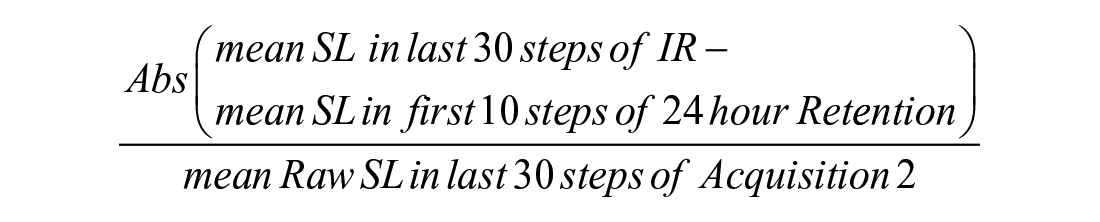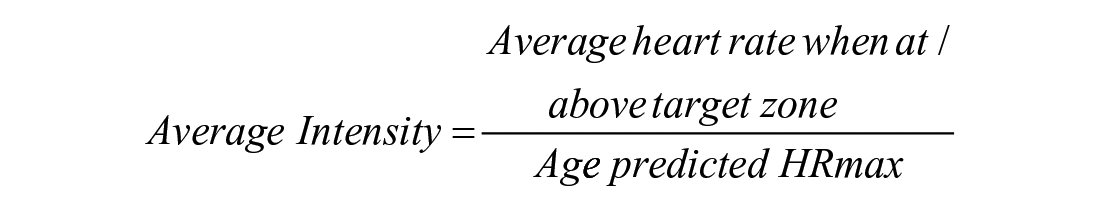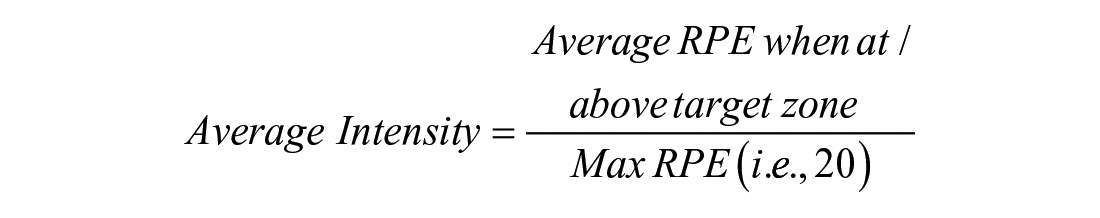Abstract
Background
Exercise priming, pairing high intensity exercise with a motor learning task, improves retention of upper extremity tasks in individuals after stroke, but has shown no benefit to locomotor learning. This difference may relate to the type of learning studied. Upper extremity studies used explicit, strategic tasks; locomotor studies used implicit sensorimotor adaptation (split-belt treadmill). Since walking is an important rehabilitation goal, it is crucial to understand under which circumstances exercise priming may improve retention of a newly learned walking pattern.
Objective
Determine the impact of exercise priming on explicit, strategic locomotor learning task retention in chronic stroke survivors.
Methods
Chronic stroke survivors (>6 months) performed 2 treadmill walking sessions. Visual feedback was used to train increased step length. Participants were assigned to control group (no exercise), continuous exercise (5 minutes high intensity), or long-interval exercise (15 minutes high/moderate intervals). After day 1 learning, participants either rested or performed exercise. On day 2, retention of the learned walking pattern was tested.
Results
All groups learned on day 1 (P < .001). The 2 priming groups showed significant changes in blood lactate and heart rate after exercise priming, the resting control group did not (P < .001). On day 2, there was no significant between-group difference in cued or un-cued task retention (P = .963 and .287, respectively).
Conclusions
Exercise priming did not affect retention of an explicit locomotor task in chronic stroke survivors. Further work should explore subgroups of individuals for whom priming may have selective clinical benefit to locomotor learning.
ClinicalTrials.gov Identifier: NCT03726047
Introduction
Stroke has a devastating impact on sensory motor function, especially walking.1 -3 Many stroke survivors report a goal of improving their walking during rehabilitation. 4 Yet 6 to 12 months after stroke, over 50% of survivors have persistent deficits that lead to decreased independence, 5 including in walking and caring for themselves. Regaining and improving movement after stroke, including walking, is dependent on creating persistent changes in the movement (motor learning) through practice and neuroplastic changes.6 -11 However, individuals with stroke show deficits in motor learning,12 -14 making it more challenging to maximize their benefit from rehabilitation interventions. Given the importance of walking to everyday function and the ramifications of stroke-related walking deficits to safety and independence,15,16 it is crucial to understand factors that may enhance locomotor learning in individuals with chronic stroke.
Exercise priming (pairing a bout of intense exercise with a motor learning task) has been studied as a potential mechanism to improve motor learning and neuroplastic changes beyond the effects of practice alone. This proposed phenomenon has been investigated both in neurotypical adults17 -22 and in individuals after stroke.6,23 -26 While possible mechanisms for the effects of exercise priming are not fully understood, the release during exercise of brain-derived neurotrophic factor (BDNF) and other chemical mediators6,18,27,28 is thought to play a key role.
Several common themes have emerged from this body of work so far. First, the benefit of priming to learning/retention may not depend on the exact modality of exercise (such as treadmill walking/running versus cycling),29 -31 as long as the physiologic stimulus is of an intensity greater than 70% of age-predicted maximal heart rate, HRmax.20,21,24,29,32 Additionally, most of the studies showing improvement in motor learning/retention with exercise priming have found a greater benefit to task retention (measured between 24 hours and several days after first task exposure) than to the initial learning process.17,19,21,23,24,27,33,34 Enhancement to learning retention after exercise priming appears to be greatest when the exercise is delivered after the initial task learning,23,29,34 during memory consolidation. In some cases, the response to priming 24 hours or longer after initial task exposure is even more robust than at earlier time points.17,20,22,27,30,31,33 Given the importance of creating and sustaining better movement patterns during post-stroke rehabilitation, it is crucial to understand how priming may aid retention as well as initial learning.
Most of the above work in motor learning/retention and exercise priming has involved upper extremity tasks such as visuomotor rotations or reaching to targets.17,19 -24,27,33,34 By contrast, much less work has explored learning associated with the functionally crucial task of walking, and the results of these studies have been very different than those found in the upper extremity. The first work in the area of exercise priming in walking was by Helm et al 18 and Charalambous et al, 35 who found no benefit of priming to locomotor retention (split-belt treadmill walking) in young typical adults.18,35 Subsequently, in another study, Charalambous et al 25 found no benefit of exercise priming to split-belt treadmill walking retention in individuals with chronic stroke.
The contrast between the results seen by Helm et al and Charalambous et al versus previous studies in the upper extremity could be due in part to the type of learning that occurs with split-belt treadmill walking versus the previously-used reaching tasks. Learning during split-belt treadmill walking occurs through the cerebellar-mediated process of implicit sensorimotor adaptation with minimal contributions from any explicit or strategic cognitive processes.11,36,37 By contrast, the previous upper extremity work relied largely on visually-guided reaching tasks and visuomotor rotations that have been found to utilize predominantly explicit strategy.38,39 Since explicit strategic learning is generally thought to be mediated through the prefrontal cortex,17,28,38,39 and the neurotrophic factors and other chemical mediators released during exercise have been shown to reach and be taken up in the prefrontal region,17,29 it is possible that exercise priming may target the neural structures associated with explicit strategic learning more than those associated with implicit sensorimotor adaptation. As yet, no groups have studied the effect of exercise priming on the learning/retention of an explicit, strategic locomotor learning task (a task where the learning mechanisms would be expected to be more similar to the tasks in the upper extremity priming literature.)
Given this possible effect of learning type on the amenability of the learning/consolidation process to exercise priming, we chose to investigate the possible impact of exercise priming on retention when used with an explicit, strategically-oriented locomotor learning task that has been previously studied in our lab.40 -43 The purpose of this study, thus, was to determine the impact of exercise priming on retention of an explicit, strategic locomotor learning task in individuals with chronic stroke. We hypothesized that exercise priming would lead to greater 24-hour task retention than no priming.
Methods
Participants
Fifty-six community-dwelling stroke survivors enrolled in the study (Figure 1). Participants were recruited from local physical therapy clinics, support groups, advertisements, and the university’s Stroke Research Registry. All participants provided written informed consent as required by the university’s Institutional Review Board (approval number 1139080-12). Prior to the start of any study activities, this project was registered as a clinical trial (www.clinicaltrials.gov, identifier NCT03726047). Inclusion and exclusion criteria were similar to those described previously.40,41 To be included, participants had to meet the following criteria: (1) age 18 to 85 years old, (2) single unilateral chronic (>6 months) stroke, confirmed by magnetic resonance imaging or computerized tomography imaging, (3) ability to walk 10 m without assistance of another person, (4) resting heart rate 40 to 100 bpm, (5) resting blood pressure 90/60 to 185/100 mmHg. Participants were excluded if they had: (1) cerebellar stroke, (2) neurological conditions other than stroke, (3) hemi-spatial neglect, (4) visual deficits due to stroke (5) inability to communicate with or understand the instructions, (6) pain that limited walking, (7) coronary artery bypass graft or myocardial infarction within past 3 months, and (8) unexplained dizziness in last 6 months. Participants initially recruited for the study were randomized by computer algorithm into 2 groups: control (CTL, n = 18), and 5-minute continuous aerobic exercise priming (EX-C, n = 18). A third, non-randomized group was subsequently added to explore possible differing effects of long interval aerobic exercise priming (EX-LI, n = 21).
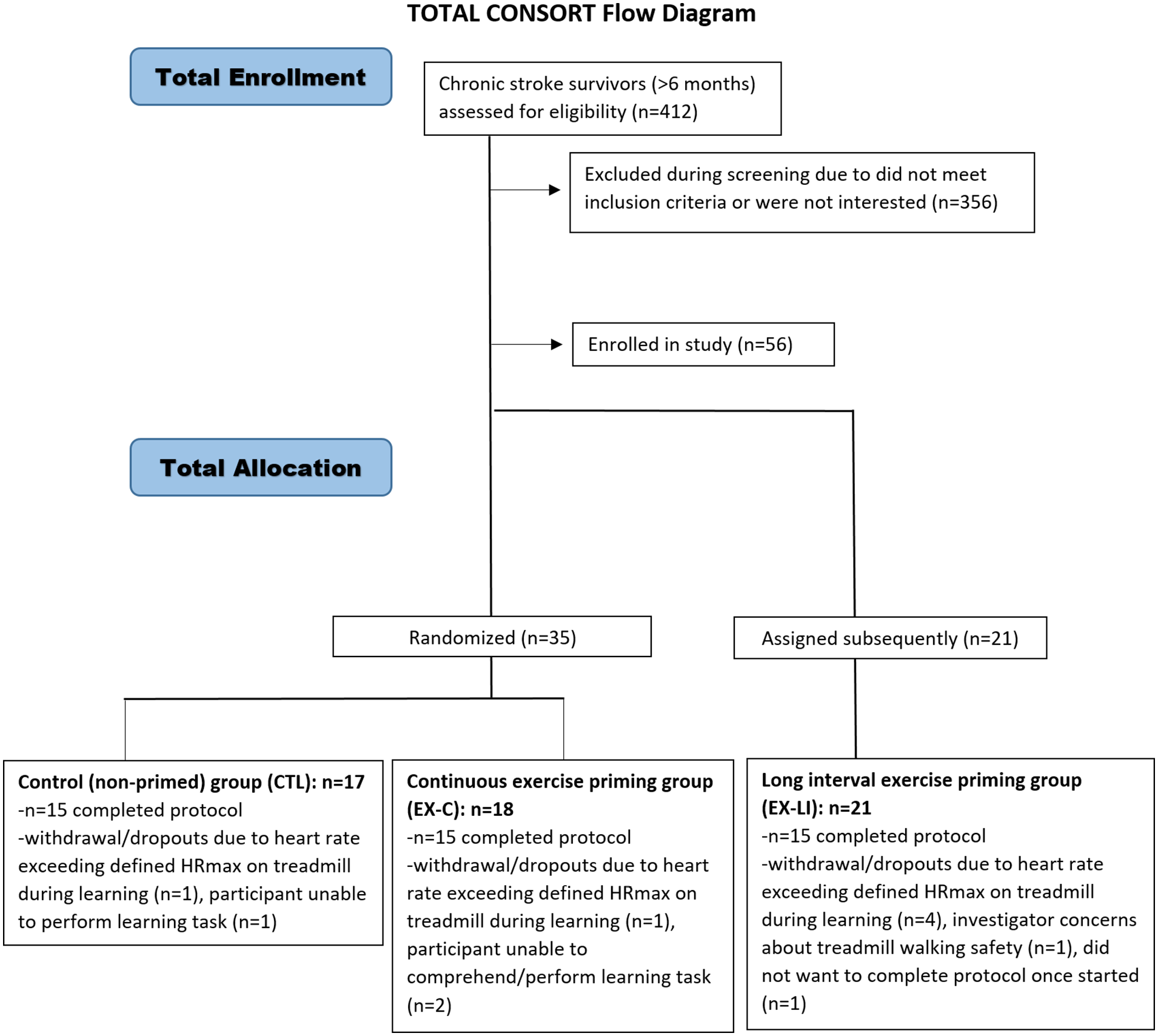
CONSORT flow diagram.
Experimental Design
Locomotor Learning Protocol
The motor learning portion of the study consisted of 2 sessions of treadmill walking with a 20″ computer monitor placed at eye level ~60 cm in front of the treadmill. The 2 sessions were scheduled 24 hours apart, and each participant walked on the treadmill at their comfortable self-selected walking speed. The motor learning paradigm did not differ across the 3 groups. Participants walked on a dual-belt treadmill instrumented with force plates with 6 degrees of freedom (Bertec Corporation). Motion capture data were collected from 7 passive markers placed on bilateral heels, lateral malleoli, fifth metatarsal heads, and left first metatarsal head. An 8-camera Vicon Motion Capture system (Vicon MX, Los Angeles, CA, USA) was used to collect the kinematic data at 100 Hz, and kinetic data were collected via the treadmill force plates at 1000 Hz.
All participants walked while lightly holding the front handrail and while wearing a ceiling-mounted harness for safety. The harness did not provide body-weight support and was intended to prevent falling if a participant lost their balance. The real-time visual display was created using the Motion Monitor Toolbox (Innovative Sports Training Inc., Chicago, IL, USA), as described in previous work.40 -42 See Figure 2 for apparatus and protocol details.
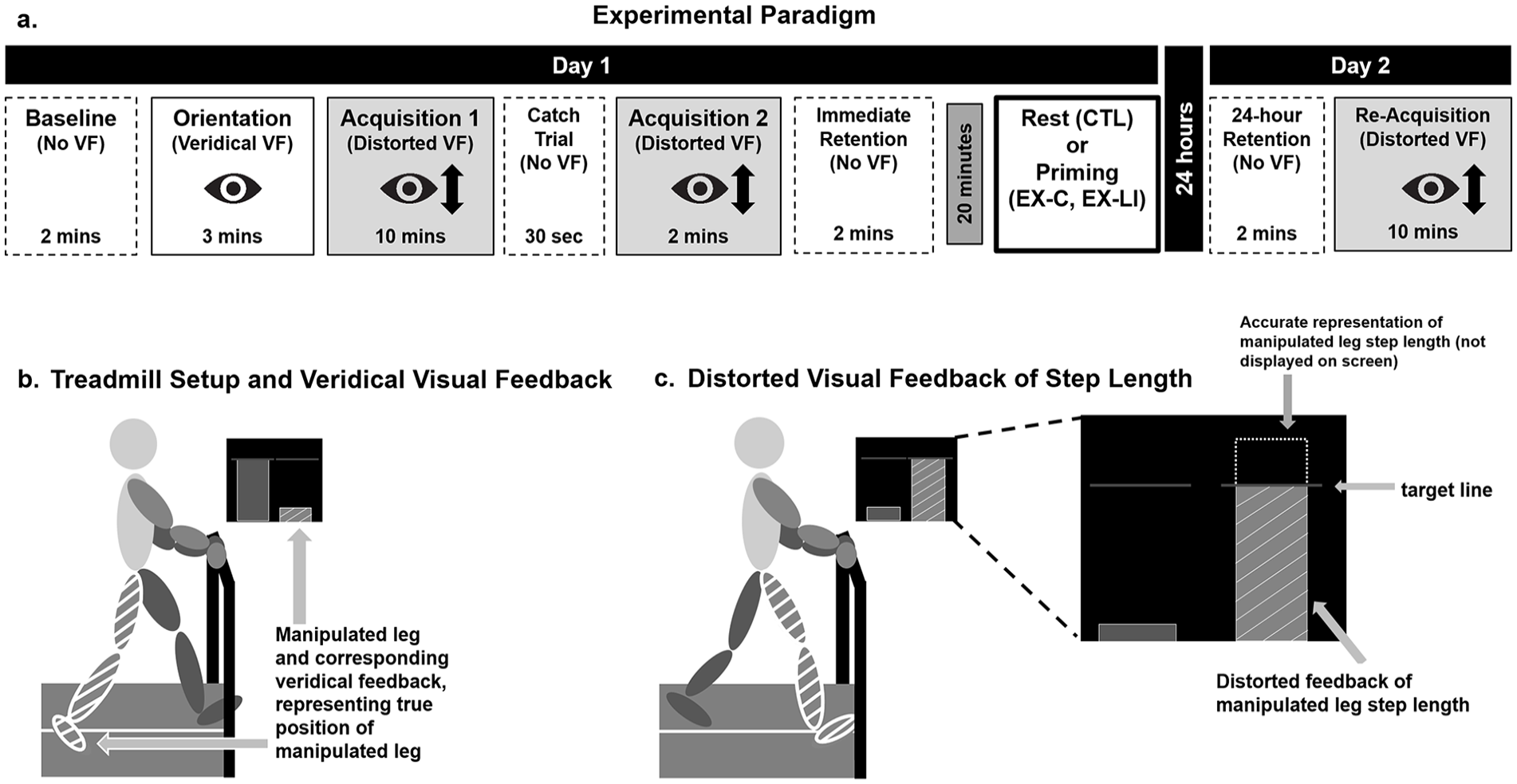
Study paradigm and apparatus.
Day 1 was divided into 6 walking phases: Baseline, Orientation, Acquisition 1, Catch Trial, Acquisition 2, and Immediate Retention. Day 2 was divided into 2 walking phases: 24-hour Retention and Re-Acquisition (Figure 2). The walking paradigm and the instructions described below were adapted from previous work. 41
During Baseline, participants were asked to walk normally without any visual display. The purpose of this phase was to determine baseline step length asymmetry and create the visual display. The leg with shorter step length at baseline was designated as the manipulated leg.
During Orientation, Acquisition 1, Acquisition 2 (day 1), and Re-Acquisition (day 2), participants received visual feedback as described previously.40 -42 The display consisted of 2 bars, a blue bar for the left leg and a green bar for the right leg. The height of each bar increased in real-time during the swing phase, and the bar height on the screen at the point of heel strike was maintained until the start of the next swing phase on the same side. The display also consisted of 2 horizontal lines representing the participant’s baseline step length. The Orientation phase introduced participants to the visual display and the concept of step length, defined as “the distance from one heel to the other when the front heel hits the ground.” All participants were told “the green bar will represent your right leg and the blue bar will represent your left leg. You will also see 2 horizontal lines across the screen. These lines represent your baseline step length and will serve as a target line later.” Throughout Orientation, participants were encouraged to walk while changing the step length of the manipulated leg, first taking the “longest possible step” with each leg, and then a shorter step. This (1) allowed participants to gain a better understanding of the interaction between the step length and the visual display and (2) verified that participants could physically take a sufficiently long step with the manipulated leg to respond to the feedback during the upcoming acquisition conditions.
During Acquisition 1 and 2, participants learned a new walking pattern using the visual feedback but with the bar height distorted as previously described. 41 During these 2 phases, real-time visual feedback for the bar of the manipulated leg was distorted (shortened by 15%). The step length bar thus provided feedback that the participant’s step was 15% shorter than the true step length (Figure 2). To reach the target line, participants learned to take a 15% longer step with the manipulated leg during these phases. Participants were asked to step such that each step length hit the target step length line on the screen as precisely as possible. To make the task as explicit as possible, participants were informed about the distortion, the goal, and the strategy they would need to counteract the distortion (increasing the step length on the manipulated leg). Between Acquisition 1 and Acquisition 2 participants performed a 30-second Catch Trial without visual feedback (not included in the present analysis.)
During Immediate Retention on day 1 and 24-hour Retention on day 2, participants re-created the learned pattern without visual feedback. Participants were asked to “walk how you were walking while you watched the display. So, whatever you were doing to get the bars to hit the line, I want you to recreate that.” Twenty-four-hour retention on day 2 was followed by Re-Acquisition, where participants re-learned the same walking pattern with the same display and instructions as on day 1.
Exercise Priming Protocol
Twenty minutes (±5 minutes) after completing the treadmill walking on day 1, the control group rested while the exercise groups performed brief bout/bouts of exercise on a whole-body ergometer. The continuous exercise group (EX-C) performed a 5-minute high intensity bout, with a target heart rate range of 70% to 85% of age-predicted maximal heart rate (HRmax = 220 – age). 44 For individuals on beta blocker medications (for whom heart rate may not be a reliable indicator of exertion during exercise 45 ) high intensity was defined as reporting a rate of perceived exertion (RPE) ≥13 on a 6 to 20 RPE scale. 46 The interval exercise group (EX-LI) started with a 1-minute warm-up (<60% HRmax) followed by three 3-minute bouts (70%-85% HRmax) that were interspersed with two 2-minute low-intensity (<70% HRmax) bouts. Participants in the EX-LI group were encouraged to be active throughout the 15-minute exercise bout, but were allowed a rest break if requested or if the heart rate exceeded 85% HRmax.
Total time spent in the target zone was determined by adding together any time spent in or above the target zone. Heart rate was monitored every 15 seconds via a chest strap monitor linked to a wrist watch display visible to study staff (Polar Electro Inc., Lake Success, NY, USA) and RPE was recorded every 30 seconds. Blood lactate levels were collected, in accordance with previous work,18,25,35 using a finger-stick automated portable Lactate Plus analyzer and test strips (Noval Medical, Waltham, MA, USA) 2 minutes before and immediately after the exercise.18,21,25,27,34,35 The blood lactate levels were also taken from a subset of individuals within the control group to demonstrate that no increase in lactate occurred with rest.
Data and Statistical Analysis
Data analysis was done using custom MATLAB code (MathWorks, Natick, MA, USA). For most participants, kinetic data were used to identify gait events, with heel strike defined as the first point where the downward vertical ground reaction force exceeded 20N. For participants where kinetic data were compromised, gait events were identified using kinematic data, as described by Zeni et al. 47 Step length was defined and calculated by finding the sagittal-plane distance between the heel markers on the 2 feet at heel strike. Statistical analysis was carried out in SPSS v26 (IBM, Chicago, IL), with an alpha of .05.
Variables of Interest: Locomotor Learning and Retention
To verify that all groups were able to learn to change their stepping behavior, a mixed repeated-measures analysis of variance (ANOVA) was performed comparing percent error in the first 10 steps of Acquisition 1 (Early Acquisition) and the last 30 steps of Acquisition 2 (Late Acquisition) for all groups. Additionally, though no day 1 between-groups differences in learning were expected, a 1-way ANOVA was performed to compare percent error in Late Acquisition across all groups.
Two different types of retention of the learned gait pattern were measured: cued (with visual feedback) and un-cued (without visual feedback). A greater decrease in either retention measure between day 1 and day 2 indicated greater “forgetting” (poorer retention) of the newly-learned pattern, whereas a smaller decrease indicated less forgetting (better retention).
For cued retention, forgetting was the absolute value of the difference in percent error between the last 30 steps of Acquisition 2 (day 1) and the first ten steps of Re-Acquisition (day 2). Percent error (PE) is an accuracy measure that has been used in prior literature40,41 and is defined as the difference between target step length and actual step length of the manipulated leg on each step.
Cued retention was calculated as follows:
(Abs = absolute value)
For un-cued retention, forgetting was defined as the absolute value of the difference (cm) between the step length of the last 30 steps of Immediate Retention (day 1) and the first ten steps of 24-hour Retention (day 2). In order to fairly compare the results of those who learned more versus less during the day 1 acquisition conditions, both retention measures were normalized to each participant’s learning by dividing by the amount learned during acquisition (average of the last 30 steps of Acquisition 2).
Un-cued retention was calculated as follows:
(Abs = absolute value; SL = step length; IR = Immediate Retention)
Two 1-way ANOVAs were performed to compare differences across all 3 groups in cued and un-cued retention.
Variables of Interest: Exercise Priming
We used 3 exercise priming metrics in this study that are consistent with recent work.25 -27,35 First, time spent at or above the “target zone” of 70% to 85% HRmax or 13 RPE (for individuals on beta blockers) was quantified. Second, average intensity was calculated as follows:
or
Lastly, changes in blood lactate levels were calculated from pre- to post-exercise. For all priming variables, between-group comparisons were made using the Kruskal Wallis test, due to heterogeneity of variance across the groups. Since it was found that the CTL group did not spend any time in the heart rate or perceived exertion target zones during their seated rest at the end of day 1 (as expected), the value for time in zone and average intensity was zero for this group.
Results
Fifty-six individuals >6 months post-stroke were enrolled in the study. Eleven participants were withdrawn due to: inability to safely complete/understand the learning task, the heart rate while walking on the treadmill exceeded predefined limits, or the participant decided to withdraw (Figure 1). Of the remaining 45 participants, there were 15 in each group: control with no priming (CTL), continuous exercise priming (EX-C), and long interval exercise priming (EX-LI). No significant between-group differences were found in age, sex, time since stroke, use of beta blockers, or self-selected treadmill speed (Table 1). There was a significant difference between the groups’ lower extremity Fugl-Meyer scores, such that the EX-LI group had higher Fugl-Meyer scores (milder neuromuscular impairments) than the other 2 groups.
Participant Characteristics.
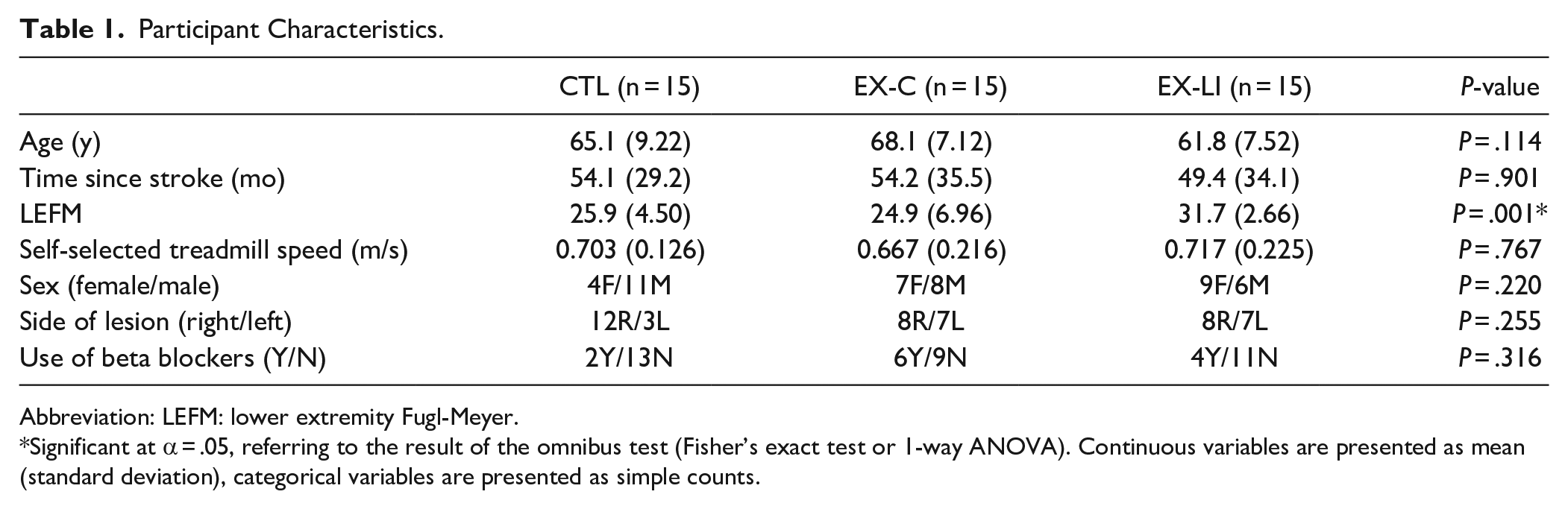
Abbreviation: LEFM: lower extremity Fugl-Meyer.
Significant at α = .05, referring to the result of the omnibus test (Fisher’s exact test or 1-way ANOVA). Continuous variables are presented as mean (standard deviation), categorical variables are presented as simple counts.
Locomotor Learning
Success in learning the novel locomotor task on day 1 was compared for each group, by examining percent error in Early Acquisition versus Late Acquisition (Figure 3). Each group demonstrated a significant difference between Early and Late Acquisition (main effect of time F(1,42) = 34.4, P < .001, partial η2 = .45), indicating that all groups learned to change their walking patterns in response to the visual feedback. However, there was no main effect of group (F(2,42) = 0.802, P = .455), and no group by time interaction (F(2,42) = 0.664, P = .520), indicating the groups learned a similar amount between the start of Acquisition 1 and the end of Acquisition 2.
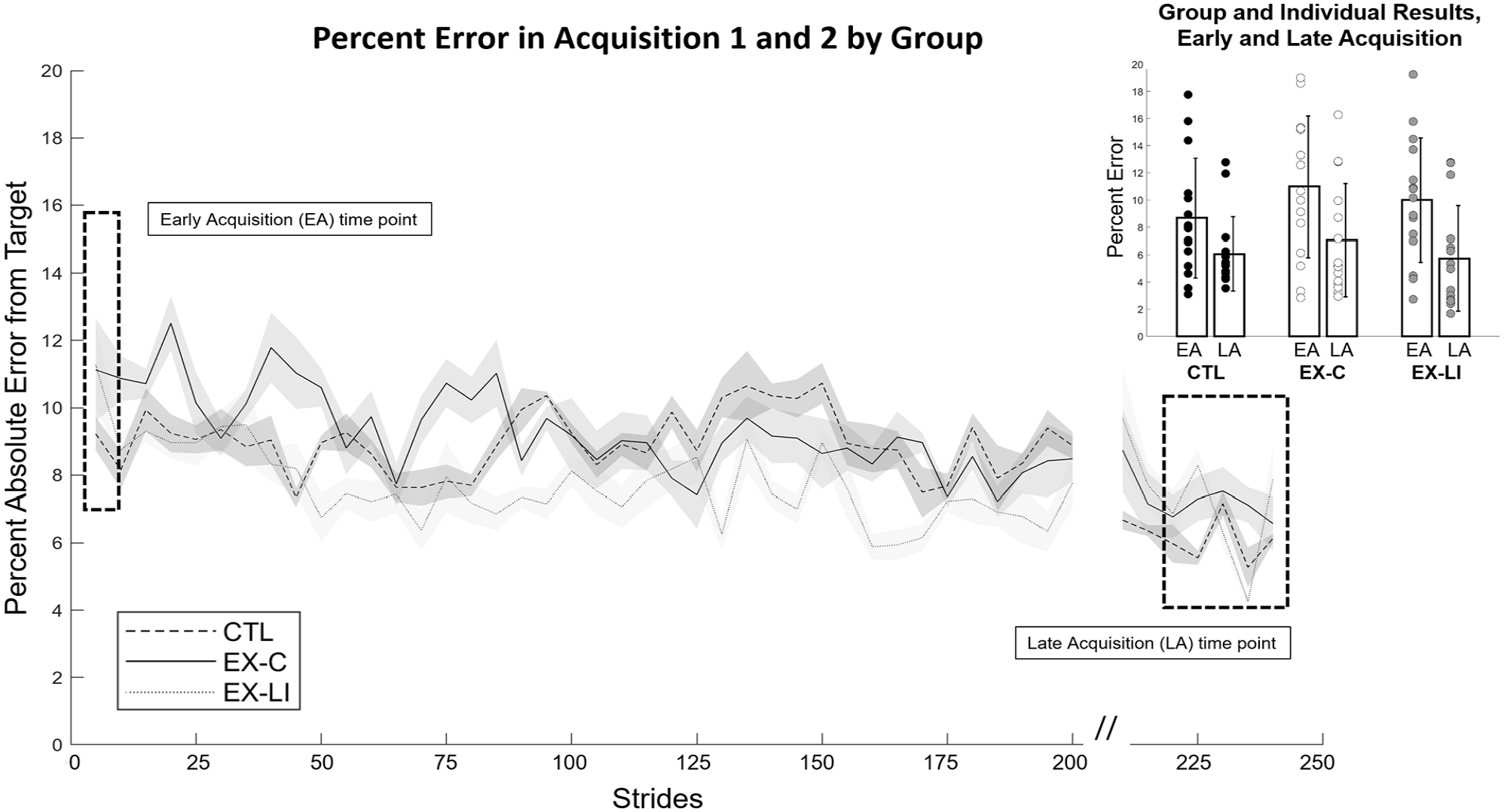
Day 1 motor task learning by group, with time series and inset of key time points (Early Acquisition and Late Acquisition). Hash marks between Acquisition 1 and Acquisition 2 indicate that the intervening Catch Trial is not shown.
After confirming that all groups were able to change their walking in response to visual feedback, a 1-way ANOVA verified that there was no between-group difference in percent error at the end of Acquisition 2 (F(2,42) = 0.547, P = .583); all groups ended Acquisition 2 at a similar level of accuracy (Figure 3). The time series of percent error in the manipulated leg during the acquisition phases is presented in Figure 3, with key time points for this analysis indicated on the figure and an inset showing group means and individual results.
Locomotor Retention
Once it was determined that all 3 groups were able to change their step length from baseline and had similar acquisition of the new walking pattern, both cued (visual feedback) and un-cued (no feedback) retention were compared across the 3 groups. For un-cued retention, the 1-way ANOVA comparing percent change in step length in Immediate Retention versus 24-hour Retention was not significant (F(2,42) = 1.287, P = .287, Figure 4A). This result indicates that un-cued 24-hour retention was not different between groups; there was no benefit of exercise priming.
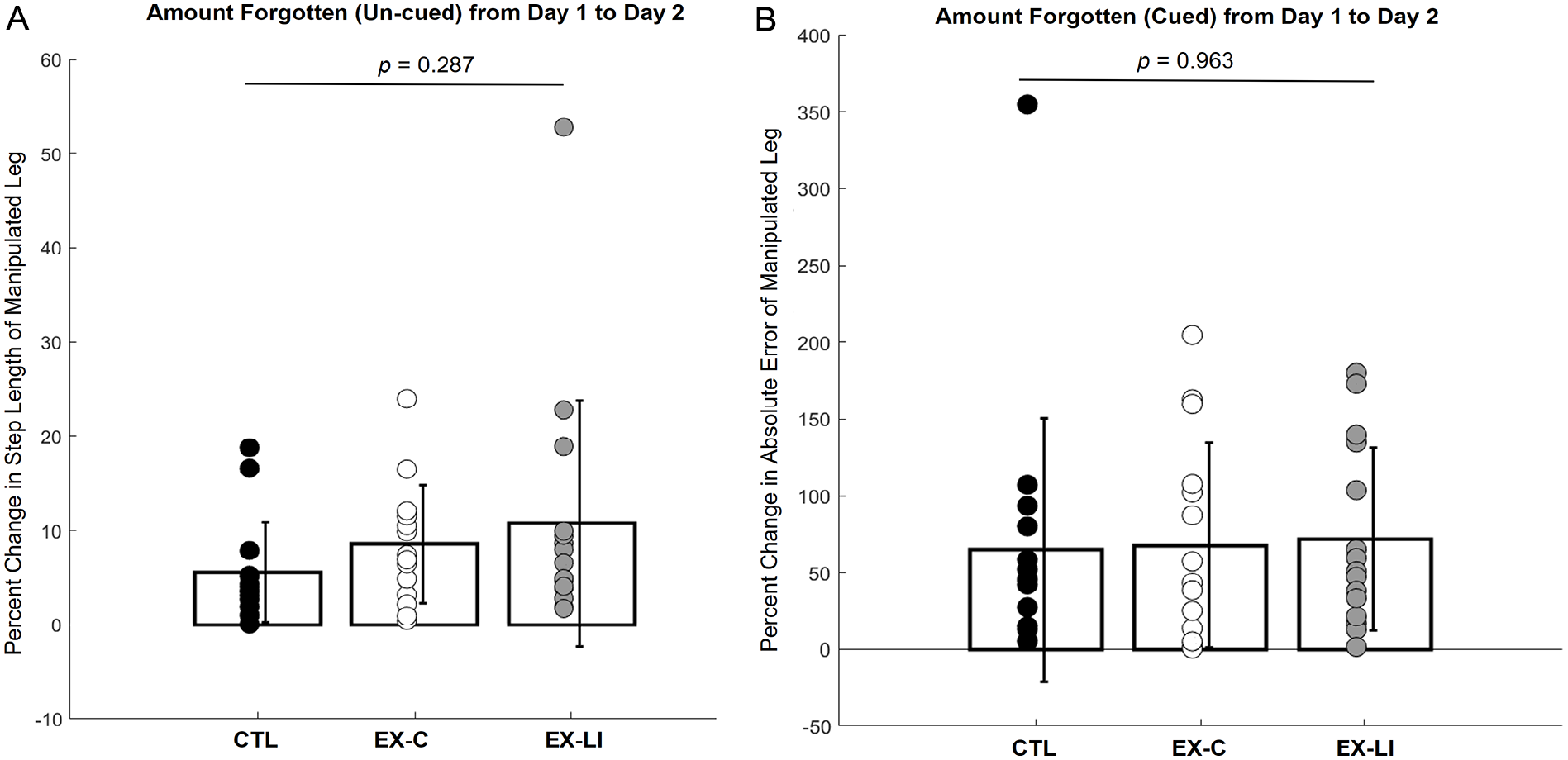
Task retention from day 1 to day 2. (A) Un-cued retention, showing difference in step length (step length forgetting) between day 1 and day 2. (B) Cued retention, showing the difference in percent error (percent error forgetting) between day 1 and day 2. Significance value for each variable’s 1-way ANOVA is shown at the top of each plot, showing no between-group difference for either measure of retention.
Next, cued retention was assessed using a 1-way ANOVA to compare percent error in Acquisition 2 versus Re-Acquisition, with no significant between groups differences found (F(2,42) = 0.037, P = .963, Figure 4B). This result indicates that cued 24-hour retention was not different between groups; there was no benefit of exercise priming. Taken together, the lack of between-group differences in either cued or un-cued retention between day 1 and day 2 indicates that there was no effect of exercise priming on locomotor task retention.
Exercise Priming
The expected physiological effects of priming were verified by comparing change in lactate (pre- to post-exercise), time spent in target heart rate or RPE zone (at or above 70% HRmax or RPE of 13), and average exercise intensity (Table 2). Significant between-group differences were found for all 3 exercise priming variables, including change in lactate levels (P = .001, η2 = .37), time spent at or above target zone (P < .001, η2 = .87), and average exercise intensity (P < .001, η2 = .68). Post-hoc Mann-Whitney U tests with Bonferroni corrections further revealed significant pairwise differences between the CTL and both exercise priming groups (EX-C and EX-LI) for all 3 variables (Table 2): lactate (CTL vs EX-C P < .001, η2 = .60; CTL vs EX-LI P < .001, η2 = .59), time in target zone (CTL vs EX-C P < .001, η2 = .83; CTL vs EX-LI P < .001, η2 = .83), average intensity (CTL vs EX-C P < .001, η2 = .83; CTL vs EX-LI P < .001, η2 = .83). EX-C and EX-LI differed from each other only in the time spent in the target zone (P < .001, η2 = .52), while change in lactate and average intensity did not differ (P = .62 and .19, respectively). The significant difference in time spent in target zone was an expected result given the longer priming duration for the EX-LI group.
Exercise Priming Variables.
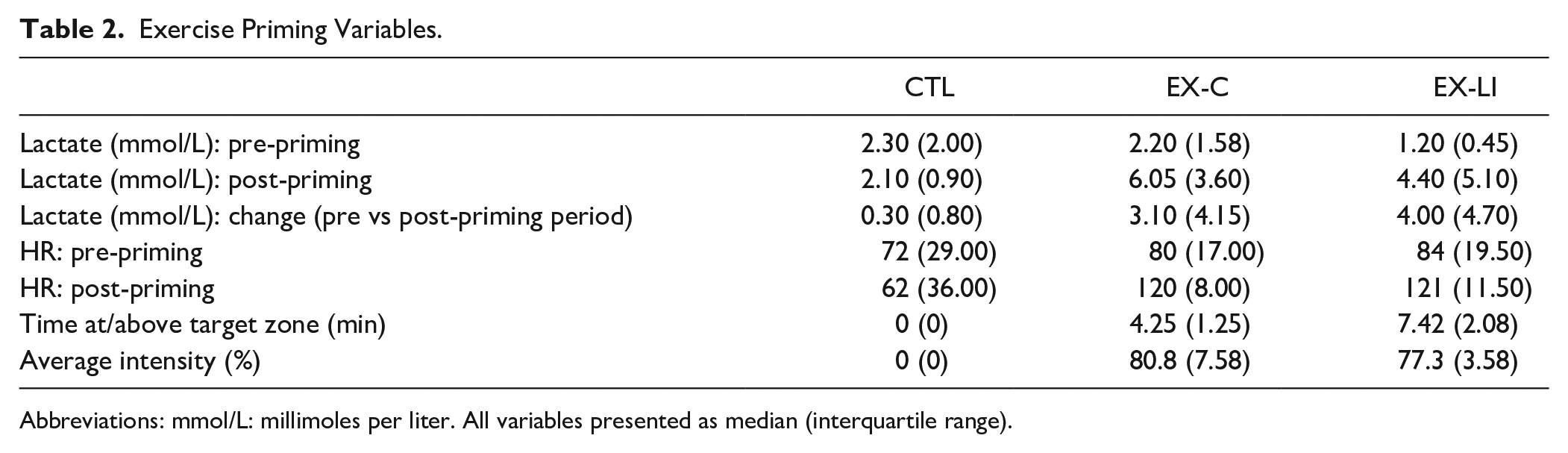
Abbreviations: mmol/L: millimoles per liter. All variables presented as median (interquartile range).
Discussion
The purpose of this study was to determine the impact of exercise priming, pairing a bout of intense exercise with motor learning, on retention of an explicit, strategic locomotor learning task in individuals with chronic stroke. We hypothesized that exercise priming would improve 24-hour retention of the task. Surprisingly, our findings did not support this hypothesis. Instead, the results presented here, including a no-priming group, a 5-minute priming group, and a 15-minute priming group, indicate that exercise priming provided after initial exposure to the explicit locomotor task (within the consolidation period of learning) does not improve 24-hour retention.
These results are in agreement with the previous work coupling exercise priming with a sensorimotor adaptation locomotor learning task, both in individuals with chronic stroke 25 and in young neurotypical individuals.18,35 However, these results stand in contrast to earlier studies which found that priming improved retention of upper extremity reaching tasks, both in young neurotypical participants19,21,22,27,31,34 and in those with chronic stroke.23,24 Previous authors have posited that the effects of exercise priming cannot be assumed to be generalizable to all motor tasks. As Snow et al 48 warned, “. . .exercise effects on motor behavior are not universal, and may be task-dependent, and/or reliant on the outcome measure used to assess motor performance.” In the case of the present results, there are multiple potential factors that could help explain our findings relative to prior work.
Type of Motor Learning Task
The nature of the present task (volitionally changing step length during walking in response to visual feedback) was very different from previous instances in which exercise priming was found to aid retention of a motor task. Previous upper extremity protocols were almost always performed with 1 limb (including image-tracing, isometric pinching, or reaching for targets), and performed in a seated position.19,21,23,27,28,30,31,34 By contrast, the present locomotor learning protocol involved the continuous full-body task of walking, with bilateral coordination demands as well as much greater posture and balance demands than seated tasks. It is possible that these elements of walking affected the impact of exercise priming on retention. In fact, these results are consistent with previous work involving whole-body motor learning tasks, such as split-belt treadmill walking, standing balance, or sport-simulation tasks. In these tasks with greater postural and coordination demands than the upper extremity learning tasks, exercise priming has not been shown to improve motor learning or retention in neurotypical individuals or those with chronic stroke.18,25,29,32,35 It has been suggested based on EEG results that seated, single-limb visuomotor tasks such as those in previous exercise priming work in the upper extremity may predominantly involve the prefrontal cortex. 17 By contrast, gait and postural tasks have been found to be related to a wide range of brain areas, including the primary motor cortex, visual cortex, cerebellum, basal ganglia, and brainstem.49 -51 While the addition of the explicit, strategic visual feedback task would be expected to increase the prefrontal contribution to the task, 50 the difference in the neuroanatomical areas associated with walking versus seated reaching could have affected the impact of exercise priming on task retention.
Exercise Priming Regimen
Another possible explanation for our current results could be the details of the exercise priming regimen. In this study, 2 groups performed full-body exercise priming. We utilized both a 5-minute priming group (EX-C) similar to prior locomotor studies18,25,35 and a 15-minute priming group (EX-LI) similar to previous upper extremity literature.22,23,27,30,32,34 The study design initially included the no-exercise CTL group and the EX-C group, with the EX-LI group added after initial analysis 52 to address the possibility that the priming parameters were responsible for the negative results. Changes in heart rate, RPE, and blood lactate after the priming regimen provide physiological evidence that the EX-C and EX-LI groups successfully completed exercise at a level associated in the upper extremity literature with greater motor task retention, and that the resting CTL group experienced no such changes.
While the 5-minute continuous protocol used by the EX-C group is newer, it was developed specifically for use in populations with lower aerobic endurance (such as those with chronic stroke), and has consistently demonstrated physiological responses consistent with those seen from longer protocols.18,25,26,35,53 Past responses to 5-minute priming in healthy young adults including significantly increased serum BDNF 18 and increases in serum lactate in excess of 10 mmol/L. 35 This magnitude of increase in lactate is notable, because 10 mmol/L is a commonly-used benchmark of high-intensity exercise in previous priming studies in young healthy adults.19,21,27 Additionally, 5-minute priming in individuals with chronic stroke has been shown to result in increased motor cortical excitability, 53 a biomarker associated with high intensity related to neuroplasticity.28,34,54,55 These results all indicate that the 5-minute priming protocol provides comparable physiologic stimulus to the 15-minute protocol used in the present study as well as previous upper extremity work. Nonetheless, to rule out the possibility that the length of the exercise bout was a factor in our results, we added a group that completed the same 15 minute interval exercise that has been used in previous studies of upper extremity motor learning that have found a positive impact of exercise on motor learning.19,22,23,27,30,34
While lactate change in our priming groups did not reach 10 mmol/L, no prior priming studies in individuals with chronic stroke have achieved this increase – studies either did not measure lactate in these individuals6,23,24 or found smaller yet statistically significant lactate increases that are very similar in magnitude to the changes found in the present study.25,26 These previous studies, and others, demonstrate that exercise regimens that achieve >70% of age-predicted maximal heart rate in people with chronic stroke are unable to elicit lactate changes >10 mmol/L.25,26,43,56,57 Nevertheless, the changes in lactate found with these high-intensity protocols in people post-stroke were associated with electroencephalographic changes and changes in circulating neurotrophic factors, indicating neuroplastic change.25,26,43,56,57
Limitations
There are several important limitations to the current study. While our results clearly show no between-group differences that would indicate that exercise priming enhanced locomotor learning or retention, there was considerable individual variability, and we did not have a sufficient sample size to explore which characteristics may make an individual more likely to benefit from exercise priming associated with motor learning. Perhaps exercise priming in an explicit strategic locomotor task could be beneficial for stroke survivors who have particular difficulty with this type of learning. Our previous work indicates that fluid cognition is related to difficulties with retention of an explicit strategically-learned walking pattern in people with chronic stroke. 41 Future studies should therefore test the effect of exercise priming on retention of an explicit, strategically-learned walking pattern in stroke survivors with poor fluid cognition. Second, the study design of this clinical trial was 2-fold: a randomized controlled trial, followed by the addition of another experimental group with a longer priming duration. The initial randomized controlled trial, which randomly assigned participants to control (no priming) or to a 5-minute exercise priming bout, did not find a benefit of priming to 24-hour task retention. To rule out any possibility that the exercise duration was too short for sufficient priming, we expanded the experiment by testing a third group. This third group completed the same locomotor learning protocol, but with a 15-minute high intensity interval exercise regimen, as seen in some of the previous upper extremity studies.19,22,23,27,30,32,34 While the addition of the third group was out of an abundance of caution to ensure that the negative results of the initial trial were not due to insufficient priming, the non-randomized nature of this additional group does make for a more complicated experimental design. Additionally, the nature of our motor task required that individuals be able to walk for multiple bouts of 5 to 10 minutes without another person’s assistance. Thus, we are unable to generalize our results to stroke survivors with more severe mobility deficits. Finally, our participants were all at least 6 months post-stroke. While this is a substantial and growing population, our focus on chronic stroke may make it difficult to generalize our results to those in the first 6 months of post-stroke recovery.
Conclusions/Implications
This study explored the impact of a single bout of high-intensity exercise on retention of an explicit, strategically-learned walking pattern in people with chronic stroke. While we did not detect a benefit of exercise priming to task retention, further work should continue to investigate additional functionally-relevant motor tasks, as well as specific characteristics of individuals who may be more or less likely to benefit from exercise priming to enhance motor learning after stroke. Given the importance of motor learning to post-stroke rehabilitation, it is critical that we understand factors that can enhance learning.
Footnotes
Acknowledgements
The authors wish to acknowledge the time and contributions of our study participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health [grant numbers NIH 2R01HD078330-05A1 and 1F31NS111806-01A1].