Abstract
Background. Traumatic brain injury (TBI) is associated with altered white matter organization and impaired cognitive functioning. Objective. We aimed to investigate changes in white matter and cognitive functioning following computerized cognitive training. Methods. Sixteen adolescents with moderate-to-severe TBI (age 15.6 ± 1.8 years, 1.2-4.6 years postinjury) completed the 8-week BrainGames program and diffusion weighted imaging (DWI) and cognitive assessment at time point 1 (before training) and time point 2 (after training). Sixteen healthy controls (HC) (age 15.6 ± 1.8 years) completed DWI assessment at time point 1 and cognitive assessment at time point 1 and 2. Fixel-based analyses were used to examine fractional anisotropy (FA), mean diffusivity (MD), and fiber cross-section (FC) on a whole brain level and in tracts of interest. Results. Patients with TBI showed cognitive impairments and extensive areas with decreased FA and increased MD together with an increase in FC in the body of the corpus callosum and left superior longitudinal fasciculus (SLF) at time point 1. Patients improved significantly on the inhibition measure at time point 2, whereas the HC group remained unchanged. No training-induced changes were observed on the group level in diffusion metrics. Exploratory correlations were found between improvements on verbal working memory and reduced MD of the left SLF and between increased performance on an information processing speed task and increased FA of the right precentral gyrus. Conclusions. Results are indicative of positive effects of BrainGames on cognitive functioning and provide preliminary evidence for neuroplasticity associated with cognitive improvements following cognitive intervention in TBI.
Keywords
Introduction
Traumatic brain injury (TBI) is a leading cause of disability in children and adolescents worldwide and has a detrimental impact on the developing brain. 1 Previous research using diffusion weighted imaging (DWI) reported widespread white matter alterations in young patients with TBI. Large effect sizes are found for changes in white matter organization of the corpus callosum, internal capsules, temporal lobes, frontal lobes, uncinate fasciculi, and dorsolateral regions (for a meta-analysis see Roberts et al 2 ). Sustaining TBI during childhood—a “critical period” of white matter development 3 —interrupts white matter maturation and leads to severe impairments in daily life functioning. 2 Cognitive impairments are common after TBI, in particular problems with higher-order cognitive functions such as attention, working memory and executive functioning.4,5 Since childhood TBI often results in lifelong negative outcomes and low quality of life in older age, 6 it is crucial to ameliorate these cognitive deficits in young patients.
Indeed, cognitive rehabilitation is now considered a standard practice for TBI. Numerous recent studies and reviews highlighted that rehabilitative cognitive training programs are a promising approach for improving cognitive functioning in brain injury.7-10 In addition, the biological mechanisms that underlie cognitive improvements are recently being mapped utilizing magnetic resonance imaging (MRI) techniques. Functional MRI (fMRI) studies have shown functional changes in brain regions after cognitive training (i.e. functional neuroplasticity) in patients with brain injury. Reorganization of functional systems after training include changes in metabolic rate, cortical excitability, or blood flow.11-13 The structural neural basis of training-induced cognitive improvements on the other hand remains largely unclear. A recent review by Caeyenberghs et al 14 found evidence for structural neuroplasticity in patients with brain injury; however, the majority of previous studies focused on the effect of motor training programs (eg, balance training) on grey matter density. Most studies investigating cognitive training-related changes in white matter were conducted in healthy adults.15-19 For example, Takeuchi and colleagues 18 showed training-related increases in fractional anisotropy (FA) in regions close to the intraparietal sulcus and the body of the corpus callosum after a 2-month working memory training. They suggest that increased myelination might be the underlying mechanism of training-related increases in FA, which in turn enhances information flow between neural circuits leading to improved cognitive performance. A plethora of studies has investigated white matter changes using diffusion tensor models despite its limited ability to disentangle multiple diffusion directions within a single voxel.20,21 In addition, studies performing voxel-based analysis of diffusion MRI data often use tract-based spatial statistics (TBSS), 22 whereby analyses are performed within a limited white matter skeleton (based on voxels with high FA). 23 It is generally known, however, that FA can be reduced in regions with crossing fibers or white matter damage. 24 Therefore, TBSS is not a suitable DWI analysis technique for the study of white matter alterations in patients with TBI, because diffuse axonal injuries typically occur at the junction between gray and white matter. This gray matter–white matter boundary is not included in the skeleton (because of the lower FA values) resulting in the exclusion of these regions and thus leading to the inability to find group differences. Fixel-based analysis (FBA) is a recently developed whole brain analysis technique that allows the examination of white matter organization of multiple fiber populations within a voxel (or fixels) based on constrained spherical deconvolution. 25 Moreover, registration across subjects is performed on fiber orientation distribution (FOD) images that contain high contrast within white matter, enabling analyses across the majority of the white matter. 23
The objectives of this study were 3-fold. The first aim was to replicate previous findings of white matter alterations in a sample of young patients in the chronic stage of TBI recovery. To this end, we compared the novel diffusion metric fiber cross-section (FC) between a group of patients and healthy controls using FBA. In addition, we examined diffusion tensor metrics FA and mean diffusivity (MD) across the whole brain, taking advantage of FOD registration, smoothing, and statistical connectivity–based enhancement 23 from the fixel-based pipeline. Based on previous studies, we hypothesized to find lower FA and FC values and higher MD, because of the white matter damage. 2 The second aim was to investigate training-related improvements in cognition and alterations in white matter in young patients with TBI after an intensive 8-week cognitive training intervention called BrainGames. This computerized restorative training has shown promising effects on cognitive functioning in patients with TBI. 26 To complement the whole brain approach, we also applied a region-of-interest–based approach in the longitudinal analyses, because training-related changes in white matter are expected to be subtle. 27 We selected the following tracts based on previous training studies15-18 and anatomical knowledge: genu of the corpus callosum, cingulum and parahippocampal part of the cingulum and three segments of the superior longitudinal fasciculus (SLF). We expected to find training-related improvements in cognitive functioning and increases in FA, together with decreases in MD, with the strongest effects in the SLF.16,17,28 Our third aim was to examine the relationship between changes in diffusion metrics and changes in cognition in young patients with TBI. As alterations in white matter reorganization might underlie improvements in cognition, we expected a significant correlation between white matter changes and cognitive improvements. Using the FBA technique, we anticipated to achieve higher sensitivity to TBI-induced changes and training-induced alterations in white matter compared with using the diffusion tensor model and TBSS.
Methods
Participants
Eligible patients were recruited from the Child Rehabilitation Center of the University Hospital at Ghent, Belgium and the Rehabilitation Center for children and adolescents at Pulderbos, Belgium between March 2015 and January 2017. Sixteen patients with TBI enrolled in the cognitive training intervention (9 boys and 7 girls, mean age = 15.6 ± 1.8 years, range 11.1-17.5 years). All patients sustained moderate to severe TBI as defined by the Mayo Classification System for TBI severity. 29 Specifically, patients were recruited based on the presence of one or more of the following criteria: loss of consciousness of ≥30 minutes, lowest Glasgow Coma Scale score <13 in the first 24 hours, or evidence of hematoma, contusion, brain stem injury, or subarachnoid hemorrhage at the time of injury. Patients were all in the chronic stage of recovery, with a time since injury (TSI) of at least 1 year (mean TSI = 2.3 years ± 1.0). Additional eligibility criteria included a minimum age of 10 years at time of injury and presence of diffuse axonal injury (DAI) as determined by a neuroradiologist based on a susceptibility weighted image (SWI) scan at the time of our study (see Table 1 for patient characteristics). Following the injury, the patients underwent a multidisciplinary rehabilitation program (mean duration of 11.9 ± 6.2 months). They received physiotherapy (5 h/wk), occupational therapy (5 h/wk), speech therapy (3 h/wk), neuropsychological support (2-3 h/wk) and special schooling (5 h/wk). One patient did not receive rehabilitation in the acute phase postinjury. All patients attended a regular school after rehabilitation. Moreover, they had adequate linguistic-receptive and cognitive abilities to perform the cognitive training program independently at home. Seventy-seven percent of the patient sample received additional academic and/or psychosocial support. For every patient, an age (maximum ±6 months) and gender-matched control participant was recruited via social contacts (9 boys and 7 girls, mean age 15.6 ± 1.8 years, range 10.7-17.7 years). There were no significant group differences in gender, χ2(1) = 0, P = 1.00, or age, t(30) = −0.107, P = .92). Both groups underwent neuropsychological testing and MRI scanning at time point 1 (baseline, pretraining session). Next, patients followed the 8-week cognitive training intervention. Controls were not engaged in any sort of training (passive controls). After 8 weeks, both groups underwent neuropsychological testing and the patients underwent a second scanning session (posttraining session) to evaluate training-related changes in white matter. The study was approved by the Ethics Committee of the Ghent University Hospital (#2014/0540) and written informed consent was obtained from both parents and participant in accordance with the Declaration of Helsinki.
Patients’ Characteristics.
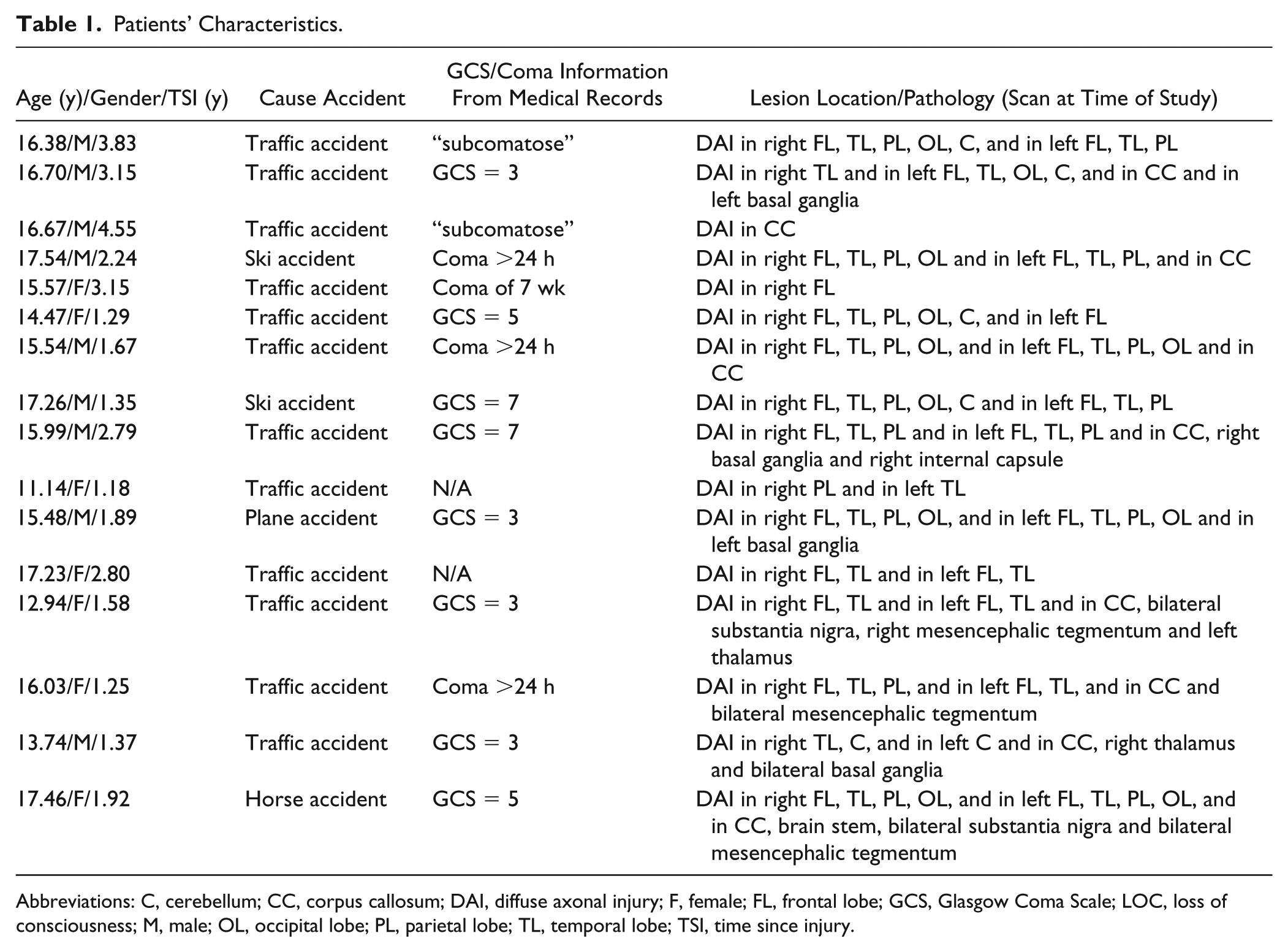
Abbreviations: C, cerebellum; CC, corpus callosum; DAI, diffuse axonal injury; F, female; FL, frontal lobe; GCS, Glasgow Coma Scale; LOC, loss of consciousness; M, male; OL, occipital lobe; PL, parietal lobe; TL, temporal lobe; TSI, time since injury.
Cognitive Training Intervention
In this study, we used BrainGames as cognitive training intervention. BrainGames is a computerized training program developed to ameliorate deficits in attention, working memory, and executive functioning in children with TBI. 25 This task-based restorative training intervention aims to improve cognitive functioning through repetition of demanding cognitive tasks with an increasing difficulty. Previous research has established the effectiveness of restorative cognitive training interventions in patients with acquired brain injury (ABI).7,10,30 A detailed description of this training program and preliminary efficacy results are reported elsewhere. 26 In short, BrainGames is an iPad application that consists of 8 games designed to train different aspects of cognitive functioning, including sustained, selective, and divided attention, inhibitory control, cognitive flexibility, verbal and visuospatial working memory, updating, and processing speed. The majority of the games are adaptations of well-known neuropsychological tests and/or existing cognitive training materials. Task difficulty increases adaptively depending on the participant’s level of performance, similar to previous cognitive training studies. 31 The dosage frequency is 5 sessions per week, and duration per session is 40 minutes, whereby 4 games are presented in a fixed order. In this study, BrainGames training was performed in a home-based environment and weekly phone calls were performed to monitor training motivation and provide assistance in case of difficulties/questions. All training data were sent to a backend server, which could be checked by the researchers to monitor training compliance. The average training completion (calculated as the percentage of successfully completed games) was 89.8% (SD = 9.37, range = 75% to 100%). The interested reader is referred to the Supplemental Material for a detailed description of this training program.
Cognitive Assessment
We used a battery of cognitive tasks to assess cognitive functioning at time points 1 and 2: the Digit Span backward (DSb) to assess verbal working memory (number of items correct) and Coding to examine information processing speed and associative nonverbal learning (number of items correct within 120 seconds) from the Dutch Wechsler Intelligence Scale for Children, Third Edition (WISC-III-NL). 32 The Spatial Span backward (SSb) was administered for visuospatial working memory assessment (number of items correct) and Stockings of Cambridge (SOC) for planning and problem solving (total number of moves needed to copy target constructions) from the computerized Cambridge Automated Neuropsychological Test Battery (CANTAB). A computerized Flanker test was used to measure response inhibition and selective attention by computing conflict cost (reaction time on incongruent minus congruent trials) from the Psychology Experiment Building Language (PEBL) battery. 33 Finally, executive functioning in daily life was assessed with the Dutch version of the Behavior Rating Inventory of Executive Function (BRIEF) 34 using the parent report form. The BRIEF comprises an overall general index score and two summary index scores, that is, metacognition index (MCI) and behavioral regulation index (BRI). 34 With regard to the BRIEF, only the MCI was selected for the present study, because the subscales of this index (Initiate, Working Memory, Plan-Organize, Organization of Materials, and Monitor) tap into cognitive components of executive functioning. A higher score on this questionnaire denotes more reported executive difficulties in daily life.
The cognitive assessment battery consisted of tests that tap into the same cognitive constructs as the training games (eg, the Flanker test and the Stingray game), while other tests had no resemblance to any of the training games (eg, SOC test). Notwithstanding the similarity between certain training games and cognitive tests, the training games utilized completely different stimuli in a game environment, bringing the resemblance to a minimum.
DWI Acquisition and Preprocessing
Imaging was performed using a Siemens 3.0 tesla Magnetom Trio whole-body scanner equipped with a 32-channel head coil. Diffusion weighted (DW) single-shot echo planar images were acquired with a twice-refocused spin echo sequence (60 contiguous transversal slices, field of view [FoV] = 240 × 240 mm2, voxel size = 2.5 × 2.5 × 2.5 mm3, repetition time [TR] = 10800 ms, echo time [TE] = 83 ms). Diffusion gradients were applied along 64 noncollinear directions (b = 1200 s/mm2) and one volume without diffusion weighting was obtained. In addition, a high-resolution 3-dimensional T1-weighted image was acquired at 1 mm isotropic resolution and an SWI scan for the patients was obtained to determine the DAIs.
DW data were preprocessed using MRtrix3. 35 Preprocessing of DW data included denoising, 36 removing of Gibbs ringing artifact,37,38 motion and eddy current–induced distortion correction, 39 and bias field correction. 40
From the preprocessed data, fiber orientation distributions (FODs) were obtained using multi-tissue constrained spherical deconvolution 41 using response functions for white matter and cerebrospinal fluid. To achieve spatial correspondence between the participants, a study-specific group template was generated using an iterative registration and averaging approach based on the FODs of all subjects (both controls and patients). 25
For the longitudinal analyses within the patient group, the paired nature of the data was taken into account; first a subject-specific template of the two time points was created for each participant and a group-specific template was then generated based on these subject templates. In both experiments, cross-sectional and longitudinal, the FODs of each subject were warped to the corresponding population template in a single step to avoid accumulation of interpolation errors.
Finally, whole brain probabilistic tractography was performed on the population template, generating 20 million streamlines and subsequently filtered down to 2 million using spherical-deconvolution informed filtering of tractograms (SIFT) to reduce seeding biases.42,43 These tractograms were used for connectivity- based smoothing and statistical inference. 23
Fiber Bundle Cross-Section
In the spatial normalization step, FODs were deformed to achieve the spatial correspondence. This implies that the underlying fiber populations may be expanded or contracted during the warping to the template. Therefore, differences in fiber cross-sectional area could be investigated using information derived from the applied transformation. The local volumetric changes of the FOD during the registration are given by the determinant of the local linear transformation F, then the change of the cross-sectional area for a given fiber orientation
Tensor-Derived Metrics
The FBA approach has been used to investigate microstructural changes in white matter relying on the hypothesis that FOD amplitudes are proportional to the intra-axonal volume of axons oriented in that direction. 44 This hypothesis holds for high b values (≥3000 s/mm2) but given the rather low diffusion weighing in the present study (b = 1200), we could not assure this assumption to be correct. Despite this limitation, it was possible to take advantage of the FBA approach while using tensor-derived parameters as fractional anisotropy (FA) and mean diffusivity (MD) by mapping these measures to all the fixels belonging to a voxel and incorporating the connectivity information into the statistical analysis. 23
Tract-of-Interest–Based Analyses
For the longitudinal analyses, diffusion parameters were examined in 11 tracts of interest (see Figure 1): genu of the corpus callosum (CC), left and right cingulum and parahippocampal part of the cingulum and 3 segments of the left and right superior longitudinal fasciculus (SLF). Group-generic tracts were delineated on the longitudinal group template according to previously validated tractography protocols.45-47 The tract density map 48 was calculated and thresholded using a parameter-free method 49 to define the final fixels in the tracts of interest. Next, the tracts were mapped to subject space using the inverse of the transformations that were obtained during the spatial normalization of the FBA. Finally, the mean FA, MD, and FC were calculated for each of the tracts in subject space.
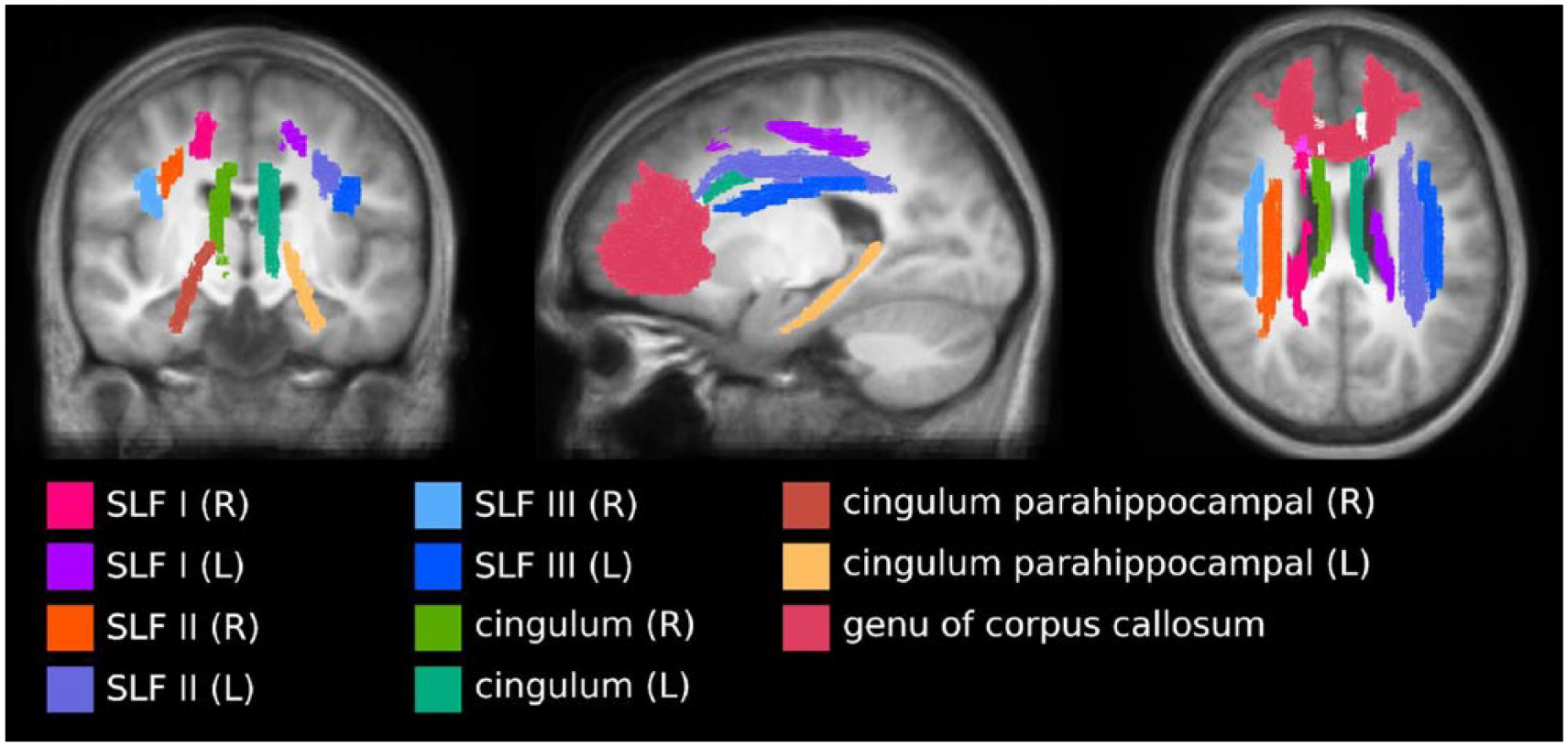
Group-generic tracts-of-interest created based on the group-specific longitudinal template that were warped to subject space.
Statistical Analyses
To examine group differences and training-related improvements in cognitive functioning, 2 (groups) × 2 (time points) repeated-measures analyses of covariance were used with age as covariate for all cognitive measures. False discovery rate (FDR) corrections were made to correct for multiple comparisons. The significance threshold was set at PFDR < .05. With regard to the FBA, nonparametric permutation tests (5000 permutations) were used to assess differences in FC, FA and MD between (a) TBI patients and controls and (b) patients pretraining versus patients posttraining. The permutation-based test statistics were calculated using connectivity-based fixel enhancement (CFE) 23 and included age and estimated intracranial volume (calculated using FreeSurfer 5.3 50 ) as nuisance covariates in the cross-sectional analysis. To control for false positives due to the number of tests, family-wise error (FWE) correction was used. Fixels with PFWE < .05 were selected and mapped to their corresponding streamlines to determine the white matter regions with significant changes. Significant fixels were anatomically identified using the JHU ICBM-DTI-81 white-matter atlas in FSL. 51 Training-related effects on FA, MD, and FC in the tracts-of-interest were investigated using paired-sample t tests within the patient group while applying FDR-corrections. Finally, associations between change scores (post – pretraining) of cognitive measures and change scores (post – pretraining) of diffusion metrics (both tract-based and fixel-based) within the patient group were performed using partial correlations with age as covariate. Because of the large amount of correlations (11 tracts × 3 diffusion metrics × 6 cognitive tasks), an exploratory significance threshold of P ≤ .001 was used when investigating the tracts-of-interest correlations. Therefore, these analyses should be considered as exploratory. An FWE-correction was applied with the fixel-based correlations.
Results
Group Differences and Training-Related Changes in Cognitive Measures
When comparing young patients with TBI to controls, we found significant lower performance in measures of inhibition (Flanker test, F(1,29) = 11.84, P = .002, η2 = .29), planning (SOC, F(1,29) = 8.14, P = .008, η2 = .20) and executive functioning in daily life (parent report MCI, F(1,29) = 28.01, P < .001, η2 = .49). No significant group differences were found for verbal (DSb) and spatial (SSb) working memory or information processing speed (Coding) (all P values >.05) (see Figure 2 and Table 2).
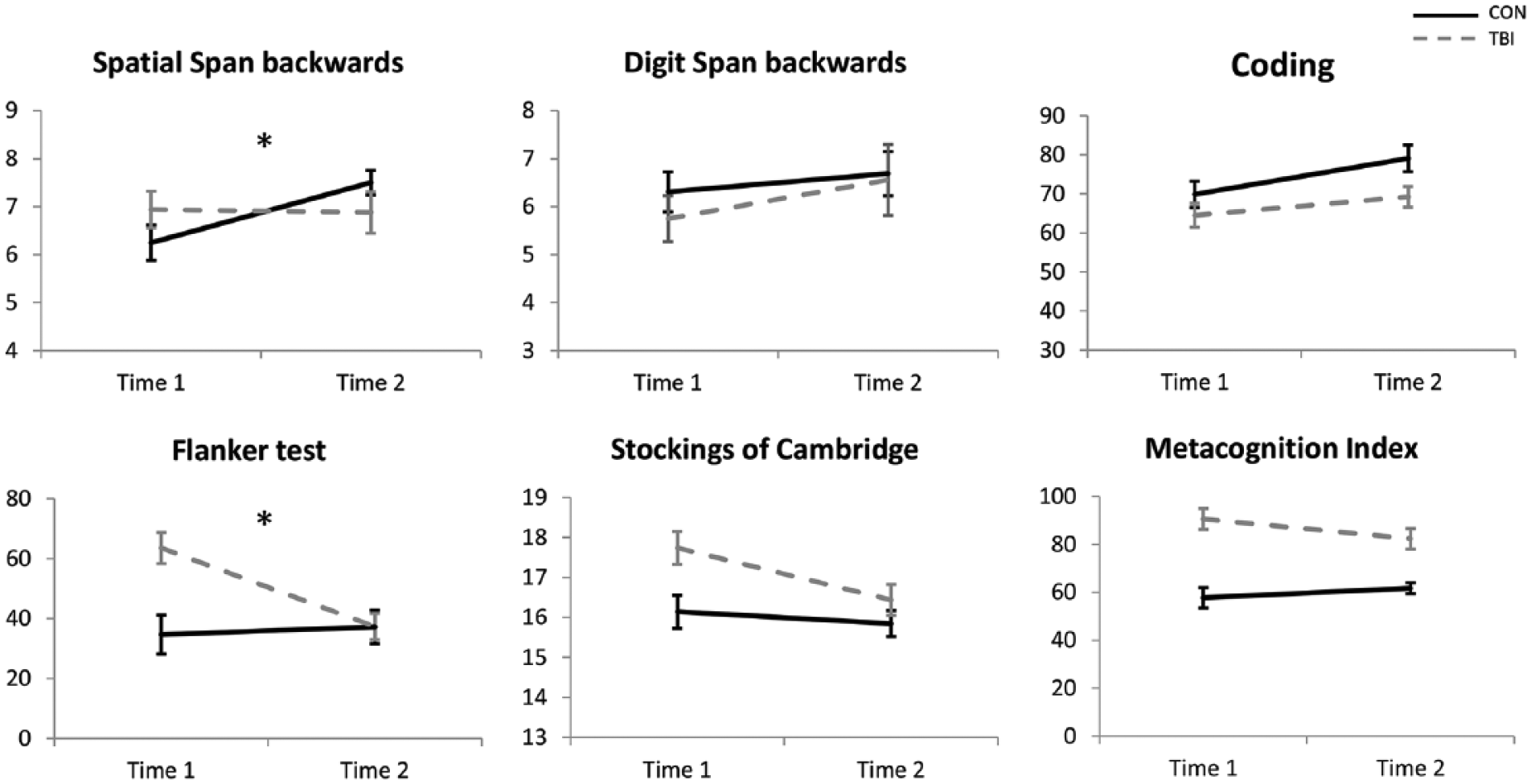
Group by time interaction effects of the cognitive measures. Top row: higher score = better score, bottom row: lower score = better score. Error bars represent standard errors and *PFDR < .05.
Results of Repeated-Measures Analyses of Covariance of Cognitive Measures With Age as Covariate of Nuisance.
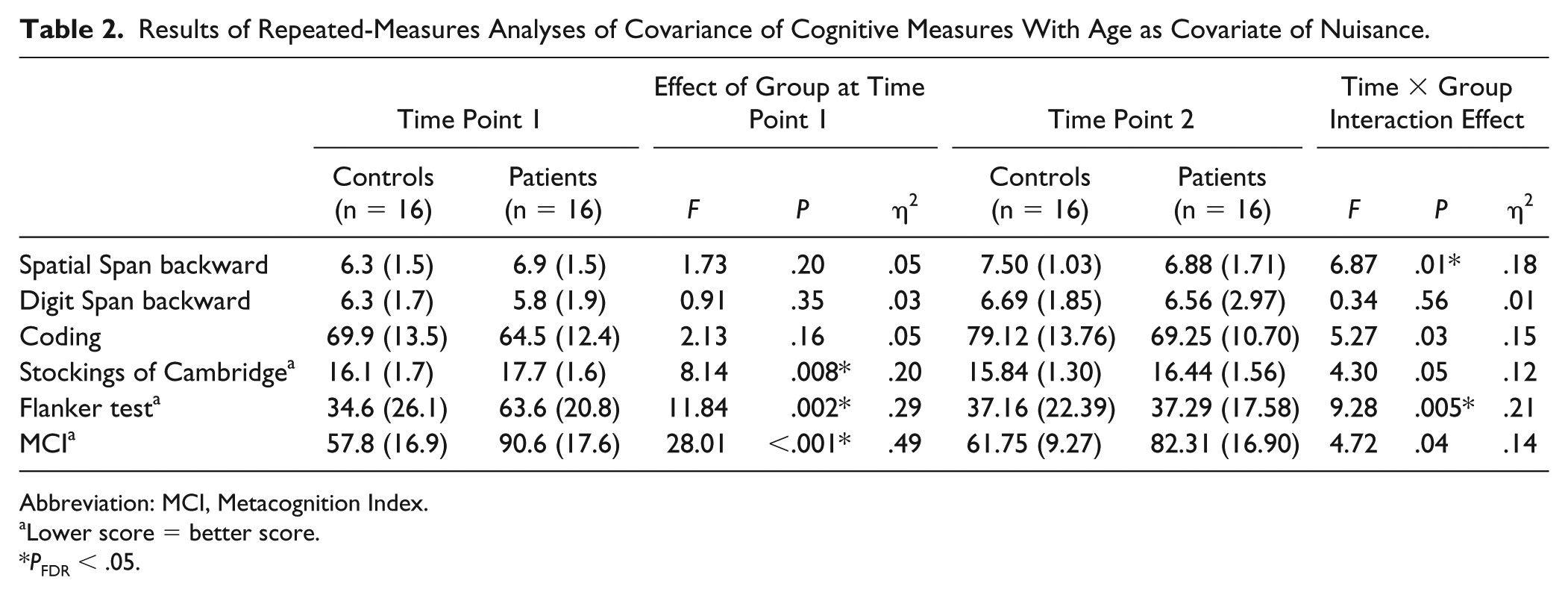
Abbreviation: MCI, Metacognition Index.
Lower score = better score.
PFDR < .05.
Longitudinal analyses demonstrated significant time by group interaction effects for the Flanker test (F(1,29) = 9.28, P = .005, η2 = .21), SOC (F(1,29) = 4.30, P = .05, η2 = .12), MCI (F(1,29) = 4.72, P = .04, η2 = 14), Coding (F(1,29) = 5.27, P = .03, η2 = .15), and the SSb (F(1,29) = 6.87, P = .01, η2 = .19). However, only the Flanker test and SSb remained statistically significant after FDR-correction (PFDR < .05). Post hoc t tests revealed a significant improvement on the Flanker test after training in young patients with TBI (P = .001), while no significant change was observed in the control group (P = .69). The significant group difference at pretraining (P = .002) disappeared after training (P = .99). With regard to the SSb, the control group improved significantly (due to practice effects) and performance of patients remained the same over time (P = .85). No significant group differences were found pre- (P = .20) or posttraining (P = .15). An overview of the results can be found in Table 2.
Cross-Sectional Analyses of White Matter
Fixel-based analyses showed significant alterations in diffusion metrics in patients with TBI when compared to healthy controls (see Figure 3). A significant decrease in FA was revealed across the brain of TBI patients, with reductions up to >40% in the genu and splenium of the CC. Other regions with significant reductions in FA included body of the CC, SLF, anterior limb of the internal capsule, sagittal stratum, posterior thalamic radiation (all bilateral) and the left cerebral peduncle. Large and spatially extensive increases in MD in patients with TBI compared with controls were also observed, partially overlapping the areas with decreased FA and group differences were more pronounced in the left hemisphere. Regions with increased MD included splenium and body of the CC, SLF, superior and posterior corona radiata and cerebral peduncles (all bilateral) and the left sagittal stratum, retrolenticular part of internal capsule, fornix, and stria terminalis. Finally, an increase in FC was found in the left SLF and the body of the CC in the TBI group compared with controls. No significant results were found in the opposite directions (controls > patient for MD and FC and controls < patients for FA, all PFWE > .05).
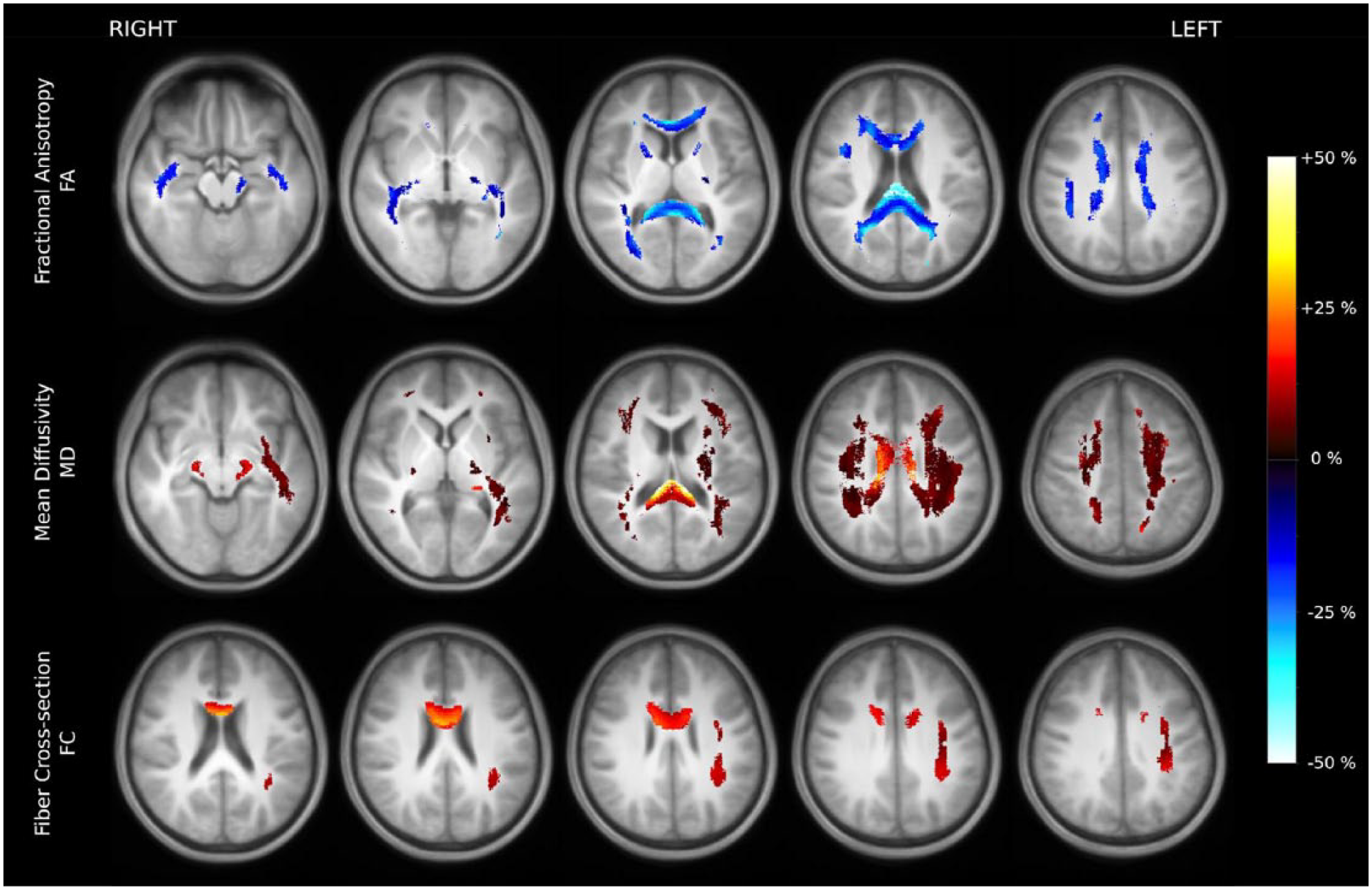
Significant alterations in fractional anisotropy (FA), mean diffucivity (MD), and fiber cross-section (FC) in patients with traumatic brain injury when compared to healthy controls. Significant fixels (family-wise error corrected P < .05) are depicted on the group-template and colored by percentage increase/decrease compared to the control group.
Training-Related Changes in White Matter
Using FBA, we failed to observe significant changes in FC, FA, or MD values in patients with TBI when comparing time point 1 with time point 2. Using our tracts-of-interest approach, results of the paired t tests revealed no significant changes in FC, FA, or MD values of the tracts (all PFDRs >.05). Using an uncorrected exploratory threshold of P < .05, only a decrease in FC was found in the left SLF I (P = .03). Mean and standard deviations of all measures before and after training are presented in the Supplemental Material.
Correlations Between Training-Induced Changes in Cognitive Functioning and Changes in White Matter
Investigating the relationship between changes in cognitive performance and alterations in diffusion metrics within patients, a significant negative correlation was found between change score on the digit span backward and change score in MD of the left SLF II and (r = −0.772, P = .001, see Figure 4A). In other words, improved scores on the Digit Span backward were associated with decreases in MD in the left SLF II. No other correlations between change scores of the cognitive tasks and change scores of FA or MD of any of the other tracts-of-interest were significant (Ps >.001).
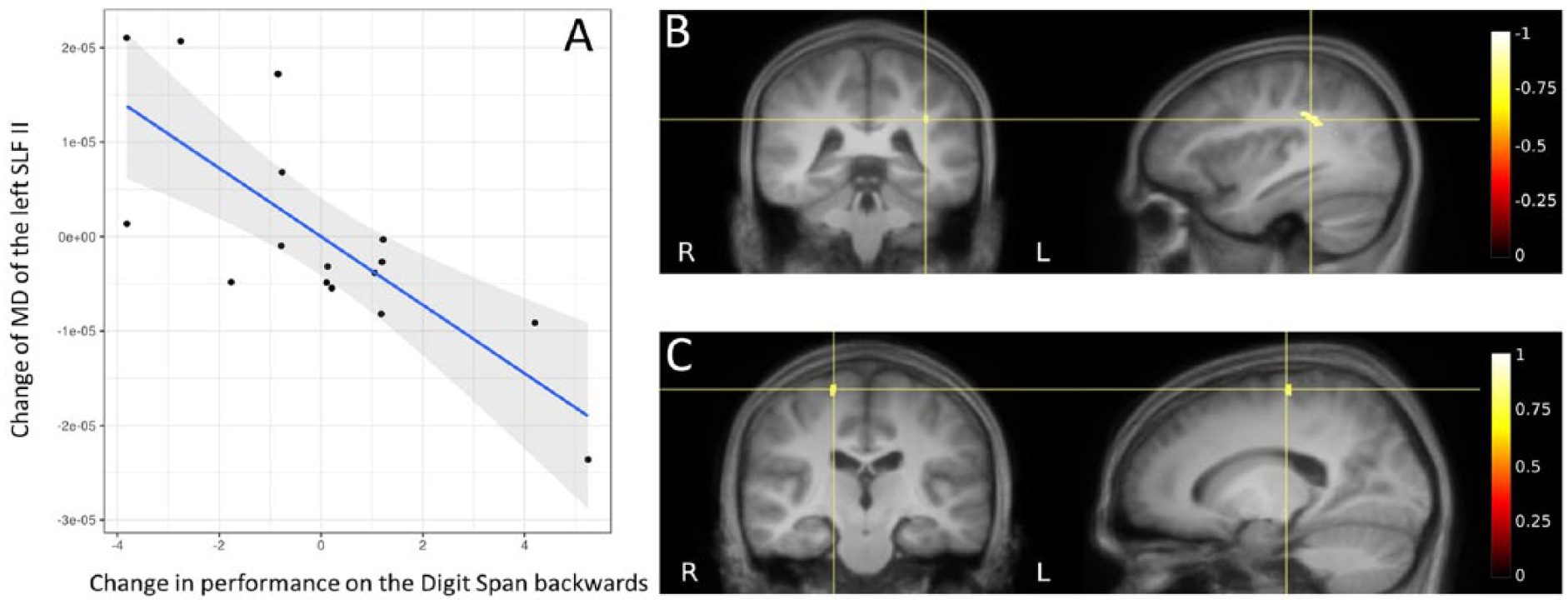
(A) Significant negative correlation between change score of mean diffusivity in the left superior longitudinal fasciculus II and the change score of digit span backward, taking age into account. (B) Significant negative correlation between the change in mean diffusivity in the left superior longitudinal fasciculus II and the change in performance on the digit span backward and (C) significant positive correlation between the change in fractional anisotropy (FA) in the right precentral gyrus and change in performance on the Coding task in patients with traumatic brain injury (TBI) after an 8-week cognitive training, taking age into account.
Complementary to the tract-of-interest-based approach, a significant negative correlation was found between changes in performance of the digit span backward and MD of a cluster of fixels in the area of the left SLF II (see Figure 4B). In addition, a positive correlation was found between changes on the Coding task and changes in FA in a cluster of fixels in the right precentral gyrus (see Figure 4C). In other words, increased FA in the right precentral gyrus coincided with improvements on the Coding task.
Discussion
Consistent with previous studies (for a meta-analysis see Babikian and Asarnow 52 ), we demonstrated cognitive impairments in young patients with TBI compared with controls before training. With regard to training-related changes in cognitive performance, our results showed cognitive improvements after training in patients with TBI compared with a passive control group of healthy controls, with effect sizes ranging from small to moderate (η2 = .01-.21). This is also in line with previous studies examining the effectiveness of computerized cognitive training interventions in patients with acquired brain injury (ABI) (η2 = .01-.11) using similar outcome measures.9,53,54
In accordance with previous research,2,55 extensive areas with decreased FA and increased MD were found in adolescent patients with TBI when compared with controls, with the strongest alterations localized in the corpus callosum. Neural plasticity is expected to be more extensive in pediatric patients with focal brain injuries compared with adults, due to the flexibility if the immature brain. 56 On the other hand, diffuse damage to the pediatric brain has been found to lead to more negative outcomes compared with mature brains. In addition, research has shown that injuries in adolescent patients seem to be followed by limited changes in white matter metrics over time compared with younger children. 57 The heterogeneity in changes over time among young patients is often considerable as there is a complex interplay of normal development, secondary injury mechanisms (eg, degenerative patterns, developmental arrest) and recovery processes.
For this study, we also calculated FC, a macroscopic specific measure representing the volumetric size of a fiber bundle. Our results revealed an increased FC in the body of the corpus callosum and the left SLF in patients with TBI compared with controls. This increase in FC is possibly the result of inflammation-related swelling after sustaining a brain injury. Extensive research has shown that inflammation can persist for years after TBI. 58 Inflammation results in increased tissue water, which can consequently lead to an increased MD and decreased FA values. 59 In line with this hypothesis, the fixels with increased FC showed overlap to a great extent with the fixels presenting decreased FA. Future research using FBA is warranted to accurately determine changes in fiber density using a scan protocol with a high diffusion weighing (b = 3000 mm2/s).
Unexpectedly, no significant training-induced alterations in white matter could be identified with the whole brain FBA approach or the tracts-of-interest method on a group level. We can conclude that the intervention had no effect on spared tissue, which would form part of a compensatory response if present. The interpretation of our findings is less clear for damaged tissue. The lack of change in FBA metrics over time may be due to an absence of training effects, but it may also reflect an inability of lesioned tissue to undergo plasticity changes or repair. Nonsignificant findings in diffusion metrics were also observed in other clinical research.19,60-62 The absent training-related changes in white matter could be the result of an insufficient effect of the cognitive training intervention. Resch et al 30 examined the effective components of cognitive rehabilitation for children and adolescents with ABI and concluded that a multicomponent rehabilitation is most promising to improve cognitive and psychosocial functioning. It is possible that BrainGames did not result in white matter changes due to the lack of compensatory training elements. Another important question is whether training-related changes in cognitive functioning are actually associated with long-lasting alterations of the underlying white matter structure. Improvements in cognition could also be driven by functional changes in the brain within the limits of the brain’s structural constraints. 63 In support of this idea, significant changes in brain activation, together with a lack of significant findings in diffusion metrics were observed in other cognitive training research in patients with brain injury.60-62 Lampit et al 61 argue that functional changes may precede structural and cognitive change. It is conceivable that longer periods of cognitive training might ultimately alter white matter structure. It is important to note that contrary to this idea, Yuan et al 64 showed significant alterations in the structural connectome of children with TBI after a 10-week cognitive training program. This cognitive intervention however, was a combination of metacognitive strategy coaching and computerized attention tasks to improve attention deficits.
Interestingly, correlations could be observed between improvements on cognitive tasks and white matter alterations. Specifically, improvements in verbal working memory was associated with a decreased MD in the left SLF. The SLF is known to connect parietofrontal cortical regions, which play a key role in working memory and executive functioning. Previous studies have shown that the SLF is subject to change as a result of cognitive training in healthy adults.16,17,28 In addition, a significant correlation was found between performance on the coding task, a measure for information processing speed, and FA in a small cluster of fixels in the precentral gyrus, corresponding to primary motor cortex. This result could be explained by the high motor load of the Coding task, that is, fine-motor dexterity, speed, and accuracy are important prerequisites to perform well on this test. The observed changes in diffusion metrics as a response to cognitive training could be caused by myelin formation or remodeling, axonal sprouting or increased axonal packing density. 65 Tissue-specific scans, such as a myelin sensitive scan sequence, 66 and diffusion metrics such as fiber density 44 could help disentangle the underlying neurobiological processes in future studies. These are exploratory findings amidst multiple comparisons; however, the effect size of the correlations were moderate to strong. These results suggest that patients who show improvements at the behavioral level, also exhibit training-related alterations in diffusion metrics. These findings could become useful biomarker candidates in the setting where no MRI changes are seen at the group level. There is high intersubject variability in the TBI group, with some patients showing training-related changes and others showing no effects at all. Additionally, our sample of patients with TBI was heterogeneous with regards to clinical profile, for example, different type/location of injury, different time since injury, different extent of damage, onset of trauma during a “critical period” of development, and so on. Heterogeneity of patient samples is inherent to TBI research and reflects the real-world heterogeneity of TBI. However, based on the correlation results, it is important to examine whether subgroups of patients (based on injury severity/location, time since injury, age at injury, functional improvements or other measures) are more sensitive to training-related change compared with others. In line with this, we recently found in the same patient sample that patients with DAI in the deep brain nuclei showed less training benefit on cognitive measures. 67
There are some limitations to this study. First, there is only a small number of participants included in this study. The effect of the training was only statistically significant for the measure of inhibition after controlling for multiple comparisons, notwithstanding the moderate effect sizes of two other measures. This is likely the result of a lack of power due to the small sample size. Secondly, the group of TBI patients is heterogeneous with regards to clinical history, which might influence the effectiveness of cognitive training. In the present study, we only included age at testing as covariate. Other possibly confounding variables such as time since injury and educational level were not included given the strong interrelationship between these variables. Future studies with larger sample sizes could examine different possible influencing factors on training effectivity. Finally, using the passive control group of typically developing children, we were able to control for practice effects on the cognitive measures. However, future studies should also include cognitive measures and diffusion metrics at both time points from an active control group of patients with TBI. This data will enable us (a) to adequately correct for spontaneous recovery, maturation, practice effects on the cognitive tasks and (b) to take expectancy effects and effects of other additional academic and/or psychosocial support into account.
Supplemental Material
Supplemental_Material – Supplemental material for Cognitive Training in Young Patients With Traumatic Brain Injury: A Fixel-Based Analysis
Supplemental material, Supplemental_Material for Cognitive Training in Young Patients With Traumatic Brain Injury: A Fixel-Based Analysis by Helena Verhelst, Diana Giraldo, Catharine Vander Linden, Guy Vingerhoets, Ben Jeurissen and Karen Caeyenberghs in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant (#01N00214) from the Special Research Fund (BOF) from the University of Ghent. BJ is a postdoctoral fellow of the Research Foundation Flanders (FWO Vlaanderen) (grant #12M3116N).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
