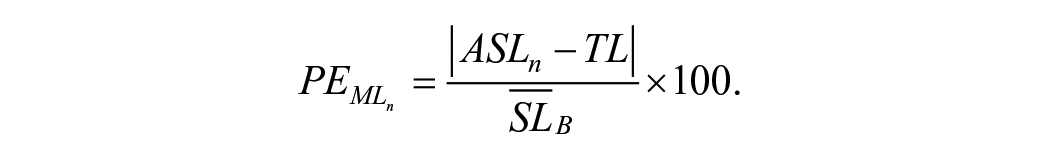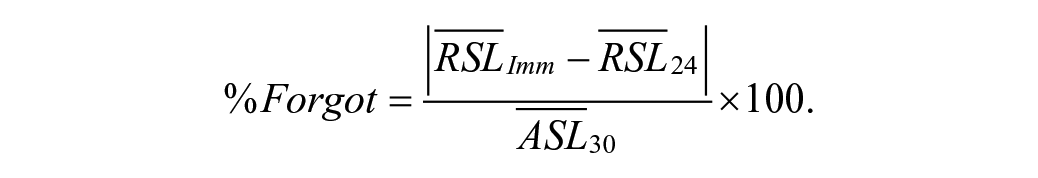Abstract
Background
There is significant variability in poststroke locomotor learning that is poorly understood and affects individual responses to rehabilitation interventions. Cognitive abilities relate to upper extremity motor learning in neurologically intact adults, but have not been studied in poststroke locomotor learning.
Objective
To understand the relationship between locomotor learning and retention and cognition after stroke.
Methods
Participants with chronic (>6 months) stroke participated in 3 testing sessions. During the first session, participants walked on a treadmill and learned a new walking pattern through visual feedback about their step length. During the second session, participants walked on a treadmill and 24-hour retention was assessed. Physical and cognitive tests, including the Fugl-Meyer-Lower Extremity (FM-LE), Fluid Cognition Composite Score (FCCS) from the NIH Toolbox -Cognition Battery, and Spatial Addition from the Wechsler Memory Scale-IV, were completed in the third session. Two sequential regression models were completed: one with learning and one with retention as the dependent variables. Age, physical impairment (ie, FM-LE), and cognitive measures (ie, FCCS and Spatial Addition) were the independent variables.
Results
Forty-nine and 34 participants were included in the learning and retention models, respectively. After accounting for age and FM-LE, cognitive measures explained a significant portion of variability in learning (R2 = 0.17, P = .008; overall model R2 = 0.31, P = .002) and retention (ΔR2 = 0.17, P = .023; overall model R2 = 0.44, P = .002).
Conclusions
Cognitive abilities appear to be an important factor for understanding locomotor learning and retention after stroke. This has significant implications for incorporating locomotor learning principles into the development of personalized rehabilitation interventions after stroke.
Introduction
Improving one’s walking ability after stroke is a common goal of patients and rehabilitation professionals. 1 As a result, significant time and money are spent on improving walking during stroke rehabilitation. Despite this, only 50% of individuals regain the ability to walk independently after their stroke. 2 This is in part due to highly variable responses to interventions across individuals.3,4 Since motor learning is the foundation of most rehabilitation interventions, differences in how well individuals learn likely contributes to these varied responses. Thus, understanding sources of individual variability during locomotor learning can improve the field’s understanding of varied responses to rehabilitation interventions and ultimately lead to the delivery of personalized rehabilitation interventions to improve walking after stroke. However, individual differences during locomotor learning have been largely ignored. The few studies that have sought to understand variability in locomotor learning after stroke have focused on a limited number of factors, specifically demographic information and sensorimotor impairment.5,6 These factors are unable to explain a significant portion of the observed variability in locomotor learning. Thus, it remains unclear what factors predict how well an individual will be able to learn and retain a new walking pattern after stroke.
Although cognition has been found to relate to motor outcomes after stroke,7 -11 it has been unexplored related to locomotor learning after stroke. In neurologically intact adults, however, lower cognitive abilities have been found to relate to poorer motor learning and retention of upper extremity tasks.12 -20 Specifically, visuospatial working memory, or one’s ability to temporarily store visual and/or spatial information for manipulation, 21 has drawn significant attention in past work. In upper extremity sequence learning13,15,16 and visuomotor rotation tasks,12,14,17,18 neurologically intact adults with lower visuospatial working memory scores have been found to learn and retain new motor patterns worse than individuals with higher visuospatial working memory scores. However, visuospatial working memory is highly related to other domains of cognition, specifically attention and executive functions.13,21,22 Additionally, other work has found that nonverbal reasoning, 13 processing speed, 23 decision making, 23 delayed memory, 20 and visuospatial construction ability19,24 are related to upper extremity motor learning. Thus, it is unclear if the relationship between motor learning and cognition is specific to visuospatial working memory. All of the domains of cognition that have been found to relate to motor learning can broadly be classified as fluid cognitive abilities, which is a global cognitive measure reflecting one’s abilities to problem solving and learn new things in the absence of prior experience. 25 Thus, it is possible that global fluid cognition rather than visuospatial working memory specifically is related to motor learning.
The work described above has all been conducted in upper extremity tasks. Therefore, despite the high prevalence of cognitive impairments after stroke, 7 it is unclear if cognition is related to locomotor learning similarly. Because of the unique demands of locomotion (ie, high levels of postural control and bilateral coordination), it is possible that cognition does not have the same relationship with locomotor learning as it does with motor learning involving upper extremity tasks. However, there is evidence that underlying learning processes that occur in the upper extremity and during locomotion are likely the same. For example, sensorimotor adaptation occurs during upper extremity tasks26 -29 and locomotion.30,31 Similarly, explicit, strategic learning processes have been observed in both upper extremity tasks28,32 -34 and locomotion.35,36 Thus, despite the unique demands of walking, the relationship between cognition and locomotor learning may exist due to similar underlying learning processes. Importantly, cognition is believed to relate to explicit motor learning processes in the upper extremity. This is highlighted by the work of Christou et al, 17 which found that a visuospatial working memory task was specifically related to the explicit, strategic component of visuomotor adaption. The involvement of the dorsolateral prefrontal cortex in both visuospatial working memory and explicit, strategic learning is the hypothesized mechanism of this relationship.12,14,37 Although much less is known about the regions of the brain involved in explicit locomotor learning, there is evidence for activity in the prefrontal cortex when learning a new walking pattern through explicit cues38,39 and during complex walking tasks, such as obstacle navigation. 40 Therefore, it is possible that cognition, specifically visuospatial working memory, has the same relationship with explicit locomotor learning due to overlapping activation in the prefrontal cortex; however, this is currently unknown.
Thus, the purpose of this work was to understand the relationship between cognition (ie, visuospatial working memory and fluid cognitive abilities) and explicit locomotor learning and retention after stroke. To do this, we used a visually guided walking paradigm that primarily results in explicit, strategic learning35,41 to assess locomotor learning abilities after stroke. Additionally, we used a standardized measure of visuospatial working memory and of fluid cognitive abilities to measure cognition. We hypothesized that cognition, specifically visuospatial working memory and fluid cognition, would explain a significant portion of variability in learning and retention of a new, strategically learned walking pattern after accounting for age and physical impairment.
Methods
Participants
Community-dwelling stroke survivors were recruited from local physical therapy and physician practices, support groups, and through advertisements. To be included, participants had to meet the following criteria: (1) 18 to 85 years old, (2) magnetic resonance imaging (MRI) evidence of a single unilateral chronic (>6 months) stroke, (3) able to walk 10 meters without physical assistance, (4) resting heart rate within 40 to 100 beats per minute, and (5) resting blood pressure within 90/60 and 185/100 mm Hg. Participants were excluded if they met the following criteria: (1) MRI evidence of a cerebellar stroke, (2) neurologic conditions other than stroke, (3) changes in vision following their stroke via self-report, (4) neglect via clinical observation, or (5) inability to communicate with investigators via questions 1b and c of the NIH Stroke Scale, (6) history of coronary artery bypass graft or myocardial infarction in the past 3 months, (7) unexplained dizziness in the last 6 months, or (8) pain that limited walking. Participants signed an informed consent approved by the Human Subjects Review Board at the University of Delaware.
Experimental Protocol
Participants completed a 3-day protocol that included 2 days of treadmill walking and 1 day of physical and cognitive testing. The 2 sessions of treadmill walking were performed 24 hours (±2 hours) apart, while the third session consisting of clinical and cognitive tests was performed 1 to 7 days after the second treadmill walking session (Figure 1A). Cognitive tests were administered on a separate day to minimize the potential impact of activity before the cognitive assessments. The range of time between Day 2 and Day 3 (1-7 days) was to ensure that participants were able to complete the 3-day protocol. On average, subjects completed the cognitive and physical tests 2.4 ± 2.1 days after Day 2.
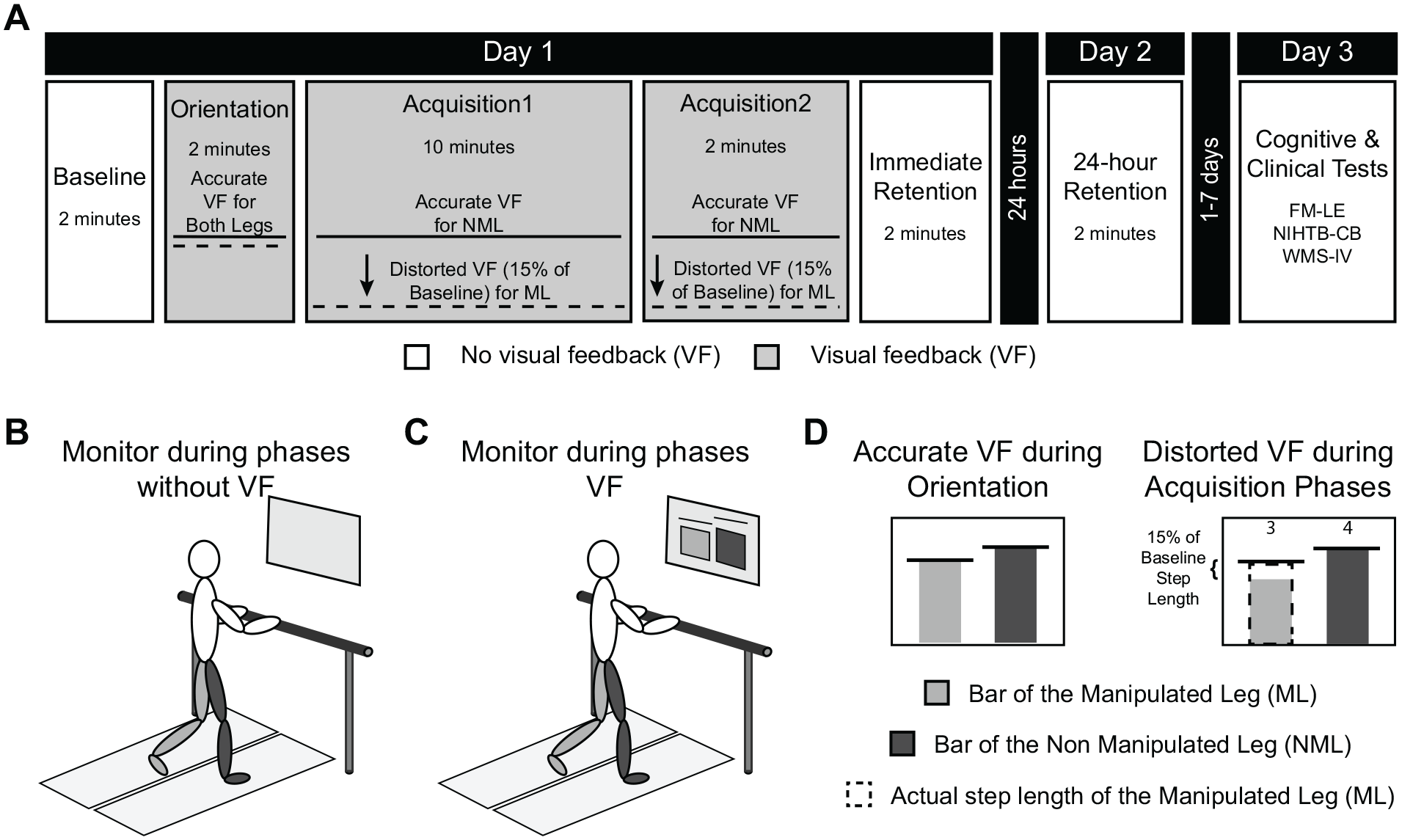
Experimental protocol and setup. Participants after stroke completed a 3-day protocol that included 2 days of treadmill walking and 1 day of clinical and cognitive testing (A). During Baseline, Immediate Retention, and 24-hour Retention, no visual feedback (VF) was provided (B), while during Orientation and Acquisition 1 and Acquisition 2 a bar graph was displayed on the computer monitor in front of the treadmill (C). During Orientation the feedback was accurate, but during Acquisition 1 and Acquisition 2, the visual feedback of one of the bars was distorted to make it look as if though that leg was taking a shorter step than they actually were (D). This resulted in participants learning to a new walking pattern where they took a longer than usual step with one leg. During Immediate Retention and 24-hour Retention, participants were instructed to walk with the pattern they had learned during the acquisition phases. The final day included the lower extremity portion of the Fugl-Meyer (FM-LE), the NIH Toolbox Cognitive Battery (NIHTB-CB), and the Wechsler Memory Scale-IV (WMS-IV).
Locomotor Learning Paradigm
During the first 2 days of the study, participants walked on a dual-belt treadmill instrumented with dual 6 degrees of freedom force plates (Bertec Corporation) while collecting motion capture data from 6 markers located on bilateral heels, lateral malleoli, and fifth metatarsal heads. kinematic data were collected at 100 Hz using an 8-camera Vicon Motion Capture system (Vicon MX), and kinetic data were collected at 1000 Hz. While walking on the treadmill, participants walked at a comfortable, self-selected walking speed with a safety harness and a light grip on the handrail in front of the treadmill. Throughout the treadmill walking, a 20-inch computer monitor was positioned in front of the treadmill.
On Day 1, participants completed 5 phases: Baseline, Orientation, Acquisition 1, Acquisition 2, and Immediate Retention. Day 2 consisted of 24-hour Retention (Figure 1A). During Baseline, Immediate Retention, and 24-hour Retention, no visual feedback was displayed on the monitor (Figure 1B). During Orientation, Acquisition 1, and Acquisition 2, real-time visual feedback was displayed on the monitor using the Motion Monitor Toolbox (Innovative Sports Training Inc; Figure 1C).
During Baseline, subjects were instructed to walk normally. During this time, average step length was calculated for each leg. The leg that naturally took the shorter step was identified and will be referred to as the Manipulated Leg (ML). The other leg will be referred to as the Non-Manipulated Leg (NML). The calculated step lengths were used to create the visual feedback display that participants watched during Orientation, Acquisition 1, and Acquisition 2.
During Orientation, Acquisition 1, and Acquisition 2, a bar graph was displayed on the monitor. This graph displayed real-time information about the participants step length and has been described previously.35,41 Briefly, there were 2 bars and 2 horizontal lines representing the participants step length during the Baseline phase on the display. Prior to Orientation, participants were verbally informed about the display. The Orientation phase was 2 minutes long and consisted of 3 separate walking bouts with accurate visual feedback (Figure 1D). First, participants were instructed to walk normally and watch the display. Next, they were instructed to take the longest possible step. Finally, participants were again asked to walk normally and watch the display for 1 minute. This phase ensured that participants understood the display and were physically able to increase their step length enough to complete the learning task.
During Acquisition 1 and Acquisition 2, the same bar graph was displayed; however, there were also numbers positioned above each bar that increased when the bar hit the target line. During these phases, the visual feedback was distorted so that the bar that represented the ML looked as if they were taking a shorter step than they actually were (Figure 1D). Specifically, the bar of the ML was shortened by 15% of the baseline step length of the ML, with the goal of increasing the step length by 15%. When subjects increased their step length enough to get within ±2% the horizontal target line, the numbers above the bar increased. Participants were instructed to get these numbers as high as possible by getting the bars to hit the target line. Additionally, participants were told that the bars would be inaccurate. This was done to maximize the amount of explicit learning processes occurring since past work suggests that detailed instructions during this paradigm facilitates explicit learning. 35
Last, during Immediate Retention on Day 1 and 24-hour Retention on Day 2, no visual feedback was provided and subjects were instructed to walk how you were walking while you watched the display. These instructions were provided during the retention phases, because tasks learned via explicit, strategic learning are recalled when prompted by a verbal or visual cue.32,35
Physical and Cognitive Testing
On Day 3, participants completed a series of physical and cognitive tests. The assessor was not aware of the results from the locomotor learning task prior to administering these tests. First, the lower extremity portion of the Fugl-Meyer (FM-LE) was performed. 42 This reflects physical impairment after stroke, where high scores indicate less physical impairment. This measure has been found to be reliable and valid in individuals poststroke. 43 Next, participants completed 5 subtests from the Cognitive Battery of the NIH Toolbox for Assessment of Neurological and Behavioral Function (NIHTB-CB) in the NIH Toolbox iPad App (version 1.19.2160).44,45 The 5 subtests that were administered were (1) Flanker Inhibitory Control and Attention Test, which measures attention and executive functions; (2) Dimensional Change Card Sort Test, which measures executive functions; (3) List Sorting Working Memory Test, which measures working memory; (4) Pattern Comparison Processing Speed Test, which measures processing speed; and (5) Picture Sequence Memory Test, which measures episodic memory.46 -49 These subtests combine to create a Fluid Cognition Composite Score (FCCS). 48 This composite score is reported as a Standard Score, where 100 is average and the standard deviation is 15. The NIHTB-CB and FCCS have good evidence for reliability and validity in individuals with stroke.47,50,51 Last, the Spatial Addition subtest from the Wechsler Memory Scale-IV (WMS-IV) was administered. The WMS-IV is commonly used to assess memory 52 and has strong evidence for validity in both neurologically intact adults53,54 and those with acquired cognitive disorders. 55 The Spatial Addition subtest was specifically selected as it is 1 of the 2 measures of the WMS-IV that measures visuospatial working memory.53,56 For both of these cognitive measures, a higher score indicates higher cognitive function.
Behavioral Data Analysis
Analysis of behavioral data was performed with a custom-written Matlab code (MathWorks). Step length of the ML was used to calculate measures of learning and retention. Step length of the ML was defined as the sagittal distance between heel markers when heel strike occurred, which was identified as the first time point when the force exceeded 20 N. Participants were asked to take their longest possible step during Orientation to ensured that participants were physically able to increase their step length by at least 15% of their step length during Baseline. Individuals who were physically unable to increase their ML step length sufficiently were excluded from the analysis.
Percent error of the ML (PE) was our measure of learning and represents the relative amount of error during learning as a percent of the subjects’ baseline step length, reflecting how accurately participants learned the new walking pattern. It was calculated as
The numerator was the absolute value of the difference between the step length of step n during the Acquisition phases (
To quantify retention, we calculated the Percent Forgotten (%Forgot) as follows:
The numerator was the absolute value of the difference between
Statistical Analysis
All statistical analyses were performed in SPSS v26 (IBM) with an α of 0.05. Prior to completing our primary analyses, we confirmed that participants learned and retained the new walking pattern. To confirm that learning occurred, we compared the average step length of the ML during Baseline and with the average step length during the last 30 strides of Acquisition 2 using a paired t test. Similarly, we confirmed that retention occurred by comparing the average step length of the ML during 24-hour Retention and during the last 30 strides of Acquisition 2.
For our primary analysis, 2 sequential regression models tested the relationship between the primary outcomes of learning and retention with cognition, after accounting for covariates. One model examined the relationship between learning and cognition with the average PE over the last 30 strides of Acquisition 2 as the dependent variable, while the other examined the relationship between retention and cognition with %Forget as the dependent variable. Both models included 4 predictors that were entered in 3 sequential blocks. The first block consisted of one predictor: age. Similarly, the second block consisted of a single predictor: FM-LE. Finally, block 3 included the cognitive measures (ie, FCCS and Spatial Addition). The scores included in the model from these cognitive assessments were not age-corrected as age was included in the first block. All assumptions for regression were tested, including normality, homoscedasticity, and no multicollinearity. Multicollinearity was assessed with the variance inflation factors (VIF), values greater than 10 would indicate a concern for multicollinearity. All VIF values were less than 2 for both regressions; thus, the analysis was conducted as detailed above. Additionally, a correlation matrix of all predictors are presented in the Supplemental Table S1. Normality was initially violated for both models. One outlier was identified (defined as a residual beyond 3 standard deviations from the mean) in each model and were therefore removed, resulting in the assumptions being satisfied. Sensitivity analyses revealed that the results were consistent whether the outlier was included or not; since the excluded results met all assumptions they are presented here. Here we present the R2 and change in R2 (ΔR2) for the model as each block is added. The P value associated with the whole model indicates if the model with all entered predictors is significant, while the P value associated with the ΔR2 reflects whether the addition of the new block of predictors significantly improves the model. Last, we will present the standardized coefficients for both of the complete models (ie, the learning and retention regression for all 3 blocks of predictors).
Results
Thirty-five individuals after stroke were enrolled in the study; however, 15 additional participants who were enrolled in a separate study completed the same protocol on Day 1 and the same physical and cognitive tests on Day 3 (ie, these subjects completed a different protocol on Day 2). Thus, these additional 15 individuals were included in the regression model for learning, bringing the total sample for the learning model to 50. However, one outlier was identified, resulting in 49 participants (64.9 ± 9.9 year old; 23 females) being included in the final analysis of learning. After removal of an outlier from the retention analysis, 34 individuals (63.7 ± 10.0 years old; 14 females) were included in the retention regression.
When instructed to take the longest step possible during Orientation, all subjects increased the step length of the ML by more than 15% of their baseline step length (28.3 ± 14.0%); thus, no participants were excluded from the analysis due to inability to physically complete the task. Additional demographic, clinical, and behavioral information of those included in the learning and retention analyses are presented in Table 1. To summarize, scores on the cognitive tests indicate that our sample had below average cognitive abilities. This is specifically illustrated by scores on the FCCS, which is reported as a standard score (ie, 100 is average and 15 is the standard deviation of the population). In our sample, 22 out of 49 individuals were more than 1 standard deviation below average on the FCCS (ie, less than 85). This was expected given the prevalence of cognitive deficits following stroke. Additionally, on average our sample included individuals who had mild physical impairment as measured by the FM-LE (26.0 ± 5.9). Last, the characteristics of those in the learning and retention analysis were not significantly different (P > .05).
Description of Participants Included in the Regression Examining the Relationship Between Cognition and Learning (n = 49) and Participants Included in the Regression Examining the Relationship Between Cognition and Retention (n = 34).
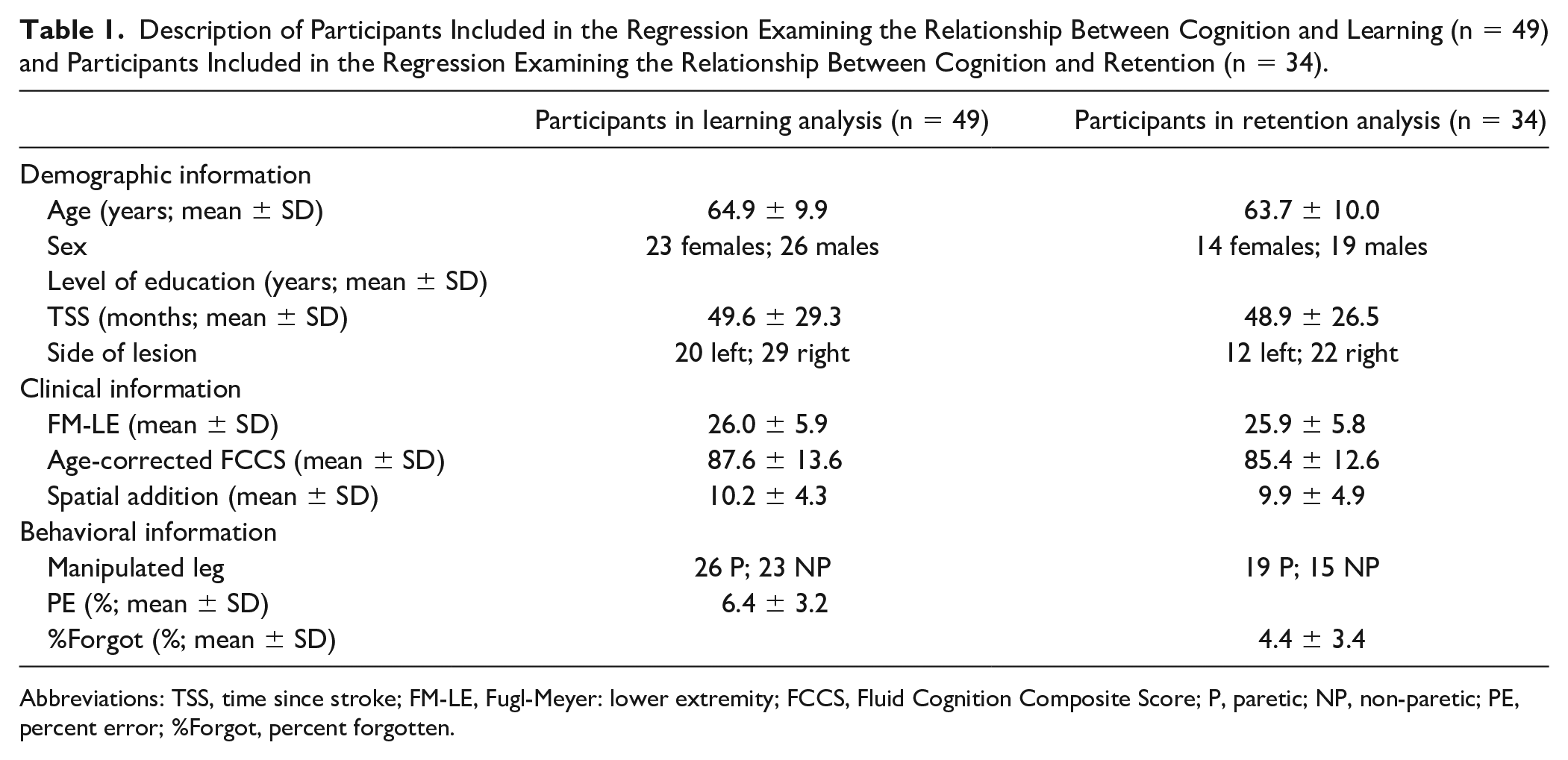
Abbreviations: TSS, time since stroke; FM-LE, Fugl-Meyer: lower extremity; FCCS, Fluid Cognition Composite Score; P, paretic; NP, non-paretic; PE, percent error; %Forgot, percent forgotten.
As a whole, participants after stroke learned a new walking pattern that was significantly different from their walking pattern at Baseline (t[48] = −13.8, P < .001; Figure 2A). Similarly, as a group, participants after stroke retained a walking pattern that was not significantly different than the pattern that was learned (t[33] = −0.3, P = .78; Figure 2B). This suggests that the group learned and retained the new walking pattern; however, as we have previously observed in neurologically intact adults,35,41 there was substantial intersubject variability in learning and retention that was explored in our primary analyses.
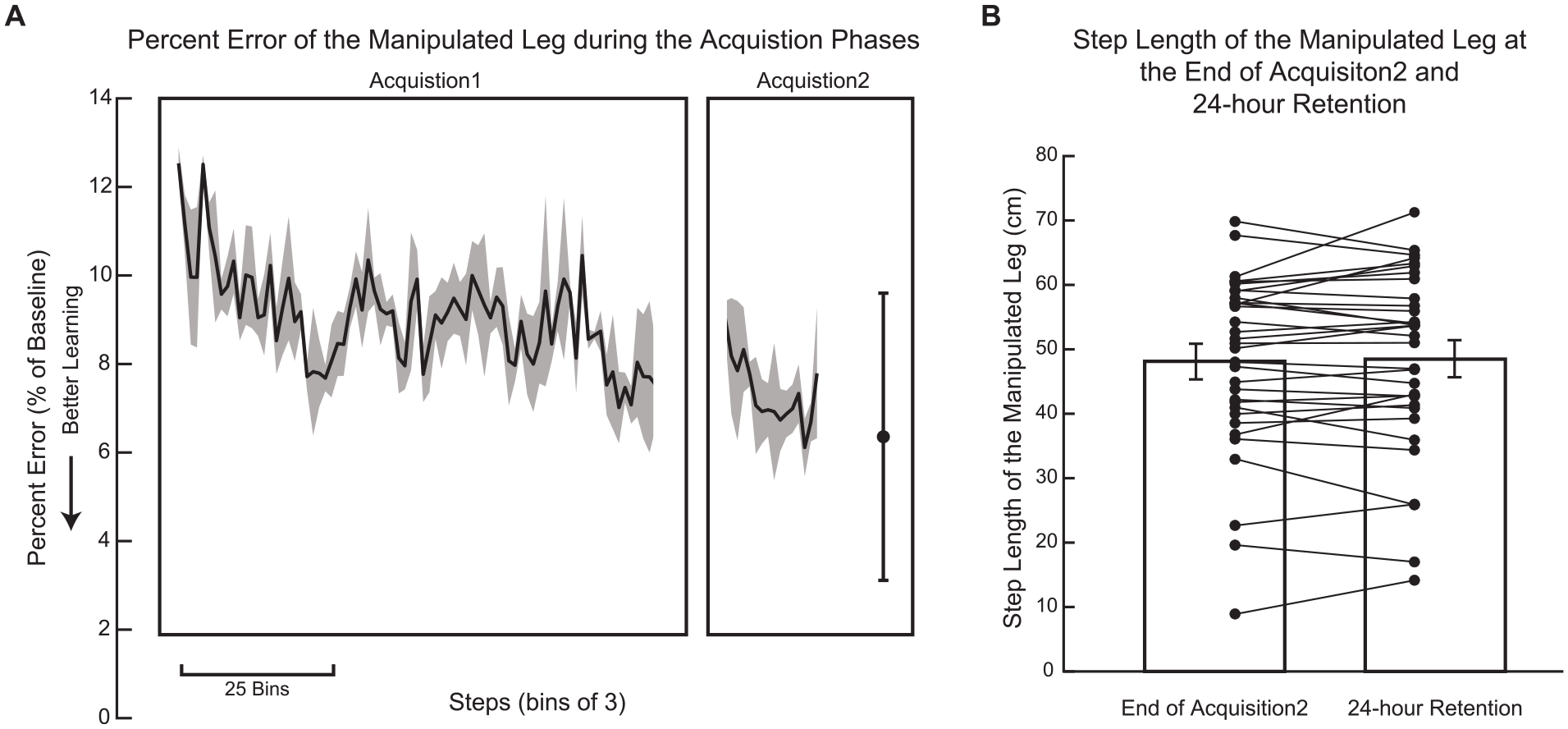
Learning and retention. (A) Percent Error decreased throughout Acquisition 1 and Acquisition 2 (n = 49), suggesting that they learned a new pattern over the course of these walking bouts. Solid line represents the group average Percent Error in bins of 3 steps, while the shaded region is the standard error of the mean. Data in both Acquisition 1 and Acquisition 2 are truncated to the participant with the fewest number of steps. The final point represents the average Percent Error during the last 30 steps of Acquisition 2 with error bars indicating the standard deviation. (B) Mean step length of the manipulated leg during the end of Acquisition 2 and during 24-hour Retention was not significantly different (n = 34), suggesting that the learned walking pattern was retained. Error bars represent standard error of the mean and individual points represent each participant.
When examining learning, the first block, age, was not a significant predictor of PE (R2 = 0.03, P = .24). The second block, FM-LE, was significant after accounting for age (ΔR2 = 0.11, P = .02). Last, the third block containing FCCS and Spatial Addition was a significant predictor of learning above and beyond age and physical impairment (ΔR2 = 0.17, P = .008). These results are presented in Table 2. The final model with all 4 predictors explained a significant portion of variability in learning (R2 = 0.31, P = .002). As indicated by their standardized coefficients, FM-LE (β = −0.34, P = .01) and FCCS (β = −0.39, P = .008) were the only significant predictors of learning (Table 3). As indicated by the standardized coefficients, these results suggest that less physical impairment and higher fluid cognition were associated with better learning. For completeness, this analysis was also performed with the 34 individuals who were included in the retention analysis detailed below. The results of this analysis mirror the results when 49 subjects were included. Specifically, the addition of the cognitive assessments resulted in a significant increase in R2 (ΔR2 = 0.16, P = .04). Also FCCS, but not Spatial Addition, was a significant predictor in the final model (β= −0.41, P = .02, and β = −0.02, P = .89, respectively).
Results of the Sequential Regression Examining the Relationship Between Locomotor Learning/Retention and Cognition. a
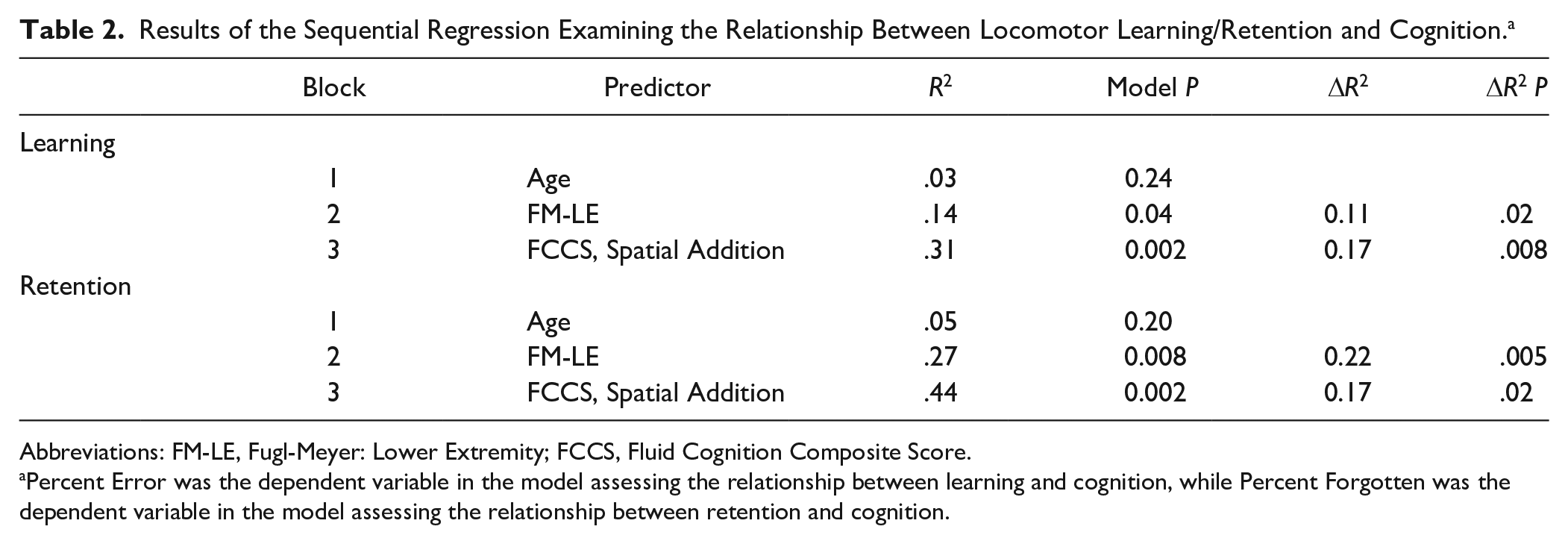
Abbreviations: FM-LE, Fugl-Meyer: Lower Extremity; FCCS, Fluid Cognition Composite Score.
Percent Error was the dependent variable in the model assessing the relationship between learning and cognition, while Percent Forgotten was the dependent variable in the model assessing the relationship between retention and cognition.
Standardized Coefficients for All Variables in the Final Model Examining the Relationship Between (1) Learning and Cognition and (2) Retention and Cognition.
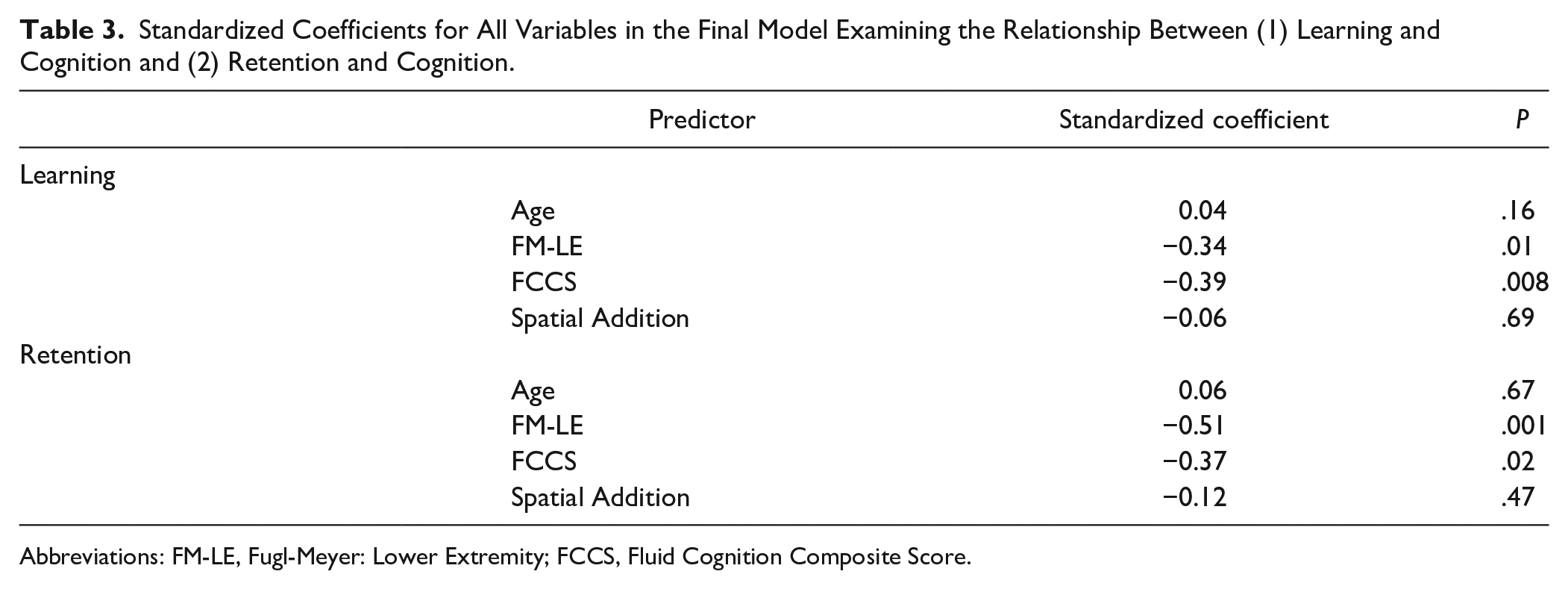
Abbreviations: FM-LE, Fugl-Meyer: Lower Extremity; FCCS, Fluid Cognition Composite Score.
When examining retention, we found a similar pattern, where the first block, age, was not significant predictor of %Forgot (ΔR2 = 0.05, P = .20). Physical impairment (ie, FM-LE) in the second block was significant (ΔR2 = 0.22, P = .005). Last, the addition of the third block with FCCS and Spatial Addition was a significant predictor of retention above and beyond age and physical impairment (ΔR2 = 0.17, P = .023). These results are presented in Table 2. The final model with all 4 predictors explained a significant portion of variability in learning (R2 = 0.44, P = .002). As indicated by the standardized coefficients (Table 3), FM-LE (β = −0.51, P = .001) and FCCS (β = −0.38, P = .02) were the only significant predictors. Again these standardized coefficients indicate that less physical impairment and better fluid cognition were associated with better retention.
As an exploratory analysis, we examined which subtest of the FCCS was most related to learning and retention. Due to the exploratory nature of this analysis α was set to 0.1. This analysis showed that Flanker Inhibitory Control and Attention Test (ρ = −0.49, P < .001), Dimensional Change Card Sort Test (ρ = −0.30, P = .04), and Pattern Comparison Processing Speed Test (ρ = −0.27, P = .06) were related to learning. Additionally, Flanker Inhibitory Control and Attention Test was related to retention (ρ = −0.32, P = .07). Complete results are presented in the Supplemental Table S2.
Discussion
The purpose of this work was to examine the relationship between locomotor learning/retention and cognition. We found that cognition explained a significant portion of variability in locomotor learning and retention above and beyond that explained by age and physical impairment. As evident by the coefficients, fluid cognition but not visuospatial working memory was a significant predictor of locomotor learning and retention. Specifically, individuals after stroke who had lower fluid cognitive abilities did not learn or retain the new walking pattern as well as individuals who had higher fluid cognitive abilities. This is in contrast to past work which has found that visuospatial working memory is the relevant domain of cognition.12 -18 While previous work has shown that cognitive abilities are related to motor function,7 -11 this is the first study to demonstrate the impact of fluid cognitive abilities on locomotor learning after stroke. The implications of these findings are discussed below.
Clinical Implications
Our finding that cognition is a significant predictor of locomotor learning and retention is consistent with previous upper extremity work in neurologically intact adults.12 -20,24 In the current work, we used a visually guided walking task that is largely explicit.35,41 Thus, this work suggests that during a task that is largely explicit, some individuals after stroke will be able to learn and retain well, while others will not. The most common techniques used in gait rehabilitation are those that rely on explicit, strategic learning techniques, such as verbal and visual cues.57,58 However, based on our results, using explicit learning techniques clinically would be effective for individuals with higher fluid cognitive abilities.
From this work, however, we do not know if this relationship holds when the task is practiced for a longer period of time. It is possible that individuals with lower cognitive abilities would do as well as those with higher cognitive abilities if they were provided more practice; thus, future work to examine the effect of increased practice on this relationship would facilitate the delivery of effective rehabilitation of walking for individuals after stroke. Additionally, this work does not speak to the relationship between implicit learning processes and cognition during locomotion. Past work in the upper extremity has found that the relationship between cognition and motor learning is unique to explicit, strategic learning rather than implicit forms of learning,12,14,17 but this has not been tested during locomotion. Future work examining the relationship between cognition and different learning processes during locomotion is needed to direct the development of customized rehabilitation interventions that are rooted in an understanding of motor learning for individuals after stroke.
Potential Mechanisms of the Cognition and Locomotor Learning/Retention Relationship
Past work has suggested that the relationship between upper extremity motor learning tasks and visuospatial working memory is due to overlapping activation of the dorsolateral prefrontal cortex.12,14 In this work, however, we did not find visuospatial working memory specifically to be a significant predictor of learning or retention. Instead, we found fluid cognitive abilities to be a significant predictor. It is possible that the discrepancy in our results compared to previous results from the upper extremity tasks may exist because this region of the brain does not overlap during this task due to differences in the learning task or in locomotion itself. Last, it is also possible that we did not observe the same relationship that was previous observed due to the involvement of the dorsolateral prefrontal cortex in numerous domains of cognition. We will discuss each of these 3 possibilities below.
First, it is possible that the learning occurring in the task in the current work compared to upper extremity motor learning is different; thus, resulting in different aspects of cognition being important. Past work from our laboratory has found that there are both explicit and implicit learning processes occurring during the visually guided walking task; however, explicit learning processes appear to contribute the most to the learned walking pattern.35,41 Similarly, visuomotor rotation involves multiple learning processes, including explicit learning processes, which have been specifically identified as the learning process related to visuospatial working memory.12,14,17 This was theorized to be due to overlapping brain activation in the dorsolateral prefrontal cortex.12,14 We hypothesized that because our learning task was explicit, the dorsolateral prefrontal cortex would be involved in our locomotor learning task, and thus, there may be a relationship with visuospatial working memory as has been previously observed. This hypothesis is supported by the work of Vitorio et al, 38 which found that the prefrontal cortex was active while learning a new walking pattern in response to auditory cues. 38 However, we do not have evidence that the dorsolateral prefrontal cortex was engaged in the visually guided learning task used in the current work. Therefore, it is possible that the dorsolateral prefrontal cortex was not active, thus explaining why visuospatial working memory was not a significant predictor of locomotor learning and retention.
Second, the unique demands of walking compared to upper extremity tasks may have also resulted in different regions of the brain being active. Importantly, past work has shown that the dorsolateral prefrontal cortex is not highly active during steady-state walking. 59 Therefore, the limited role of this region of the brain during walking may help explain the discrepancy between our results and past results in the upper extremity. Additionally, locomotion requires greater postural control and balance than seated upper extremity tasks that have been used in past work, resulting in an important role for the cerebellum. 60 It is also clear that the cerebellum has neural connections to the prefrontal cortex and is active in a number of fluid cognitive functions, such as problem solving, executive functions, and visuospatial working memory.61,62 Although the exact role of the cerebellum in cognition is unclear, it is accepted that the cerebellum plays some role in cognition broadly. 61 Given our current understanding of the role of the cerebellum in cognition broadly, the overlapping activation of the cerebellum during locomotion and during cognition globally may explain the relationship between fluid cognition (a global measure of cognition) and locomotor learning. Additionally, past work suggests that midline structures of the cerebellum are involved in locomotion 60 and that more lateral regions of the cerebellum are involved in visuospatial working memory62,63; thus, this may explain why visuospatial working memory alone was not related to our locomotor learning task.
Last, the dorsolateral prefrontal cortex is involved in cognitive processes other than visuospatial working memory. For example, the dorsolateral prefrontal cortex is thought to be involved in attention and general fluid cognitive abilities as well as visuospatial working memory.64,65 Thus, even if the dorsolateral prefrontal cortex is involved in the specific learning task used in this work, it is possible that the relationship we observe between fluid cognition and locomotor learning and retention is due to overlapping activation of the dorsolateral prefrontal cortex. While our results are clear that fluid cognitive abilities, rather than visuospatial working memory specifically, is related to locomotor learning and retention, the mechanism is less clear. The above discussion is based on the evidence that we have from past work regarding locations of the brain involved in locomotion, locomotor learning, explicit learning, and cognitive function; however, future work with functional near-infrared spectroscopy and other techniques that can provide insight into the active regions of the brain during both fluid cognitive testing and locomotor learning is warranted to help understand the mechanism of this relationship.
Assessment of Cognition
Cognition is an extremely complex construct that involves highly correlated domains.21,22 As a result, there are many ways to assess cognition. The current work and past work have used a variety of different tasks and assessments to measure cognition, and specifically visuospatial working memory, thus making it challenging to compare results. This issue is particularly pertinent for visuospatial working memory since it is highly related to other cognitive functions21,22 and involves regions of the brain known to be involved in other cognitive functions.64,65 Thus, it is possible that the measures of visuospatial working memory used in this work and past work reflect visuospatial working memory differently and may capture aspects of other domains of cognition in different manners. This issue is further exacerbated by the use of a single assessment to reflect very complex cognitive domains in both the present and past work. Because of the strong relationships between various cognitive domains, a more robust way to measure cognition would be to use existing tools, such as the Repeated Battery of Neuropsychological Status and the Wechsler Memory Scale, that use multiple assessments to reflect specific cognitive domains. Future work utilizing this type of approach would aid in advancing our understanding of the relationship between motor learning and specific cognitive domains.
Additionally in the current work, fluid cognition was measured as a composite score of 5 subtests. This construct is extremely broad which could be viewed as a strength or a detriment to this work. The relationship between cognition and locomotor learning found in this work is important as it has implications for the structure of rehabilitation interventions after stroke. The fact that this relationship exists between a broad, global measure of cognition is valuable as it may be easier for rehabilitation professionals to use this information to guide rehabilitation interventions. For example, established screening tools provide rehabilitation professionals with a broad understanding of cognitive function; thus, a relationship between locomotor learning and general fluid cognition, rather than a specific cognitive domain that requires more detailed assessment, would enable clinicians to use this information to guide the delivery of rehabilitation interventions more easily. Future work to examine the relationship between cognition and locomotor learning using commonly used clinical screening tools would be valuable to the clinical utility of these findings. Conversely, the broad nature of these findings are limited in their ability to point to a specific mechanism that explains the relationship between cognition and locomotor learning. This limitation highlights the importance of future work that uses techniques such as functional near-infrared spectroscopy and electroencephalogram to understand the regions of the brain involved during locomotor learning and to identify specific cognitive domains that may explain this relationship.
In an effort to guide this future work, we examined the correlation of the 5 subtests of the FCCS and our metrics of learning and retention in an exploratory analysis. Based on our exploratory analysis, it seems that attention, measured by Flanker Inhibitory Control and Attention Test, may be a domain of cognition that is particularly important for understanding variability in locomotor learning and retention. This finding is particularly interesting given the strong relationship between attention and visuospatial working memory21,22 and the involvement of the dorsolateral prefrontal cortex in attention. 65 It is possible that these 2 domains of cognition are so tightly linked that distinguishing between these domains has not been successful in past work. However, it is important to note that like other domains of cognition, attention is a complex construct; thus, performing factor analyses with multiple cognitive assessments to quantify each specific cognitive domain would provide a more robust assessment of attention and other cognitive domains. Therefore, the findings of this exploratory analysis should be interpreted cautiously and used as a guide for future research examining the relationship between various cognitive domains, including attention and locomotor learning.
Limitations
The work adds important information to the body of literature on cognition and locomotor learning; however, it is not without limitations. First, we did not include all factors that may explain variability in learning and retaining a new walking pattern. For example, patient information, such as time since stroke and level of education, and genetic information, such as brain-derived neurotrophic factor genotype, may be important predictors of locomotor learning and should be explored in future work. Additionally, the cognitive tests were administered after the motor testing; therefore, it is possible that completion of the motor testing influenced performance on the cognitive testing. Although this is unlikely due to the duration between motor testing and cognitive testing (2.4 ± 2.1 days), it is a possibility. Next, we manipulated the leg that took a shorter step for every participant. As a result, some participants were required to change their step length on the paretic leg, while others were required to change their step length on the nonparetic leg. While this is similar to what is done in clinical practice, it is possible that this affected the relationship between FM-LE and learning/retention, thereby affecting our results. Additionally, while we included a fairly broad sample in this work (ie, we did not have inclusion criteria related to gait speed or cognition), we did exclude individuals who had evidence of neglect or reported vision changes after their stroke. These individuals were excluded to ensure that all participants could see the visually display; however, it does limit the generalizability of our findings to stroke survivors who have these conditions. Last, although the FCCS has evidence for validity in adults with stroke,50,51 it is a global measure of cognition; therefore, we are unable to conclude what specific domain of cognition is most important for to this relationship.
Conclusions
This work is the first to examine the relationship between cognition and locomotor learning after stroke. Based on our results, fluid cognition relates to the ability of individuals after stroke to learn and remember a new walking pattern that was learned through an explicit, strategic learning paradigm. This provides a foundation for developing our understanding of the complex relationship between locomotor learning and cognition. Coupled with future work, this work will facilitate the development and prescription of interventions that capitalize on the learning processes each individual can successfully accomplish in order to provide more effective and efficient rehabilitation interventions to improve gait after stroke.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211001025 – Supplemental material for Fluid Cognitive Abilities Are Important for Learning and Retention of a New, Explicitly Learned Walking Pattern in Individuals After Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683211001025 for Fluid Cognitive Abilities Are Important for Learning and Retention of a New, Explicitly Learned Walking Pattern in Individuals After Stroke by Margaret A. French, Matthew L. Cohen, Ryan T. Pohlig and Darcy S. Reisman in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683211001025 – Supplemental material for Fluid Cognitive Abilities Are Important for Learning and Retention of a New, Explicitly Learned Walking Pattern in Individuals After Stroke
Supplemental material, sj-docx-2-nnr-10.1177_15459683211001025 for Fluid Cognitive Abilities Are Important for Learning and Retention of a New, Explicitly Learned Walking Pattern in Individuals After Stroke by Margaret A. French, Matthew L. Cohen, Ryan T. Pohlig and Darcy S. Reisman in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank the participants of this study and the undergraduate research assistants who aided with data collection, processing, and analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (2R01HD078330-05A1, S10RR028114-01, and F31NS111806) and The Foundation for Physical Therapy Research (2016 Florence P Kendall Scholarship, 2018 Promotion of Doctoral Studies [PODS] Level I Award, and 2019 Promotion of Doctoral Studies [PODS] Level II Award).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.