Abstract
Background
Traumatic brain injury (TBI) can cause sensorimotor deficits, and recovery is slow and incomplete. There are no effective pharmacological treatments for recovery from TBI, but research indicates potential for anti-Nogo-A antibody (Ab) therapy. This Ab neutralizes Nogo-A, an endogenous transmembrane protein that inhibits neuronal plasticity and regeneration.
Objective
We hypothesized that anti-Nogo-A Ab treatment following TBI results in disinhibited axonal growth from the contralesional cortex, the establishment of new compensatory neuronal connections, and improved function.
Methods
We modeled TBI in rats using the controlled cortical impact method, resulting in focal brain damage and motor deficits like those observed in humans with a moderate cortical TBI. Rats were trained on the skilled forelimb reaching task and the horizontal ladder rung walking task. They were then given a TBI, targeting the caudal forelimb motor cortex, and randomly divided into 3 groups: TBI-only, TBI + Anti-Nogo-A Ab, and TBI + Control Ab. Testing resumed 3 days after TBI and continued for 8 weeks, when rats received an injection of the anterograde neuronal tracer, biotinylated dextran amine (BDA), into the corresponding area contralateral to the TBI.
Results
We observed significant improvement in rats that received anti-Nogo-A Ab treatment post-TBI compared to controls. Analysis of BDA-positive axons revealed that anti-Nogo-A Ab treatment resulted in cortico-rubral plasticity to the deafferented red nucleus. Conclusions. Anti-Nogo-A Ab treatment may improve functional recovery via neuronal plasticity to brain areas important for skilled movements, and this treatment shows promise to improve outcomes in humans who have suffered a TBI.
Introduction
Traumatic brain injury (TBI) is a major cause of disability and can result in long-term or permanent deficits in sensory and motor functions.1-3 Recovery after a TBI is slow and often incomplete, due in part to the limited regenerative capacity in the central nervous system (CNS). Compensatory sprouting of uninjured axons, which may underlie functional recovery, is restricted by the presence of myelin-associated inhibitory proteins, among which the most well-characterized is Nogo-A.4-7 Numerous animal studies have demonstrated that suppression of Nogo-A signaling leads to enhanced neuronal fiber generation, and consequently, Nogo-A has been identified as a therapeutic target in models of CNS injury, such as spinal cord injury (SCI) and ischemic stroke (see Schwab and Strittmatter 8 for review). We therefore sought to determine if inhibition of Nogo-A signaling may also be a useful approach to ameliorate deficits from a TBI.
Studies of SCI in rodents have shown that treatment with anti-Nogo-A antibodies results in improved functional recovery, enhanced fiber regeneration, compensatory sprouting, and the formation of new functional connections in the spinal cord (see Zörner & Schwab 9 for review). Our laboratory and others have used rodent models of ischemic stroke and shown that anti-Nogo-A treatment improves spatial memory performance and sensorimotor recovery, and also induces neuroplastic changes that may underlie the functional improvement.10-20 Anti-Nogo-A treatment has also been investigated in a rat model of TBI. Following a fluid-percussion injury, treatment with anti-Nogo-A antibodies was found to enhance cognitive function at 4 weeks post-injury. However, this treatment had no effect on restoring motor function, and did not alter axonal sprouting. 21 Importantly, in our previous studies of anti-Nogo-A treatment following stroke, sensorimotor recovery, and neuroplasticity were not observed until several weeks after the cessation of treatment and more than 4 weeks post-injury.13,14,16,18,22 Thus, we hypothesized that sensorimotor recovery correlates with neuronal plasticity which requires more than 4 weeks for new axonal connections to form.
In the current study we utilized the controlled cortical impact (CCI) model of TBI to test the efficacy of anti-Nogo-A therapy on sensorimotor functional recovery. This method produces a less variable and more focal injury in comparison to fluid percussion injury, 23 and allowed us to induce a consistent sensorimotor impairment in the contralesional forelimb. We used the skilled forelimb reaching task and the horizontal ladder walk task to assess functional recovery using a long-term endpoint of 8 weeks post-injury to allow for growth of new axons. After completion of behavioral testing an anterograde neuronal tracer, biotinylated dextran amine (BDA), was injected into the contralesional forelimb motor cortex to determine if axonal plasticity had occurred. Our results showed an enhancement of sensorimotor functional recovery in rats receiving anti-Nogo-A treatment, and this corresponded with an increase in BDA-labeled, midline-crossing fibers to the deafferented red nucleus, an important nucleus for motor control.24,25 Our results indicate that anti-Nogo-A treatment restores sensorimotor function following a TBI, perhaps through the enhancement of compensatory neuroplasticity within motor circuits.
Methods
All experiments were approved by the Institutional Animal Care and Use Committee and conducted in the AAALAC accredited animal facility of Edward Hines Jr. Veterans Affairs Hospital. The studies were performed in compliance with the Department of Agriculture Animal Welfare Act regulations and the National Institutes of Health’s Public Health Service Policy. The ARRIVE guidelines 2.0 for reporting animal research were followed for the preparation of this manuscript. 26
Subjects
A total of 24 male, Long Evans, black-hooded rats (HsdBlu:LE), 6 weeks of age, were purchased from Envigo (Indianapolis, IN). One subject died early in the study and was excluded from the results. Rats were pair-housed in standard cages, in a temperature- and humidity-controlled room, on a 12-hour light-dark cycle. Rats had ad libitum access to water and were ration-fed with standard laboratory diet (Teklad 20185, Envigo) to maintain approximately 95% of their predicted weight by age, starting 1 week after arrival in the animal facility.
Experimental Timeline
The experimental timeline is illustrated in Figure 1. After 1 week of acclimation and handling, rats began training on the skilled forelimb reaching task. The rats were required to establish a limb preference and achieve baseline proficiency on the reaching task (described below). Then, baseline performance was also measured on the horizontal ladder walk task. All subjects then received a TBI followed by 2 days of recovery. On postoperative day (POD) 3 the rats were tested a second time on the horizontal ladder walk task and began testing on the skilled forelimb reaching task (5 days/week, for 8 weeks following TBI). On POD 7, rats were randomly assigned to 1 of 3 groups: TBI-only (n = 8), TBI + Anti-Nogo-A antibody (Ab, n = 7), or TBI + Control Ab (n = 8). Treatment groups had osmotic pumps surgically implanted, which delivered Ab intracerebroventricularly (ICV), continuously until they were removed 2 weeks later, on POD 21. On POD 56 the rats were tested a third time on the horizontal ladder walk task. All behavior testing was conducted by the same investigator, who was blind to the treatment groups. After behavior testing, rats received an injection of the anterograde neuronal tracer, BDA, into the caudal forelimb motor cortex contralateral to the TBI. Two weeks later the rats were euthanized, and brains were processed for analyses of lesion size and neuronal plasticity.
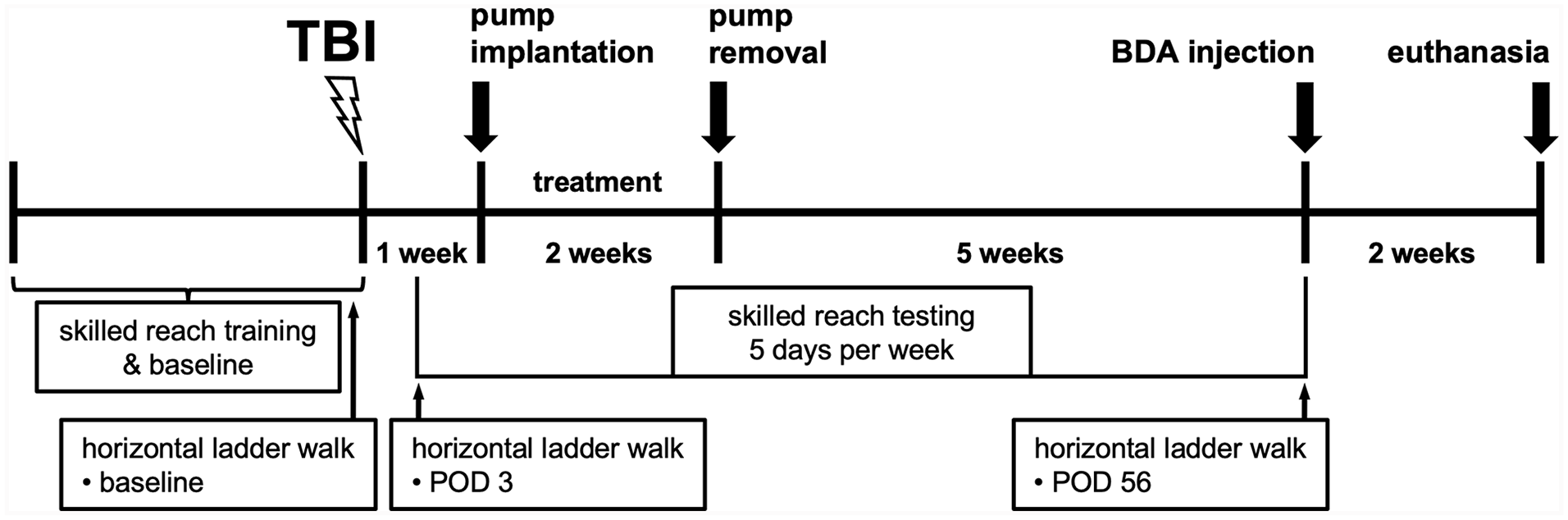
Experimental timeline. Baseline performance was assessed on the skilled forelimb reaching task and the horizontal ladder walk task prior to traumatic brain injury (TBI). Rats were treated with anti-Nogo-A or control Ab for 2 weeks, beginning 1 week after TBI, and 1 group had TBI-only, with no treatment. Behavior testing resumed on postoperative day (POD) 3, the skilled forelimb reaching task was assessed 5 days per week across the following 8 weeks, and the horizontal ladder walk task was examined on POD 3 and POD 56. Rats then received an injection of the anterograde neuronal tracer, biotinylated dextran amine (BDA), into the contralesional cortex and were euthanized 2 weeks later.
Skilled Forelimb Reaching Task
The skilled forelimb reaching task is a sensitive measure of voluntary, precise forelimb and digit use in both normal functioning and brain damaged rats,22,27-31 and its use has been validated for the measurement of motor functional recovery following neurological injury such as ischemic stroke.13-18 Skilled reach testing was conducted during the light phase, in a transparent, acrylic chamber (30 cm × 36 cm × 30 cm) with a rectangular opening (1.5 cm × 3 cm) in 1 wall, and a short shelf outside of the box, beneath the opening. Individually, rats were placed into the chamber and the investigator placed round, 45 mg food pellets (5TUT, #1811251, Test Diet, St. Louis, MO, USA), 1 at a time, onto the shelf 1.5 cm from the opening. Rats were trained daily (Mon-Fri) until they learned to reach through the window, grasp the pellet, and bring it into their mouth (a success). Rats were required to establish a limb preference and achieve a baseline score of 16 successful reaches out of 20 trials for 3 consecutive days. After meeting criteria, all subjects received a TBI (described below). Testing (20 trials per day, 5 days/week) resumed 3 days after TBI and continued until 8 weeks after TBI. The average success score per week was the dependent measure for analysis.
Reaching Error Analysis
The rats were video recorded while performing the skilled forelimb reaching task on 3 days: baseline (before TBI), POD 3 (before treatment), and POD 56. Four subjects (3 TBI-only and 1 TBI + Anti-Nogo-A Ab) were excluded from this analysis due to poor recording quality. Recordings were analyzed frame by frame, and each trial was categorized, as shown in Figure 4, adapted from Tsai et al. 32 These categories provided a numerical scale (1-8) to score the severity of impairment in the use of the forelimb. Errors were scored by an investigator blind to experimental groups. The average score across 20 trials on each day was the dependent measure for analysis.
Horizontal Ladder Walk Task
The horizontal ladder walk task is used in rodents to assess skilled walking, limb placement, and limb coordination, 33 and is suitable to assess lesions of the motor system, as demonstrated in rodent models of cortical injury 34 and pyramidotomy. 35 The apparatus was a 1-m long walkway with sidewalls made of transparent acrylic, and an open platform on each end. The walkway was elevated 15 cm above the tabletop below, and the floor consisted of metal rungs (3 mm diameter), spaced unevenly apart at a minimum distance of 1 cm and a maximum distance of 3 cm between rungs. Rats were first acclimated to the apparatus with 3 trials, traversing the walkway freely and without reinforcement. The rats were tested (and video recorded) on 3 different days (pre-TBI baseline, POD 3, and POD 56) with 3 trials each day. Recordings were analyzed by an investigator blind to experimental groups, scoring the number of foot faults per 10 steps. A foot fault was characterized as a total miss or slip from the rung or a misplacement of the paw on the rung (ie, using the wrist or digits instead of the plantar surface). A separate score was counted for each of the 4 limbs: forelimb 1 (the impaired forelimb), rear limb 1 (ipsilateral to the impaired forelimb), forelimb 2 (contralateral to the impaired forelimb), and rear limb 2 (contralateral to the impaired forelimb).
TBI—CCI
After meeting criteria on the skilled forelimb reaching task all subjects received a TBI to the caudal forelimb motor cortex associated with their preferred forelimb via CCI, as previously described31,36,37 and adapted for Long-Evans rats.38,39 Rats were anesthetized with isoflurane (2%-3% in oxygen) and body temperature was maintained between 36.5°C and 37.5°C for the duration of the procedure. Rats were shaved at the surgical site and placed in a stereotaxic instrument. A midline incision exposed the cranium, and a 4 mm diameter craniotomy was performed directly over the (preferred) caudal forelimb motor cortex, at a position of 0.75 mm anterior and 2.75 mm lateral to bregma. The TBI was delivered by a CCI injury device (MyNeuroLab, Impact One Stereotaxic Impactor; Leica Microsystems Inc., Buffalo Grove, IL, USA), consisting of a small bore, double acting, pneumatic piston cylinder (3 mm diameter) mounted on a stereotaxic micro-manipulator, angled 22.0° away from vertical so that the flat impactor tip was perpendicular to the surface of the brain. The cylinder struck the brain at 2.5 m/s, at a depth of 1.7 mm below the cortical surface, for 250 ms. After impact, the skull fragment was replaced, and the scalp was closed with 4-0 monofilament nylon suture.
Intracerebroventricular (ICV) Ab Treatment
One week following TBI, rats in the Ab treatment groups were anesthetized with isoflurane and had an osmotic pump (Alzet model 2ML2; Durect Corporation, Cupertino, CA, USA) surgically implanted, subcutaneously in the mid-scapular area, as previously reported.10,13 The pump was attached to a catheter that led subcutaneously to a cannula leading into the ipsilesional lateral cerebral ventricle. This method of infusion into the cerebrospinal fluid (CSF) has been shown to distribute the anti-Nogo-A Ab throughout the CNS.14,40 The cannula was placed in a 2 mm burr hole, 1.3 mm lateral, 0.8 mm posterior, and 3.8 mm dorsoventral, relative to bregma. The pumps contained 2.5 mg/ml of either purified mouse monoclonal anti-Nogo-A Ab (11C7; IgG1) or control Ab (anti-bromodeoxyuridine; IgG1), which was infused at a rate of 5.0 µL/hour. After 2 weeks, the pump, cannula, and tubing were surgically removed under isoflurane anesthesia. Rats in the TBI-only group did not have surgery to implant osmotic pumps.
Biotin Dextran Amine Neuroanatomical Tracing
After completion of behavioral testing, rats were anesthetized with isoflurane and placed into the stereotaxic instrument as described above. A midline scalp incision was made to expose the skull, and a 2 mm burr hole was made to expose the caudal forelimb motor cortex contralateral to the TBI. Two 1 µL aliquots of 10% BDA (10 000 MW, lysine fixable, Invitrogen) solution in 0.01 M phosphate buffer (pH 7.2) were injected at a depth of 1.5 mm into the caudal forelimb motor cortex at the following coordinates: 2.5 mm lateral/1.0 mm anterior, and 3.0 mm lateral/0.5 mm anterior, relative to bregma. The scalp was then closed with 4-0 monofilament nylon suture. The depth and coordinates were based on prior studies,14,38,39 and were chosen to target layer 5 of the caudal forelimb motor cortex and to cover the approximate cortical area that was targeted by the TBI in the contralateral hemisphere.
BDA Histochemistry
BDA histochemistry was performed as previously described by our laboratory.14,17,18,32 Two weeks after BDA injections, rats were euthanized with sodium pentobarbital (100 mg/kg; i.p.) and transcardially perfused with heparinized PBS solution followed by 4% paraformaldehyde. Brains were extracted, post-fixed for 24 hours in 4% paraformaldehyde, cryoprotected by sinking in 30% sucrose, frozen, and sectioned coronally at 40 µm using a cryostat. Endogenous peroxidases were blocked via incubation with 0.1% hydrogen peroxide solution. Sections were then washed and incubated overnight with an avidin-biotin-peroxidase complex (ABC elite, Vector Labs), washed again, and pre-incubated with 0.4% ammonium nickel sulfate, followed by 0.015% 3,3′-diaminobenzidine, and finally reacted with 0.004% hydrogen peroxide. The reaction was stopped by washing with tris buffer, and the sections were then mounted on slides and air dried before being lightly counterstained with Nissl stain.
Lesion Size Analysis
Nissl-stained coronal sections were quantitatively analyzed every 400 µm between +4.7 mm and −5.2 mm from bregma, using a computer-interfaced imaging system (Image J, NIH, Bethesda, MD, USA), as described previously.14,18,22,32 All slides were coded, and investigators were blind to the treatment groups. Lesion size was expressed as a percent of the intact contralateral hemispheric area (total area of the intact contralateral hemisphere minus total area of the ipsilesional hemisphere divided by the total area of the intact contralateral hemisphere). Four subjects (2 TBI-only and 2 TBI + Control Ab) were excluded from lesion size analysis due to tissue damage.
Neuroplasticity Analysis
BDA-labeled fibers were quantified at the level of the red nucleus, using the atlas of Paxinos and Watson 41 to identify anatomical structures. All slides were coded, and investigators were blind to the treatment groups. The cortico-efferent projections to the red nucleus were quantified ipsi- and contralaterally to the injection site, as described previously.14,35 Labeled corticofugal fibers in the cerebral peduncle ipsilateral to the injection site were counted, and this number was used to correct for variance among animals in BDA tracing. The corticorubral projection to the contralateral red nucleus was analyzed by counting all BDA-positive fibers crossing the midline on each section through the rostral-caudal extent of the red nucleus. To normalize for staining differences, the number of midline-crossing BDA-positive fibers was divided by the total number of corticofugal fibers in the cerebral peduncle and multiplied by 10 000 to determine the red nucleus midline fiber crossing index. Histological analyses of BDA-positive fibers included only 5 subjects per group, as some subjects were excluded due to either tissue damage, poor BDA transport, or insufficient injection site uptake.
Statistical Analysis
All analyses were performed using GraphPad Prism, version 9.5.1 (GraphPad Software, LLC, Boston, MA, USA). Analyses of lesion size and BDA-labeled fibers were conducted by 1-way analysis of variance (ANOVA), followed by Tukey’s post-hoc to correct for multiple comparisons. The skilled reaching task and skilled ladder walk task data were analyzed using 2-way repeated measures ANOVA, followed by Tukey’s post-hoc to correct for multiple comparisons. A P < .05 was considered significant. There were no violations of normality or homogeneity of variance in any of the analyses.
Results
Lesion Size Was Consistent in All Experimental Groups
Figure 2 shows images of Nissl-stained coronal sections from a representative rat in each group. The lesions were localized to the sensorimotor cortex corresponding to each rat’s preferred forelimb. Quantification of lesion size as a percentage of the non-lesioned hemisphere revealed no statistical difference between experimental groups (TBI-only, 10.15% ± 2.48; Control Ab, 13.01% ± 2.31; Anti-Nogo-A Ab, 10.37% ± 2.21; P > .05).
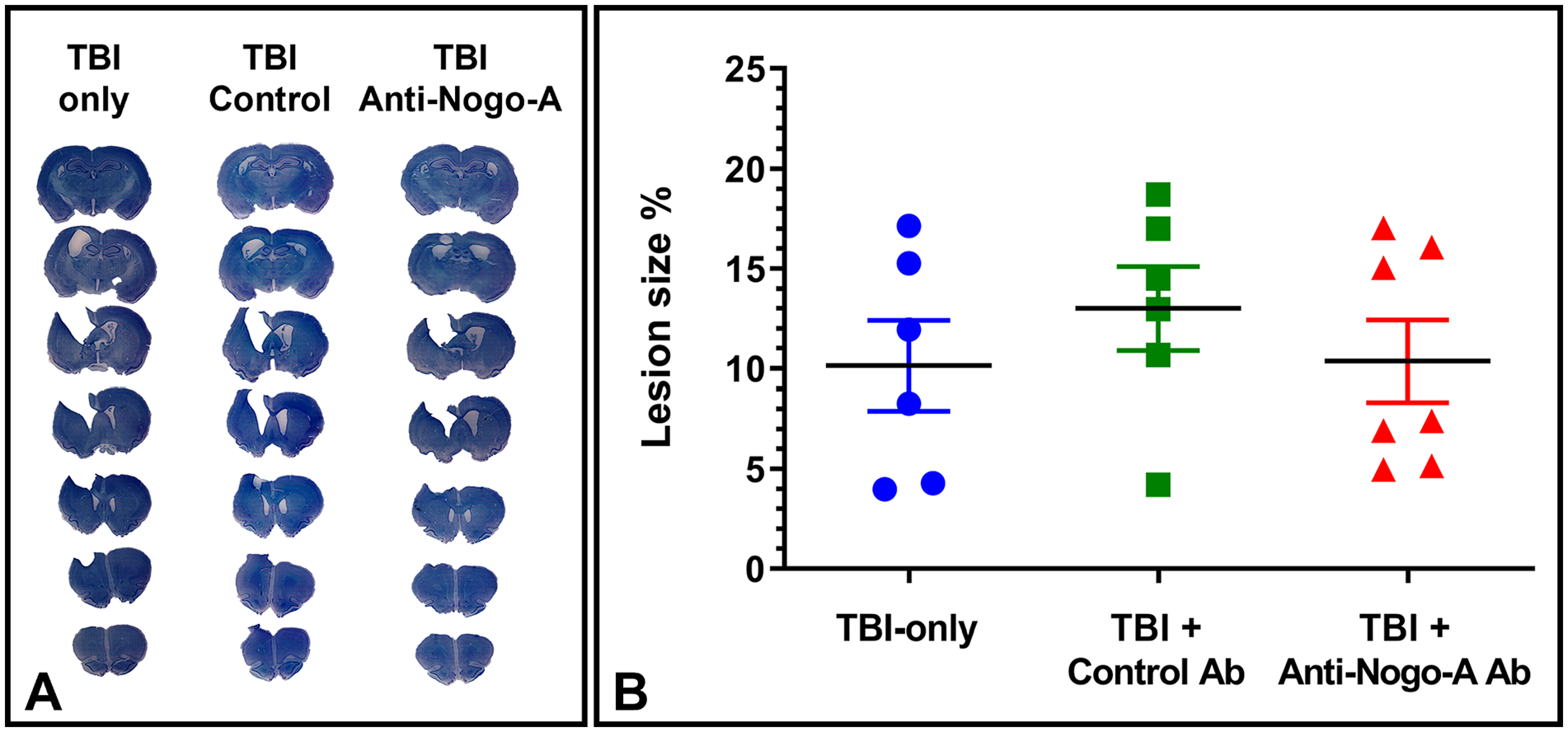
TBI lesion size. (A) Representative examples of Nissl-stained coronal sections showing the typical TBI lesion for all 3 groups of rats. (B) Quantification of lesion size as a percentage of the non-lesioned cortex. One-way ANOVA indicated no difference in the lesion size between groups (P > .05).
Anti-Nogo-A Ab Treatment Enhanced Recovery on the Skilled Forelimb Reaching Task
Success scores were averaged across 5 days into weekly blocks and analyzed via 2-way repeated measures ANOVA, revealing a significant main effect of week [F(4,86) = 72.19, P < .0001] and a significant group by week interaction [F(16,160) = 3.455, P < .0001] (Figure 3). After training on the skilled reaching task, baseline success scores did not differ between experimental groups. After TBI (prior to treatment) success scores were significantly decreased in all subjects, again with no statistical difference between groups. All groups demonstrated improvement from week 1 to week 8. However, Anti-Nogo-A Ab treated rats had significantly improved success scores compared to both TBI-only and Control Ab groups at weeks 6, 7, and 8 (P < .05). Anti-Nogo-A Ab rats improved to approximately 70% of their pre-TBI baseline, while Control Ab and TBI-only rats recovered only to approximately 40% of pre-TBI baseline, with no significant difference between control groups.
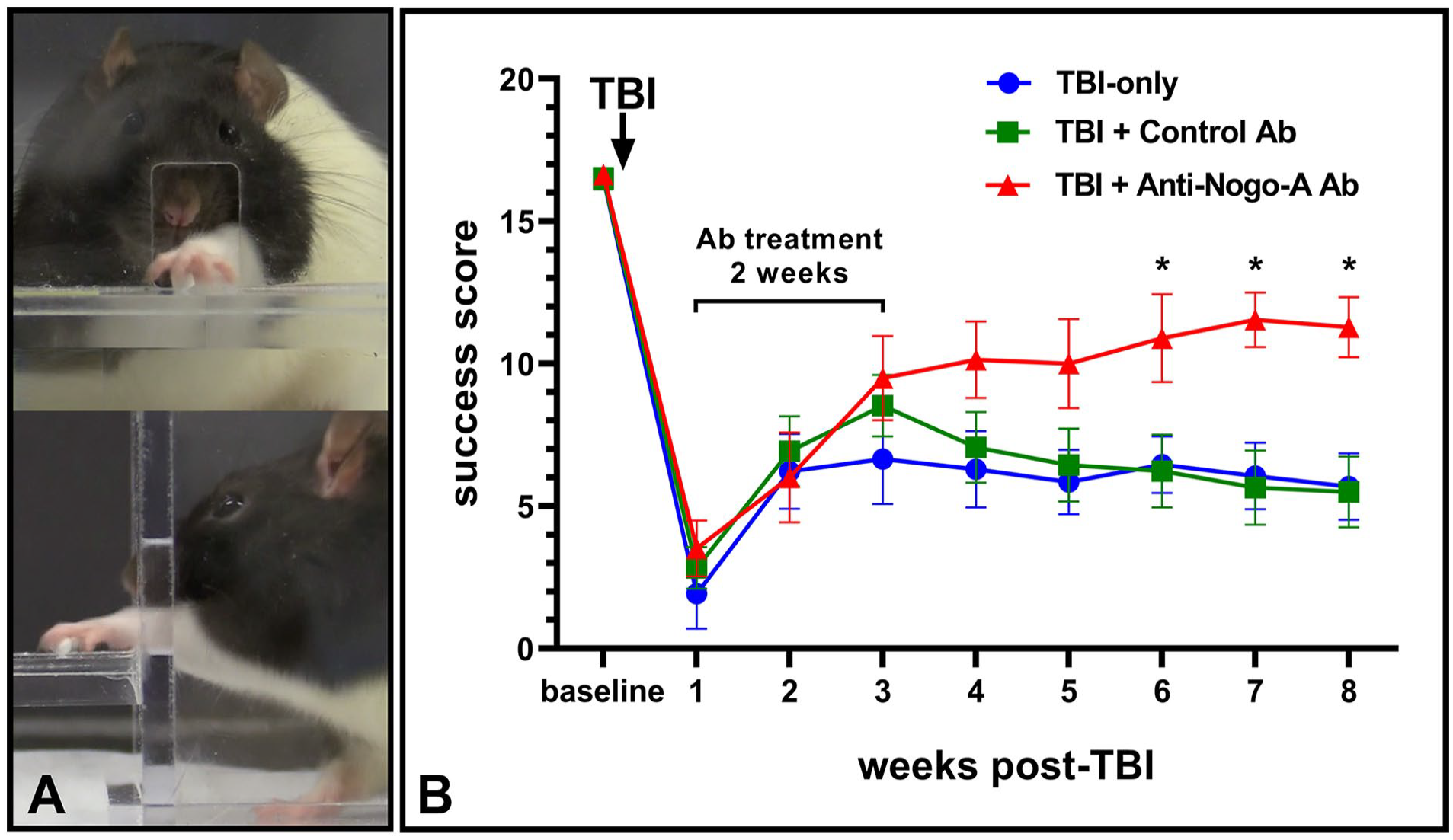
Skilled forelimb reaching task. (A) Front and side views of a rat reaching through the window and grasping a pellet. (B) Success scores on the reaching task at baseline and across 8 weeks of testing after TBI. Two-way repeated measures ANOVA revealed a significant effect of week (P < .0001), and a significant week by group interaction (P < .0001). There was no difference between groups at baseline. Deficits were observed at week 1 post-TBI, before the treatment began, with no significant difference between groups. Rats receiving Anti-Nogo-A Ab treatment exhibited improvement in comparison to both Control Ab and TBI-only groups, with significant differences evident at weeks 6, 7, and 8 (*P < .05). Error bars = ±SEM.
Reach Error Analysis Revealed Functional Improvement in Anti-Nogo-A Ab Treated Rats
Analysis of the forelimb reaching errors indicated that the severity of the errors was decreased in the Anti-Nogo-A Ab treated rats 8 weeks after TBI. Reaching errors were categorized as shown in Figure 4, and the heatmap shows trial by trial (1-20) results for individual subjects on each day, with each category color coded (eg, white = most severe deficit; dark blue = successful reach). The average score on each day was analyzed via 2-way repeated measures ANOVA, revealing a significant main effect of day [F(2,28) = 94.21, P < .0001], a significant main effect of group [F(2,16) = 6.019, P = .0112], and a significant group by day interaction [F(4,32) = 4.961, P = .0032]. At pre-TBI baseline there was no significant difference in score between groups. All groups had >80% successful reaches, with some minor deficits in their unsuccessful reaches. On POD 3 there was no significant difference in score between groups. All groups exhibited poor success and made errors that were predominantly in the more severe categories (ie, failure to extend limb, or failure to grasp), represented by lighter colors. On POD 56, the Anti-Nogo-A Ab treated rats had a significantly higher score compared to both TBI-only and Control Ab groups (P < 0.05). Anti-Nogo-A Ab treated rats not only demonstrated a greater number of successful reaches, but their errors were improved to less severe categories (ie, dropping the pellet during retraction).
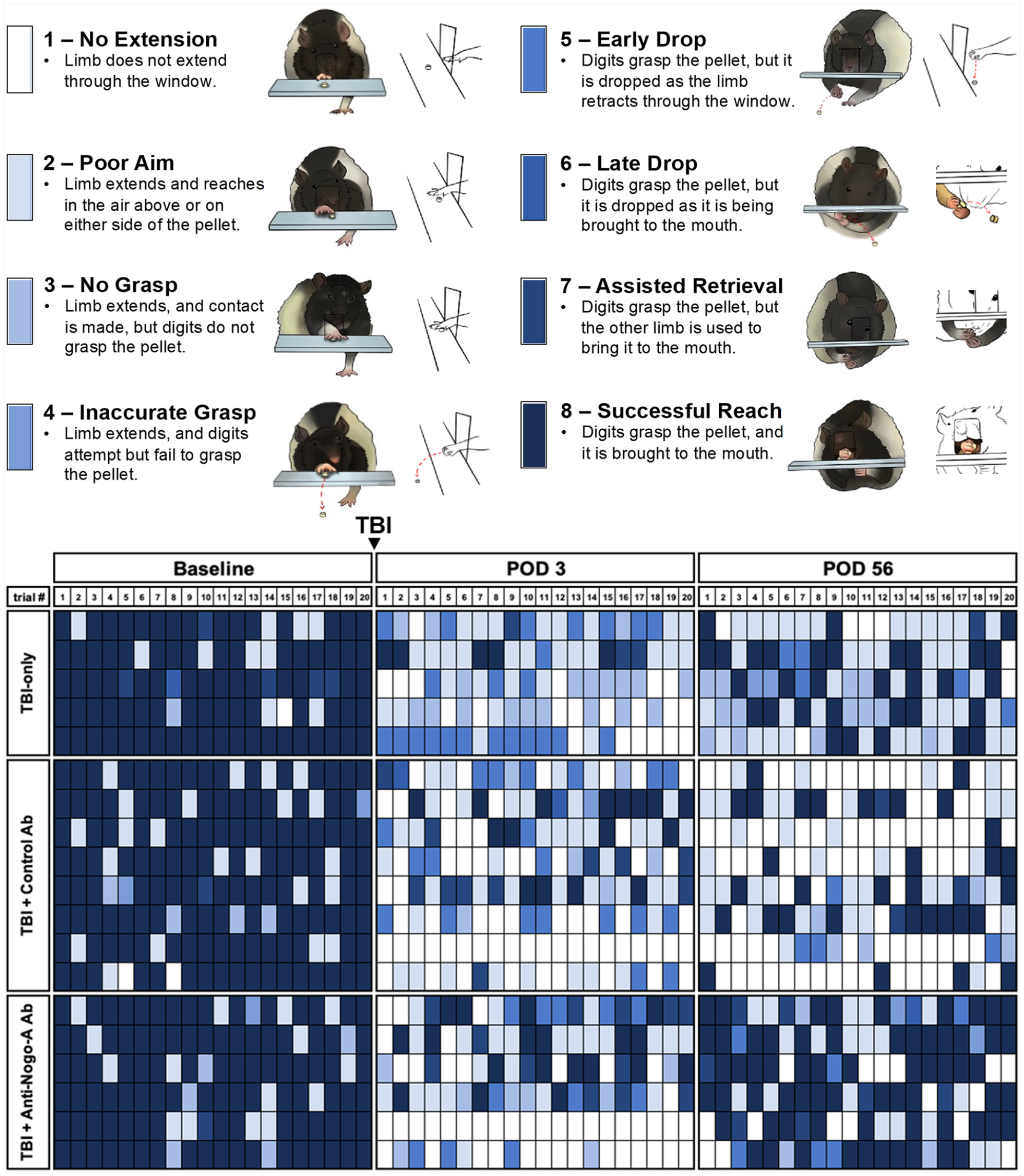
Heatmap illustrating categories of reaching errors. Rats were recorded and analyzed at pre-TBI baseline, POD 3 after TBI, and POD 56 after TBI. The result of each of the 20 trials was categorized and color-coded from white to dark blue, as illustrated, with white indicating the most severe deficit, and dark blue a successful reach. Each row represents a different subject, each column represents a different trial. The preponderance of darker shades observed in the lower right panel indicates that at POD 56, the Anti-Nogo-A Ab group made more successful reaches compared to controls, but also exhibited a shift to less-severe categories of errors.
Anti-Nogo-A Ab Treatment Improved Recovery on the Horizontal Ladder Walk Task
Foot slips on the horizontal ladder walk task were analyzed using 2-way repeated measures ANOVA, revealing a significant main effect of day [F(2,33) = 278.4, P < 0.0001] and a significant group by day interaction [F(4,38) = 4.360, P = .005] in the lesion-impaired forelimb (Figure 5). At baseline, rats made very few foot slips, and there was no significant difference between groups. On POD 3 (prior to treatment), all rats demonstrated an increase in foot slips, but there was no statistical difference between groups. However, on POD 56, the Anti-Nogo-A Ab group made significantly fewer foot slips in comparison to both the Control Ab group (P = .002) and the TBI-only group (P = .007). When examining the limbs not directly affected by the cortical lesion (ie, the forelimb contralateral to the impaired forelimb, the rear limb ipsilateral to the impaired forelimb, or the rear limb contralateral to the impaired forelimb), there were no significant differences between groups on any day (Figure 5).
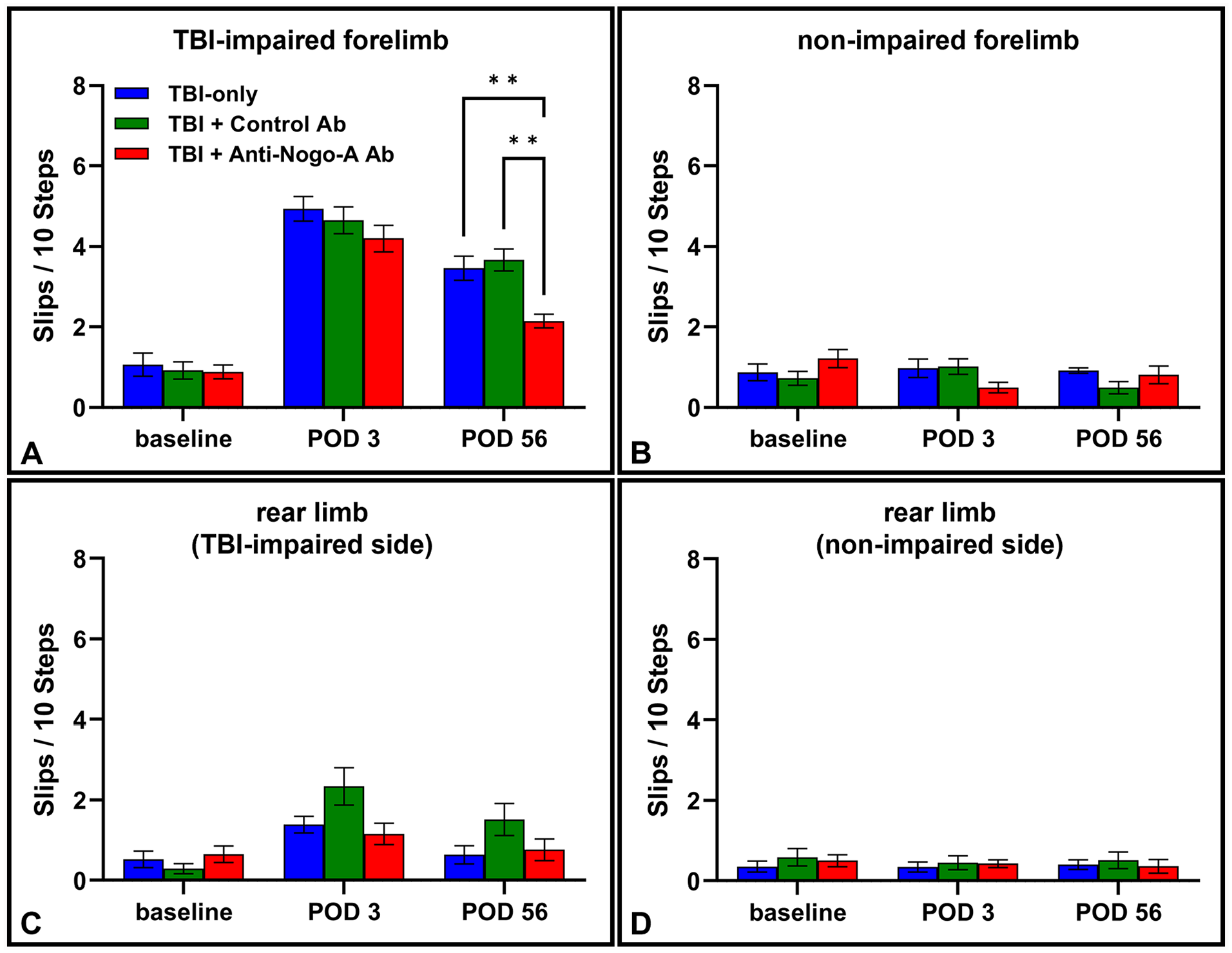
The skilled horizontal ladder walk task. (A) Slips/10 steps at baseline, POD 3 after TBI, and POD 56 after TBI, in the TBI-impaired forelimb. Two-way repeated measures ANOVA revealed a significant main effect of day (P < .0001), and a significant day by group interaction (P < .01). There was no difference between groups at baseline. Deficits were observed on POD 3, before the treatment began, with no significant difference between groups. On POD 56, rats receiving Anti-Nogo-A Ab treatment exhibited significant improvement in comparison to both Control Ab and TBI-only groups (**P < .01). There were no significant differences between groups on any day in the contralateral forelimb (B), the rear limb ipsilateral to the TBI-affected forelimb (C), or the rear limb contralateral to the TBI-affected forelimb (D). Error bars = ±SEM.
Anti-Nogo-A Ab Treated Rats Exhibited Increased Fiber Crossings in the Red Nucleus
Figure 6 shows representative images of BDA-labeled corticofugal fibers crossing the midline at the level of the red nucleus, and the quantification of the crossing index. One-way ANOVA revealed a significant main effect of group [F(2,12) = 4.977, P < .05]. Tukey’s multiple comparison test confirmed the crossing index was significantly increased in the Anti-Nogo-A Ab treated rats in comparison to the Control Ab treated rats and the TBI-only rats (P < .05), indicating cortico-rubral axonal plasticity to the deafferented red nucleus.
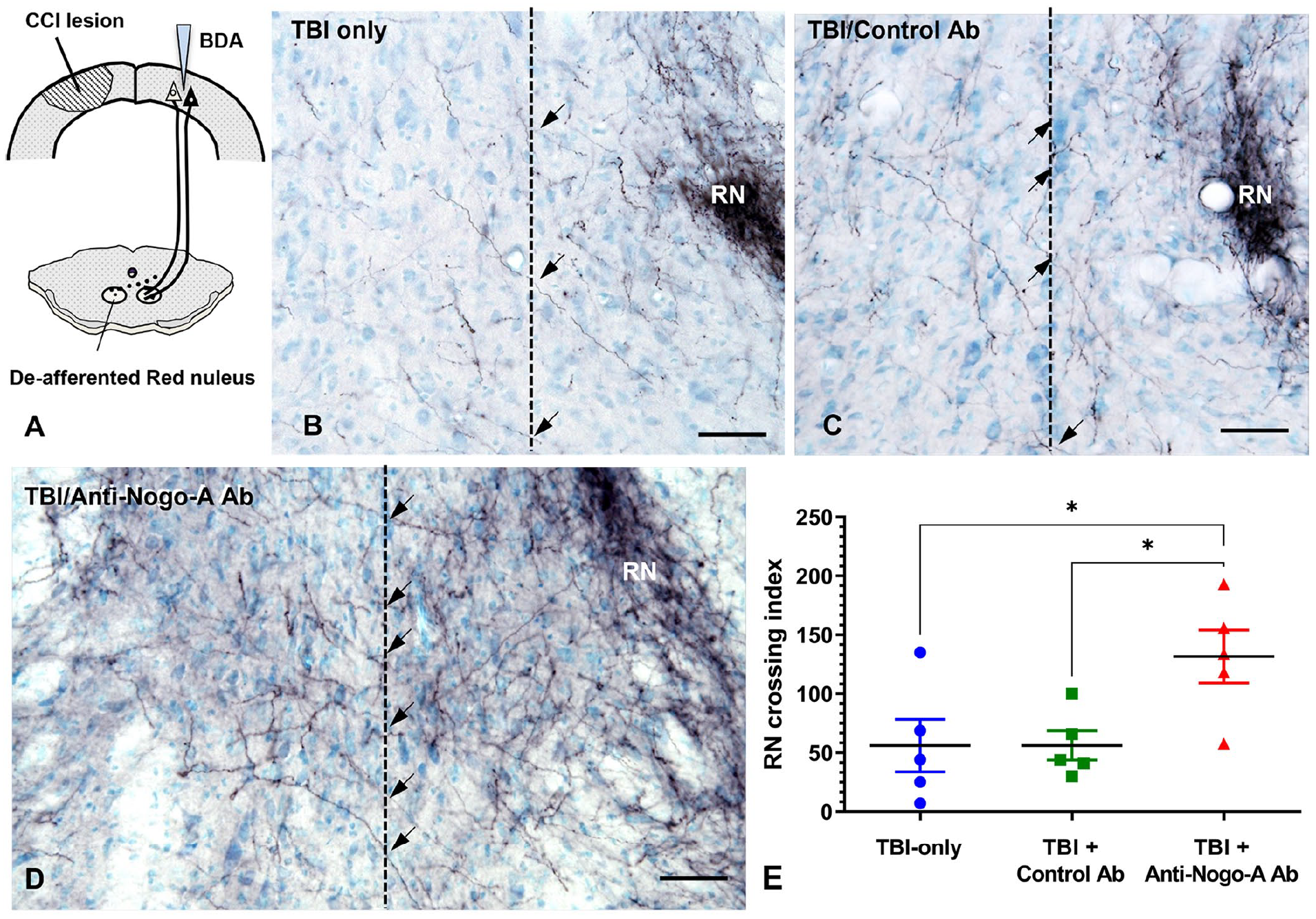
Neuroplasticity in the red nucleus. (A) After a unilateral CCI lesion to the caudal forelimb motor cortex, an anterograde neuronal tracer (BDA) was injected into the contralateral cortex. We hypothesize that neurons in the spared cortex sprout new axons, which cross the midline (dotted line) to innervate the deafferented subcortical motor neurons in the red nucleus (RN). Representative images of the RN demonstrate midline crossing fibers (arrows) observed in TBI-only (B), Control Ab (C), and Anti-Nogo-A Ab (D) rats. Bars on images indicate 50 µm. (E) One-way ANOVA of the standardized total of BDA-labeled midline crossing corticofugal fibers at the level of the RN revealed a significant main effect of group (P < .05). The RN crossing index was increased in rats receiving the Anti-Nogo-A antibody relative to the Control Ab and TBI-only groups (*P < .05). Error bars = ±SEM.
Discussion
Our results show that treatment with an anti-Nogo-A Ab beginning 1 week after TBI enhanced sensorimotor recovery in the impaired forelimb. Furthermore, we observed an increase in midline-crossing fibers to the red nucleus in Anti-Nogo-A Ab treated rats, indicating that the treatment leads to increased neuronal plasticity from the contralesional sensorimotor cortex to deafferented neurons in the red nucleus, a midbrain region important for motor control.24,25
We used the CCI method to induce a TBI in rats, which allowed for a consistent, reproducible lesion specifically targeting each rat’s preferred forelimb motor control area.28,30,31,36,37 Function was assessed using a pair of tasks that have been used extensively in rodent models of acquired brain injury.14-18,28,30,31 Our results from the skilled ladder walk task confirmed the specificity of the injury, with all rats demonstrating significant impairment in the targeted forelimb, while demonstrating normal function in all other limbs. The skilled reaching task has particular translational value, because the component movements of reaching and their topography are very similar in rats and humans, 42 and reaching abnormalities following CNS injury are also similar to those observed in humans. 43 Additionally, the neural circuitry involved in reach-to-grasp behavior has been well-characterized in rodents, 44 and is highly conserved between rats and humans. 45 Furthermore, the skilled reaching task can be used to measure performance with a success score but can also be used for kinematic analyses 46 and for categorical analyses of error type, 32 which can provide additional information about the nature of recovery.
We observed a profound deficit in the reaching success score across all groups during week 1 after TBI, and all groups demonstrated some level of improvement by week 8. However, the TBI-only and the Control Ab treated rats recovered to only about 40% of their baseline scores, while the Anti-Nogo-A Ab treated rats recovered to about 70% of their baseline scores. By week 6 after TBI, the Anti-Nogo-A Ab group was significantly improved in comparison to controls. This level of functional improvement agrees with prior reports of anti-Nogo-A treatment in rats with lesions to the sensorimotor cortex via cortical aspiration34,38 or ischemic stroke.13,14,16-18,22 One previous study of anti-Nogo-A treatment in rats following a TBI reported no improvement in motor function, 21 but only measured effects up to 4 weeks after TBI. In the current study, we did not detect a significant difference between groups until 6 weeks after TBI, suggesting that recovery depends on neuroplasticity that most likely requires weeks for the establishment of new functional synaptic connections. We have previously shown that new connections established following anti-Nogo-A treatment are indeed functional, 38 and it has also been demonstrated that inactivation of newly sprouted axonal connections reverses functional recovery. 19
We also conducted a detailed examination of performance on the skilled forelimb reaching task with an analysis of error types. On POD 3 all rats exhibited a preponderance of the most severe error categories, characterized by lack of limb extension through the window and a failure to grasp the pellet. On POD 56 the Anti-Nogo-A treated rats demonstrated not only an improvement in their success scores, but also a shift to less-severe error types, characterized by the ability to reach and grasp the pellet, but a failure to successfully complete the process of bringing the pellet to their mouth. This suggests that the anti-Nogo-A treatment had a dramatic effect on recovery of proximal motor function, but some evidence of distal motor impairment remained, which corresponds with prior results of anti-Nogo-A treatment following a cortical aspiration lesion. 34 It is possible that the rats may have continued to improve with additional testing.
It should be noted that recovery and neuroplastic responses to injury can vary across aspiration-, ischemic-, and CCI-lesion models.30,47,48 It has been shown that rehabilitation strategies known to be effective in animal models of stroke are not sufficient for recovery in animal models of TBI, and that enhanced recovery and neuroplasticity following TBI requires an approach that includes an intense and varied rehabilitation regime.30,47 However, in the current study we observed enhanced recovery from TBI in Anti-Nogo-A treated rats in spite of having a very limited rehabilitation regime. In fact, we believe the effects of rehabilitation are negligible in the current study, since rats were only allowed 20 reaches per day, 5 days per week, and were tested on the skilled ladder walk task after TBI only twice, on POD 3 and POD 56. By comparison, the aforementioned studies allowed rats 100 reaches daily along with additional rehabilitative treatment after TBI.30,47 Indeed, the functional recovery and neuroplasticity that we observed following TBI, absent significant rehabilitation, highlights the effectiveness of the anti-Nogo-A Ab treatment.
Nogo-A has been well-characterized as an inhibitor of axon growth using various rodent models of neuronal injury (see Pernet & Schwab 49 for review). Nogo-A signaling is induced through its binding to the S1PR2 receptor, 50 which activates the small GTPase Rho-A, and in turn the Rho-associated protein kinase (ROCK), which ultimately leads to cytoskeletal reorganization which mediates growth cone collapse and arrested growth of axons. 51 Nogo-A may also affect axon growth indirectly via effects on vascular remodeling after an injury. Genetic deletion of either Nogo-A or the S1PR2 receptor, or Ab-mediated neutralization of Nogo-A resulted in blood vessel regrowth in the periinfarct region of adult mice post-stroke. 52 Additionally, Nogo-A may affect neuronal plasticity by fine-tuning the balance between excitation and inhibition at the synapse, as Nogo-A was found to have a rapid effect on the strengthening of inhibitory synaptic transmission by restricting GABA receptor diffusion and promoting its synaptic localization. 53
The current study suggests that the neutralization of Nogo-A following a unilateral TBI to the caudal forelimb motor cortex leads to enhanced motor functional recovery via the strengthening of bilateral motor circuits. Rather than regenerating damaged axons, neuronal plasticity occurs from the contralesional cortex whereby axons from the homotypic forelimb region descend to re-innervate the deafferented red nucleus. This mechanism suggests that anti-Nogo-A Ab treatment may be particularly beneficial for humans with a unilateral cortical TBI, where the contralesional sensorimotor cortex can be leveraged for functional recovery. Notably, our study used only male rats, as epidemiological studies consistently report a higher incidence of TBI among males. 54 However, many TBIs may be unreported in women, and with the increasing participation of women in the military and in contact sports, the incidence may be increasing in females. 55 Undoubtedly, it will be necessary for future experiments to include female subjects.
Importantly, anti-Nogo-A Ab treatment is effective even when given 1-week after TBI, greatly enhancing the treatment window. Studies in stroke have even shown a much longer treatment window of 2 months,18,22 which may be applicable to TBI. While the effectiveness of anti-Nogo-A Ab treatment has been demonstrated in animal models, translation of this treatment to humans is currently underway. Promising results have been reported in 1 study of anti-Nogo-A Ab treatment for acute SCI in humans, 56 and new advances have led to a non-invasive, intranasal delivery method for anti-Nogo-A Abs, which may greatly improve future clinical trials. 57
Footnotes
Acknowledgements
The authors also thank Professor Martin Schwab for his generous gift of anti-Nogo-A antibody (11C7).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the VA Polytrauma and Traumatic Brain Injury Rehabilitation Research Fellowship from the Department of Veterans Affairs, the Paul Kalmanovitz Central Nervous System Repair Research Program, NIH/NS115759, and the Department of Veterans Affairs.
