Abstract
Background
Postural instability and gait dysfunction (PIGD) is a cardinal symptom of Parkinson’s disease (PD) and is exacerbated under dual-task conditions. Dual-task training (DTT), enhances gait performance, however it is time and cost intensive. Digitizing DTT via the Dual-task Augmented Reality Treatment (DART) platform can expand the availability of an effective intervention to address PIGD.
Objective
The aim of this project was to evaluate DART in the treatment of PIGD in people with PD compared to a Traditional DTT intervention. It was hypothesized that both groups would exhibit significant improvements in gait, and the improvements for the DART group would be non-inferior to Traditional DTT.
Methods
A single-blind randomized controlled trial was conducted with 47 PD participants with PIGD. Both groups completed 16 therapeutic sessions over 8 weeks; the DART platform delivered DTT via the Microsoft HoloLens2. Primary outcomes included clinical ratings and single- and dual-task gait biomechanical outcomes.
Results
Clinical measures of PD symptoms remained stable for DART and Traditional DTT groups. However, both groups exhibited a significant increase in gait velocity, cadence, and step length during single- and multiple dual-task conditions following the interventions. Improvements in gait velocity in the DART group were non-inferior to Traditional DTT under the majority of conditions.
Conclusion
Non-inferior improvements in gait parameters across groups provides evidence of the DART platform being an effective digital therapeutic capable of improving PIGD. Effective digital delivery of DTT has the potential to increase use and accessibility to a promising, yet underutilized and difficult to administer, intervention for PIGD.
Clinical Trial Registration
ClinicalTrials.gov Dual-task Augmented Reality Treatment for Parkinson’s Disease (DART) NCT04634331; posted November 18, 2020.
Introduction
Postural instability is a cardinal motor symptom of Parkinson’s disease (PD) as nearly 80% of people with PD (PwPD) fall at least once per year and many experience multiple falls during daily activities. 1 Falls, coupled with cognitive decline, are frequently cited as a leading cause for loss of independence and hospitalization in PwPD. 2 Multiple theories regarding the mechanism underlying dual-task interference exist 3 ; fundamentally, voluntary movement is not wholly automatic and the addition of an attention-demanding cognitive task re-allocates attentional resources and generally results in performance degradation. Task degradation is further compromised in PwPD as the connectivity between the striatum and the motor cortex during self-initiated movement is lessened due to PD. 4 The cognitive component of routine daily activities contributes to increased fall rates in PwPD relative to healthy peers. 5
Dual-task interference in PwPD has been well-defined with declines in gait velocity, step length, and arm swing under dual-task conditions.6 -8 Over the past decade, positive results from several randomized clinical trials in PwPD indicate that training can improve performance. Dual-task training (DTT) produced positive changes in gait, balance, physical activity, and dual-task cognitive performance in PwPD relative to a usual care control group. 9 Two recent reviews indicate that DTT is suitable for PwPD and is effective in improving gait function.10,11
Despite overwhelming support for DTT in improving gait performance for PwPD, fundamental barriers in its delivery have prevented widespread adoption (eg, time intensive and timing of DTT relative to On\Off medication periods). 12 Although >90% of surveyed rehabilitation providers reported having PD-specific training, 12 providers reported challenges with implementing DTT in PwPD. Obstacles included, time needed to create tailored interventional programs and keeping the intervention challenging and engaging. 12 The known benefits associated with DTT coupled with the barriers in delivering DTT make it ideally suited to be a digital therapeutic (DTx).
A subdivision of digital health, a DTx is defined as “using technology to deliver evidence-based therapeutic interventions to patients that are driven by software to prevent, manage, or treat a medical disorder or disease. DTxs are used. . .to optimize patient care.” 13 In order for a digital intervention to be considered a DTx, it should undergo a rigorous evaluation relative to a treatment or intervention previously supported by clinical trial evidence.14,15 Generally, to be considered a DTx the digital therapy should be equivalent or not inferior to the evidence-based reference therapy.
Head-mounted augmented reality (AR) devices, such as the Microsoft HoloLens2 (HL2), are suited to deliver DTT as they are untethered devices that place digital objects in the real-world of the user while possessing the capability to recognize verbal responses and accurately and objectively evaluate gait function.16 -18 The fusing of the real and digital worlds creates a unique, enhanced environment that is conducive to delivering DTT scenarios to facilitate rehabilitation training. The Dual-task Augmented Reality Treatment (DART) platform 18 was designed to simultaneously deliver a proven DTT intervention 6 and objectively measure gait and cognitive performance during training. The DART platform was modeled after our successful randomized clinical trial in which an 8-week therapist-led DTT intervention led to improvements in single- and dual-task gait velocity. 6 The aim of this project was to compare gait and disease-specific outcomes from an 8-week DTT intervention delivered via one-on-one physical therapy compared to the digital delivery of DTT using the DART platform. Considering the DART modules were based on cognitive and motor activities from our previous successful clinical trial, it was hypothesized that the improvements in gait function in the DART group would be non-inferior to the Traditional DTT.
Methods
A single-blind randomized controlled trial was completed. Eligible participants were randomized to the DART or Traditional DTT group. The Traditional DTT group participated in one-on-one sessions with a physical therapist for 8 weeks (16 session); those in the DART group completed that same number of treatment sessions. Outcomes were gathered on 3 occasions: Baseline (BL), end of treatment (EOT), and 8 weeks post-EOT (EOT + 8). Participants were assessed and trained in the “On-medication” state, defined as taking their anti-parkinsonian medication 1 hour prior to the evaluation.
Participants
Participants were recruited from the Center of Neurological Restoration at the Cleveland Clinic. Inclusion criteria were as follows: confirmed diagnosis of idiopathic PD by a physician or physician extender using the UK Brain Bank criteria, 19 Hoehn and Yahr I to III, 20 ability to ambulate at least 10 minutes without the use of an assistive device, and self-reported gait or balance deficits. Exclusion criteria included: surgical procedures for PD including deep brain stimulation or focused ultrasound, a diagnosis of dementia or neurocognitive deficit by a physician or physician extender that would impair the ability to provide consent, a musculoskeletal or cardiopulmonary condition that would restrict walking activity, other neurological disorder(s) such as stroke or multiple sclerosis, and 3 or more errors on the Short Portable Mental Status Questionnaire. 21 All participants completed the informed consent process approved by the Cleveland Clinic Institutional Review Board.
Sample Size Calculation
To evaluate effectiveness between the Traditional DTT and DART groups, a power analysis was conducted using data from our previous therapist-led DTT study,6,22 where the single-task velocity effect was anticipated to be at least 90% of the previous treatment effect, which equated to 0.16 m/s. Results indicated a sample size of N = 17 to 30 in each group was required to achieve a power of 70% to 80% at a significance level of 5% (α = .05; RStudio 2017-power-paired-test) 23 ; therefore, N = 25 for each group was selected to achieve a power of 80%.
Outcome Metrics
All outcomes were gathered by a blinded assessor and blinded technician at BL, EOT, and EOT + 8 weeks timepoints. The following were gathered at each assessment: (1) Movement Disorder Society-Unified Parkinson’s disease Rating Scale motor subscale (MDS-UPDRS III), (2) System Usability Scale (SUS), (3) seated, single-task cognitive assessment, (4) single- and dual-task biomechanical gait analysis.
Movement Disorder Society-Unified Parkinson’s Disease Rating Scale Motor Subscale
The MDS-UPDRS III is a clinician-rated scale of global motor function in PD. 24
System Usability Scale
To determine the usability of the DART platform as an intervention, all participants in the DART group completed the 10-item SUS at EOT. 25 The SUS asks the user to rate the ease of use of a system, system complexity, ease of learning to use the system and confidence in using the system among other general usability characteristics. Scores for the SUS range from 0 to 100; a score of 100 indicates the highest score for usability. A raw score of 68 on the SUS represents a score at the 50th percentile. 26
Single-Task Cognitive Assessment
Consistent with our previous clinical trial methods, 6 single-task cognitive function over a range of cognitive domains was evaluated at each assessment while seated. The following cognitive tasks were randomly administered: N-back (working memory), Serial 7 subtraction (attention), Digit Recall (working memory), Controlled Oral Word Association (COWA, verbal fluency), and visual Stroop test (executive function).
All cognitive tasks were 60 seconds in duration. The N-back (1- and 2-back) asked the participant to recall the letter that was presented 1 or 2 letters previously. 27 For the serial 7 subtraction, participant’s counted backward by 7 for 60 seconds starting at a random number between 300 and 400. 28 Digit recall, a measure of working memory, required the recall of an increasingly complex string of numbers until failure. 29 The COWA test of verbal fluency required the verbalization of as many words as possible beginning with “C, F, or L” (randomized). 30 The visual Stroop test assesses inhibition and executive function through the presentation of a color word printed in an incongruent color ink. 31 The Stroop was displayed on a screen with a new word presented every 1.5 seconds for 60 seconds.
Biomechanical Gait Analysis
The Computer Assisted Rehabilitation Environment (CAREN, Motekforce Link, Amsterdam Netherlands), complete with a three dimensional gait analysis system, was used to quantify gait performance. Participants walked on the CAREN system in the self-paced mode in which treadmill speed was controlled by the anterior-posterior position of the pelvis relative to the center of the treadmill. 32 The position data, sampled at 100 Hz, were gathered from 32 retro-reflective markers placed on anatomic landmarks. 33 Data were filtered using a second order low-pass Butterworth filter with a 6 Hz cut-off frequency.
Gait cycles were defined as heel strike to heel strike of the same foot by using the anterior-posterior heel marker position data. Step length was calculated for each gait cycle and the average for each trial was used for analysis. Cadence and velocity were averaged over the course of the trial.
As part of the informed consent visit, familiarization walking trials with the self-paced treadmill were completed. At each assessment visit, participants were asked to demonstrate the ability to start, stop, and change speeds on the treadmill prior to initiating data collection. The gait assessment at each outcome time point consisted of single- and dual-task conditions. To ensure the participant was comfortable walking on the treadmill, 2 single-task walking trials of 60 seconds were completed during each outcome session; the greater velocity of the 2 trials was used in the subsequent analyses. For the dual-task gait paradigms, the participant walked on the CAREN system while simultaneously performing each of the cognitive assessments completed under single task conditions (1-back, 2-back, Serial 7’s, Digit Recall, COWA Verbal Fluency, and Stroop). Consistent with single-task gait conditions, participants walked for 60 seconds under dual-task conditions. Participants were provided rest between individual trials as needed. Total data collection time was approximately 45 minutes.
Randomization
Following the informed consent process, participants were randomized via a computerized-randomization scheme to either the Traditional DTT or DART group.
Intervention
Traditional DTT
The Traditional DTT training was conducted as in our previous randomized clinical trial. 6 Participants completed 45-minutes of simultaneous gait and cognitive training administered one-on-one by a physical therapist. Interventions were tailored to the participant’s needs, and a high amplitude, high velocity training approach was employed. 34
Cognitive tasks focused on: attention, memory, language, and executive function. 6 For each participant, specific cognitive tasks were selected with respect to performance during the BL testing session dual-task gait assessment. Notably, cognitive tasks completed during training differed from cognitive tasks used for data collection. The complexity of the cognitive task was participant specific based on individual’s cognitive abilities and performance during treatment.
Dual-Task Augmented Reality Treatment
The DART protocol leveraged the hardware and software capabilities of the HL2 to deliver therapeutic protocols that mirror Traditional DTT interventions. An overview of the DART platform is provided in Figure 1; hardware and software have been described in detail in a previous technical publication. 18 Briefly, the DART platform consists of (1) Clinician Dashboard displayed on an Apple iPad (Apple, Cupertino, CA, USA) and (2) DART application delivered via the HL2. The 2 devices communicate via a HIPPA-compliant server.
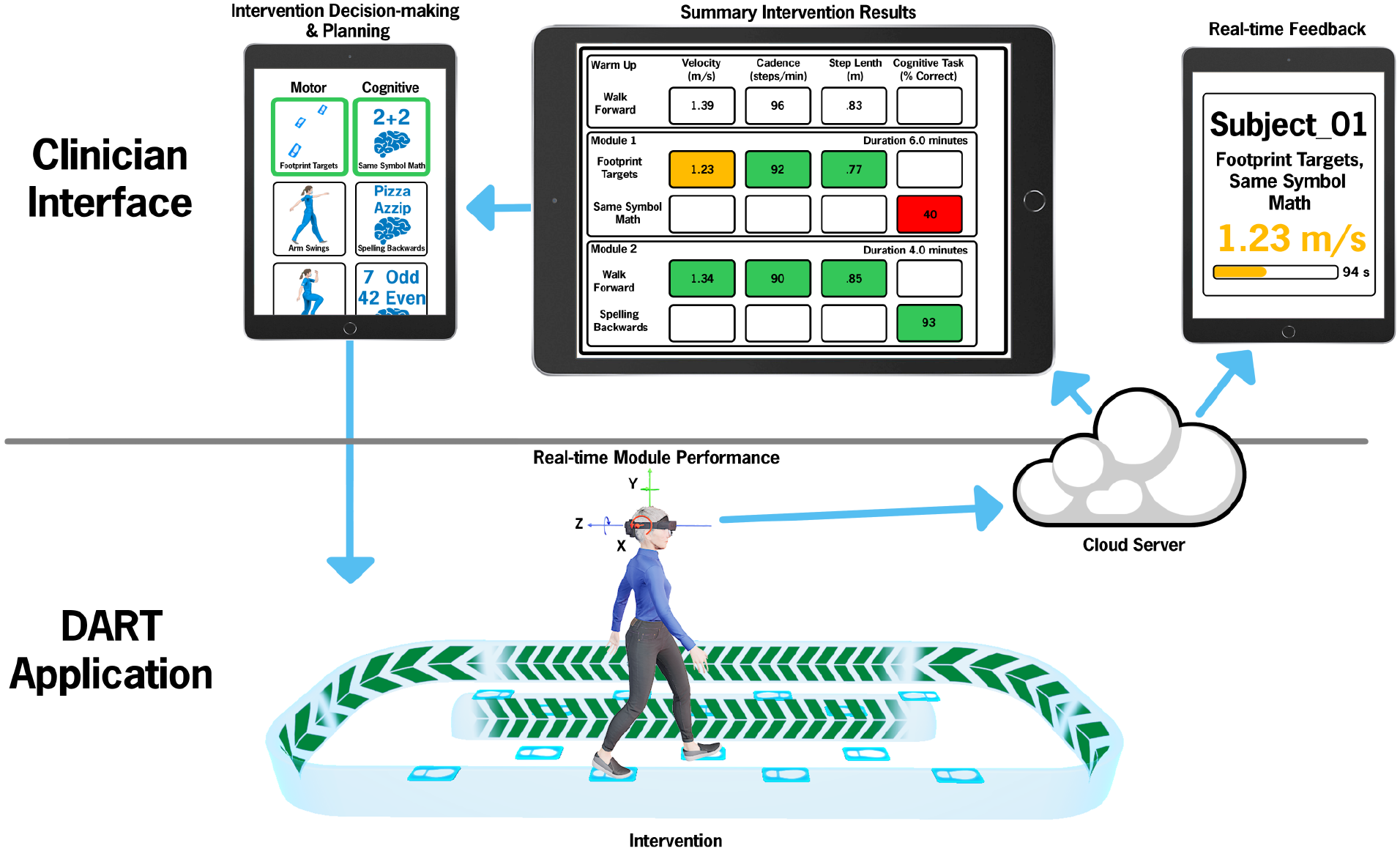
Overview of the DART platform. The Clinician Interface is displayed on a tablet device. The “Intervention Decision-making and Planning” (upper left panel) depicts the therapist creating an individualized therapy session by pairing physical and cognitive tasks to meet the needs of the participant. During the intervention, the pre-programmed intervention is delivered to the participant via the DART application on the HL2. In the “Real-time Module Performance”, the participant is performing the Footprint Targets + an attention task (mathematical questions). The IMU data from the HL2 are sent to the server and used to calculate gait and turning variables. Gait velocity and module progression are provided in real-time to the therapist iPad as “Real-time Feedback”. Following module completion, data from the HL2 are sent to a HIPPA compliant cloud and summary gait and turning metrics are computed and displayed within the “Summary Intervention Results” portion of the Clinician Dashboard (not all metrics are displayed). The therapist uses the summary results to inform the programming of the subsequent session.
Fourteen motor and 17 cognitive tasks were translated into digital modules delivered by the DART platform. The same motor and cognitive tasks available to the Traditional DTT group were digitized and developed for use. Prior to the therapy session, a physical therapist created a participant-specific DART treatment schedule, which took approximately 5 to 7 minutes of the therapist’s time per patient. The session consisted of 8 to 12 dual-task modules in which a motor task was paired with a cognitive task. Within a single module, both the motor and cognitive task could be adjusted for difficulty. For example, in the Footprint Target motor task, depicted in Figure 1, the distance between the footprint targets could be adapted to create a participant-specific step length goal. Similarly, the difficulty of the cognitive tasks was based on capabilities of the participant (ie, spelling 3, 4, or 5+ letter words backwards). The personalized approach to DART content programming enabled the physical therapist to scale motor and cognitive tasks to appropriately challenge the participant based on objective outcomes provided by the HL2. The DART training protocols were programmed for 45-minute session, thus total therapy time across groups was identical.
The DART application was self-administered (ie, the subject progressed through each module at their own pace and could repeat instructions as needed) and semi-supervised (ie, the therapist was in the room with 1 or more participants at a time, but did not intervene or provide guidance during the DART session). The digital DTT content was delivered via a holographic avatar, Donna. Donna was created to guide participants through the specific task requirements and provide visual demonstration of motor tasks. Principles of virtual reality avatar development, 35 were adapted to ensure Donna was relatable and realistic in the delivery of digital DTT. The participant saw the avatar in the lens of the HL2 headset and was presented auditory instructions for the motor and cognitive tasks through the built-in HL2 speakers. The DART platform was interactive as the participant could ask Donna to repeat directions or movement patterns, etc. The therapist monitored the progress of each task in real-time via the Clinician Dashboard. The same physical therapist who delivered Traditional DTT monitored DART sessions to ensure safety of the participant and meet compliance guidelines of the IRB.
Therapeutic Progression
Traditional DTT
Consistent with clinical practice, the physical therapist used their best clinical judgement when grading task difficulty. The physical therapist generally increased the difficulty of the cognitive task when the participant was able to accurately respond to the cognitive task approximately 80% to 85% of the time while maintaining high amplitude, high velocity movements. Consistent with standard clinical practice, the therapist provided the subject with performance feedback (ie, your stride length was longer today, etc.) following the session and areas of continued focus (ie, continue to focus on heel strike, etc.).
Dual-Task Augmented Reality Treatment
Following the completion of a DART session, the physical therapist viewed summary biomechanical performance data on the Clinician Dashboard of the iPad for the purpose of reviewing outcomes with participants and programming the next session to appropriately progress physical and cognitive demands of the task(s). Validated algorithms calculated average gait velocity (m/s), average step length (m), average cadence (steps/minutes), peak turn velocity (°/seconds), average turn velocity (° /seconds), and number of turns16,17 as well as accuracy on discrete cognitive tasks automatically scored through voice recognition software. Gait outcomes were color-coded based on decline from single-task performance: green = 90+% of the single-task performance, yellow = 80% to 89%, and red if <80%. Increments of 10% were modeled from the Timed Up and Go (TUG) subtask of the Mini-Balance Evaluation Systems Test (Mini-BESTest) where a 10% worsening in performance on the TUG cognitive task represented an impairment in dual-tasking abilities. 36 Discrete cognitive tasks were color coded as follows: green = 85% to 100%, yellow = 70% to 84% correct, and red = <70% correct. Figure 2 depicts representative post-session motor and cognitive data from a single module during week 1 of the intervention and illustrates how the data were used to inform module selection and grading. Similar to the Traditional DTT group, the therapist provided performance feedback following the session. The therapist used the objective gait and cognitive metrics provided by the DART platform to provide targeted feedback (ie, the difference in turning velocity under 2 dual-task conditions, etc.).
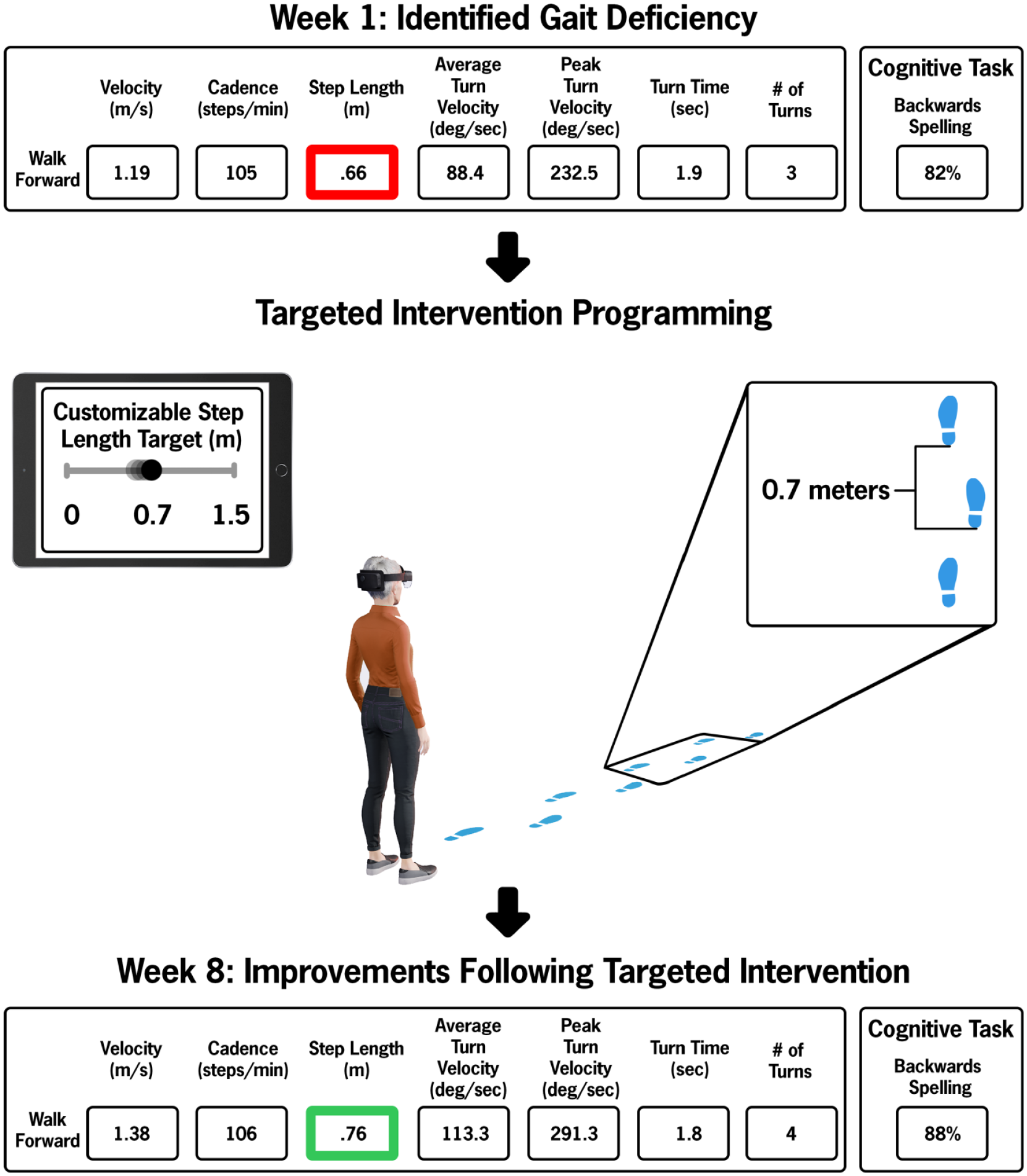
Representative data and subsequent intervention module from the DART application.
Statistical Analysis
The primary analysis was to assess whether improvement was exhibited over time across both treatment groups for outcomes of velocity, cadence, step length, and MDS-UPDRS III. Secondarily, if velocity metrics exhibited significant improvement from BL to EOT, a non-inferiority test was conducted to determine whether the improvement in velocity in the DART group was as good as or better than the improvement in the Traditional DTT group.
Improvement over time was assessed using linear mixed models (LMM) constructed with main effects for time and randomization group, an interaction term between the 2, and random intercept terms for each participant. The LMMs were constructed separately for each dual-task cognitive test and the single-task walk. Outcomes assessed for improvement include velocity, cadence, step length, and MDS-UPDRS III scores. Denominator degrees of freedom were estimated using the Kenward-Roger approximation. If time was found to be significant, pairwise comparisons were then conducted between BL and EOT and also BL and EOT + 8 to assess where differences occurred. If there was a significant group by time interaction effect, pairwise comparisons were conducted separately between each group, otherwise data were collapsed across groups.
For all conditions in which gait velocity exhibited significant improvement from BL to EOT, the improvement between treatment groups was compared using non-inferiority tests. 37 Traditional superiority tests are capable of establishing a difference in treatments, but fail to establish similarity. A non-inferiority test seeks to determine whether a new treatment is as good as or better than an existing treatment, and is constructed using a 1-sided t-test to compare whether the mean improvement of the new treatment (DART) is greater than the mean improvement of the pre-existing treatment (Traditional DTT) minus some noninferiority margin of clinical indifference. The noninferiorty margin is defined as the maximum acceptable difference in treatment effects where the treatment effects could still be considered equal. To ensure that differences between groups were not meaningful, the inferiority margin was set using a previously reported minimal clinically important difference (MCID) of 0.06 m/s. 38 The non-inferiority tests were conducted for gait velocity only, as cadence and step length do not have established MCID values.
The significance of all statistical tests was conducted using a .05 level of significance. Pairwise comparisons from the LMMS were Bonferroni corrected within each outcome to maintain a per outcome family-wise type I error rate of 5%. All statistical analyses were conducted using RStudio 2023.03.0, R version 4.2.3.
Results
The consort diagram, Figure 3, summarizes study and participant flow. Fifty-one participants were randomized. Four participants withdrew from the study, and 47 participants participated in BL and EOT assessments and were ultimately included in the data analysis. Demographics are provided in Table 1. There were no meaningful differences between the groups.
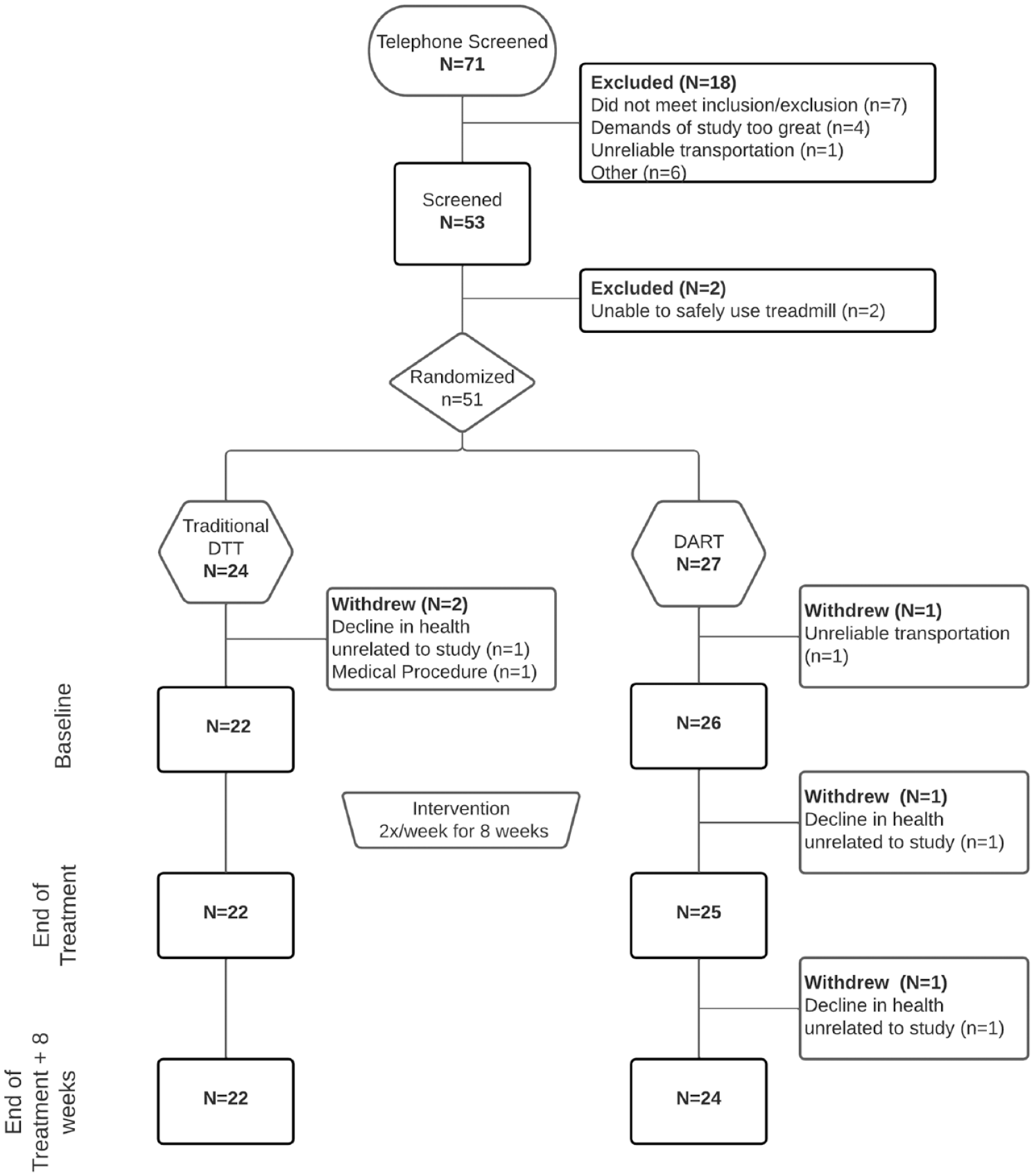
Study consort diagram.
Participant Demographics.
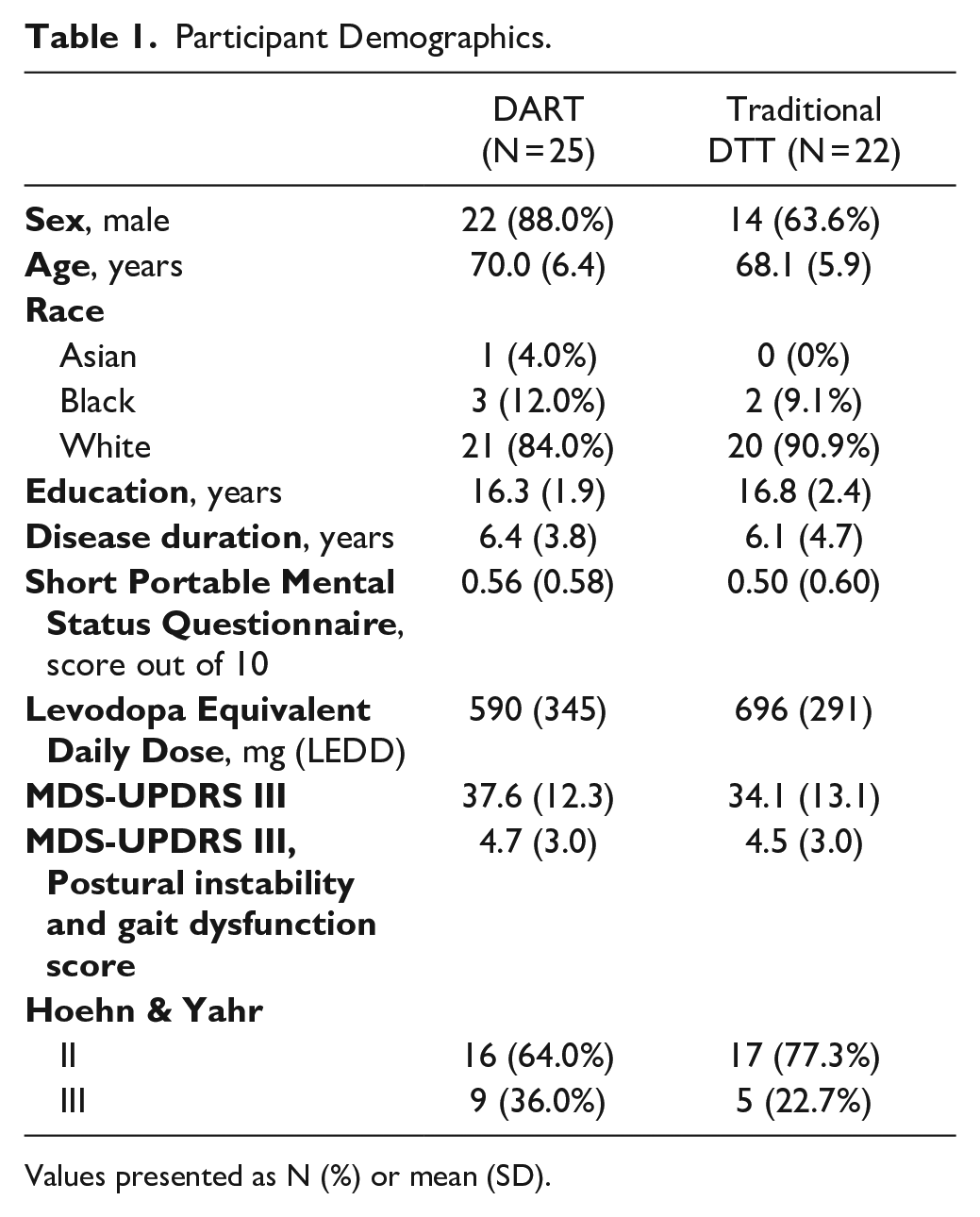
Values presented as N (%) or mean (SD).
Subject Retention, Adherence, and Adverse Events
Over the course of the 8-week intervention, our retention rate was 92% (47 participants finished the 8-week intervention/51 were consented). The withdraws were evenly distributed between groups (N = 2 DTT; N = 2 DART). One additional participant withdrew during the 8-week follow up in the DART group. The reasons for the withdraws were unrelated to the study. In the 47 individuals who completed the study, they completed 100% of the intervention sessions.
One DART participant fell without injury during training. He continued the session and participation in subsequent sessions was not impacted.
Movement Disorder Society-Unified Parkinson’s Disease Rating Scale Motor Subscale
Mean (SD) MDS-UPDRS III scores in the Traditional DTT group were 34.1 (13.1), 34.9 (11.4), and 36.4 (11.3) at BL, EOT, and EOT + 8, respectively. Mean (SD) MDS-UPDRS III scores in the DART group were 37.6 (12.3), 36.6 (12.8), and 38.0 (11.5) at the 3 assessment time points. There was no significant main effect of time, group, or group by time interaction.
Gait Variables From CAREN Testing
All biomechanical gait variables gathered during single- and dual-task CAREN testing are provided in Table 2.
Biomechanical Gait Outcomes by Group and Time.
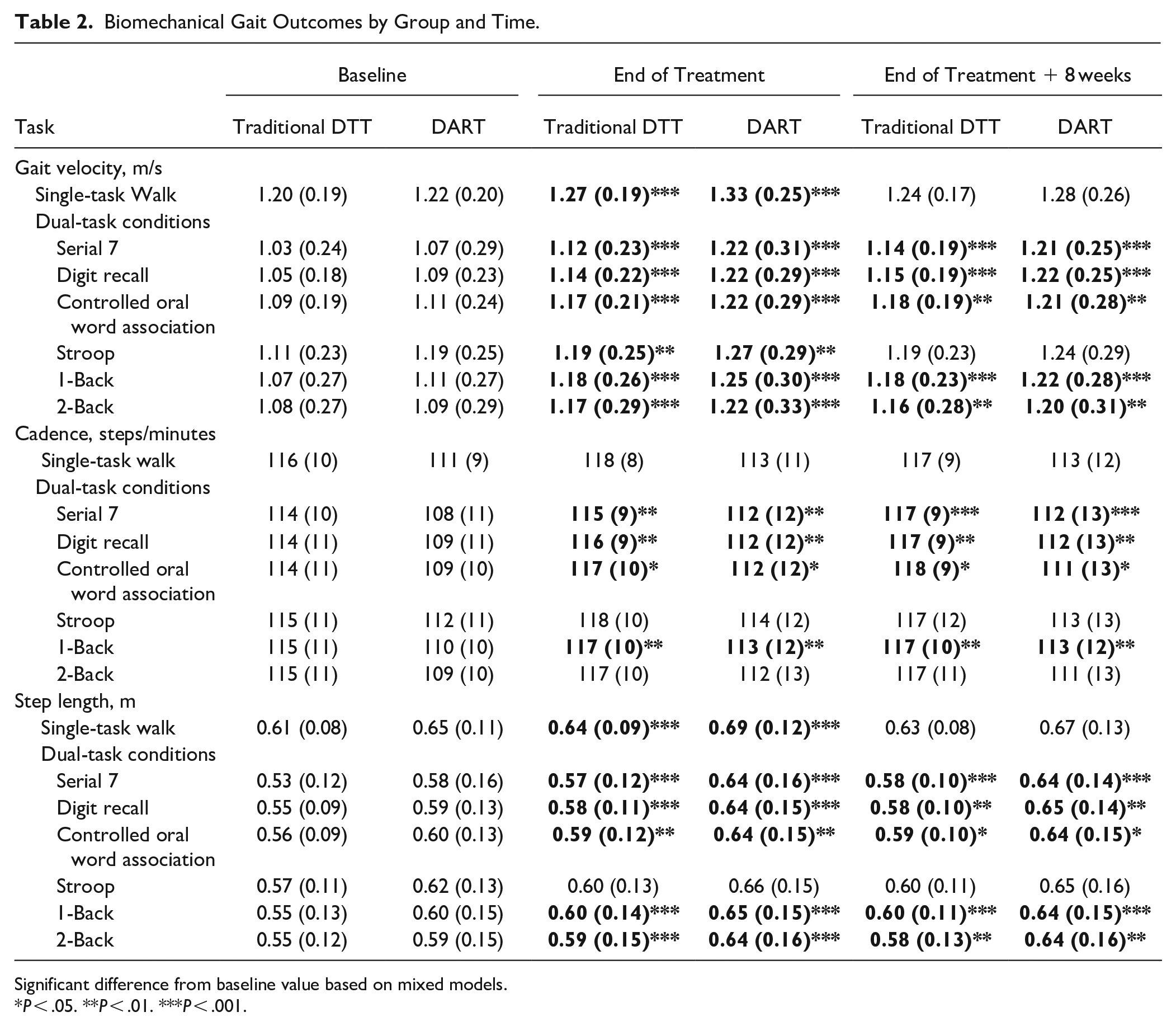
Significant difference from baseline value based on mixed models.
P < .05. **P < .01. ***P < .001.
Gait Velocity
Across all dual-task walks and the single-task walk, gait velocity significantly improved from BL to EOT (P < .01). The improvement was largely maintained at EOT+8; significance from BL to EOT + 8 was achieved (P < .01) in all but 2 tasks (single-task walk, P = .15, Stroop, P = .22). Figure 4 displays data from both groups during the Serial 7 walking. There were no significant interaction effects for gait velocity.
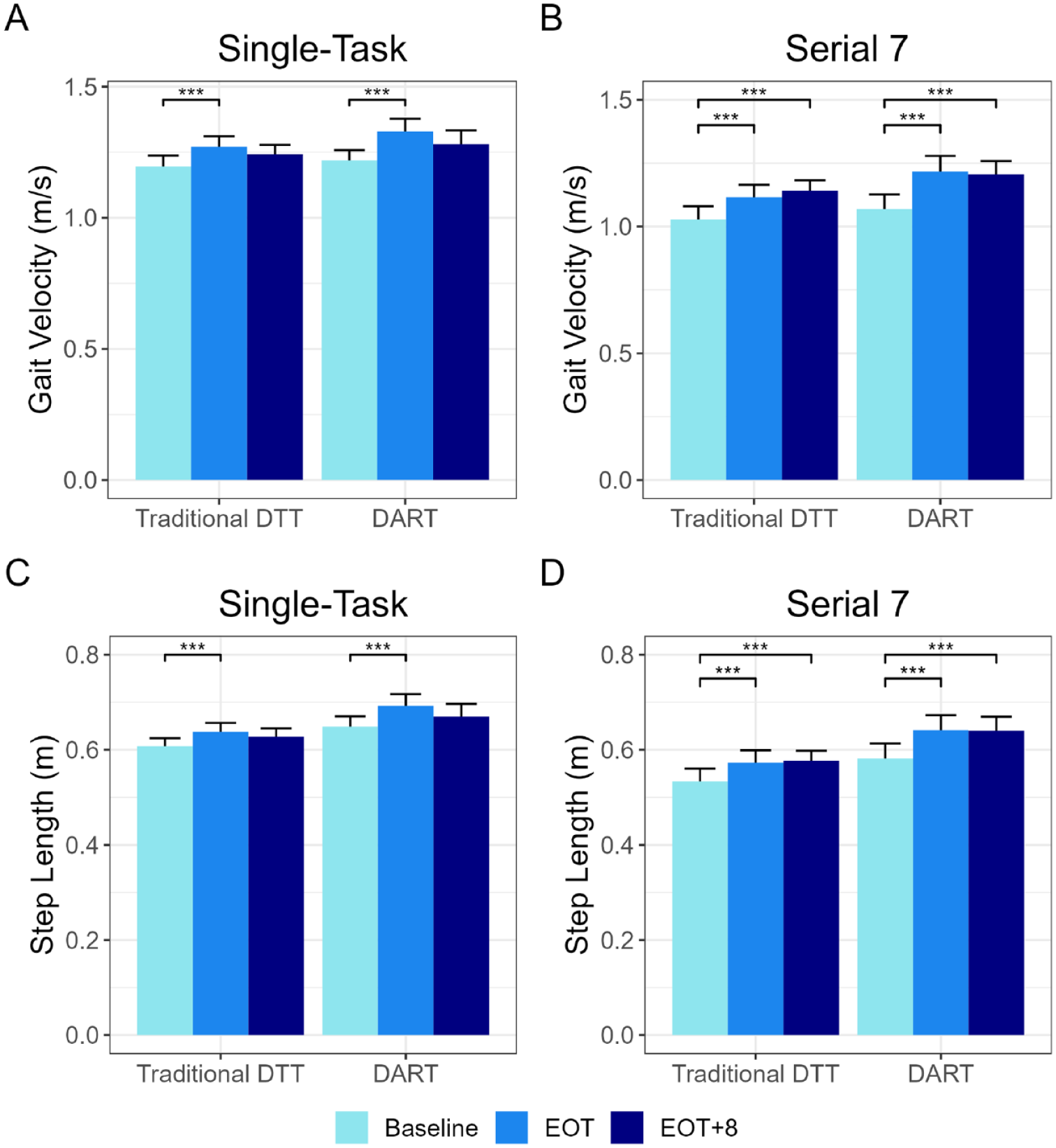
Mean and standard error of participant outcomes by group for (A) Single-task gait velocity, (B) Dual-task Serial 7 gait velocity, (C) Single-task gait step length, (D) Dual-task Serial 7 step length. A (***) denotes a significant improvement from BL (P < .001). During single-task conditions, gait velocity and step length significantly improved from BL to EOT in both groups. During the Serial 7 task, gait velocity and step length significantly improved from BL to EOT and BL to EOT + 8 in both groups.
When assessing non-inferiority, the mean improvement in the DART group always numerically exceeded the improvement in the Traditional DTT group. Non-inferiority testing revealed that the DART group was non-inferior to the Traditional DTT group for all tasks (P < .05) except 1-Back and Stroop (P = .053 and .08, respectively). Supplemental Table 1 provides details of the analysis.
Cadence
Across all tasks, mean cadence values increased from BL to EOT and EOT + 8 and reached statistical significance on the 1-Back, Digit Recall, Serial 7’s, and Controlled Oral Word Association tasks at EOT (P < .05 for all) and EOT + 8 (P < .05 for all). There were no significant interaction effects for cadence.
Step Length
Across all tasks, participant mean step length values increased from BL to EOT and EOT + 8. At EOT, improvement reached statistical significance in all but 1 of the dual-task conditions (P < .01 for all except Stroop, P = .06) and significance was largely maintained at EOT + 8 (P < .05 for all except single-task walk, P = .18). There were no significant interaction effects for step length.
Single-Task and Dual-Task Cognitive Performance
Cognitive performance was stable for both groups following the 8-week intervention. However, from BL to EOT during single-task conditions, performance of the Serial 7 task, number of correct responses over 60 seconds, increased from 10.1 to 12.4 responses (22.9%) and 10.5 to 12.0 responses (14.3%) for the DART and Traditional DTT groups, respectively, (P = .002).
Usability of the DART Platform
Participants in the DART group (n = 25) evaluated the usability of the platform as a treatment intervention for PD using the SUS. Mean score for the SUS was 82.7 (9.04).
Discussion
Utilizing the DART platform or engaging in Traditional DTT with a physical therapist resulted in significant improvements in gait speed and gait kinematics, but no change in the clinical MDS-UPDRS III. Non-inferiority in gait velocity across both groups provides support for the DART platform to be considered a DTx for the treatment of gait impairments associated with PD. To our knowledge, this is the first DTx for PD that has undergone the rigorous evaluation relative to an accepted intervention using a randomized controlled trial (RCT) design.14,15
While the interventions resulted in a statistical improvement in gait speed, cadence, and step length, it is important to examine the clinical relevance of these improvements. Improvement in single- and dual-task gait speed performance from BL to EOT ranged from 0.08 to 0.12 m/s and from 0.09 to 0.15 m/s in the Traditional DTT and DART groups, respectively. In every condition, improvements in gait speed exceeded the small (0.06 m/s) to moderate (0.14 m/s) clinically important differences in gait speed for PwPD, 38 suggesting that the improvement in gait speed was statistically improved and, importantly, clinically meaningful. Furthermore, prior to training, mean gait speed was below 1.1 m/s in 5 out of the 6 dual-task conditions, which is relevant since gait speed below 1.1 m/s is predictive of falls in community-dwelling PwPD. 39 Following the 8-week training, mean gait velocity under single- and dual-task conditions increased to >1.1 m/s under all dual-task conditions and remained above the cut-off value at the 8-week follow-up, representing a decreased risk of falls in this cohort. Thus, the clinical implication of improvements in gait speed under both single- and dual-task conditions exceeding the PD-specific clinically important difference and 1.1 m/s fall cut-off value suggest that our findings have meaningful implications in those with PD as many daily gait activities could be considered dual-task conditions.
Despite support for the use of DTT to address PD related gait impairments, 40 it has not been widely implemented clinically.41,42 A recent report from Domingos et al 12 sought to identify clinical barriers to the adoption of DTT. Primary difficulties preventing adoption included patient and training dynamics (eg, poor compliance and motivation, lack of tailored interventions and poor motivation). 12 One of the key barriers summarized by Domingos and colleagues was the tailoring of patient-specific interventions and keeping the interventions challenging and engaging: “Every Parkinson’s patient is different, we can’t generalize. . .” 12 It is contended that a DTx such as the DART platform is suited to create, deliver and monitor patient-specific DTT interventions by providing a platform that guides the creation and tailoring of an individualized intervention. Currently, the DART platform provides 238 combinations of dual-tasks that can be used to tailor treatment to individual patient needs and preference. Within the 238 combinations, motor and cognitive tasks can be graded (ie, the number and type of obstacles in the obstacle course and the difficulty of the math problems) for an expansive number of module options. Another challenge of tailoring the intervention is appropriate exercise progression. Clinically, exercise progression during Traditional DTT is primarily based on clinical expertise and subjective observation. In contrast, the color-coded feedback from the DART platform provides a guideline to progression by providing objective feedback and dual-task interference with the calculation of dual-task loss. While it does take 5 to 7 minutes to program the DART platform for a single participant, arguably this time is gained during the treatment session because there are no periods of inefficiencies, such as the therapist physically arranging an obstacle course or foot print targets, and there are no periods of inactivity during transition to the next task.
Another training issue noted by Dominogos was poor compliance and motivation. 12 Our data indicate that retention and adherence were high with use of the DART platform, as our withdraw rate was less than 10%, which is comparable or superior to similar therapeutic interventional studies of similar duration.43,44 Additionally, of the 47 individuals who completed the study, both the DART and Traditional DTT groups had an attendance rate of 100% and for those in the DART group, they endorsed a high rate of usability and satisfaction with the DART platform. In terms of usability, PwPD using the DART platform group scored its usability as “good-to-excellent.” 45 This places the DART platform’s usability as a treatment tool in the 90 to 95th percentile. 26 These feasibility metrics indicate that the issue of compliance and motivation can be addressed through the use of a well-designed technology platform.
Barriers to DTT adoption, including accessibility, provided the initial rationale to develop and rigorously evaluate the DART platform as a DTx. Recently, the W.H.A.T. Digital Therapeutic Validation Criteria rubric was introduced as a means to identifying a DTx.
46
Briefly, the designation of DTx should be evaluated by determining if it:
Our initial usability data, 18 and others, 49 support the feasibility of using head-mounted AR devices in PwPD. Individuals with PD typically experience no motion sickness, find the device relatively comfortable, 49 and report positive usability scores following single bouts of activity. 18 However, preliminary efficacy of head-mounted AR devices in treating PD symptoms has been mixed.49 -52 A plausible reason for the mixed results is that previous studies deliver the intervention (predominately external cues to alleviate gait dysfunction) over a single session. It is difficult to envision a single-session could alter behavior and central nervous system plasticity in those with a neurological disease such as PD. Altering CNS function in those with neurological disease generally requires a longer intervention period that consists of a salient, challenging, repetitious environment. 53 The DART intervention was built upon neuroplastic principles and modeled after a successful DTT clinical trial.6,22 The cognitive and motor training modules were heavily informed by the tasks used in our earlier DTT trial. During the digitization of these training modules, attention was paid to creating tasks that were relevant, understandable, and demonstrable by a digital avatar and could be scaled in terms of difficulty over the course of the intervention. These considerations, in particular the latter, were likely critical in maintaining the interest and appropriately challenging the participants completing DART training. Therefore, the resultant DART training replicated, by using more objective methods and outcomes, the personalized nature of one-on-one DTT through the creation of engaging and relevant therapy exercises.
Currently, DTx’s are regulated by the US Food and Drug Administration (FDA) and classified into the category of Software as a Medical Device, hence, the software itself, rather than the hardware it is deployed on, is a medical device. To our knowledge, the DART platform is the first PD gait focused DTx to undergo and pass the rigorous scientific validation that has been recommended for the evaluation of DTx. A FDA submission is currently in preparation. The cost of a HL2 system is approximately $3500 (at the time of this study), which is a comparable cost to other rehabilitation equipment, such as a treadmill or stationary bicycle and makes procurement and use feasible for most rehabilitation centers. Notably, the DART platform will not take the place of a physical therapist. Rather, the DART platform will serve as enabling technology in which a single therapist may be able to deliver DTT to and monitor multiple PwPD performing patient specific therapy designed to improve PD related gait dysfunction.
Conclusion
Augmented Reality training is safe and effective for PwPD. Employing a technology development approach that targeted those with a neurological disease as the end-user resulted in high usability scores and an overall improvement in gait under single-and dual-task conditions. As the field of DTx is rapidly evolving, it is imperative that rigorous validation principles, such as those noted in the W.H.A.T. rubric, are employed. The DART platform is an enabling technology that addresses barriers to implementing DTT and the process employed to create and validate the DART platform should be considered a model for the development of future DTx aimed at countering the effects of PD. The current study population, while having gait dysfunction, was able to perform the majority of the digitally and physically prescribed motor tasks. Future iterations of DART should consider adaptations to address those more severely affected patients. Finally, a major value proposition of a DTx is the potential to bring an effective intervention to the patient, a future project is planned to evaluate the safety and effectiveness of PwPD using DART in a home environment.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231184190 – Supplemental material for A Randomized Clinical Trial to Evaluate a Digital Therapeutic to Enhance Gait Function in Individuals With Parkinson’s Disease
Supplemental material, sj-docx-1-nnr-10.1177_15459683231184190 for A Randomized Clinical Trial to Evaluate a Digital Therapeutic to Enhance Gait Function in Individuals With Parkinson’s Disease by Jay L. Alberts, Ryan D. Kaya, Amanda L. Penko, Matthew Streicher, Eric M. Zimmerman, Sara Davidson, Benjamin L. Walter and Anson B. Rosenfeldt in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Kathryn Scelina, Mike Buss, and Logan Scelina for their assistance with technology development and quality assurance testing.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Author Contributions
A.B.R. and J.L.A. designed the study. A.B.R. and R.D.K. developed and delivered the intervention. A.L.P. and M.S. performed data collection. E.M.Z. performed the statistical analysis. A.B.R. and J.L.A. wrote the first draft of the manuscript. All authors (A.B.R., R.D.K., A.L.P., M.S.., E.M.Z., B.L.W., and J.L.A.) reviewed, contributed in manuscript preparation, and approved the final version. All authors agree on the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.L.A. and A.B.R. have authored intellectual property associated with the DART platform.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Michael J. Fox Foundation for Parkinson’s Research (grant number 18159) and the Edward and Barbara Bell Family Endowed Chair.]
Data Availability
Correspondence and request for materials should be addressed to Jay L. Alberts.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
