Abstract
Background:
Previous studies have found that high-frequency repetitive transcranial magnetic stimulation (rTMS) of the cerebellar hemisphere could improve swallowing function, but whether intermittent theta burst stimulation (iTBS), which has similar excitatory effect and higher efficiency, can also improve swallowing function for dysphagia after stroke remains unclear.
Objective:
This trial aimed to explore the efficacy and safety of bilateral cerebellar transcranial magnetic stimulation with iTBS for dysphagia after stroke.
Methods:
Seventy patients with dysphagia after stroke were divided into 2 treatment groups: true bilateral cerebellar iTBS and sham bilateral cerebellar iTBS. The true iTBS group underwent ten 100% resting motor threshold (RMT) iTBS sessions for 2 weeks. In the sham iTBS group, the parameters were the same except that the figure-eight coil was perpendicular to the skull. Both groups received traditional swallowing rehabilitation treatment 5 times a week for 2 weeks. Swallowing function was assessed with the Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS), Penetration/Aspiration Scale (PAS), Standardized Swallowing Assessment (SSA), and Functional Oral Intake Scale (FOIS) at baseline, 2 weeks after the intervention, and at 4 weeks of follow-up.
Results:
There were significant time and group interaction effects in both multi-factorial adjusted and unadjusted FEDSS, PAS, SSA, and FOIS score (P < .001). In the pairwise comparison of the swallowing parameters among the 2 groups, the FEDSS, PAS, SSA, and FOIS scores at 2 weeks and 4 weeks showed a significantly higher improvement in the iTBS simulation group than sham group (P < .05). In both the true iTBS and sham iTBS stimulation groups, all FEDSS, PAS, SSA, and FOIS scores were significantly improved over time (P < .001).
Conclusions:
The present study suggested that as a more efficient TMS stimulation mode, iTBS could efficiently improve swallowing function by stimulating the bilateral cerebellar hemisphere. In addition, 100% resting motor threshold bilateral cerebellar iTBS is a relatively safe treatment.
Clinical Trial Registration:
Effect analysis of repeated transcranial magnetic stimulation of cerebellar on dysphagia after stroke. www.chictr.org.cn. Identifier: ChiCTR2100042092.
Keywords
Introduction
Dysphagia is one of the common complications post-stroke, and the incidence of dysphagia in acute stroke patients is 37%–78%. 1 Dysphagia causes severe complications such as dehydration, aspiration, pneumonia, and malnutrition. 2 Despite dysphagia care efforts, traditional swallowing training is not supported by strong evidence showing it can improve swallowing function or decrease the associated complications.3,4
The cerebellum communicates with the brainstem with 3 peduncles and it modulates the coordination and balance of movement and postural control. 5 Sensory from the oral, pharynx, and esophagus is transmitted to the nucleus tractus solitarius, then communicates with motor nuclei nucleus ambiguus. 6 This network of interneurons constituted the central pattern generator (CPG) of swallowing in the medulla oblongata. 7 After the cerebrum initiates swallowing, the cerebellum plays an important role in the precise and coordinated control of swallowing. 8 The cerebellum regulates swallowing mainly through the cerebellar cortical pathway, involving the dentate pathway and fastigial pathway. Dentate pathway: The cerebellar cortex communicates with the bilateral deep dentate nuclei of the cerebellum, and afferent fibers are transmitted to the contralateral primary motor cortex via thalamic synaptic connections. 9 Fastigial pathway: The cerebellar connects with the fastigial nuclei, and transmitted to the ipsilateral primary motor cortex via CPG in medulla oblongata. 10 Stimulation of the cerebellum may modulate the excitability of the bilateral cortex by dentate and fastigial pathway, thus regulating swallowing function. Bernston and Martner reported the stimulation of fastigial nucleus in cats elicited oral behaviors critical for grooming and chewing and feeding behavior could be better organized.11,12 Besides, in human functional brain imaging studies, the cerebellum, thalamus, cingulate gyrus and all areas of both primary sensorimotor cortex are significantly activated, when performing swallowing-related tasks. 13 The activation of the bilateral dentate nucleus, the declive and culmen, and right fastigial nucleus has also been identified during coughing task in healthy adults. 14 The cerebellar dentate nucleus, fastigial nucleus, and cerebello-cortical pathway may play an important role in the regulation of swallowing function.
In 2001, Jayasekeran first discovered that single-pulse TMS stimulation of the cerebellum and electrodes in the pharyngeal could record motor evoked potential (MEP). More importantly, it was found that paired cerebellar pulses could increase the amplitude of cortical MEP. 15 The cerebellum could facilitate cortical excitability. Some previous studies have shown that cerebellar 10 Hz rTMS improved MEP amplitude in the swallowing cortex 16 and completely reversed the suppressive MEP of a “virtual lesion” in health. 17 Zhong et al. 18 performed three-arm study comparing the ipsilateral hemisphere, contralateral hemisphere, and cerebellum in stroke patients with dysphagia and revealed that cerebellar rTMS improve swallowing function just as the hemisphere. The aforementioned studies suggest that the cerebellum may be a new target for non-invasive neural modulation of swallowing disorders.
Positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) showed bilateral cerebellar hemisphere activation during swallowing.19,20 Moreover, Sasegbon found that both unilateral and bilateral cerebellar rTMS activated the bilateral cerebral cortex, and bilateral rTMS was significantly more effective than unilateral rTMS. 21 Bilateral cerebellar stimulation may be a more effective treatment.
In recent years, with the rapid development and improvement of repetitive transcranial magnetic stimulation (rTMS), this technology has been widely used for various aspects of mental psychology, stroke rehabilitation, and other applications, and it has achieved remarkable results. 22 Theta burst stimulation (TBS) is a new stimulation model of TMS that simulates endogenous oscillating θ frequencies in the hippocampus during work and learning and it can produce rapid and lasting effects with a weaker stimulus and a shorter time, including continuous TBS (cTBS) and intermittent TBS (iTBS). 23 cTBS reduces excitability, while iTBS improves the excitability of the stimulated cortex, and the effect can persist for 60 min. 24 TBS improves dysphagia, mainly focusing on the deglutition motor area in the cerebral hemisphere. fMRI studies have reported that iTBS could activate the cortex of the suprahyoid muscle in healthy people by facilitating cortical excitability.25–28 Cosentino found that 5 consecutive days of iTBS over the right swallowing motor cortex improved the dysphagia outcome and severity scale (DOSS) scores at the 1 and 3 months follow-ups in elderly individuals. 29 Compared with traditional rTMS, TBS is more effective in improving cerebral function. 30 In a single treatment of treatment-resistant depression in adults, iTBS takes only 3 minutes, while standard 10 Hz rTMS takes 37.5 minutes. 31 In our study, iTBS requiring only 190 s for one standard run. However, high-frequency rTMS usually takes 10–20 minutes. 32 iTBS can induce longer cerebral cortex excitability with lower stimulation intensity and in a shorter time than rTMS, thus achieving ideal therapeutic effects. 33
However, there is no relevant research on cerebellar iTBS for dysphagia post-stroke. Therefore, we hypothesized that bilateral cerebellar iTBS stimulation could improve swallowing function like high-frequency rTMS does. This paper aimed to study the efficacy and safety of bilateral cerebellar iTBS for dysphagia after stroke to provide an objective basis for the clinical promotion of this treatment approach.
Materials and Methods
Experimental Design and Participants
This study was designed as a two-arm, randomized, double-blind, sham-controlled clinical trial. The 2 study arms were bilateral cerebellar true iTBS and sham iTBS. Participants were recruited from the Rehabilitation Department of Yue Bei People’s Hospital in Guangdong Province, China, from January 2021 to June 2021. The inclusion criteria were as follows: (1) Stroke patients with first onset, including unilateral infarction or hemorrhage, confirmed by computed tomography (CT) or magnetic resonance imaging (MRI); (2) dysphagia measured by fiberoptic endoscopic evaluation of swallowing (FEES) or a video fluoroscopic swallowing study (VFSS); (3) duration after stroke 1 week to 6 months; (4) age 18 to 85, male or female; (5) medically stable; (6) without dementia (Mini-Mental State Examination >23), 34 able to cooperate with various examinations and rehabilitation training; and (7) consent obtained from the patient and their family, and informed consent was signed. Exclusion criteria included the following: (1) dysphagia not caused by stroke; (2) acute stroke with hemorrhage conversion; (3) contraindications for rTMS, such as metal stents or pacemakers installed in the heart, metal implants in the skull, epilepsy, cochlear implants, etc.; and (4) a malignant tumor. All subjects signed the informed consent form before enrollment. The trial was approved by the ethics committee of Yue Bei People’s Hospital. This clinical study follows the Consolidated Standards of Reporting Trials (CONSORT) guidelines. 35
Randomization and Masking
The participants were randomly allocated (1:1) to groups receiving bilateral cerebellum stimulation, either true iTBS or sham iTBS. Randomization was performed by a staff member of Yuebei People’s Hospital who did not participate in the implementation or evaluation of the trial using a pseudorandom number generated by a computer. Opaque, sealed envelopes were used to mask the randomization tables and they contained a paper labeled with a random identification number and the assignment code of the participant. The speech therapist performing the intervention was aware of the treatment condition, but the participants and outcome evaluators were masked to the treatment condition. We instructed the participants to avoid discussing their group assignment with the staff or the other participants until the end of the experiment.
Intervention
These 2 groups of patients received 10 true or sham iTBS sessions, 5 times a week for 2 weeks,32,36 performed by the same experienced speech therapist. Both groups received the same amount of traditional dysphagia therapy, such as oropharyngeal strength training, sensory stimulation training, Masako and Mendelson maneuvers, Shaker exercises, vocal cord training, breathing training, and cough protection training, for 30 minutes once a day, 5 times a week for 2 weeks after the iTBS. These exercises were performed by another experienced speech therapist unaware of their group assignment.
The magnetic stimulator used for the iTBS was a MagPro CCY-I stimulator (YIRUIDE Company, Wuhan, China) with a diameter of 9 cm figure-eight coil. Due to the absence of pharyngeal catheter electrodes, stimulation hot spots cannot be precisely located in this study. However, according to previous studies, we localized stimulation sites by measuring the distance 4.3 cm to lateral and 2.4 cm below the inion.15,16 Although not precise enough, the stimulation range of the figure-eight coil is about 2 to 3 cm2, 37 which could cover the hot spots area with a high probability. To determine the stimulus intensity, we measured the MEP of the hand and used 100% RMT of the hand as the stimulus intensity for this study. 38 RMT was measured using single-pulse TMS. The coil targeted the M1 region of the hemisphere by moving slightly within 1 cm anteriorly and 6 cm laterally of the Cz point to obtain the maximum MEP recording. RMT was measured in the abductor pollicis brevis muscle on the side of the undamaged cortical hemisphere. RMT was defined as the minimum stimulus intensity required to elicit 5 responses of 50 μV or more in 10 consecutive trials.
True iTBS was given as follows: The patient maintained a comfortable lying or sitting position. The stimulus mode of iTBS consisted of 3 pulses of stimulation delivered at 50 Hz and repeated at 5 Hz, in which a 2 s train of TBS was repeated every 10 s for a total of 190 s (600 pulses in total). (Figure 1). The stimulation intensity was 100% RMT. The stimulation site was the pharyngeal motor representation of the cerebellum (4.3 cm to lateral and 2.4 cm below the inion). 16 The left and right sides were each stimulated with 600 pulses, first on the right side and then on the left side (Figure 2-true). The stimulation coil was hand-fixed (190 s for 1 side), due to lack of neuronavigation and the metal arm is not strong enough to fix coil.
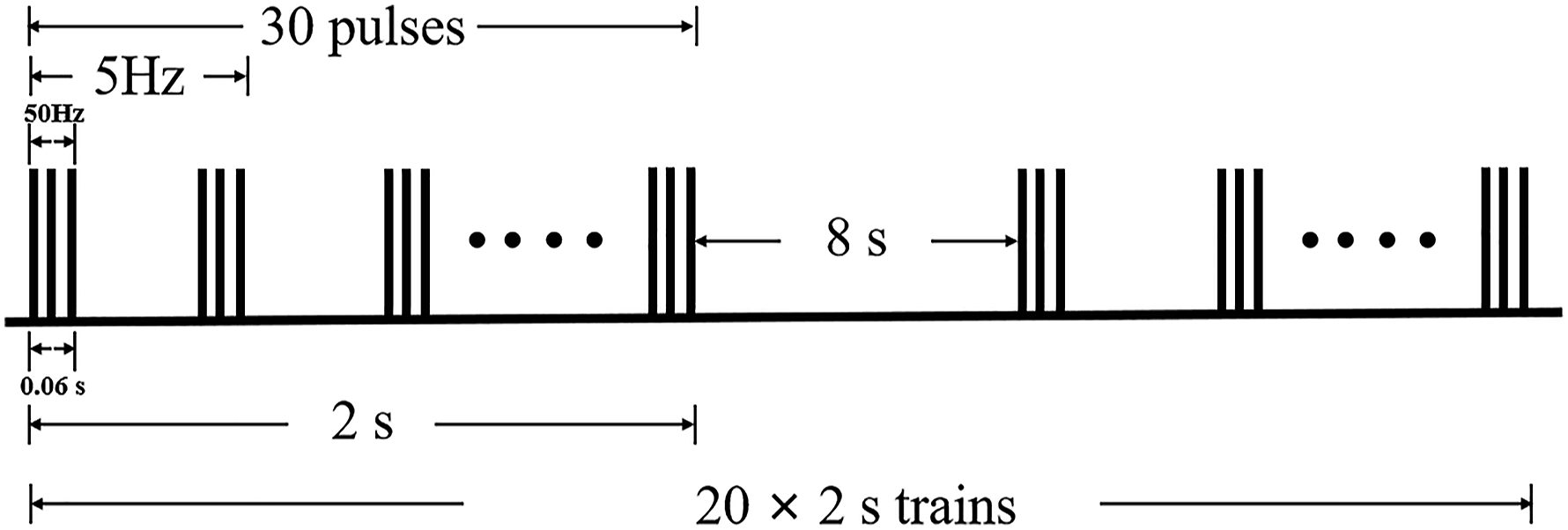
iTBS pulse schematic. A 2 s train of TBS was repeated every 10 s for a total of 190 s (600 pulses in total).
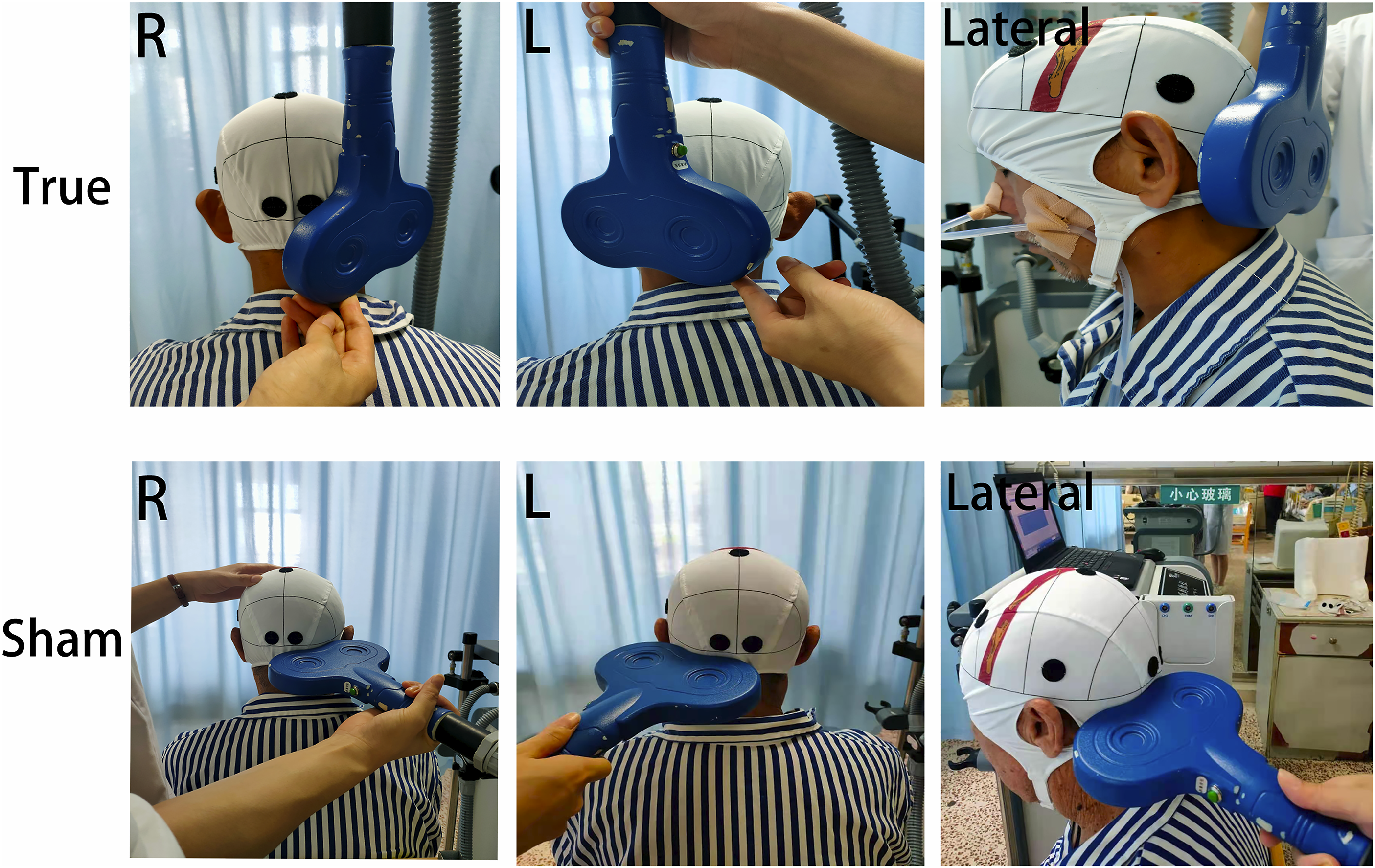
Shows that the figure-eight coil was tangentially close to the cerebellum stimulation point in the true iTBS group and perpendicular to the cerebellum in the sham group. R: Stimulating the right cerebellum. L: Stimulating the left cerebellum.Lateral: lateral plan.
Sham iTBS was given: the equipment, stimulation site, treatment time, and frequency were all the same as the iTBS group, but the probe was placed perpendicular to the patient’s skull so that the signal could not pass through the skull to act on the brain, so it produced the identical “click” sound as active treatment, without any therapeutic effect 39 (Figure 2-sham).
Outcome Measurements
All included patients were measured at 3 different times: before (baseline) and after treatment (2 weeks) and at the follow-up (4 weeks after treatment). The primary outcome was the Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS), measured by the same attending physician; secondary outcomes included the other dysphagia rating scales, such as the Water Swallow Test (WST), Penetration-Aspiration Scale (PAS), Standardized Swallowing Assess-ment (SSA), Functional Oral Intake Scale (FOIS), and adverse events.
Fiberoptic Endoscopic Dysphagia Severity Scale
The FEDSS is a scoring scale based on the results of the flexible endoscopic evaluation of swallowing (FEES) examination. Under flexible endoscopy, the patient’s salivary secretion was observed. Then, the patient ate 3 kinds of food with different textures, namely, soft solid foods, liquids, or pudding. The FEDSS scale is divided into 1 to 6 points, with the best score of 1 and the worst score of 6. The scores were based on the risk of foods with varying dietary consistency and salivary penetration or aspiration observed on endoscopy. 40
Penetration-Aspiration Scale
The severity of dysphagia was scored using an 8-subscale, the Penetration/inhalation scale (PAS). This scale is widely used as a semiquantitative assessment of penetration and aspiration for endoscopic or radiological measurements, with higher scores indicating more severe injuries. 41
Standardized Swallowing Assessment
The SSA consisted of 3 parts: The first part was a clinical examination, which is divided into 8 to 23 points, including consciousness; head, neck, and trunk postural control; breathing patterns; lip and soft palate movement; laryngeal function; pharyngeal reflex; and spontaneous cough. In the second part, with a total score of 6–11 points, the patient was instructed to swallow 5 mL of water 3 times, and then observed to see whether there was water within the mouth, repeated swallowing, coughing, wheezing, etc. In the third part, the total score is 5–12 points. If the above 2 parts are not abnormal, the patient is allowed to swallow 60 mL of water, observe the time required for swallowing all the water, and observe whether there is coughing, wheezing and aspiration during or after swallowing. The score of this scale is 19–46 points, and the higher the score is, the worse the swallowing function. 42
Functional Oral Intake Scale
The FOIS is graded in 7 levels according to safety and efficiency: Level 1: Could not eat by mouth. Level 2: Tube-dependent feeding, minimal attempt to ingest food or fluids. Level 3: Tube-dependent feeding, with mouth feeding of a single texture of food or liquid. Level 4: Single-texture food eaten entirely orally. Level 5: Complete oral intake of a variety of textures, but it requires special preparation or compensation. Level 6: Complete oral feeding requires no special preparation, but there are special food restrictions. Level 7: Complete oral feeding with no restrictions. 43
Sample Size
Based on a pilot study of bilateral cerebellum stimulation, we observed that the mean improvement in FEDSS was 1.21 in the experimental group and .42 in the control, with a standard deviation of 1.02. In this study, for a 5% type I error level with 80% power and an allocation ratio of 1:1, the sample size was estimated to be at least 28 individuals for each group. Considering 20% loss at the 1-month follow-up, 34 individuals were included in each group.
Statistical Analysis
All data were analyzed with SPSS 25.0 (SPSS Inc., Chicago, IL, USA). The mean ± standard deviation was used for the quantitative data as descriptive statistics. Proportions were used for the qualitative data. The Shapiro–Wilk test was used to evaluate whether the variables were normally distributed. Age and SSA were normally distributed at baseline, but SSA was not at posttreatment or follow-up in the study. Student’s t-tests were used for comparisons between groups at baseline. The other variables were not normally distributed. Nonparametric Mann–Whitney tests were used at baseline. Qualitative data were tested by chi-square tests. Statistical analysis of the scores in each test was performed with the generalized estimation equation (GEE) with time as the within-subject factor, the treatment condition (true and sham iTBS) as the between-subject measure, time and treatment condition as interaction effects. Further pairwise comparisons were performed, with P values corrected for Bonferroni. A post hoc stroke type and clinical stage subgroup analysis was performed to explore the consistency of the results. To control for confounding factors, baseline factors such as stroke type, stroke site, stroke affected hemisphere, clinical stage, ADL, and MMSE were used as covariates and corrected. P < .05 was considered significant for all statistical analyses.
Results
Ninety patients were assessed for eligibility, 16 did not meet the inclusion criteria, and 4 refused to participate. Seventy patients were randomized into either the “iTBS” (n = 35) or “Sham” (n = 35) groups. 2 patients in the iTBs group and 4 in the sham group dropped out during the 4-week follow-up. Three of the 6 patients were unwilling to return to the hospital for review despite the provision of transport subsidy, 2 of them in sham group because of discomfort from FESS examination and the other in iTBS group for unknown reasons. Three patients could not be contacted by phone, message, Email, and WeChat (1 in true group, 2 in sham group). The remaining participants (n = 64) were treated as intended and completed the study (Figure 3).
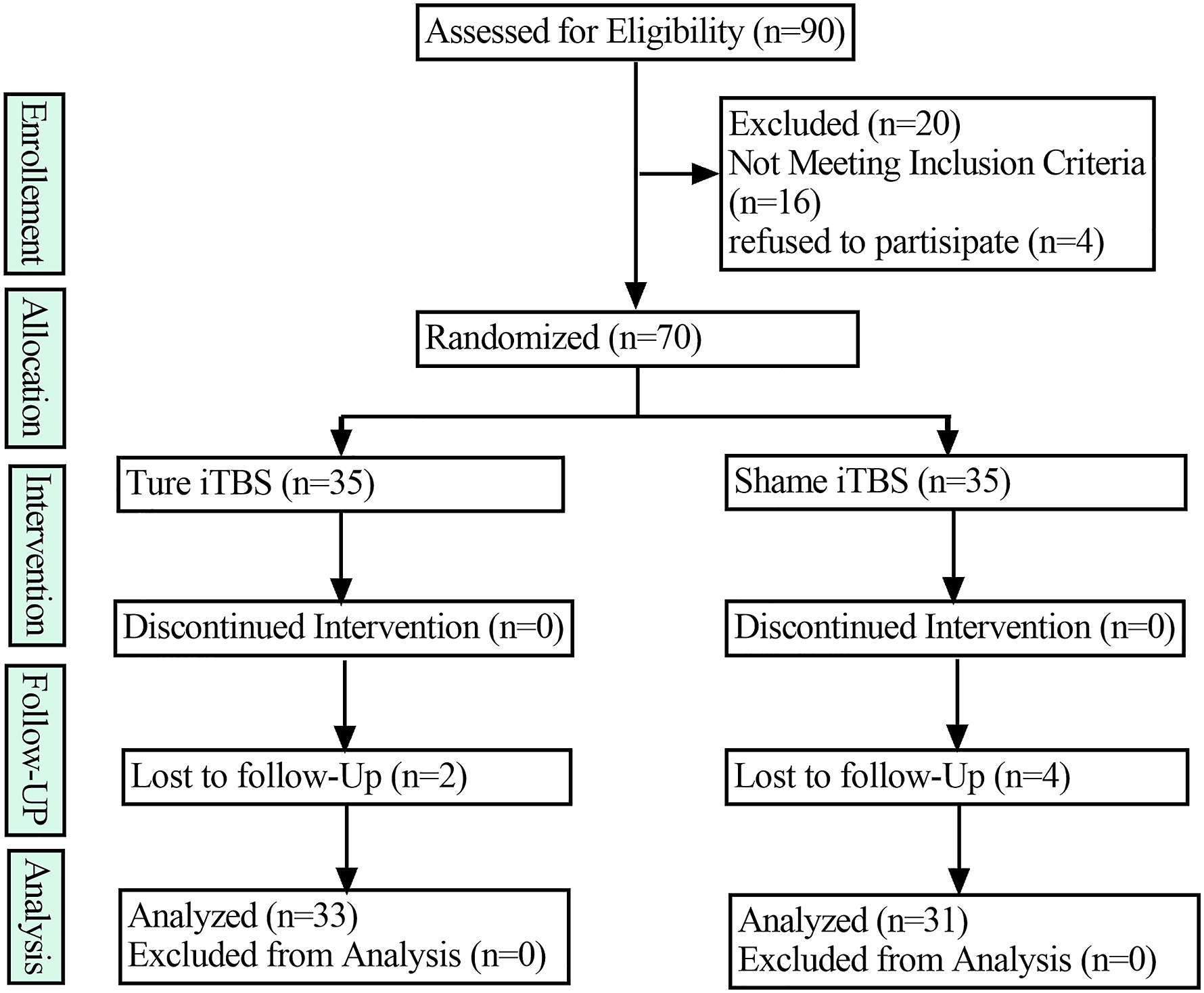
Participant flow diagram.
In the true iTBS group (average age 63.42 ± 10.35, 11 males and 22 females) and sham iTBS group (average age 65.9 ± 11.42, 7 men and 24 women), there were no significant differences in the distribution of sex, age, type of stroke, affected hemisphere, duration after stroke onset, site of stroke, drug using, ADL (Activities of Daily Living), Mini-Mental State Examination (MMSE) scores and clinical staging of stroke. The clinical staging are defined as acute stage within 3 weeks, subacute stage from 3 weeks to 3 months, chronic stage more than 3 months. 44 For the swallowing function assessment at baseline, the FOIS, WST, FEDSS, PAS, and SSA scores showed no significant difference between the 2 groups (see Table 1).
Characteristics of the Patients at Baseline.
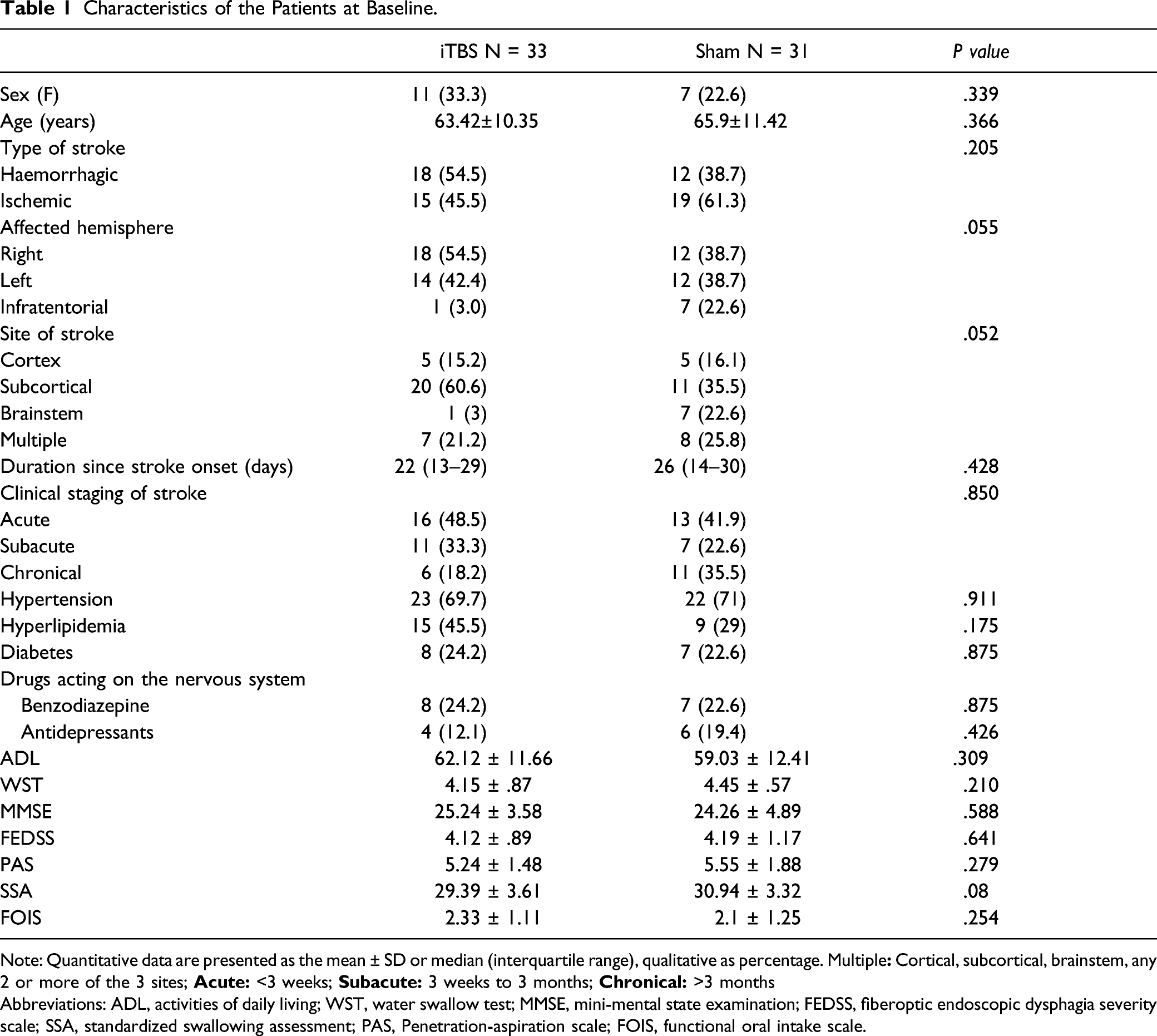
Note: Quantitative data are presented as the mean ± SD or median (interquartile range), qualitative as percentage. Multiple
Abbreviations: ADL, activities of daily living; WST, water swallow test; MMSE, mini-mental state examination; FEDSS, fiberoptic endoscopic dysphagia severity scale; SSA, standardized swallowing assessment; PAS, Penetration-aspiration scale; FOIS, functional oral intake scale.
In the one-way unadjusted GEE analysis, all FEDSS, PAS, SSA, and FOIS scores in both the true iTBS and sham iTBS stimulation groups significantly improved over time (P < .001). (Figure 4, Table 2). Adding prespecified baseline characteristics as covariates to adjust, all primary and secondary outcome remained improved in the iTBS and sham groups (P < .001) (Table 2). The effect of iTBS was consistent between adjusted and unadjusted analysis.
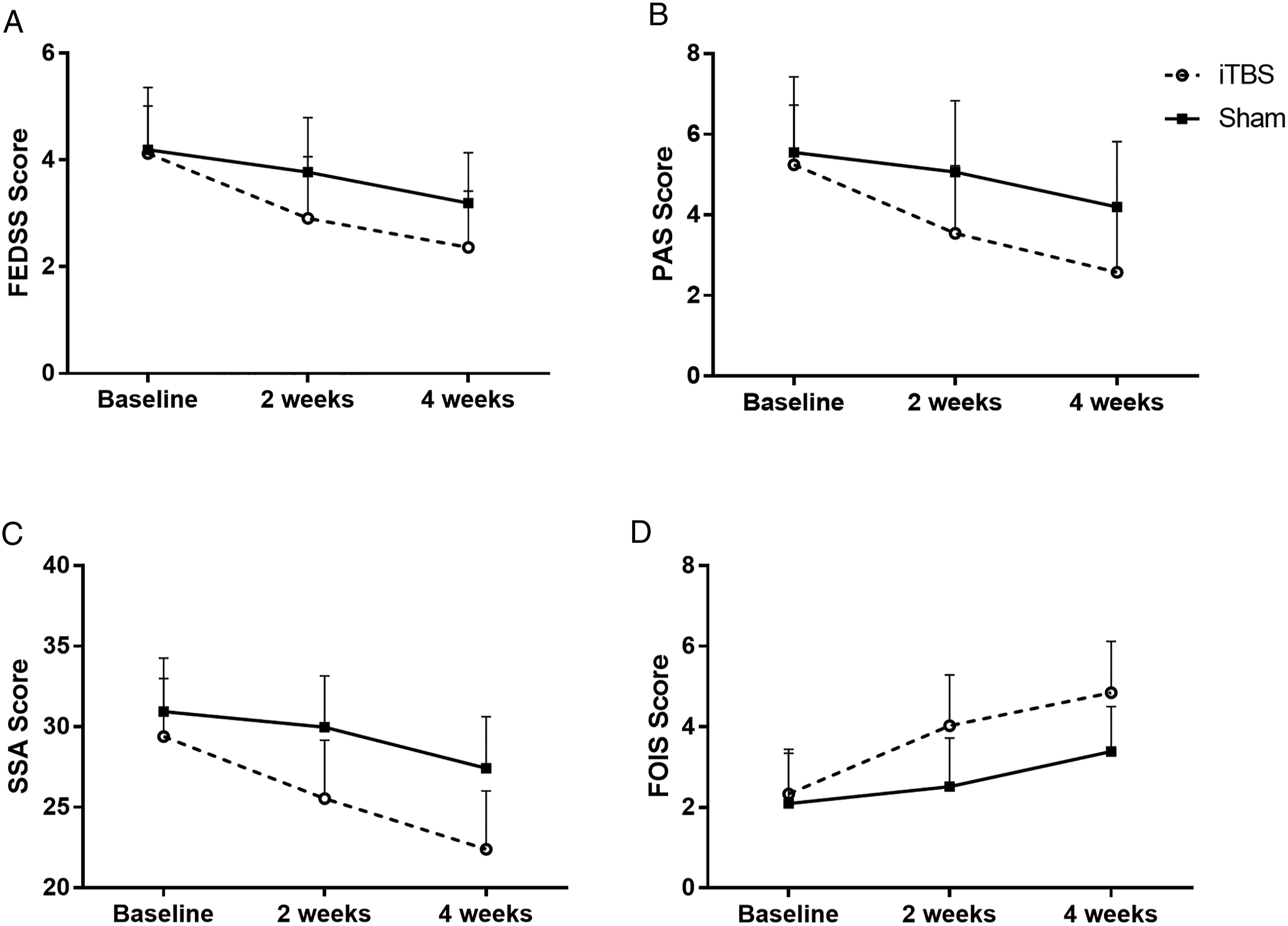
The mean FEDSS (A), PAS (B), SSA (C), and FOIS (D) scores at the 3 evaluation points in the true iTBS and sham groups. The data are described as the mean ± SD. Both groups showed significant improvements over time (P < .05). The iTBS group improved more than the sham group at the 2-week treatment and 4-week follow-up (P < .05).
Main Effect and Interaction Effect of Swallowing Function in iTBS and Shame Group.
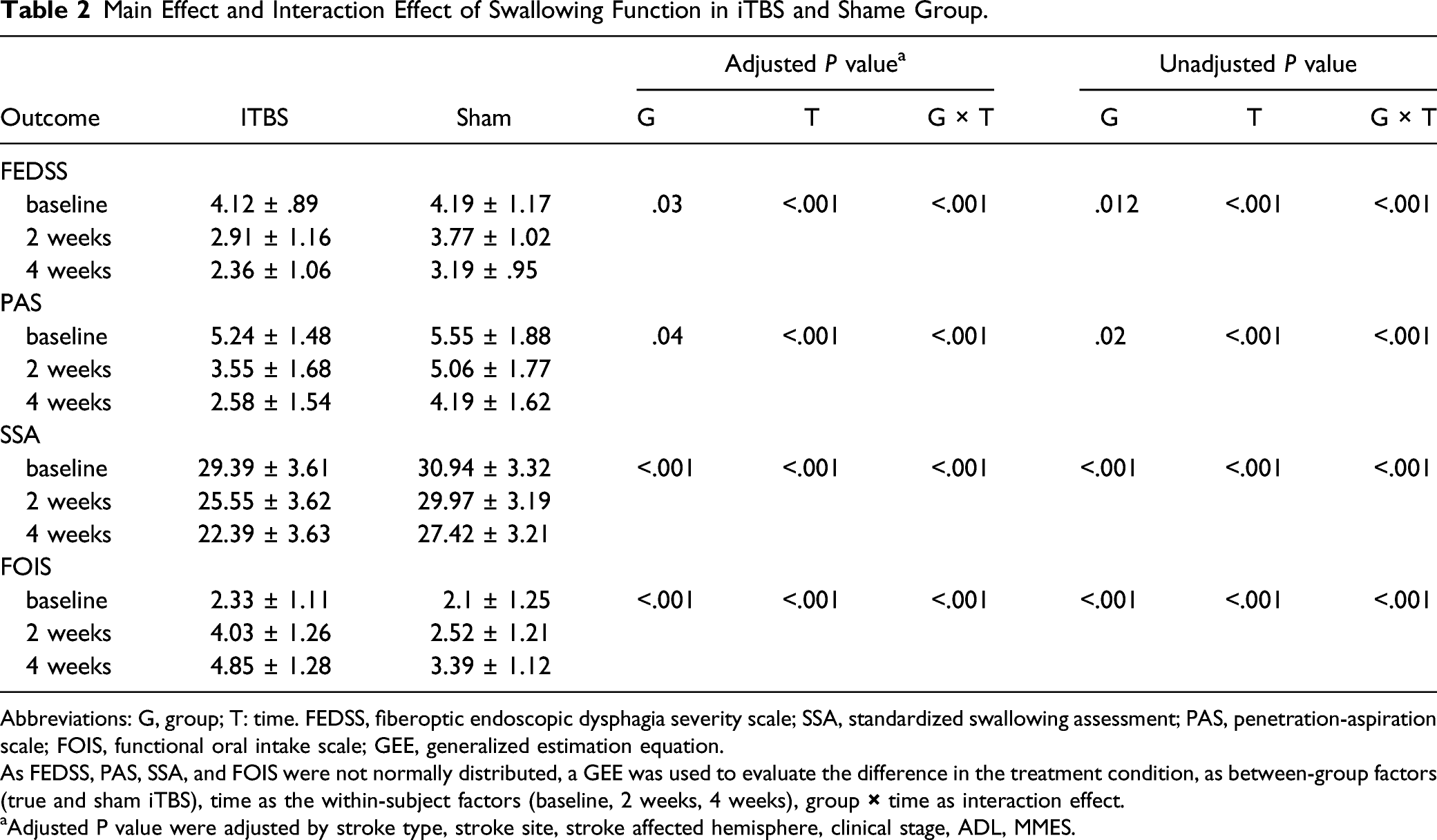
Abbreviations: G, group; T: time. FEDSS, fiberoptic endoscopic dysphagia severity scale; SSA, standardized swallowing assessment; PAS, penetration-aspiration scale; FOIS, functional oral intake scale; GEE, generalized estimation equation.
As FEDSS, PAS, SSA, and FOIS were not normally distributed, a GEE was used to evaluate the difference in the treatment condition, as between-group factors (true and sham iTBS), time as the within-subject factors (baseline, 2 weeks, 4 weeks), group
Adjusted P value were adjusted by stroke type, stroke site, stroke affected hemisphere, clinical stage, ADL, MMSE.
There were significant time and group interaction effects in both unadjusted and adjusted analysis for all FEDSS, PAS, SSA, and FOIS scores (P < .001) (Table 2). Further pairwise comparisons were made. For FEDSS score, iTBS reduced .72 points more than sham after 2 weeks (P = .02), and .69 points after 4 weeks (P = .03) in multi-factor adjusted. The similar improvement was seen in PAS, SSA, and FOIS scores (P < .001) (Table 3). In both iBTS and Sham groups, compared to baseline, FEDSS score improved after 2 weeks treatment (P < .001), and further improvement after 4 weeks of treatment, compared to 2 weeks (P < .001). The PAS, SSA, and FOIS scores in both iTBS and sham group were also significantly different, 2 weeks better than baseline, 4 weeks better than 2 weeks (P < .001) (Figure 4).
Comparison Between iTBS and Sham at 3 Measurement Time.
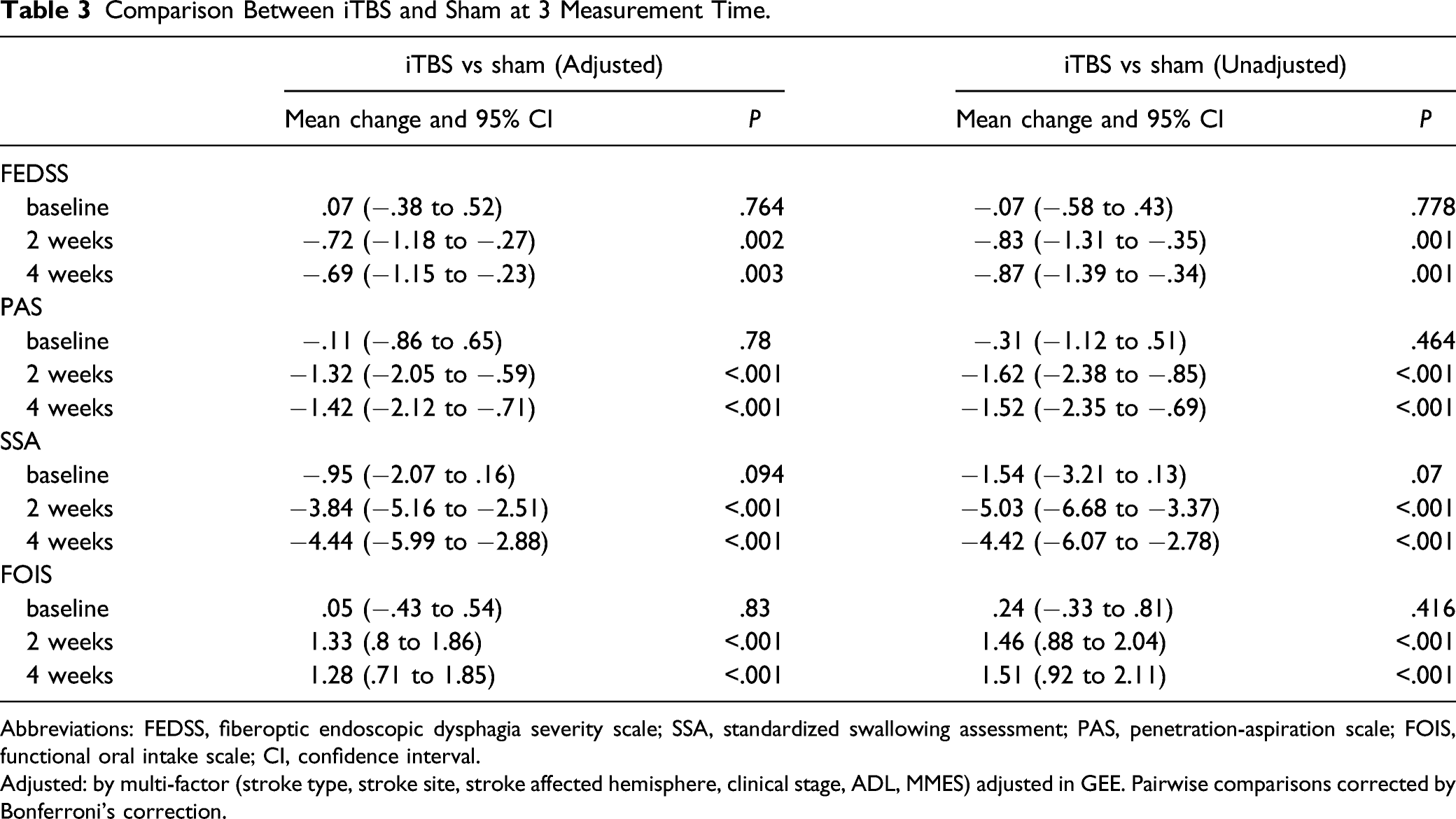
Abbreviations: FEDSS, fiberoptic endoscopic dysphagia severity scale; SSA, standardized swallowing assessment; PAS, penetration-aspiration scale; FOIS, functional oral intake scale; CI, confidence interval.
Adjusted: by multi-factor (stroke type, stroke site, stroke affected hemisphere, clinical stage, ADL, MMSE) adjusted in GEE. Pairwise comparisons corrected by Bonferroni’s correction.
The subgroup analysis shown a significant heterogeneity in clinical stage (P = .026 for interaction). The benefit of iTBS appeared to be attenuated in chronical stage. No significant heterogeneity was observed in stroke type (P = .353 for interaction) (Figure 5).
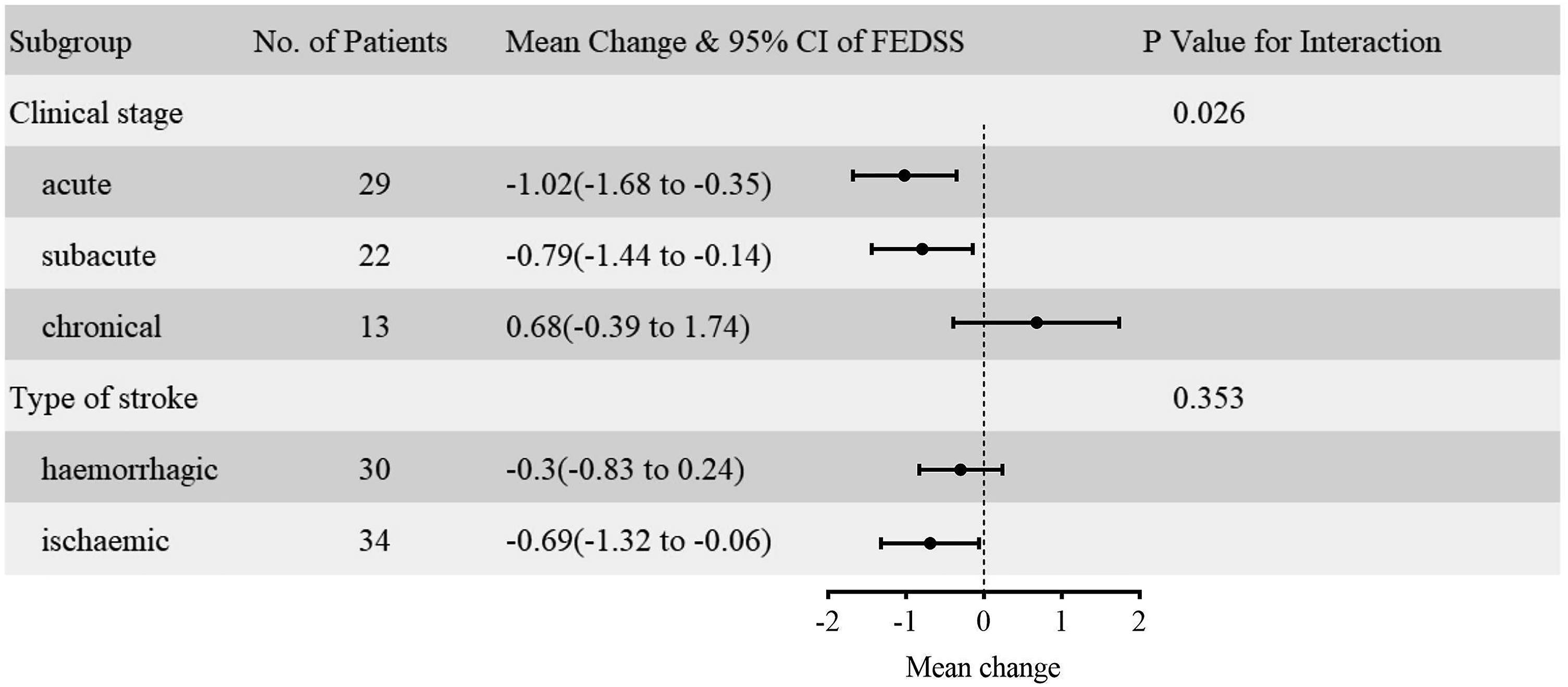
The subgroup analysis of clinical stage and type of stroke. P value is not adjusted by covariates.
During the treatment period, 3 participants in the true iTBS group had slight dizziness, which was tolerable, and the experiment could be continued. No tinnitus or headache was observed during iTBS stimulation. None of the participants withdrew from the study due to the occurrence of seizures or other serious adverse reactions during the treatment period.
Discussion
To our knowledge, this is the first randomized, double-blind, sham-controlled trial to investigate the effects of bilateral cerebellar iTBS stimulation on swallowing function in stroke patients with dysphagia. Our study revealed that bilateral cerebellar iTBS combined with swallowing exercises reduced the FEDSS, PAS, and SSA score and improved the FOIS score significantly after 2 weeks of treatment. Furthermore, improvement was also observed at the 4-week follow-up. Figure 6 directly shows the change in the FEDSS and PAS under the fiber endoscope.
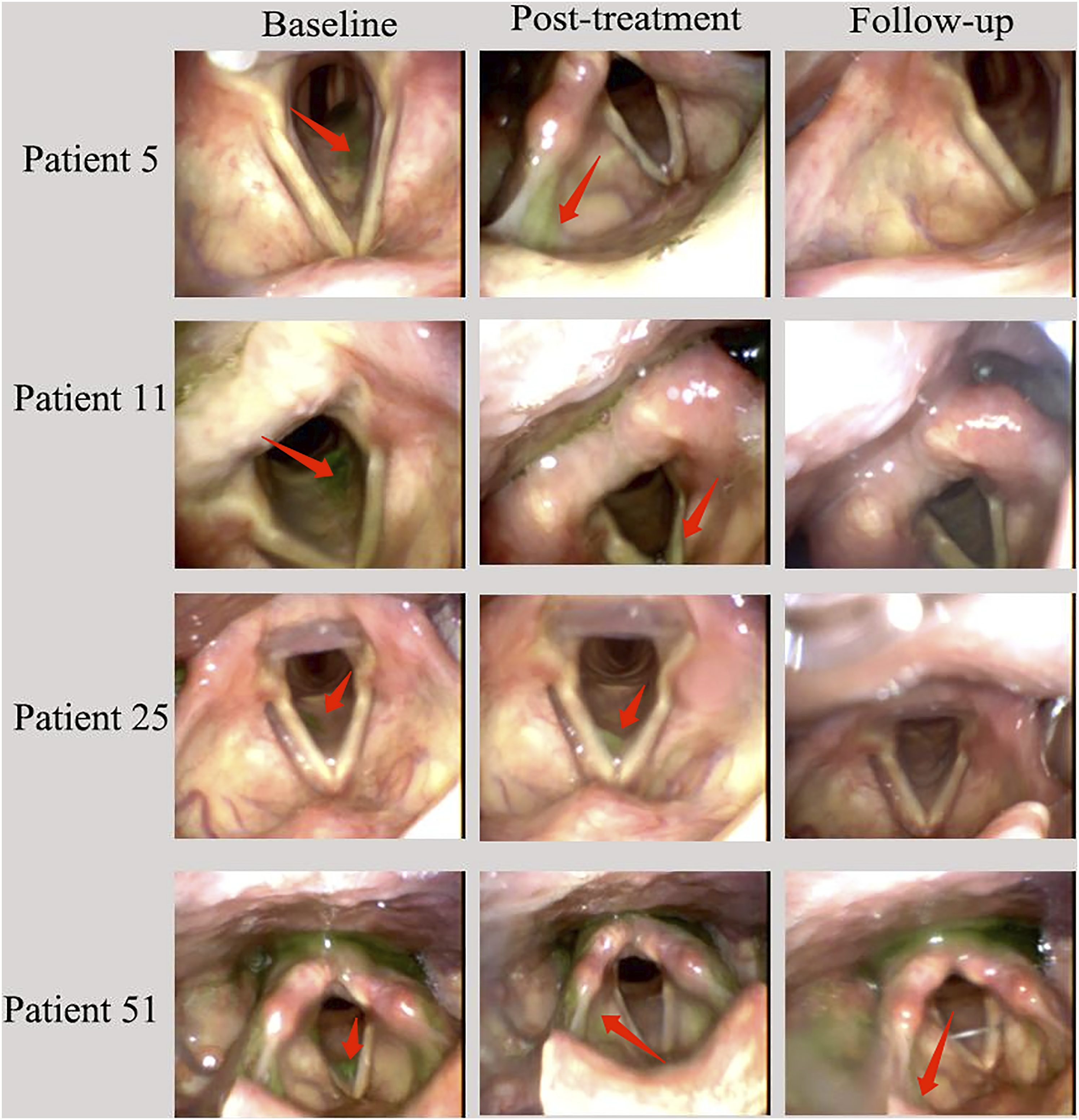
Fiberoptic endoscopic images of 4 patients (patients 5 and 11 in the real iTBS group and 2 in the sham group) at baseline, posttreatment, and follow-up. At baseline, green puree was aspirated into the subglottic airway in all 4 patients. A protective cough reflex was observed in patients 5 and 25, in which the bolus was cleared out of the airway, but not in patients 11 and 5. Therefore, the FEDSS and PAS scores were 4 points and 6 points, respectively, for patients 5 and 25, 5 points for the FEDSS and 7 points for the PAS for patients 11 and 51. After 2 weeks of treatment, there was no aspiration into the airway, but there was penetration to the vocal cords (patients 11 and 25, FEDSS 3 points, PAS 4 points) and the laryngeal vestibule (patients 5 and 51, FEDSS 3 points, PAS 3 points). In the follow-up, no penetration or aspiration was observed (FEDSS 1 point, PAS 1 point), except for patient 51, who still had a small amount of aspiration in the vestibular larynx but could clear it out (FEDSS 2 points, PAS 2 points).
The mean change of between iTBS and sham was .72 points at 2 weeks and .69 points at 4 weeks. Although the degree of improvement was less than 1 point, we still considered it clinically significant. First, after 2 weeks of treatment, 84.8% patients in iTBS group improve more than 1 FEDSS point, while 38.7% in sham group. Second, in clinical terms, a one-point increase in the 6-point scale makes the difference between nasogastric tube (score 5) and return to initial oral feeding (score 4), or parenteral fluid supplementation (score 3) vs oral fluid intake (score 2). Third, a FEDSS predictive model indicated that, for every 1 point improvement in FEDSS, the incidence of pneumonia can be 2-fold reduced and the odds for the necessity of endotracheal intubation reduced by a factor of 2.5. 40 Besides, the second outcome (PAS, SSA, FOIS) show consistent improvement as FEDSS. Therefore, in this study, the clinical significance of bilateral cerebellar iTBS stimulation in patients with post-stroke dysphagia was evident.
Both the iTBS and sham groups improved swallowing, partly due to the effect of spontaneous recovery and traditional swallowing treatment. However, after multi-factorial correction, there was still a significant difference between the 2 groups, with the iTBS group being more effective than the sham stimulation group. It indicates that bilateral cerebellar iTBS can improve swallowing function. This is similar to the results of previous studies of cerebellar rTMS stimulation. Sasegbon reported bilateral cerebellar 10 Hz rTMS better activates the cortical brainstem tracts to the pharynx than unilateral cerebellar. 21 In zhong’s study, cerebellar 5 Hz rTMS with 40 45-pulse trains, inter-train interval of 21 s, 5 days a week for 2 weeks, also improved FEDSS scores for post-stroke dysphagia. The biggest advantage of iTBS stimulation over rTMS is its high efficiency, requiring only 190 s for 1 standard run. However, high-frequency rTMS usually takes 10–20 minutes, depending on the frequency and the total number of pulses. 32
The subgroup analysis of clinical stage show the acute and subacute phases are more effective than in the chronic phase. This difference was significant difference. This is similar to the optimal time for recovery of motor function after stroke. The best time for motor recovery after stroke is the first 3 months. After 3 months the rate of recovery decreases, but partial recovery is still possible. 45 Subgroup analysis of stroke types showed that recovery of swallowing in ischemic appeared to be superior to hemorrhage. However, the differences were not significant difference. As there was only 1 case in brainstem stroke and infratentorial stroke for the iTBS group, subgroup analysis based on stroke site and affected hemisphere was not appropriate.
In our study, some of the patients enrolled had mild cognitive impairment, but were able to cooperate with the examination and treatment, as required by the inclusion criteria. Cognitive impairment can affect the recovery of other functions. 46 However, the cognitive impairment in the iTBS and sham groups was not statistically significant at baseline. As limited of sample size, so we did not include secondary influences such as MMSE, hypertension, diabetes mellitus, hyperlipidemia, and drugs in the statistical analysis to correct for covariates.
The control of swallowing involves several brain regions including bilateral cortex, limbic system, basal ganglia, thalamus, brainstem, and cerebellum.47,48 The cerebellum is richly connected directly or indirectly to the cortex, basal ganglia, thalamus, and brainstem. Afferent nerve of cerebellar cortex and dentate nucleus enters the contralateral motor cortex via a synaptic connection with the thalamus. 49 Efferent nerves from the fastigial nucleus contact the central pattern generator (CPG) in the brainstem. 49 CPG regulates swallowing and interfaces with the swallowing cortex on both sides of the cerebral hemisphere. 7 The richly connected network implies that the cerebellar modulator could indirectly regulate the swallowing cortex, basal ganglia, CPG, etc. In Jayasekeran’s study of cerebellar interactions between cerebellar and pharyngeal motor cortical activity, it was found that cerebellar TMS stimulation facilitates corticobulbar projections. 15 Besides, Vasant found that cerebellar 10 Hz rTMS increased cortico-pharyngeal MEP amplitude by 52% and lasted 30 minutes, compared to cerebellar sham stimulation, which indicate cerebellar stimulation can produce sustained increases in corticobulbar excitability. 16 iTBS, as a new stimulation mode of TMS, could induce the long-term potentiation (LTP) effect of synaptic transmission function and increase the excitability of the cerebellar. 50 The excited cerebellar cortex, dentate and dentate nuclei regulate the restoration of swallowing, through afferent and efferent nerves, in contact with the cortex, thalamus, basal ganglia and CPG. Mosier thought the cerebellum may be responsible for feed-forward mechanisms and controlling timing, sequencing, and internal coordination. 51 But, Through which circuits do the activated cerebellum activate or inhibit other brain regions, is still clear.
No severe adverse events were observed during the treatment period, and only 3 participants in the true iTBS group had slight dizziness. A review of the safety and efficacy of theta burst stimulation revealed that there was no significant increase in adverse events in TBS compared with traditional TMS, such as a mild headache, local pain, dizziness, palpitations and nausea. 52
There are some limitations to this study. First, the limitation of experimental conditions, such as the absence of pharyngeal catheter to measure the MEP of pharynx to precisely locate the hot spot of cerebellum; The stimulation coil was hand-fixed, hardly to ensure same position for the entire duration, due to lack of neuronavigation and the metal arm is not strong enough to fix coil; We could not use more advanced sham coil due to lack of equipment instead of 90° coil tilt, which is not the most adequate blinding. If possible, use more accurate experimental equipment to reduce experimental error and bias. Second, The rehabilitation process was not strictly controlled, and for ethical reasons, patients with limb paralysis received the appropriate physical and occupational therapy, while patients without the appropriate dysfunction did not receive physical and occupational therapy. It may has influence on the result. Thirdly, For safety reasons, our study excluded patients with hemorrhagic transformation after cerebral ischemia, but a major cause of dysphagia is hemorrhagic transformation. So our study did not reflect the effect and safety of patients with hemorrhagic transformation. Forth, the effect of bilateral cerebellar iTBS in our study was evaluated based on the FEDSS and clinical scale, not on electrophysiological or functional images, such as MEP, functional near-infrared spectroscopy (fNRIS), fMRI, PET, etc., due to a lack of relevant equipment available for experimentation. Therefore, no mechanistic studies were conducted. This is our future studies.
Conclusions
The present study suggested that as a more efficient TMS stimulation mode, iTBS could better improve swallowing function compared to sham stimulation, by stimulating the bilateral cerebellar hemisphere when combined with traditional dysphagia treatment for dysphagia post-stroke.
Footnotes
Acknowledgements
We thank Professor Li Ye of Guangxi Medical University for providing valuable revision comments and statistician Yiyang Liu of Yue Bei People’s Hospital for his guidance on statistical analysis. The authors are grateful to the participants, suppliers, and administrative staff at Yue Bei People’s Hospital and The First Affiliated Hospital of Guangxi Medical University who made the trial possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Science and Technology Department of Guangdong Province (No. 201803010).
Informed consent
The participants signed written informed consent forms. Any potentially identifiable images or data in this study were authorized in writing by the patient.
Ethics statement
Human trials were involved, and the study was approved by the ethics committee of Yue Bei People’s Hospital (No. KY-2020-213).
Author Contributions
Huiyu Liu, Pu Wang and Jianwen Xu were involved in the conception of the trial, supervised the study, and modified the manuscript. Jinzhu Rao, Fang Li, and Lida Zhong performed data sorting and analysis, manuscript writing and editing. Jing Wang and Yang Peng performed data collection. The authors declare that there is no conflict of interest. All authors have consented to the submitted version of the manuscript.
