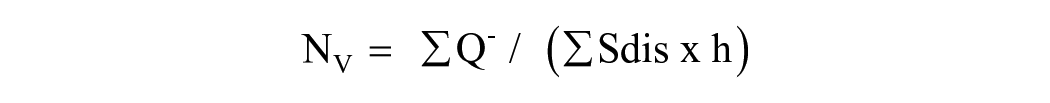Abstract
Purpose
Exercise reduces cognitive deficits in traumatic brain injury (TBI), but early post-trauma exercise is often discouraged due to potential harm. The purpose was to evaluate the interaction between pre- and post-injury physical exercise on cognition, neuronal survival and inflammation.
Methods
Rats were either sham-operated and kept sedentary (Sham) or subjected to controlled cortical impact injury and then distributed into sedentary (Tbi), pre-injury exercise (Pre-Tbi), post-injury exercise with early (24 hours, Tbi-early) or late (6 days, Tbi-late) onset, and a combination of pre- and post-injury exercise with early (Pre-Tbi-early) or late (Pre-Tbi-late) onset. Object recognition memory, hippocampal volume, neuronal survival (NeuN+) in the hippocampus and perirhinal cortex, and microglial activity (Iba-1) in the hippocampus were evaluated.
Results
All exercise conditions, except TBI-early, attenuated the significant memory impairment at 24-hour retention caused by TBI. Additionally, Pre-TBI-early treatment led to memory improvement at 3-hour retention. Pre-TBI reduced neuronal death and microglial activation in the hippocampus. TBI-late, but not TBI-early, mitigated hippocampal volume loss, loss of mature neurons in the hippocampus, and inflammation. Combining pre-injury and early-onset exercise reduced memory deficits but did not affect neuronal death or microglial activation. Combining pre-injury and late-onset exercise had a similar memory-enhancing effect than late post-injury treatment alone, albeit with reduced effects on neuronal density and neuroinflammation.
Conclusions
Pre-TBI physical exercise reduces the necessary onset delay of post-TBI exercise to obtain cognitive benefits, yet the exact mechanisms underlying this reduction require further research.
Introduction
Every year, millions worldwide experience traumatic brain injury (TBI), 1 with many facing long-term cognitive issues including attention and memory deficits, 2 and increased risk of dementia. 3 This underscores the need for treatment not only in the acute phase but also long-term. Several non-pharmacological therapies such as environmental enrichment4–6 and physical exercise (PE)7–9 have been suggested as a potential therapeutic approach to improve cognitive recovery following mild, moderate, and severe TBI.
Rest post-TBI has traditionally been recommended, but recent data challenges prolonged rest, especially after mild TBI. 10 Identifying the ideal timing for transitioning from rest to PE remains unclear, as it may depend on factors such as injury severity and pre-injury fitness. Tailoring interventions to individual patient characteristics has been proposed to address this variability. 11 The benefits of post-injury PE appear to be linked to a decrease in the neurodegenerative processes, primarily a reduction of inflammation12,13 and neuronal death,12–18 as well as an increase in regenerative processes, mainly an increase in neuroplasticity13,19,20 and neurogenesis.12,13,15
In animal models, post-injury PE has been shown to alleviate memory deficits, but controversy exists regarding the optimal delay to start PE. While benefits are generally observed with longer delays (over a week),12,13,17,19 contradictory results arise with shorter delays, with some papers reporting benefits,12,14–16,21 and others an exacerbation of the deficits.13,18,19 Although experimental procedures may contribute to these discrepancies, the nature of events occurring after the initial insult may also play a role. TBI pathogenesis occurs in two phases: a primary injury caused by direct mechanical forces and a secondary damage that develops later, offering a window for therapeutic interventions. It is suggested 22 that the significant mitochondrial damage and impact on brain metabolism 23 occurring in the initial hours and days after injury could be exacerbated by PE during the acute post-injury phase, since the energy demand associated to PE adds to an already compromised metabolism. Thus, starting PE too early might worsen cognitive consequences. However, since the secondary injury triggering cognitive issues involves early-activated processes, initiating post-injury PE earlier may improve long-term prognosis.
Pre-injury PE can mitigate the consequences of trauma on the brain, particularly in the initial hours or days. It helps stabilize brain metabolism post-impact24–27 and diminishes the initial inflammatory reaction,24,26–30 reducing neuronal loss24,26,28–30 and memory deficits.25,26,29–31
As far as we know, the interaction between pre- and post-injury PE on memory has not been studied. Given that pre-injury PE mitigates some of the early effects of brain trauma, it is possible that this training may allow earlier initiation of post-injury PE, enhancing its beneficial effects without negative consequences. Considering that usual inpatient rehabilitation is mainly inactive or sedentary, 32 this could be relevant when trying to adjust the post-injury treatment to the specific characteristics of the individual patients.
This study aims to investigate how pre-injury training influences the impact of post-injury PE treatment on memory, neuroprotection, and microglial activation. Specifically, it explores whether pre-injury PE could shorten the delay required to initiate post-injury PE treatment.
Materials and Methods
Animal procedures followed European and local laws (2010/63/EU; Real Decreto 1386/2018) and the protocols were approved by Animal Experimentation Commission of Autonomous Government of Catalonia (No. 9734-P2). 82 six-week-old male Sprague rats (Charles River Laboratories; Abresle, France) weighing 249.09±22.09 g were used. Animals were individually housed and maintained under controlled conditions on a 12 hours light-dark cycle (lights on at 8:00 a.m.), temperature 20 to 22°C and 40% to 70% humidity. Water was available ad libitum, while food was provided in a fixed amount (30 g/day) to prevent overfeeding without caloric restriction, promoting better long-term health compared to ad libitum conditions.
The animals were randomly divided into seven groups (Figure 1): (1) Sham: sham-surgery and sedentary condition; (2) Tbi: controlled cortical impact (CCI) surgery and sedentary condition; (3) Pre-Tbi: voluntary exercise for 18 days before CCI surgery and post-TBI sedentary condition; (4) Tbi-early: CCI surgery and voluntary exercise for 14 days from 24 hpi; (5) Tbi-late: CCI surgery and voluntary exercise for 9 days from 6 dpi; (6) Pre-Tbi-early: voluntary exercise for 18 days before CCI surgery and from 24 hpi (14 days); (7) Pre-Tbi-late: voluntary exercise for 18 days before CCI surgery and from 6 dpi (9 days).
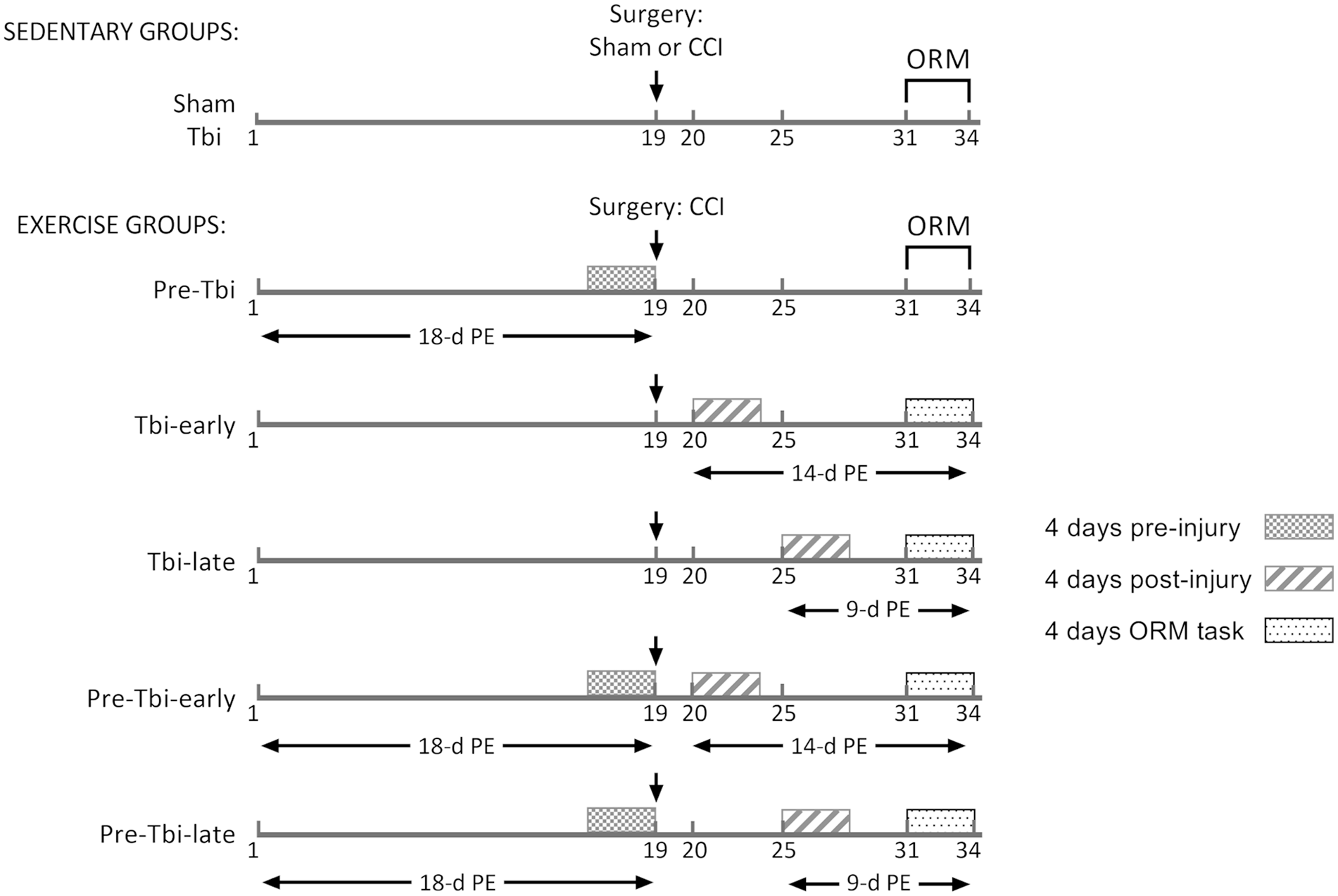
Timeline of the experimental procedures.
Stereotaxic Surgery and TBI
TBI was induced by means of CCI (Pittsburgh Precision Instruments, Inc, Pittsburgh, PA), as described previously. 15 Anaesthesia was induced with 5% isoflurane (Forane, Abbot Laboratories, SA, Madrid, Spain) in oxygen (1 l/minute) for 5 minutes. The animals were then placed in a stereotaxic frame (David Kopf Instruments, Tujunga, CA), and the anaesthesia was maintained by delivering 2% isoflurane in oxygen (0.8 l/minute) through a nose mask. The scalp was incised along the midline, and after exposing the skull, a craniectomy (4 mm diameter) was performed over the right hemisphere (4.5 mm posterior to bregma and 3 mm from midline). A 3-mm diameter rod impacted into the right hemisphere at a speed of 6.0 m/second, reaching a depth of 2.0 mm below the dura matter layer and persisting for 150 millisecond. Sham animals underwent similar procedures, but no impact was applied. To control for post-operative pain a non-invasive and stress-free oral treatment with buprenorphine (0.4 mg/kg) mixed in Nutella® (2 g/kg) was administered to all the animals 1 hour pre- and 23 hours post-surgery. 33
Physical Exercise
During sedentary periods, animals were kept in their standard cages, while for periods of PE activity, they were maintained in cages (48 × 26 × 20 cm) connected to a 37 cm diameter running wheel (Rat Wheel W/Brake, ENV-042: Med Associates Inc, St. Albans, VT, USA). The time and distance run were recorded daily and analyzed in three different periods: (1) the last 4 days before surgery (for those animals with pre-TBI training), (2) the initial 4 days of PE post-surgery, (3) the 4 days during memory training.
Object Recognition Memory Task
The Object Recognition Memory (ORM) task started 12 dpi as described previously. 12 Sessions were recorded via software Anymaze (Stoelting Europe, Dublin, Ireland) and manually analyzed in a blinded procedure.
Training began with three sessions (12 minutes each) of habituation to the experimental box, two of which were held on the same day, separated by a 90-minute interval, and the third carried out on the following day. Locomotor activity during these sessions was recorded. To assess potential anxiety reactions to novel objects, a neophobia test was conducted 90 minutes after the last habituation session. During this test, animals were allowed to explore a box containing an unfamiliar object for 10 minutes, and latency to initial exploration was recorded.
The subsequent day, a 15-minute acquisition session took place, during which animals explored two identical objects positioned 10 cm from the walls. Retention sessions (5-minute) occurred at 3 and 24 hours post-acquisition, involving one familiar object from the acquisition session and one novel object. Object assignments and positions were randomized to prevent bias. The position of the familiar object was reversed between the 3-hour and the 24-hour test. Exploration time for each object was recorded.
To analyze cognitive performance while accounting for differences in total exploration time, a discrimination index was computed using the formula: [((time exploring the novel object – time exploring the familiar object)/total time spent on both objects) × 100]. 34 An index higher than zero indicates good recall of the familiar object, as rats naturally tend to explore novel objects. A 10-second minimum exploration time during the acquisition session was required for statistical analyses inclusion. 35
Tissue Processing
Animals were euthanized 24 hours post-memory test with sodium pentobarbital (200 mg/kg), followed by 4% paraformaldehyde (PFA) intracardial perfusion. Brains were extracted, post-fixed in PFA for 3 hours, rinsed, cryoprotected in 30% sucrose/PB, frozen and stored at −80°C. Coronal cryostat sections (40 μm) were taken between −2.76 and −4.48 anteroposterior coordinates from bregma. 36 Each animal provided nine series of free-floating sections and one series of gelatin-coated slides, facilitating systematic random sampling.
Nissl-stained sections were digitized, and hippocampal area was measured using FIJI image analysis software. 37 Dorsal hippocampal volume was estimated by multiplying total surface areas by section thickness. An interhemispheric ratio [(ipsilateral volume/contralateral volume) × 100] was calculated to estimate hippocampus volume loss.
Two series of free-floating slices from six animals per group were processed for NeuN, a marker for mature neurons, 38 and Iba1, expressed by microglia/macrophage cells, 39 following the procedure previously described. 12
For NeuN and Iba-1 immunohistochemistry, endogenous peroxidases were blocked via incubation with 0.3% hydrogen peroxide solution. Sections were washed and nonspecific binding was blocked with bovine serum solution for 1 hour and incubated overnight with primary antibody (Mouse anti-NeuN, MAB377, Sigma, 1:500 or rabbit anti-Iba-1, GTX100042 Genetex, AntibodyBCN, 1:1000). Following washes, sections were incubated in biotinylated secondary antibody (NeuN, Goat anti-Mouse IgG (H+L), SAB 4600004, Sigma, 1:1000 or Iba-1, Goat anti-Rabbit A1664 Thermo Fisher Scientific, 1:500), and then in a streptavidin-biotin horseradish peroxidase complex (1:3600, SA-HRP conjugate, NEL 750001EA: Perkin Elmer, Tres Cantos, Spain) dilution followed by 3,3’-diaminobenzidine. Sections were mounted on slides, dehydrated and cleared, and then cover-slipped.
Quantification of NeuN+ Cells
Serial digitized images of NeuN-stained sections were obtained at 10× magnification for the hilus of dentate gyrus (Hil) of hippocampus and the perirhinal cortex (PRhc) using a DSLR camera (EOS 6D Mark II, Canon) coupled to a microscope (Axio Imager A1, Carl Zeiss Iberia). FIJI software 37 stitched partial images together to create whole Hil or PRhc images for each hemisphere.
In two or three slices between −2.76 and −4.20 from bregma, 36 Hil region was outlined and NeuN+ cells were quantified manually due to the relatively low quantity of neurons in this region. The density of neurons was calculated by dividing the number of NeuN+ cells by the area of the Hil. To assess neuronal loss in the ipsilateral hemisphere to the lesion, a ratio between hemispheres was calculated [(ipsilateral density/contralateral density) × 100] for each slice, followed by the mean of these ratios across all examined slices.
Due to the high neuronal density of the PRhc, stereological principles were applied using a random uniform systematic sampling system. The PRhc area was manually outlined, and a grid with counting frames of 1245 μm2 was randomly superimposed. Cells were manually counted in every 25 frames, excluding those touching the right and lower edges but including those touching the left and upper edges to avoid edge effects. Neuron density was calculated using a formula adapted from optical dissector calculations 40 :
Where Nv, numerical density; ∑Q−, sum of quantified cells; ∑Sdis, sum of quantified dissectors (counting frames) by dissector area; h, height of the slice (given a value of 1 as quantifications were carried out on microphotographs).
Finally, the neuron density ratio was calculated using the same method as in the Hil.
Quantification of Iba-1 Staining
Serial digitized images of three Iba-1-stained sections between −2.76 and −4.20 from bregma 36 were obtained at 5× magnification for the dorsal hippocampus using the camera and microscope previously described. Consistency in illumination parameters were ensured, and a post hoc correction was applied to prevent vignetting effects. 41 FIJI software 37 stitched partial images together to create whole dorsal hippocampus images for each hemisphere. To assess the mean intensity and the percentage of area stained, the following steps were followed: Black and white inversion was performed so that higher values indicated higher staining. The contralateral dorsal hippocampus was manually outlined. An automatic threshold (Huang dark option) was applied, and the mean gray value and percentage of area were measured. The ipsilateral dorsal hippocampus was manually outlined, and the contralateral hemisphere’s minimum value was set as a threshold. The mean gray value and percentage of area were measured.
Similar to NeuN analysis, a ratio was calculated [(ipsilateral/contralateral) × 100] for each slice, followed by the mean of these ratios across all examined slices.
Statistical Analyses
The analyses were conducted using the statistical programming language R 3.6.3 42 and the graphical interface Jamovi. 43 Outliers were identified using box-plot analyses, confirmed with a one-sample t-test against the group mean, and subsequently excluded from the corresponding analyses. Groups differences were examined through a one-way analysis of variance using a between-group design, and post-hoc comparisons were conducted with Tukey correction. If homogeneity of variances was lacking, Welch and Games-Howell corrections were applied. One-sample t-tests were used to determine whether group mean values significantly differed from the reference value (0 for the discrimination index and 100 for the interhemispheric ratio). Mean and SEM of all the variables can be found in the Supplemental Material. Statistical significance was set at P < .05.
Results
Three animals died due to complications from anaesthesia, and four were excluded from the analysis due to abnormally large lesions not comparable with those of the other animals (1 Tbi-sed, 1 Tbi-late, 1 Pre-Tbi-early, 1 Pre-Tbi-late). The final sample comprised 75 rats: Sham (11), Tbi (12), Pre-Tbi (12), Tbi-early (11), Tbi-late (9), Pre-Tbi-early (10), and Pre-Tbi-late (10).
Exercise Behavior
During the pre-injury period, no differences between groups were found (Figure 2A). In the first 4 days post-injury (Figure 2B), differences between groups in the mean distance run [F(3,17.76) = 10.57; P < .001] were found. Pre-injury PE (Pre-Tbi-early and Pre-Tbi-late) groups ran higher distances than non-pre-injury groups (Tbi-early P = .031, P = .003; Tbi-late P = .036, P = .006). Similar results were obtained for the time spent running for both the whole groups [F(3,18.08) = 11.37; P < .001], and the post-hoc comparisons (Tbi-early P = .015, P = .003; Tbi-late P = .011, P = .009).
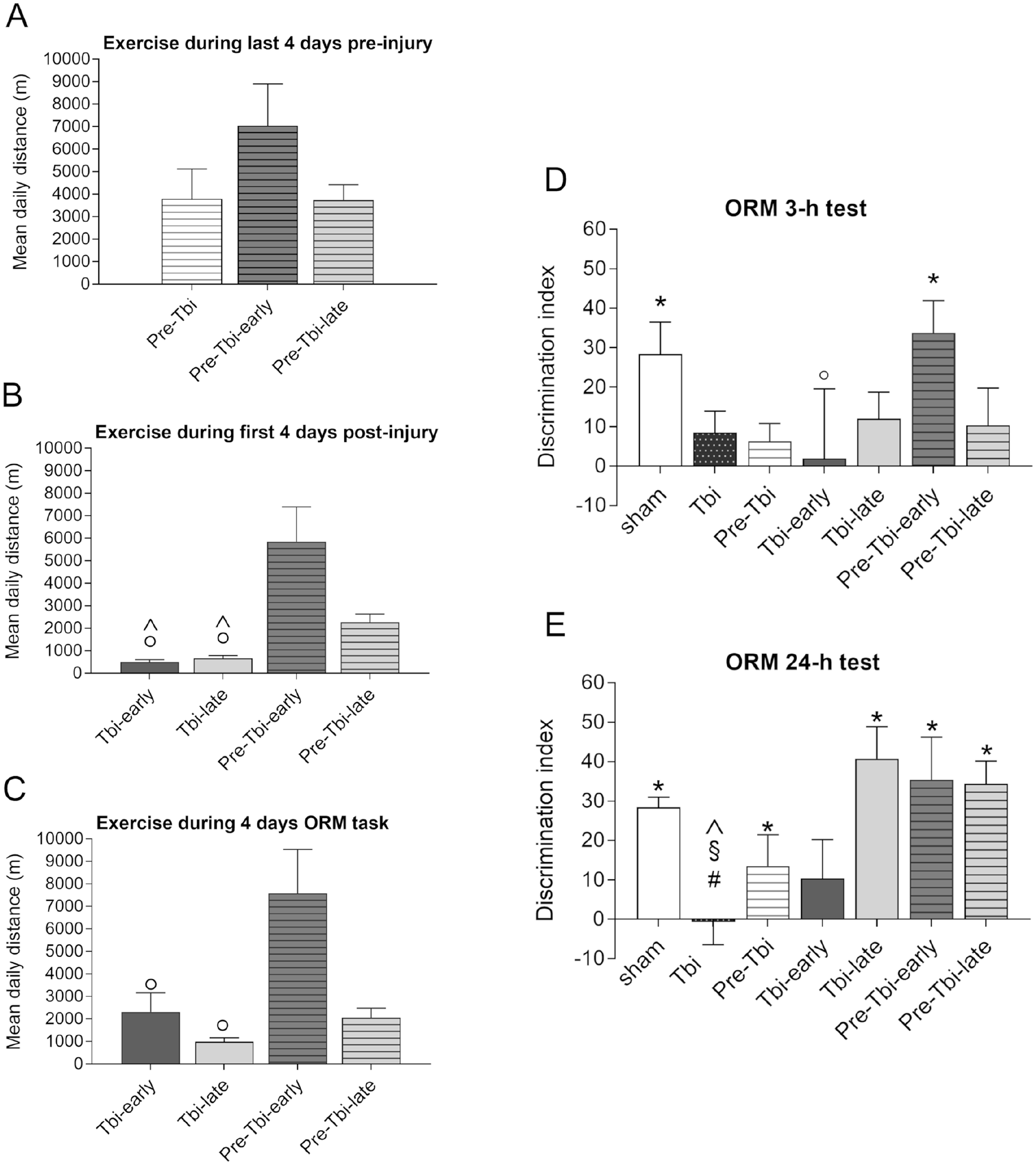
Mean (+SEM) daily distance (m) run in the last 4 days pre-injury (A), the four first days post-injury (B) and in the 4 days during the ORM task (C) for each exercising groups. Mean (+SEM) values of discrimination index for each experimental group in the retention tests of the object recognition memory task carried out 3 hours (D) and 24 hours (E) after acquisition.
Regarding the 4 days corresponding to the ORM (Figure 2C), differences between groups were observed in the mean distance run [F(3,17.11) = 4.95; P = .012] and in the time spent running [F(3,17.23) = 4.94; P = .012]. Specifically, Pre-Tbi-early group ran a greater distance than Tbi-early (P = .039) and Tbi-late (P = .034) groups and spent more time running than Tbi-late (P = .021) group.
Object Recognition Memory
Due to computer issues, data from three animals were lost in all ORM sessions (1 Tbi, 2 Pre-Tbi-early), from one animal in the first retention session (Tbi-early), and from three animals in the second one (2 Sham, 1 Tbi-late).
Group differences were detected in the third habituation session [F(6,27.6) = 4.44; P = .003]. The Pre-Tbi group showed higher locomotion compared to the Tbi-late (P = .016) and Pre-Tbi-late (P = .035) groups. No group differences were detected in the neophobia test.
In the acquisition session, between groups analysis [F(6,63) = 5.04; P < .001] and post-hoc comparisons revealed that total exploration time was lower in the Tbi-early (P = .006) and Pre-Tbi-early (P < .001) groups compared to the Sham group, and in the Pre-Tbi-early compared to the Tbi group (P = .008).
In the 3-hour retention session (Figure 2D), discrimination indices were significantly higher than 0 only in the Sham [t(10) = 3.525; P = .005] and Pre-Tbi-early [t(7) = 4.171; P = .004] groups. Between-groups differences were detected [F(6,63) = 2.68; P = .022], indicating that the Pre-Tbi-early group had a higher discrimination index than the Tbi-early group (P = .045).
For the 24-hour retention session (Figure 2E), one-sample t-tests revealed discrimination indices above 0 in the Sham (P < .001), Pre-Tbi (P = .017), Pre-Tbi-early (P < .001), Tbi-late (P = .002), and Pre-Tbi-late (P = .013) groups. Between-groups analyses [F(6,25.51) = 4.64; P = .017] and post-hoc comparisons indicated a lower discrimination index in the Tbi group compared to the Sham (P = .007), Tbi-late (P = .015) and Pre-Tbi-late (P = .006) groups. No between-groups differences were detected in the total time of exploration in the two retention sessions.
Hippocampal Volume
One Pre-Tbi-late subject’s tissue was damaged and excluded from staining. All CCI rats exhibited a lesion cavity over the ipsilateral parietal lobe and a deformation of the dorsal hippocampus (see Figure 3). Ipsilateral hippocampal volume reductions compared to the contralateral side were found in the Tbi [t(11) = −6.63; P < .001], Pre-Tbi [t(10) = −5.38; P < .001], Tbi-early [t(10) = −4.68; P = .001], Tbi-late [t(7) = −3; P = .02], Pre-Tbi-early [t(8) = −7.71; P < .001] and Pre-Tbi-late [t(8) = −5.25; P = .005] groups, but not in the Sham group. Between-groups analysis [F(6,25.41) = 26.53; P < .001] and post-hoc comparisons showed a higher volume ratio in the Sham group compared to the Tbi (P < .001), Pre-Tbi (P = .002), Tbi-early (P = .008), Pre-Tbi-early (P < .001) and Pre-Tbi-late (P = .005) groups, and in the Tbi-late compared to the Tbi (P = .004), Tbi-early (P = .032) and Pre-Tbi-early (P = .002) groups. No differences were found between the Sham and Tbi-late groups.
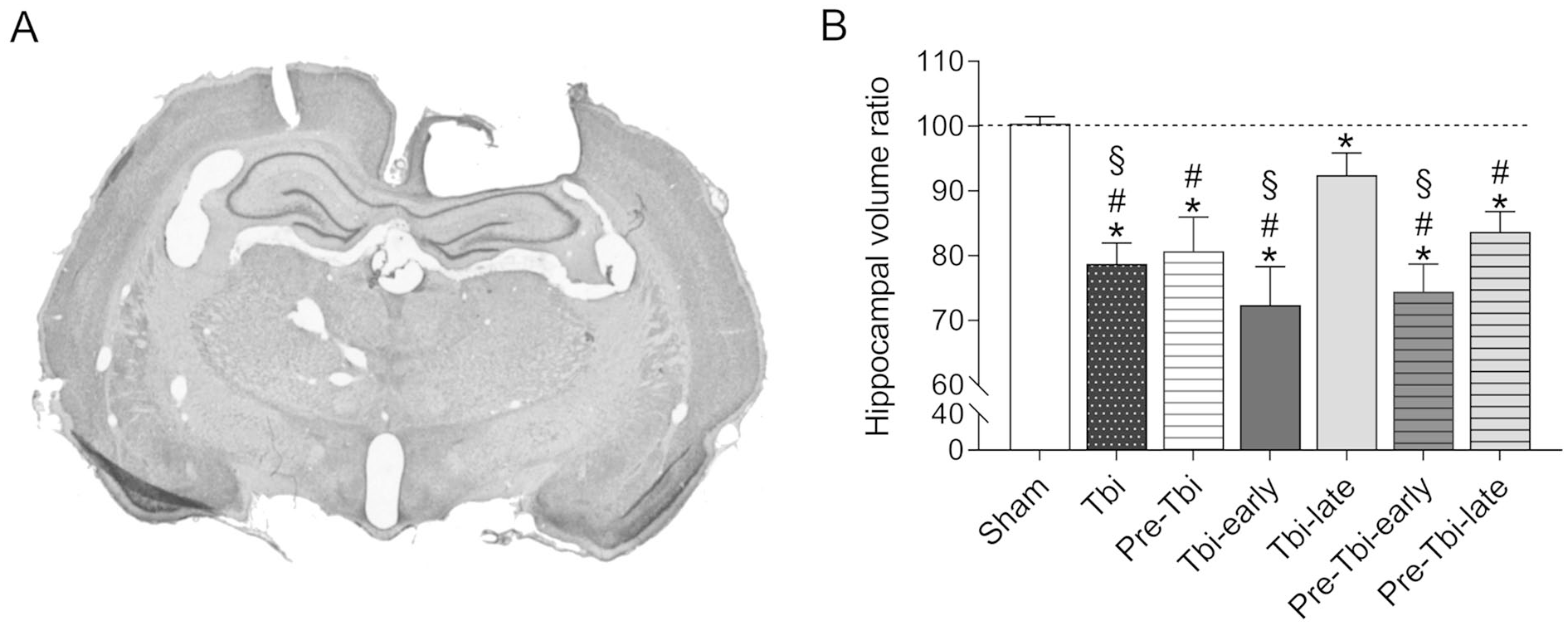
Representative microphotography of a coronal section stained for cresyl violet in an animal with TBI (A). Mean (+SEM) interhemispheric ratio of hippocampal volume for each experimental group (B).
Density of NeuN+ Cells
Figure 4B depicts the mean interhemispheric ratio of NeuN+ cells density in the Hil. One-sample t-tests showed that the Tbi [t(5) = −5.12; P = .004], Pre-Tbi [t(5) = −3.23; P = .023], Tbi-early [t(5) = −19.5; P < .001], Pre-Tbi-early [t(5) = −15.6; P < .001] and Pre-Tbi-late [t(5) = −3.34; P = .021], but not the Sham and Tbi-late groups, had lower ratios than 100. Between-group analysis [F(6,14.8) = 57.9; P < .001] and post-hoc comparisons showed lower ratios in the Tbi (P = .022), Tbi-early (P < .001) and Pre-Tbi-early groups (P < .001) compared to the Sham group, and in the Tbi-early (P = .013) and Pre-Tbi-early (P = .004) groups compared to the Tbi-late group.
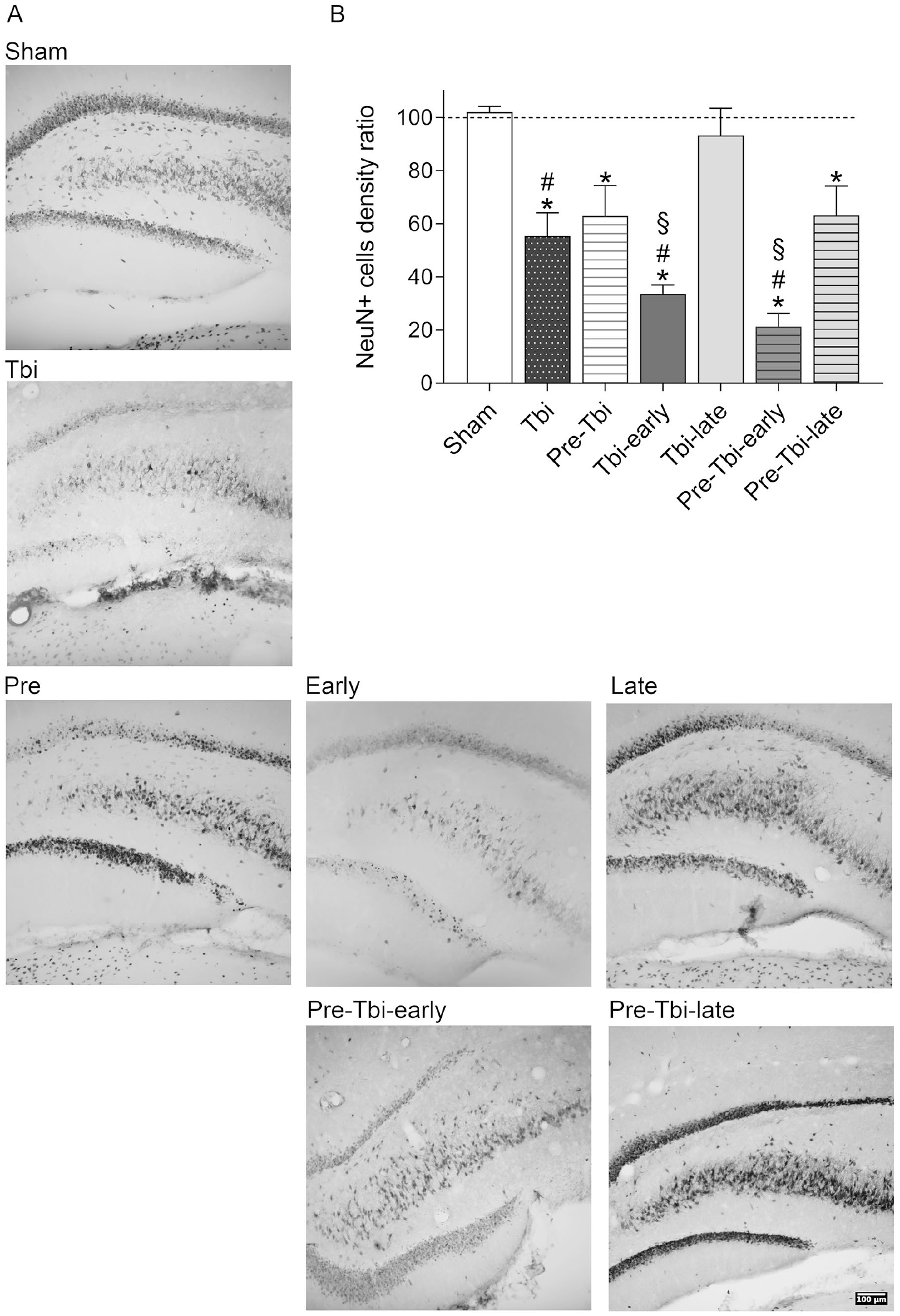
Detail of representative stitched images of the ipsilateral hilus of the dentate gyrus of NeuN immunostained slices for each experimental group (A). Mean (+SEM) interhemispheric ratio of NeuN+ cells density in the hilus of dentate gyrus in each experimental group (B).
Figure 5B depicts the mean interhemispheric NeuN+ cells density ratio in the PRhc. One-sample t-tests showed that ratios of the Tbi [t(5) = −13.7; P < .001], Pre-Tbi [t(5) = −5.07; P = .004], Tbi-early [t(5) = −4.19; P = .009], and Pre-Tbi-early [t(5) = −4.72; P = .005] groups, but not the Sham, Tbi-late and Pre-Tbi-late groups, were lower than 100. Between-group analysis [F(6,14.34) = 34.7; P < .001] and post-hoc comparisons showed lower ratios in the Tbi (P < .001), Pre-Tbi (P = .019), Tbi-early (P = .045) and Pre-Tbi-early (P = .032) compared to the Sham group, and in the Tbi (P < .001), Pre-Tbi (P = .009), Tbi-early (P = .026) and Pre-Tbi-early (P = .022) groups compared to the Tbi-late group.
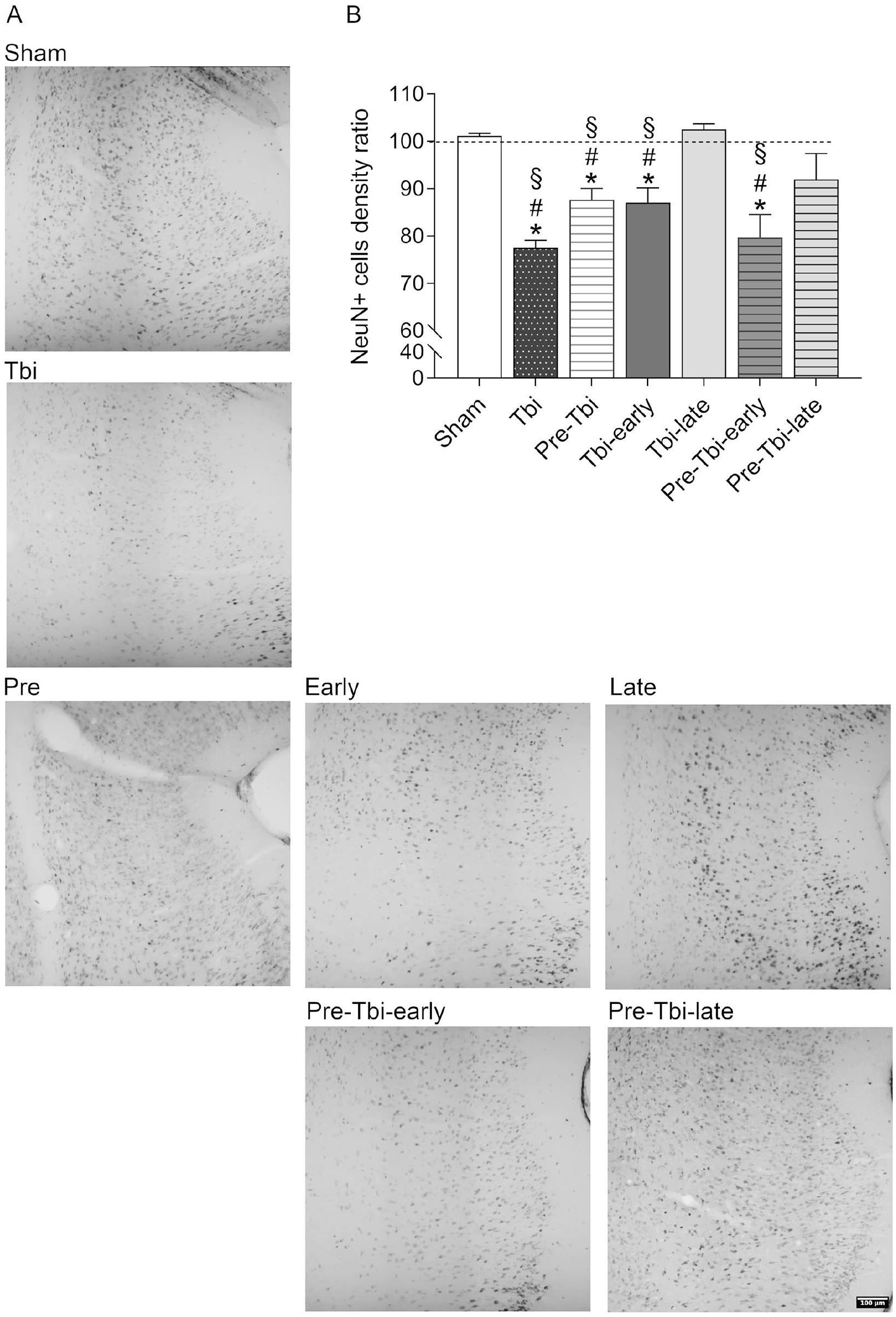
Detail of representative stitched images of the ipsilateral perirhinal cortex of NeuN immunostained slices for each experimental group (A). Mean (+SEM) interhemispheric ratio of NeuN+ cells density in the perirhinal cortex in each experimental group (B).
Iba Staining in Dorsal Hippocampus
For the mean intensity ratio (Figure 6B top), one-sample t-tests showed that the Tbi [t(5) = 7.79; P < .001], Tbi-early [t(5) = 9.46; P < .001], Pre-Tbi-early [t(5) = 7.08; P < .001] and Pre-Tbi-late [t(5) = 4.32; P = .008] groups, but not the Sham, Pre-Tbi and Tbi-late groups, were higher than 100. Between-groups analysis [F(6,14.21) = 28.35; P < .001] and post-hoc comparisons showed higher ratios in the Tbi (P = .021), Tbi-early (P = .019) and Pre-Tbi-early (0.006) groups compared to the Sham group, and in the Tbi (0.005), Tbi-early (P = .001) and Pre-Tbi-early (P = .008) groups compared to the Tbi-late group.
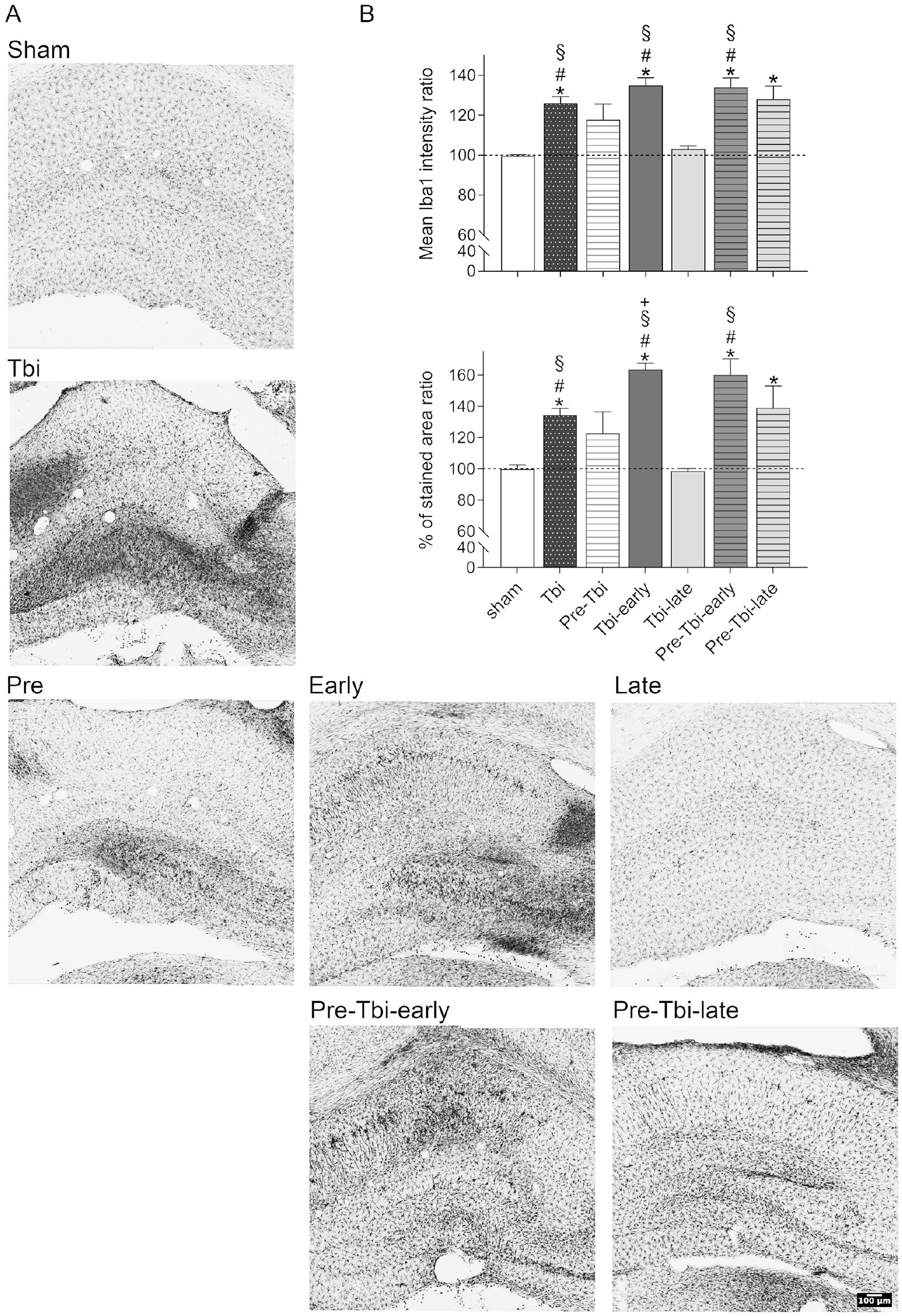
Detail of representative stitched images of the ipsilateral hippocampus of Iba-1 immunostained slices for each experimental group (A). Mean (+SEM) interhemispheric ratio of the mean intensity (top) and percentage of stained area (bottom) for the Iba-1 staining in the hippocampus in each experimental group (B).
For the percentage of stained area ratio (Figure 6B bottom), one-sample t-tests showed ratios exceeding 100 in the Tbi [t(5) = 7.89; P < .001], Tbi-early [t(5) = 15.55; P < .001], Pre-Tbi-early [t(5) = 5.82; P = .002] and Pre-Tbi-late [t(5) = 2.79; P = .039] groups, but not in the Sham, Pre-Tbi and Tbi-late groups. Between groups analysis [F(6,14.73) = 41.145; P < .001] and post-hoc comparisons showed higher ratios in the Tbi (P = .002), Tbi-early (P < .001) and Pre-Tbi-early (P = .014) groups compared to the Sham group, and in the Tbi (P = .002), Tbi-early (P < .001) and Pre-Tbi-early (P = .014) groups compared to the Tbi-late group. Additionally, the Tbi-early group showed a higher ratio than the Tbi (P = .008) group.
Discussion
Our findings emphasize the effectiveness of PE in treating cognitive deficits post-TBI, highlighting the importance of two factors: the interval between the injury and the onset of PE, and the interaction with prior training.
Effects of CCI
The TBI led to short (3 hours) and long-term (24 hours) memory deficits in the ORM assessed 2 weeks post-trauma. These outcomes were not due to differences in habituation, neophobia, or acquisition sessions. CCI also resulted in reduced volume of the dorsal hippocampus and neuronal loss in both the Hil and PRhc, with increased microglial reactivity in the dorsal hippocampus. Neuronal loss in the PRhc may contribute to recall impairment at 3 hours, as it is involved in recognizing familiarity rather than long-term memory consolidation. 44 Long-term memory deficits may be attributed to TBI-induced changes in the hippocampus, which is crucial for memory retrieval during retention. 45 Its involvement becomes more pronounced when the memory test has a significant spatial component, 46 as evidenced by our 24 hours test where the location of the familiar object changes between the first and second tests. These results were expected given TBI’s consistent association with memory deficits in ORM and other tasks,12,14,15,19,47 neurodegeneration in the PRhc 15 and hippocampus,12–15,29,48 and increased microglial activity in the hippocampus, possibly indicating an inflammatory response.12,48,49
Exercise as Post-Training Treatment After a TBI: The Earlier, the Better?
Given the evidence that PE diminishes neurodegeneration after a TBI by reducing neuroinflammation12,13 and the associated neuronal death,12–16 initiating PE earlier may yield greater benefits. In this study, we investigated two post-TBI PE protocols (Tbi-early and Tbi-late groups). However, early initiation of PE did not produce any cognitive or histological improvements compared to untreated injured subjects. The lack of ORM improvement does not appear to be linked to reduced exploration during acquisition, as all animals met criteria, and the Pre-Tbi-early group, with similar exploration values, performed well in both retention sessions. Previous experiments with short post-trauma delays (1-2 days), yielded conflicting results: some positive,16,21,28 while others negative or none, 19 suggesting caution in recommending early treatment. In contrast, delayed PE reversed memory deficits and reduced TBI-associated histological changes, indicating neuroprotective and anti-inflammatory effects. Our results confirm prior findings on the benefits of post-TBI PE, especially with delayed onset.12,15,17,19,49,50 The variation in treatment efficacy based on the onset delay of post-injury PE might be linked to events in the initial hours and days post-TBI, marked by extensive mitochondrial damage and impaired cerebral metabolism. 23 Our findings support the idea of a time window after TBI, during which metabolic changes from the trauma and metabolic needs of PE may interact, compromising PE efficacy and potentially causing harmful effects. As time elapses post-trauma, compromised cerebral metabolism decreases, reducing its interference with PE’s energetic demands. The timing of this interaction depends on factors such as injury severity, treatment intensity, and pre-trauma fitness. The window for obtaining benefits from post-training PE has been poorly studied, likely due to the challenge of equalizing all temporal variables that influence PE effects. Essentially, initiating treatment with different delays but maintaining the same duration implies that the cognitive evaluation cannot be carried out at the same time points post-trauma in all the groups. 19 Conversely, when treatment delays vary but evaluation time and treatment duration are kept constant, a pause without PE is required. 13 Thus, these designs imply either evaluating at different stages of secondary damage evolution or introducing a significant temporal gap between PE cessation and cognitive assessment. Our design lacks any treatment interruption of the post-TBI treatment, with cognitive tests conducted at the same post-injury moment, this, however, results in differences in treatment duration since this treatment was initiated either early or late after CCI. This parallels the study by Chen et al., 21 using a closed-head injury model with mice and forced PE. In their study, the early initiation group began PE after 48 hours and continued for 14 days, while the delayed initiation group started at 9 days and exercised for 7 days. In contrast to our findings, their study demonstrated beneficial effects from early PE on behavior and histology after TBI, with late initiation showing no such benefits. Various factors, including animal type (rats vs mice), TBI method (CCI vs closed head impact) and severity, timing of PE initiation (24 hours vs 48 hours) and duration, and PE protocol (voluntary vs forced), may contribute to these disparate outcomes.
Is Early Onset of PE Always Harmful After TBI?
Our findings demonstrate that 18 days of pre-trauma PE (Pre-Tbi group) alleviate long-term memory deficits. Similar outcomes have been reported in prior studies using various tasks, such as the Morris water maze, with both voluntary and forced PE.24,25,29–31 Histologically, pre-trauma PE did not alter the hippocampal volume reduction induced by TBI, but partially mitigated neuronal density loss in the ipsilateral Hil and reduced microglial reactivity in the hippocampus. Similar neuroprotective effects have been previously described.24,26,28–30 Thus, given the beneficial effects observed with pre-injury treatment at both behavioral and histological levels, we hypothesize that pre-injury PE could enable an earlier initiation of post-trauma treatment to harness its benefits from an early stage after the trauma.
The combined PE programs yielded unexpected results. Early post-TBI PE (Pre-Tbi-early group) improved cognitive function by reducing short-term and long-term memory deficits in trained subjects. However, this cognitive improvement was not accompanied by neuroprotective effects on the PRhc or the Hil, nor by a decrease in microglial reactivity in the hippocampus. Conversely, combining prior training with delayed post-injury PE (Pre-Tbi-late) produced similar cognitive effects but with less neuroprotection and reduced impact on neuroinflammation than delayed post-treatment alone (Tbi-late). These outcomes might be linked to the amount of PE. Animals with pre-injury training exhibited significantly higher levels of PE post-TBI compared to those without prior training. This disparity stems from the fact that subjects with pre-injury PE are already accustomed to the running wheel and are more physically fit. Then, the increased metabolic demands associated with high levels of PE may have counteracted the reduction produced by pre-trauma PE treatment during the energy crisis post-TBI. This imbalance might be more pronounced in the early stages post-TBI and gradually diminish as the delay between trauma and initiation of PE increases. Consequently, the Pre-Tbi-early group did not demonstrate neuroprotection, while the Pre-Tbi-late group exhibited intermediate neuroprotection between the Pre-Tbi-early and Tbi-late groups.
If neuroprotection was compromised in the Pre-Tbi-early group, another mechanism must explain the cognitive benefits observed. Previous studies indicate that PE not only activates neuroprotection but also stimulates neurorepair processes like neurogenesis12,15 and neurotrophins such as BDNF. 50 Studies have shown a positive correlation between the volume of voluntary PE and the increase in various neurotrophins, and other molecules related to plasticity.20,51 Therefore, the beneficial effects of the combined treatment could be explained by an enhancement of repairing mechanisms rather than neuroprotective mechanisms. Further experiments are needed to explore this alternative explanation.
Conclusions
Overall, PE may seem like a relatively simple treatment, but numerous factors influence its effects, including the type of PE, as well as its frequency, duration, intensity, volume, etc. Specifically, our findings demonstrate that both the delay in treatment initiation and its interaction with previous training determine the efficacy and underlying mechanisms of PE as a treatment after TBI, and support the notion that the parameters of PE as a treatment after TBI should be tailored to individual patient characteristics.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241270023 – Supplemental material for Treating Traumatic Brain Injury with Exercise: Onset Delay and Previous Training as Key Factors Determining its Efficacy
Supplemental material, sj-docx-1-nnr-10.1177_15459683241270023 for Treating Traumatic Brain Injury with Exercise: Onset Delay and Previous Training as Key Factors Determining its Efficacy by Tanit Sánchez-Martín, David Costa-Miserachs, Margalida Coll-Andreu, Isabel Portell-Cortés, Soleil García-Brito and Meritxell Torras-Garcia in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministerio de Economía y Competitividad (PSI2014-55087-R).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.