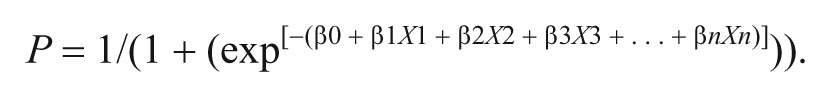Abstract
Background. Early prognosis, adequate goal setting, and referral are important for stroke management. Objective. To investigate if independent gait 6 months poststroke can be accurately predicted within the first 72 hours poststroke, based on simple clinical bedside tests. Reassessment on days 5 and 9 was used to check whether accuracy changed over time. Methods. In 154 first-ever ischemic stroke patients unable to walk independently, 19 demographic and clinical variables were assessed within 72 hours and again on days 5 and 9 poststroke. Multivariable logistic modeling was applied to identify early prognostic factors for regaining independent gait, defined as ≥4 points on the Functional Ambulation Categories. Results. Multivariable modeling showed that patients with an independent sitting balance (Trunk Control Test–sitting; 30 seconds) and strength of the hemiparetic leg (Motricity Index leg; eg, visible contraction for all 3 items, or movement against resistance but weaker for 1 item) on day 2 poststroke had a 98% probability of achieving independent gait at 6 months. Absence of these features in the first 72 hours was associated with a probability of 27%, declining to 10% by day 9. Conclusions. Accurate prediction of independent gait performance can be made soon after stroke, using 2 simple bedside tests: “sitting balance” and “strength of the hemiparetic leg.” This knowledge is useful for making early clinical decisions regarding treatment goals and discharge planning at hospital stroke units.
Introduction
Regaining independent gait is considered a primary goal in stroke rehabilitation. 1 A number of prospective cohort studies have shown that approximately 60% 2,3 to 80% 4,5 of stroke patients are able to walk independently at 6 months poststroke. Early accurate prediction of the outcome in terms of independent gait poststroke is paramount for stroke management, in order to: (a) facilitate an early start of proper discharge planning, (b) inform clients and their relatives properly, (c) set realistic multidisciplinary therapeutic goals, 6 and (d) anticipate possible consequences such as home adjustments and the need for community support. 7 A number of prognostic studies suggest that age, 8-10 severity of sensory and motor dysfunction of the paretic leg, 9,11 homonymous hemianopsia, 9,11 incontinence for micturation, 8,10 sitting balance, 10,12-14 initial disability in activities of daily living and ambulation, 2,10,12 level of consciousness at admission, 8 and the number of days between stroke onset and first assessment 15 are independently associated with gait outcomes 6 months after stroke. 12
Unfortunately, it is difficult to compare results of these prognostic studies due to differences in patient characteristics, the candidate determinants selected for regression modeling, measurement instruments used and definitions of gait, as well as poststroke timing of physical assessments, ranging from 7 days 10 to 14 days. 11,12 It is particularly the moment when clinical determinants are measured after a stroke that influences the accuracy of prediction, since early spontaneous neurological recovery as a function of time is an important confounder in understanding prognostic models. 3,16 This finding suggests that determinants such as continence, sitting balance, and severity of hemiplegia should be measured preferably at a fixed moment after stroke and not at nonfixed moments such as the time of admission to a stroke rehabilitation ward. 16,17
The first aim of the present study was to investigate if independent gait at 6 months poststroke can be accurately predicted within the first 72 hours after stroke, in order to optimize early goal setting and referral policy in hospital stroke units. The second objective was to investigate the effects of early reassessment on days 5 and 9 on the accuracy of predicting outcomes in terms of regaining independent gait at 6 months poststroke.
Materials and Methods
Design and Procedures
The EPOS study (acronym for Early Prediction of Functional Outcome after Stroke) is a prospective cohort study with an intensive repeated-measurements design during the first 2 weeks poststroke. Patients were recruited from 9 hospital stroke units in the Netherlands. Assessments were performed within 72 hours and on days 5 and 9 poststroke. The main pragmatic reasons for (re)testing every 3 days were that daily testing would intervene in the usual rehabilitation program for included patients at (sub)acute stroke units, whereas previous studies have shown that the added value of day-to-day change is limited. Considering the nonlinear recovery pattern observed early poststroke as well as the early discharge policy of patients with stroke from hospital stroke units to other care facilities, we restricted to reassessment of the candidate determinants up to day 9 poststroke. Final outcome was measured at 6 months poststroke. All assessments were performed by trained physical therapists from each participating stroke unit, and patients received physical therapy treatment according to the Dutch guidelines for physical therapists. 18 The EPOS study was approved by the ethics committees of the participating hospitals.
Subjects
Stroke was defined according to the World Health Organization criteria. 19 The Bamford classification was used to record the size and site of the infarct, based on clinical features. 20 Patients were included when they met the following criteria: (a) having suffered an ischemic first-ever anterior circulation stroke; (b) suffering from hemiparesis within 72 hours poststroke, even after application of recombinant Tissue Plasminogen Activator; (c) no disabling medical history (ie, a premorbid Barthel Index score ≥19); (d) aged 18 years or older; (e) no severe deficits of communication, memory, or understanding; (f) being unable to walk independently (ie, Functional Ambulation Categories [FAC] <4) within 72 hours; and (g) giving written informed consent to participate.
Dependent Variable
The outcome variable was independent gait, measured by the FAC. The FAC is a reliable and valid tool to classify the level of physical support needed by patients to ambulate safely after a stroke, and it comprises 6 levels (0-5), ranging from “nonfunctional ambulation” (0) to “ambulate independently, on level and nonlevel surfaces including stairs and inclines” (5). 21-23 For the present study, the FAC score was dichotomized into 0 (dependent gait, FAC <4) and 1 (independent gait, FAC ≥4). 22,23
Independent Variables
The 19 variables used to develop a prediction model included gender, age, body mass index, social support, comorbidity (Cumulative Illness Rating Scale 24 ), hemisphere of stroke, days between stroke onset and first assessment, type of stroke (Bamford classification 20 ), urinary incontinence (Barthel Index 25 item 1), sitting balance (Trunk Control Test item 3 [TCT-s] 26,27 ; ie, sitting unsupported for 30 seconds), muscle strength of the upper (Motricity Index [MI] arm 26,28 ) and lower paretic limb (MI leg), synergism of upper (Fugl-Meyer [FM] arm 29 ) and lower extremity (FM leg), and 5 neurological functions: consciousness at onset (National Institutes of Health Stroke Scale [NIHSS] 30 item 1A; no/yes), extinction or inattention (NIHSS item 11; yes/no), hemianopsia (NIHSS item 3; yes/no), conjugate deviation (NIHSS item 2; yes/no), and sensory loss (NIHSS item 8; yes/no).
Data Analysis
Only subjects with completed baseline and 6 month assessments were included in statistical analysis. Baseline demographics, stroke characteristics, and severity of impairments caused by the stroke were recorded. Candidate determinants were preferably dichotomized (0/1) on the basis of clinical grounds. Otherwise, the optimal cutoff point for each determinant was established by applying a receiver–operating characteristic analysis. Sensitivity/1 − specificity and area under the curve for each cutoff score were used to estimate the optimal dichotomization and was estimated for each individual variable. In case of missing values for day 5 or 9, the last value was carried forward.
Subsequently, bivariable logistic regression analysis was applied to calculate odds ratios (OR) with 95% confidence intervals (95% CI) for each individual variable to identify statistically significant determinants related to the dichotomized FAC score at 6 months. To prevent omission bias, determinants with a liberal significance level of p ≤ .10 were selected for further analysis. Collinearity diagnostics (rs ) were used to prevent spurious relationships in the multivariate regression model. If the correlation coefficient (rs ) was ≥.70, only one of these determinants was selected for further use in the multivariate analysis.
The retained determinants were used for forward stepwise multivariable logistic regression analysis. These analyses were tested 2-tailed using critical P values for entry and removal of ≤.05 and ≥.10 respectively. The probabilities of developing independent gait at 6 months poststroke were calculated from the multivariable model using the constant and regression coefficients of the included determinants in the following equation:
Finally, 2-way contingency tables were used to calculate sensitivity, specificity, and positive predictive value (PPV) and negative predictive value (NPV), including their 95% CIs, for the derived model within 72 hours and on days 5 and 9 poststroke. Statistics were performed using SPSS version 15.
Results
Between February 2007 and November 2009, 221 patients were recruited. Thirty-five patients were lost to follow-up, for reasons including death (N = 21), withdrawal (N = 3), recurrent stroke (N = 5), or other reasons such as migration (N = 2). Another 32 were excluded from the analysis because they achieved independent gait within 72 hours. Hence, a total of 154 patients were available for further analysis. Patient demographics and associated stroke characteristics are shown in Table 1. At 6 months poststroke, 122 patients (79%) were able to walk independently (FAC ≥4).
Characteristics of Patients Measured Within 72 Hours Poststroke
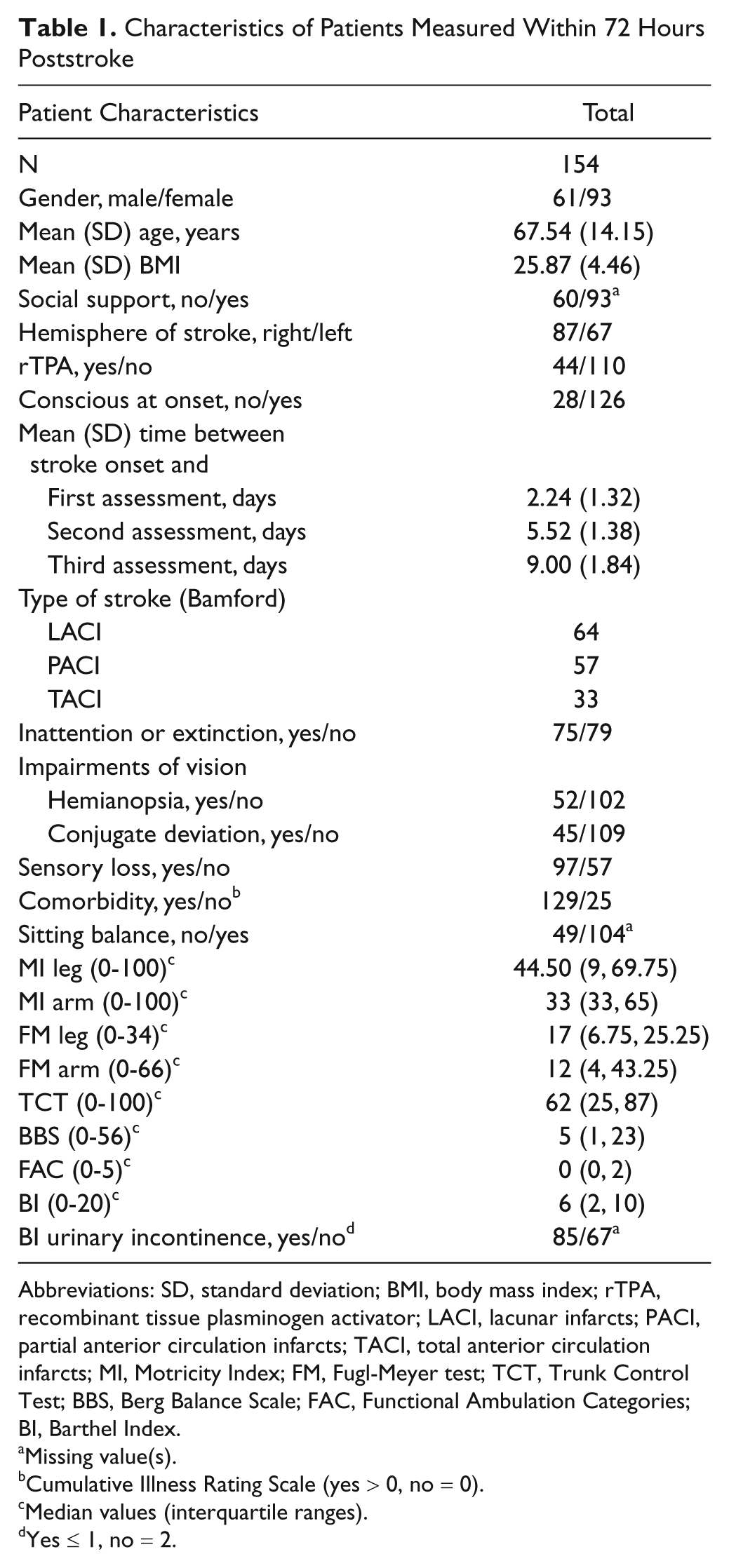
Abbreviations: SD, standard deviation; BMI, body mass index; rTPA, recombinant tissue plasminogen activator; LACI, lacunar infarcts; PACI, partial anterior circulation infarcts; TACI, total anterior circulation infarcts; MI, Motricity Index; FM, Fugl-Meyer test; TCT, Trunk Control Test; BBS, Berg Balance Scale; FAC, Functional Ambulation Categories; BI, Barthel Index.
Missing value(s).
Cumulative Illness Rating Scale (yes > 0, no = 0).
Median values (interquartile ranges).
Yes ≤ 1, no = 2.
Bivariable Association Between Dependent Variable and Independent Variables
Table 2 shows ORs and their 95% CIs for independent gait at 6 months, as determined by bivariable logistic regression analysis for the assessment within 72 hours after stroke onset. Of 19 candidate variables, 15 were significantly related to the return of independent gait at 6 months poststroke. Collinearity diagnostics revealed a high level of association between the MI leg score and the FM leg score (rs = .81, p = .01). The MI arm score was strongly associated with the FM arm score (rs = .84, p = .01). The MI leg and arm scores were selected for regression modeling, based on clinical applicability (ie, MI takes less time to administer than the motor part of FM).
Impairments and Disabilities Within 72 Hours Associated With Independent Gait 6 Months Poststroke as Determined by Bivariable Logistic Regression Analysis (N = 154)
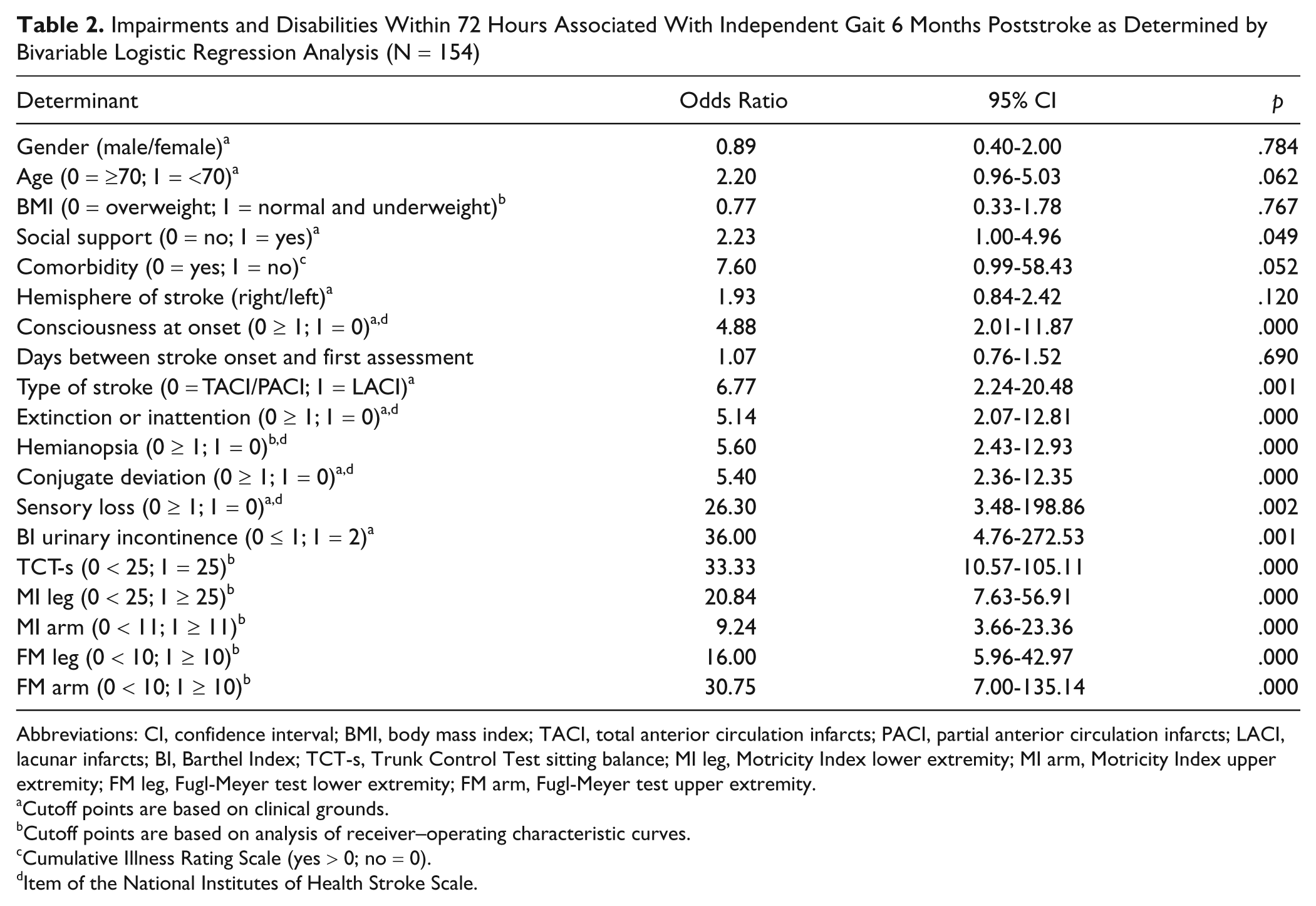
Abbreviations: CI, confidence interval; BMI, body mass index; TACI, total anterior circulation infarcts; PACI, partial anterior circulation infarcts; LACI, lacunar infarcts; BI, Barthel Index; TCT-s, Trunk Control Test sitting balance; MI leg, Motricity Index lower extremity; MI arm, Motricity Index upper extremity; FM leg, Fugl-Meyer test lower extremity; FM arm, Fugl-Meyer test upper extremity.
Cutoff points are based on clinical grounds.
Cutoff points are based on analysis of receiver–operating characteristic curves.
Cumulative Illness Rating Scale (yes > 0; no = 0).
Item of the National Institutes of Health Stroke Scale.
Multivariable Modeling
Table 3 shows the variables included in the prediction model and the probabilities of achieving independent gait 6 months poststroke. Testing all 15 significant candidate determinants simultaneously in a multivariable regression analysis resulted in a final model including 2 significant variables: TCT sitting balance and MI leg score. Based on regression coefficients and constant, the maximum probability of achieving independent gait was estimated at 98% if the patients had a maximum TCT-s score of 25 (ie, independent sitting for 30 seconds) and an MI leg score of ≥25 (eg, visible contraction but no movement for all 3 test items, or movement against resistance but weaker than the nonparetic side for one item) within 72 hours poststroke, whereas a probability of 27% was found for patients who did not reach this level within 72 hours after stroke onset. An analysis of the time dependency of these probabilities for the former group showed that probabilities had remained about the same on days 5 and 9, whereas in the latter group, the probability of achieving independent gait was reduced to 23% on day 5 and to 10% on day 9.
Probabilities of Achieving Independent Gait Six Months Post Stroke (N = 154)
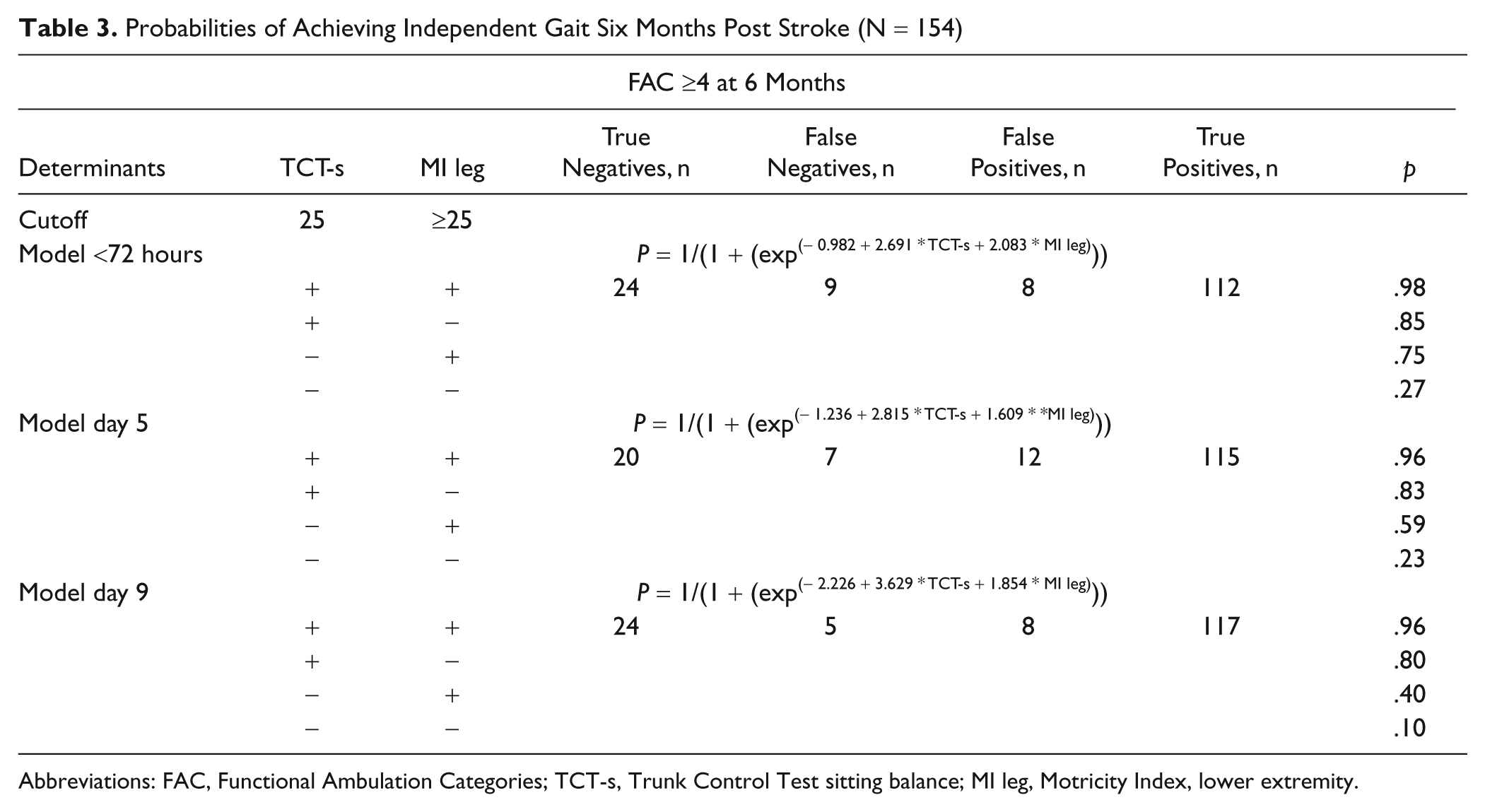
Abbreviations: FAC, Functional Ambulation Categories; TCT-s, Trunk Control Test sitting balance; MI leg, Motricity Index, lower extremity.
The sensitivity of the model ranged from 0.93 (95% CI = 0.86-0.96) on day 2 to 0.94 (95% CI = 0.87-0.97) on day 9, whereas specificity ranged from 0.63 (95% CI = 0.43-0.78) on day 5 to 0.83 (95% CI = 0.64-0.93) at day 9. The PPV changed from 0.93 (95% CI = 0.85-0.96) on day 2 to 0.96 (95% CI = 0.90-0.98) on day 9, whereas the NPV changed from 0.63 (95% CI = 0.57-0.82) on day 5 to 0.75 (95% CI = 0.56-0.88) on day 9.
Discussion
To the best of our knowledge, this is the first study to investigate prospectively the accuracy of predicting independent gait within 72 hours after stroke onset in nonambulatory stroke patients. The present study showed that accurate prediction within 72 hours is attainable at hospital stroke units by means of 2 simple bedside tests: sitting balance and muscle strength of the paretic leg. Those nonambulatory patients who regained their sitting balance as assessed by the TCT-s and developed some voluntary movement of the hip, knee, and/or ankle as assessed by the MI leg score (≥25 points) within the first 72 hours poststroke had about a 98% chance of regaining independent gait within 6 months. In contrast, those patients who were unable to sit independently for 30 seconds and were not or hardly able to contract the muscles of the paretic lower limb within 72 hours had a probability of about 27% of achieving independent gait. Early reassessment of sitting balance and lower limb strength on days 5 and 9 showed that if sitting ability and lower limb strength failed to recover, the probability of regaining independent gait declined to 23% on day 5 and 10% on day 9 poststroke.
The aforementioned findings allow valid prediction of independent gait after stroke and are important for clinicians to improve their discharge policy at hospital stroke units and to support the standardization of care processes in clinical pathways after stroke. These findings on the early prediction of outcome poststroke are important, since it takes advantage of a decreased length of stay at a hospital stroke unit and could help to tailor rehabilitation interventions.
Comparing our findings with those of other studies is difficult because of the lack of prognostic studies investigating the accuracy of prediction within 72 hours. However, a number of prospective studies have shown that muscle strength of the hemiplegic leg 9,11 and sitting balance, 10,13 when measured in the second to fourth week after stroke, are significantly associated with improvement of walking ability 3 and achieving independent gait 12,14,17 at 6 months. Obviously, the early control of sitting balance as a prerequisite for regaining standing balance and gait is an important factor for the final outcome at 6 months. 14,17 The importance of balance control for gait is also supported by a study by Kollen and colleagues, 3 who showed that improvement in standing balance was the most important variable associated with improvement of gait performance as measured with the FAC.
Since the proportion of false positives (≈7%) was clearly smaller than the proportion of false negatives (≈27%) within 2 days, the present study suggests that our model is generally somewhat pessimistic and illustrates that some patients with an initially poor sitting balance and a severe paresis of the hemiplegic limb will nevertheless regain independent gait. 12 The increasing accuracy of prediction over time may reflect underlying intrinsic neurological mechanisms such as elevation of diaschisis after stroke. 16,31 This elevation is generally completed within the time window used in this study, 31 making it virtually impossible to improve the accuracy of prediction based on the 2 variables of our model. For example, our model does not take into account the ability or inability of patients to use compensatory strategies. This is supported by a number of recent longitudinally conducted studies showing that gait recovery is closely related to learning to use compensatory movement strategies. 3,31,32 For instance, patients learn to keep their balance by shifting their center of gravity to the nonparetic side, 32 while significant change in motor control on the paretic side is almost lacking. 33 Obviously, patients learn to cope with existing neurological deficits. 33
Future studies should not only investigate the optimal timing of clinical assessments at hospital stroke units but also try to gain insight into recovery profiles in the early poststroke period. 34 For this purpose, cohort studies should use an intensive repeated-measures design, allowing clinicians to increase their understanding of the longitudinal relationships between early changes in body functions, such as coordination, compensation strategies, and improved gait performance, preferably by including kinematic and electromyography measures. Simultaneously, neuroimaging techniques such as functional magnetic resonance imaging should be added to increase our knowledge about the neurophysiological mechanisms that accompany gait recovery.
The present study was subject to some limitations, such as being restricted to first-ever anterior circulation strokes and the lack of cross-validation of the derived prediction model in an independent sample of stroke patients. In addition, the influence of some cognitive and emotional disabilities such as depression 35 and anxiety on recovery were not addressed in the present study, even though they could negatively influence independent gait. Finally, although recovery of walking function mainly occurs within 6 months poststroke, 2,36 about 10% of the patients still show significant functional changes after these 6 months. 37 Hence, we cannot exclude that patients who are nonambulatory within the first 6 months may still be dependent in this chronic phase poststroke. 37,38
In conclusion, the 2 simple tests of “sitting balance” and “strength of the paretic leg” allow early and accurate prediction within 2 days poststroke of the recovery of walking ability after 6 months for nonambulatory patients who score positive on both tests. Patients with a poor prognosis should be reassessed with these 2 tests on day 9 to improve the accuracy of prediction.
EPOS Investigators
Executive committee: G. Kwakkel (PhD), principal investigator, Department of Rehabilitation Medicine, VU University Medical Centre, Amsterdam; and co–principal investigator, B. C. Harmeling–Van der Wel, Department of Rehabilitation Medicine, Erasmus MC, Rotterdam. Steering committee and data management: J. M. Veerbeek and R. Nijland, Department of Rehabilitation Medicine, VU University Medical Centre, Amsterdam. Monitoring board: M. A. Van der Beek, UMC Utrecht; W. A. M. Cornelissen, AMC Amsterdam; A. A. G. Goos, Franciscus Hospital, Roosendaal; C. S. Steeg, UMC Sint Radboud, Nijmegen; J. M. Timmermans, LUMC, Leiden; R. Tichelaar, Amphia Hospital, Breda.
Footnotes
Acknowledgements
We thank all EPOS assessors at the stroke units of the participating university centers and local hospitals (ie, AMC, Amsterdam; Erasmus MC, Rotterdam; LUMC, Leiden; UMC Sint Radboud, Nijmegen; UMC Utrecht; VUmc, Amsterdam; Amphia Hospital Breda; Diaconessen Hospital, Leiden; Franciscus Hospital, Roosendaal) and at the associated nursing homes (ie, St Jacob, Zonnehuis and Cordaan/Berkenstede in Amsterdam, Laurens Antonius Binnenweg and Reumaverpleeghuis in Rotterdam, Albert van Koningsbruggen in Utrecht, and Wiekendaal in Roosendaal) for performing the measurements, and we thank all patients who participated in the study. Finally, we thank E. Blaauw and M. R. Wirtz, students at Hogeschool Amsterdam, for their contribution to the data management.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was part of the EPOS research project funded by the “Wetenschappelijk College Fysiotherapie” (Grant Number 33368) of the Royal Dutch Society for Physical Therapy (KNGF), the Netherlands.