Abstract
Background. Priming results in a type of implicit memory that prepares the brain for a more plastic response, thereby changing behavior. New evidence in neurorehabilitation points to the use of priming interventions to optimize functional gains of the upper extremity in poststroke individuals. Objective. To determine the effects of priming on task-oriented training on upper extremity outcomes (body function and activity) in chronic stroke. Methods. The PubMed, CINAHL, Web of Science, EMBASE, and PEDro databases were searched in October 2019. Outcome data were pooled into categories of measures considering the International Classification Functional (ICF) classifications of body function and activity. Means and standard deviations for each group were used to determine group effect sizes by calculating mean differences (MDs) and 95% confidence intervals via a fixed effects model. Heterogeneity among the included studies for each factor evaluated was measured using the I2 statistic. Results. Thirty-six studies with 814 patients undergoing various types of task-oriented training were included in the analysis. Of these studies, 17 were associated with stimulation priming, 12 with sensory priming, 4 with movement priming, and 3 with action observation priming. Stimulation priming showed moderate-quality evidence of body function. Only the Wolf Motor Function Test (time) in the activity domain showed low-quality evidence. However, gains in motor function and in use of extremity members were measured by the Fugl-Meyer Assessment (UE-FMA). Regarding sensory priming, we found moderate-quality evidence and effect size for UE-FMA, corresponding to the body function domain (MD 4.77, 95% CI 3.25-6.29, Z = 6.15, P < .0001), and for the Action Research Arm Test, corresponding to the activity domain (MD 7.47, 95% CI 4.52-10.42, Z = 4.96, P < .0001). Despite the low-quality evidence, we found an effect size (MD 8.64, 95% CI 10.85-16.43, Z = 2.17, P = .003) in movement priming. Evidence for action observation priming was inconclusive. Conclusion. Combining priming and task-oriented training for the upper extremities of chronic stroke patients can be a promising intervention strategy. Studies that identify which priming techniques combined with task-oriented training for upper extremity function in chronic stroke yield effective outcomes in each ICF domain are needed and may be beneficial for the recovery of upper extremities poststroke.
Introduction
Clinical practice in neurorehabilitation relies on the assumption that motor learning contributes to motor recovery after injury. 1 A type of implicit learning used in clinical practice involves priming methods, which can be delivered prior to or in conjunction with the main intervention, where a previous stimulus can change a behavior.2,3 Priming theory presupposes that using an intervention strategy before or concomitant with another strategy leads to an improvement in neural activity before or during motor training through an increase in excitability and/or the normalization of inhibition, which may facilitate long-term potentiation or long-term depression-like mechanisms.2-4
However, the neural mechanisms related to motor priming vary according to the priming method, which can include (1) stimulation priming (repetitive transcranial magnetic stimulation [rTMS], transcranial direct current stimulation [tDCS], paired associative stimulation [PAS], and peripheral nerve stimulation [PNS]); (2) motor imagery and action observation (mirror therapy, computer-directed imagery, imagery directed by audiotape or video); (3) sensory priming (PNS, repetitive sensory stimulation); (4) movement priming (bilateral or unilateral movements; mirror symmetric active or passive movements; any type of exercise); and (5) pharmacology-based priming.2,3
Priming is used in the rehabilitation of poststroke patients to maximize motor gains of the upper extremity (UE), as 40% of such patients in the chronic phase present with some reduction in UE functional use. 5 According to a systematic review, effects on UE motor outcomes have been reported for several rehabilitation approaches. 6 Despite improvements in motor function with conventional motor therapies, the transfer of functional gains is more difficult to realize in everyday life; therefore, strategies that relate to real environments are promising for more effective functional transfer. 6 Among these treatment strategies is task-oriented training (TOT), which involves the active training of motor tasks performed within a clear functional context that includes complex whole task or pretask movements of the whole limb or a limb segment. This training is characterized by a high number of repetitions performed within a single session.7,8 According to the literature, TOT results in neuroplastic changes and is critical for improving motor and functional recovery. 9
Although there is evidence of the positive effects of different physiotherapy strategies, mainly TOT, new evidence points to the ability of priming stimuli to facilitate functional gains in motor and cognitive (memory, attention, and concentration) function, as these strategies prime the brain for a more plastic response. 10 In the context of neurorehabilitation, it is worth emphasizing the importance of a more comprehensive view of functional gains. Namely, a biopsychosocial approach must be considered that integrates the biological, individual, and social dimensions of health and that involves the interactions among the three main components of the International Classification of Functioning, Disability, and Health (ICF): functions and body structures, activity and participation, and personal and environmental factors. 11 According to the literature, the optimal therapeutic recovery period is within the first weeks to months poststroke. 12 In this sense, there remains a need to determine the optimal treatment for chronic stroke patients. Thus, the objective of this review was to determine the effects of priming for TOT on UE outcomes (body functions and activity) in chronic stroke patients.
Methods
This review followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement 13 and the recommendations from the Cochrane Collaboration. 14 The State of the Art through Systematic Review (StArt) guidelines were used 15 to systematize and organize the search and data extraction. The systematic review was registered in the International Prospective Register of Systematic Reviews (PROSPERO, Identifier: CRD42017074215).
Data Sources and Search Strategy
A literature search of the following electronic databases was performed: PubMed (via National Library of Medicine), CINAHL with Full Text (EBSCO), Web of Science (Thomson Scientific/ISI Web Services), EMBASE, and Physiotherapy Evidence Database (PEDro). The MeSH database was searched to identify the appropriate terms, and these terms were included in the search strategy (Supplementary Material Table). Publications in English and Portuguese were accepted. The publication period for the search was not limited.
Eligibility Criteria
This systematic review included randomized, pseudorandomized controlled trials and randomized controlled crossover trials with outcomes related to UE motor function. The study criteria for inclusion in this systematic review and meta-analysis were as follows: (1) randomized, pseudorandomized controlled trials, randomized crossover trials; (2) inclusion of an experimental group receiving a type of priming combined with TOT and a control group that received sham or no priming combined with TOT; (3) inclusion of individuals (>18 years old) who were chronic stroke survivors with brain ischemia/hemorrhage (>6 months), confirmed through imaging; (4) evaluation of 1 of 5 types of noninvasive priming (stimulation priming, motor imagery, action observation, sensory priming, or movement priming) immediately before or concurrent with the TOT; (5) assessment of at least one outcome of UE motor function related to the ICF domains, such as structure and body function or activity, were also included; and (6) inclusion of patients who received training to facilitate repetition and for a functional purpose. Studies were excluded if they used any other type of intervention that could impair the interpretation of the effects of TOT or verified the effects of associated interventions with TOT on other body parts, such as the lower limb.
The TOT protocol had to contain active repetition of motor sequences with functional objectives. Functional objectives should include complex movements involving various joints or muscular groups oriented toward motor performance assessments. 7 The training needed to contain different training components used in combination. 16 For example, functional goals, feedback, follow-up, and distributed practice (postintervention). 16
Selection Process
Through October 2019, 2 reviewers (ESMS and GNO) independently performed the selection process considering the inclusion criteria regarding study design, participants, intervention, and outcomes of interest. Potentially eligible articles were initially selected based on their titles and abstracts. Then, the full text of these articles were retrieved to verify whether they met all the inclusion criteria. In cases of disagreements between the 2 reviewers, a third reviewer (GLS) was consulted to determine the study eligibility. Authors were contacted when articles were not available or if there was important missing information.
Data Analysis
Data Extraction
A standardized electronic data extraction form was used to obtain key information regarding the participants, interventions and outcomes of interest. The data extracted from each primary study were as follows: priming category, authors, year of study, sample size, and the population characteristics of each group (sex, age, type of stroke, poststroke time). For experimental and control interventions, the types of priming and priming parameters such as frequency, intensity, polarity, pulse duration, duty cycle, location of stimuli, intervention duration, number of sessions per week, and total number of sessions were extracted. For the type of motor training and the motor training parameters, the type of task, repetitions, time for each task, intervention duration, number of sessions per week, and total number of sessions were extracted. The details of the UE intervention protocol were extracted as the intervention dose quantified by length of treatment sessions and period, number of treatment sessions, and the intensity of interventions.
In addition, measurement tools, time between measurements, ICF domain of measurements, mean and standard deviation of measurements, effect size, main results, and methodological quality score were collected from the studies. The reviewers’ contacts with corresponding authors of the primary studies to obtain information about missing data and details of the applied intervention were also documented.
Methodological Quality Assessment
The methodological quality of the included studies was assessed by the PEDro scale. 17 This scale uses a score with a total of 11 points based on the internal validity (10 items) and external validity (1 item). As recommended by the PEDro scale scoring guidelines, item 1 was not included in the final score. The total score was grouped into levels: good (7-10), fair (4-6), and poor (0-3). Two reviewers (GLS and CT) independently scored the articles, and the interrater reliability (kappa statistic) was 0.76 (P < .001), which indicates adequate agreement between the investigators regarding both assessment scales. 18
Assessment of Risk of Bias
The risk of bias (methodological quality assessment) of the studies included in the review was assessed by the Cochrane Collaboration’s tool. This tool assesses seven domains (generation of the allocation sequence, concealment of the allocation sequence, blinding, incomplete outcome data, selective outcome reporting and other biases). The risk of bias was classified as low, high, or unclear. 14
Statistical Analysis
Meta-analysis of the included studies was conducted after data pooling with the Cochrane Collaboration software (Review Manager 5.3). 19 Means and standard deviations (SDs) for each group (priming intervention and control groups) were used to determine group effect sizes by calculating mean differences (MDs) and 95% confidence intervals (95% CIs) using a fixed-effects model. When the data were presented as a percentage 31 or mean28,48 of change or only as the mean22,43 and interquartile27,41,47 or minimum/maximum, 24 the mean and the associated SD were obtained, as suggested by the Cochrane Collaboration. 14 Moreover, the heterogeneity among the included studies for each aspect evaluated was measured using the I2 statistic; values less than 30% indicated low heterogeneity, 30% to 50% indicated moderate heterogeneity, 50% to 75% indicated substantial heterogeneity, and 75% or more indicated considerable heterogeneity. When heterogeneity values were greater than 50, a random-effects model was applied.
Studies that did not provide enough information for data extraction were excluded from the meta-analysis but were still included in the descriptive review. The studies were grouped based on the type of priming and outcome measure. In studies with 3 groups, the data from the group that had undergone priming with the associated TOT and the group that underwent only the TOT were selected for analysis. The means and SDs of each group at baseline and at the end of treatment for each included study were used for the analyses.
Outcome data were pooled into categories of outcome measures considering the ICF classifications of body function and activity. For the structure and function ICF domain, the Fugl-Meyer Assessment (UE-FMA), force control, range of movement, and the modified Ashworth scale score were considered the outcome measures. In addition, for the activity ICF domain, the outcome measures used were the Wolf Motor Function Test (WMFT), the Action Research Arm Test (ARAT), the Motor Activity Log (MAL), the Jebsen-Taylor Hand Function Test (JTHFT), and the Box and Block Test (BBT). According to the literature, these outcome measures have been widely used to assess UE motor function and activity. 20 As improvements in some measures can be indicated by higher and lower values, differences between groups can be expressed as negative and positive values. Thus, all negative values were converted into positive values. Therefore, for the purpose of the statistical analyses, data from all studies were transformed such that a positive value represented an effect in favor of the intervention and a negative value represented an effect in favor of the comparison.
In cases where the evaluation measures fell into more than one domain, measures were included in only one of these domains according to the description in the included article. For example, if the included study specified the use of the Fugl-Meyer Assessment to assess body function, the activity domain was not considered, even given that the assessment includes some items related to mobility.
Quality of Evidence
We used the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) system 14 to assess the quality of evidence. The GRADE system provides critical components (study limitations, imprecision, inconsistency, indirectness, and publication bias) of this assessment. 14 Although the quality of evidence represents a continuum, the GRADE system assesses the quality of a body of evidence as high (the confidence in the estimate of the effect is very unlikely to change), moderate (an important impact on the confidence in the estimate of the effect; the estimate may change), low (an important impact on the confidence in the estimate of effect; the estimate is very likely to change), or very low (any estimate of the effect is very uncertain). 14
Results
A total of 3227 articles were identified through an electronic search. After the selection process, 125 full-text articles were assessed for eligibility, and 36 articles fulfilled all the inclusion criteria (Figure 1). Of those 36 studies, 31 presented high methodological quality4,21-50 and 5 studies presented fair quality.51-55 All included studies had a score ≥6 on the PEDro scale (Table 1).
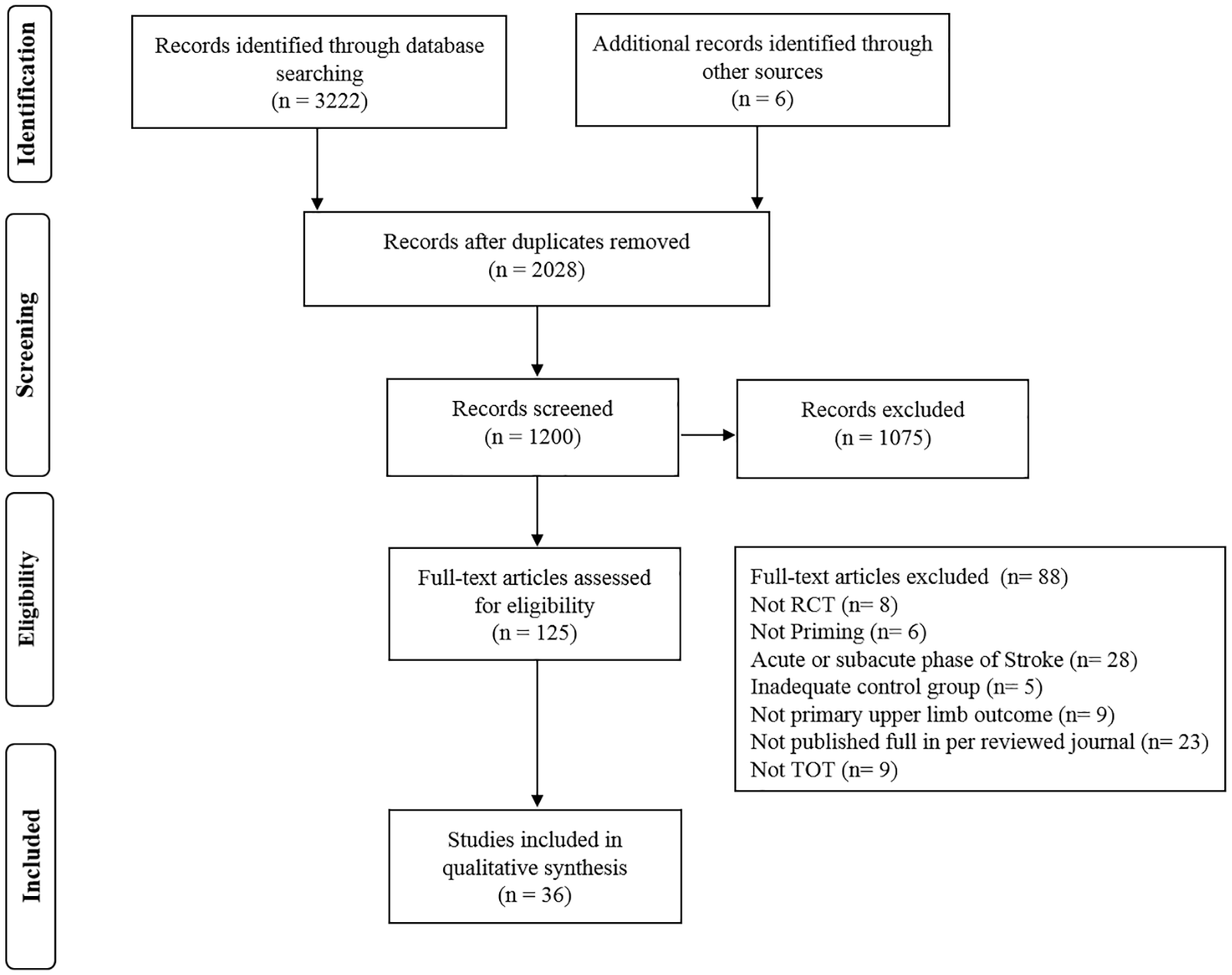
Study flow diagram.
Methodological Quality of the Included Studies (Physiotherapy Evidence Database [PEDro] Scale). a
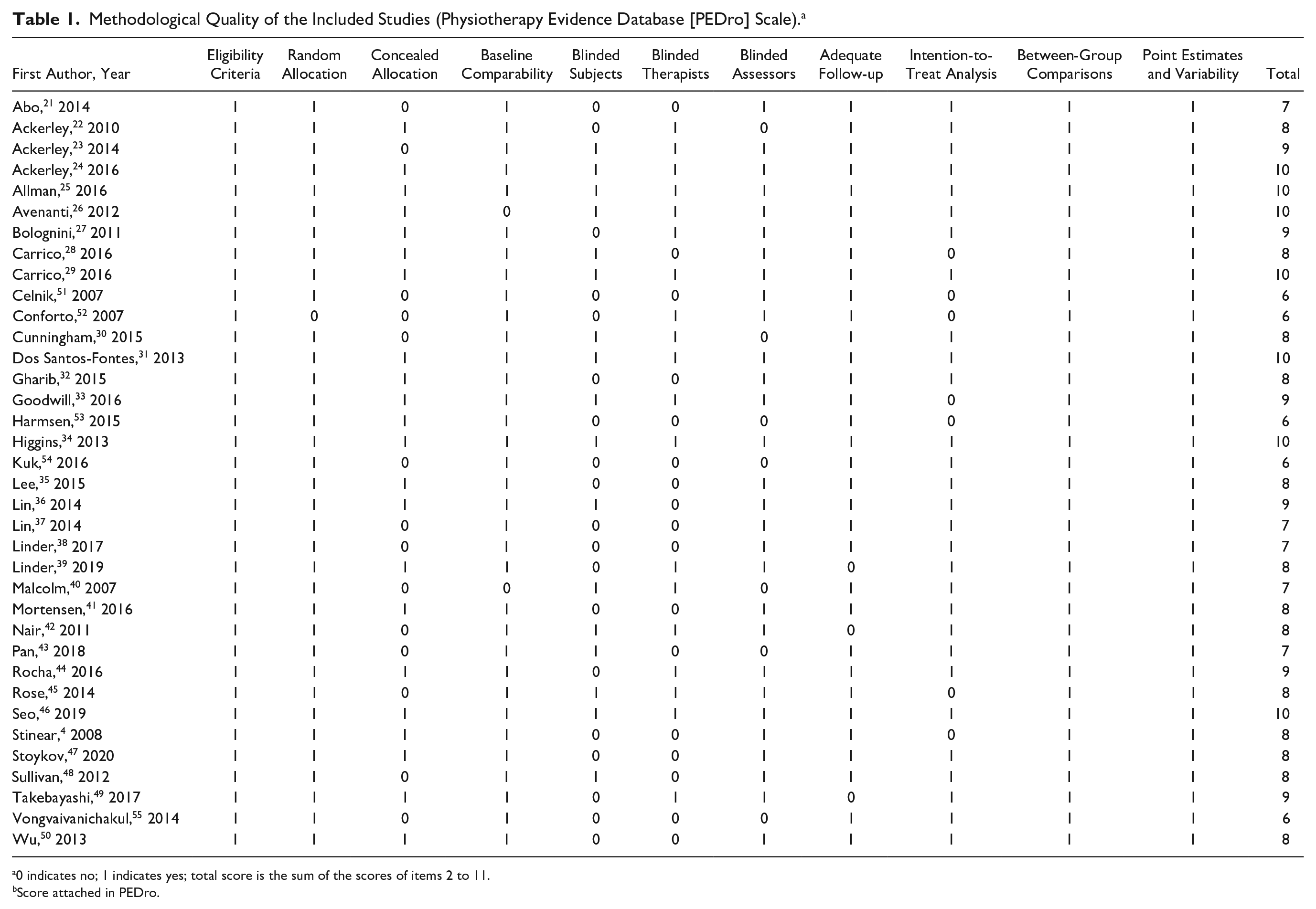
0 indicates no; 1 indicates yes; total score is the sum of the scores of items 2 to 11.
Score attached in PEDro.
Description of Studies
A summary of the included studies is shown in Table 2. The studies included a total of 814 participants, ranging from 9 to 66 participants each. The poststroke time ranged from 8.7 to 478 months. Regarding interventions, the studies varied in the number of sessions per week, session duration, protocol duration, and follow-up duration; the details are shown in Tables 3 and 4. The number of sessions per week ranged from 1 session per day22,23,25,27-29,41,51,53 to 5 sessions per week,24,26,35-37,50 with the total number ranging from 1 session22,23,41,51-53,55 to 40 sessions. 48 Thirteen studies only performed an evaluation after the intervention.21-23,32,36,37,42,48,49,52-55 Five studies had a follow-up of less than 1 month.33,41,46,47,51 Eight studies had follow-up periods of 1 month,4,27-30,34,44,45 4 studies assessed their participants after 3 months,24-26,35 4 studies performed 4 months of follow-up,31,38,39,43 and Malcolm et al 40 and Wu et al 50 had a 6-month follow-up period.
Demographic Characteristics of the Participants in the Selected Studies.
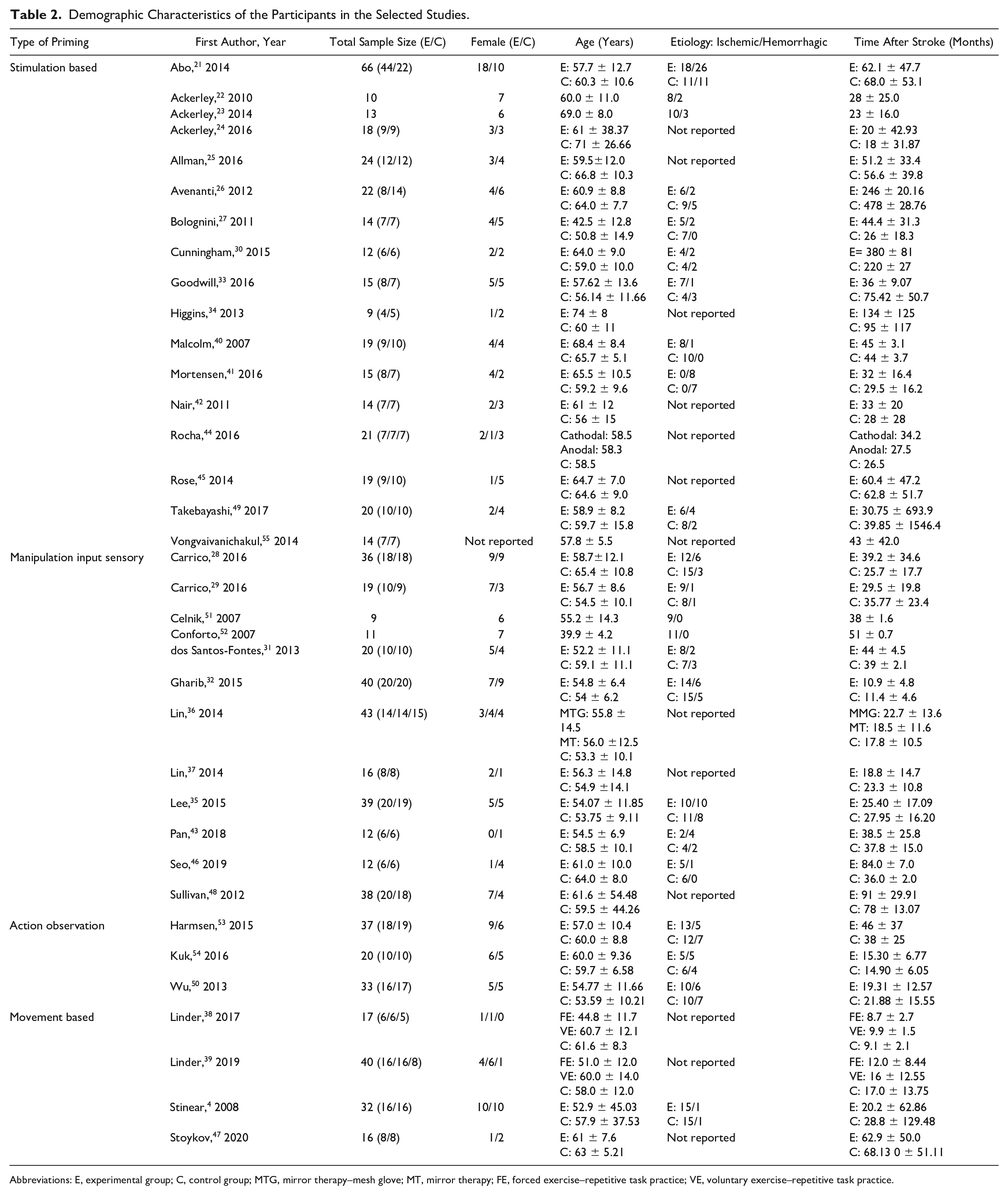
Abbreviations: E, experimental group; C, control group; MTG, mirror therapy–mesh glove; MT, mirror therapy; FE, forced exercise–repetitive task practice; VE, voluntary exercise–repetitive task practice.
Characteristics of Priming Interventions.
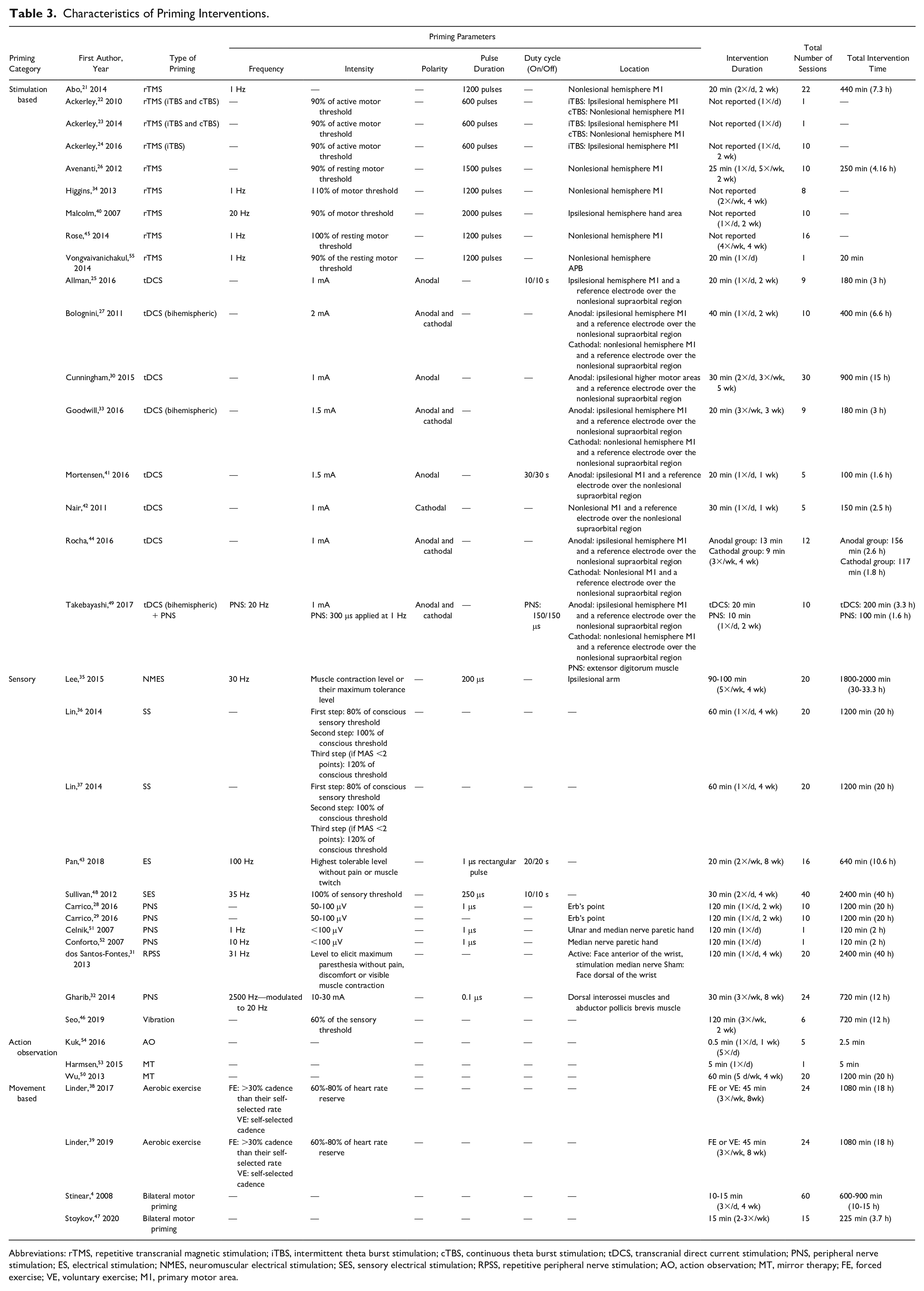
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; iTBS, intermittent theta burst stimulation; cTBS, continuous theta burst stimulation; tDCS, transcranial direct current stimulation; PNS, peripheral nerve stimulation; ES, electrical stimulation; NMES, neuromuscular electrical stimulation; SES, sensory electrical stimulation; RPSS, repetitive peripheral nerve stimulation; AO, action observation; MT, mirror therapy; FE, forced exercise; VE, voluntary exercise; M1, primary motor area.
Characteristics of Motor Training Intervention.
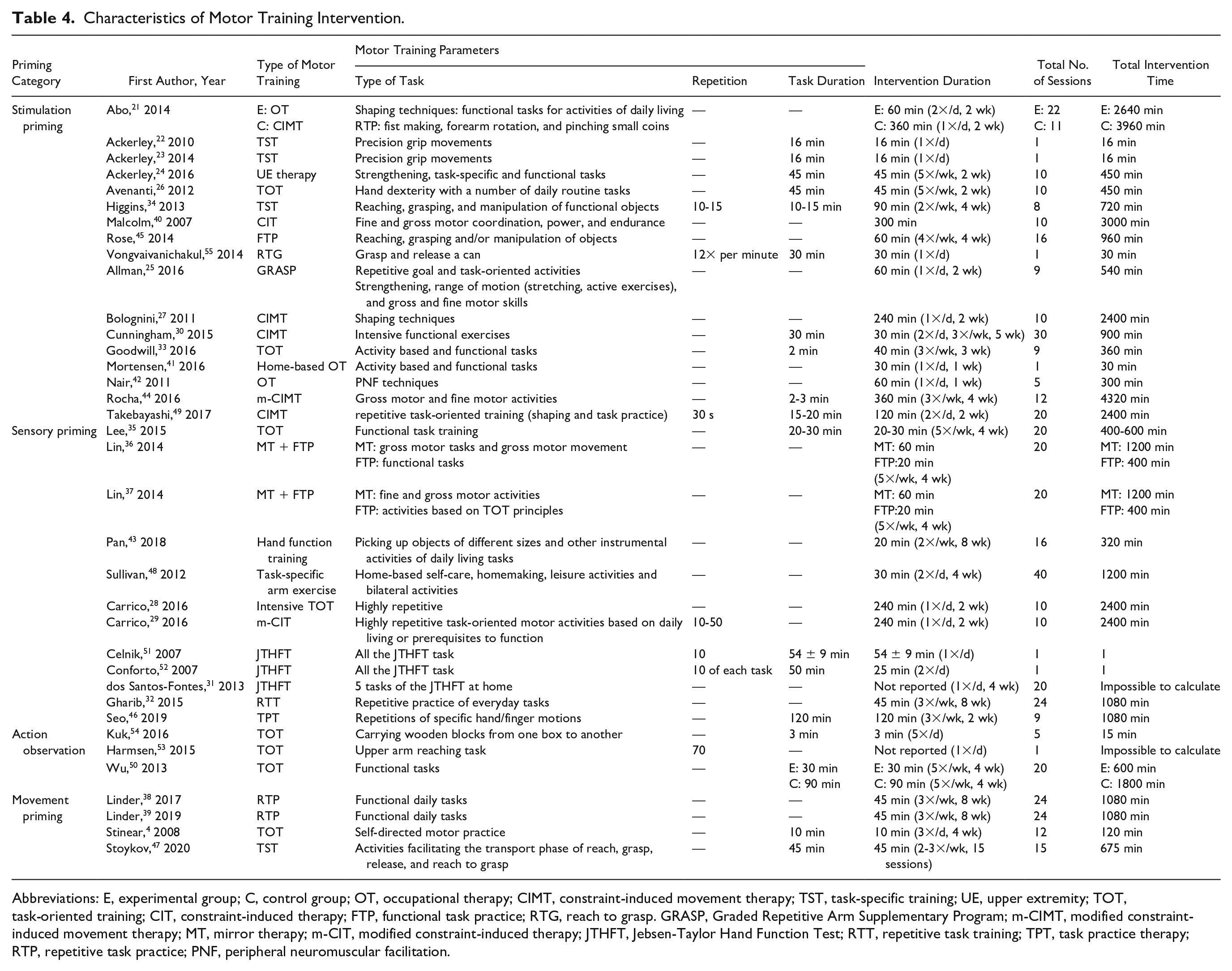
Abbreviations: E, experimental group; C, control group; OT, occupational therapy; CIMT, constraint-induced movement therapy; TST, task-specific training; UE, upper extremity; TOT, task-oriented training; CIT, constraint-induced therapy; FTP, functional task practice; RTG, reach to grasp. GRASP, Graded Repetitive Arm Supplementary Program; m-CIMT, modified constraint-induced movement therapy; MT, mirror therapy; m-CIT, modified constraint-induced therapy; JTHFT, Jebsen-Taylor Hand Function Test; RTT, repetitive task training; TPT, task practice therapy; RTP, repetitive task practice; PNF, peripheral neuromuscular facilitation.
Outcomes Measures
Twenty-two different outcome measures were identified as primary and secondary outcomes in the 36 included studies. Of the 22 outcome measures, 6 were classified as structure and body function, and 8 activity domains were classified according to the ICF model (Table 5). The most common assessment tools for the structure and body function domain were the UE-FMA, identified in 20 studies4,21,25,27-30,35,36,38,39,42-45,47-50 (55.5%) of the total 36 included. Considering the activity domain, the WMFT was the most used tool, which was reported in 12 studies21,25,28,29,34,35,38-40,45,46,55 (33%). However, some studies used the MAL27,30,34-36,40,44,45,48-50 and/or the ARAT22-25,28,29,37,45 to analyze the activity domain of the ICF.
Characteristics of the Assessments.
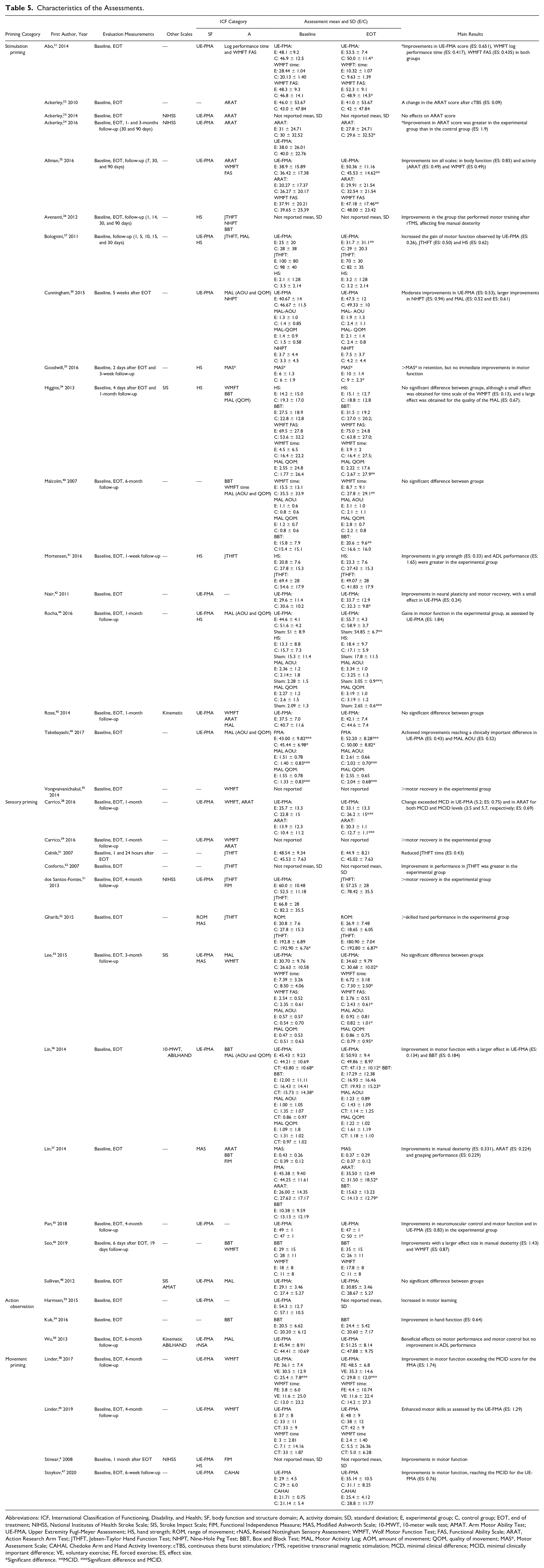
Abbreviations: ICF, International Classification of Functioning, Disability, and Health; SF, body function and structure domain; A, activity domain; SD, standard deviation; E, experimental group; C, control group; EOT, end of treatment; NIHSS, National Institutes of Health Stroke Scale; SIS, Stroke Impact Scale; FIM, Functional Independence Measure; MAS, Modified Ashworth Scale; 10-MWT, 10-meter walk test; AMAT. Arm Motor Ability Test; UE-FMA, Upper Extremity Fugl-Meyer Assessment; HS, hand strength; ROM, range of movement; rNAS, Revised Nottingham Sensory Assessment; WMFT, Wolf Motor Function Test; FAS, Functional Ability Scale; ARAT, Action Research Arm Test; JTHFT, Jebsen-Taylor Hand Function Test; NHPT, Nine-Hole Peg Test; BBT, Box and Block Test; MAL, Motor Activity Log; AOM, amount of movement; QOM, quality of movement; MAS*, Motor Assessment Scale; CAHAI, Chedoke Arm and Hand Activity Inventory; cTBS, continuous theta burst stimulation; rTMS, repetitive transcranial magnetic stimulation; MCD, minimal clinical difference; MCID, minimal clinically important difference; VE, voluntary exercise; FE, forced exercise; ES, effect size.
Significant difference. **MCID. ***Significant difference and MCID.
Priming Interventions Protocol
Regarding priming, studies varied in both the parameters and types of priming used (Table 3). Overall, 17 studies included stimulation priming, of which 9 were performed with rTMS21-24,26,34,40,45,55 and 8 with tDCS.25,27,30,33,41,42,44,49. Twelve studies performed sensory priming, of which 5 used PNS,28,29,32,51,52 2 used somatosensory stimulation (SS),36,37 and 5 studies used one of the following stimuli: electrical stimulation (ES) 43 , repetitive peripheral somatosensory stimulation (RPSS), 31 neuromuscular electrical stimulation (NMES), 35 vibration 46 or somatosensory electrical stimulation (SES). 48 Four studies used movement priming via aerobic exercise38,39 and bilateral motor priming.4,47 Three studies used action observation priming, of which one study employed action observation 54 and 2 studies employed mirror therapy.50,53
Task-Oriented Training Protocol
The TOT varied among the studies and included constraint-induced movement therapy (CIMT), mirror therapy and/or only functional exercises with a higher number of repetitions (Table 4). Among the studies, a variety of TOT parameters were used (Table 4).
Assessment of Risk of Bias
The methodological quality of the included studies was evaluated by analyzing each dimension in the risk of bias analysis. A summary and graph of the detailed analyses per study are provided in Supplementary Figures S1 and S2, respectively.
Allocation
The randomization of participants via random sequence generation was adequately reported in most studies; randomization was not performed in only 2 studies.42,52 Allocation concealment was not adequately reported by 12 studies.21,22,27,30,38-40,48,51,52,54,55
Blinding
Five studies did not report blinding with respect to the outcome assessment.40,43,53-55 Twenty-one studies did not report the blinding of participants and therapists.4,22,27,30,32,35-39,43,44,47-55
Incomplete outcome data
Most studies adequately reported how missing data points were handled. Two studies29,33 were inconsistent in reporting how the missing data were handled.
Selective reporting
Few studies4,23,26,52,53 reported the outcomes for all measurements taken.
Quantitative Analysis
Eighteen of the 36 included studies reported postintervention means and standard deviations for primary and secondary outcome measures. Nine studies included data for which postintervention means and standard deviations could be calculated.22,24,27,28,31,41,43,47,48 Some studies were not included in all meta-analyses for the following reasons: they presented values regarding differences between post- and pretreatment periods and did not present baseline data,4,29,45 they presented values in plots,23,26,27,52,55 the mean and standard deviation were not able to be calculated, 33 or they performed a kinematic analysis. 53 The meta-analysis is shown according to body function and activity ICF domains for stimulation, sensory priming and movement priming. An overview of the meta-analysis data postintervention is presented in Figures 2, 3, and 4. The results of the quality of the evidence are presented in Table 6.
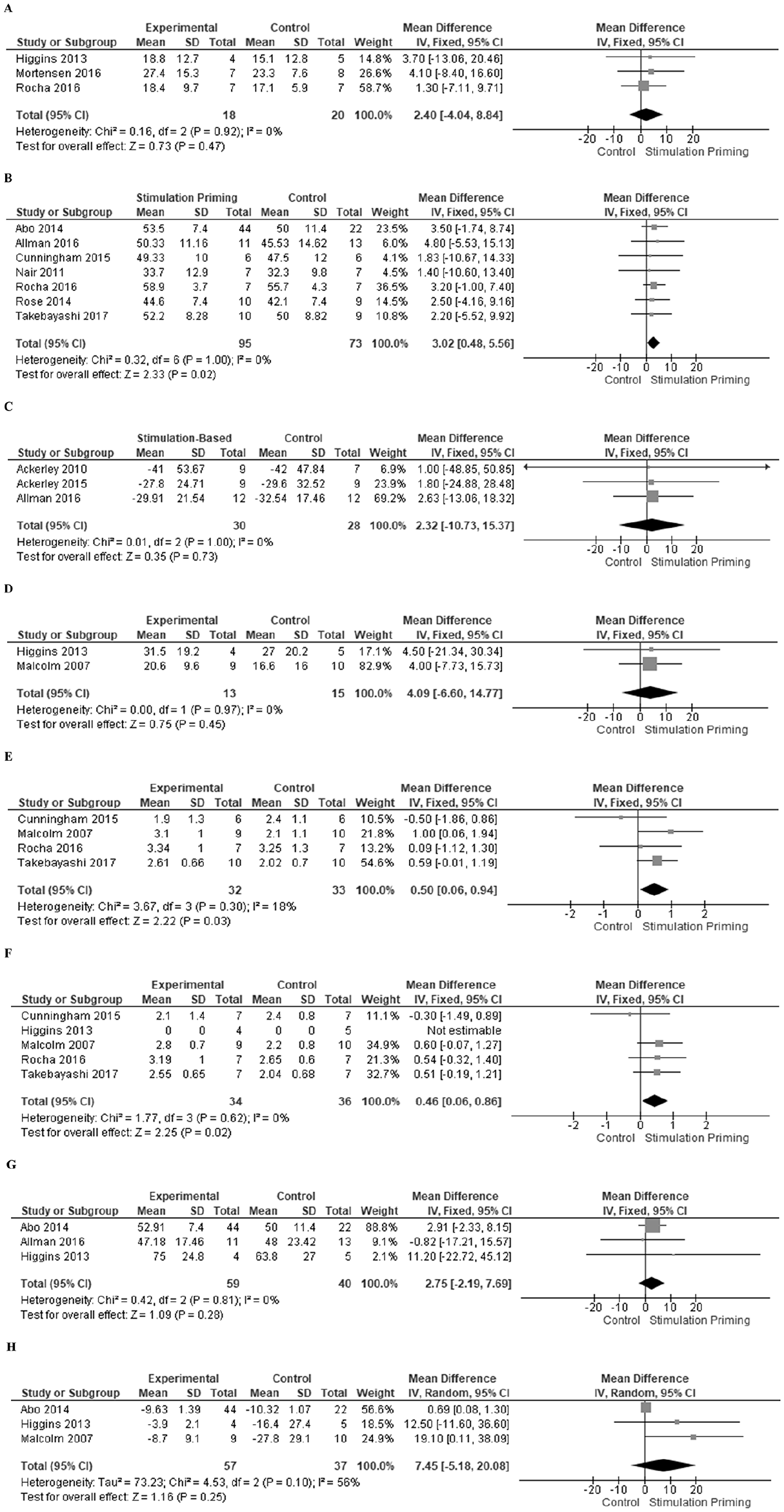
Forest plot of postintervention summary effect sizes of Stimulation Priming for Body Function and Activity ICF domain outcome measures: (A) Hand strength (kg/F); (B) Upper Extremity-Fugl Meyer Assessment; (C) Action Research Arm Test; (D) Box and Block Test (seconds); (E) Motor Activity Log–Amount of Use; (F) Motor Activity Log–Quality of Movement; (G) Wolf Motor Function Test–Functional Ability Scale; (H) Wolf Motor Function Test–time(seconds). ICF International Classification of Functioning, Disability, and Health.
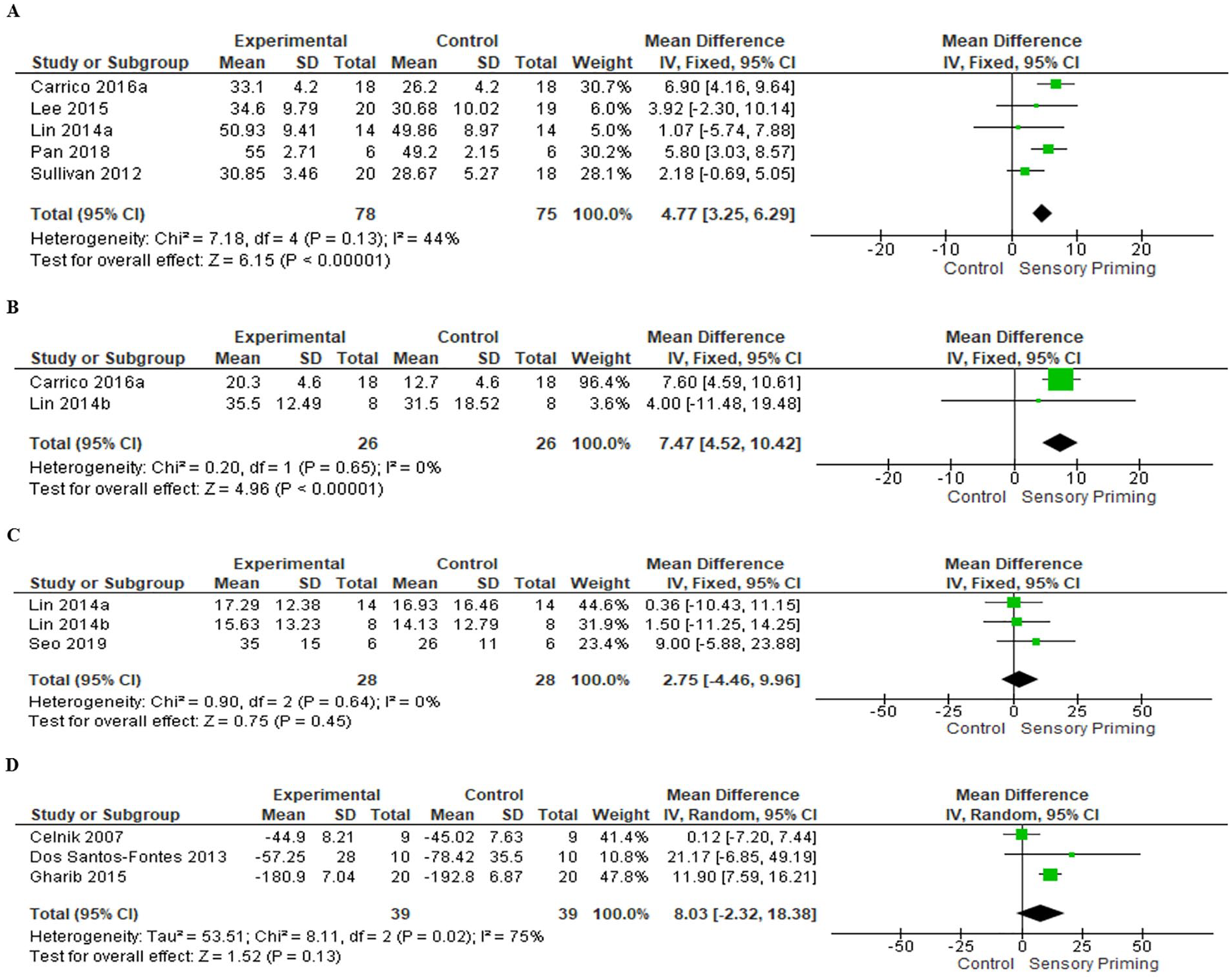
Forest plot of postintervention summary effect sizes of Sensory Priming for Body Function and Activity ICF domain outcome measures: (A) Upper Extremity-Fugl Meyer Assessment; (B) Action Research Arm Test; (C) Box and Block Test (seconds); (D) Jebsen-Taylor Hand Function Test (seconds). ICF, International Classification of Functioning, Disability and Health.
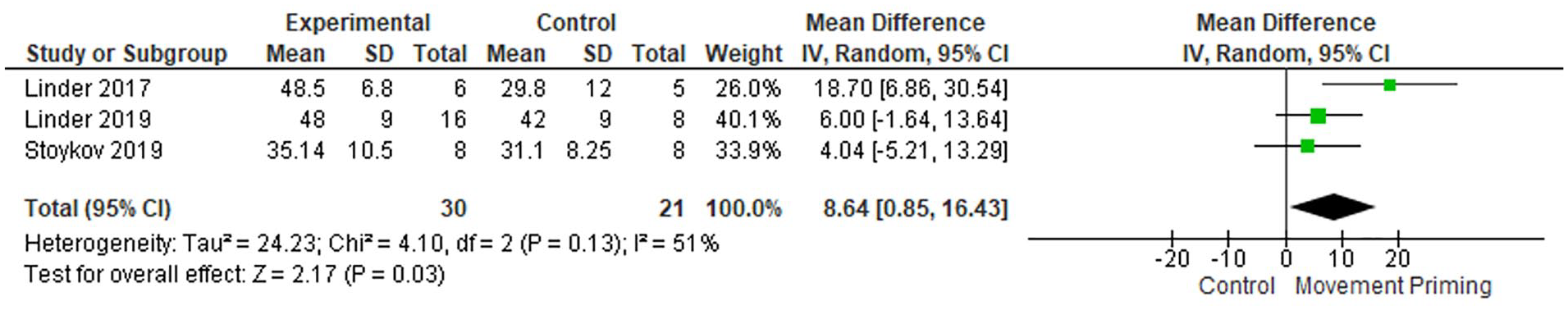
Forest plot of post-intervention summary effect sizes of Movement Priming for Body Function and Activity ICF domain outcome measures: Upper Extremity Fugl-Meyer Assessment. ICF, International Classification of Functioning, Disability and Health.
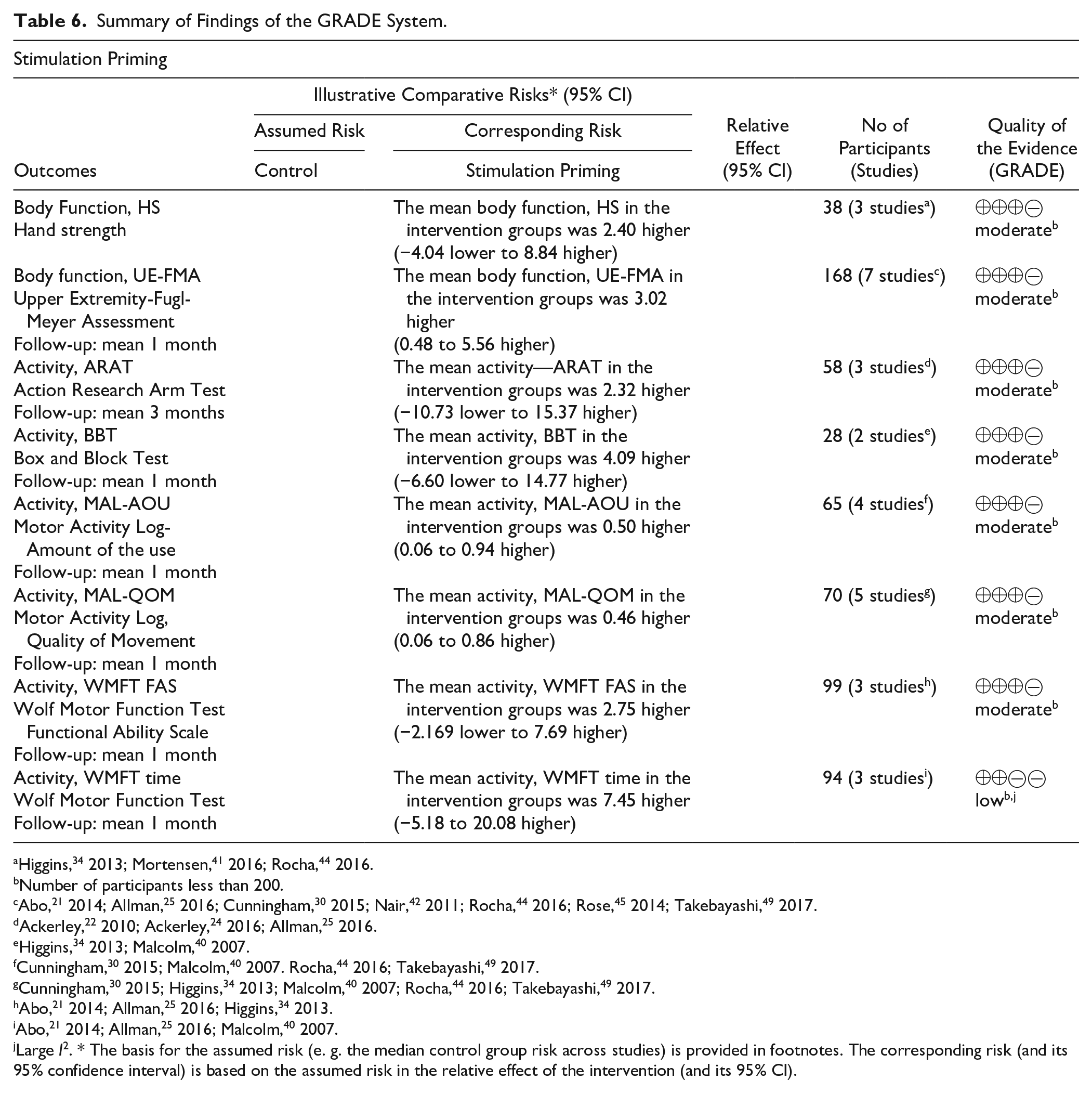
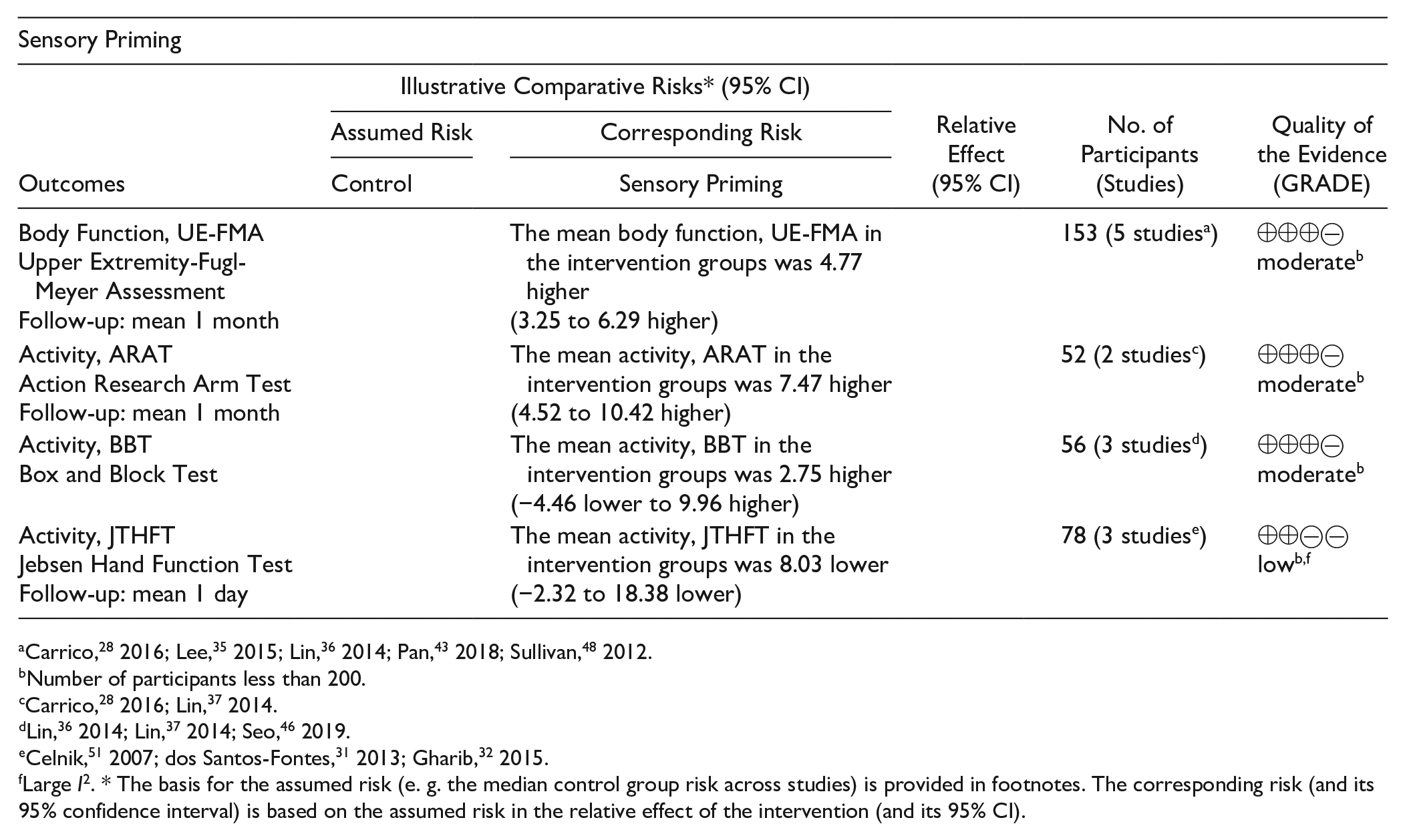
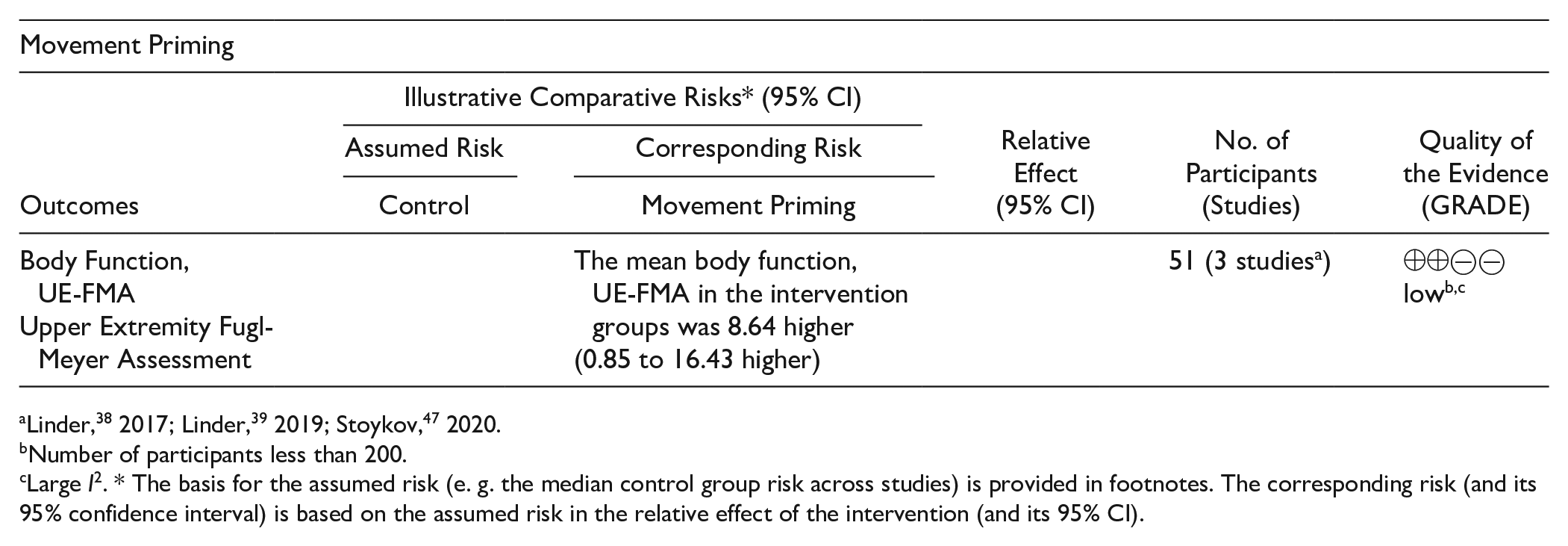
Summary of Findings of the GRADE System.
Number of participants less than 200.
Abo, 21 2014; Allman, 25 2016; Cunningham, 30 2015; Nair, 42 2011; Rocha, 44 2016; Rose, 45 2014; Takebayashi, 49 2017.
Large I2. * The basis for the assumed risk (e. g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the relative effect of the intervention (and its 95% CI).
Number of participants less than 200.
Large I2. * The basis for the assumed risk (e. g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the relative effect of the intervention (and its 95% CI).
Number of participants less than 200.
Large I2. * The basis for the assumed risk (e. g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the relative effect of the intervention (and its 95% CI).
Effects on Body Functions
For studies with stimulation priming, grip strength was assessed by three studies34,41,44 and presented low heterogeneity (I2 = 0%) and moderate-quality evidence with a nonsignificant effect size (38 patients, MD 2.40, 95% CI −4.04 to 8.84, Z = 0.73, P = .48; Figure 2A). Motor impairment assessed by the UE-FMA was investigated in 7 studies.21,25,27,30,42,44,45,49 The heterogeneity among these studies was low (I2 = 0%), with moderate-quality evidence and a significant effect size favoring priming (168 patients, MD 3.02, 95% CI 0.48 to 5.56, Z = 2.33, P = .02; Figure 2B). The studies21,44,45,49 with large weights in effect size all included CIMT.
For studies with sensory priming, 5 studies28,35,36,43,48 investigated participants by the UE-FMA, among which there was moderate-quality evidence and moderate heterogeneity (I2 = 44%). The analysis revealed significant effect sizes favoring priming (153 patients, MD 4.77, 95% CI 3.25 to 6.29, Z = 6.15, P < .0001; Figure 3A). One study 28 with a greater weight of the effect size employed PNS with longer motor training time, and the included individuals had severe impairment. Lin et al 36 reported the lowest efficacy in favor of priming, and the included individuals had mild impairment.
Among studies that employed movement priming, 3 studies38,39,47 investigated participants by the UE-FMA. There was low-quality evidence and substantial heterogeneity (I2 = 51%), with significant effect sizes favoring priming (51 patients, MD 8.64, 95% CI 0.85 to 16.43, Z = 2.17, P = .03; Figure 4). One study 39 with a greater weight of the effect size employed aerobic exercise with longer motor training time.
Effects on Activity
In studies with stimulation priming, the activity ICF domain was evaluated by the ARAT (3 studies22,24,25), BBT (2 studies34,40), MAL-AOU (4 studies30,40,44,49), MAL-QOM (5 studies30,34,40,44,49), WMFT FAS (3 studies21,25,34) and WMFT-time (4 studies21,25,34,40). Studies had low heterogeneity at I2 = 0% for ARAT, BBT, MAL-QOM, and WMFT FAS and I2 = 18% for MAL-AOM; otherwise, for WMFT-time corresponded to substantial heterogeneity (I²=56%) in the data. In the ARAT, moderate-quality evidence and a nonsignificant effect size were observed (28 patients, MD 2.32, 95% CI −10.73 to 15.37, Z = 0.35, P = .73; Figure 2C). For BBT, moderate-quality evidence and a nonsignificant effect size for priming were found (28 patients, MD 4.09, 95% CI −6.60 to 14.7, Z = 0.75, P = .45; Figure 2D). Significant effect sizes for MAL-AOM (65 patients, MD 0.50, 95% CI 0.06 to 0.94, Z = 2.22, P = .03; Figure 2E) and MAL-QOM (70 patients, MD 0.46, 95% CI 0.06 to 0.86, Z = 2.25, P = .03; Figure 2F) with moderate-quality evidence were demonstrated. The study by Malcolm et al 40 reported a greater effect size in both analyses, with more intensive motor training. A nonsignificant effect size for the WMFT FAS with moderate-quality evidence was observed (99 patients, MD 2.75, 95% CI −2.19 to 7.69, Z = 1.09, P = .28; Figure 2G). For the WMFT time, a nonsignificant effect size with low-quality evidence was observed (94 patients, MD 7.45, 95% CI −5.18 to 20.08, Z = 1.16, P = .25; Figure 2H).
For sensory priming, the ICF domains were evaluated by the ARAT (2 studies28,37), BBT (3 studies36,37,46), and JTHFT (3 studies31,32,51). The heterogeneity was low (I2 = 0%) for ARAT and BBT analyses and considerable (I2 = 75%) for JTHFT analyses. A significant effect size in favor of priming with moderate quality was observed for the ARAT (52 patients, MD 7.47, 95% CI 4.52 to 10.42, Z = 4.96, P < .0001; Figure 3B). For BBT, moderate-quality evidence and a nonsignificant effect size for priming were observed (56 patients, MD 2.75, 95% CI −4.46 to 9.96, Z = 0.75, P = 0.45; Figure 3C). Carrico et al 28 reported a greater effect size in the ARAT analysis, with more intensive priming and motor intervention. For JTHFT, we observed low-quality evidence and a nonsignificant effect size for priming (78 patients, MD 8.03, 95% CI −2.32 to 18.38, Z = 1.52, P = .13; Figure 3D).
Discussion
This systematic review and meta-analysis suggest that priming associated with TOT may be a promising adjuvant for rehabilitation after hemiparetic chronic poststroke. Stimulation priming combined with TOT provided moderate-quality evidence of improved motor limb outcomes with a significant effect on UE-FMA (body function) and MAL (activity). Regarding sensory priming, significant effects on UE-FMA (body function) and ARAT (activity) with moderate-quality evidence were observed. For movement priming, significant effects on body function (UE-FMA) with low-quality evidence were observed. Although important effects of the association of the different types of priming with TOT were observed, it must be noted that small sample sizes and heterogeneous outcome measures, intervention parameters, and dosages may influence the quality of evidence, which limits data extrapolation.
Outcomes Measures
For the outcome measures, most studies obtained moderate effect sizes, highlighting the importance of priming. However, although the use of one type of measurement was frequently observed (55% of studies used UE-FMA), heterogeneity and inconsistency in measurements among studies impaired the pooling of data and, consequently, the subsequent data analysis. For example, the BBT and the ARAT assess motor performance; however, the BBT focuses on gross dexterity, while the ARAT focuses on gross gripping function. Thus, caution should be taken in data interpretation. Other factors may have also influenced the outcomes, such as the time since stroke onset, sample size and type of intervention, which can influence the choice of the outcome measures in the studies. 20 Thus, these results should be interpreted with caution.
Priming Interventions Protocol
Stimulation Priming
The meta-analysis revealed moderate-quality evidence for all analyses, except for WMFTtime, and a significant effect on the activity domain measured by MAL (AOU and QOM) and body function quantified by UE-FMA. On the other hand, nonsignificant effects and moderate-quality evidence were identified for the ARAT, BBT, grip strength and WMFT (FAS and time). The main types of stimulation priming involved rTMS and tDCS.
Differences between tDCS and rTMS interventions are related to mechanisms of action, with TMS acting as a neurostimulator and tDCS acting as a neuromodulator. Moreover, TMS presents better spatial and temporal resolution with better established protocols. On the other hand, tDCS is easier to use and to apply simultaneously with behavioral tasks. Despite their differences, both were described to induce long-term after-effects (months) on cortical excitability, which may translate into behavioral impacts. Moreover, tDCS and rTMS promote motor recovery, particularly when combined with suitable behavioral interventions. 56
Previous studies demonstrated that the application of rTMS prior to or during a conventional physical therapy protocol resulted in no difference. However, the association of tDCS with rehabilitation therapies increased the efficacy of both techniques and contributed to a greater benefit in the rehabilitation of the UE. 57 Stimulation priming through rTMS and tDCS may induce a short-term and long-term therapeutic effect on motor functional recovery in the injured UE of stroke patients 58 and should influence motor recovery using the low or high frequency generated by rTMS. 58
In the studies that used tDCS as stimulation priming, the anodal polarity had a greater effect than the cathodal polarity of tDCS on increasing motor function, as evaluated by the FMA in chronic stroke patients. Corroborating these findings, Tedesco Triccas et al 59 also demonstrated that anodal tDCS applied over the ipsilesional hemisphere provided greater gains than motor training. According to the literature, the balance of excitability between hemispheres is associated with better recovery. However, after stroke, the ipsilesional hemisphere presents decreased excitability, while the contralesional hemisphere presents increased excitability 2 . Thus, these findings of the effects of tDCS polarity could be related to the anodal polarity promoting neural excitability of the ipsilesional hemisphere but the cathodal polarity promoting an inhibitory effect. 57
Although significant effects were observed on the activity of the ICF domain assessed by MAL, no effects were observed for other measures, such as the ARAT, BBT, and WMFT. A possible reason for the nonsignificant effect is the variety of parameter protocols adopted across individual studies, as previously described.2,57 According to Malcolm et al, 40 possible reasons for not achieving adjuvant effects are as follows: (1) transient effects on rTMS-induced cortical networks did not establish effective changes through learning; (2) the dose or intensity of rTMS was insufficient; (3) stimulation locus or the area of stimulation was not adequate; and (4) the effect of adjuvant therapy did not result in behavioral changes.
A possible explanation for the nonsignificant effect could be related to the inclusion criteria that were used to select the patients, which were based on clinical measures of motor impairment. However, according to the literature, clinical measures alone make it difficult to accurately predict recovery and motor outcomes. Researchers have suggested that neurophysiological (motor-evoked potentials after stimulation of the primary motor cortex) and neuroimaging (cortical pathway integrity and activity) biomarkers may more accurately predict recovery and motor outcomes after stroke. 60 Thus, patients in whom the intervention was ineffective may have been included, which could lead to a difference between the experimental and control groups.
Sensory Priming
The meta-analysis showed that sensory priming had a significant effect on body function, with low-quality evidence evaluated by the UE-FMA. However, the activity ICF domain evaluated by the BBT showed a nonsignificant effect and moderate-quality evidence, the ARAT showed a significant effect and moderate-quality evidence, and the JTHFT showed a significant effect and low-quality evidence. A possible reason for the lack of an observed effect when considering the BBT may be that this type of assessment evaluates a specific skill, that is, manual dexterity, while the ARAT evaluates motor performance. In addition, the most commonly used UE treatment describes evidence regarding improvements in arm motor function, not specifically manual dexterity. 61
Electrical stimulation through surface electrodes acts on motor neurons, inducing depolarization of lower motor neurons and promoting muscle contractions in the muscles that have intact motor innervation. 62 Electrical stimulation is generally used in the wrist muscles, which may be one reason why no improvement was obtained in activities of daily living among individuals with impaired elbow extension. 62 A systematic review of randomized clinical trials reported low- to moderate-quality evidence suggesting that somatosensory stimulation does not improve motor involvement of the hand or participation in the activity. However, the available literature may be insufficient to confirm this effect. 63
The mechanisms by which peripheral electrical stimulation acts on the cortical motor area are still unclear, but when used alone or as an adjuvant strategy, such stimulation could facilitate the recovery of motor function in stroke patients. 64 Pan et al 43 showed that peripheral electrical stimulation administered prior to each hand function training session for 8 weeks had the potential to improve neuromuscular control and hand function in chronic stroke individuals. Neuromuscular electrical stimulation as a motor learning tool reduces UE motor impairment and increases the quality of life after stroke when applied alone or in association with motor training. 65 Studies with PNS showed effects in favor of priming.
Movement Priming
One study that met our criteria employed movement priming for UE in individuals after chronic stroke.
3
Physical activity has a positive effect on brain function, health,
66
and cognition because of alterations in molecular signaling pathways that act directly on the central nervous system.CNS
10
Therefore, most previous studies have demonstrated that exercise can improve skill learning in young healthy individuals
67
and cognition and motor function in poststroke individuals.
68
High-intensity aerobic exercise performed immediately after motor training improves skill retention,
67
and the same exercise performed immediately before motor task practice is thought to facilitate learning.
10
In addition, the case study by Valkenborghs et al
69
showed an improvement in UE motor function in the subacute and chronic stages of stroke when aerobic exercise was combined with cycle ergometers with task
Action Observation Priming
Action observation priming promotes activation of the mirror-neuron system by the patient observing motor task performance, either on a video or via a live demonstration. After observing for a period of time, the individuals may or may not be asked to perform the same action. According to the literature, this practice may induce neural plasticity by promoting activation of the damaged motor circuits and providing access to multiple brain areas, thus facilitating motor relearning. Action observation has been applied alone or in conjunction with other therapies to stimulate motor relearning. 70 However, the present review could not clarify the effects of the association of this type of priming with TOT, since the included studies employed different types of action observation priming, such as video observation 54 and mirror therapy50,53, leading to heterogeneity of the outcome measures.
Task-Oriented Training Protocol
The included studies used different TOT protocols, such as functional daily tasks, CIMT, and shaping techniques. Most studies differed in the number of repetitions, sets, sessions, types of functional task and duration of intervention. Poor methodological quality and low treatment doses and durations may negatively affect the estimated effects. 71
One of the main interventions based on the principles of specific task training is CIMT. 72 The clinical improvement observed may be supported by previous studies that demonstrated that CIMT increases the size of the motor cortical maps of the paretic upper extremity 73 and recruits new pathways to the injured cerebral hemisphere. Thus, these studies present level A evidence that CIMT results in an increase in use and an improvement of motor function of the paretic side 72 associated with the mechanisms of neural plasticity. 73 Additionally, one of the pillars of CIMT is shaping, which should include intensive, repetitive TOT. 74 According to the literature, a linear relationship between the number of repetitions and the recovery of motor function has been observed. 75 Tasks should be repeated at least 300 times a day for 2 weeks, without causing pain or fatigue, to obtain significant improvements in motor function and in the use of the affected UE after stroke, 75 and factors such as intensity, duration, appropriate use of training and motivation directly influence the success of interventions. 16
A significant benefit of high-intensity and high-dose exercise has been observed. 76 In this systematic review and meta-analysis, the studies that employed higher doses reported effects in favor of the priming intervention and TOT. In addition to the evidence that TOT leads to improved UE motor function, there is a need for additional robust studies to determine the optimal protocol parameters.
Study Limitations
The results presented in this study require further investigation for several reasons. First, the effects of priming associated with TOT were limited to body function and activity because no study evaluated other domains of ICF; it should be noted that the included studies might not have published all the details of their intervention and individuals. Second, most of the studies did not assess other ICF domains as a primary or secondary outcome. Third, the results of this meta-analysis are limited to specific types of priming, such as stimulation and sensory priming, due to the reduced number of studies using action and observation and movement priming. Last, the variety of priming and TOT protocols had low power of their analyses.
Conclusion
Combining priming (stimulation, sensory, and movement priming) and TOT for the treatment of upper extremity chronic stroke patients may be a promising intervention strategy, as it confers gains in motor function according to the UE-FMA. Regarding the activity domain, the results are inconclusive, as some studies demonstrated gains favoring an association between priming (stimulation and sensory) and TOT while other studies reported similar effects among experimental and control interventions. For action observation priming, there is insufficient evidence to support its combined use. In this sense, future studies should include other outcome measures for body function and the activity domain and additional evaluations of action observation intervention as priming.
Supplemental Material
Supplementary_material – Supplemental material for The Effect of Priming on Outcomes of Task-Oriented Training for the Upper Extremity in Chronic Stroke: A Systematic Review and Meta-analysis
Supplemental material, Supplementary_material for The Effect of Priming on Outcomes of Task-Oriented Training for the Upper Extremity in Chronic Stroke: A Systematic Review and Meta-analysis by Erika Shirley Moreira da Silva, Gabriela Nagai Ocamoto, PT, Gabriela Lopes dos Santos-Maia, Roberta de Fátima Carreira Moreira Padovez, Claudia Trevisan, Marcos Amaral de Noronha, Natalia Duarte Pereira, Alexandra Borstad and Thiago Luiz Russo in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo; grants: 2017/25185-4 and 2017/22173-5) and was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–Brasil (CAPES)–Finance Code 001.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
