Abstract
Background
Preconditioning with cathodal high-definition transcranial direct current stimulation (HD-tDCS) can potentiate cortical plasticity induced by intermittent theta burst stimulation (iTBS) and enhance the after-effects of iTBS in healthy people. However, it is unclear whether this multi-modal protocol can enhance upper limb function in patients with stroke.
Objective
The aim of this study was to investigate whether priming iTBS with cathodal HD-tDCS over the ipsilesional M1 can augment upper limb motor recovery in poststroke patients.
Methods
A total of 66 patients with subacute stroke were randomly allocated into 3 groups. Group 1 received priming iTBS with HD-tDCS (referred to as the tDCS + iTBS group), Group 2 received non-priming iTBS (the iTBS group), and Group 3 received sham stimulation applied to the ipsilesional M1. One session was performed per day, 5 days per week, for 3 consecutive weeks. In Group 1, iTBS was preceded by a 20-minute session of cathodal HD-tDCS at a 10-minute interval. The primary outcome measure was the Fugl-Meyer Assessment-Upper Extremity (FMA-UE) score. Moreover, the secondary outcome measures for muscle strength and spasticity were the Motricity Index-Upper Extremity (MI-UE) and the Modified Ashworth Scale Upper-Extremity (MAS-UE), respectively, and the Hong Kong Version of the Functional Test for the Hemiplegic Upper Extremity (FTHUE-HK) and the Modified Barthel Index (MBI) for activity and participation.
Results
Significant differences were detected in the changes in FMA-UE, MI-UE, and MBI scores between the 3 groups from baseline to post-intervention (χ2FMA-UE = 10.856, P = .004; χ2MI-UE = 6.783, P = .034; χ2MBI = 9.608, P = .008). Post hoc comparisons revealed that the priming iTBS group demonstrated substantial improvements in FMA-UE (P = .004), MI-UE (P = .028), and MBI (P = 0.006) compared with those in the sham group. However, no significant difference was observed between the iTBS group and the sham group. Moreover, no significant differences were found in the changes in MAS-UE or FTHUE-HK between the groups.
Conclusions
Priming iTBS with HD-tDCS over the ipsilesional M1 cortex had beneficial effects on augmenting upper limb motor recovery and enhancing daily participation among subacute stroke patients.
Keywords
Introduction
In recent years, repetitive transcranial magnetic stimulation (rTMS) has gained a lot of attention in treating upper limb dysfunction of patients with stroke because it can regulate brain plasticity. Long-term potentiation- (LTP-) like facilitation or long-term depression- (LTD-) like suppression can be induced by different frequencies of rTMS.1-3 Excitatory rTMS stimulation, delivered to the ipsilesional the primary motor cortex (M1), is a promising treatment to boost poststroke motor recovery during upper limb training.4-7 Particularly, Kim’s study evaluated motor performance and cortical excitability and demonstrated the beneficial effect of an excitatory rTMS over the affected hemisphere to boost the upper-limb motor recovery of stroke patients. 5 This improvement would be attributable to alterations in excitability in the M1 of the affected hemisphere. In addition, Gao et al 8 reported in an animal experiment that infarct volumes of a rat model with acute stroke were decreased substantially after 7 sessions of 20 Hz-rTMS on the lesional hemisphere.
Intermittent theta burst stimulation (iTBS) is a unique rTMS pattern that employs high-frequency bursts (eg, 50 Hz) repeated at the theta frequency 9 to increase the cortical excitability of the stimulated M1, which is delivered intermittently. 10 It has a shorter stimulation time (3 minutes for 600 pulses) and comparable therapeutic effects to high-frequency rTMS paradigms.11,12 In stroke patients, iTBS to the ipsilesional M1 has been demonstrated to enhance upper limb outcomes and boost grip strength recovery in acute and chronic stroke patients.13-15 However, the response to TBS protocols, such as all current methods of non-invasive brain stimulation, varies greatly between individuals. 16 Nevertheless, the therapeutic effect of TBS is still open to debate.
The pre-existing synaptic excitatory state of the stimulus targets is a critical factor that results in variability in the response to TBS. 17 Prior synaptic activity influences subsequent neuroplasticity via dynamic modulation of the threshold of synaptic plasticity. 18 On the one hand, reduced postsynaptic activity lowers the threshold for preferred LTP; on the other hand, increased postsynaptic activity raises the threshold for preferential LTD. This mechanism is termed the plasticity of synaptic plasticity for homeostasis or metaplasticity.19-21
Therefore, a priming test design followed by a conditioning session has been used to test homeostatic patterns in the human M1. 17 For instance, an inhibitory priming procedure using 1-Hz rTMS or continuous theta burst stimulation (cTBS) may stabilize or even enhance the facilitative effect of subsequent excitatory conditioning stimulation.18,22 Additionally preconditioning with transcranial direct current stimulation (tDCS) can help to sensitize the motor cortex to the conditioning effects of 5-Hz rTMS.23-25 In addition, previous study by our team have confirmed that the preintervention with cathodal high-definition (HD) tDCS before iTBS has a greater effect on augmenting the peak amplitude of motor-evoked potentials at the stimulated site than does iTBS alone for healthy people. 2 These findings indicate that tDCS preconditioning may be a viable method for increasing the sensitivity of corticospinal projection neurons to rTMS modulation.
However, whether priming iTBS with cathodal HD-tDCS over the ipsilateral M1 can enhance motor recovery in the upper extremity in patients with stroke is still unknown. Therefore, this randomized controlled trial aimed to propose an innovative scheme of applying preconditioning with cathodal HD-tDCS before an iTBS session among poststroke patients. Our hypothesis was that individuals experiencing upper limb dysfunction who underwent 15 sessions of the priming treatment, would exhibit superior recovery from upper limb motor impairment than those who received sham stimulation or iTBS alone.
Materials and Methods
Participants
A total of 66 participants with poststroke hand dysfunction were recruited from December 2021 to December 2022 in this randomized, sham-controlled, assessor-blinded study. All the subjects were inpatients at Wuxi Central Rehabilitation Hospital. This study was approved by the Ethics Committee of Wuxi Mental Health Center (Wuxi Central Rehabilitation Hospital; No. WXMH-CIRBS2021LLky142) and registered in the Chinese Clinical Trial Registry (No. ChiCTR2200063397). The CONSORT reporting guidelines for clinical trials were followed in this study (Figure 1). Each participant provided written informed consent before participating and answered the safety questionnaire about contraindications to rTMS.
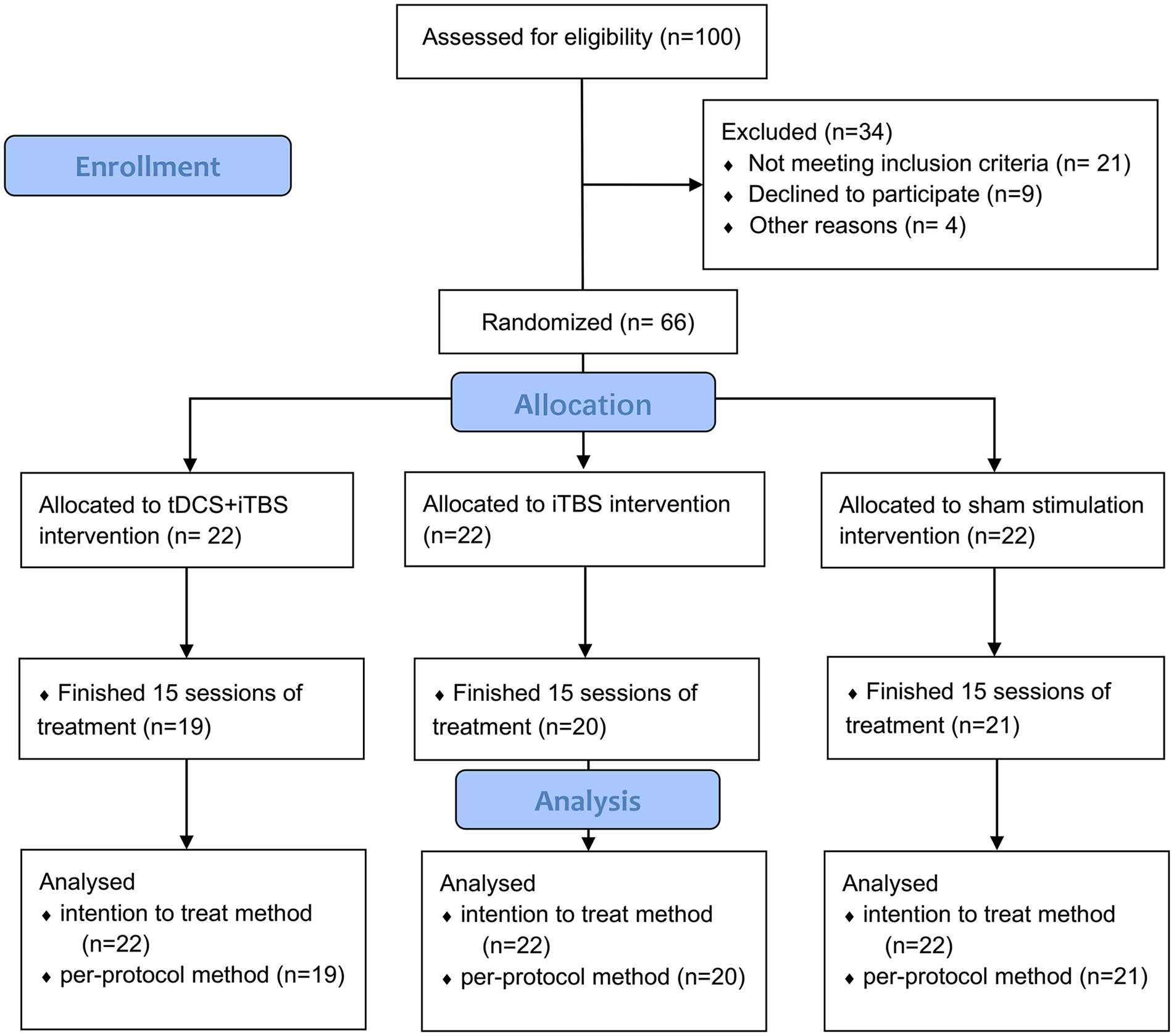
CONSORT diagram of patient flow through the study.
Inclusion/Exclusion Criteria
The inclusion criteria for participants were as follows: (1) met the diagnostic criteria for ischemic or hemorrhagic stroke using computed tomography or magnetic resonance imaging; (2) experienced a first-ever, ischemic or hemorrhagic stroke with unilateral hemispheric involvement; (3) aged 35 to 75 years; (4) with post-stroke onset from 2 weeks to 3 months; (5) Brunnstrom stage I-IV; and (6) signed informed consent to participate in the study.
The exclusion criteria were as follows: patients who had (1) any contraindications involving rTMS, as listed in the safety questionnaire 26 ; (2) severe cognitive and communication impairment (ie, a score of <17/30 on the Mini-Mental Status Examination) that had difficulty in following the therapist’s instructions; (3) hemiparetic upper limb mobility is hampered by excruciating pain or considerable stiffness (ie, a modified Ashworth score >3, specifically the shoulder adductor, elbow flexor, and wrist palmar flexor); and (4) serious cervical spine lesions, including severe cervical spinal stenosis and cervical spine instability.
Prior Sample Size Estimation
Based on a previous study, 12 the observed increases in FMA-UE scores after the intervention were 6.5 ± 2.88, 5.29 ± 2.81, and 3.5 ± 2.06 points for the priming iTBS, nonpriming iTBS, and sham stimulation groups respectively. We determined the requisite sample size for the 1-way analysis of variance (ANOVA) F Test employing PASS 15 software. The effect size was 0.44, with a power of 0.80 and a 2-sided alpha of .05, a mini mum of 18 patients per group was necessary. To accommodate a potential 15% dropout rate, our targeted sample size was set at a minimum of 66 patients.
Randomization and Blinding
In this trial, 3 parallel groups received the following interventions: Group 1-preconditioning with cathodal HD-tDCS before the iTBS session (ie, HD-tDCS + iTBS), Group 2-non preconditioning before the iTBS session (ie, sham HD-tDCS + iTBS), and Group 3-sham stimulation (ie, sham HD-tDCS + sham iTBS). After completing the baseline assessments, participants were randomly assigned to 1 of the 3 groups at a 1:1:1 ratio, with the allocation sequence being kept confidential from all the researchers and assessors. Participants underwent 15 sessions, 5 times per week for 3 weeks. The primary outcome and secondary outcomes were assessed within 3 days before (T0) the intervention and immediately after (T1) 15 sessions in the 3 groups.
Both the participants and the therapist were blinded to the allocation of interventions. The assessment of outcome measures was carried out by a highly experienced therapist who was unaware of the participants’ group allocation. The HD-tDCS and iTBS protocols were administered by an investigator who was not involved in the clinical assessment or data analysis processes.
Routine Rehabilitation Treatment
The study participants underwent an extensive 3-week neurological rehabilitation program comprising standardized physical therapy and occupational therapy protocols administered by experienced therapists. This comprehensive program comprised 15 sessions conducted 5 days a week. Its primary goal was to improve diverse facets of physical function, such as strength, flexibility, transfers, posture, balance, coordination, and activities of daily living, with a particular focus on enhancing upper limb movements. The physical therapy sessions spanned 40 minutes, while the occupational therapy sessions lasted 30 minutes, immediately following the iTBS treatment.
Cathodal HD-tDCS
The tDCS equipment (VC-8000 series) used a 4 × 1 HD-tDCS adaptor (Soterix Medical, New York, NY, USA) and 5 small round electrodes (l cm2). Before the intervention, different sizes of caps were selected according to the size of the subjects’ heads so that the EEG caps were close to the heads and the electrodes were accurately positioned. The central cathode electrode was placed on the scalp over the hand area in the ipsilesional M1, and the 4 return electrodes (3.5 cm away from the central electrode) were placed to form a circular current loop coated with a conductive paste (Figure 2). Following a connection quality check, tDCS was carried out for 20 minutes at a current intensity of 1.5 mA. The current of the sham stimulus gradually increased to 1.5 mA in the first 15 seconds and subsequently declined to 0 in the next 15 seconds; the other parameter settings were the same as those used for the real stimulus.
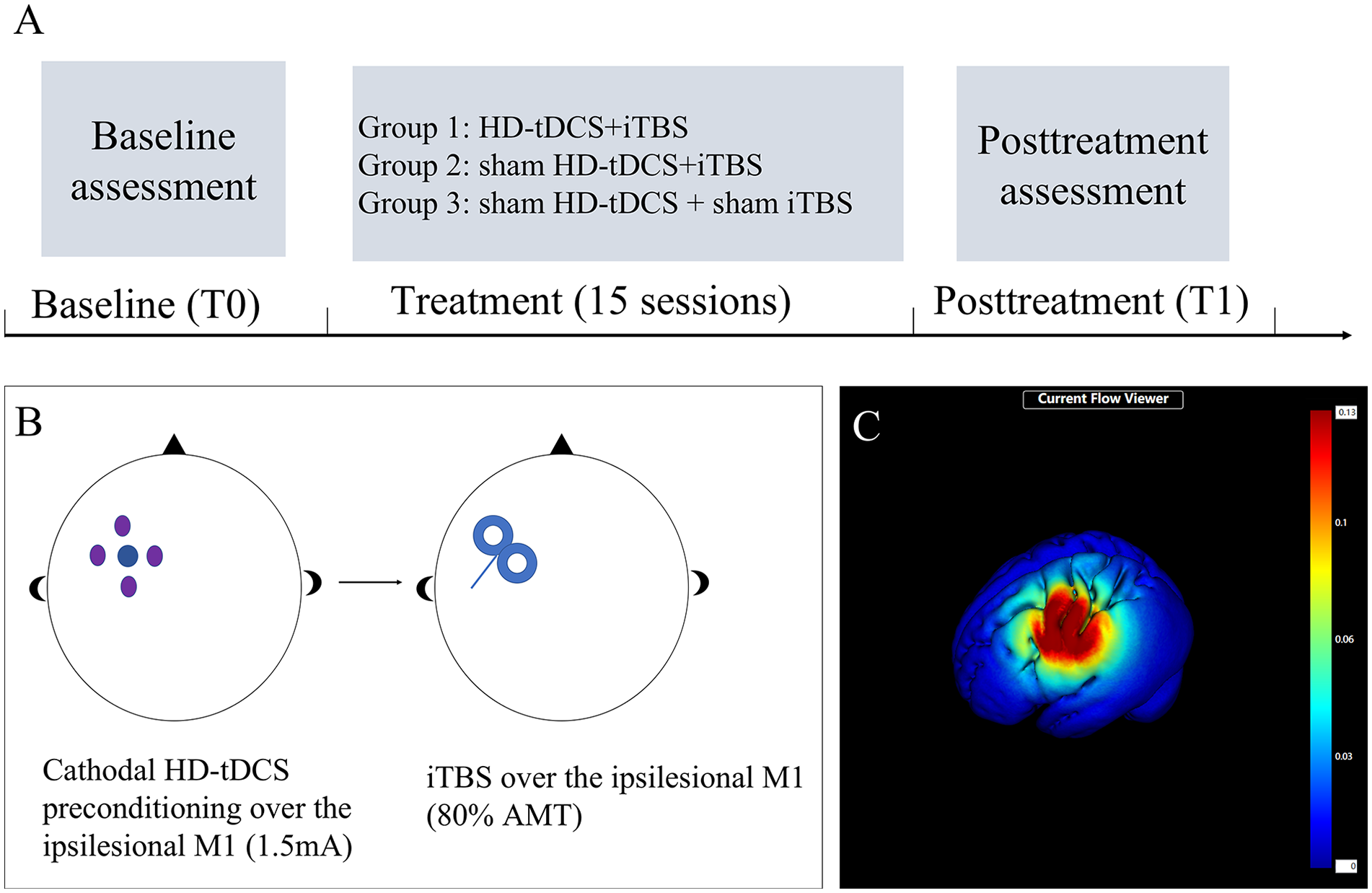
Study design and methods. (A) Schematic diagram of the study design. (B) Priming iTBS with cathodal HD-tDCS. (C) Electrical field induced by cathodal HD-tDCS and iTBS stimulation.
Intermittent Theta Burst Stimulation
A Magneuro® (Vishee Medical Technology Co., Ltd., Nanjing, China) with a 70-mm figure-8-shaped stimulation coil was used to perform the iTBS protocol. 2 The coil was positioned tangentially to the scalp over the hand area in the ipsilesional M1. The handle was held backwards and laterally at an angle of 45° to the sagittal plane to generate a posterior-to-anterior current flow in the brain. By measuring the active motion threshold (AMT) of the contralesional M1, the AMT was defined as the minimum output of stimulation that produced a motor-evoked potential (MEP) of >200 μV in at least 5 out of 10 trials with 10% maximal voluntary contraction of the abductor pollicis brevis. 27 The stimulation intensity was set at 80% of the AMT. The iTBS stimulation pattern consisted of 3 pulses at 50 Hz repeated at 5 Hz; a 2 seconds train of TBS was repeated every 10 seconds for 192 seconds. 10 A customized sham coil was used in the iTBS sham treatments, which guaranteed that each participant heard the same clicking noises at the same frequencies and locations.
Both the HD-tDCS and the iTBS sessions were performed on the ipsilesional M1, which is the mirror motor hotspot of the contralesional hemisphere. 28 There was a 10-minute interval between HD-tDCS preconditioning and subsequent iTBS.
Primary Outcome
The Fugl-Meyer Assessment-Upper Extremity (FMA-UE) was used to observe the degree of upper limb motor impairment 29 ; this scale consists of 33 items with a total score of 66. The higher the score is, the better the upper limb motor function.
Secondary Outcomes
(1) Stroke severity and disability were evaluated using the National Institutes of Health Stroke Scale (NIHSS) 30 ; (2) The Hong Kong Version of the Functional Test for the Hemiplegic Upper Extremity (FTHUE-HK) was used to determine a stroke patient’s ability to use the upper limbs in daily life tasks during recovery; (3) The Upper Extremity Strength Index (Motricity Index-Upper Extremity, MI-UE) was used to assess muscle strength on the hemiplegic side; (4) The Modified Barthel Index (MBI) was used to assess daily living activity, with a total of 10 items and a total score of 100; and (5) The Modified Ashworth Scale for Upper-Extremity (MAS-UE) was used to evaluate the muscle tension of the hemiplegic biceps and pectoralis major.
Data Analysis
The statistical analysis was performed using SPSS 22.0, while GraphPad Prism 8 was used for data visualization. P < .05 indicated statistical significance (2-tailed). Categorical data were analyzed using the chi-square test and are reported as counts. Normality was assessed using the Shapiro-Wilk test, and homogeneity of variance was evaluated using the Levene test for continuous data. For normally distributed data with homogeneous variances, intergroup comparisons of demographic and baseline data were conducted using 1-way ANOVA. For the outcome measures involving continuous data (FMA-UE, MI-UE, and MBI scores), within-group comparisons were assessed using paired t tests, while intergroup comparisons were evaluated using covariance analysis with the baseline FMA-UE, MI-UE, and MBI scores as covariates. In cases where the outcome data violated the assumptions of a normal distribution or homogeneity of variance, nonparametric tests such as the Wilcoxon signed-rank sum test and the Kruskal‒Wallis rank sum test were employed for within-group and intergroup comparisons, respectively. The results of these nonparametric tests are reported as the median (interquartile range [IQR]). In instances where a significant intergroup effect was observed, post hoc comparisons were adjusted using the Bonferroni correction method. Both Per-Protocol and Intention-To-Treat (ITT) analyses were also conducted on the continuous data.
Results
A total of 66 eligible patients met the inclusion and exclusion criteria and participated in the study. However, during the intervention, 6 subjects (3 from the tDCS + iTBS group, 2 from the iTBS group, and 1 from the sham group) withdrew from the trial. A total of 60 patients successfully completed the trial (Figure 1). The patient was classified as MEP+ if MEPs with a peak-to-peak amplitude ≧50 μV could be elicited in at least 4 out of 8 consecutive trials while the abductor pollicis brevis muscle on the affected side remained at rest. Conversely, if this criterion was not met with stimuli delivered at maximum intensity, the patient was classified as MEP-. 31 There were no significant differences in demographic or clinical characteristics among the 3 groups (Table 1, P > .05). The treatment protocol was well tolerated, and no significant adverse effects were reported in either group.
Demographic and Clinical Characteristics of the 3 Groups (n = 60).
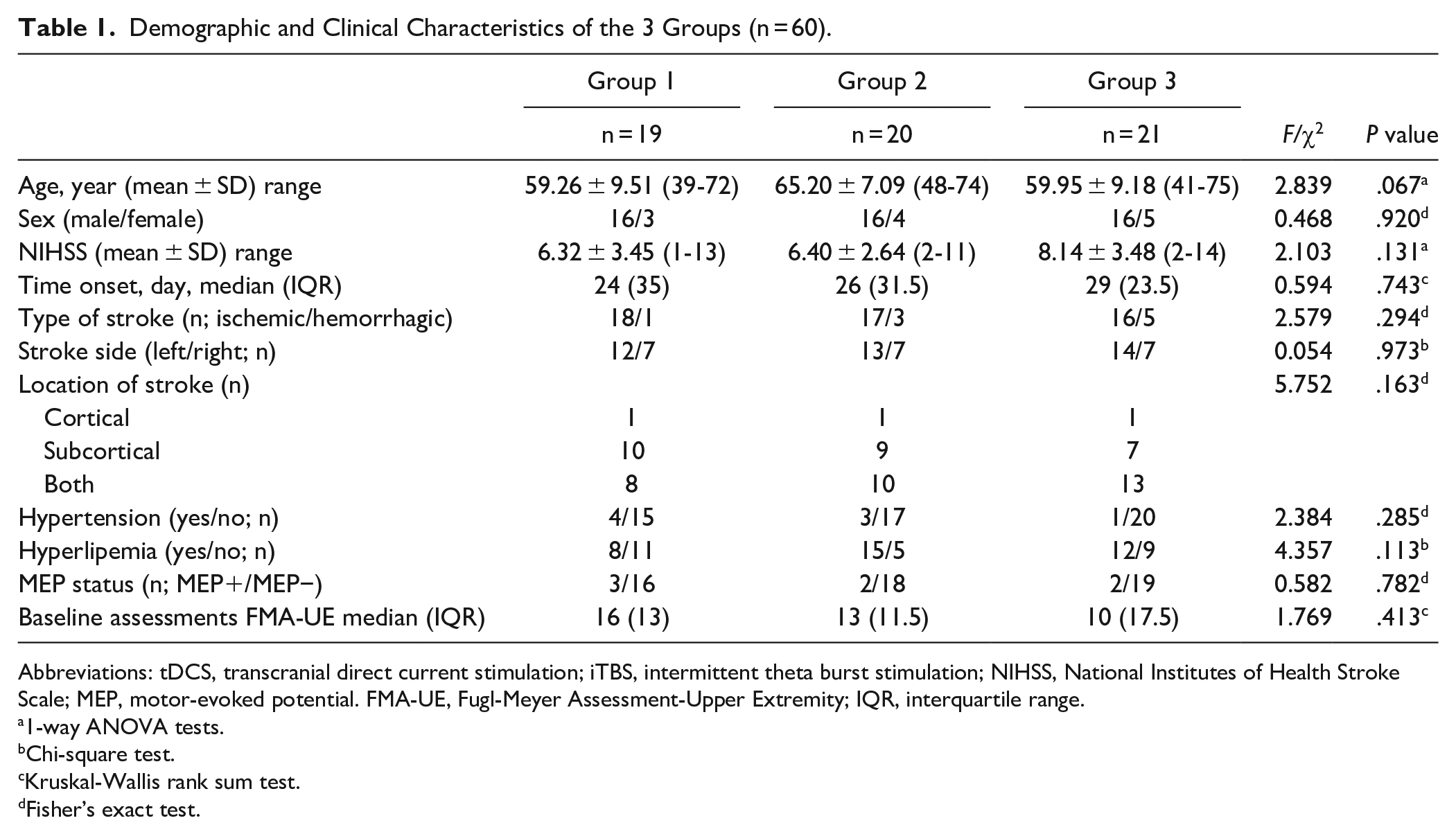
Abbreviations: tDCS, transcranial direct current stimulation; iTBS, intermittent theta burst stimulation; NIHSS, National Institutes of Health Stroke Scale; MEP, motor-evoked potential. FMA-UE, Fugl-Meyer Assessment-Upper Extremity; IQR, interquartile range.
1-way ANOVA tests.
Chi-square test.
Kruskal-Wallis rank sum test.
Fisher’s exact test.
The FMA-UE, MI-UE, and MBI scores were not normally distributed in any of the groups. To address this, we employed the Wilcoxon matched pairs with signed ranks test for within-group comparisons and the Kruskal‒Wallis rank sum test for intergroup comparisons. The results, presented in Table 2, were reported as medians (IQRs). At baseline (n = 60), no significant differences were found in FMA-UE (χ2 = 1.769, P = .413), MI-UE (χ2 = 2.355, P = .308), and MBI (χ2 = 2.925, P = .232) scores among the 3 groups. After the intervention, we found statistically significant within-group differences in FMA-UE (Z ≥ 3.824, P < .001), MI-UE (Z ≥ 3.628, P < .001), and MBI (Z ≥ 3.824, P < .001) scores among the 3 groups.
Changes in Clinical Outcomes Before and After Intervention in Per-protocol Method (n = 60).
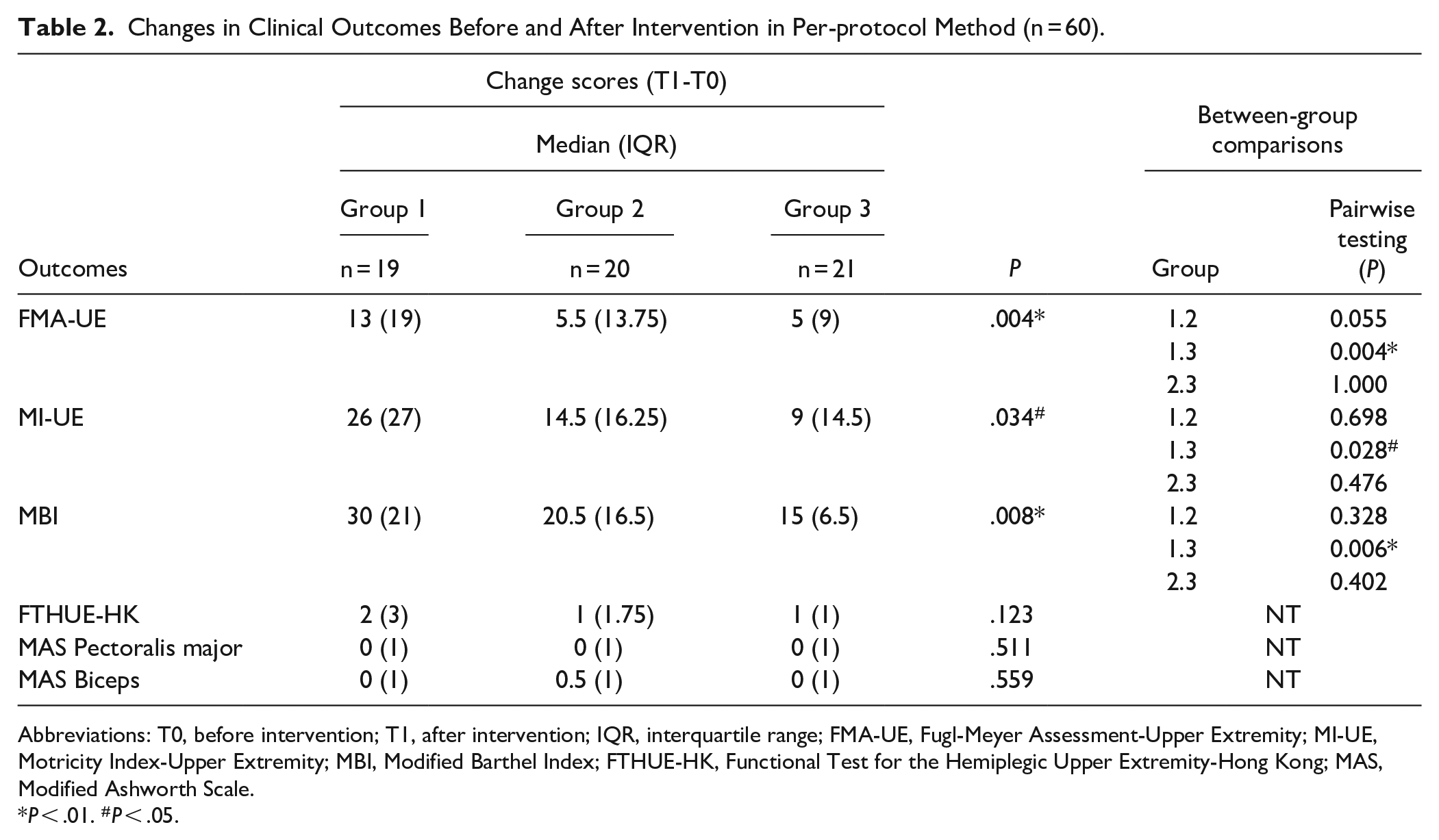
Abbreviations: T0, before intervention; T1, after intervention; IQR, interquartile range; FMA-UE, Fugl-Meyer Assessment-Upper Extremity; MI-UE, Motricity Index-Upper Extremity; MBI, Modified Barthel Index; FTHUE-HK, Functional Test for the Hemiplegic Upper Extremity-Hong Kong; MAS, Modified Ashworth Scale.
P < .01. #P < .05.
Significantly, the Kruskal-Wallis rank sum tests revealed significant differences in FMA-UE, MI-UE, and MBI score changes between the groups from baseline to post-intervention (χ2FMA-UE = 10.856, P = .004; χ2MI-UE = 6.783, P = .034; χ2MBI = 9.608, P = .008; n = 60; Figure 3). Post hoc comparisons revealed that the tDCS + iTBS group showed significantly greater improvement than the sham group in FMA-UE (P = .004), MI-UE (P = .028), and MBI (P = .006; Figure 3), while no significant difference was observed between the iTBS group and the sham group.
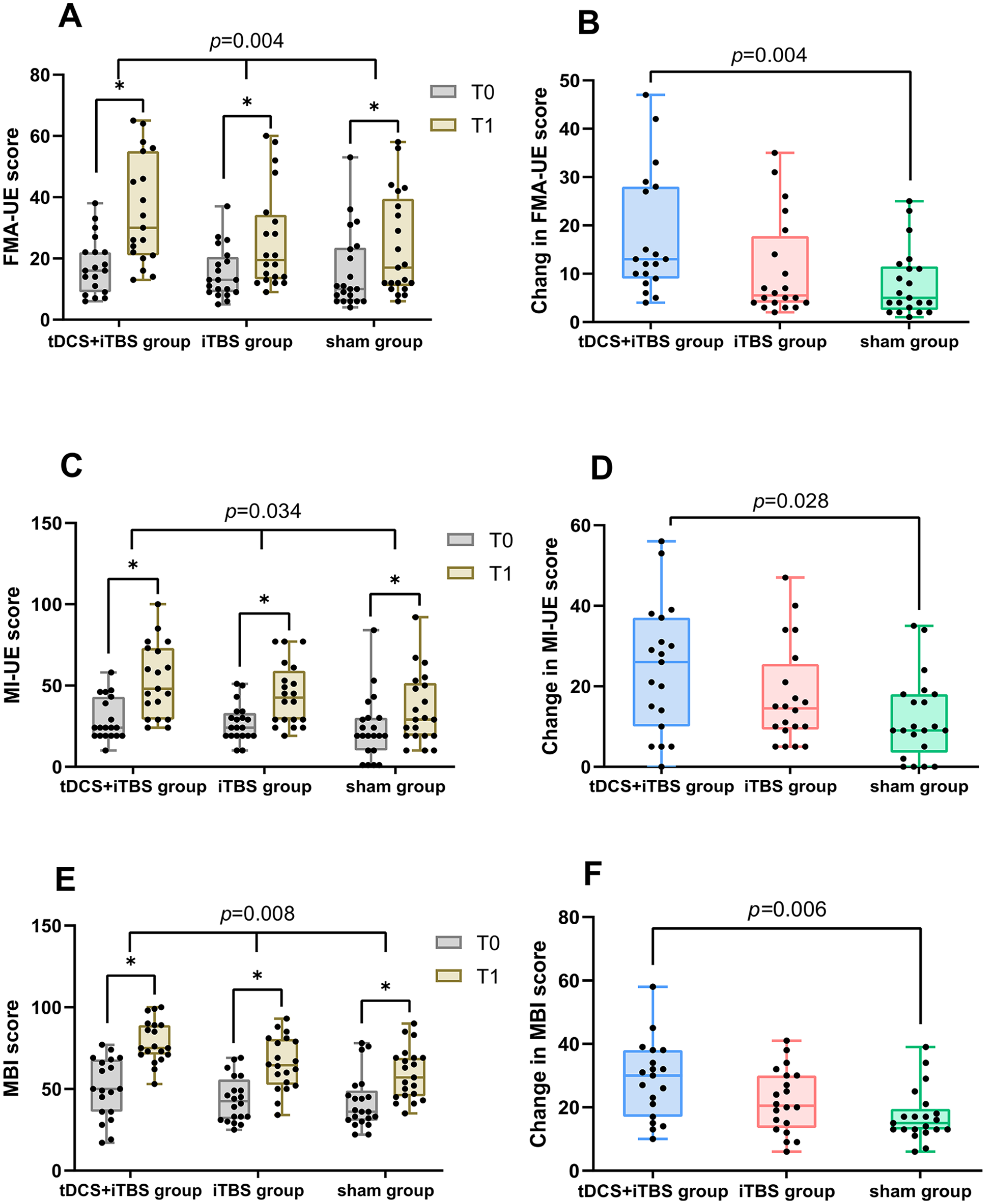
Comparison of improvements in FMA-UE, MI-UE, and MBI scores among the 3 groups before and after the intervention (n = 60). T0: Before intervention; T1: After intervention; (A) (C) (E) Significant between-group differences were observed in terms of changes in the FMA-UE score (P = .004), MI-UE score (P = .034), and MBI score (P = .008). Furthermore, compared to those at the T0 time point, the FMA-UE score, MI-UE score, and MBI score significantly improved in all 3 groups at the T1 time point (*P < .001). (B) (D) (F) According to pairwise comparisons of the FMA-UE difference, MI-UE difference, and MBI difference, the tDCS + iTBS group exhibited significantly greater improvement than the sham group (P = .004, P = .028, P = .006). The data points are participants.
As presented in Table 2, there were no significant differences in the changes in the FTHUE-HK (χ2 = 4.189, P = .123) or MAS-UE (χ2pectoralis major = 1.343, P = .511; χ2biceps = 1.162, P = .559) scores among the 3 groups.
Additionally, the ITT analysis yielded comparable results with a sample size of 66 (see Table 3).
Changes in Clinical Outcomes Before and After the Intervention According to the Intention-to-treat Method (n = 66).
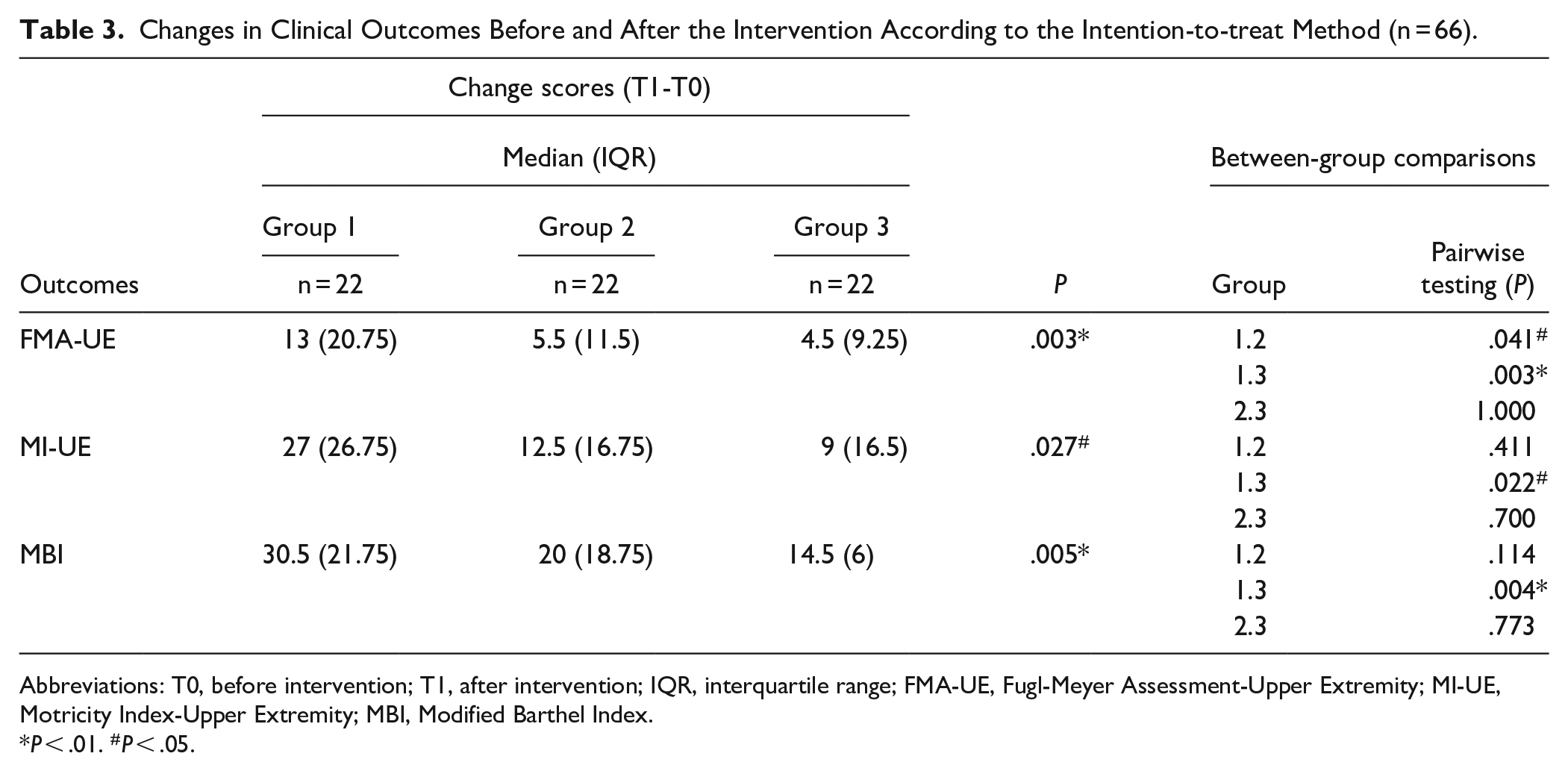
Abbreviations: T0, before intervention; T1, after intervention; IQR, interquartile range; FMA-UE, Fugl-Meyer Assessment-Upper Extremity; MI-UE, Motricity Index-Upper Extremity; MBI, Modified Barthel Index.
P < .01. #P < .05.
Discussion
The present study revealed that priming iTBS with cathodal HD-tDCS achieved a superior effect compared with sham stimulation in boosting upper limb motor recovery among patients with subacute stroke, as demonstrated by the increase in FMA-UE and MI-UE scores. However, no difference was found between the non-priming iTBS group and the priming iTBS group. The changes in FTHUE-HK and MAS did not differ among the 3 groups. To our knowledge, this is the first randomized controlled study to investigate the effectiveness of preconditioning with cathodal HD-tDCS before iTBS over the ipsilesional M1 cortex for augmenting upper limb motor recovery in patients with subacute stroke.
Repetitive transcranial magnetic stimulation (rTMS) has become widely used to treat upper limb paresis with stroke, however, evidence of applying facilitatory rTMS on the ipsilesional hemisphere for upper extremity motor recovery remains limited. 32 Thus, the current research results on its effectiveness are inconsistent. Our research results show that nonpriming iTBS did not augment the gains from the upper limb motor function. This finding is consistent with the results of previous reports. 33 Several possible reasons may be related to the fact that nonpriming iTBS did not appear more effective than sham stimulations. First, it may have been masked by differential responses to rTMS based on lesion location, 34 and our trial was not adequately powered to detect it; second, the majority of stroke patients in our study exhibited notably severe upper limb impairments, evidenced by the absence of MEP, a key biomarker of corticospinal tract integrity in the affected upper limb. Severe poststroke arm impairment is generally associated with lower structural reserve. 35 In line with our findings, there few studies are achieving positive responses to non-invasive brain stimulation in stroke patients with severe impairments.36,37 It indicates that relatively severe patients are likely more difficult responders. These results may be attributed to the lower structural reserve spared by stroke injury, which limited the ability of iTBS over the affected M1 cortex to enhance synaptic responses. In other words, the amount of improvement that can be achieved or demonstrated in patients with severe upper limb impairments may be limited. Alternatively, longer periods of treatment may be needed at this stage for a plasticity-modifying intervention to produce a measurable effect. 33 Although non-priming iTBS stimulation did not show significant benefits in our study, priming iTBS with HD-tDCS stimulation effectively boosts motor function recovery.
A possible factor affecting responsiveness may be attributed to the brain activity before the stimulation. 17 Hence, the present study provided a possible strategy for adopting a preconditioning protocol to augment the effect of iTBS on the M1 cortex. We used preconditioning HD-tDCS before iTBS sessions for subacute stroke patients. The findings suggested that priming iTBS with HD-tDCS on motor outcomes was superior to sham stimulation, but not to nonpriming iTBS. This result was consistent with the findings of previous priming trials of iTBS with cTBS trials for chronic stroke patients, which showed that priming iTBS was more efficient than nonpriming iTBS particularly in patients with severe upper limb impairment (FMA-UE scores <39.5). 12 The positive effect of the priming iTBS protocol in our study was also detected in subacute stroke participants with relatively severe hemiplegia. The significant improvement in FMA-UE score observed in the majority of stroke patients who were MEP negative in our study is noteworthy, which contrasts with prior research indicating limited upper limb functional recovery in MEP-negative individuals. 38 The role of preconditioning can be explained by the mechanism of synaptic homeostasis plasticity.17,19 This finding indicates that pre-stimulus synaptic activation states influence the aftereffect of TBS stimulation. This mechanism has been shown by some preliminary studies in healthy young adults to sensitize the motor cortex through cathodal HD-tDCS preconditioning, thereby leading to greater cortical plasticity changes induced by iTBS or HF rTMS.18,23 Importantly, our results suggest that this priming iTBS approach is also effective at boosting motor function recovery in subacute patients with lower-functioning upper limbs.
Furthermore, the enhancements observed in MBI scores among participants receiving priming iTBS were greater than those observed among participants who received sham stimulation. However, the changes in MBI did not significantly differ between the nonpriming iTBS group and the sham stimulation group. Therefore, this study indicated that the benefits gained from priming iTBS interventions may involve addressing activities of daily living and improving participation across subacute stroke patients. However, for improving upper limb spasticity, priming and nonpriming iTBS were not superior to sham stimulation in the subacute stage. To our knowledge, this is the first study to test the changes in upper limb spasticity after priming with the iTBS protocol. A reduction in upper limb spasticity after a single or repeat iTBS session over the M1 cortex was reported in chronic stroke patients.13,39,40 Upper limb spasticity increases gradually from the acute to the subacute stage, thereby peaking in the chronic stage during the pathophysiological event of a stroke. Therefore, the potential effects of reducing upper limb spasticity may be difficult to observe in the subacute stage.
This study has several limitations. First, due to the immediate postintervention assessment, long-term follow-up data concerning improvements in upper limb function were not available. Second, despite the prevalent absence of ipsilateral M1 MEPs in most participants, evaluating both MEPs and the ipsilateral silence period (iSP) before and after treatment is crucial for understanding neurophysiological changes and assessing the effectiveness of interventions. The iSP serves as an important indicator in assessing interhemispheric motor inhibition between cerebral hemispheres.41,42 These assessments could elucidate the underlying mechanisms associated with balancing corticomotor excitability, which involves facilitating the ipsilateral cortical excitability and reducing hemispheric inhibition subsequent to priming iTBS over the ipsilateral M1.
Conclusions
This randomized controlled study explored the potential impact and safety of multimodal non-invasive brain stimulation on the functional recovery of the hemiparetic upper limbs. Priming iTBS with HD-tDCS over the ipsilesional M1 had beneficial efficacy in improving upper limb function, thereby increasing daily participation. Furthermore, no patients suffered significant acute side effects after receiving either priming iTBS or non-priming iTBS. Therefore, priming iTBS with HD-tDCS may be a potential and safe intervention option as an adjuvant therapy that could enhance the therapeutic efficacy of neurorehabilitation in stroke patients. Thus, further studies with neurophysiological or neuroimaging techniques are warranted to explore the underlying mechanism involved.
Footnotes
Acknowledgements
The authors would like to extend their sincere thanks to the participants of this study. Special appreciation is given to Dr. Hanjin Liu from the First Affiliated Hospital of Sun Yat-sen University for his guidance throughout the study, and to Dr. Huaide Qiu from Nanjing Normal University of Special Education for his invaluable contribution to the data analysis.
Author Contributions
Li Bian: Data curation; Project administration; and Writing—original draft; Li Zhang: Formal analysis andWriting—original draft; Guilan Huang: Investigation; Project administration; and Supervision; Da Song: Supervision andVisualization; Kai Zheng: Conceptualization andFunding acquisition; Xinlei Xu: Supervision andValidation; Wenjun Dai: Software; Supervision; and Validation; Caili Ren Funding acquisition; Methodology; Project administration; Supervision; and Writing—review & editing; Ying Shen Conceptualization; Funding acquisition; Methodology; and Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Program of China ( No. 2022YFC2009700), the National Natural Foundation of China (No.82302882), the Natural Science Foundation of Jiangsu Province (No. BK20201138), the Social Development Project of Jiangsu Province (No. BE2022700), the Key Project of Jiangsu Province’s Key Research and Development Program (No.BE2023023-2), the Competitive Project of Jiangsu Province’s Key Research and Development Program (No.BE2023034), and the Top Talent Support Program for young and middle-aged people of Wuxi Health Committee.
