Abstract
Background
Cerebellum shares robust di-synaptic dentato-thalamo-cortical (DTC) connections with the contralateral motor cortex. Preclinical studies have shown that DTC are excitatory in nature. Structural integrity of DTC is associated with better upper extremity (UE) motor function in people with stroke, indicating DTC are important for cerebellar influences on movement. However, there is a lack of understanding of physiologic influence of DTC in humans, largely due to difficulty in accessing the dentate nucleus.
Objective
Characterize DTC physiology using dentate nucleus deep brain stimulation (DBS) combined with transcranial magnetic stimulation (TMS) in stroke.
Methods
Nine chronic stroke survivors with moderate-to-severe UE impairment (Fugl-Meyer 13-38) underwent a paired DBS–TMS experiment before receiving experimental dentate nucleus DBS in our first-in-human phase I trial (Baker et al., 2023, Nature Medicine). Conditioning DBS pulses were given to dentate nucleus 1 to 10 ms prior to supra-threshold TMS pulses given to ipsilesional motor cortex. Effects were assessed on motor evoked potentials (MEPs). Size of DBS-conditioned MEPs was expressed relative to TMS MEPs, where values >1 indicate facilitation.
Results
Dentate nucleus DBS led to facilitation of MEPs at short-latency intervals (3.5 and 5 ms, P = .049 and .021, respectively), a phenomenon we have termed dentato-cortical facilitation (DCF). Higher DCF was observed among patients with more severe UE impairment. Diffusion tensor imaging revealed microstructure of thalamo-cortical portion of DTC was related to higher corticomotor excitability.
Conclusions
Our in vivo investigation reveals for the first time in humans the intrinsic excitatory properties of DTC, which can serve as a novel therapeutic target for post-stroke motor recovery.
Keywords
Introduction
Stroke is a leading cause of long-term disability among adults. There are an estimated 8 million stroke survivors living in the United States. 1 More than two-thirds experience chronic upper extremity (UE) paresis despite intensive rehabilitation, and of them up to 50% experience moderate-to-severe paresis which significantly limits functional independence and ability to perform daily tasks.2,3 There remains a critical need for novel interventions to capitalize on intrinsic repair mechanisms of the brain and promote rehabilitation outcomes.
Cerebellum may be a useful target to promote post-stroke UE motor outcomes because its di-synaptic dentato-thalamo-cortical (DTC) connections are largely spared in a typical middle cerebral artery stroke. 4 Studies in animal models have revealed the excitatory potential of DTC for contralateral motor cortical excitability at short-latency intervals (~3 ms).5-7 Preclinical studies have shown invasive deep brain stimulation (DBS) of DTC delivered in conjunction with motor skill training in rodent stroke models can promote greater return of forelimb motor skill than motor training delivered alone.8,9 Mechanisms may include increased motor cortical excitability, map reorganization, and higher synaptogenesis and dendritic arborization.9-11 In humans, the role of the cerebellum in post-stroke UE movement has been investigated using neuroimaging. Functional magnetic resonance imaging (MRI) studies have demonstrated increased cerebellar activation during paretic hand movement in chronic stroke. 12 Those who exhibit larger increases in cerebellar activation also make higher motor gains in rehabilitation.13,14 Diffusion tensor imaging (DTI) has also revealed a close association between microstructural integrity of DTC and UE motor function. Individuals possessing higher DTC integrity have more favorable paretic UE motor status, regardless of residual microstructural integrity of the corticospinal tract (CST).15,16
Our recent phase I clinical trial of dentate nucleus DBS has indicated for the first time in humans the significance of targeting DTC in stroke. Chronic stroke survivors with moderate-to-severe UE motor impairment received at least 4 months of dentate nucleus DBS delivered in conjunction with UE rehabilitation. Findings demonstrated that DBS was safe and feasible to deliver and could elicit clinically meaningful improvement of UE motor impairment and functional ability scores in association with increased ipsilesional motor cortical metabolism measured by positron emission tomography. 17
Our phase I trial 17 however did not report the physiology of DTC and motor cortices. The present study sought to capitalize on this opportunity from the parent trial for direct investigation of DTC physiology and its real-time effect on motor cortical excitability, which otherwise remains elusive in humans due to difficulty to access the dentate nucleus. Cerebellar-cortical physiology has generally been studied using non-invasive approaches including transcranial electrical and transcranial magnetic stimulation (TMS) in humans. 18 In neurologically intact adults, transcranial cerebellar stimulation leads to inhibition of motor evoked potentials (MEPs) elicited from the contralateral motor cortex at 5 to 7 ms latency, known as cerebellar inhibition (CBI).19-21 CBI is believed to reflect Purkinje cell mediated inhibition of DTC output to the motor cortex. 21 However, preclinical studies have revealed DTC to be excitatory in nature.7,22,23
The present study sought to harmonize evidence through direct investigation of DTC physiology using dentate nucleus DBS and measurement of its effect on motor cortical MEPs using TMS. Pairing of invasive DBS and non-invasive TMS is novel in stroke. Conditioning DBS pulses were delivered to dentate nucleus through externalized leads a few milliseconds prior to TMS pulses given over the ipsilesional motor cortex. The expectation was that DBS would lead to facilitation (not inhibition) of MEPs at short-latency intervals, which would be consistent with the fast excitatory nature of DTC transmission revealed before in animal studies. 7 We also expected the degree of facilitation would vary with the level of motor impairment severity and microstructural integrity of CST and DTC. In vivo characterization of DTC physiology holds significance for design and refinement of dentate nucleus DBS for post-stroke motor recovery and offers an important biomarker of response to our own efforts to translate novel DBS treatments targeting the cerebellum.4,17,24,25
Methods
Participants
Participants were enrolled in a parent phase I clinical trial evaluating the safety and feasibility of dentate nucleus DBS in chronic stroke (clinicaltrials.gov NCT02835443). Eligibility was determined based on selection criteria for the parent trial. The parent trial was performed under an investigational device exemption granted by the United States Food and Drug Administration (FDA, #G150237). The trial was approved to enroll 12 individuals 25 to 75 years of age, who were 12 to 36 months post index, ischemic stroke affecting the middle cerebral artery territory. The trial was initially approved to only include severely impaired participants, but this requirement was later modified to also allow inclusion of moderate-to-severely impaired individuals. Severe and moderate-to-severe UE impairment was defined by UE Fugl-Meyer (UEFM) score ≤42 (out of best 66).26,27 Participants however still needed to possess sufficient motor ability to engage in rehabilitation (indicated by score of ≥1 on elbow flexion/extension or finger flexion/extension items of UEFM). Participants were also required to possess evidence of residual ipsilesional corticomotor physiology, that is, MEP+ status, defined by the presence of criterion-sized (≥100 µV) MEPs elicited using TMS in paretic forearm, wrist, or finger extensor muscles. 28 Participants were excluded for excessive UE spasticity/contracture (Modified Ashworth Scale = 4), significant cognitive impairment (Mini Mental State Examination score <24 out of 30), 29 hemorrhagic stroke, severe pain, loss of sensation or neglect, and any other major neurological or psychiatric condition. Participants were also excluded for surgical risks (eg, severe cardiovascular disorder) and contraindications to MRI or TMS (eg, seizure and pacemaker). 30 All research activities were approved by the Cleveland Clinic Institutional Review Board (16-498). Participants provided written informed consent in line with the Declaration of Helsinki.
Clinical Assessment of Motor Status
Motor status of the UE was assessed before implant surgery by an occupational therapist. UEFM scale was utilized as a measure of motor impairment. UEFM has excellent reliability, consistency, and validity.31-33 Volitional movement of the shoulder, elbow, forearm, wrist, and hand is tested in and out of synergies, and each movement is graded on a 3-point ordinal scale (0, cannot perform; 1, perform partially; and 2, perform fully) for a total maximum score of 66.
Unilateral Cerebellar DBS Implant
All participants underwent surgery for implantation of DBS leads in the dentate nucleus contralateral to the ipsilesional (stroke) hemisphere with computed tomography and MRI guidance. The intended target was output of the dentate nucleus as it projects to the superior cerebellar peduncle. The trajectory was planned using standard stereotactic techniques with entry from the occipital surface of cerebellum using a lateral-to-medial approach. 3-D intra-operative images were co-registered to preoperative plan to confirm or refine lead placement. The study used VerciseTM DBS system developed by Boston Scientific Corp. (Marlborough, MA, USA). Five participants were implanted with the Vercise Cartesia 8-contact directional lead that has a distal active tip, a proximal omnidirectional contact and 2 levels of 3 radially arranged directional contacts in between. The other subjects had a linear 8-contact lead that has 8 omnidirectional contacts. The lead was connected to a temporary percutaneous extension, which was externalized at the shoulder for 1 week, during which paired DBS and TMS experiments were conducted. The temporary percutaneous extension was removed, and proximal end of implanted DBS lead was connected to a new extension tunneled to the implantable pulse generator in the chest during a second surgery at end of 1 week.
Paired DBS and TMS Experiment
Paired DBS–TMS experiment was conducted during the week-long externalization period that immediately followed DBS implantation. As summarized in Figure 1A, DBS pulses were delivered using an external clinical neurostimulation device attached to the percutaneous extension at the shoulder (MEE2000, Nihon Kohden, Tokyo, Japan). TMS was delivered as single monophasic pulses to the ipsilesional motor cortex using a figure-of-8 coil with an inner loop diameter of 7 cm (Magstim 200 2 , Magstim, Inc., Whitland, Wales, UK). The coil was held tangentially to the head with the handle oriented 45° posterolateral to the interhemispheric fissure. Application of TMS was guided using MRI stereotaxy (Brainsight, Rogue Research Inc., Montreal, QC, Canada). TMS responses (MEPs) were collected from paretic finger extensor muscle—extensor digitorum communis (EDC). If MEPs could not be elicited in EDC given the severe/moderate-to-severe impairment status of the sample, a forearm/wrist flexor muscle was chosen. Surface electromyography (EMG) electrodes (Ag–AgCl, 8 mm diameter) were affixed in a belly-belly montage. Sampling rate was 4000 Hz and data was band-pass filtered online between 10 and 2000 Hz (PowerLab 4/35T, ADInstruments Inc., Colorado Springs, CO, USA). LabChart 8 was used to visualize data online and store data for offline processing (Figure 1).
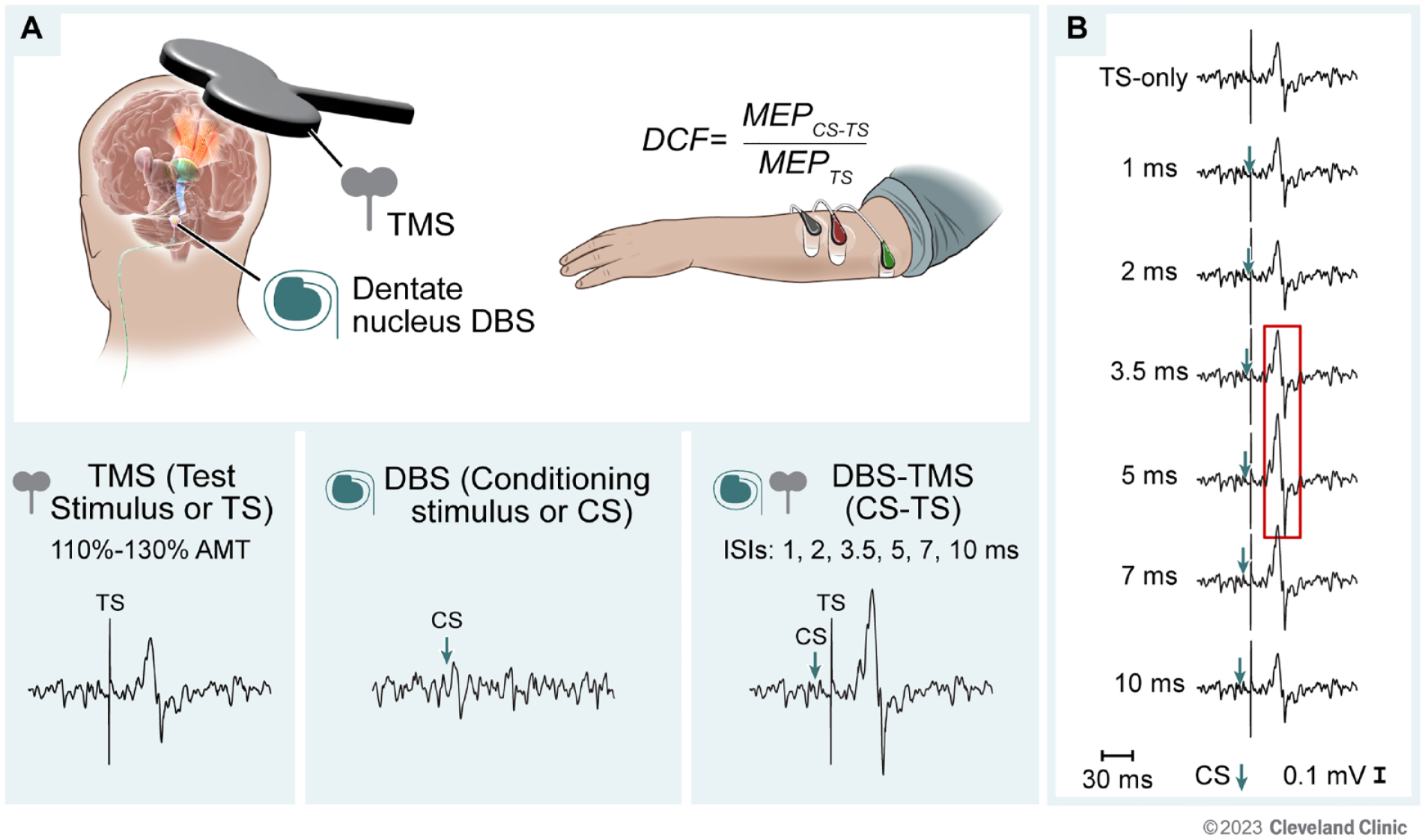
Experimental paradigm. (A) Experimental setup for paired deep brain stimulation (DBS) and transcranial magnetic stimulation (TMS). Direct stimulation of dentate nucleus was paired with TMS of the contralateral (stroke-affected or ipsilesional) motor cortex. Surface electromyography was collected from the paretic test muscle (extensor digitorum communis muscle shown in figure). TMS pulse given as test stimulus (TS), DBS pulse given as conditioning stimulus (CS), and paired CS-TS pulses at different inter-stimulus intervals (ISIs) were collected. Dentato-cortical facilitation (DCF) was calculated as the ratio of the motor evoked potential (MEP) amplitudes from paired CS-TS trials relative to average MEP from TS-only trials. (B) Schematic of hypothesized changes in MEP amplitude at each ISI. MEPs were expected to be facilitated at 3.5 and 5 ms ISIs.
TMS motor hotspot was identified as the site that consistently elicits the largest MEPs in contralateral test muscle at the lowest TMS intensity. The lowest TMS intensity required to produce criterion-sized MEPs (≥100 µV, peak-to-peak amplitude) in slightly contracted (20% ± 5% maximal voluntary contraction) test muscle in ≥6 of 10 trials was called the active motor threshold (AMT). 34 Participants received real-time visual feedback of ongoing EMG signal and required contraction level to improve consistency of volitional activity. Because severely impaired participants have smaller and inconsistent MEPs in paretic muscles, we used several safeguards to accurately identify MEPs. First, MEPs had to meet a size criterion of ≥100 µV amplitude (peak-to-peak) even in the presence of slight background muscle contraction (to increase likelihood of eliciting MEPs in paretic muscles in severely impaired participants). Second, MEPs had to be evident within a narrow post-stimulus time window of 15 to 60 ms, consistent with the latencies for testing of UE muscles. Third, we overlaid consecutive trials to confirm consistency of the onset and the shape of MEP waveforms, a control approach adopted for identifying smaller MEPs in severely impaired survivors.28,35 Participants were given frequent rest breaks (every 5 trials) to avoid fatigue-related confounds.
During the paired DBS and TMS experiment, conditioning DBS pulses were given to dentate nucleus just prior to TMS pulses to the ipsilesional motor cortex. To allow opportunity (room) for modulation of MEPs with conditioning DBS, supra-threshold TMS test stimulus (TS) intensity was identified using a stimulus-response curve. 36 TMS was delivered at intensities ranging from 90% to 150% AMT (in a randomized order) while MEPs were acquired in the test muscle (10 trials at each intensity). The curve was visualized online to ensure maximal MEPs were acquired in each participant. The TMS intensity that produced MEPs ~50% of size of the difference between the largest and the smallest MEPs was defined as TS. TS intensities ranged between 110% and 130% AMT. If high AMT values precluded collection of the stimulus-response curve, TS was set at 100% maximum device output. DBS conditioning stimuli (CS) consisted of single (n = 5 subjects) or double- or triple-pulse bursts (n = 4 subjects, intra-burst frequency 1000 Hz) delivered to the dentate nucleus. DBS lead contacts chosen to deliver CS were identified based on anatomic proximity to the dentate nucleus output. CS pulses were delivered at intensities of 4 to 8 mA with pulse width 90 to 200 µs.
Paired DBS and TMS was investigated over 10 experimental blocks. The paradigm consisted of delivering CS-TS pairs with CS preceding TS at varying interstimulus intervals (ISIs)—1, 2, 3.5, 5, 7, and 10 ms. Each experimental block was comprised of 6 CS-TS pairs (one pair per ISI), 5 TS pulses, and 3 CS pulses/bursts, delivered in a pseudorandomized order. A total of 60 CS-TS pairs (10 pairs per ISI), 50 TS trials, and 30 CS trials were collected in every participant. In 2 participants, however, only 7 experimental blocks could be collected including 42 CS-TS pairs (7 pairs per ISI), 56 TS trials, and 21 CS trials due to post-operative fatigue and drowsiness.
Peak-to-peak MEP amplitude for paired CS-TS, TS-only, and CS-only trials was extracted for analysis. For each ISI, size of conditioned MEPs collected from paired CS-TS trials was expressed relative to the average size of test MEP collected from TS-only trials. A ratio greater than 1 indicates MEPs are facilitated with CS-TS relative to TS alone, a phenomenon which we call DCF. A ratio smaller than 1 indicates MEPs are inhibited with CS-TS relative to TS alone (termed CBI in previous studies). 19 With the expectation that peak change in MEPs may occur at different ISIs across participants, we also identified the maximal effect at short latencies whether seen at 3.5 or 5 ms inter-stimulus intervals, called maximal DCF or DCFMAX. Due to the polyphasic nature of MEPs often seen in stroke, MEP area was extracted as a secondary outcome using a custom-written MATLAB script (The MathWorks, Inc., Natick, MA). Paired CS-TS, TS-only, and CS-only trials were rectified and averaged for analysis to increase signal-to-noise ratio.37-39 MEP onset was defined as the timepoint when the averaged rectified signal rose above 1SD of the mean background for 5 ms and MEP offset was defined as the timepoint that the signal fell below the same threshold.35,38 MEP area was calculated as the area under the curve between onset and offset, normalized to the background area of same duration to control for differences in contraction levels. 39 DCF based on MEP area was calculated for each ISI.
Imaging
All participants underwent a T1-weighted MRI at 3 Tesla (T; Prisma, Siemens Healthineers, Erlangen, Germany). A high angular resolution diffusion imaging (HARDI) scan was also acquired at 7T in 6 participants (Magnetom, Siemens Healthineers, Erlangen, Germany) and 3T in 3 participants who had contraindications to 7T MRI. Image acquisition included: T1-weighted magnetization prepared 2 rapid acquisition gradient echo (MPRAGE) with 0.75 mm isotropic voxels at 7T and 1 mm isotropic voxels at 3T. Lesions were manually segmented from the 3T MRI using FSL (version 6.0, fsl.fmrib.ox.ac.uk) 40 and BrainLab software (BrainLab AG, Munich, Germany) and volumes were calculated. HARDI acquisition using multi-slice echo planar imaging included 8 b = 0 volumes and 71 non-collinear diffusion-weighting gradients with b = 1000 s/mm2 (1.3 mm isotropic voxels at 7T and 2 mm isotropic voxels at 3T). Data acquired at 7T was corrected for susceptibility-induced distortions using FSL topup41,42 and eddy current-induced distortions and subject movements using FSL eddy. 43 Data acquired at 3T was pre-processed using iterative motion correction that included updating diffusion gradient directions.44,45
Probabilistic tractography was used to define DTC and CST pathways, incorporating both local and global information to improve accuracy, and specialized partial differential equation solvers for fast results. 46 DTC tract was defined between ventrolateral posterior (VLp) nucleus of thalamus and motor cortical regions. VLp seed was defined as a disc (radius = 2 mm, thickness = 0.75 mm for 7T and 1 mm for 3T MRIs) manually localized based on thalamic segmentation of T1-weighted MRI offered in FreeSurfer (version 7.1.1, surfer.nmr.mgh.harvard.edu). 47 The segment of DTC from dentate nucleus to VLp could not be reconstructed due to lack of volumes spanning the cerebellum in 3 participants. Motor cortical regions were defined using the Human Motor Area Template, 48 including primary motor, dorsal premotor, supplementary motor, and primary somatosensory cortices. Primary, premotor and supplementary motor areas were also studied collectively as a single motor region. CSTs were defined across the posterior limb of the internal capsule (PLIC), labeled using Johns Hopkins University’s ICBM-DTI-81 white-matter atlas in FSL. 49 Targets were defined as inferior-most slice of PLIC and seeds were defined 11 mm superior to the target. Cortical, VLp, and CST regions were non-linearly warped to align with DTI before performing tractography. Structural integrity for DTC and CST was quantified using metrics known to reliably reflect the microstructural status of white matter, including average fractional anisotropy (FA), transverse diffusivity, mean diffusivity, and longitudinal diffusivity.50,51 Decreased FA values and increased directional diffusivities are indicative of post-stroke white matter damage and degeneration. 52
Statistical Analysis
Normality of data was assessed using the Shapiro–Wilks test. Based on the a priori hypothesis that dentate nucleus DBS would lead to facilitation of MEPs at short latencies (3.5 and 5 ms) via DTC, we used 1-sample t-tests to compare DCF and DCFMAX values against 1 [representing no difference between conditioned (CS-TS) MEPs vs test (TS) MEPs]. We did not perform a repeated-measures ANOVA with dedicated post hoc testing due to the exploratory nature of this experiment and the understanding that we would have insufficient power with the small sample size and variable MEPs from severe and moderate-to-severely impaired stroke survivors. To determine the impact of motor impairment severity, we used independent t-tests to compare DCF and DCFMAX values across participants with preserved versus non-preserved distal movement. Preservation of distal movement was defined as UEFM wrist and hand sub-score ≥4 out of 24, whereas sub-score less than that indicated non-preserved movement. We also evaluated exploratory bivariate correlation between white matter structural integrity of DTC and CST with DCF (and other TMS metrics) using Pearson’s coefficient. All tests were 2-tailed with alpha-level set at 0.05 (uncorrected due to exploratory nature of the study).
Results
A total of 12 participants were enrolled in the parent phase I trial. However, this experiment included 9 of the 12 participants, 57.7 ± 7.5 years of age (mean ± SD, 2 females) who had experienced an index stroke 28.2 ± 8.0 months earlier. The first 2 participants of the parent trial could not undergo this paired DBS and TMS experiment because safety and feasibility of DTC DBS was still being determined and leads were not externalized. Another participant could not undergo this experiment due to the need to shorten externalization considering COVID-19 pandemic-related restrictions. Clinico-demographic characteristics are presented in Table 1 and lesion locations are displayed in Supplemental Figure 1. Participants had moderate-to-severe UE motor impairment characterized by UEFM score of 23.4 ± 7.2 (range 13-38). Five participants had preserved distal movement (UEFM mean ± SEM 28.2 ± 2.5) while 4 did not (UEFM 17.5 ± 1.6, t(7) = −3.36, P = .012). Post-operative imaging demonstrated DBS contacts were localized within the region of the dentate nucleus (reported previously). 25
Participant Demographics and Clinical Information.
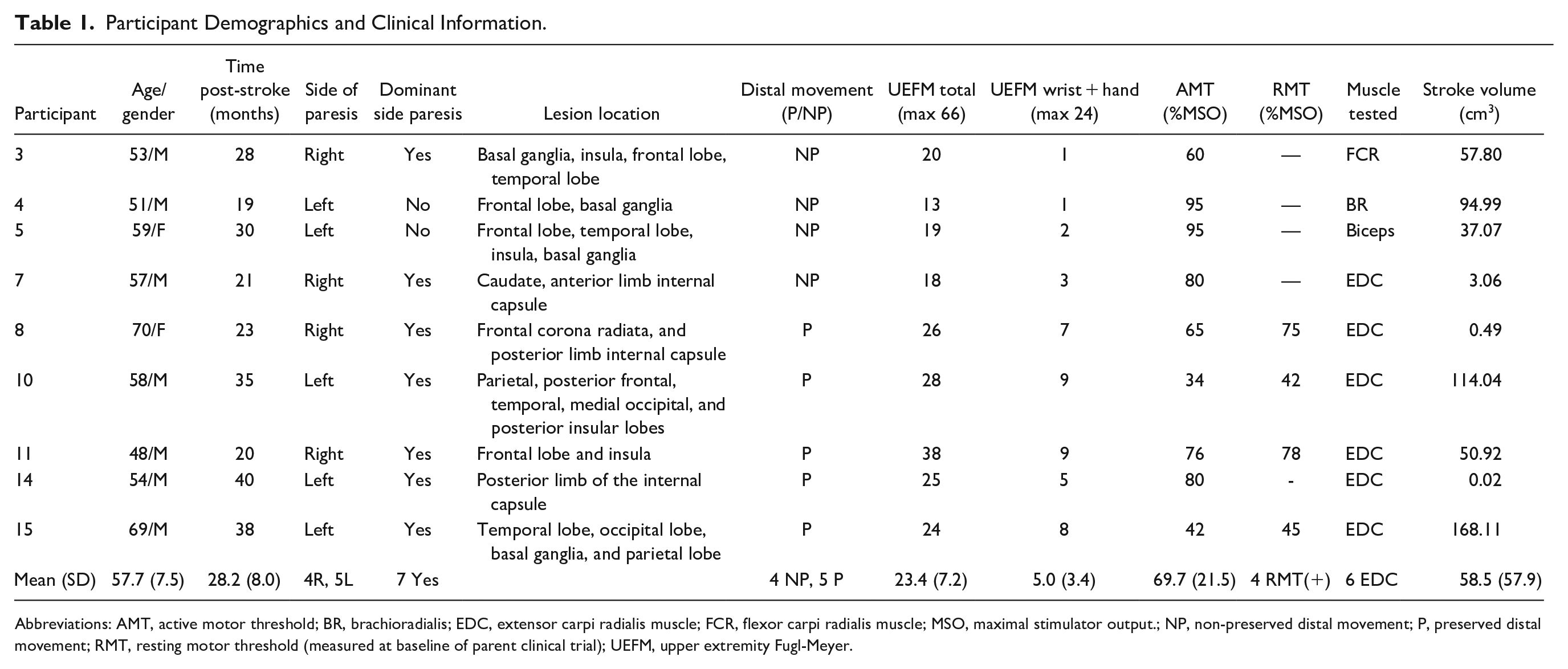
Abbreviations: AMT, active motor threshold; BR, brachioradialis; EDC, extensor carpi radialis muscle; FCR, flexor carpi radialis muscle; MSO, maximal stimulator output.; NP, non-preserved distal movement; P, preserved distal movement; RMT, resting motor threshold (measured at baseline of parent clinical trial); UEFM, upper extremity Fugl-Meyer.
All participants underwent the paired DBS–TMS experiment without any adverse effect. The test muscle was paretic EDC in 6 participants, and flexor carpi radialis, brachioradialis, or biceps brachii in 1 participant each. Ipsilesional AMT (mean ± SD) was 69.7% ± 21.5%, ranging from 34% to 95% maximum device output. Test or TS MEP size was 0.54 ± 0.58 mV with a range from 0.08 to 1.85 mV. Participants with preserved distal movement had larger TS MEPs than participants without distal preservation, though the difference between the 2 subgroups was not significant (mean ± SEM, 0.83 ± 0.29 mV vs 0.16 ± 0.03 mV; t(7) = −2.06, P = .078).
Conditioning DBS pulses led to facilitation of MEPs at short-latency intervals. Conditioned MEP (CS-TS) to test MEP (TS) ratio, that is, DCF, exceeded 1 at 3.5 and 5 ms ISIs (1.09 ± 0.04, t(8) = 2.33, P = .049 and 1.11 ± 0.04, t(8) = 2.88, P = .021; Figure 2A). The maximal ratio found at 3.5 or 5 ms ISI (termed DCFMAX) also exceeded one (1.16 ± 0.03, t(8) = 4.74, P = .001; Figure 2C). However, DCF calculated from MEP area was not significantly different than one (all P > .09).
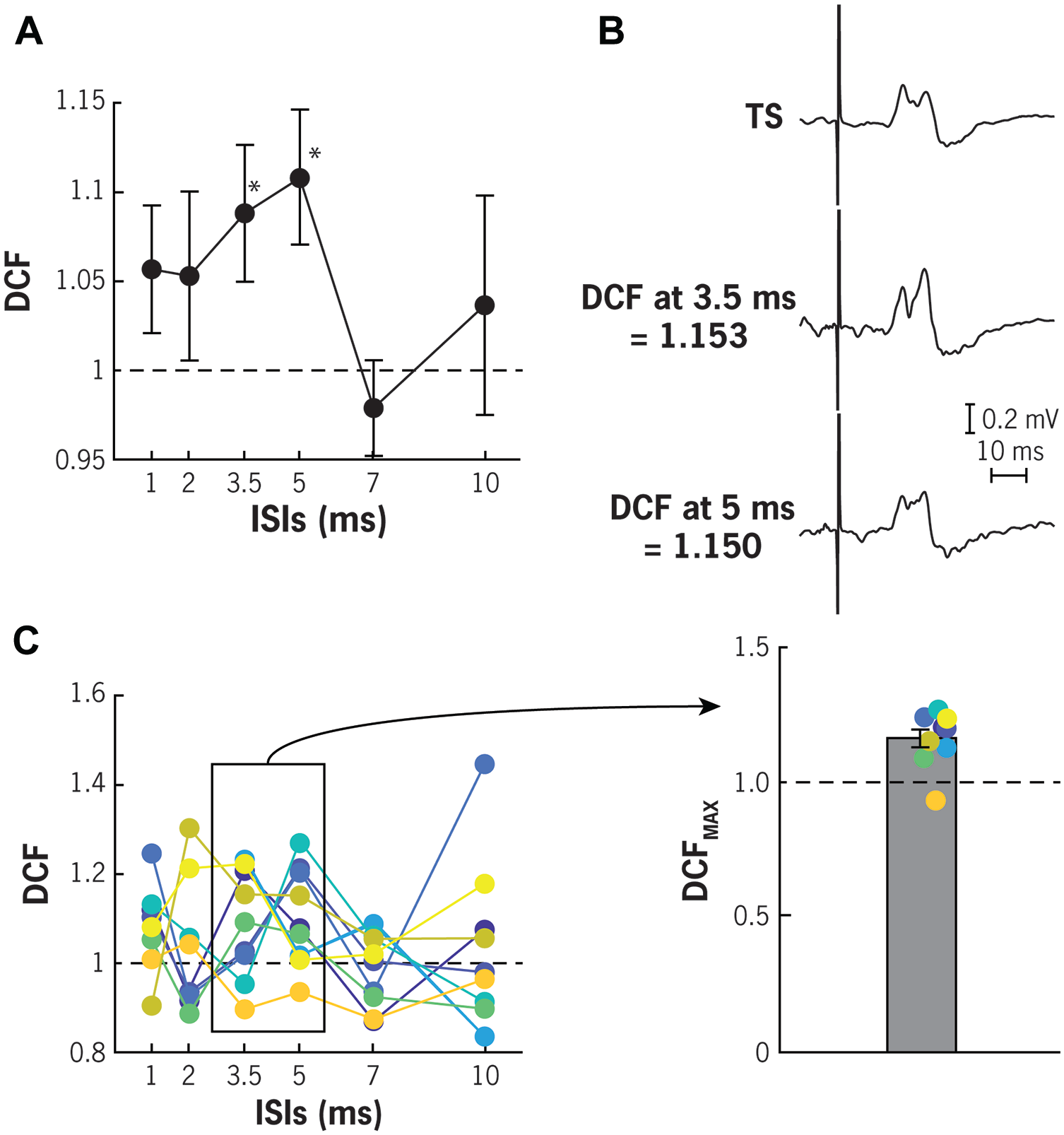
Dentato-cortical facilitation (DCF) across different inter-stimulus intervals (ISIs). (A) Potentiation of motor evoked potentials (MEPs) was observed when conditioning DBS pulses were given directly to the dentate nucleus 3.5 and 5 ms before TMS was given over the motor cortex (* denotes P < .05 compared to 1). (B) Example average MEP traces from a participant showing increase in MEP amplitudes with DBS conditioning at 3.5 and 5 ms ISIs in comparison to unconditioned TMS-only or test stimulus (TS) MEPs. (C) Maximal DCF or DCFMAX extracted from 3.5 to 5 ms ISIs also shows potentiation of MEPs (individual data shown in color).
Conditioning DBS pulses led to greater facilitation of MEPs in more severely impaired participants. Participants without distal preservation had DCFMAX values that exceeded 1 (1.19 ± 0.02, t(3) = 8.51, P = .003), whereas participants with distal preservation had DCFMAX values that failed to exceed one (1.14 ± 0.06, t(4) = 2.29, P = .084; Figure 3A). Differences between participants with and without distal preservation were not significant with regards to DCFMAX. Since test MEP amplitudes may influence the degree of conditioning seen in paired pulse paradigms, 21 we also examined the relationship between TS MEP amplitude and DCFMAX. There was no significant association between TS MEP amplitude and DCFMAX (Spearman rank correlation, ρ = 0.12, P = .78, Figure 3B), which indicates that evidence of higher DCFMAX in more severely impaired participants cannot be ascribed to their smaller MEPs.
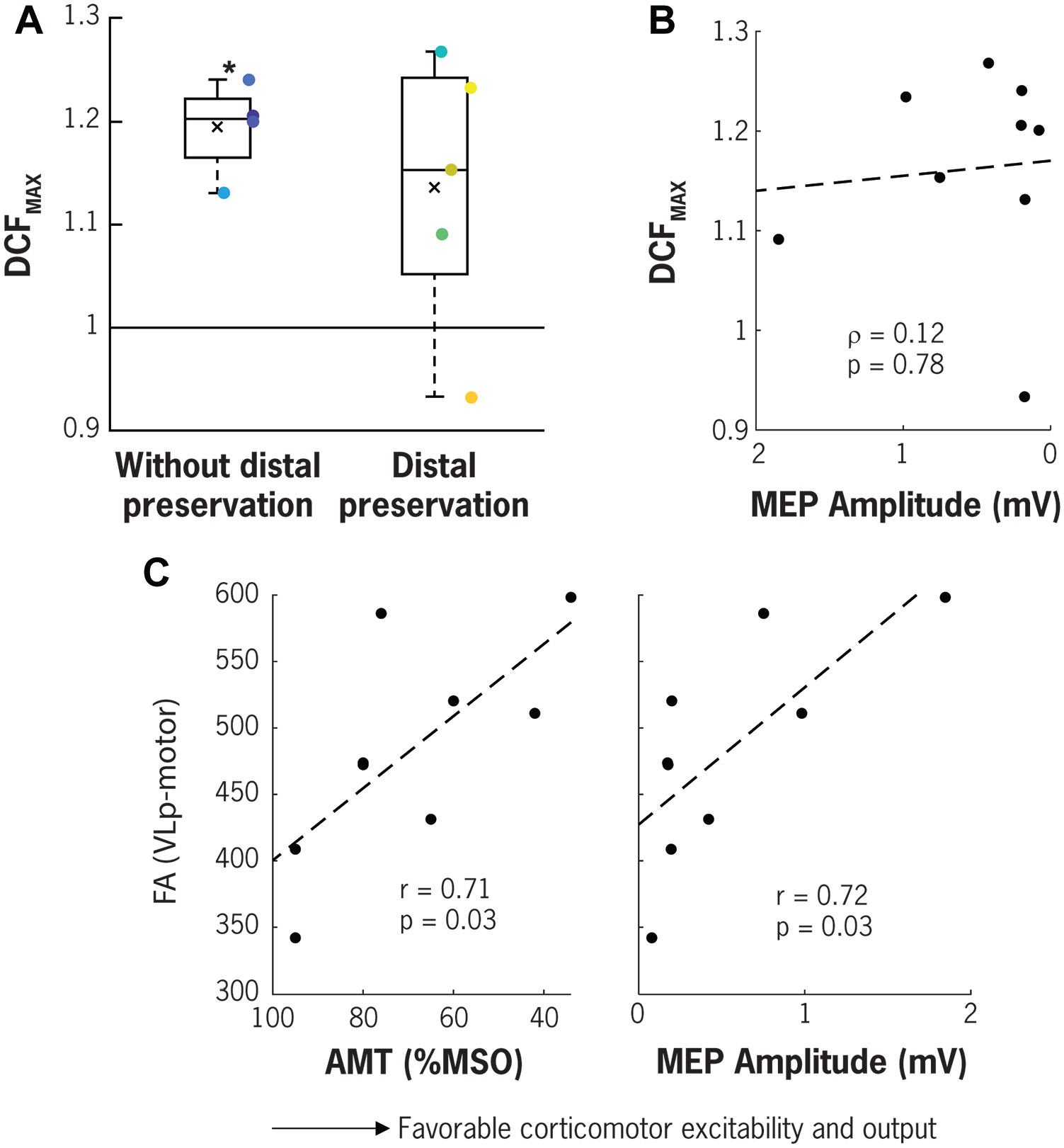
Influence of impairment severity and white matter tract structural integrity on dentato-cortical facilitation (DCF), corticomotor excitability and output. (A) Participants lacking criterion distal movement show significant potentiation of MEPs with DBS conditioning of dentate nucleus, whereas participants with preserved distal movement do not (* indicate P < .05 when compared to 1). (B) TMS-only MEP amplitudes collected at 110% to 130% AMT (or 100% MSO) do not correlate with DCFMAX. (C) Participants with more favorable corticomotor excitability and output indexed by lower AMT and higher MEP amplitude (collected at 110%-130% AMT or 100% MSO) also have higher structural integrity of DTC indexed by FA of white matter tract from VLp to combined motor cortical region.
Structural integrity of DTC pathways was associated with higher corticomotor excitability and output. Individuals who had higher FA of thalamo-cortical portion of the DTC (ie, between VLp and primary motor, dorsal premotor, supplementary motor, somatosensory cortices, and combined motor cortical regions) also had lower AMT values and larger MEP amplitudes (r value range −0.73 to −0.62, p value range 0.025 to 0.072 and r value range 0.60 to 0.72, p value range 0.027 to 0.085, respectively). Figure 3C shows the bivariate correlation of FA measured between VLp and the combined motor cortical regions and the ipsilesional AMT values and MEP size. Structural integrity of residual CST pathways was also associated with higher corticomotor excitability and output. Individuals who had higher FA at PLIC also had lower AMT and larger MEP amplitudes (r = −0.66 and 0.85, P = .05 and .004, respectively). There was no correlation however between microstructural integrity of DTC and DTC physiology indexed as DCF (all P > .07).
Discussion
The present study reports the findings of the first-in-human in vivo experiment investigating physiologic influence of DTC on motor cortical excitability and output assayed using a combination of DBS and TMS in stroke. Participants had moderate-to-severe post-stroke UE impairment which offered an opportunity to characterize the influence of the degree of impairment severity and white matter microstructural integrity on DTC physiology. Our findings reveal that: (1) DBS of dentate nucleus leads to facilitation of MEPs from the contralateral motor cortex at short latency intervals (3.5 and 5 ms), a phenomenon we call DTC facilitation or DCF, consistent with the fast excitatory nature of DTC transmission revealed before in preclinical studies;7,23 (2) participants with more severe distal impairment show higher DCF, indicating potentially a greater facilitatory cerebellar role in the presence of severe injury; and (3) higher microstructural integrity of thalamocortical portion of DTC is associated with higher corticomotor excitability but not DCF, suggesting thalamocortical pathway integrity is likely critical for corticomotor physiology and that dentato-thalamic portion of DTC may make higher contribution to DCF. Our findings of acute DCF revealed in the context of a cross-sectional experiment are in line with our phase I trial findings of higher ipsilesional motor cortical metabolism following chronic DBS. 17 Increased ipsilesional motor cortical excitability with brief DBS given to dentate nucleus may predict its potential to elicit motor cortical reorganization following long-term DBS therapy and may offer more opportunities for refined DTC targeting to promote UE motor function in moderate/severely impaired stroke survivors.
Our findings of DTC physiology revealed using invasive targeting also have the potential to inform the underpinnings of CBI tested using non-invasive stimulation. 19 Direct targeting of dentate nucleus in this study led to facilitation of MEPs (DCF) at short latency intervals (3.5 and 5 ms) in contrast to inhibition of MEPs (CBI) seen with transcranial targeting at 5 to 7 ms latencies,19-21 which supports the theory that CBI results from an indirect activation of DTC by way of superficial inhibitory Purkinje-cell neurons 21 (Figure 4). A previous study of transcranial electrical stimulation has also reported a facilitatory effect of the cerebellum on motor cortical output. 53 Facilitation of MEPs was observed at 3 ms ISI, followed by inhibition (CBI) at 5 to 7 ms ISIs in able-bodied individuals. The facilitatory effect however required careful titration of CS and TS intensities, specific stimulation parameters and unique TMS coil orientation. The later inhibitory (CBI) effect was much more consistent like in other CBI studies. 18 Our results of early facilitation (DCF) witnessed at 3.5 to 5 ms reflect the natural fast excitatory pace of DTC transmission revealed before in preclinical studies,7,10,11,22 without confounding off-target effects of transcranial targeting.
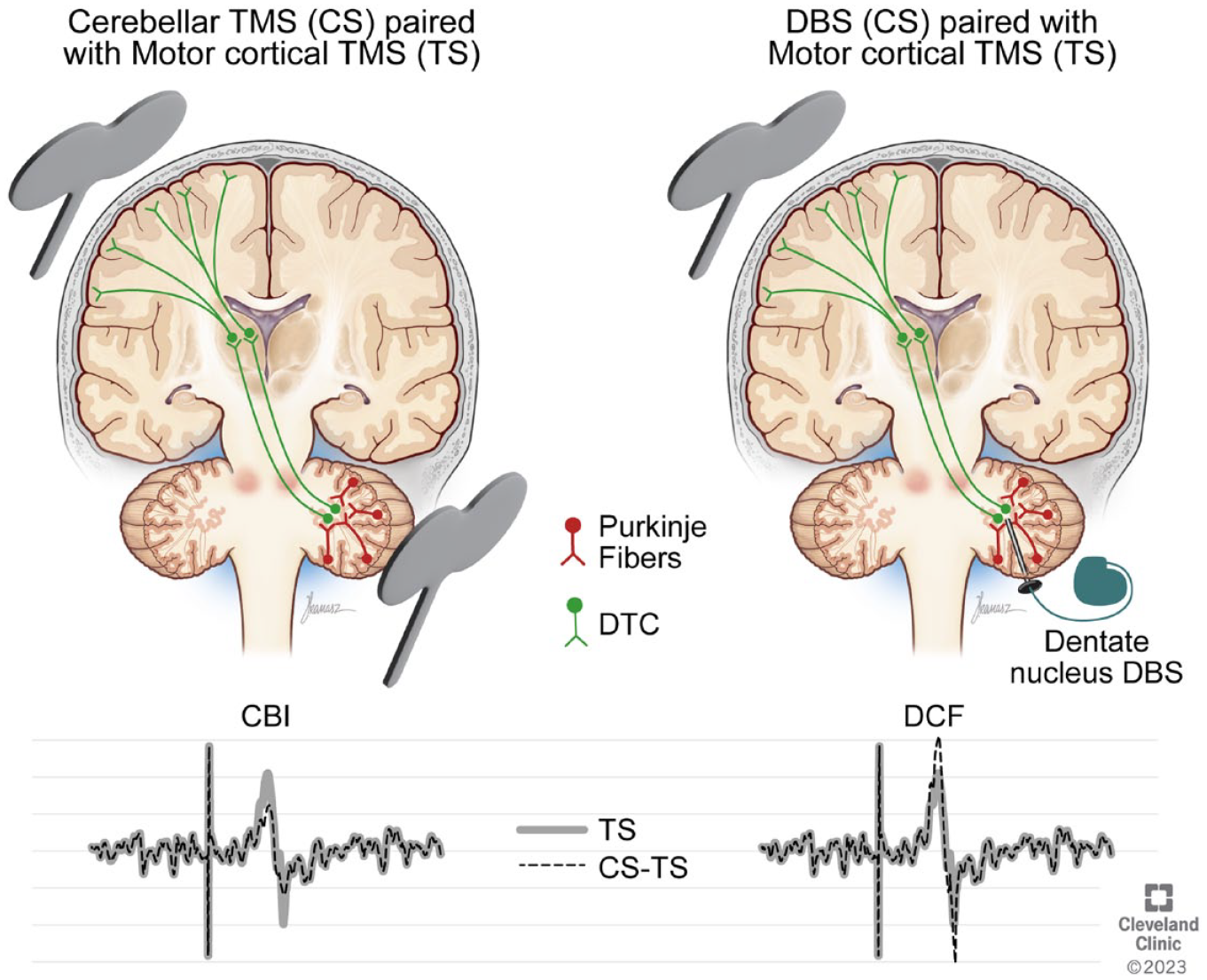
Putative effects of indirect cerebellar stimulation achieved with transcranial targeting (left) versus direct dentate nucleus targeting with deep brain stimulation (right) on motor cortical excitability. Indirect stimulation may activate dentato-thalamo-cortical (DTC) pathways via superficial inhibitory Purkinje neurons, which results in a net inhibitory effect on motor cortical output evident as reduced motor evoked potentials (MEPs). Dentate nucleus stimulation may directly activate the excitatory DTC pathways, resulting in facilitation of MEPs. Dashed line depicts MEP from paired stimulation (CS-TS), while thick gray line depicts MEP from motor cortical stimulation (TS).
Our investigation in the context of stroke also highlights the clinical value of DCF. We found DCF is higher among stroke survivors with more severe distal impairment. Increased physiologic response from DTC may help compensate for severe ipsilesional loss in a typical middle cerebral artery stroke. Cerebellar output can remain chronically hypoactive due to crossed cerebellar diaschisis after stroke 54 which is associated with poor motor function. 55 A chronically hypoactive state may have more “room” for gain with exogenous (DBS) stimuli explaining higher DCF seen in more impaired participants. On the other hand, less impaired individuals may have better cerebellar output to begin with, making it less likely for them to exhibit acute facilitation in this cross-sectional experiment. Results of the parent clinical trial (which demonstrate that less impaired participants make higher motor gains with chronic DBS) supports this position. 17 Taken together, it is conceivable that acute DCF may be higher in more impaired individuals due to there being more room for MEP gain, but stronger cerebellar-cortical connectivity in less impaired individuals may render DTC pathways responsive to chronic DBS entrainment over months for higher functional gains. Longitudinal study of how DTC physiology evolves with chronic DBS in association with motor gains will further support our position.
Our imaging results indicate that microstructural integrity of DTC is associated with corticomotor excitability and output but not DCF. Relationship between DTC integrity and motor cortical activity in stroke has been witnessed before 16 and our findings underscore the importance of DTC for corticomotor physiology. Characterization of DTC integrity along with CST integrity may strengthen potential to predict motor gains in stroke. Nevertheless, structural integrity of DTC was not related to DCF. Of note, structural integrity was only analyzed for the thalamo-cortical portion of DTC (due to missing data in 3 participants). The dentato-thalamic portion of DTC likely makes greater contribution to cerebellar-cortical physiology, which remains to be investigated, whereas the thalamo-cortical portion may have higher relevance for corticomotor physiology. Studying both neurophysiology and structural integrity of DTC can provide complementary information, given that physiology of pathways may be disproportionately affected compared to structure.
The extent of DCF seen in our stroke sample with direct dentate nucleus stimulation (7%-27%) is smaller than extent of CBI seen in neurologically unimpaired adults with indirect transcranial targeting (~40%-50%). 19 Diminished cerebellar-motor cortical physiology may be a feature of stroke. In a published conference abstract, authors note that CBI from the ipsilesional motor cortex is reduced compared to CBI from the unaffected, contralesional motor cortex in persons with stroke. 56 Stroke may limit DTC excitability and corticomotor physiology and thereby extent of DCF albeit direct targeting may obviate off-target effects and serve as robust resource to facilitate motor cortex. A head-to-head comparison of indirect (TMS–TMS) and direct (DBS–TMS) methodologies is compelling but could not be performed due to the need for special regulatory approvals required for all procedures in the parent trial and on account of safety issues surrounding TMS application directly over cerebellar DBS implant. 30
We were limited by the sample size of the parent phase I trial that was granted approval to only enroll 12 participants for investigation of safety and feasibility of a novel dentate nucleus DBS approach. We were also constrained by the impairment status of the sample which was approved at first to include only severely impaired participants due to justification of greater need of testing a new implant. A severely impaired sample meant MEPs had to be collected in slightly contracted state of the muscle, which can reduce cerebellar output (and thereby DCF). 21 We had no other option but to test proximal muscles in the first few patients. Proximal muscles have smaller, more variable MEPs than intrinsic hand muscles which are common to test for evaluation of CBI. 18 Smaller MEPs in some participants may have introduced variability. Differences in TS MEP amplitude between participants with and without preserved distal movement (albeit non-significant) also may have influenced DCF. Non-invasive CBI is more easily elicited with smaller TS MEP size (0.5 mV vs 2 mV). 21 Although there was no association between TS MEP size and DCF in our study, confounding influence of test MEP size and thereby differences in baseline impairment status cannot be ignored. Lingering effects of anesthesia during externalization and likely “stun effect” from surgery also may have led to diminished cerebellar and corticomotor excitability (6 out of 9 participants had smaller MEPs or higher AMTs during externalization period compared to their pre-surgical baseline). Unknown sex-based differences in DCF could also have influenced the results in our small sample (n = 2 female).
Conclusions
Our in vivo investigation of dentate nucleus DBS paired with TMS in humans reveals for the first time direct, excitatory effects of DTC on motor cortical output at early latencies, called DCF. Conducted in the context of stroke, this study demonstrates the value of DCF among severely impaired survivors indicating greater cerebellar role in the presence of severe injury. Overall, our early-phase findings have potential to create opportunities for novel neuromodulation approaches that promote UE motor function in stroke.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683231219265 – Supplemental material for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors
Supplemental material, sj-pdf-1-nnr-10.1177_15459683231219265 for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors by Xin Li, Kenneth B. Baker, Kyle O’Laughlin, Jacqueline Chen, Olivia Hogue, Andre G. Machado and Ela B. Plow in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-2-nnr-10.1177_15459683231219265 – Supplemental material for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors
Supplemental material, sj-pdf-2-nnr-10.1177_15459683231219265 for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors by Xin Li, Kenneth B. Baker, Kyle O’Laughlin, Jacqueline Chen, Olivia Hogue, Andre G. Machado and Ela B. Plow in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-3-nnr-10.1177_15459683231219265 – Supplemental material for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors
Supplemental material, sj-pdf-3-nnr-10.1177_15459683231219265 for Paired DBS and TMS Reveals Dentato-Cortical Facilitation Underlying Upper Extremity Movement in Chronic Stroke Survivors by Xin Li, Kenneth B. Baker, Kyle O’Laughlin, Jacqueline Chen, Olivia Hogue, Andre G. Machado and Ela B. Plow in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank Ms. Alexandria Wyant for enrollment, coordination, and management and Drs. Robert Chen, Kelsey Baker, and David Cunningham for technical input. We would also like to thank Dr. Yin-Liang Lin for help in early data collection.
Author Contributions
Xin Li: Data curation; Formal analysis; Investigation; Visualization; Writing—original draft; Writing—review & editing. Kenneth B. Baker: Conceptualization; Funding acquisition; Investigation; Methodology; Writing—original draft; Writing—review & editing. Kyle O’Laughlin: Data curation; Investigation; Methodology; Writing—review & editing. Jacqueline Chen: Formal analysis; Methodology; Writing—original draft; Writing—review & editing. Olivia Hogue: Formal analysis; Writing—review & editing. Andre G. Machado: Conceptualization; Funding acquisition; Writing—original draft; Writing—review & editing. Ela B. Plow: Conceptualization; Data curation; Funding acquisition; Investigation; Methodology; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Andre G. Machado is a consultant and has intellectual property licensed to Enspire DBS. Kenneth B. Baker is a consultant to Enspire DBS. Enspire DBS funded part of the clinical trial. The remaining authors report no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health BRAIN initiative [grant number UH3NS100543] and Enspire DBS. EP’s role was also supported by National Institutes of Health [grant number R01HD098073]. All funding sources did not have a role in study design, in the collection, analysis, and interpretation of data, in the writing of the report, and in the decision to submit the article for publication.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
