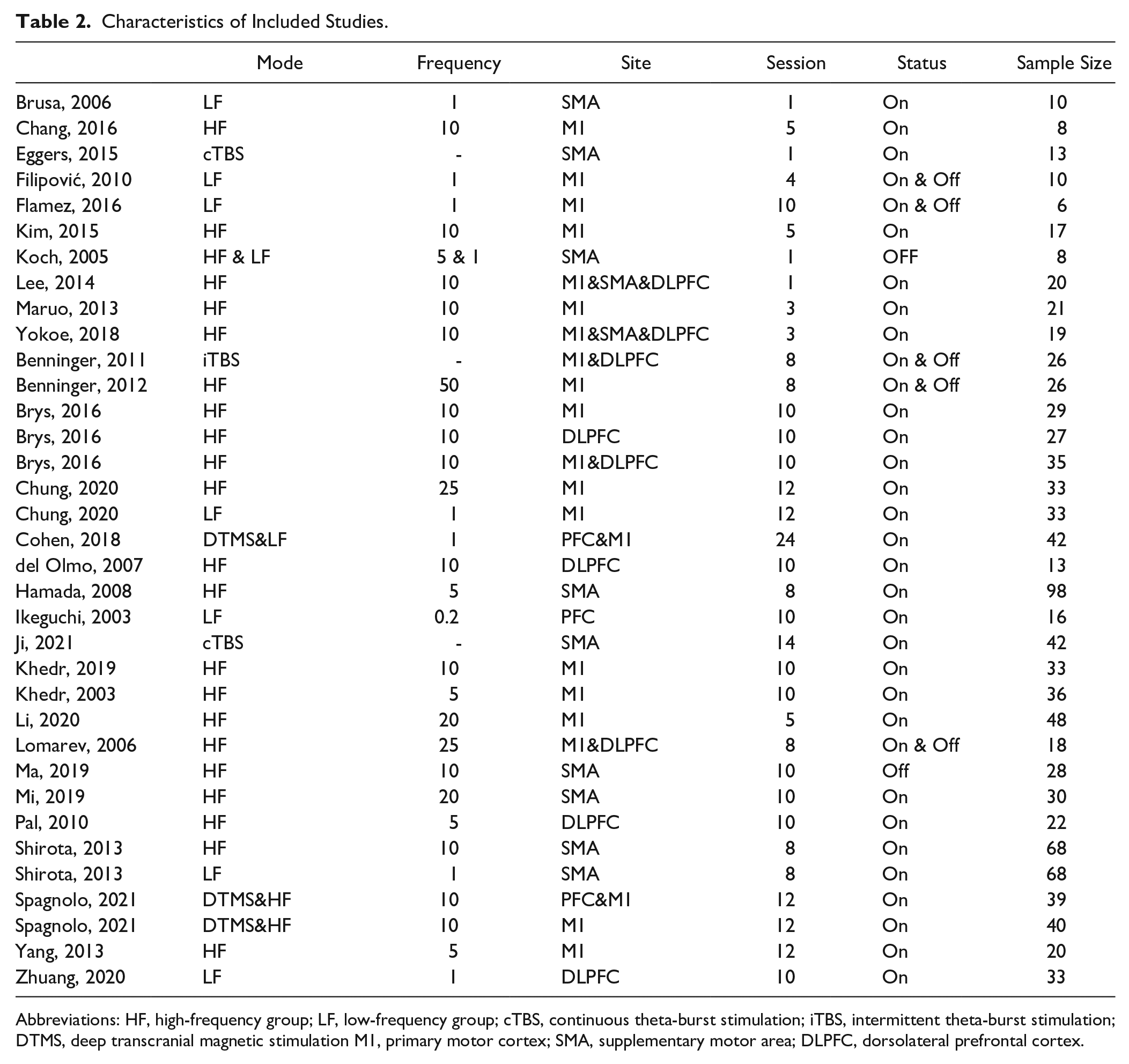
Abbreviations: HF, high-frequency group; LF, low-frequency group; cTBS, continuous theta-burst stimulation; iTBS, intermittent theta-burst stimulation; DTMS, deep transcranial magnetic stimulation M1, primary motor cortex; SMA, supplementary motor area; DLPFC, dorsolateral prefrontal cortex.
A total of 1048 patients were included in this review, and random-effects analysis revealed a pooled SMD of 0.64, 95%CI [0.47, 0.80] (P < 0.0001, I2=64%) (Figure 2, Figure S1), indicating an overall medium effect size favoring active rTMS over sham rTMS in relief of motor symptoms (P < 0.00001).
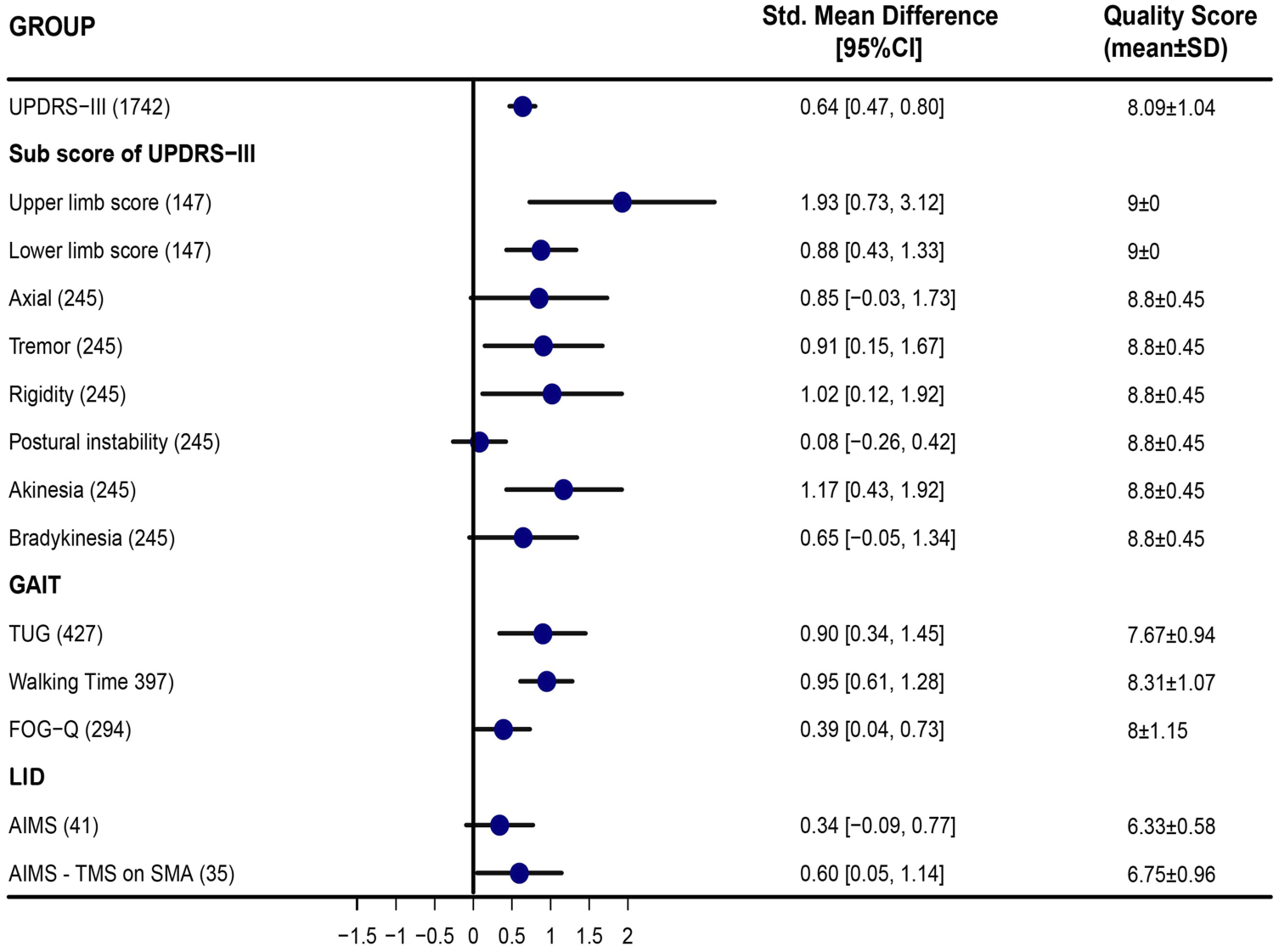
Meta-analysis of TMS for motor symptoms in parkinsonism. Items of subscores of UPDRS-III, Upper limb score (UPDRS-III item 20-25), Lower limb score (UPDRS-III item 20, 22, 26, 29), Axial score (UPDRS-III item 27–30), Tremor (UPDRS-III item 20, 21), Rigidity (UPDRS-III item 22), Postural instability (UPDRS-III item 30), Akinesia (UPDRS-III item 23–26), Bradykinesia (UPDRS-III item 31). TUG, Timed up and go test. FOG-Q, Freezing of Gait Questionnaire score. AIMS, Abnormal Involuntary Movement Scale.
PD is characterized by symptom heterogeneity and four trials present the measurements of subscores of UPDRS-III. Our analysis indicated significant amelioration of upper and lower limb scores, (SMD 1.93, 95%CI [0.73, 3.12] and SMD 0.88, 95%CI [0.43, 1.33]), tremor (SMD 0.91, 95%CI [0.15, 1.67]), akinesia (SMD 1.17, 95%CI [0.43, 1.92) and rigidity symptoms (SMD 1.02, 95%CI [0.12, 1.92]) (Figure 2).All of these trials involved selected M1 or SMA as the target area of high-frequency TMS, and the clinical evaluations were made when PD patients were “ON” status. In addition, the Gait symptom is one of the most common and debilitating symptoms in PD. 48 Pharmacological treatment for gait disorders is inconclusive. Seven studies focused on gait symptoms, assessed walking performance, including walking time, walking speed, TUG (Timed Up and Go), and FoG-Q (Freezing of Gait Questionnaire score). Our analysis demonstrated that rTMS could significantly reduce walking time (SMD 0.95, 95%CI [0.61, 1.28]), TUG (SMD 0.90, 95%CI [0.34, 1.45]) and FOG-Q score (SMD 0.39 95%CI [0.04, 0.73]). In other words, rTMS can improve the walking performances of PD patients. Moreover, the management of levodopa-induced dyskinesias (LID) remains a huge challenge during chronic dopaminergic treatment. Four trials investigating the effect of TMS for LID were included. Our results exhibited a significant amelioration of LID symptoms when stimulations were focused on SMA (SMD 0.60, 95%CI [0.05, 1.15]). (Figure 2)
Our subgroup analysis indicated a significant therapeutic effect on M1 (SMD 0.64, 95% CI [0.42, 0.87]). And there was a difference in effect size among different combinations of rTMS frequency and rTMS site. The combination of high frequency and targeting M1 showed a significant effect (SMD 0.79,95%CI [0.52, 1.07]). Moreover, multiple-session stimulation showed a therapeutical effect (SMD 0.67, 95% CI [0.50, 0.85]). A novel stimulation mode deep transcranial magnetic stimulation (dTMS) was also involved in our analysis. The result showed dTMS significantly improved PD motor function (SMD 0.46, 95%CI [0.14, 0.75]). Besides, we made a comparison between different sites and frequencies. Stimulation on M1 shows a better therapeutic effect (SMD 0.95 95%CI [0.33, 1.57]) than on SMA (Figure S2). Comparison between stimulation on M1 and DLPFC showed no significant results, which is consistent with our subgroup analysis (Figure S2). Another subgroup analysis of the on-off phenomenon during assessment showed that the therapeutic effect in on-state (SMD 0.74, 95%CI [0.52, 0.96]) was larger than off-state (SMD 0.09, 95%CI [-0.17, 0.35]) (Figure 3).
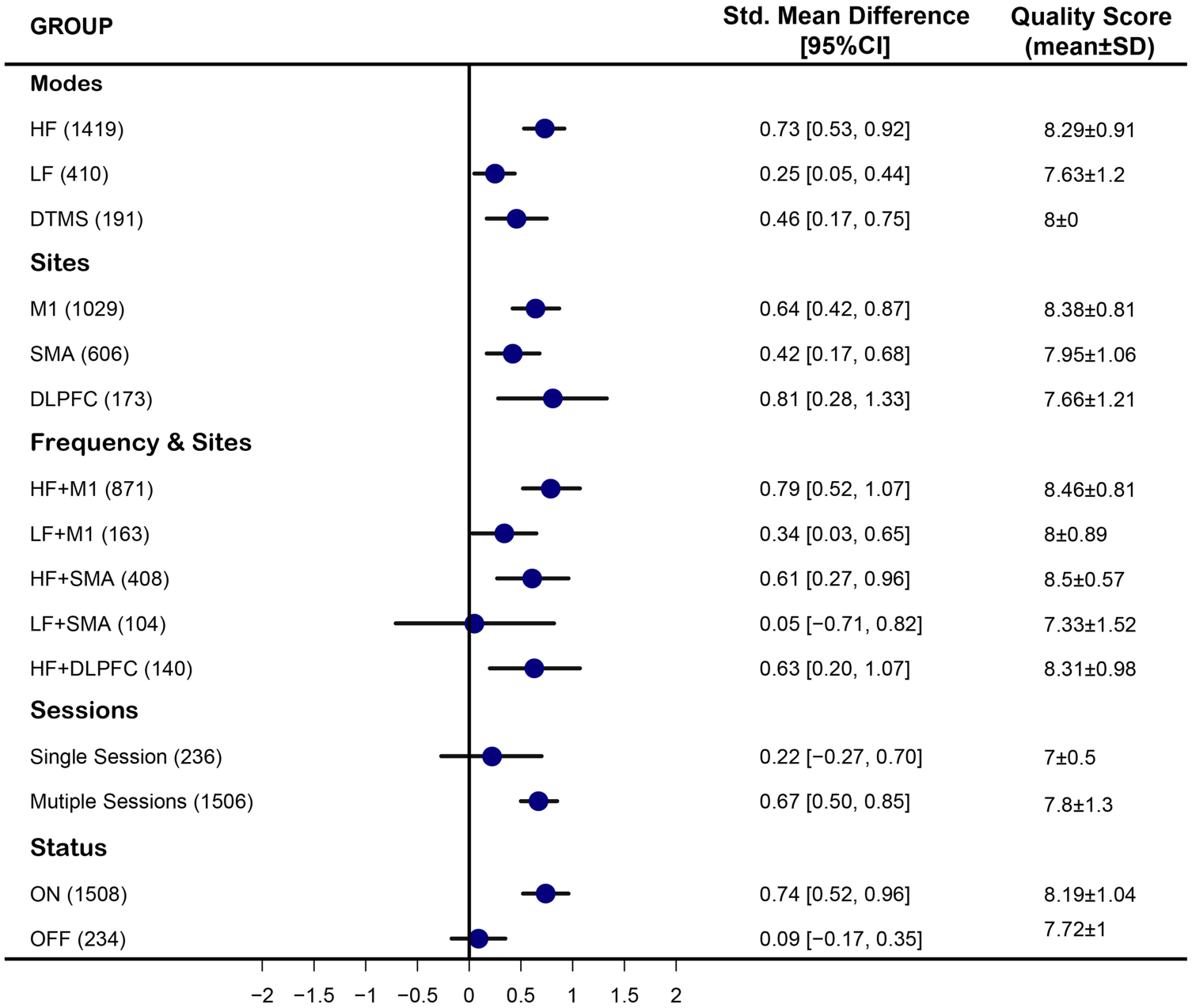
Subgroup meta-analysis of TMS for motor symptoms in parkinsonism.M1, the primary motor cortex. SMA, supplementary motor area. DLPFC, Dorsolateral Prefrontal Cortex. HF, high frequency. LF, low frequency. DTMS, deep transcranial magnetic stimulation.
Discussion
The meta-analysis showed that rTMS, especially multiple-sessions HF-rTMS on bilateral M1 region, is effective for global PD motor performance and can significantly reduce the UPDRS-III score of PD patients (SMD 0.64, 95%CI [0.47, 0.80]), which is consistent with the previous reports and the Evidence-based Guidelines.9,49,50 In addition, various motor symptoms such as akinesia, rigidity, tremor, gait impairment, and LID can be relieved by specific rTMS modes in PD subjects. Our present work could give a new insight into TMS intervention for Parkinson’s disease.
Akinetic-rigid and tremor are two major subtypes of PD symptoms. Our results showed HF rTMS on M1 or SMA can improve limb function and relieve both Akinetic-rigid and tremor symptoms. The study of Eman M. Khedr enrolled a large sample of 52 PD patients and concluded Akinetic-rigid patients benefited from excitatory stimulation instead of inhibitory stimulation, whereas tremor-dominant patients received no significant effect from either stimulation. 51 The different responses between two subtypes could result from underlying neural mechanisms. Akinetic-rigid patients always exhibit more changed neural activities in the mesolimbic cortex, an over-inhibition of neuronal activity in the motor thalamus, and consequently the hypoactivity in cortical motor and association areas.52,53 Whereas tremor-dominant patients exhibited reduced motor thresholds and short ipsilateral silent periods. 54 They were also found to display more changed activities in the cerebellar regions. 55 However, inhibitory stimulation targeting cerebellar failed to modify the tremor symptoms of PD patients in the studies of Matteo Bologna and Zhen Ni, which indicated abnormal oscillatory activity might occur in more structures and mechanism warrants further investigation.55,56
Gait impairment is a major risk factor for falls and contributes greatly to reduced mobility and quality of life. 48 The SMA plays an important part in neural resources linking from motor domains. Increasing evidence points to that the SMA is associated with anticipatory postural adjustments, which is essential in gait initiation and PD-FOG.57,58 Our results demonstrated an improvement in walking speed, TUG, and Fog-Q scores, especially after excitatory stimulation on primary motor cortex of lower the limb (M1-LL) and SMA. Despite not being calculated in the present study, step amplitude is expected to be rescued after excitatory stimulation as well. This is because, in PD subjects, the change in step amplitude and walking speed often occur in the same direction. Similarly, Maruo et al 32 observed improvements in both step amplitude and gait speed after three daily high-frequency rTMS sessions.
Chronic levodopa therapy in patients with PD is commonly complicated by the development of LID, 59 which occurs in 40% of patients after 4–6 years. LIDs are characterized by dysfunctional coupling between the right inferior frontal cortex (IFC) and SMA and deficient motor cortex plasticity. Abnormal overactivation of SMA and insufficient thalamocortical inhibition were observed in neuroimaging and experimental studies in PD with LID.60,61 In our analysis, low-frequency stimulation on SMA could relieve LID symptoms. Other studies also showed inhibitory stimulation on SMA and cerebellar, instead of the primary motor cortex, leads decrease of global abnormal involuntary movements scores. 12 Besides, the stage of the disease should be taken into account, as several late-stage studies found no significant clinical change in LID and motor function.20,36
The subgroup analysis for the stimulation site showed the therapeutic effect of rTMS intervention over M1, SMA, and DLPFC. The distinct results can be due to the difference in the included trials and data extraction, as several positive-result studies published in recent years.15,23,33,43 The novel dTMS was also included in the analysis, and the result showed a significant motor function improvement, which can be attributed to the deep and wide stimulation area. Besides, consistent with the previous reports, we found multiple-session is the better choice for the long-term benefit and more permanent changes. Overall, giving appropriate intervention based on abnormal neural circuits underlying specific symptoms could bring better relief for PD patients.
Several limitations of our works should be addressed. First, some uncontrolled variables, such as medication use, disease stage, age, and sex could confound the results. Given the large population involved, we can expect that any differences in background factors will be evenly distributed through random allocation. Second, the method used for sham stimulation can be various. The sham stimulation protocols can be control stimulation sites, 34 wrong TMS coil angles19,30,31 and matched sham coil.13,17 Finally, as with any meta-analysis, the possibility of publishing bias is also a problem. The funnel plot of our meta-analysis looks asymmetrical. At the same time, when the Egger’s rank correlation method and the Egger weighted regression method are applied to the data, the bias of the publication is visible.
In summary, rTMS therapy has been demonstrated to be an effective treatment for motor symptoms of PD, and multi-session HF stimulation on M1 can be an optimal stimulation protocol. We found that HF TMS on M1 and SMA has beneficial effects on limb function, tremor, and akinesia symptoms in PD patients. The individual differences in pathophysiology likely play an important role in impacting the treatment outcomes. Future studies should focus on optimal stimulation protocols for clinical subtypes and different symptoms. It may also be reasonable to conclude that the rTMS therapy may yield greater benefits if the dose and stimulation protocols are individualized to address specific symptoms.
Supplemental Material
sj-tif-1-nnr-10.1177_15459683221095034 – Supplemental material for Effects of Repetitive Transcranial Magnetic Stimulation on Motor Symptoms in Parkinson’s Disease: A Meta-Analysis
Supplemental material, sj-tif-1-nnr-10.1177_15459683221095034 for Effects of Repetitive Transcranial Magnetic Stimulation on Motor Symptoms in Parkinson’s Disease: A Meta-Analysis by Ruoyu Li, Yijing He, Wenting Qin, Zhuoyu Zhang, Junhui Su, Qiang Guan, Yuhui Chen and Lingjing Jin in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-2-nnr-10.1177_15459683221095034 – Supplemental material for Effects of Repetitive Transcranial Magnetic Stimulation on Motor Symptoms in Parkinson’s Disease: A Meta-Analysis
Supplemental material, sj-tif-2-nnr-10.1177_15459683221095034 for Effects of Repetitive Transcranial Magnetic Stimulation on Motor Symptoms in Parkinson’s Disease: A Meta-Analysis by Ruoyu Li, Yijing He, Wenting Qin, Zhuoyu Zhang, Junhui Su, Qiang Guan, Yuhui Chen and Lingjing Jin in Neurorehabilitation and Neural Repair
Footnotes
Author’s Contribution
Li, He, and Jin initiated and organized the project. Li, He, and Qin reviewed references and conducted this meta-analysis. Li, He, and Zhang reviewed references and interpreted data. Li, He, and Su drafted the manuscript. Jin, Chen, and Guan revised the manuscript. All authors have given final approval of the current version to be published. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China(2018YFC1314700), Clinical Research Project of Shanghai Tongji Hospital (ITJ(ZD) 1901), outstanding academic leaders plan of Shanghai Municipal Science and Technology Committee (20XD1403400), and science and technology innovation program of Shanghai Municipal Science and Technology(19441908000).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.