Abstract
Background. Neuropathic pain often follows spinal cord injury (SCI). Objective. To compare the effect of repetitive transcranial magnetic stimulation (rTMS) applied over different motor cortex targets (hand vs leg area) versus sham stimulation on neuropathic pain and local neurophysiological changes in patients with SCI. Methods. A total of 16 patients with complete or incomplete motor SCI and chronic neuropathic pain participated in a double-blind, cross-over randomized study. Three single sessions of sham or active rTMS (10 Hz, total of 2000 stimuli) were applied in random order over the hand or leg area with a minimal 2-week interval. The main outcome measures were the numeric rating scale for pain sensation and parameters derived from motor mapping of the first dorsal interosseous muscle, including maximal amplitude of evoked response as well as map area, volume, and location. Results. rTMS applied to either the hand or the leg area, but not sham stimulation, induced a significant but equivalent reduction in pain for the first 48 hours postintervention (P < .05). Participants with an incomplete lesion showed greater analgesia than those with a complete lesion (21% vs 3%, respectively; P < .05). The main change observed for motor map measurements was an increase in corticospinal excitability after stimulation of the hand area (P = .04) but not for the other conditions. Conclusion. rTMS applied over the hand or leg motor cortex decreased neuropathic pain regardless of any change in cortical excitability, suggesting that the analgesic effect is not associated with local changes at the motor cortex level itself.
Chronic pain is a significant problem after a spinal cord injury (SCI), affecting more than 80% of patients.1-3 Pain is associated with less engagement in physical activity and a lower level of community reintegration. 4 Moreover, pain is considered by those with SCI as one of the most difficult problems to deal with. 2 Several types of pain occur in these patients (musculoskeletal, visceral, or neuropathic), but neuropathic pain is recognized as the most resistant to treatment. 5 Two types of neuropathic pain are observed in SCI patients: the first type occurring in a dermatomal distribution at the level of injury (referred to as at-level pain) and the second type occurring in a more diffuse distribution below the level of injury (referred to as below-level pain). Although both types of neuropathic pain are severe and persistent, there may be some differences in underlying mechanisms. 2
Repetitive transcranial magnetic stimulation (rTMS) is a noninvasive method that induces immediate and lasting changes in cortical excitability. 6 In recent years, studies have shown that rTMS applied over the motor cortex (M1) can alleviate neuropathic pain from diverse origins, such as SCI, poststroke, amputation, or brachial avulsion pain, with pain reduction between 10% and 40%.7,8 However, the mechanisms of action of the rTMS on pain are still not clearly defined. Studies have demonstrated sensorimotor cortical reorganization after a nervous system lesion, such as a shift of cortical representations or increased excitability of adjacent representations.9-12 Those changes have been correlated with neuropathic pain.13-15 Some authors have suggested that local excitability changes induced by rTMS (eg, restored intracortical inhibition in the motor cortex area corresponding to the painful limb) are associated with the level of analgesia.7,16 Therefore, most studies have tried to target the motor cortical area corresponding to the painful zone.17-19
In contrast, it has been proposed that activation of distant structures involved in pain, such as the thalamus or brainstem periaqueductal gray matter, could explain the analgesic effect of rTMS. 20 Lefaucheur et al 21 obtained better results for facial pain when stimulating the hand cortical area. Moreover, it has been proposed that the endogenous opioid system could play a role in the analgesic effect of rTMS.22,23 In this scenario, targeting cortical areas corresponding to the painful zone might not be necessary.
In a recent meta-analysis, Leung et al 8 showed that those with different medical conditions do not respond similarly to rTMS. Until now, only 2 studies have specifically investigated the analgesic effects of rTMS (using repeated sessions) on neuropathic pain from a spinal origin.18,24 Defrin et al 18 stimulated over-the-leg motor representation (M1 area corresponding to the painful zone), whereas Kang et al 24 stimulated over-the-hand motor representation (M1 area outside of the painful zone). The therapeutic efficacy of rTMS compared with a placebo has not been clearly demonstrated in any of these studies, although they both reported some results in favor of the real rTMS treatment (both studies had a limited sample size [n = 11] with quite heterogeneous samples that are representative of the population of patients with a SCI).
The aim of the present study was to evaluate the immediate and long-term analgesic effects of a single rTMS session applied over the leg versus the hand motor area (compared with sham stimulation) to inform the choice of a stimulation site for future clinical studies using repeated sessions. The secondary aim was to document the local neurophysiological changes induced by rTMS and to assess whether these changes are associated with the analgesic effects. Based on results suggesting that descending inhibitory mechanisms play a role in the analgesic effect of rTMS,22,23 our primary hypothesis was that no significant difference would be observed between the 2 different cortical targets.
Methods
Participants
Participants were recruited from the outpatient population of our rehabilitation center (Institut de Réadaptation en Déficience Physique de Québec, Canada). Inclusion criteria were (1) complete or incomplete SCI preserving hand muscle function and (2) neuropathic pain for at least 3 months. Presence of other types of pain (musculoskeletal/visceral) was not an exclusion criterion, but neuropathic pain had to be reported as the most disturbing pain. Location of the neuropathic pain (at or below level) was assessed but was not used as an exclusion criterion. Because of contraindications for the rTMS/TMS and MRI, patients having the following conditions were excluded: (1) any kind of metal implant in the head (excepted dental implants); (2) implanted stimulator, including cardiac pacemaker or drug delivery system; and (3) personal or familial history of epilepsy. Almost all patients (14/16) were being treated with various medications, including antidepressants, anticonvulsants, or opioids. They were instructed not to change the medication throughout the study or to report any change. The study protocol and consent forms were reviewed and approved by our local ethics committee (IRDPQ, project #2009-154). All participants provided written informed consent.
In all, 18 patients with SCI participated in the study. One patient withdrew from the study before the first rTMS session for medical reasons (unexpected finding of a subacute hematoma on MRI), and another one was excluded at the first TMS session (resting motor threshold [rMT] close to 100% of maximal stimulator output). Therefore, 16 participants completed the study (mean age = 50 ± 9 years). Among them 12 patients had paraplegia, and 4 patients had tetraplegia; 8 patients had motor-complete SCI. Also, 6 patients developed syringomyelia following their initial injury (see Table 1 for detailed patient characteristics and description of baseline pain).
Characteristics of the Participants, Pain at Baseline (Intensity and Quality), and Pain Change (Intensity).
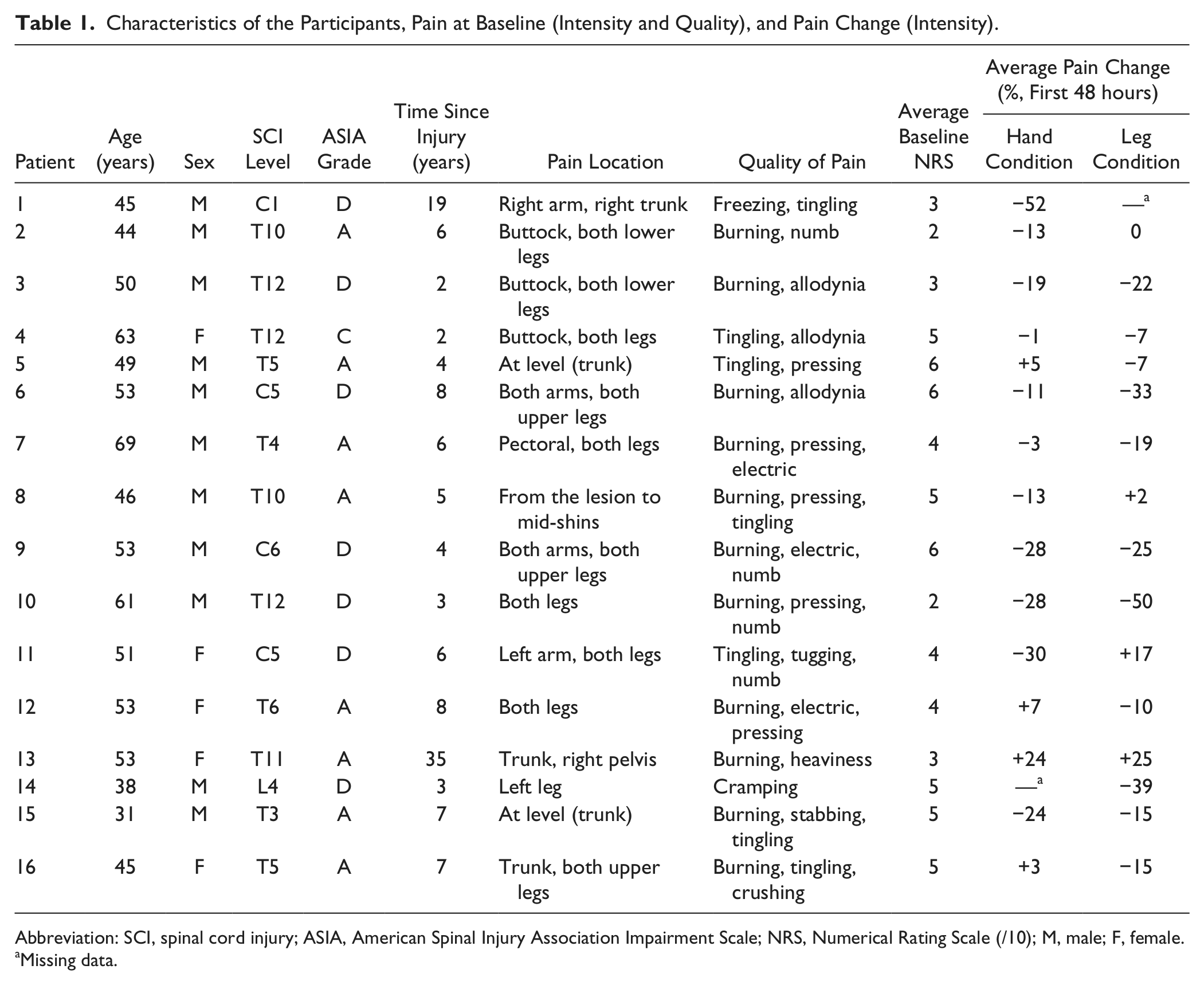
Abbreviation: SCI, spinal cord injury; ASIA, American Spinal Injury Association Impairment Scale; NRS, Numerical Rating Scale (/10); M, male; F, female.
Missing data.
Experimental Procedure
A quasiexperimental cross-over design was used, each participant being exposed to 2 active rTMS sessions (hand/leg M1 area) and 1 sham rTMS session in a randomized, counterbalanced order. The sessions were all at least 2 weeks apart. The patients were not informed that they would receive sham stimulation but were rather told that 3 sessions of rTMS, using different parameters of stimulation, would be tested to compare their efficacy in relieving pain. In those who reported more pain on one side, the stimulation was applied over the motor cortex contralateral to that side. In those who reported similar pain on both sides, the stimulation was applied over the motor cortex contralateral to the dominant-hand side. Prior to the first laboratory session, patients had to undergo an MRI examination to obtain a 3D reconstruction of their brain, on which a grid (sites spaced by 10 mm) was applied to guide TMS via a neuronavigation system (Brainsight, Rogue Research). The use of a neuronavigation system ensured accurate positioning of the coil throughout each individual session and across the different sessions.
Experimental sessions took place in a quiet room. Patients were seated in a comfortable armchair or in their wheelchair. After skin preparation, 2 Ag/AgCl surface recording electrodes (1 cm2 recording area) were placed over the first dorsal interosseous (FDI) muscle in a belly-tendon montage. EMG signals were amplified, bandpass filtered (20-1000 Hz), digitized at a sampling rate of 2000 Hz (Power1401 Interface; Cambridge Electronic Design, Cambridge, UK), and stored on a computer for offline analyses. A coregistration procedure was also performed with the neuronavigation system. The FDI hotspot was then located, and the rMT was determined (lowest stimulation intensity evoking peak-to-peak motor-evoked potentials [MEPs] >50 µV in at least 6/12 trials). The single-pulse TMS was delivered through a figure-of-eight coil (70 mm in diameter) connected to a Magstim SuperRapid2 stimulator. The coil was held tangential to the scalp, the handle pointing 45° backward and laterally.
After the participants were prepared, each experimental session comprised 3 phases: (1) pre-rTMS assessment, (2) rTMS application, and (3) post-rTMS assessment. During the pre-rTMS and post-rTMS assessments, the following dependent variables were assessed: (1) immediate pain intensity and (2) TMS motor map of the FDI representation.
Pre-TMS and Post-rTMS Assessments
Pain Intensity
Immediate effects were evaluated using a Numeric Rating Scale (NRS) at 0 and 20 minutes post-rTMS. NRS consists of a 11-point scale, where 0 represents no pain sensation and 10 the worst possible pain. Long-term effects were evaluated by a logbook that patients had to fill out a week before and a week after each session. In this logbook, they had to quantify their average and worst pain each day on 2 different NRSs.
FDI Motor Mapping
The different sites of the grid implemented over M1 were stimulated successively. In all, 6 TMS pulses (12 at the hotspot) were applied at each site, at 110% rMT, separated by intervals of 4 to 6 seconds (jittered). For each site, the peak-to-peak amplitude of the recorded MEP was measured and averaged using custom analysis software (IsotopCM, Mathomic Solutions, Québec, Canada). The different TMS variables were then extracted: (1) maximal MEP amplitude, (2) map area, (3) normalized map volume, and (4) center of gravity (CoG). CoG was computed for the mediolateral (x) and anteroposterior (y) coordinates relative to the vertex (expressed in mm) using the following formula: CoG x = (Σxi × MEP i )/ΣMEP i and CoG y = (Σyi × MEP i )/ΣMEP i , where MEP i represents the mean amplitude of the MEPs produced at one site. 25
The researcher running the pre-post assessment (as well as data analysis) was blind relative to the applied rTMS protocol (as was the participant), with the rTMS application being performed by a different researcher.
rTMS Application
rTMS was also applied using a Magstim SuperRapid2 stimulator, but this time with a 70-mm figure-of-eight Airfilm Coil (or sham Airfilm Coil). Active rTMS consisted in applying 40 trains of 5 seconds each at 10 Hz (25-s intertrain interval), for a total of 2000 stimulations applied in 20 minutes. The intensity of the stimulation was set at 90% of the FDI rMT when applied over the hand area and 110% of the FDI rMT over the leg area. The TMS intensity was estimated from Saisanen et al, 26 who compared rMTs of hand and leg muscles in healthy individuals. It is worth noting that the intensity of stimulation was corrected to take into account the difference in output between the regular figure-of-eight coil used for mapping and the air film coil used for rTMS. FDI hotspot position was targeted for the hand area condition, whereas the intersection point of the motor strip and the interhemispheric line was targeted for the leg area. The coil was kept steady during rTMS via mechanical fixation (with corrections by the operator during intertrain intervals if necessary based on neuronavigation feedback). Sham rTMS, using a sham coil (mimicking the noise and scalp sensations), was applied over the hand area using the same parameters.
Analysis and Expected Results
All statistical analyses were performed with SPSS 13.0 software (IBM, Chicago, IL). Because of the limited sample size and of the large interindividual variability observed, nonparametric tests were selected. Wilcoxon exact tests were used to compare pre-TMS and post-rTMS measures for each variable. Friedman tests were used to compare the level of change for each variable (post vs pre) across conditions (sham/active hand/active leg area). For pain intensity, the percentage of change within each individual was calculated as follows: [(Post-rTMS − Pre-rTMS Pain Score)/(Pre-rTMS Pain Score)] × 100. Although the NRS is an ordinal scale, it has been shown that adjusting for the baseline pain score by calculating the percentage change in the NRS resulted in a more linear and consistent relationship between the reported change in pain and the patients’ perception of their improvement. 27 For immediate pain, the pre-rTMS pain score was assessed prior to the rTMS intervention. For long-term pain, the pre-rTMS pain score was the average of the 7-day pain ratings prior to the experimental session. For TMS variables (MEP amplitude and CoG location), the level of change was computed as the difference between post-rTMS and pre-rTMS. The Mann-Whitney exact test was used to perform subgroup analyses based on clinical profile (complete vs incomplete lesion). To evaluate the difference in participant’s perception of treatment across conditions, a proportion test (R) was used. Spearman correlational analyses were used to assess relationships between the change in pain for the first 48 hours and the change on neurophysiological variables following stimulation (MEP amplitude and CoG shift). All analyses were performed with an α threshold at .05.
Results
Pain Intensity
Short-term Effect
Only the 2 active conditions showed significant time (pre-post change) effects. Immediately after intervention, there was significant pain reduction for the hand condition (P = .021), a nonsignificant trend toward decrease for the leg condition (P = .086), and no change in the sham condition (P = .148). Also, 20 minutes after the intervention, both active conditions showed a significant pain reduction (P = .003, hand, and P = .047, leg), without any difference for the sham condition (P = .180). However, the level of pre-post change was not significantly different across conditions, neither immediately (P = .452) nor 20 minutes after stimulation (P = .846; see Figure 1A).
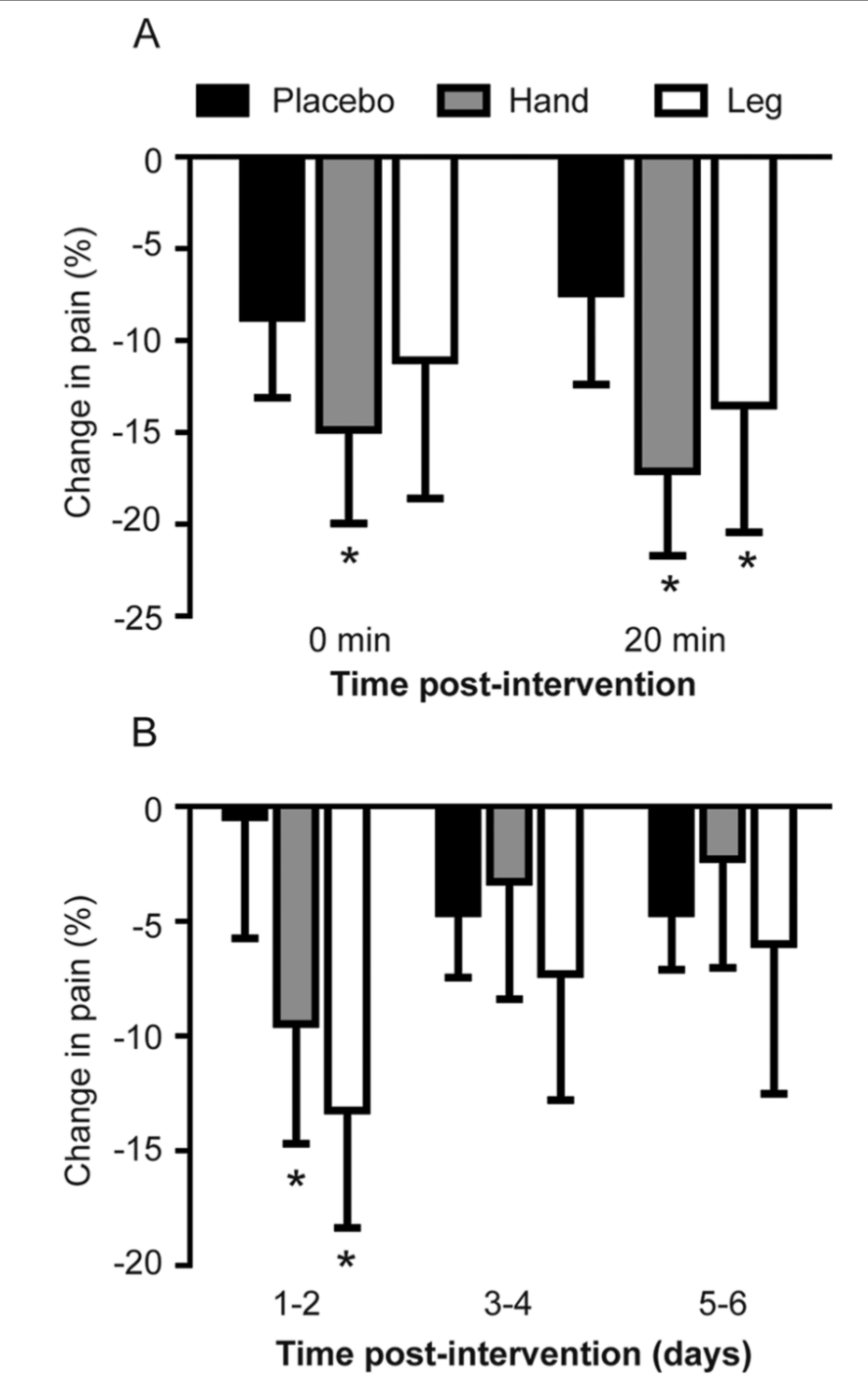
Average percentage of pain reduction induced under each condition. Error bars show standard error of the mean. Asterisks indicate a significant pre-post difference for a given condition (all P < .050). A. Short-term pain. B. Long-term average pain.
Long-term Effect
Both active conditions showed a significant time effect (pre-post change) on the average pain level for the first 48 hours only (P = .021, hand, and P = .008, leg), whereas no significant difference was observed for the sham condition (P = .719). However, the level of change in pain was not significantly different across conditions (P > .05; see Figure 1B). When looking at the worst pain level, only the hand condition showed a significant time effect for the first 96 hours (P = .028, 0-48 hours, and P = .008, 48-96 hours). Again, no significant effect of condition on the level of pain was found (P > .05).
Subgroup analyses showed a significant effect of the type of lesion (complete vs incomplete) on average pain reduction for the first 48 hours. Those with incomplete lesions had a greater reduction of pain for the hand condition (P = .018) than those with a complete lesion. There was a nonsignificant trend for the leg condition (P = .054) and no effect in the sham condition (P = 1.00; see Figure 2).
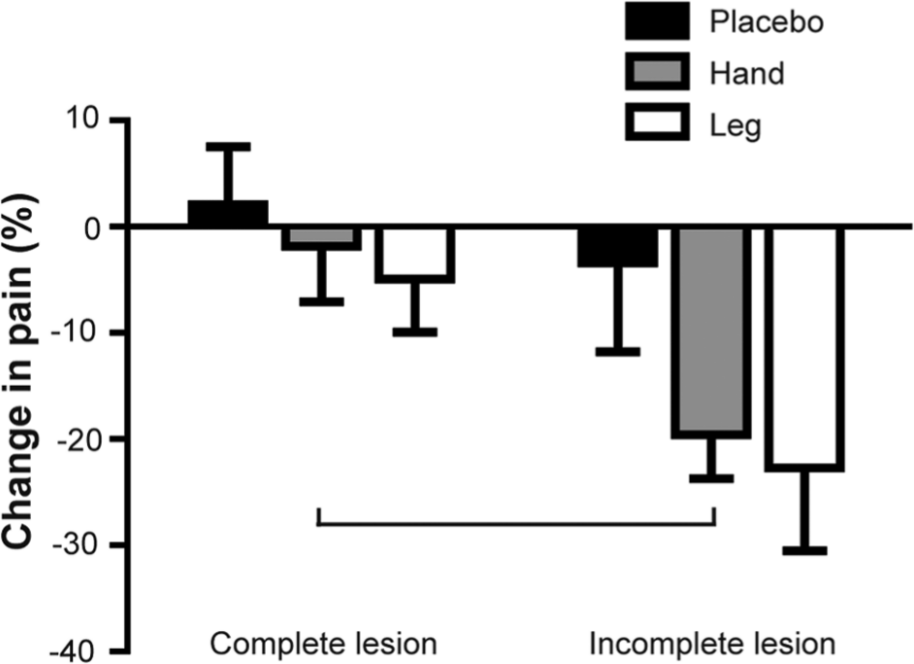
Percentage of average pain reduction according to the type of lesion for the first 48 hours. Error bars show the standard error of the mean. The horizontal bar indicates a significant difference between subgroups for the hand condition (P = .018). A nonsignificant trend was observed for the leg condition (P = .054).
FDI Motor Mapping
Maximal MEP Amplitude
For maximal MEP amplitude, only the hand condition showed a significant time effect (increase of 0.34 ± 0.54 mV; average pre-rTMS amplitude = 0.80 ± 0.67 mV; P = .035). The Friedman test used to compare the level of change across conditions showed a significant difference (P = .030), but post hoc analyses failed to reveal significant differences between a specific pair of conditions (see Figure 3).
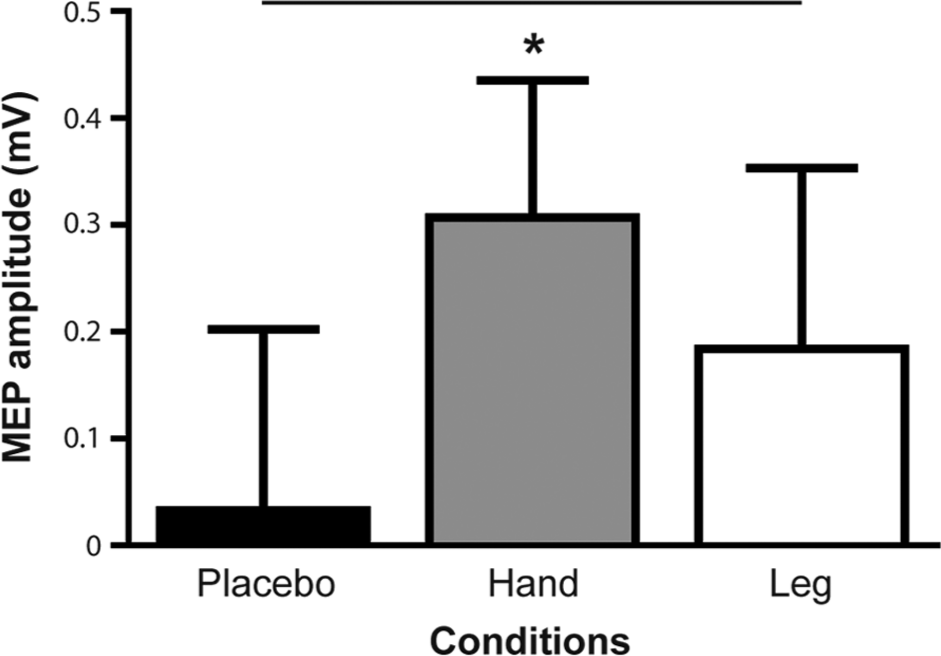
Maximal motor-evoked potential (MEPs) amplitude increase post-rTMS. Error bars show standard error of the mean. Asterisk indicates a significant pre-post difference for a given condition (P = .035). Horizontal bars indicate a significant difference in the corticospinal excitability change between conditions (P = .030, but post hoc analyses failed to reveal significant differences between a specific pair of conditions).
Map Area and Map Volume
For map area and normalized map volume, there was no significant effect of time (P > .05) or condition (P = .510, map area, and P = .607, map volume).
Center of Gravity
Figure 4 illustrates CoG shifts on the mediolateral axis for each condition. There was no significant effect of time for any condition (P > .05), but a significant condition effect was found (P = .023). Post hoc analyses showed differences in changes in map location between sham and hand conditions (P = .019) as well as between hand and leg conditions (P = .019). On the anteroposterior axis, there was no significant effect of time and no significant condition effect (P = .584).
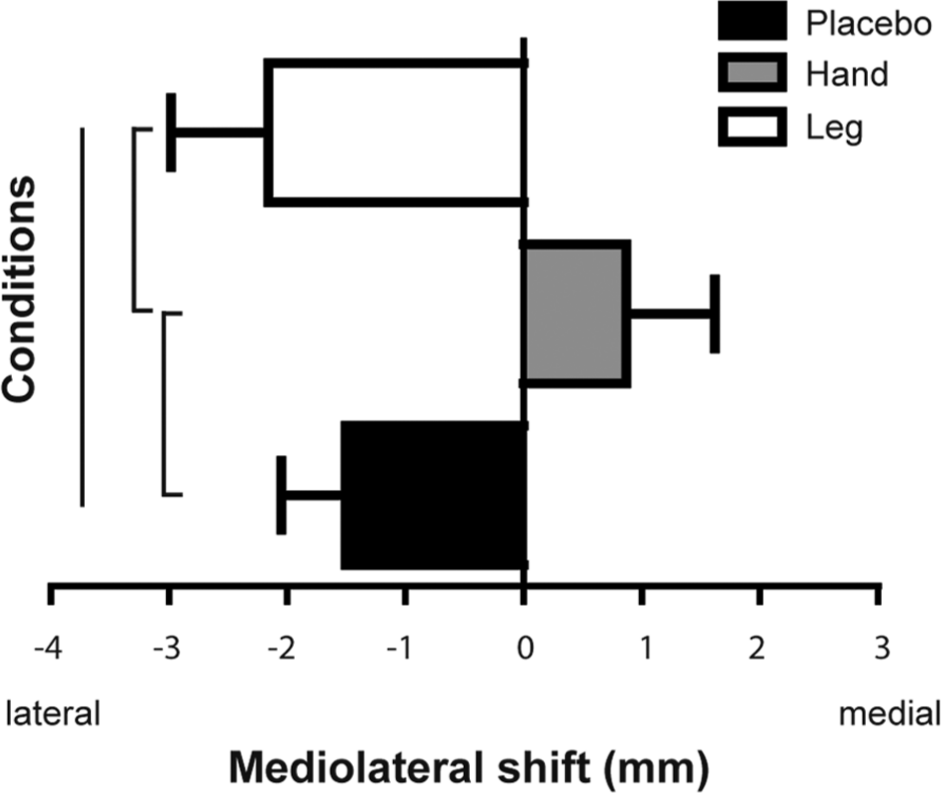
Shift of the center of gravity (CoG) on mediolateral axis (X). Error bars show standard error of the mean. Vertical bars indicate a significant difference between conditions as well as the significant results of post hoc comparisons (all P < .025).
Correlational Analyses
There was no significant association between average pain for the first 48 hours and the changes in MEP amplitude (placebo: P = 0.449; hand: P = .446; and leg: P = .516) nor between average pain for the first 48 hours and the CoG shifts (placebo: P = .214; hand: P = .478; and leg: P = .318).
General Remarks
Most of the participants (n = 7) reported mild discomfort (2.6 ± 2.8 on a 10-point scale) during TMS measures or rTMS interventions. It was mostly a result of the pressure applied by the coil and the facial muscle twitching. To ensure that participants were blinded to the condition, their perception of treatment effectiveness was asked 20 minutes after the intervention. There was no significant difference between conditions (P > .05).
Discussion
The primary aim of the study was to evaluate the analgesic effect of rTMS applied over 2 different M1 stimulation targets (hand and leg area), compared with a sham stimulation, on neuropathic pain following a SCI. Only active conditions (on both targeted areas) induced significant pre-post reductions in average and worst pain ratings after a single treatment, whereas sham stimulation did not induce any significant change. In contrast to the observations of Kang et al, 24 who found that rTMS reduced pain 2 weeks after the first intervention, most of the analgesic effect was observed immediately after the intervention and was of short duration (48 hours). The intensity of pain reduction for the first 48 hours was about 10% for active conditions versus 2% for the sham condition. It is important to note that our results failed to show statistically significant differences in the level of pain reduction across conditions (even compared with sham stimulation) and therefore need to be interpreted cautiously. However, the pattern of significant pre-post differences observed was very consistent from one variable to another (significant differences were always observed only for the 2 active conditions and never for the sham condition). The lack of significant effect between conditions might be attributable to the limited sample size as well as to the heterogeneity of the SCI population. Most of the previous studies reported between 10% and 40% of pain reduction after rTMS.8,28 Patients with different medical conditions respond differently to rTMS. 8 For example, pain reduction observed following a single session for trigeminal nerve origin was 20% versus 15% for spinal cord origin. 8 It is important to note that our results suggest that even within a given clinical population, the effectiveness of stimulation may vary according to the characteristics of the lesion (eg, pain reduction for active conditions for complete [3.4%] vs incomplete [21%] injury). Although these changes can appear small, it should be kept in mind that a clinically meaningful level of analgesia is not necessarily expected after a single session of rTMS. Some rTMS studies have shown greater effects with repeated sessions. For example, Khedr et al 29 showed much greater pain relief after the fifth rTMS session than after a single one.
The secondary aim was to document the neurophysiological changes underlying the analgesic effects of rTMS. It is important to emphasize the fact that we characterized the neurophysiological effects of rTMS by looking at a spared hand muscle representation, regardless of whether the hand or leg area was stimulated. Repeated stimulation of one representation can result in increased motor output from this representation, a process that can be referred to as “within-representation plasticity.” 30 As expected, our results show that high-frequency stimulation of the hand area induced an increase in corticospinal excitability. 31 Because half of our patients had complete motor lesions (and several others had important lower-limb paresis), it was not possible to look at within-representation plasticity for the leg area. However, rTMS has been shown to also induce across-representation plasticity, although interactions between leg and hand areas are still unclear. 30 Therefore, hand representation was used to probe whether stimulation of the hand and leg areas resulted in different local effects at the motor cortex level. Our results demonstrate that the local neurophysiological changes were dependent on the site of stimulation. The amplitude of maximal MEPs only increased for hand area stimulation, and the CoG shifted in opposite directions between the 2 active conditions (although these shifts were very small). However, there was no significant effect for map area and map volume.
Several mechanisms have been reported to explain the analgesic effects of rTMS, including local changes at the motor cortex level itself (in particular, restoration of intracortical inhibition) 16 as well as distant effects such as the following: (1) activation of perigenual cingulate and orbitofrontal areas modulating the emotional appraisal of pain; (2) top-down activation of brainstem periaqueductal gray matter driving descending inhibition toward the spinal cord; and (3) triggering of mechanisms resulting in the secretion of endogenous opioids.20,22,23 The fact that we observed differences between the 2 active conditions regarding local motor cortex effects while pain was reduced similarly for both conditions does not support a causal relationship between local changes and pain relief and rather suggests that the analgesic effect is mediated by distant effects. Supporting this hypothesis, correlational analyses between the analgesic effect of the first 48 hours and the neurophysiological variables (MEP amplitude and CoG shift) showed no association. It is, however, important to mention that potentially relevant measurements (such as intracortical inhibition) were not taken due to the duration of the TMS mapping procedure. The fact that patients with an incomplete spinal lesion exhibited a much better analgesia than those with a complete lesion also supports the role of descending inhibition mechanisms in the analgesia produced by rTMS. Unfortunately, no MRI of the spinal cord was obtained in the present study. Future studies looking at the relationship between spared spinal pathways and the effect of rTMS on pain in a larger sample of patients with incomplete SCI would allow a better understanding of the mechanisms underlying the analgesic effect. Another limitation of this study is that pain ratings were not obtained separately for each limb. Given that stimulation was performed unilaterally for hand area, it would have been relevant to assess whether a difference was perceived between sides for patients with bilateral pain (stimulation of leg area might have induced bilateral effects because of leg area location). If diffuse noxious inhibitory controls are responsible for the observed analgesia, we should expect bilateral effects despite unilateral stimulation. 32
Focusing on patients with incomplete SCI might also allow better control of the stimulation parameters for the leg area. In the present study, the intensity of stimulation for the leg area was set at 110% of rMT of the FDI muscle. This intensity was chosen according to Säisäsen et al, 26 who compared the intensity of the stimulator for rMT of hand muscles versus leg muscles in healthy adults (in an attempt to match the 90% of rMT used for the FDI). 26 Absence of recording of evoked responses in the leg area might have led to a less-optimal selection of the stimulation site and of the intensity (nevertheless, the effect on pain was comparable to that obtained with hand area stimulation). Another limitation that should be pointed out is that factors such as mood or spasticity have not been taken into account. Studies have shown that rTMS may also have an impact on these 2 factors.33-35 However, Defrin et al 18 pointed out that reduction in the depression score did not suffice to explain pain reduction. Unfortunately, there were not enough patients with spasticity in the present study to allow us to use spasticity as a secondary variable to verify whether this effect is dependent on the targeted motor area. Based on the limited available data, the leg area appears as an appropriate target for the reduction of both pain and spasticity using subthreshold high-frequency rTMS. Despite requiring higher stimulation intensity, it was not considered as more painful or uncomfortable by the participants.
Conclusion
In summary, active rTMS applied over the motor cortex decreased neuropathic pain regardless of the motor region stimulated and of the changes in cortical excitability, suggesting that the analgesic effect is not associated with local changes at the motor cortex level itself. Pain reduction then probably relies on distant effects such as top-down activation of brainstem periaqueductal gray matter driving descending inhibition, a hypothesis supported by the observation that analgesia was mainly noted in patients with an incomplete spinal lesion. Comprehensive neuroimaging assessments are needed in future studies investigating the effect induced by repeated applications of rTMS on neuropathic pain in order to improve our understanding of its underlying mechanisms and to guide clinical use.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (CIHR), Grant Number MOP-79370. C. Mercier was supported by salary awards from the CIHR and the Fonds de recherche du Québec, Santé (FRQS). F. Jetté was supported by a fellowship from Université Laval and H. B. Meziane by a fellowship from the Réseau Provincial de Recherche en Adaptation-Réadaptation (REPAR-FRQS). Support was provided by the Consortium d’Imagerie en Neuroscience et Santé Mentale de Québec (CINQ) for MRI acquisition.
