Abstract
Background
A single bout of aerobic exercise (AE) can produce changes in neurophysiological and behavioral measures in healthy individuals and those with stroke. However, the effects of AE-priming effects on neuroplasticity markers and behavioral measures are unclear.
Objectives
This systematic review aimed to examine the effects of AE on neuroplasticity measures, such as corticomotor excitability (CME), molecular markers, cortical activation, motor learning, and performance in stroke.
Methods
A literature search was performed in MEDLINE, CINAHL, Scopus, and PsycINFO databases. Randomized and non-randomized studies incorporating acute AE in stroke were selected. Two reviewers independently assessed the risk of bias and methodological rigor of the studies and extracted data on participant characteristics, exercise interventions, and neuroplasticity related outcomes. The quality of transcranial magnetic stimulation reported methods was assessed using a standardized checklist.
Results
A total of 16 studies were found suitable for inclusion. Our findings suggest mixed evidence for the effects of AE on CME, limited to no effects on intracortical inhibition and facilitation and some evidence for modulating brain derived neurotrophic factor levels, motor learning, and cortical activation. Exercise intensities in the moderate to vigorous range showed a trend towards better effects on neuroplasticity measures.
Conclusion
It appears that choosing a moderate to vigorous exercise paradigm for at least 20 to 30 minutes may induce changes in some neuroplasticity parameters in stroke. However, these findings necessitate prudent consideration as the studies were diverse and had moderate methodological quality. There is a need for a consensus on an exercise priming paradigm and for good-quality, larger controlled studies.
Keywords
Introduction
Stroke is a leading cause of adult-onset disability 1 and results in significant motor and cognitive impairments which persist well beyond the acute phase. 2 Currently available rehabilitative therapies focus on improving function by promoting dynamic experience-dependent neuroplasticity, as it is established that the post-stroke brain can undergo rewiring in response to training or therapy. 3 Although rehabilitation can improve motor and functional outcomes and quality of life, 4 recovery is often incomplete. 5 Motor recovery is partly dependent on underlying neural plasticity and cortical reorganization which are affected in stroke. 6 There is a growing need for novel interventions that can promote neural substrates of biological recovery for reducing motor impairment following stroke.
Aerobic exercise (AE) is a well-known rehabilitation adjunct for improving cardiovascular health in individuals with stroke. 7 In the past decade or so, AE is being explored for facilitating neuroplasticity, motor learning, and movement recovery. 8 Recent work has suggested that acute exercise can improve functional connectivity and neuroplasticity of the cortical regions involved in performance of a working memory task in older adults. 9 Recently, AE has been considered as a “priming”10 approach to create an optimal learning environment within the primary motor cortex (M1) and other cortical regions that are involved in movement. Motor priming refers to a process where a stimulus facilitates a subsequent change in movement behavior, that may occur in conjunction with an alteration in corticomotor excitability (CME) as measured by transcranial magnetic stimulation (TMS).10,11 Specifically, an increase in CME may be reflected as an increased excitability of the descending corticospinal projections from the M1 and/or a decrease in gamma-aminobutyric acid (GABA)-mediated intracortical inhibition and/or an increase in glutamate mediated intracortical facilitation (ICF) within the M1. 11 Exercise priming studies in healthy individuals have consistently shown a decrease in short interval intracortical inhibition (SICI) for a non-exercised upper extremity muscle. 12 A decrease in intracortical inhibition can occur in parallel with motor learning, and is thought to be a precursor for long-term potentiation (LTP)-like effects within the M1.11,13 Studies have shown that acute AE can increase levels of circulating neurotrophins, such as brain derived neurotrophic factor (BDNF), 14 insulin-like growth factor (IGF-1), 15 and vascular endothelial growth factor (VEGF). 16 More recently, the secretion of BDNF in response to activity or exercise has emerged as a critical component for regulating motor learning and formation of motor memories.17,18 Peripheral serum or plasma-based neurotrophin levels are however considered proxy markers as they may not reflect cerebral contributions to circulating levels. 19 In addition to upregulating neurotrophins, acute AE is also thought to modulate catecholamine levels of dopamine, nor-epinephrine, glutamate, GABA, and lactate.20 -24 Several lines of evidence show that acute exercise when performed immediately before or after a motor task, can improve performance on skilled motor tasks25 -27 and facilitate motor learning and retention.26,28 Changes in TMS measures, neurotrophin and catecholamine levels are thought to render the cortical environment within the M1 more responsive to motor learning. 29 Taken together, acute AE mediated changes via different neurophysiological mechanisms are thought to prime the motor system and make it more responsive to LTP-like synaptic plasticity which underlies behavioral therapy and motor learning.30,31
Considering the above, acute AE can be used as a potential priming tool to enhance the neuroplasticity of the motor systems in conjunction with rehabilitation modalities in clinical populations such as stroke. This type of exercise could create a therapeutic window to facilitate neuroplasticity, 11 and behavioral interventions targeting motor relearning could be incorporated during this window. Since the effects of exercise could occur in a non-exercised limb, it is thought that CME within the ipsilesional M1 could be modulated by exercising less affected muscles in individuals post stroke.32,33 However, several knowledge gaps remain. The bulk of the research investigating the neurophysiological effects of AE priming is in healthy adults and it is not completely known if these findings are translatable to individuals with stroke. The neurophysiological responses to AE in stroke may be affected by factors such as reduced physical activity and fitness levels, an altered physiological response to exercise and an inability to reach the target exercise intensity. Over the last decade, AE priming is being increasingly explored in stroke. However, the findings are inconsistent and are derived from studies with small sample sizes. Moreover, studies in stroke are limited to few outcomes, so it is difficult to precisely identify the neurophysiological associations between exercise induced change in systemic and molecular metrics (i.e., CME, neurotrophins, and functional connectivity) with change in motor behavior and learning. It is also unknown if exercise can normalize the interhemispheric imbalance in CME, which is commonly observed post-stroke. 34 Given that the neural effects of acute AE have not been summarized comprehensively, we aimed to synthesize the evidence pertaining to exercise priming and its effects on multiple neuroplasticity related outcomes in stroke.
Methods
Search Strategy and Selection Criteria
This systematic review is based on a search that was conducted in May’21 and updated in March’22 and included the following databases: MEDLINE/PubMed, CINAHL, Scopus, and PsycINFO with no restrictions on publication dates. The keywords included a combination of terms related to “aerobic exercise,” “stroke,” “neuroplasticity,” “brain derived neurotrophic factor,” “BDNF,” “motor learning,” “corticomotor excitability,” “motor evoked potential (MEP),” “non-invasive brain stimulation,” “cortical activation,” and related MeSH terms (see Table 1 for search strategy). The review protocol was registered in PROSPERO (CRD42021249342).
Search Strategy.

Search strings used in MEDLINE and replicated in other databases.
Experimental studies were eligible for inclusion if they included (i) adults with a diagnosis of stroke; (ii) a single session of AE (ie, dynamic exercise involving large muscle groups resulting in increase in heart rate, energy expenditure); (iii) any measure of neuroplasticity such as CME as obtained by TMS, and/or neurotrophins or molecular markers, such as BDNF, growth factors, catecholamines, hormones, and/or measures of motor learning and performance, such as speed, accuracy, time to perform task, reaction time, and/or neuroimaging measures such as cortical activation as measured by functional near-infrared spectroscopy (NIRS) or electroencephalography (EEG). Exclusion criteria included: (i) other neurological populations; (ii) studies that incorporated >1 session of AE; (iii) studies that incorporated other forms of exercise such as resistance training; (iv) outcomes not related to neuroplasticity (eg, heart rate variability); (v) non-English articles (no expertise for translation); (vi) conference proceedings; (vii) animal studies; and (viii) case reports.
Data Extraction and Analyses
The citations obtained by the database searches were imported into Rayyan. 35 Two review authors independently screened all titles and abstracts for including eligible articles. Figure 1 shows the PRISMA flow chart of the study. Data extraction for each study included participants’ characteristics, intervention characteristics, that is, exercise intensity, duration, type, outcome characteristics, that is, type of neuroplasticity assessment; and main findings pertaining to the outcome of interest. In addition, for studies that utilized molecular markers (BDNF, VEGF, IGF-1, and lactate), data was extracted with respect to sample preparation, storage, and assay characteristics. A descriptive analysis of the results was performed. A meta-analysis was not feasible as there was substantial heterogeneity between the studies in terms of study designs, control groups, exercise protocols, and outcome assessed (eg, difference in reporting metrics, MEP latency vs S–R curve metrics).
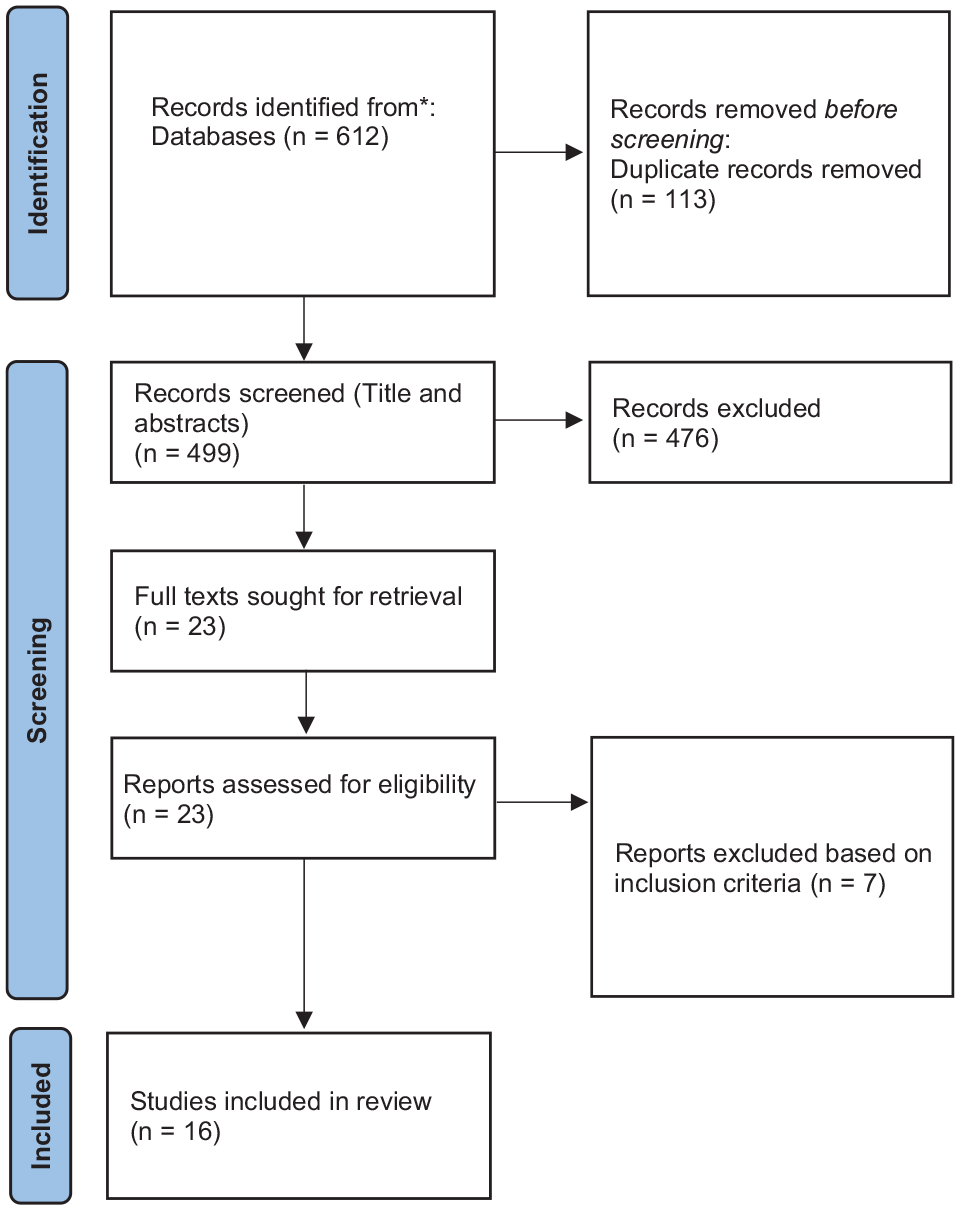
PRISMA flow diagram.
Effect sizes were computed for each study using the formula meanpost − meanpre/SDpre. The corresponding author was contacted for more information when the reported data was not sufficient for the purpose of this review. If data were still unavailable, effect sizes were not calculated, but the results were reported in the narrative synthesis. CME related measures included: MEP latency (duration from stimulus artifact to MEP onset); MEP amplitude (peak-to-peak amplitude in raw values (mV) or relative to peripheral M-wave amplitude (MEPmax) or expressed as a slope of stimulus–response (S–R) curve or area under the curve). Molecular markers (BDNF, IGF-1, VEGF, and cortisol) were quantified based on reported serum levels. Cortical activation was quantified based on reported oxyhemoglobin concentration levels (arbitrary units). Effect sizes were classified as 0.14 (small), 0.31 (medium), and 0.61 (large) based on guidelines for rehabilitation research. 36
Quality Assessment and Risk of Bias
The methodological quality for randomized and non-randomized studies was assessed using the modified Downs and Black checklist 37 which includes 27 questions. We used the following criteria for rating quality: 24 to 28 (excellent), 19 to 23 (good), 14 to 18 (moderate), and <14 (poor). 38
The TMS quality checklist was used to assess methodological quality of TMS based studies.39,40 The total scores for reported and controlled items were expressed as a percentage of the maximum possible points. 40 Scores greater than 75% were considered as high quality, 50 to 75% as moderate quality, and lower than 50% as poor quality. 40 The revised Cochrane risk of bias tool for randomized trials 2.0 41 (Figures 2 and 3) was used to assess risk of bias for randomized parallel-group and cross-over studies by the reviewers independently.
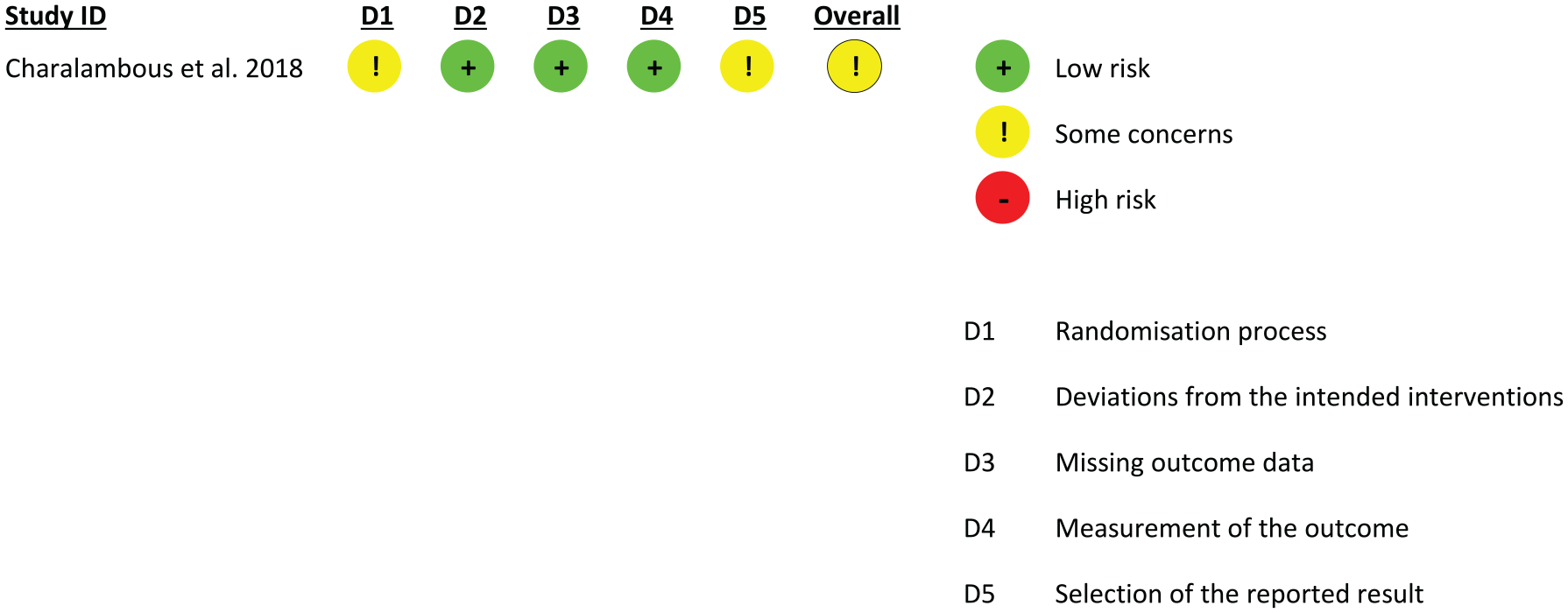
Cochrane risk of bias 2 for randomized controlled trials.
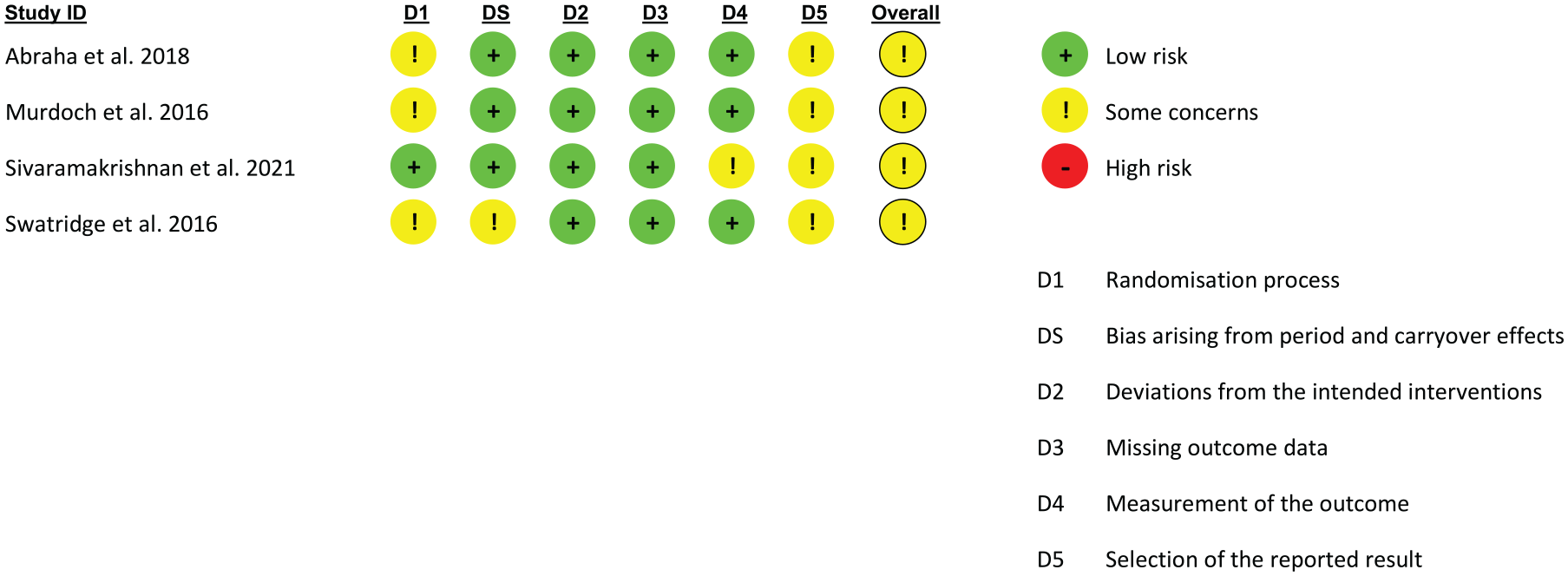
Cochrane risk of bias 2 for randomized cross-over trials.
Results
Search Results
Our search yielded a total of 612 articles (Figure 1). After removing 113 duplicates, we identified 499 articles for title and abstract screening. We removed 476 articles and screened the remaining 23 articles’ full texts. From these 7 articles were excluded due to either wrong population/outcome/intervention or study duration. We finally included 16 articles in the systematic review based on the selection criteria.
Methodological Quality and Risk of Bias
Downs and Black Scores
The overall quality assessment scores for each study are shown in Table 2. Supplemental Table S1 in the electronic format provides the breakdown of scores. The Downs and Black scores ranged from 15 to 22 [mean (SD) = 18.28 (1.5)] with most studies rated as having moderate quality. Overall, the authors did not control for factors relating to external validity, blinding, randomization, concealed allocation, and power.
Characteristics of the Included Studies.
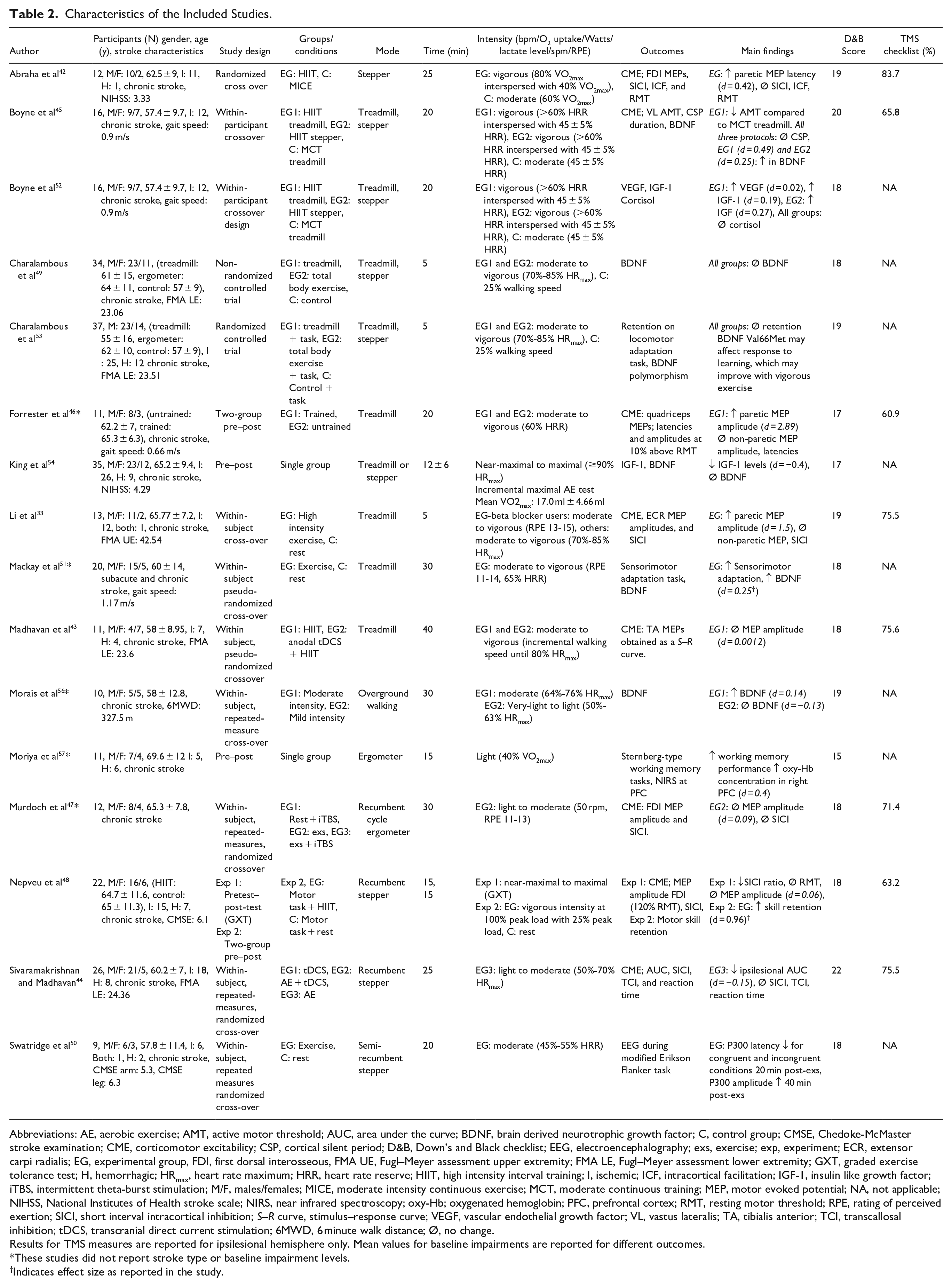
Abbreviations: AE, aerobic exercise; AMT, active motor threshold; AUC, area under the curve; BDNF, brain derived neurotrophic growth factor; C, control group; CMSE, Chedoke-McMaster stroke examination; CME, corticomotor excitability; CSP, cortical silent period; D&B, Down’s and Black checklist; EEG, electroencephalography; exs, exercise; exp, experiment; ECR, extensor carpi radialis; EG, experimental group, FDI, first dorsal interosseous, FMA UE, Fugl–Meyer assessment upper extremity; FMA LE, Fugl–Meyer assessment lower extremity; GXT, graded exercise tolerance test; H, hemorrhagic; HRmax, heart rate maximum; HRR, heart rate reserve; HIIT, high intensity interval training; I, ischemic; ICF, intracortical facilitation; IGF-1, insulin like growth factor; iTBS, intermittent theta-burst stimulation; M/F, males/females; MICE, moderate intensity continuous exercise; MCT, moderate continuous training; MEP, motor evoked potential; NA, not applicable; NIHSS, National Institutes of Health stroke scale; NIRS, near infrared spectroscopy; oxy-Hb; oxygenated hemoglobin; PFC, prefrontal cortex; RMT, resting motor threshold; RPE, rating of perceived exertion; SICI, short interval intracortical inhibition; S–R curve, stimulus–response curve; VEGF, vascular endothelial growth factor; VL, vastus lateralis; TA, tibialis anterior; TCI, transcallosal inhibition; tDCS, transcranial direct current stimulation; 6MWD, 6 minute walk distance; Ø, no change.
Results for TMS measures are reported for ipsilesional hemisphere only. Mean values for baseline impairments are reported for different outcomes.
These studies did not report stroke type or baseline impairment levels.
Indicates effect size as reported in the study.
TMS Quality Checklist
The TMS quality checklist was used for 8 studies (Table 2 and Supplemental Table S2) and total scores ranged from 60.95% to 83.6% [mean (SD) = 71.4% (7.6)]. Four studies were of high quality33,42 -44 and 4 studies were of moderate quality.45 -48 Most of the studies scored well on the TMS-related methodological factors but there was limited information on participant factors such as handedness/footedness, prescribed medication, use of central nervous system acting drugs and co-existing medical conditions which could affect CME.
Risk of Bias
The RoB 2.0 tool was used for 1 randomized controlled trial 49 and 4 randomized cross-over trials42,44,47,50 respectively (Figures 2 and 3). Together, overall RoB for all studies was rated as “some concerns” with respect to domains relating to randomization and concealed allocation (n = 4), period and carryover effects (n = 1), measurement of the outcome (n = 1), and reporting (n = 5). Most studies did not report if participants were blinded to the intervention and outcome assessors were blinded to the allocation. Finally, the studies did not have a pre-specified analysis plan raising some concerns about selective reporting of results.
Study Characteristics
A detailed summary of the individual studies is provided in Table 2. Briefly, almost all studies recruited individuals with chronic stroke [total sample size: 245 subjects (67% males), age range: 55-69.6 years] while 1 study 51 recruited individuals with both subacute and chronic stroke. The studies utilized pre–post, within-subject repeated measures crossover, randomized controlled, and randomized crossover designs. The experiments evaluated the effects of a single AE session, or compared AE at different intensities or to rest, or compared AE with NIBS such as intermittent theta burst stimulation or transcranial direct current stimulation or compared AE with motor tasks. Details from the AE-only conditions were included from studies that combined AE with other interventions such as NIBS. Two groups of authors45,49,52,53 reported data from the same cohort of participants who underwent the same intervention in 2 different studies. Since the reported outcomes were different for these studies, they were considered as separate studies with respect to the neurophysiological effects.
Exercise Protocol
A graded exercise test (GXT) until exhaustion was performed to measure maximal oxygen consumption either prior to the intervention42,45 or as an intervention on its own.48,54 One study incorporated a submaximal test on a recumbent stepper to determine the rate of work prior to the intervention. 50 Therefore, 5 studies incorporated either maximal or sub-maximal cardiopulmonary exercise tests.42,45,48,50,54
Since exercise intensities were reported differently, we incorporated the ACSM guidelines 55 as follows: very light exercise (<57% age predicted heart rate maximum (HRmax)/<37% VO2max), light exercise (57%-63% HRmax/37%-45% VO2max), moderate exercise (64%-76% HRmax/46%-63% VO2max), vigorous exercise (77%-95% HRmax/64%-90% VO2max), and near-maximal to maximal intensity exercise (≥96% HRmax/≥91% VO2max). Therefore, 2 studies used near-maximal to maximal intensity exercise, that is, GXT,48,54 3 studies used vigorous exercise,42,45,48 5 studies used moderate to vigorous exercise,33,43,46,51,53 4 studies used moderate exercise,42,45,50,56 2 studies used light to moderate exercise,44,47 and 2 studies used very light or light exercise.56,57 Six studies used a bike/ergometer or recumbent stepper,42,44,47,48,50,57 4 studies used a treadmill,33,43,46,51 and 3 studies used both modes.45,49,54 One study incorporated overground walking. 56 The total duration of AE ranged from 5 to 40 minutes.
Exercise-Induced Response in Neuroplasticity Outcomes
With respect to neuroplasticity outcomes, 8 studies evaluated the effects of exercise on CME measures, 5 studies evaluated BDNF levels, 2 studies evaluated IGF-1 levels, 1 study evaluated VEGF levels, 1 study evaluated cortisol levels, 4 studies investigated motor learning/performance and retention effects, 1 study measured cortical activation with NIRS, and 1 study assessed event related potentials with EEG.
Single and Paired Pulse TMS Measures
Eight studies investigated the effects of an acute bout of exercise on CME where MEPs were recorded from either an upper limb33,42,47,48 or lower limb43 -46 M1 representation.
MEP related parameters: The results between studies are mixed and inconsistent. One study reported a significant increase in ipsilesional MEP latency for a non-exercised muscle only in the vigorous intensity group when compared to moderate intensity 42 [medium effect size, d = 0.42]. However, another study did not observe any differences in ipsilesional MEP latency between 2 groups of subjects who underwent a single AE bout. 46 These groups included trained individuals in a long term treadmill exercise program and untrained individuals. 46 This study, however reported a significant increase in ipsilesional MEP amplitude for an exercised muscle [large effect size, d = 2.89 (trained group)]. 46 Another study reported a significant increase in MEP amplitude in a non-exercised muscle with AE [large effect size, d = 1.5 (exercise group)], 33 3 studies reported no change in MEP amplitude or S–R curve slope43,47,48 and another study reported a significant decrease in ipsilesional area under the curve for an exercised muscle following AE [d = −0.15]. 44 A consistent feature of all studies was that no change in contralesional CME was found despite differences in exercise intensities.33,42 -44,46,48
Motor threshold: One study reported no effects of AE on ipsilesional resting motor threshold, 42 however, another reported a decrease in ipsilesional active motor threshold for an exercised muscle following vigorous exercise when compared to moderate exercise. 45
Silent period: Only 1 study evaluated silent period duration and reported no change with vigorous or moderate intensity exercise. 45
Intracortical circuits: Five studies evaluated the effects of exercise on SICI and 4 reported no effects following vigorous/moderate/low intensity exercise.33,42,44,47 Another study reported a non-significant decrease in SICI in the ipsilesional hemisphere, but a significant increase in the interhemispheric SICI ratio following a graded exercise test. 48 Only 1 study assessed the effects of AE on ICF and reported no effects. 42
Interhemispheric inhibition: Only 1 study evaluated transcallosal inhibition following moderate intensity exercise and reported no change. 44
Molecular Markers
Table 3 describes the procedures that were used for measuring molecular markers.
Description of Procedures for Testing Molecular Markers.
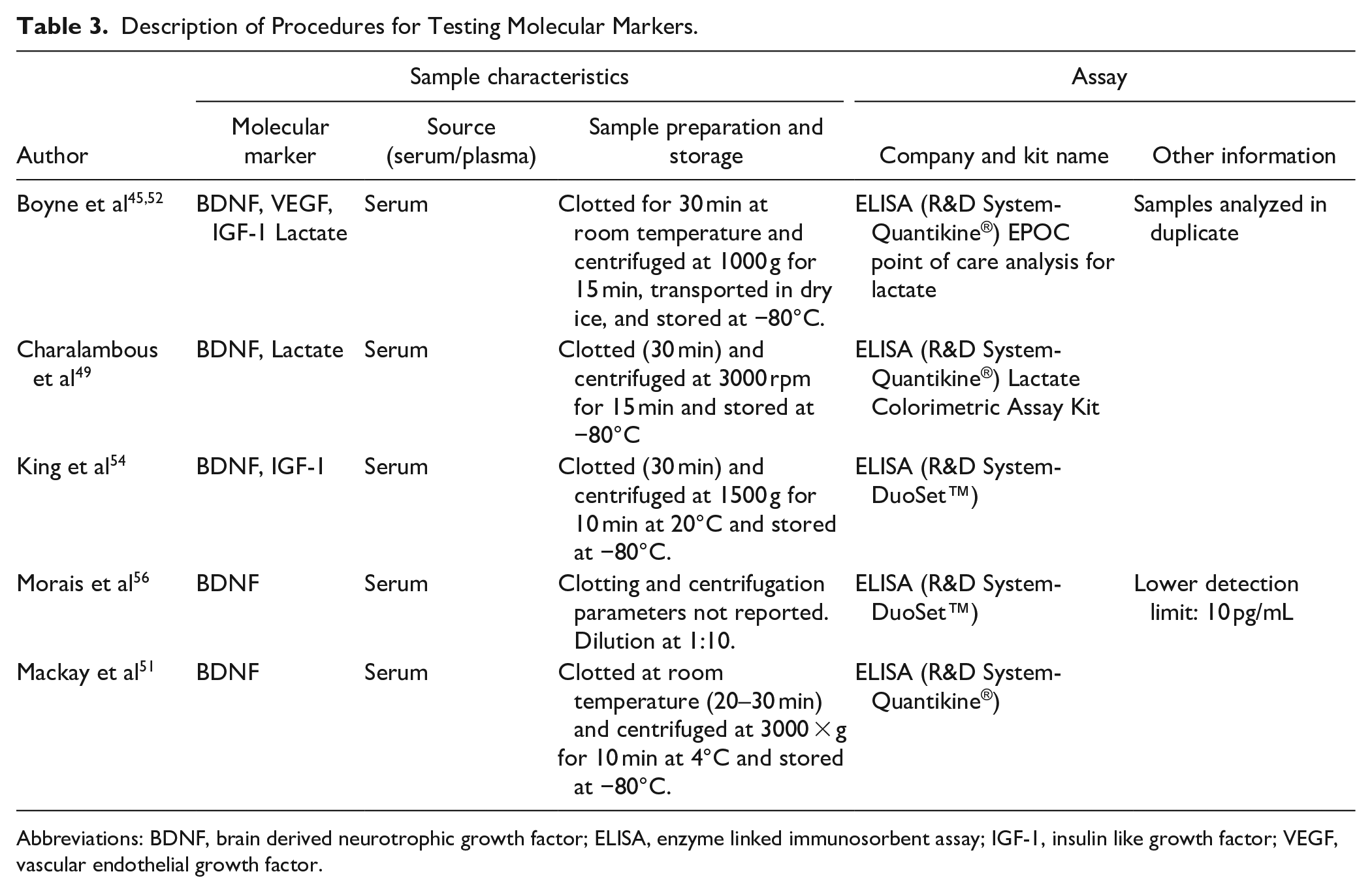
Abbreviations: BDNF, brain derived neurotrophic growth factor; ELISA, enzyme linked immunosorbent assay; IGF-1, insulin like growth factor; VEGF, vascular endothelial growth factor.
BDNF: One study reported an increase in BDNF levels following 20 minutes of vigorous treadmill exercise (medium effect size, d = 0.49) and stepping exercise (medium effect size, d = 0.25), but no change following moderate intensity treadmill exercise. 45 Another study reported no change in BDNF following 5 minutes of moderate-vigorous treadmill or stepping exercise compared to low-intensity walking on a treadmill (control). 53 King et al 54 reported no change in BDNF following a graded exercise test. Mackay et al 51 reported an increase in BDNF following moderate-vigorous exercise (medium effect size, d = 0.25) when compared to rest. Morais et al 56 reported an increase in BDNF with moderate intensity (small effect size, d = 0.14) and not light intensity overground walking exercise.
IGF-1: Boyne et al 52 report an increase in IGF-1 with vigorous treadmill (medium effect size, d = 0.19) and vigorous stepping (medium effect size, d = 0.27) compared to moderate intensity treadmill exercise. Another study reported a decrease in IGF-1 (d = −0.4) following an incremental GXT. 54
VEGF: Boyne et al 52 report an increase in VEGF following vigorous treadmill (small effect size, d = 0.08) but not vigorous stepper exercise.
Cortisol: One study evaluated effects of AE on cortisol and found no change with vigorous treadmill/stepper exercise or moderate intensity exercise. 52
Motor Performance, Learning, and Retention
In 1 study, participants performed a split belt locomotor task either before moderate-vigorous exercise (stepper group) or after exercise (low intensity control and moderate-vigorous treadmill groups). 53 There was no change in 24-hour retention between all 3 groups. 53 Mackay et al 51 found that moderate-vigorous intensity treadmill exercise improved sensorimotor adaptation on an upper extremity task (medium effect size, d = 0.24) when compared to rest. Nepveu et al 48 found that 24-hour retention improved when an upper extremity hand grasping time-on-target motor task was administered before vigorous stepping exercise (large effect size, d = 0.96) but not rest. Another study found no change in simple and choice ankle reaction time following light-moderate intensity stepping exercise. 44
Cortical Activation
Moriya et al 57 reported that light intensity exercise increased oxygenation of the right prefrontal cortex as measured by NIRS (medium effect size, d = 0.4) during performance of a working memory task. Swatridge et al 50 found that moderate intensity exercise when compared to rest improved EEG correlates of attention (P300 amplitude) and speed of processing (P300 latency) for approximately 40 minutes following exercise.
Discussion
The findings from this review suggest that moderate to vigorous intensity acute AE for 20 to 30 minutes is likely to induce changes in some biological mediators of neuroplasticity and behavioral endpoints in individuals with stroke, however these findings need to be carefully interpreted. There is mixed evidence for the effects of AE on CME, limited to no effects on intracortical inhibition and facilitation (SICI and ICF), and some evidence for modulating BDNF levels, measures of motor learning and cortical activation. Most of the studies utilized small samples demonstrating low statistical power and moderate methodological quality. Thus, definitive conclusions regarding the extent of neural and behavioral effects obtained by AE cannot be drawn due to the lack of consistent data and the vast heterogeneity between studies. Our findings tentatively suggest that acute AE can potentially modulate neuroplasticity indices in stroke based on preliminary evidence from the included studies.
Exercise and TMS Measures
In this review, only 2 out of 8 studies reported positive findings with large effect sizes for increased MEP amplitude within the ipsilesional M1 after acute AE.33,46 Although these studies report significant effects, the samples were small, and the findings need to be replicated in larger studies to optimally characterize AE-related effects on MEP amplitude. Some studies also reported inhibitory effects of exercise on the ipsilesional M1 such as decreased CME 44 and increased MEP latency. 42 Studies that measured CME were of moderate to high quality as per the TMS checklist, however there was a high methodological variability between the TMS protocols. Most of the studies reported MEP parameters that were calculated from a single stimulus intensity and only 2 studies assessed CME with S–R curves.43,44 A S–R curve can capture the relationship between the size of responses and stimulus intensities, and the slope of the sigmoidal curve has been reported to correlate with glutamate levels, 58 suggesting that this measure may be more sensitive to capture acute exercise induced effects. One factor that may explain changes in neuroplasticity is the state of the target muscle during exercise (ie, involved in exercise vs rest). A recent systematic review found strong evidence to suggest no effects of AE-based priming on CME of a non-exercised muscle in healthy individuals. 12 However, studies included in this review reported facilitatory and inhibitory effects on CME for both exercised and non-exercised muscle representations. Perhaps other stroke-specific factors may underlie the differences in CME responses and need further characterization in future studies.
The effects of AE on intracortical circuits were assessed by 5 studies, and except for 1 study 48 which reported a significantly increased inter-hemispheric SICI ratio following a GXT, other studies did not report significant findings on SICI or ICF. These findings contrast with AE-based studies in healthy individuals where there is robust evidence to suggest that AE can modulate SICI. 12 A decrease in SICI with exercise is thought to reflect a modulation within the GABA-A receptors and GABA levels, which could serve as a precursor to practice dependent plasticity. 59 Intracortical inhibition is known to depend on the state of the target muscle (ie, rest vs active), 60 and controlling the level of pre-stimulus background electromyography (EMG) before and after an intervention may be more suitable to detect subtle changes in SICI. Three studies reported methods where pre-stimulus EMG amplitude was monitored for sub-threshold muscle activity,33,42,47 however no study reported comparisons for background EMG pre and post exercise. While individuals with stroke may show similar change in SICI, it is possible that current techniques for measuring muscle activation need better characterization to capture these changes.
A further consideration to explain these findings is stroke-specific severity as neurophysiological measures of CME, intracortical mechanisms and interhemispheric inhibition are expected to change with recovery and use or non-use of the affected limb. Many individuals with stroke, especially those with more severe lesions of the corticospinal tracts may not have a measurable MEP from the ipsilesional hemisphere. In this review, most of the included studies had small samples, and in some, the number of individuals with elicitable ipsilesional MEPs was even smaller. Findings from recent work on acute exercise in elderly adults may extend to those with stroke, as concomitant aging influences may affect the responses of the M1 to exercise. 61 Taken together, all these factors may have accounted for the mixed findings with respect to the directionality of AE induced effects on TMS.
Exercise and Molecular Markers
In this review, BDNF, IGF-1, and VEGF were measured across studies, however BDNF was more commonly investigated (n = 5). Although somewhat inconsistent, preliminary findings from this review suggest that serum BDNF levels could potentially increase with moderate to vigorous intensity exercise. Five of the included studies measured BDNF levels and while some reported positive findings45,51,56 with small to medium effect sizes, other studies did not report an effect. Studies that reported positive findings in neurotrophin levels resulted from either vigorous exercise or moderate to vigorous intensity exercise that was performed for 20 to 30 minutes. This finding aligns with other studies which recommend that vigorous intensity exercise at approximately 70% HRmax, 62 for a duration of ≥ 30 minutes may be required to show a change in BDNF levels. 63 In this review, there was an inconsistency in reporting the methods used for sample preparation and centrifugation which could explain some of the variability in the results. Methodological differences in BDNF sampling such as the time of blood withdrawal, centrifugation temperature and storage, skill of the tester in performing the enzyme linked immunosorbent assays (ELISA), and type of ELISA kits may introduce bias and affect comparisons across studies.
Another factor to consider is a genetic variation wherein individuals expressing the met allele on their BDNF gene (val66met) may show a 25% reduction in activity-dependent secretion of BDNF.64,65 It has been reported that this polymorphism may negatively affect post-stroke recovery and motor learning,66,67 however more studies are required to elucidate this relationship. Given that recent evidence suggests that 36 sessions of HIIT increases BDNF levels in individuals post-stroke, 68 it is plausible that long term vigorous exercise may improve motor relearning rates in those with the polymorphism. These findings are encouraging, and open new avenues for future studies with larger samples, and pre-allocation stratification of individuals based on the presence of the polymorphism.
Exercise and Motor Learning
Similar to findings related to BDNF, studies that incorporated moderate—vigorous AE (20-30 minutes) improved motor skill retention 48 and sensorimotor adaptation 51 with medium to large (0.24-0.96) effect sizes respectively. In both these studies, the tasks were upper extremity based, and the exercise conditions involved treadmill walking or stepping.48,51 Two studies assessed motor learning outcomes and reported no effects when an exercised extremity was involved either in a locomotor task 53 or an ankle reaction time task. 44 In one of these studies, vigorous AE was used for shorter duration ∼5 minutes. 53 Factors such as fatigue may have interfered with motor learning and performance in these studies. Furthermore, the type of motor task utilized, that is, split-belt treadmill walking versus upper limb time-on-target task 48 or reach adaptation 51 may affect motor learning and retention.69,70 Although vigorous AE was used in some studies, individuals with stroke may not produce blood lactate levels that correspond to vigorous exercise in healthy individuals, 53 suggesting that experimental protocols in this population may require greater fine-tuning.
Other Exercise Parameters
In recent years, post-stroke rehabilitation has focused on high-intensity interval training (HIIT) exercise as it incorporates periods of active recovery and can potentially prime the motor system by stimulating exercise induced release of BDNF. HIIT protocols may be better than moderate intensity protocols as they challenge the cardiovascular (ie, increased heart rate response) and metabolic system (ie, increased blood lactate post-exercise) to a greater extent and are likely to produce improvements in motor skill retention or other indices of neuroplasticity.71 -73 Three studies incorporated HIIT (ie, vigorous exercise) in this review, and preliminary findings suggest that a single session of HIIT can increase CME, improve motor learning and retention, and increase levels of circulating BDNF. While HIIT is promising in exercise priming studies, it is important to note that even in the high intensity interval regimens, stroke survivors may not reach their ventilatory thresholds, due to disease severity, age, and other comorbidities.
From a practical perspective, an exercise intervention seems superior to a maximal GXT for eliciting change in neurotrophin levels and/or cortical activation. Other moderating variables such as aerobic fitness levels and frequency of exercise may also affect neuroplasticity responses. Chronic exercise may be more beneficial for producing sustained effects on cortical mechanisms, BDNF levels, and related outcomes. This area of research is still in its infancy, and limited studies have evaluated the effects of long-term AE on neural measures.
Important Recommendations for Exercise Priming Studies in Stroke
Methodological Considerations
Similar to acute exercise and cognition-based studies, priming studies in stroke can incorporate a within-subjects repeated measures crossover design to minimize variability. Including an appropriately designed control condition that is matched in duration for both experimental and control groups would facilitate better comparisons. Other factors to improve methodological rigor include controlling for external validity, performing a priori power calculations, reducing selection bias (controlling for randomization and concealed allocation) and controlling for blinding.
Exercise Variables
Testing: While there is still some debate on the most suitable parameter for exercise prescription, recent research suggests that exercise intensity should be prescribed based on individual lactate levels rather than HR-based or maximal oxygen consumption methods for minimizing inter-individual variability. 74 However, measuring lactate threshold can be cumbersome, and researchers could use rating of perceived exertion (RPE) which is a good proxy for blood lactate levels, 75 and more suitable for individuals on beta-blockers.
Exercise intensity and other factors: It appears that at least 20 minutes of vigorous interval-based exercise on a treadmill or stepper may be suitable for stroke survivors to offset fatigue with active recovery periods, and possibly induce changes in neurophysiological parameters and improve ipsilesional excitability. 45 The magnitude of priming related effects may be related to exercise intensity, which suggests that future studies need to elucidate a dose-response relationship. Only 9 studies reported the actual exercise intensities of their participants in terms of HRmax or VO2max or RPE.33,42,44,45,47,48,50,51,54 One study reported the average intensity at target intensity as a % of maximum intensity. 53 Due to the inconsistency in reporting, it is imperative that studies report individual responses to exercise as a % of HRmax or VO2max for identifying if the participants truly exercised at the target zones.
Outcomes
Future exercise studies can aim to include at least 1 outcome each from neurophysiological, molecular, and behavioral domains to identify the profile of AE-induced effects. Some parameters to consider while measuring CME include controlling the state of the target muscle that is being trained or tested, using S–R curves over single-intensity measures and adhering to standardized checklists. Molecular markers such as serum BDNF and VEGF have shown to be more responsive to vigorous exercise bouts. 52 It is plausible that simple, explicit motor learning tasks such as time-on-target and reaction time tasks that do not involve an exercised extremity are more sensitive to exercise-mediated changes in learning and retention.
Limitations
A meta-analysis was not statistically feasible given the vast heterogeneity in study designs, control groups, and different methods to measure outcomes. Most of the studies had small samples which may have been underpowered to show statistical significance. Moreover, the studies reported either change scores from baseline, or P values and post-intervention parameters but not the corresponding 95% confidence intervals. Therefore, we could not obtain outcome data and variance estimates for computing effect sizes. Nevertheless, this review provides a starting point for developing future studies in identifying neurophysiological mechanisms underlying AE and their causal relationship with behavioral parameters.
Conclusion
There is encouraging evidence for incorporating moderate to vigorous intensity exercise paradigms for inducing possible neuroplasticity effects, however they warrant cautious interpretation. It appears that the choice of AE parameters and neuroplasticity outcomes may affect efficacy, and conclusions are limited by few high-quality randomized studies that evaluate different combinations of AE on a range of outcomes. Larger, controlled studies are needed to evaluate the effects of exercise priming before it translates into a post-stroke rehabilitation adjunct.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221146996 – Supplemental material for A Systematic Review on the Effects of Acute Aerobic Exercise on Neurophysiological, Molecular, and Behavioral Measures in Chronic Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683221146996 for A Systematic Review on the Effects of Acute Aerobic Exercise on Neurophysiological, Molecular, and Behavioral Measures in Chronic Stroke by Anjali Sivaramakrishnan and Sandeep K. Subramanian in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to acknowledge Rebecca Ajtai, liaison librarian to the UT Health Sciences Center San Antonio for executing the search across all databases, Nicholas Dennis and Christopher Hughen for their assistance with data extraction, Charalambos C. Charalambous and Marc Roig for their thoughtful inputs on this systematic review. We would also like to thank the authors of the included studies for sharing their data to be included in this review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
