Abstract
Background
Exercise and cognitive training have been shown to induce neuroplastic changes and modulate cognitive function following stroke. However, it remains unclear whether hybridized exercise-cognitive training facilitates cortical activity and further influences cognitive function after stroke.
Objective
The study aimed to investigate the effects of 2 hybridized exercise-cognitive trainings on neuroplastic changes and behavioral outcomes in stroke survivors with mild cognitive decline.
Methods
This study was a single-blind randomized controlled trial. Stroke survivors were randomly assigned to 1 of 3 groups: (1) sequential exercise-cognitive training (SEQ), (2) dual-task exercise-cognitive training (DUAL), or (3) control group (CON). All groups underwent training 60 min per day, 3 days per week, for a total of 12 weeks. The primary outcome was the resting-state (RS) functional connectivity (FC) in functional magnetic resonance imaging. Secondary behavioral outcomes included cognitive and physical functions.
Results
After 12 weeks of training, patients in the SEQ group (n = 21) exhibited increased RS FC between the left occipital lobe and posterior cingulate gyrus with right parietal lobe, compared to the DUAL (n = 22) and CON (n = 20) groups. Additionally, patients in the DUAL group showed increased FC of the left temporal lobe. However, changes in behavioral outcome measures were non-significant among the 3 groups (all
Conclusions
This study highlights the distinct neuroplastic mechanisms associated with 2 types of exercise-cognitive hybridized trainings. The pre-post functional magnetic resonance imaging measurements illustrated the time course of neural mechanisms for cognitive recovery in stroke survivors following different exercise-cognitive training approaches.
Keywords
Introduction
Post-stroke cognitive impairment is highly prevalent, ranging from 20% to 60%, and it can significantly impact a person’s ability to engage in everyday activities, thereby influencing their overall quality of life. 1 This impairment encompasses a spectrum, varying from focal deficits directly stemming from infarcted region or adjacent hypoperfusion tissue to more comprehensive global cognitive decline. 2 Global cognitive deterioration can manifest as deficits in attention, memory, executive functioning, or visuoconstruction (the ability to organize and manipulate spatial information), among other cognitive domains. An increasing body of literature in the realm of cognitive rehabilitation after stroke underscores the potential advantages of targeted cognitive interventions, such as cognitive or physical exercise training, in enhancing cognitive function in stroke survivors.3,4 For instance, the sequential training paradigm involves a hybrid approach where exercise and cognitive training are administered sequentially. The incorporation of aerobic exercise prior to cognitive training has been posited to elevate arousal levels and bolster memory consolidation and retrieval mechanisms. Conversely, the dual-task training paradigm involves the simultaneous execution of exercise and cognitive tasks. This approach poses heightened cognitive demands, as it necessitates the integration and coordination of 2 tasks concurrently. Dual-task training, thus, presents a more intricate cognitive challenge.
A recent systematic review has undertaken an investigation into the impact of combined interventions involving aerobic physical exercise and cognitive training, administered either sequentially or concurrently as a dual task, on cognitive function among stroke patients. 5 The outcomes of this review reveal certain indications of heightened cognitive benefits associated with the implementation of combined interventions. Noteworthy enhancements have been observed in Montreal Cognitive Assessment (MoCA) scores and performance on the Stroop task, which assesses inhibitory capacity. In addition to the cognitive advancements, the review also sheds light on improved motor functions, as evidenced by outcomes such as the 6-minute walking test, subsequent to the implementation of combined interventions. While the combined training paradigm has demonstrated its efficacy in enhancing cognitive and physical functions among stroke survivors, a comprehensive understanding of the underlying mechanistic rationale for the benefit of such hybridized training remain elusive. The necessity to delve into neuroimaging analyses is apparent, as they can elucidate the intricate neural mechanisms that come into play during combined interventions in this population. Existing literature underscores the need for neuroimaging investigations to uncover the intricate neural mechanisms underpinning the effectiveness of combined interventions among stroke survivors.5,6 Previous research has yielded insights into distinct neural mechanisms associated with singular interventions, such as computer-based cognitive or aerobic exercise, within the stroke population. For instance, a 10-week computer-assisted cognitive training program for stroke patients exhibited augmented hippocampal functional connectivity (FC) in the frontal and left parietal lobes. 7 Similarly, integrated cognitive therapy yielded increased hippocampal FC in the prefrontal gyrus and the default-mode network (DMN) among stroke patients, highlighting the potential of computer-based cognitive rehabilitation in modulating neural networks in this population. 8 Conversely, moderate-intensity aerobic treadmill exercise has been shown to activates neural circuitry in the midbrain and cerebellum, alongside frontal, temporal, and parietal cortical regions, among stroke survivors. 9
However, the underlying neural mechanism responsible for the cognitive improvements resulting from combined exercise-cognitive training in individuals with post-stroke cognitive impairment remain elusive.7,10 –12 Despite the growing body of research in this area, certain key questions remain unanswered, including the potential of cognitive training to induce cortical activation and FC alterations in specific brain regions. Additionally, there is interest in whether various forms of exercise-cognitive training might yield distinct patterns of neural plasticity. Furthermore, it is noteworthy that recent systematic review included merely 5 randomized controlled trials (RCTs) published within the years 2019 and 2020, which underscores the burgeoning interest in this area of investigation. However, the limited number of studies highlights the necessity for more robustly designed RCTs that comprehensively assess the combined impact of exercise-cognitive training on cognitive function in stroke patients. 5 In view of these considerations, the present study was undertaken with the aim of shedding light on these aspects. Specifically, the study sought to explore the effects of 2 hybridized exercise-cognitive training interventions on neuroplastic changes and resultant behavioral outcomes among stroke survivors with mild cognitive decline.
Methods
Design
The present study was designed as an interventional, single-blind RCT, conducted within the geographical context of Taiwan. Ethical approval for the study protocol was obtained from the Institutional Review Board (Approval Number: KMUHIRB-F(II)-20170040) of Kaohsiung Medical University Chung-Ho Memorial Hospital, located in Kaohsiung, Taiwan. The study protocol is duly registered with clinical.trials.gov under the registration identifier NCT03230253. Prior to participation, all enrolled patients were furnished with comprehensive written informed consent, detailing the study’s objectives, procedures, potential benefits, and any associated risks.
Patient Population
The study’s inclusion criteria encompassed individuals who met the following conditions: (1) a documented history of stroke occurring at least 6 months prior to enrollment; (2) an age range spanning from 20 to 80 years; (3) a Mini-Mental State Examination (MMSE) score below 28 or a MoCA score below 25; (4) demonstratable ability to comprehend and adhere to study instructions; (5) sufficient cardiopulmonary capacity to undertake physical activity; (6) ability to ambulate, either independently or with the assistance of mobility aids. Individuals were excluded from participation if they met any of the following criteria: (1) a medical history characterized by instability, such as recent myocardial infarction, potentially impeding their participation; (2) concurrent presence of other neurological disorders (eg, Parkinson’s disease, amyotrophic lateral sclerosis, multiple sclerosis); (3) ongoing involvement in other concurrent research projects; (4) pregnancy; (5) presence of contraindications to undergoing functional magnetic resonance imaging (fMRI) scanning, including claustrophobia, seizure history, possession of a pacemaker, existence of metallic elements (such as steel pins or plates) within the body or eyes, and severe obesity. Prospective participants were subject to these criteria to ensure that the study cohort exhibited homogeneity in terms of their post-stroke cognitive impairment status and their eligibility for the experimental interventions and assessments.
Randomization
Utilizing the Research Randomizer web-based tool, a research assistant performed randomization, which was stratified based on participants’ baseline cognitive function as determined by the MMSE. Participants were allocated randomly in a 1:1:1 ratio to the sequential exercise-cognitive training (SEQ), dual-task exercise-cognitive training (DUAL), or control group (CON), according to a predefined allocation table.
Interventions
All trainings sessions were consistently administered for 60 minutes per day, spanning 3 days per week, for a duration of 12 weeks. During the intervention, participants’ vital signs and the Borg Perceived Exertion Scale 13 were monitored and recorded. The choice of the 12-week testing window was influenced by prior research investigating cognitive function post-stroke. This timeframe is well-established within stroke research as a suitable period to capture significant alterations in cognitive function.14 –17
SEQ Training
Participants assigned to the SEQ group commenced their session with a 30-minute stationary bike exercise. This session encompassed a 3-minute warm-up phase, succeeded by 25 minutes of aerobic training, and concluded with a 2-minute cool-down phase. During the aerobic training segment, participants were guided to maintain a target heart rate falling within the range of 40% to 70% of their maximal heart rate (HRmax). Subsequent to the cycling regimen, participants engaged in a 30-minute cognitive training session utilizing the commercially available computer-based program,
DUAL Training
Participants assigned to the DUAL group engaged in simultaneous stationary bike cycling and computer-based cognitive training. The training protocol paralleled the principle employed in the SEQ group; however, the intensity of the training was tailored to accommodate the individual abilities of each participant. Specifically, participants in the DUAL group were tasked with concurrently performing aerobic exercise while engaging in cognitive tasks. The target heart rate during the aerobic segment was established at 40% to 70% of the maximal heart rate, calculated using the formula (208 − 0.7 × age).
18
As participants exhibited progress in their performance over practice sessions, the exercise intensity was systematically augmented. The monitoring and recording of vital signs, alongside the utilization of the Borg Perceived Exertion Scale, were consistently administered during each session to gauge participants’ exertion levels. Within the context of the stationary bike exercise, participants simultaneously tackled cognitive tasks using the
Recognizing that dual-task training inherently demands the integration of 2 tasks, thus imposing greater cognitive demand compared to singular tasks. To alleviate potential frustration, initially simplified versions of the cognitive tasks were introduced. The complexity of both exercise intensity and cognitive task difficulty was incrementally heightened as participants displayed advancements in their dual-task performance. This adaptive approach aimed to optimize participants’ cognitive and physical progress while ensuring their engagement and satisfaction throughout the training program.
Control Training
Participants assigned to the CON underwent a 60-minute health-related rehabilitation program during each session. The program encompassed non-aerobic physical exercise, such as muscle stretching and range of motion exercises as well as relaxation techniques. In addition, participants engaged in unstructured cognitive-related rehabilitation activities. These included activities like watching health-related videos or read newspapers or magazines, followed by answering the content-related questions posed by the therapist. The control training approach aimed to ensure that participants in the CON received comparable attention and engagement as those in the intervention groups, while the exercises were tailored toward non-aerobic physical activity and unstructured cognitive stimulation.
Primary Outcome and Experimental Procedure
The primary outcome is the resting-state (RS) FC and neural activation within the frontal, parietal, and occipital lobes. These assessments were conducted through fMRI scans carried out prior to and following the intervention. To achieve this, participants underwent fMRI scans during periods of quiet rest.
fMRI Data Acquisition and Scanning Protocol
MRI data were acquired prior to and after the interventions using a 3-Tesla MRI scanner (MAGNETOM Skyra; Siemens) featuring an 8-channel phased-array head coil. The scanning took place at Chang Gung Memorial Hospital in Kaohsiung, Taiwan. During the fMRI scans, participants were given specific instructions to maintain their eyes open and to remain completely motionless. Additionally, participants were guided not to engage in any specific thoughts during the RS functional MRI (RS-fMRI) scan. To minimize head movement and ensure imaging accuracy, comfortable foam padding was employed. Furthermore, earplugs were administered to mitigate the impact of scanner noise.
In consideration of the absence of a standardized fMRI acquisition and analysis protocol for stroke populations, our study adhered to locally established parameters. Notably, the T1-weighted magnetization-prepared rapid acquisition gradient echo sequences were utilized to capture high-resolution 3D structural brain images, with parameters including a repetition time/echo time/inversion time of 3500 ms/4.73 ms/1100 ms, acquisition of 192 sagittal slices with a voxel size of 1 × 1 × 1 mm³, and a flip angle of 7°. These parameters provide a contextual framework for our study’s imaging methodology, acknowledging the inherent variability in fMRI practices across research contexts.
RS-fMRI data were obtained through the utilization of a T2*-weighted gradient-echo echo planar imaging sequence, employing specific parameters to ensure data accuracy. These parameters encompassed a repetition time of 2500 ms, an echo time of 27 ms, a field of view measuring 200 mm × 220 mm, a flip angle of 77°, a matrix size of 64 × 64, a slice thickness of 3.4 mm, and a slab encompassing 43 slices to cover the entire brain. A total of 200 measurements were acquired during an interval of 8 minutes and 27 seconds. Participants were explicitly instructed to maintain open eyes and a motionless disposition throughout the imaging process, thereby fostering consistent data acquisition conditions.
fMRI Data Processing
The processing of fMRI data was executed using the DPARSF advanced edition (Data Processing Assistant for RS fMRI) within the MATLAB R2019a environment (MathWorks, Natick, MA, USA). The initial 10 volumes of each functional time series were excluded from analysis. The residual fMRI images underwent a series of recommended procedures, comprising realignment, co-registration, segmentation, and spatial normalization to Montreal Neurological Institute space (www.mni.mcgill.ca). The individual images were subsequently resliced into 3-mm3 isotropic voxels. To account for confounding factors, nuisance variables encompassing the averaged signals of the ventricular, white matter, and entire brain regions, alongside Friston 24 regressors, were regressed out from the fMRI data. Subsequently, a band-pass frequency filter (0.01-1.0 Hz) was applied to mitigate low-frequency drift and high-frequency noise. Finally, the filtered blood oxygen level—dependent images underwent partial smoothing using an isotropic Gaussian kernel with a full width at half maximum of 6 mm.
The images volumes underwent additional segmentation into 246 regions of interest (ROIs) utilizing the validated and reported automated anatomical labeling template (AAL), as established by Fan et al.
19
This template effectively divides 210 cortical and 36 subcortical subregions within the cerebral structure. The subsequent step involved the calculation of FC between these ROIs. This computation was based on the assessment of signal similarities within the time series by using Pearson’s correlation coefficients. Consequently, a FC matrix emerged, characterized by 6670 connections ([246 × 245]/2) within each subject. For normalization purposes, Fisher’s
The statistical analysis of FC was executed using MATLAB R2019a software. Various contrasts were scrutinized through paired-sample
Secondary Outcomes
Secondary outcomes encompassed assessments of cognitive and physical functions before and after the intervention, enabling a comprehensive evaluation of the intervention’s impact on these domains.
Cognitive Functions
Montreal Cognitive Assessment
General cognitive functions were evaluated using the MoCA, a comprehensive assessment tool encompassing multiple cognitive domains and yielding a total score of 30. The MoCA has been shown to be a feasible tool to evaluate the global cognitive function in a large population of patients with stroke. 20 The reliability and validity have been established to be good to excellent for patients with cerebrovascular diseases. 21
Wechsler Memory Scale—Third Edition (WMS—III)
The WMS—III, a standardized and reliable neuropsychological assessment tool, was employed to evaluate visuospatial and memory functions. 22 Subtests from WMS—III, including faces recognition, verbal paired associates, word lists, and spatial span, were administered to assess immediate, delayed, and working memory capabilities. 23 For instance, the faces recognition test entailed the presentation of 24 human face images, subsequently requiring participants to recognize and recall these faces. The verbal paired associates test involved memorizing 8-word pairs, while the word lists test encompassed the recollection of 12 words presented in a list format, both immediately and after a time interval of 25 to 35 minutes. The spatial span test necessitated participants to sequentially touch spatially located blocks, following the instructor’s sequential or reversed order. The test-retest reliability of these subtests has been determined to be moderate within the context of community-dwelling adults. 24
Physical Functions
Timed Up and Go (TUG)
The TUG assessed the dynamic balance ability and mobility. The participants were tasked with standing up from a chair, walking a distance of 3 m, performing a turn, returning to the chair, and sitting down. The time to complete the TUG test has been shown to be a good indicator to detect potential fallers in frail elderly. 25 The test-retest reliability of TUG on individuals with stroke was excellent. 26
6-Minute Walk Test (6MWT)
The 6MWT gauged participants’ endurance and mobility by quantifying the maximum distance covered within a 6-minute timeframe. The test-retest reliability and responsiveness have been established to be high for patients with chronic stroke. 27
Sample Size Estimates
Our sample size determination was guided by pertinent research studies7,28 focusing on a comparable stroke patient cohort and employing analogous behavioral outcome measures. We extracted Cohen’s
Under the premise of a 5% significance level, 90% statistical power, and an effect size f of 0.25, our projection yielded a sample size of 20 participants per group. Anticipating a potential attrition rate of 20% during the 6-month follow-up, our comprehensive recruitment strategy outlines the inclusion of a total of 75 participants, with each of the 3 groups accommodating 25 individuals.
Statistical Analyses
For efficacy assessment, an intention-to-treat analysis was employed. Continuous-scale outcomes underwent an initial assessment for normality, with appropriate log-transformations applied when needed. Baseline characteristics and outcome measures among the groups were analyzed using Analysis of variance and χ2 tests. To ascertain the intervention’s impact across the 3 groups, analysis of covariance (ANCOVA) was conducted. This adjustment accounted for baseline measures, furnishing an unbiased estimation of mean group differences. A significance level of .05 was adopted for all comparisons. In addition to
Results
Baseline Characteristics
Initially, 84 stroke survivors were identified and referred from the department, with 16 individuals subsequently deemed ineligible during the screening process. Ultimately, 68 stroke survivors with cognitive impairment were stratified based on their MMSE scores and subsequently randomized into the SEQ, DUAL, and CON groups. Over the 12 weeks training period, a total of 5 participants withdrew from the study due to health-related concerns or changes in decision. In total, data analysis was conducted on 63 stroke survivors, distributed among the SEQ (n = 21), DUAL (n = 22), and CON (n = 20) groups, all exhibiting mild cognitive impairment. The consolidated standards of reporting trials (CONSORT) flow diagram is depicted in Figure 1. Notably, baseline characteristics were well-matched across the 3 groups, with no significant discrepancies in baseline demographic attributes observed among the groups (
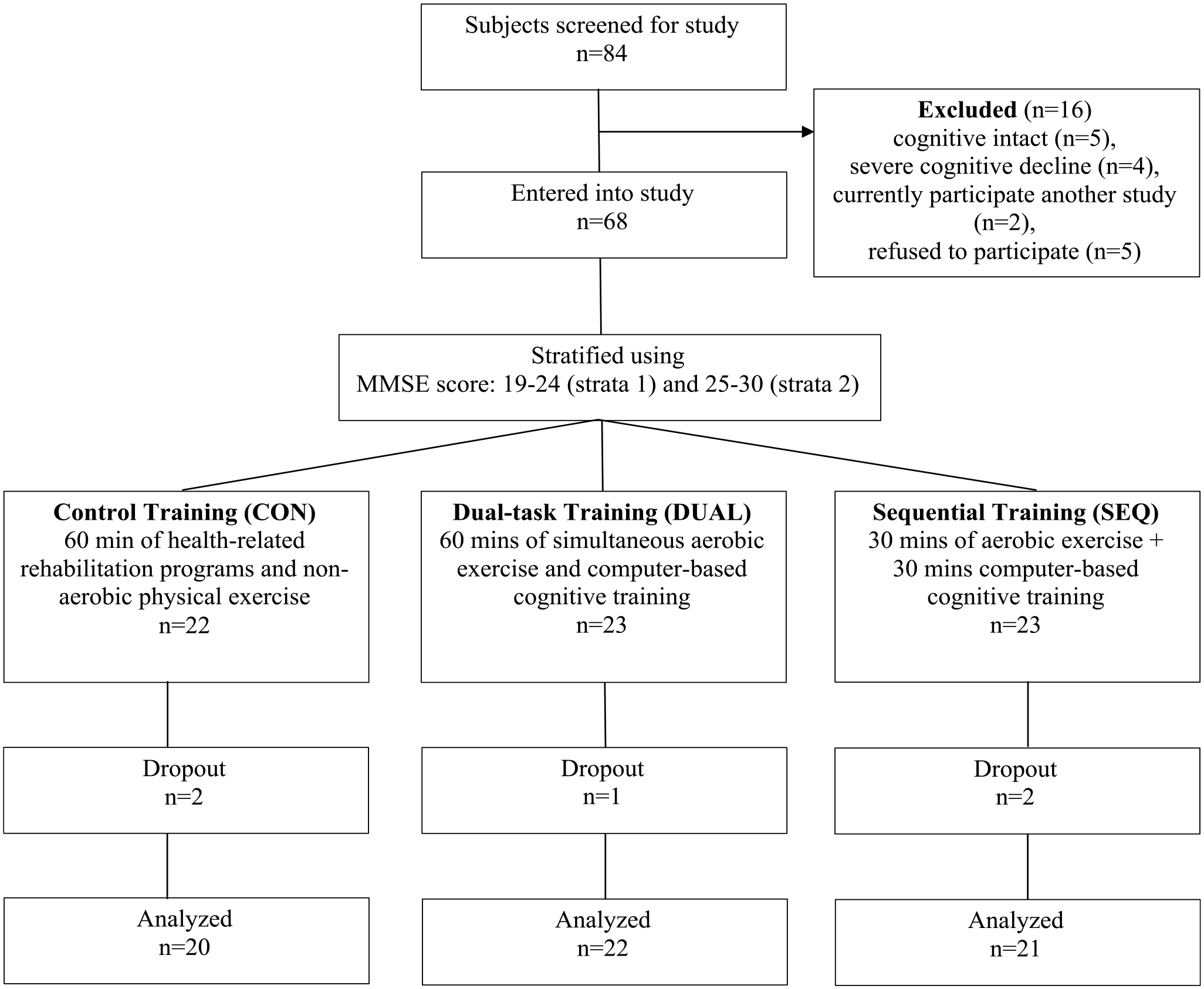
CONSORT diagram showing the flow of participants through the trial.
Demographic and Clinical Features of the Participants.
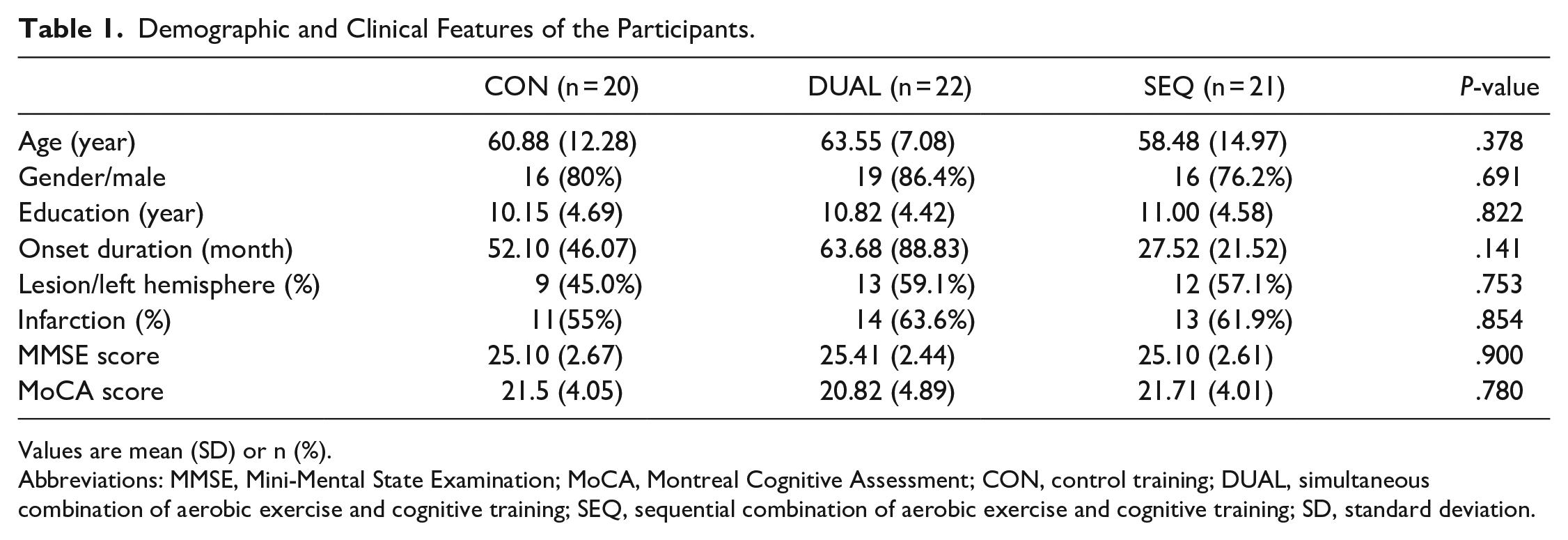
Values are mean (SD) or n (%).
Abbreviations: MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; CON, control training; DUAL, simultaneous combination of aerobic exercise and cognitive training; SEQ, sequential combination of aerobic exercise and cognitive training; SD, standard deviation.
Primary Outcome Measures
Figures 2 and 3 present the data illustrating alterations in FC between baseline and the 12-week mark. Notably, the SEQ group demonstrated higher FC between the left middle occipital gyrus (Area 199) and the right parietal lobe, specifically, the posterior cingulate gyrus, in comparison to both the CON and DUAL groups (
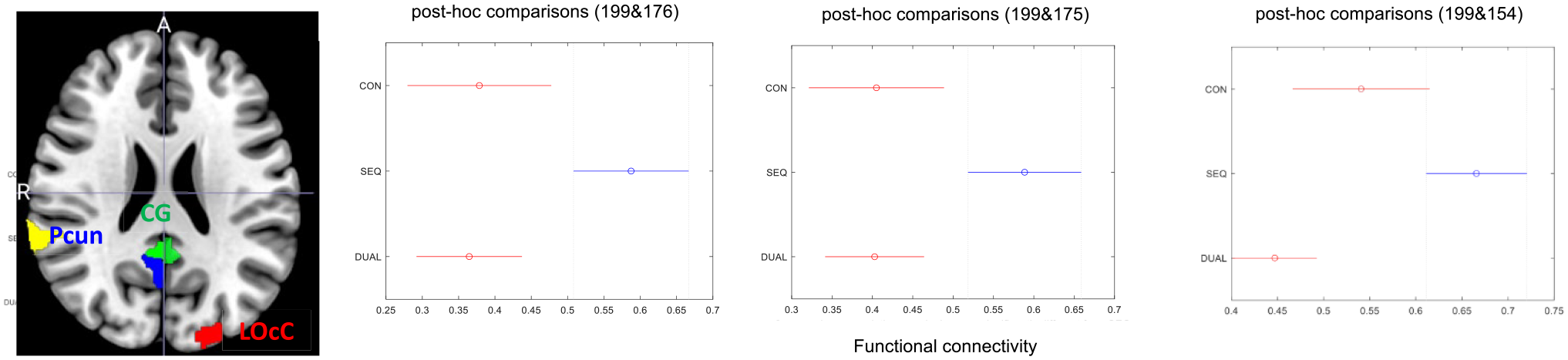
Functional magnetic resonance imaging scans show a significantly increased in resting state functional connectivity (RS FC) of the middle occipital gyrus (area 199) in patients with stroke in the SEQ group between the left occipital lobe (middle occipital gyrus) with posterior cingulate gyrus and right parietal lobe (right inferior, right superior; areas 176, 175, and 154) at 12 weeks compared to DUAL and CON group.
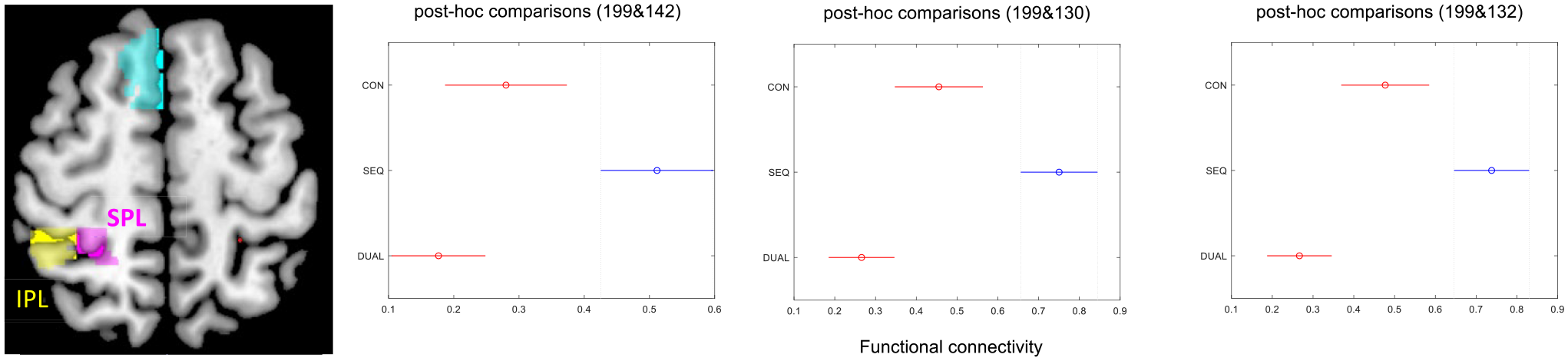
Functional magnetic resonance imaging scans show a significantly increased in resting state functional connectivity (RS FC) of the middle occipital gyrus (area 199) in patients with stroke in the SEQ group between the left occipital lobe (middle occipital gyrus) with parietal lobe (right inferior, right superior; areas 142, 130, and 132) at 12 weeks compared to DUAL and CON group.
Secondary Outcome Measures
The secondary outcomes include evaluation of cognitive and physical functions prior and following the training interventions. The ANCOVA indicated no statistically significant differences among the 3 groups in the post-training phase (all
Summary Statistics From Linear Mixed Effects Models of Behavioral Outcome Measures. Pair
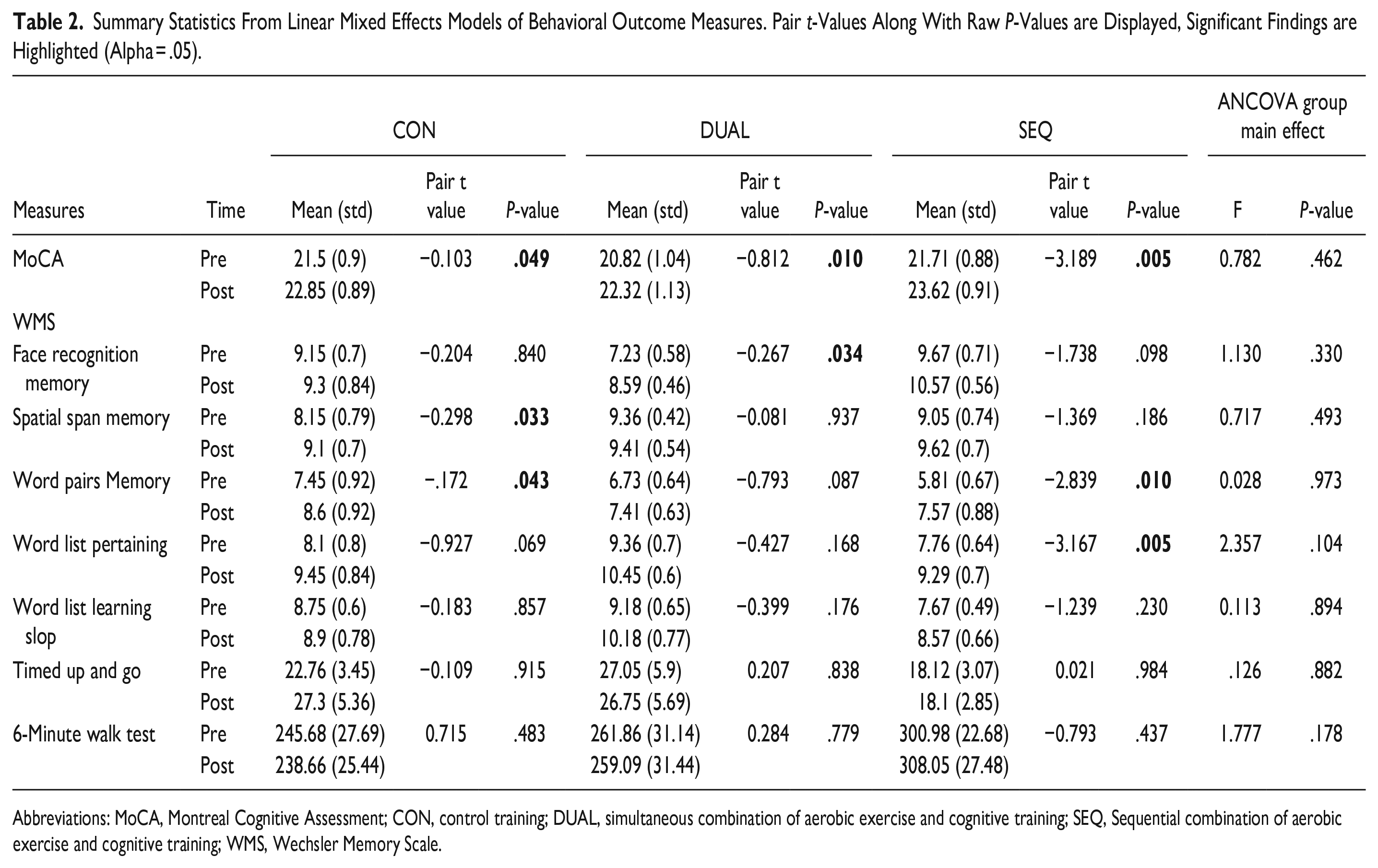
Abbreviations: MoCA, Montreal Cognitive Assessment; CON, control training; DUAL, simultaneous combination of aerobic exercise and cognitive training; SEQ, Sequential combination of aerobic exercise and cognitive training; WMS, Wechsler Memory Scale.
Notably, the SEQ group exhibited enhancements across several cognitive function outcomes after 12-week training, including MoCA (
Discussion
The present study, known as the hybrid exercise-cognitive training (HECT) trial, represents a pioneering endeavor as the first RCT aimed at evaluating the impact of 2 hybridized exercise-cognitive interventions on both brain plasticity and behavioral outcomes in stroke survivors. After 3-month of hybrid interventions, the SEQ group demonstrated higher FC between the left middle occipital gyrus and right parietal lobe, including the posterior cingulate gyrus, in comparison to both the CON and DUAL groups. Furthermore, the SEQ demonstrated higher FC between the left frontal lobe and bilateral superior temporal gyrus and sulcus. In contrast, participants in DUAL group showed increased FC within the left temporal lobe, including the inferior temporal gyrus, fusiform gyrus, and parahippocampal gyrus. While clinical outcome measures exhibited pre-post intervention effects, no significant differences were detected among the groups. The utilization of pre-post fMRI assessments offered insight into the temporal dynamics of neural mechanisms associated with distinct cognitive training approaches in stroke survivors, contributing to cognitive recovery.
Changes in FC in Strokes Survivors After Exercise-Cognitive Hybrid Trainings
The outcomes of our study have unveiled noteworthy insights into the effects of a 3-month regimen of hybrid interventions, specifically within the SEQ group. This group displayed a significantly higher levels of FC between the left middle occipital gyrus and the right parietal lobe, along with the posterior cingulate gyrus, differentiating itself from both the CON and DUAL counterparts. This novel discovery underscores the distinct potential of SEQ to elicit targeted neural responses conductive to bolstering cognitive recovery through the intricacies of neuroplasticity. These neuroplastic changes potentially contribute to the augmentation of cognitive processes such as perception and memory. Specifically, the occipital lobe serves as a hub for visual processing, encompassing visuospatial tasks, depth and distance perception, color determination, as well as memory consolidation. 30 Furthermore, our study revealed higher FC in the SEQ group between the left frontal lobe and bilateral superior temporal gyrus and sulcus. This phenomenon resonates with neuroanatomy as the posterior cingulate cortex features prominently within the DMN, displaying increased activity during the recollection of autobiographical memories and planning. 31 Moreover, the precuneus, located at the posteromedial region of the parietal lobe, not only constitutes a vital node within the DMN but also performs a pivotal role within the intricate neural framework of self-consciousness, engaging in self-referential mental representations during periods of rest. 32 Additionally, the parietal lobes are functionally bifurcated, with 1 facet dedicated to sensory perception and integration, while the other specializes in the synthesis of spatial coordinates to represent the external world. Although FC offers valuable lens into the flow of information and neural connectivity, we acknowledge its limitations and therefore have supplemented our study with a comprehensive array of neurocognitive assessments. We recognize the imperative for future research endeavors to further validate and fortify the correlation between FC variations and neurocognitive enhancements.
Multiple factors may underlie the superior effects observed within the SEQ group, warranting exploration. First, the exercise training potential to bolster cell proliferation and neurogenesis might prepare the brain for heightened synaptic plasticity upon subsequent cognitive enrichment, suggesting the preclude of exercise
Physical and Cognitive Trainings Did Not Yield Significant Group Effects in Behavioral Outcome Measures
Contrary to our initial hypotheses, the analysis did not reveal significant group effects in terms of behavioral outcome measures among the 3 participant groups. However, noteworthy within-group changes in cognitive function were observed post-training. Specifically, the SEQ group exhibited improvements in various aspects of verbal memory, as evidenced by enhancements in the word list subtest and word pair memory subtests after the 3-month training regimen, while such improvements were not significant in the DUAL and CON groups. Although no significant group effects were found, trends indicated that the SEQ group might have had a more pronounced impact on cognitive outcomes compared to the DUAL and CON groups. This lack of significant group effects could stem from a few factors. Firstly, the sensitivity of the selected behavioral outcome measures to subtle changes might be limited, despite their established psychometric properties. These measures might not fully capture the intervention effects observed at the neurophysiological level. Secondly, the targeted cognitive domains of the computer-based cognitive training in our study—such as visuospatial processing, attention, memory, and executive function—could have influenced cognitive outcomes differently across the groups. Notably, the SEQ group showed greater improvements in memory subtests of the WMS-III compared to the DUAL and CON groups, implying that the distinctive training components might have contributed to differential cognitive outcomes. Lastly, while no significant group effects were observed, significant improvements in several cognitive outcome measures were discernible in the SEQ group. This suggests that the training intensity employed in this study might not have been potent enough to differentiate behavioral changes among the distinct hybrid training modes for stroke survivors with cognitive decline. Prior research on the dose-response relationship between cognitive and physical training in stroke recovery has yet to establish an optimal training intensity for cognitive enhancement. Consequently, this study lays the foundation for future interventions to explore higher-intensity training paradigms that could potentially elicit more substantial behavioral improvement in stroke patients with cognitive decline. While group effects were not realized, sequential hybrid training demonstrated particular advantages bolstering certain cognitive outcomes among stroke survivors. Subsequent investigations should focus on refining intervention protocols and discerning the ideal training intensity to foster cognitive amelioration in this specific population.
Clinical Relevance
The clinical significance of our findings is pivotal for their meaningful interpretation in the context of patient outcomes and treatment efficacy. While statistical significance discerns group differences, clinical significance delves into whether these differences hold importance in patient outcomes and treatment effectiveness. In a previous investigation focusing on this specific cohort, we thoroughly assessed the psychometric and clinimetric attributes of the psychometric and clinimetric properties of the MoCA. Notably, minimal clinically important difference (MCID) values were estimated as 1.22 (anchor-based method) and 2.15 (distribution-based method), indicating these benchmarks as indicative of clinically meaningful changes for stroke survivors. 36 In our current study, we analyzed the pre-post changes in 3 groups: CON (21.5-22.85, diff = 1.35), DUAL (20.82-22.32, diff = 1.50), and SEQ (21.71-23.62, diff = 1.91). According to the anchor-based method, all 3 groups exceeded the threshold of 1.22, indicating a clinically important change. However, based on the distribution-based method, none of the groups surpassed the threshold of 2.15. It is crucial to consider various factors that may influence the estimation of MCID values, including age, stroke type, baseline severity, and intensity of rehabilitative intervention. 36 By estimating MCID values specific to stroke survivors using both anchor-based and distribution-based methods, we determined the thresholds that represent clinically meaningful changes in cognitive function. Meeting or exceeding these thresholds indicates that the observed changes in cognitive performance are considered significant from a clinical standpoint. Understanding the clinical relevance helps healthcare professionals and researchers interpret the practical implications of the study findings. It provides insights into whether the observed changes have meaningful effects on patients’ overall functioning, treatment decisions, and prognosis.
Study Limitations
Factors limiting our interpretation should be acknowledged, and further investigation is required. Our study may be restricted by the small sample size within each group, as neuroimaging studies requires substantial statistical power. 37 Further research with a larger sample size is encouraged. Nonetheless, the study sample examined in the present study could reflect distinct FC induced by different hybrid interventions for cognitive training, suggesting that unique combination of cognitive training paradigm does have an optimistic impact on brain activation. Another limitation could be the possibility that the concomitant use of certain medication for treatment of other health issues, such as antihypertensive drugs that might be associated with reducing cognitive impairment. 38 To mitigate this potential confounder, future trials could employ sub-group analyses with larger sample sizes to attenuate such confounding effects. Finally, secondary outcome measure should have included divided attention as dual-task outcome domains to explore the potential efficacy of dual-task training. Also, based on the theory of dual-task interference, the superior effect of dual-task training may not be observed in the short term because of the interaction of cognitive and physical tasks. 6 Future studies with larger sample sizes and longer follow-ups are needed to examine the long-term effect of dual-task training.
Conclusions
Cognitive deficits after stroke are highly associated with functional disability, institutionalization rate, mortality, and increased risk of developing dementia; targeted treatment to reserve cognitive ability is emergent. This clinical trial launched 2 novel hybrid exercise-cognitive trainings and aims to move the field forward by comparing the intervention effects of different combination approaches—combining physical exercise and cognitive training sequentially or concurrently—with active control of training. In conclusion, this randomized controlled study provides the first evidence that a sequential combination paradigm tended to be a better program than the concurrent combination one. It activated the link between the occipital cortexes and the cingulate gyrus, where these links are associated with visual processing and visual perception. Our preliminary findings underscore the feasibility and potential of sequential hybrid training to foster central plasticity in mild cognitive decline after stroke. The addition of neuroimaging analyses shed light on the specific neural mechanisms involved in the different intervention paradigm. This study lays the foundation for clinical insights, demonstrating neurobiological adaptation post-specific hybrid training, thereby guiding personalized cognitive rehabilitation strategies according to individual cognitive profiles and needs. The hybrid training modalities appear as promising non-pharmacological tools capable of stimulating latent or underutilized brain circuits in chronic stroke survivors.
Footnotes
Acknowledgements
We thank and acknowledge the support from the research team and the staff from our recruiting sites who contributed to setting up and running this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Science and Technology Council (109-2314-B-182-027-MY3, NSTC111-2314-B-182-037-MY3), Chang Gung Memorial Hospital Research Council (BMRP553), National Health Research Institutes (NHRI-EX112-11105PI), Healthy Aging Research Center at Chang Gung University.)
