Abstract
Background. Evidence remains mixed as to the effectiveness of repetitive transcranial magnetic stimulation (rTMS) in treating mild cognitive impairment (MCI) in patients with Parkinson’s disease (PD). Objective. In this study, we examined the short- and long-term effects of patterned rTMS. Methods. We randomly assigned 35 patients with PD with MCI to two groups. One group received intermittent theta burst stimulation (iTBS; n = 20), and the other received its sham counterpart (n = 15). The stimulations were applied over the left dorsolateral prefrontal cortex for 10 consecutive weekdays. Measurements based on the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) and Montreal Cognitive Assessment (MoCA) were conducted at three time points: at baseline, immediately after the last intervention and at 3-month follow-up. Each patient received a 99mTc-TRODAT-1 single-photon emission computed tomography (SPECT) brain scan at baseline. Results. The iTBS group exhibited significantly greater improvement than the sham group did in total RBANS and MoCA scores (p < .001 for both) immediately after intervention and at the 3-month follow-up. Radiotracer uptake in the bilateral basal ganglion in baseline SPECT was positively correlated with response to iTBS conditioning with respect to improvements in MoCA scores (p = .021). Conclusion. This randomised controlled trial provides evidence that a consecutive iTBS protocol can achieve a persistent and wide-ranging therapeutic effect in patients with PD with MCI.
Keywords
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder that is characterised by resting tremor, rigidity, gait disorder, bradykinesia and postural instability. With the degeneration of dopaminergic neurons in the substantia nigra pars reticulata and the internal segment of the globus pallidus, individuals diagnosed with PD may gradually present with motor and nonmotor symptoms. Cognitive impairment is a common nonmotor manifestation in patients with PD and comes in a spectrum ranging from PD with mild cognitive impairment (PD-MCI) to PD with dementia (PD-D). MCI might be a harbinger of dementia, 1 with a conversion rate to PD-D as high as 31%. 2
Medication, such as anticholinesterases, is the first line of treatment for PD-MCI or mild PD-D, although evidence remains limited as to the efficacy of medication in cognitive restoration. Implementing behavioural treatments, such as traditional cognitive training, is a valuable prevention strategy. 3
As a novel noninvasive treatment, repetitive transcranial magnetic stimulation (rTMS) has been noted in recent studies as having the capacity to ameliorate cognitive dysfunction in PD. 4 According to the theory that long-term potentiation (LTP) enhances synaptic transmission, rTMS can improve the excitability imbalance between the substantia nigra pars reticulata and the internal segment of the globus pallidus, further leading to beneficial neuroplasticity in the circuit underlying frontostriatal cognition.5,6 Systematic reviews have demonstrated that multiple sessions of high-frequency (10–20 Hz) rTMS over the dorsolateral prefrontal cortex (DLPFC) may improve executive function in patients with PD with cognitive deficit. 7 However, findings on the effect of rTMS on PD-MCI remain controversial, yielding heterogenous conclusion. Some studies have disagreed with recommending rTMS for cognitive treatment. 8 A randomised controlled trial (RCT) indicated that rTMS over the bilateral DLPFC cannot improve cognitive status in patients with PD-MCI. 9 Two RCTs have revealed that six sessions of rTMS in patients with PD-MCI might improve overall cognitive function, but the results were not significantly different from those of the sham group.10,11 There remains no consensus on the effect of rTMS for patients with PD with MCI.
Theta burst stimulation (TBS) 12 is a novel and patterned rTMS protocol that confers a more lasting stimulation effect for improved cognitive performance in domains such as working memory and executive function. 13 Studies have reported that prefrontal intermittent TBS can improve the working memory of healthy subjects, 14 whereas continuous TBS was associated with a decline in working memory. 15 Ekman et al 16 investigated presynaptic dopamine integrity by using single-photon emission computed tomography (SPECT) in PD and revealed under-recruitment of the frontostriatal region, including the DLPFC, in PD-MCI. 16 Monchi and colleagues reported that patients with PD and MCI exhibited decreased frontostriatal activation while performing an executive task in an fMRI study compared with those with only PD. Therefore, executive impairment in PD could be attributed to frontostriatal deficit.17,18 Our previous RCT indicated that iTBS over the left DLPFC improves cognitive function in patients with stroke and MCI. 19 On the basis of the rationale that the DLPFC is a key node of cognitive frontostriatal circuitry and that the impairment of this region leads to executive deficit in PD-MCI, in the present study, we tested the hypothesis that consecutive iTBS treatments over the left DLPFC improve cognitive function in patients with PD-MCI. We conducted a sham-controlled randomised study to examine the immediate and long-term effects of iTBS using comprehensive neuropsychological assessments.
Material and Methods
Participants
This study surveyed 62 patients with PD who were recruited from outpatient clinics or the PD unit of a tertiary medical centre. The patients were screened for eligibility to participate in this study according to their subjective expression of memory decline. Of the screened patients, 10 refused to participate and 12 did not meet the inclusion criteria (Figure 1). The remaining 40 patients satisfied the following inclusion criteria: (1) having a diagnosis of idiopathic PD according to the UK Brain Bank Society criteria for idiopathic PD
20
provided by a neurologist specialised in movement disorders; (2) receiving a stable medication dosage throughout the study period and (3) having MCI defined by the Movement Disorder Task Force Level II criteria for MCI in PD,
1
which are as follows: (i) subjective complaint of cognitive decline by patient or accompanying person; (ii) performance of one to two standard deviations below the standardised mean on at least two tests, represented by either two impaired tests in one cognitive domain or one impaired test in two different cognitive domains, on a formal, comprehensive neuropsychological test (Repeatable Battery for the Assessment of Neuropsychological Status [RBANS] in this study) and (iii) absence of dementia. The comprehensive neuropsychological assessment measured five cognitive domains with at least two tests for each domain, as suggested by the Movement Disorders Society Task Force: (1) attention and working memory; (2) executive function; (3) language; (4) memory and verbal learning and (5) visuospatial abilities. PD-dementia was defined as (1) a dementia syndrome with insidious onset and slow progression, developing within the context of established PD; (2) impairment in at least two of the four core cognitive domains (attention, memory, executive and visuospatial functions) that represents (3) a decline from premorbid level; and (4) deficits severe enough to impair daily life, independent of the impairment ascribable to motor or autonomic symptoms.
21
Patients were excluded if they had (1) a cardiac pacemaker, intracranial metal clips or deep brain stimulator or (2) a history of seizure or serious medical conditions. The protocols for rTMS administration followed in this study were in accordance with the safety guidelines for rTMS.
22
Flow of recruitment, group allocation, pre- and post-rTMS evaluation and analysis.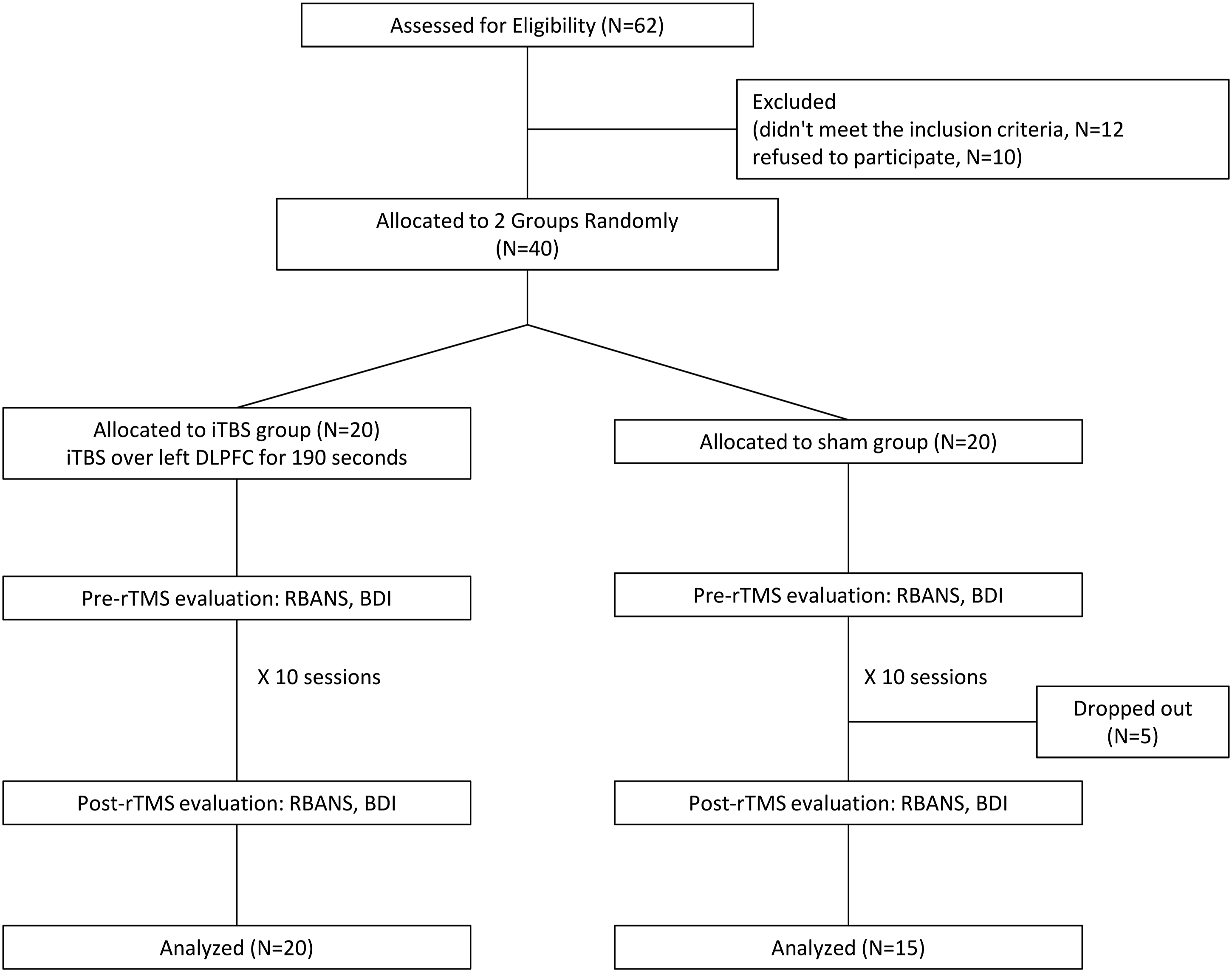
Design
This was a randomised, controlled and double-blind study. The randomisation order was computer generated and concealed in sequentially numbered opaque envelopes by a statistician who was not affiliated with this study. The 40 participants were randomly assigned to either an iTBS (n = 20) or sham group (n = 20). Each group received 10 daily sessions of treatment over the left DLPFC for 10 consecutive weekdays. Five patients in the sham group could not attend all the sessions; therefore, they were excluded from the final analysis. Their stated reasons were as follows: their families could not assist them in transportation with the hospital (three patients) and they admitted themselves to another hospital for rehabilitation during the 2-week intervention (two patients) (Figure 1). Each participant’s dose of PD medication was unaltered throughout the course of the study.
Determining Resting Motor Threshold
We used a Magstim Rapid2 stimulator (Magstim Company, Whitland, Dyfed, United Kingdom) with a 70-mm figure-8-shaped coil for stimulation. We required patients to sit on a fixed chair in a relaxed position with their eyes open. We then placed the coil over the primary motor cortex, where we recorded the largest consistent motor-evoked potential (MEP) response from the contralateral first dorsal interosseous (FDI). The resting motor threshold (RMT) for the FDI was determined to be that at which the MEP of at least a 50-
TBS Protocol and Procedure
Per standard protocol, 12 we conducted one session of iTBS every weekday for 2 consecutive weeks for a total of 10 sessions. According to the international 10–20 electroencephalography (EEG) recording system, we set (1) the target site of DLPFC to be on the F3 point of each individual participant and (2) the stimulation intensity at 100% of RMT. For the sham stimulation, we applied a placebo coil (Magstim), which delivered <5% of the magnetic output with an audible click on discharge. Because all the participants had no experience of rTMS, they did not know whether they were receiving real or sham rTMS.
Assessment of Cognitive and Depression Status
The Montreal Cognitive Assessment (MoCA), 24 RBANS 19 and Beck Depression Inventory (BDI) 25 were administered by a therapist who was blinded to group allocation at three time points: before the first intervention (which served as the baseline), immediately after the 10th session and 3 months after the intervention. The comprehensive test, that is, the RBANS, comprises five domains measuring distinct neurocognitive abilities which are the following: immediate memory, delayed memory, visuospatial and constructional abilities, language and attention. Greater cognitive function is indicated by a higher total score, which was adopted as a summary statistic for the five domains. To reduce the learning effect caused by repeated assessment, we used alternate versions of the RBANS and MoCA after intervention (at the second timepoint). In addition, because the target of rTMS modulation for cognition facilitation (DLPFC) is identical to the one used for depression modulation, whether the cognitive effect was derived from mood improvement was uncertain. Therefore, we evaluated depression status by using the BDI at baseline and postintervention.
Brain Scans and Data Processing
The decrease in dopamine active transporter (DAT) in patients with PD was measured with high sensitivity and specificity through 99mTc-TRODAT-1 SPECT.26,27 This examination serves as an ideal tool for the evaluation of PD and even as a diagnostic marker for PD. Therefore, we adopted TRODAT SPECT imaging to determine whether iTBS conditioning was related to the characteristics on SPECT, where this relationship may constitute a predisposing factor to the responsiveness of rTMS. A nuclear physicist blinded to the purpose of the study prepared the 99mTc-TRODAT-1 using a lyophilised kit, with 2 ml of tracer and a dose of 925 MBq (25 mCi), which were supplied from the Institute of Nuclear Energy Research of Taiwan. 28 At four hours after an intravenous injection of 99mTc-TRODAT-1, the technician performed SPECT scans and acquired the SPECT images using a MULTISPECT 3 gamma-camera (Siemens, German), with fanbeam collimators as well as 120 equally spaced projections over 360°. Each step took 60 s, and a 128 × 128 matrix was used. The nuclear physicists used backprojection for each image that was then reconstruction using a Ramp–Butterworth filter with a cut-off frequency of .3 per cm and an order of 10. The nuclear physicist corrected the database for photon attenuation through first-order Chang’s method with an attenuation coefficient of .12 and with attenuation ellipses, which was defined on the summed images of the entire dataset and administered to all images. 28 We set 2.9 mm as the slice thickness and in-plane resolution, calculated as the sum of three reconstructed transaxial slices and reoriented it to be parallel to the orbitomeatal line, with the highest signal in the region of the basal ganglion as the central slice.
For data processing after SPECT scan, based on the stereotactic shape and individual morphology of the basal ganglia (BG), a neuroradiologist drew the standard template of the region of interest (ROI), which comprised the putamen, caudate nucleus and occipital cortex in the composite slice of each hemisphere. Normalised uptake of BG was calculated as (ACPBG – ACPIO)/ACO, where ACPBG is the average count per pixel (ACP) of the BG ROI, ACPIO is the ACP of the ipsilateral occipital ROI and ACPO is the ACP of the occipital ROI. The results were classified into right versus left BG uptake ratio.28,29 Prior to their participation in the rTMS protocol, the patients underwent one scan to measure their bilateral BG uptake ratio
Statistical Analysis
The TBS and sham groups were compared with respect to their baseline and biographic data using a Mann–Whitney test or Chi-Square test, depending on the distribution. To determine improvements in MoCA, RBANS and BDI scores and in the attendant subscores after the stimulation, we used a Wilcoxon signed-rank test for intragroup comparisons. We conducted a linear regression for the intergroup comparisons of changes in MoCA, RBANS and BDI scores, with age, sex and baseline RBANS or MoCA scores as explanatory variables. Furthermore, we conducted a linear regression for the relationship of the BG uptake ratio with changes in MoCA, RBANS and BDI scores postintervention, with age and sex as explanatory variables. All these analyses were performed using SPSS version 22.0 (IBM, Armonk, NY, USA). Statistical significance was indicated if p < .05.
Results
Demographic and Clinical Characteristics
Demographic and Clinical Characteristics of All Patients at Baseline.
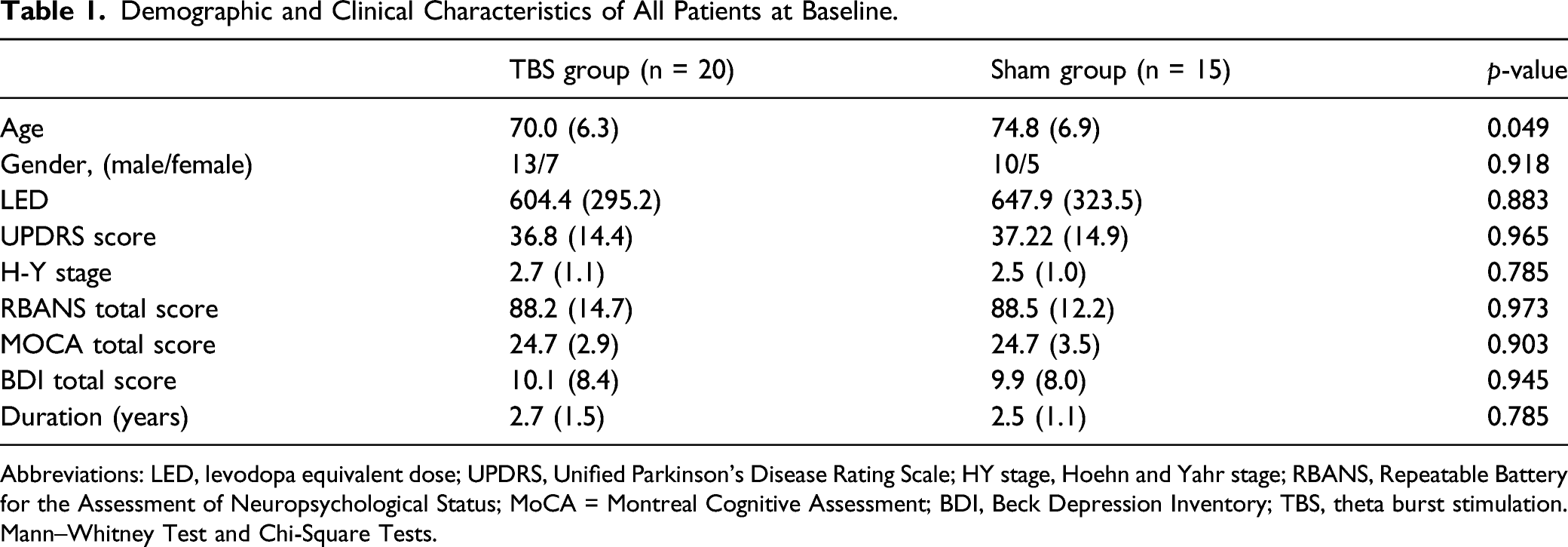
Abbreviations: LED, levodopa equivalent dose; UPDRS, Unified Parkinson's Disease Rating Scale; HY stage, Hoehn and Yahr stage; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status; MoCA = Montreal Cognitive Assessment; BDI, Beck Depression Inventory; TBS, theta burst stimulation. Mann–Whitney Test and Chi-Square Tests.
Improvements in the Various Groups
Mean (SD) Group Data of Neuropsychological Assessments in Each Group.
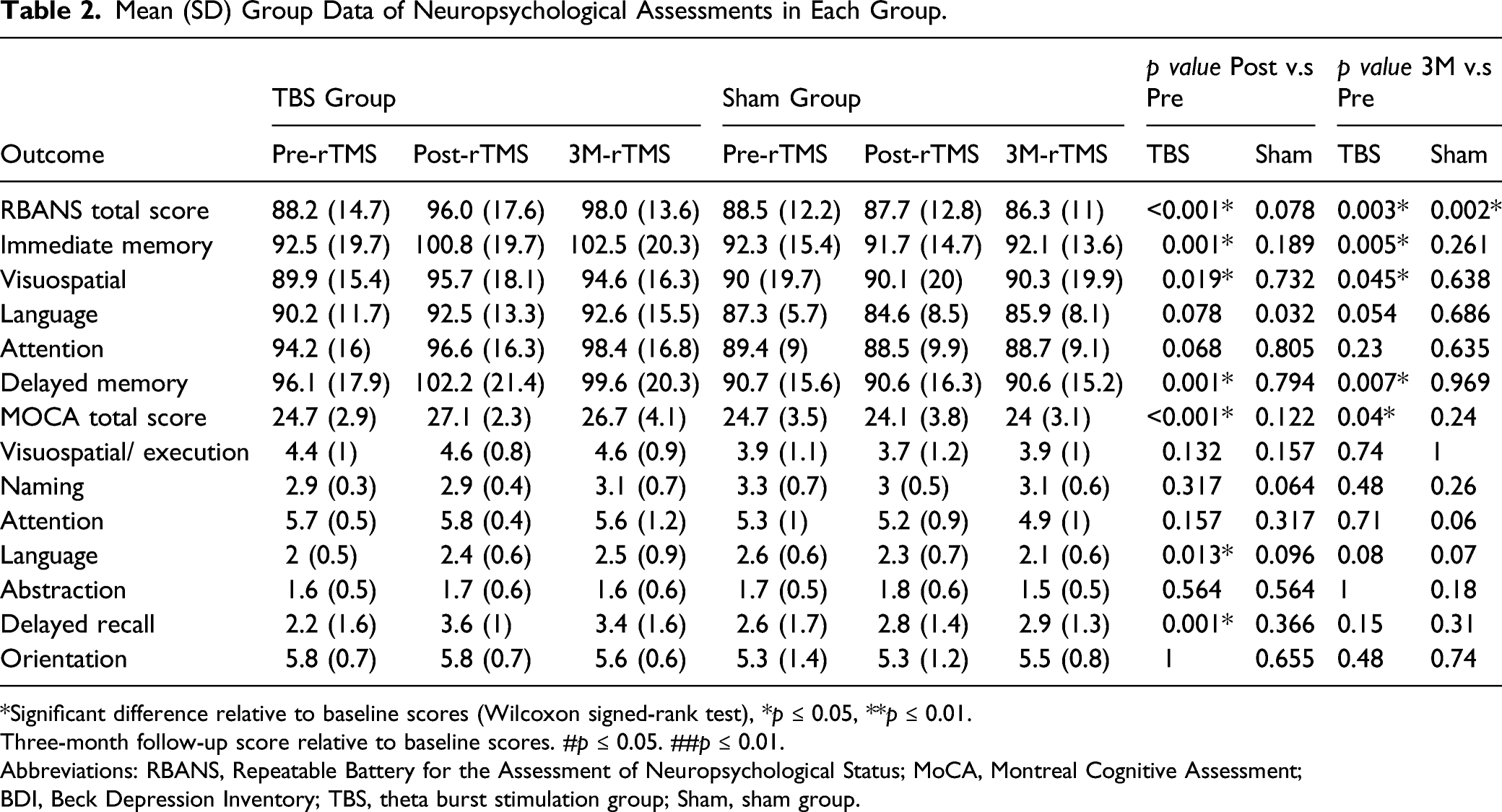
*Significant difference relative to baseline scores (Wilcoxon signed-rank test), *p ≤ 0.05, **p ≤ 0.01.
Three-month follow-up score relative to baseline scores. #p ≤ 0.05. ##p ≤ 0.01.
Abbreviations: RBANS, Repeatable Battery for the Assessment of Neuropsychological Status; MoCA, Montreal Cognitive Assessment; BDI, Beck Depression Inventory; TBS, theta burst stimulation group; Sham, sham group.
Intergroup Comparisons
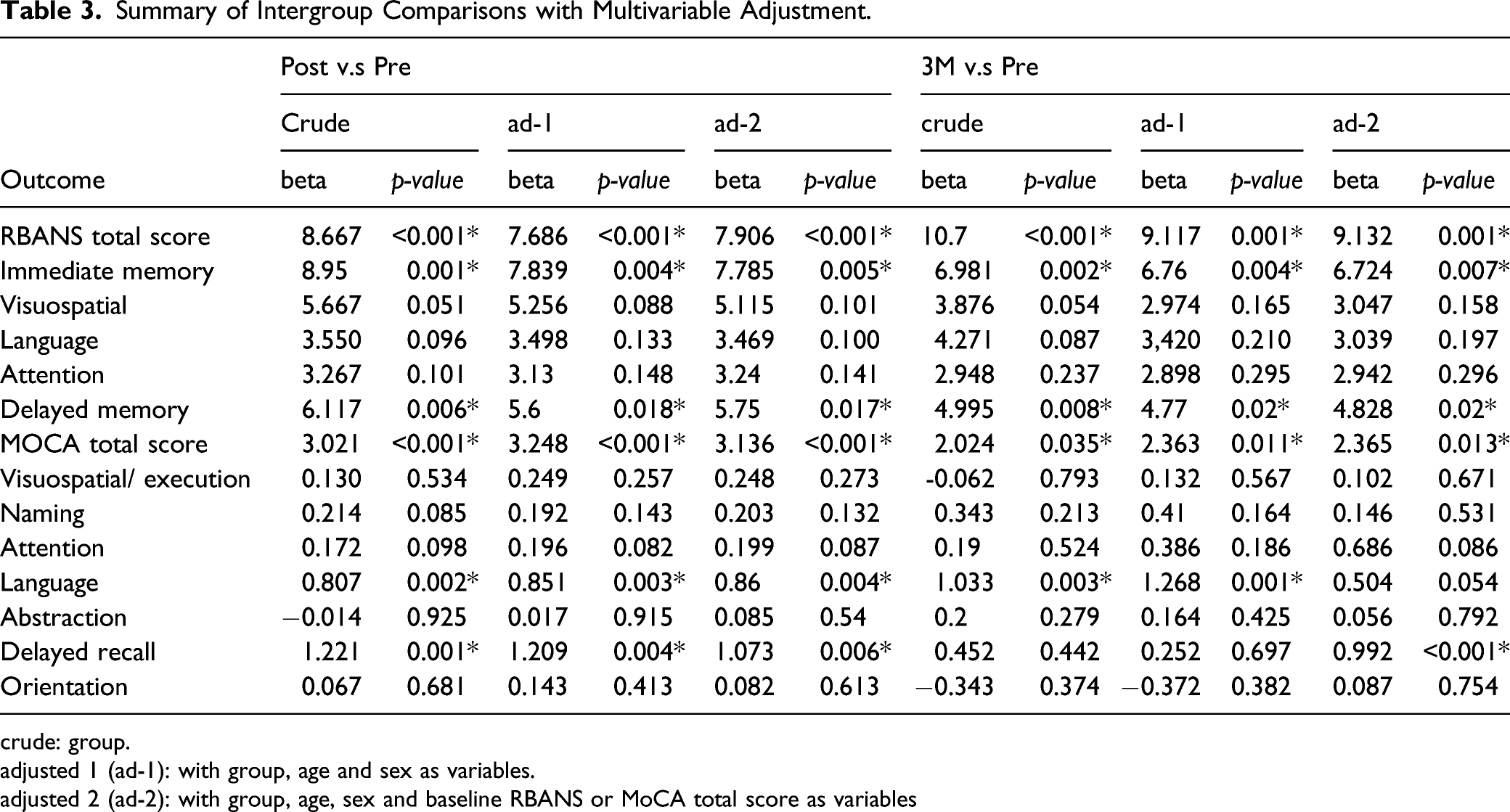
Compared with the sham group (Figure 2), the TBS group had a significantly larger poststimulation RBANS total score (p < .001) and MoCA total score (p < .001) in the linear regression, with age, sex and baseline cognitive function adjusted for. With regard to performance in each domain for the TBS group, we noted significant improvements in the IM (p = .005) and DM (p =.017) scores in the RBANS. Moreover, the significant improvements in RBANS total score (p = .001) and MoCA total score (p = .013) in the TBS group were found to persist for at least 3 months after the stimulation. Furthermore, we noted significant improvements in RBANS IM (p = .007) and DM scores (p = .02) at 3-month follow-up (Table 3). Significant mean group findings for intergroup comparisons. Postintervention score relative to baseline score *p ≤ 0.05. **p ≤ 0.01. Three-month follow-up score relative to baseline score. #p ≤ 0.05. ##p ≤ 0.01. (Linear regression analysis, with age and gender as explanatory variables). RBANS = Repeatable Battery for the Assessment of Neuropsychological Status; MoCA = Montreal Cognitive Assessment. Summary of Intergroup Comparisons with Multivariable Adjustment. crude: group. adjusted 1 (ad-1): with group, age and sex as variables. adjusted 2 (ad-2): with group, age, sex and baseline RBANS or MoCA total score as variables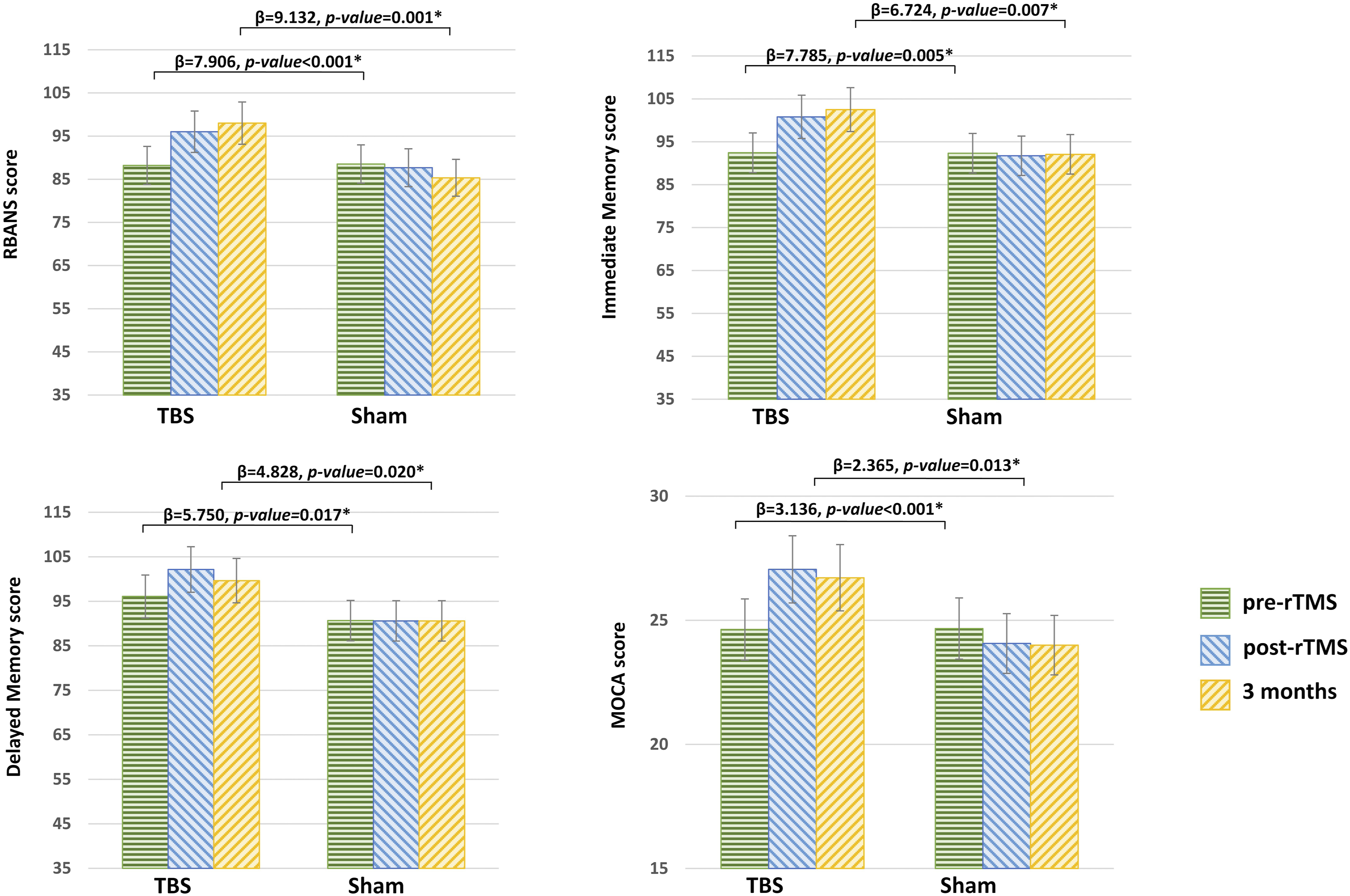
Correlation Between Tc-TRODAT SPECT Imaging and iTBS Effect
Linear regression analyses revealed significant correlations between MoCA improvement after the 10-session intervention and right caudate uptake (β = .701, p = .021), right putamen uptake (β = .653, p = .039) and left caudate uptake (β = .754, p = .021). Patients who displayed higher radiotracer uptake in the bilateral basal ganglion at baseline appeared to respond better to iTBS conditioning.
Discussion
This randomised, sham-controlled, double-blind study contributes to a growing body of evidence that consecutive iTBS over the left DLPFC has therapeutic potential for MCI in patients with PD. This benefit of iTBS extends to overall cognitive performance, immediate and delayed memory in the short posttreatment period. Notably, these significant improvements persist for up to 3 months after intervention. The iTBS, under our current protocol, exhibited a broad and persistent modulation effect on cognitive function. Furthermore, we also demonstrated the value of a SPECT scan in predicting how responsive a patient with PD-MCI will be to iTBS. This study is the first to provide evidence that 10-session iTBS modulation produces substantial and persistent cognitive improvements after applying a comprehensive set of tools for neuropsychological assessment.
PD is a type of neurodegenerative disorder related to the lack of dopamine and to the perturbation of dopamine signalling. Its principal motor symptoms of tremor and bradykinesia can be initially controlled using drugs, such as L-dopa or dopaminergic agonists. 30 However, such dopaminergic medication only marginally improves cognition. A neuroimaging study of patients with PD-MCI found that striatal dopamine depletion and reduced D2 receptor availability in the bilateral insula are associated with cognitive impairment. 31 Dopamine projections from the substantia nigra to the striatum and frontal cortex form a key mechanism for learning, attention, working memory and decision-making. 32 Therefore, rTMS is expected to mitigate cognitive decline through neuromodulation on the dopaminergic pathway. Recent evidence from an animal model revealed that 25 Hz rTMS at 110% RMT over the frontal region greatly and continually increases extracellular dopamine concentrations in the dorsolateral striatum of rats. 33 A prior study using in vivo positron emission tomography scans demonstrated that 10 Hz rTMS over the prefrontal cortex (PFC) may facilitate dopamine release in the frontostriatal circuitry, which in turn improves cognitive performance in healthy individuals. 34 For older adults with MCI, rTMS has been recognised as a safe and well-tolerated alternative intervention for improving cognition. 35 Protocol of iTBS exerts a more durable effect on cortical excitability relative to its counterparts, and we adapted this protocol due to its better response in cognitive modulation. In the field of neuropsychology, LTP-like effects mediated by DLPFC iTBS may activate cognitive corticostriatal loops, doing so through glutamatergic projection that in turn indirectly facilitates the release of dopamine in the BG. 36 According to neuroimaging evidence, cognition control has the characteristics of cognition task specificity and the lateralisation of functional difference, meaning that the left PFC is associated with the top-down control of attention, executive function 37 and working memory, 38 whereas the right PFC plays major roles in motor control inhibition and macro adjustments for cognitive flow. 37 Therefore, in this study, we used DLPFC modulation to investigate the efficacy of iTBS on the manipulation of mainstream cognitive functions.
Researchers have investigated rTMS in DLPFC to alleviate PD-related problems with cognition; results have been mixed, and high-quality RCTs have been rare. 39 Most preliminary studies have suggested the usefulness of rTMS in treating PD-MCI; however, evidence for this claim has been insufficient, and other studies have furnished opposite findings. An RCT of 25 patients with PD demonstrated that 15 Hz rTMS over the left DLPFC at 110% RMT over 10 sessions can significantly improve performance in the Stroop, Hooper and Wisconsin tests. 40 Another RCT involving 22 patients with PD evaluated the effect of 5 Hz rTMS over the left DLPFC at 90% RMT for 10 days 41 ; that RCT reported improvements in the Stroop test and mood status, which were maintained for 1 month after the treatment. Opposite results were obtained by an RCT in which iTBS at 80% active motor threshold (AMT) over the left DLPFC was administered to 26 patients with PD for eight sessions over 2 weeks. 42 That study reported that their treatment significantly improved mood but not motor symptoms or cognition. In summary, these pioneering studies have had small sample sizes, differences in stimulation intensity and the lack of a comprehensive assessment tool, which have inevitably led to conflicting results. Two recent RCTs involving patients with PD-MCI have reported only marginal improvements in the active groups, with nonsignificant differences compared with the sham groups after iTBS treatment over the left DLPFC.9-11 Both studies used comprehensive assessments; however, they followed a six-session iTBS protocol (twice daily for 3 days with a 1–2-day break in between), which may have resulted in a lower cumulative effect compared with the 10-day protocol used in the present study. Additionally, our present study featured a more potent treatment, whereby iTBS was administered at a high intensity of 100% RMT, making our findings more robust than those of previous studies. We demonstrated that iTBS exerted broad and persistent improvements in general neuropsychological condition, working memory and linguistic function.
In our present study, we applied iTBS over the left DLPFC and found that the TBS group exhibited significant improvements in IM and DM compared with the sham group. This improvement was observed both immediately after the intervention course and at 3-month follow-up. As part of working memory, IM and DM involve complex processes to manipulate information that is mediated through different cortical and subcortical structures, such as the PFC and basal ganglion. 43 When the working memory is engaged, a series of parallel loops provide the appropriate time and contexts for the execution of complex cognitive tasks. 44 In PD, the dysfunction of the cholinergic network in the nucleus basalis of Meynet contributes to memory impairments. 45 Our findings are consistent with those of previous studies on patients with stroke and healthy individuals. Persistent dysfunction of immediate and delayed recall in patients with left hemispheric stroke can be alleviated using conventional high-frequency rTMS over the left DLPFC. 7 Hoy et al 14 studied healthy individuals and, through comparisons with a sham group, reported that iTBS improved working memory. 14 The mechanisms underlying this effect may be associated with increased gamma-aminobutyric acid levels 46 in the corticostriatal circuit and with acetylcholine inputs into the hippocampus after modulation. 47
Scholars generally believe that cognitive deficit from PD profoundly interferes with cognitive planning and linguistic ability, such as phonemic fluency, semantic fluency and the grasp of pragmatics. 48 The BG–thalamocortical motor circuits through the putamen are likely to play a key role in phonemic fluency. 49 In a study of patients with dementia, high-frequency rTMS over the DLPFC improved their auditory sentence comprehension. 50 Verbal fluency requires the organisation of information retrieval and recall, self-initiation and perseveration. Verbal fluency, categorised under the subscale of initiation/perseveration in the Dementia Rating Scale, is considered a test of execution. 21 Sentence repetition reflects listening and perception abilities, grammatical skills and speech production and also measures working memory.51,52 Patients with Alzheimer’s disease and impaired sentence repetition ability may present with ending omissions or word substitutions, which are significantly correlated with performance on the Digit Span test. 53 Therefore, we speculate that our findings of improved speech fluency and repetition ability may be attributed to improved cognitive processes.
The significant correlation that we noted between cognitive improvement and striatal tracer uptake following the intervention lends support to the usefulness of SPECT as a tool for predicting how much a patient will respond to rTMS modulation. Patients who exhibit greater striatal dopamine activity at baseline are more sensitive to iTBS-DLPFC modulation. As mentioned, the improvement of cognitive function was found to be associated with the recovery of DAT binding. 54 Numerous studies have reported the close relationship between striatal dopamine activity and cognition function.55,56 Similarly, our SPECT findings imply that the striatal dopamine reserve contributes to a patient’s response to iTBS treatment with respect to improvement in cognitive function.
Our study is limited by its small sample size. Future large-scale studies are warranted to confirm the immediate and long-term effects of this intervention. Because the remote effects of rTMS based on changes in microstructure or functional connectivity remain largely unknown, our findings can be expanded in future studies through the use of neuroimaging techniques, such as functional magnetic resonance imaging, diffusion tensor imaging or SPECT, to elucidate the underlying mechanism. Further studies using a larger and more homogenous sample, a longer intervention period or more intensive courses are warranted to determine the optimal treatment protocol.
This was a standard randomised, controlled and double-blind consecutive iTBS study involving patients with PD along with MCI; a set of comprehensive assessment tools were used to measure outcomes. Our results indicated the long-term benefits of consecutive iTBS over the left DLPFC for cognitive improvement. Because MCI is common in the early stage among patients with PD and is a risk factor for the progression to dementia, this study provides a novel intervention to help clinicians more safely and effectively prevent the disease through neuromodulation. Future large-scale studies should address whether iTBS, when applied in daily clinical settings, constitutes a viable treatment alternative for cognitive deficits in patients with PD.
Footnotes
Acknowledgement
The authors thank Hsin-Yi Huang for her contribution to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Taipei Veterans General Hospital Grant (V106C-108) and the MOST Grant (107-2314-B-075-010).
Ethics Committee
The Institution Review Board of Taipei Veterans Veterans General Hospital.
