Abstract
Background
People with Parkinson’s disease (PD) are known to have motor learning difficulties. Although numerous studies have demonstrated that a single bout of aerobic exercise (AEX) can facilitate motor learning in non-disabled adults, the same beneficial effect in PD is unknown. Furthermore, associated neuroplastic changes have not been investigated.
Objectives
This study aimed to determine whether a single bout of aerobic exercise (AEX) can facilitate motor sequence learning in people with PD and to investigate the associated neurophysiological changes.
Methods
Thirty individuals with PD were recruited and randomized into the exercise group (PD + AEX) and non-exercise group (PD − AEX). At the first visit, corticomotor excitability was assessed using transcranial magnetic stimulation (TMS). All participants then performed a serial reaction time task (SRTT) followed by 20 minutes of moderately-high intensity aerobic exercise (AEX) for the PD + AEX group or rest for the PD − AEX group. The SRTT and TMS were reevaluated at 3 time points: immediately after aerobic exercise (AEX) or rest, on the second day after practice (D2), and a week after practice (D7).
Results
Both groups showed improvement throughout practice. At retention, the PD + AEX group showed improved SRTT performance on D7 compared to D2 (
Conclusion
A single session of aerobic exercise (AEX) could enhance motor sequence learning and induce neuroplastic changes. Clinicians can consider providing aerobic exercise (AEX) after motor task training for people with PD.
Clinical registration:
NCT04189887 (ClinicalTrials.gov).
Keywords
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder that is characterized by motor dysfunctions as well as motor learning difficulties. A review by Marinelli et al 1 summarized that patients with PD are specifically impaired in learning motor tasks that involve the usage of attentional resources and cognitive strategies, coinciding with the frontostriatal disruption found in this disease. Moreover, individuals with PD often demonstrate poorer retention performance (ie, poorer learning) compared to age-matched non-disabled adults, suggesting an impaired “memory consolidation” process in this patient population. 1 Compromised motor learning would limit the ability to acquire new motor skills as well as relearn old skills related to daily activities.2,3 Deficits in learning would also limit the ability of individuals with PD to generalize what they learned in the clinic back to their home environment. 4 Hence, it is crucial for clinicians to seek for intervention approaches to enhance motor learning ability for people with PD in order to maximize the training benefits.
Aerobic exercise (AEX), in addition to sleep or non-invasive brain stimulation, is one of the most commonly used approaches to facilitate motor learning in non-disabled adults.5 -7 Extensive research conducted with healthy young adults has shown that performing aerobic exercise immediately after motor skill acquisition can enhance the process of memory consolidation and facilitate learning. 8 Aerobic exercise has been shown to increase blood circulation in fronto-parietal and hippocampal areas, which are known to be important brain regions for motor learning and memory. 9 Since people with PD are found to have problems with memory consolidation, it would be interesting to determine whether aerobic exercise can induce similar beneficial effects.
To date, only 2 studies have determined the effect of aerobic exercise on motor learning in people with PD. The first study conducted by Steib et al 10 required the participants to perform a single bout of moderate intensity (60%-70% VO2max) bicycling exercise before practicing a motor adaptation task—the dynamic balance stabilometer task. The results showed that 30 minutes of moderate-intensity aerobic exercise led to better motor performance at retention tested 1 day after practice, compared to the non-aerobic resting condition. The second study conducted by the same group of authors used the same motor task except that the aerobic exercise was performed immediately after skill learning. The results again showed that adding aerobic exercise after skill acquisition led to better retention, especially when tested at 1 week after practice. 11 Despite the positive results, 1 potential confounding factor of these 2 studies was that the aerobic exercise and the motor task being practiced both required the use of lower extremity muscles, which might be too fatiguing for people with PD. Furthermore, a motor adaptation task was used in these studies instead of a motor sequence task. A motor adaptation task is to test one’s ability to compensate for (or adapt to) environmental changes, while a motor sequence task is to determine the process of combining simple individual movement elements into a unitary sequence.12,13 Neuroimaging studies have shown that, compared with motor adaptation, motor sequence learning required greater involvement of the frontostriatal circuit, which is known to be specifically impaired in PD. 12 Thus, we were especially interested in determining whether aerobic exercise can overcome neural impairment and facilitate sequence learning in people with PD. Lastly, while research has provided ample evidence that aerobic exercise could improve motor learning behaviorally, no study has investigated the related neurophysiological changes along with behavioral improvement, especially in PD. Probing the associated neural mechanisms may enable researchers to better understand the potential benefits of aerobic exercise.
To bridge these knowledge gaps, this study aimed to investigate whether a single bout of aerobic exercise could enhance motor sequence learning in people with PD. Additionally, corticomotor excitability was assessed with transcranial magnetic stimulation (TMS) to determine potential neurophysiological changes after sequence learning and aerobic exercise.
Methods
Participants
Participants diagnosed with idiopathic PD were recruited for this study. The inclusion criteria were (1) able to follow the instructions to perform the tasks, (2) no neurological disorders other than PD, and (3) no history of surgery or injury of the upper extremities in the past 6 months. To ensure the safety of receiving TMS, participants were excluded if they had (1) deep brain stimulation or pacemaker implantation, (2) a self-history of seizure or a blood-relative with a history of epilepsy, and (3) unstable or uncontrolled medical conditions. The participants were required to sign an informed consent form approved by the Research Ethics Committee of National Taiwan University Hospital before participating the study. Eligible participants were then randomized into 1 of 2 groups: PD with aerobic exercise (PD + AEX) group or PD without aerobic exercise (PD − AEX) group. The group allocation of participants with PD was determined by drawing lots from a sealed envelope. This study conformed to the principles of the World Medical Association Declaration of Helsinki.
Study Procedure
This was a cross-sectional study in which the participants visited the laboratory 3 times. On the first day of visit, all participants first underwent TMS assessment of their corticomotor excitability after the baseline evaluation, which included Montreal Cognitive Assessment (MoCA) 14 and Part III of the MDS-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS III). 15 All assessment were performed by a researcher who was trained by the principal investigator. Following TMS, the participants practiced a motor sequence task—the serial reaction time task (SRTT). Immediately after the acquisition of SRTT, participants in the PD + AEX group performed aerobic exercise with a stationary bicycle for 20 minutes, while the PD − AEX group sat and read books or magazines for 20 minutes. After the 20-minute exercise or rest period, immediate retention of the SRTT and corticomotor excitability were reassessed.
All participants were asked to return to the laboratory on the second day and a week later for the short- and long-term retention tests of the SRTT. Additionally, corticomotor excitability was assessed with TMS at these 2 visits to investigate the neurophysiological changes associated with learning. All procedures were conducted during the “ON” medication state, approximately 1 to 1.5 hours after taking the medication. The study procedure is illustrated in Figure 1.
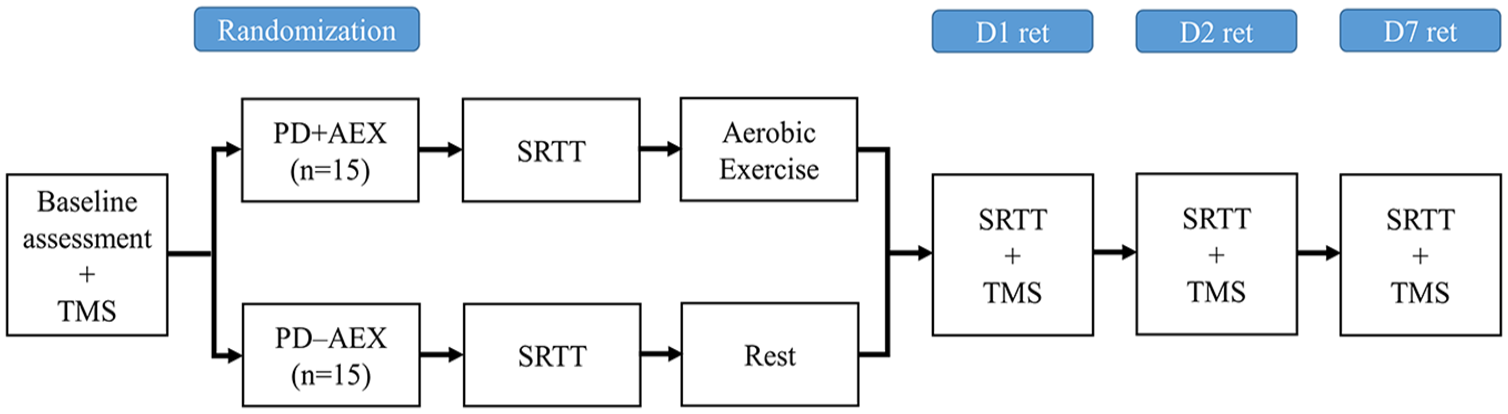
Experimental design of the study. Participants were randomized into the PD + AEX and PD − AEX groups after baseline demographic and transcranial magnetic stimulation (TMS) assessments. Immediately after practicing the serial reaction time task (SRTT), the PD + AEX group performed 20 minutes of aerobic exercise while the PD − AEX group rested. SRTT performance and TMS were reassessed on the first day after aerobic exercise or rest (D1 ret), a day after practice (D2 ret), and a week after practice (D7 ret).
Serial Reaction Time Task (SRTT)
The SRTT was used to evaluate the motor sequence learning ability. The design and implementation of the SRTT were similar to what have been reported in previous studies.16,17 To avoid the potential movement difficulty of the ring and little fingers, the participants were asked to place their index and middle fingers of both hands on the “C, V, B, N” keys of an enlarged keyboard. Four circles were presented on the computer screen, and the relative position of each circle corresponded to the specific key button on the keyboard; that is, the leftmost circle corresponded to the “C” key, the second left circle was the “V” key, the third circle “B” key, and the rightmost circle was the “N” key. In each trial, 1 of the 4 circles on the computer screen was filled with color while the other circles remained hollow, and the participants were required to press the corresponding key as accurately and as quickly as possible. Once the participants pressed a key, the next target appeared. Unknown to the participants, there was a 12-element target sequence (B-C-N-B-V-N-V-C-B-N-C-V) embedded within several random sequences (each also contained a 12-element random key press). During the acquisition period on the first day, the SRTT started with 6 random sequences followed by 84 target sequences and ended with another 6 random sequences. Adding the random sequences after the target sequences enabled researchers to contrast and determine whether the improved SRTT performance during acquisition was due to learning the sequence or learning the visuomotor association of the visual stimuli and the motor responses. 18 These 96 sequences were divided into 3 practice sessions, and the participants were given a 1-minute break between each session to reduce mental and physical fatigue.
Retention tests of the SRTT were conducted on the first day immediately after aerobic exercise or rest (D1), a day after practice (D2), and a week after practice (D7). During the retention tests, the subjects were required to perform 6 target sequences of the SRTT as accurately and as quickly as possible.
With the SRTT, the response time of each finger key press and the errors of the key presses were recorded. To further examine whether the participants retained what they had learned from the acquisition phase on the first day,
Aerobic Exercise Protocol
Participants in the PD + AEX group engaged in the aerobic exercise with a stationary bicycle after practicing the SRTT. A 5-minute warm-up, set at approximately 45% to 55% of the heart rate reserve (HRR), was first provided. Following the warm-up, the participants were required to perform 20 minutes of moderately high intensity cycling exercise with an exercise intensity set between 60% and 75% of HRR. 10 This exercise intensity was chosen based on earlier studies conducted with healthy young adults or people with PD.10,19 We did not raise the intensity higher than 75% HRR considering the fatigue and the potential autonomic dysfunctions in PD. Additionally, a recent meta-analysis suggested that exercise intensity has less of an impact on neuroplasticity in patients with neurological disorders. 9 Hence, a moderately high intensity was chosen in this study. A 5-minute cool-down period was provided after the 20-minute aerobic exercise. Throughout the exercise period, the heart rate and rating of perceived exertion (RPE)20 -23 were monitored every 5 minutes.
Measurement of Corticomotor Excitability
A TMS device (The Magstim Company Ltd., Whitland, UK) with a figure-of-eight coil was used to determine neurophysiological changes associated with sequence learning and exercise. The TMS outcomes of interest included resting and active motor evoke potentials (MEPs), cortical silent period (CSP), short intracortical inhibition and intracortical facilitation. These outcomes were chosen based on previous evidences related to neurophysiological changes after motor learning or PD.24,25 Active surface electromyographic electrodes were placed over the muscle belly of the first dorsal interosseous (FDI) of the more affected hand, and a ground electrode was placed over the styloid process of the ulna. The sampling rate of the EMG signals was set at a frequency of 12 500 Hz and a band-pass filter at 0 to 1000 Hz. The hotspot and resting motor threshold (RMT) of FDI were first determined. Under the single-pulse condition, stimuli were applied at an intensity of 120% RMT. Resting MEPs were determined with FDI at rest, while active MEPs and the CSP were recorded when FDI actively contracted around 10% to 15% of maximal voluntary contraction. Under the paired-pulse condition, the conditioning stimulus was set at 80% RMT and the testing stimulus at 120% RMT with an interstimulus interval set at 2 and 12 ms to measure short intracortical inhibition (SICI2ms) and intracortical facilitation (ICF12ms), respectively. 26
At each stimulation condition, 15 TMS pulses were applied and an average MEP value was calculated. The raw MEP values were recorded for the resting and active MEPs, while the MEP values of SICI2ms and ICF12ms were normalized to the resting MEP for further analysis. Across-day TMS measures were controlled by making sure the participants received TMS assessment at the same time of the day during “ON” medication, and assessed by the same researcher who had 2 years of experiences in performing TMS.
Sample Size Calculation
The sample size of this study was estimated from a study conducted by Duchesne et al 27 who investigated the effect of long-term aerobic training on SRTT in people with PD, and a study by Steib et al 10 who determined the effect of a single bout of aerobic exercise on improving learning a stabilometer task. The effect sizes of performance change after aerobic exercise were large (η2 = 0.136-0.251) in these 2 studies. With these effect sizes, the calculated sample size required for the current study was 8 to 12 participants per group. Considering a potential of 20% drop out rate, we decided to recruit 15 participants for each group.
Statistical Analysis
All acquired data were analyzed with SPSS 22.0 (Chicago, IL, USA). The Shapiro–Wilk test was used to determine the normality of the outcome measures. Baseline dichotomous data and continuous data were compared using the chi-square and independent
Results
SRTT Performance
Thirty participants with idiopathic PD were recruited into the study, and randomized into the PD + AEX group (n = 15) and the PD − AEX group (n = 15). Basic demographic data of the participants are provided in Table 1. The average error rate during acquisition was 3.11 ± 0.55% for the PD + AEX and 2.74 ± 0.47% for the PD − AEX group without significant group differences (
Baseline Demographic Data of the Participants.
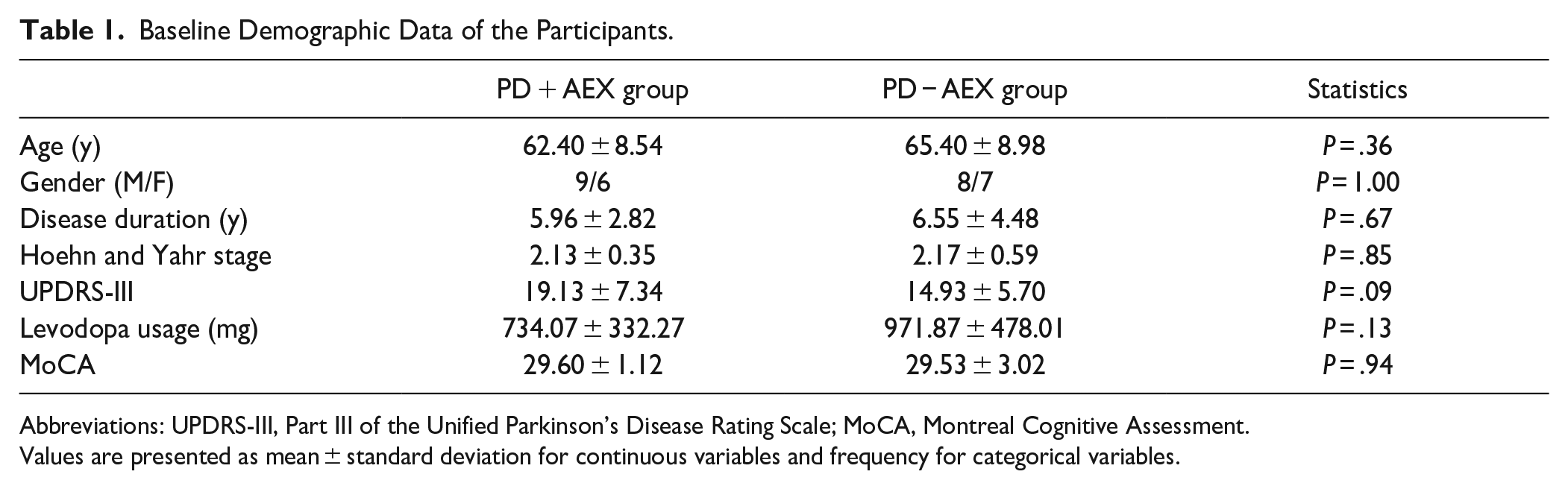
Abbreviations: UPDRS-III, Part III of the Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment.
Values are presented as mean ± standard deviation for continuous variables and frequency for categorical variables.
As for the response time during the acquisition phase, repeated measures ANOVA revealed that there was a significant block main effect (
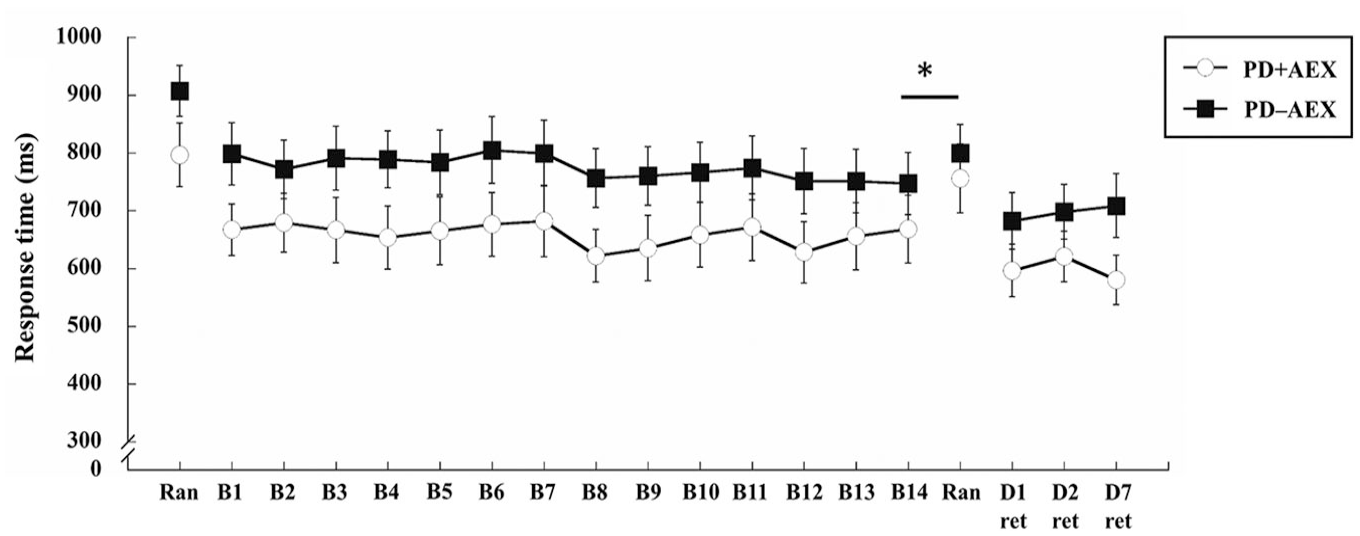
Response time of the PD + AEX and PD − AEX groups throughout practice (B1-B14), random blocks (Ran), and retention tests (ret). Range bars represent standard error.
Analysis of the retention tests showed no significant day main effect (
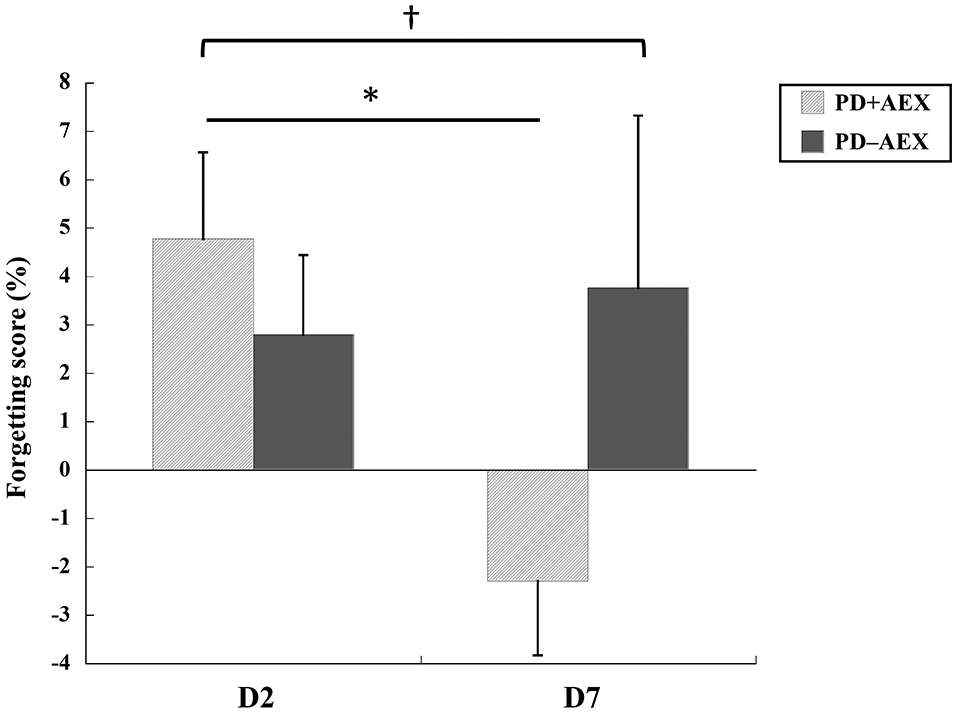
Forgetting scores of the second day (D2) and seventh day (D7) retention tests.
TMS Outcomes
Table 2 presents the TMS results at pre-test, D1, D2, and D7. The Mann–Whitney
Corticomotor Excitability Measured by Transcranial Magnetic Stimulation of the 2 Groups.
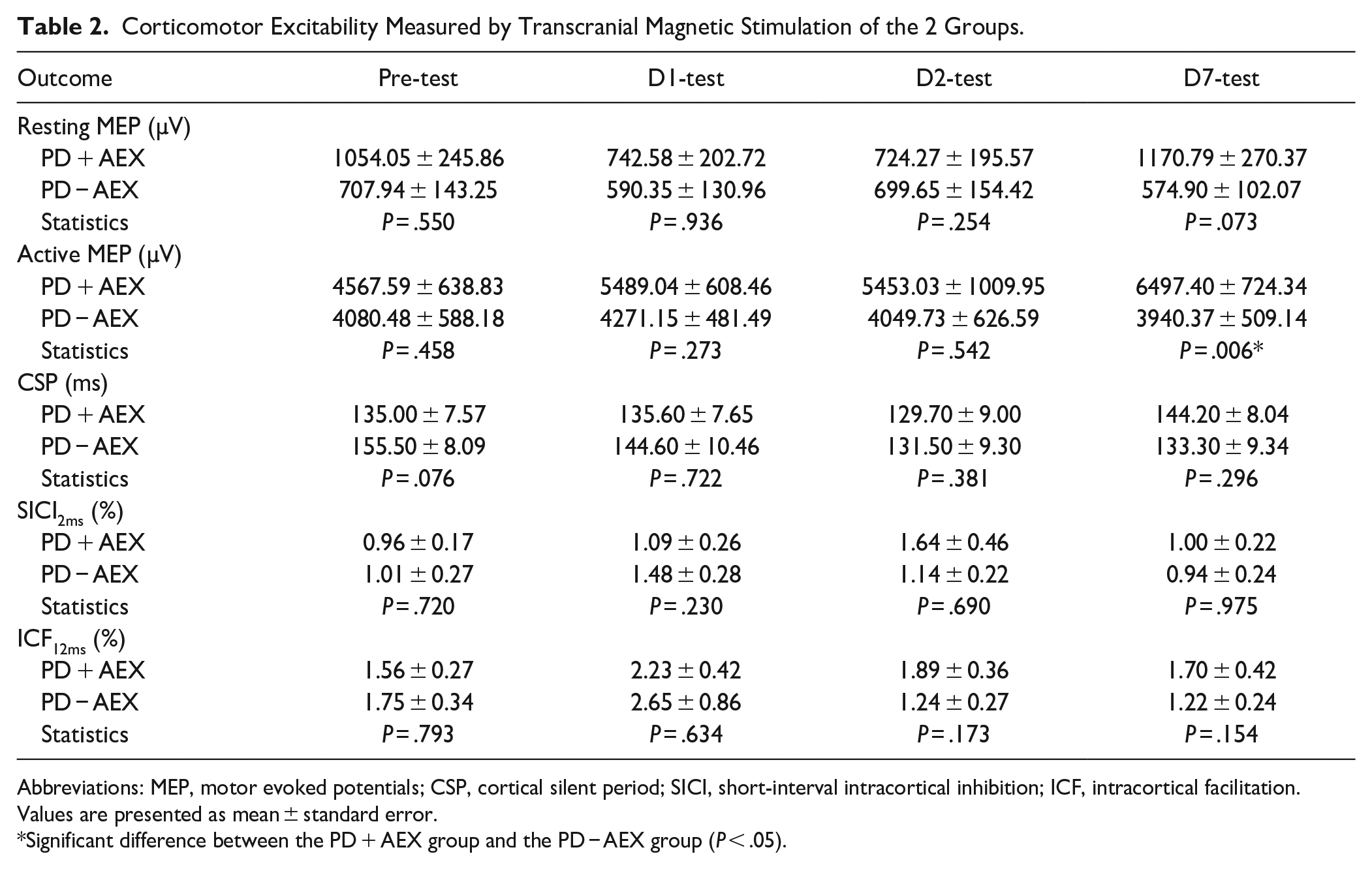
Abbreviations: MEP, motor evoked potentials; CSP, cortical silent period; SICI, short-interval intracortical inhibition; ICF, intracortical facilitation.
Values are presented as mean ± standard error.
Significant difference between the PD + AEX group and the PD − AEX group (
Discussion
This study aimed to investigate the behavioral and neurophysiological effects of a single bout of aerobic exercise on motor sequence learning in people with PD. The results showed that a single bout of moderately high intensity aerobic exercise immediately after practicing SRTT could facilitate motor learning, as demonstrated by significant reduction in forgetting on D7 as compared to D2 in the PD + AEX group, but not in the PD − AEX group. Furthermore, TMS results showed that MEP amplitudes under active contraction conditions were significantly larger on D7 for the PD + AEX group as compared to the PD − AEX group, suggesting a greater net excitability of the corticospinal tract. 29
During the acquisition phase, both groups showed a significant reduction in response time. There were also significant differences between the last block of the SRTT practice (ie, B14) and the block of random sequences. These results suggest that the participants with PD learned the 12-element target sequence. It should be noted from Figure 2 that although not statistically significant, the participants in the PD − AEX group seemed to perform slower than those in the PD + AEX group throughout the acquisition phase. Since this observation occurred before aerobic exercise was conducted and there were no apparent differences in the characteristics of patients, the observed between group differences was probably a result of randomization. To account for potential baseline differences, forgetting scores were then calculated to compare retention performance differences between the groups.
A more interesting result was observed on D7, a week after practice, in which the PD + AEX group showed a significant
The benefit of aerobic exercise appeared on D7 but not on D2, suggesting that offline learning might have occurred at a time period greater than 24 hours after practice. Memory consolidation of a newly acquired skill is found to take approximately 24 to 72 hours. 32 Hence, the beneficial effects on motor learning may become more visible after a longer period of time when plasticity-induced memory consolidation becomes more stable. 20 Another influential factor might be related to dopamine neurotransmission. It has been demonstrated that aerobic exercise can upregulate dopamine and facilitate receptor binding during some critical stages of memory formation, which may further benefit memory consolidation.19,21 Additionally, aerobic exercise can elevate the brain-derived neurotrophic factor (BDNF) level, which is also known to be responsible for memory formation. 22 BDNF can enhance the survival of dopaminergic neurons, improving dopaminergic neurotransmission and motor performance. 23 It is possible that the aerobic exercise after SRTT elevated the BDNF levels during the process of memory consolidation and thus further facilitated the retention performance of SRTT on D7. More research is needed to explore the above proposed mechanisms for enhanced learning in people with PD.
While similar findings were observed in earlier studies, 1 novelty of the present study is that a motor sequence task was used. The majority of previous studies used motor adaptation tasks, such as a hand tracking visuomotor task8,30,31 or a stabilometer balance task.10,11 Very few studies have investigated the influence of aerobic exercise on motor sequence tasks. Evidences from neuroimaging studies have suggested that motor sequence and motor adaptation tasks may involve different neural networks. While motor adaptation mainly involves the cortico-cerebellar circuits, motor sequence learning is dominated by the frontostriatal circuit.33,34 Owing to striatal impairment, people with PD seemed to have great difficulty learning motor sequence tasks, such as the SRTT. 16 It thus becomes important to demonstrate that a single bout of aerobic exercise could also facilitate motor sequence learning for people with PD.
Using TMS, this study provided preliminary evidence for neuroplastic changes induced by aerobic exercise after motor skill practice in people with PD. Interestingly, the TMS data showed that the PD + AEX group had a significantly larger active MEP amplitude than the PD − AEX group on D7 but not on D2, which coincides with the behavioral finding. Larger active MEP amplitudes indicate greater excitability of the corticospinal tract, which is a net result of increased excitation and decreased inhibition, during active contraction condition. 29 Since there were no significant group differences in CSP or SICI, we speculated that the increased MEP amplitude was primarily a result of enhanced corticomotor excitation but not decreased inhibition. In addition to the group difference on D7, there was a trend of gradual increase in the active MEP amplitude for the PD + AEX group each day (Table 2). The changes in MEP amplitude across days even exceeded the smallest detectable change of 423 to 931 µV reported in a TMS reliability study conducted on elderly adults. 35 Since there were no significant changes in other TMS parameters, we speculated that the influence of aerobic exercise might be specific to the corticospinal excitability of the active condition in people with PD. Performing the SRTT requires activation of the FDI muscles; hence, greater excitability of the corticospinal tract during active condition might denote better efficiency of neural connection and lead to better motor performance. Previous studies have suggested that increased neuronal excitability may be mediated via the increased blood lactate after aerobic exercise.9,36 Additionally, aerobic exercise can also increase dopamine, serotonin, and norepinephrine levels, which may also enhance M1 excitability. 37 To the best of our knowledge, this is the first study that investigated changes in corticomotor excitability after motor learning and aerobic exercise in people with PD. More studies are needed in this field to support our findings and understand the mechanisms.
One major limitation of the study was that we did not recruit more participants to minimize variations in the TMS data. The sample size was estimated from previous related studies using the results of motor performance after aerobic exercise, and suggested that 15 participants in each group would be sufficient. However, this may not be sufficient to compare the group differences in TMS outcomes. Owing to the non-normal distribution and great variability of the TMS data, non-parametric statistics were used, which may have limited the interpretation of the results. Future studies with larger sample sizes are warranted to support the TMS findings. Another potential limitation of the study was the design of the SRTT task. In many SRTT studies, the repeated and random sequences were interspersed throughout practice to determine the differences in sequence learning and general learning of the task, respectively. The design of SRTT was slightly modified in this study because we were interested in how aerobic exercise might affect the retention of the learned repeated target sequence. To meet our research goal, the random sequences were placed before and after practicing the target sequences, while the target sequences were tested again immediately after the aerobic exercise or rest (D1), on D2, and on D7. Future research could retest the influence of aerobic exercise on motor sequence learning using the traditional SRTT design.
Conclusion
This is the first study to investigate the behavioral and neurophysiological effects of a single bout of aerobic exercise on motor sequence learning in people with PD. The results of this study showed that aerobic exercise was effective in facilitating motor sequence learning in individuals with PD. Additionally, an increased active MEP amplitude was observed on D7, suggesting greater corticospinal excitability. Clinical implications will be that clinicians can provide a single bout of aerobic exercise after motor skill acquisition to maximize the training effects for people with PD who have mild to moderate disease severity with intact cognitive function.
Footnotes
This study was conducted at School and Graduate Institute of Physical therapy, College of Medicine, National Taiwan University.
The Institutional Review Board of National Taiwan University Hospital approved the study protocol.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of Taiwan [MOST 109-2314-B-002-128- and MOST 110-2314-B-002-048-MY3].
