Abstract
Introduction
The ability to safely ambulate in everyday environments is compromised in patients with Parkinson’s disease (PD). This leads to an increased risk of falls, disability, and reduced independence. Safe ambulation is affected by the complexity of task and the person’s abilities.1-3 For example, walking in complex environments involves avoidance of static and dynamic obstacles. This requires the utilization of motor resources and cognitive resources such as divided attention and visual scanning.4-7
The ability to walk while carrying out another task, that is, dual task (DT) walking, relies on executive function; a set of cognitive skills that are needed for planning, monitoring and executing a sequence of goal-directed complex actions. 8 DT represents a cognitive challenge since it demands the allocation of attentional resources to concomitant tasks.9-11 Deficits in executive function have been reported in patients with PD12-15 and have been linked to difficulties in walking while dual tasking.16,17 In patients with PD, the negative effects of a DT on gait properties such as gait speed and stride-to-stride variability and on the cognitive task are larger than that seen in age-matched controls.18-21
Obstacle crossing is a daily activity that involves tasks such as going up a curb or stepping over a crack in the ground. Motor planning, attention to relevant stimuli, and visual-spatial abilities are essential to successfully negotiate obstacles.4,22,23 Attention and visual-spatial deficits are common in patients with PD and manifest as difficulty to negotiate obstacles and diverse terrains. This may explain why patients with PD approach and step over obstacles more slowly and with smaller and wider steps than healthy older adults.4,7,24
The neural control of DT walking and obstacle negotiation is mediated by various regions in the brain, among them the frontal lobe. The frontal cortex, and specifically the prefrontal cortex and its related circuits, are involved in dual tasking and have been implicated in planning, organization, execution, and adjustment to the environment of walking.25,26 Recently, several studies used functional near infrared spectroscopy (fNIRS) to examine frontal lobe activity during walking and reported increase oxygenated hemoglobin (HbO2) levels during walking while DT, compared to usual walking in healthy young and older adults.27-29 These findings provide direct evidence that DT walking is associated with frontal activation. Although deficits in executive function are common among patients with PD,12,30,31 accumulating evidence suggests that patients utilize the frontal lobe, mainly regions associated with cognitive resources, to compensate for striatal impairments.32-34
To date, however, all studies used DT paradigms that involved the performance of a cognitive task during walking. Changes in frontal activation during obstacle negotiating have not been examined. In addition, most of the fNIRS studies that investigated DT walking included healthy young and older adults. Cognitive compensation in patients with PD32-34 suggest higher activation of the frontal lobe during different walking conditions, however, direct association between frontal activation and walking in patients with PD are lacking. Therefore, the aims of the present study were (
Methods
Participants
Thirty-eight healthy older adults (20 men and 18 women) and 68 patients with PD (46 men and 22 women) participated in this study. General inclusion criteria were (
Procedures
The protocol included 2 parts: (
Gait and frontal activation were assessed during 3 walking tasks: (
Functional Near Infrared Spectroscopy
Changes in oxygenated hemoglobin (HbO2) and deoxygenated hemoglobin (HHb) concentrations in the prefrontal cortex were measured with the PortaLite fNIRS system (Artinis Medical Systems, Elst, the Netherlands). The system uses near infrared light, which is transmitted at 2 wavelengths, 760 and 850 nm. Data were sampled with a frequency of 10 Hz. The PortaLite uses wireless technology (Bluetooth), allowing participants to walk without the restriction of wires. Two probes were placed on the right and left forehead of the participants. Probes were positioned at a height of 15% of the nasion-inion distance from nasion and at 7% of the head circumference to the left and right from midline, to avoid measuring the midline sinus. These locations roughly target left and right Brodmann’s areas 10, the dorsolateral and anterior prefrontal cortex (PFC).41,42 The probes were shielded from ambient light by covering the forehead with black fabric. Oxysoft version 3.0.52 (Artinis Medical Systems, Elst, the Netherlands) was used for data collection.
Based on different absorption spectra, concentration changes of HbO2 and HHb in the targeted PFC were calculated from the changes in detected light intensity using the modified Lambert-Beer law, assuming constant scattering.
43
The PortaLite has 3 transmitters and 1 receiver, with transmitter-receiver distances of 30, 35, and 40 mm. The concentrations of HbO2 and HHb were exported to MATLAB (MATLAB and Statistics Toolbox Release 2012b, The MathWorks, Inc, Natick, MA) for further data processing. A bandpass filter with frequencies of 0.01 to 0.14 Hz was used to reduce physiological noise such as heart beat and drift of the signal. To remove motion artifacts, a wavelet filter was used,44,45 followed by correlation based signal improvement (CBSI).44-46 HbO2 concentration signals of the three channels of each probe were then averaged, resulting in an HbO2 signal for the left and right PFC. For each trial, the average concentration of HbO2 during task performance, and during the 5 seconds before the task (referred to as the baseline) were calculated. Consistent with previous studies of fNIRS signals, each baseline concentration was subtracted from the average concentration during task performance to evaluate the relative change in HbO2 concentration during specific tasks.11,27,29,47-49 All trials were averaged per task, resulting in 2 HbO2 concentrations for each task (left and right PFC). Since no left-right differences were present (
Behavioral Task Performance
Gait was measured using electronic walkway with pressure sensors embedded in a carpet. The walkways were connected to a personal computer using PKMAS software (ProtoKinetics, Havertown, PA) for processing and data storing. In order to characterize the performance of usual and DT walking, 2 key measurements of walking, gait speed and stride length, were analyzed and presented. 50 For obstacle negotiation, gait speed and the duration of stepping over the obstacle for each step (in seconds) were obtained. We also calculated the percentage change in step duration (seconds) between steps over the obstacles and normal steps during the obstacle walk. Serial subtraction performance was measured by calculating the percent of correct responses from the total responses for each trial. DT cost was calculated as ([dual task − single task)/single task] × 100) for both gait measures.
Statistical Analyses
Means and standard errors were calculated for all dependent variables. Differences in levels of HbO2 between groups (ie, healthy older adults and patients with PD) and tasks (ie, usual walk, DT, and obstacle negotiation) were analyzed using a linear mixed model, including the interaction between group and task. We also included the factors age, gender, gait speed, and executive function index to control for group differences. Linear mixed models with the factors age and gender were used to analyze group and task differences in the behavioral performance outcome measures. For linear mixed models on HbO2 concentrations, random intercepts were allowed between participants since it improved model estimations. For all models, a covariance matrix was set for variance components. Post hoc test
Results
Participants
Participant characteristics are shown in Table 1. No differences in age and gender were observed between groups. Patients with PD had lower scores on all cognitive measures including the Mini-Mental State Examination and computerized cognitive tests, compared with the healthy older adults. As expected, patients with PD performed worse on tests of mobility.
Participant Characteristics. a
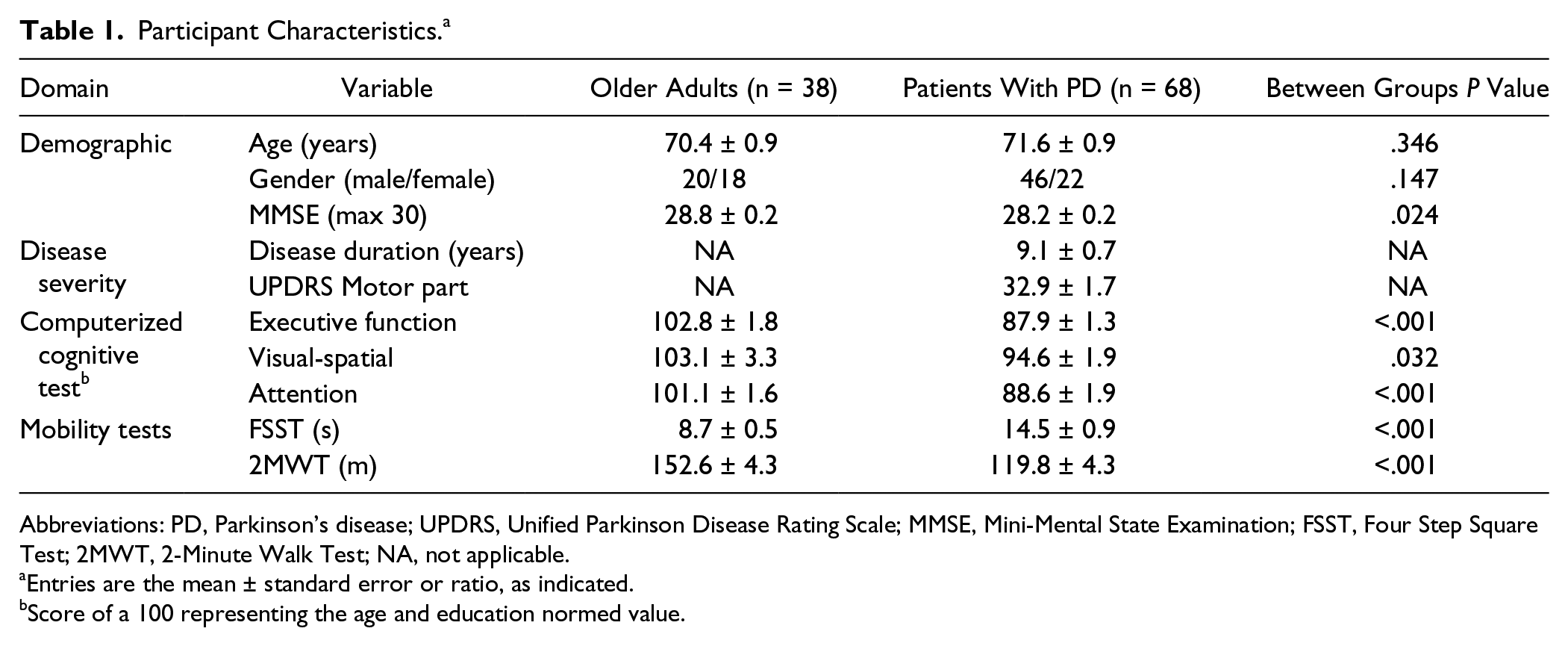
Abbreviations: PD, Parkinson’s disease; UPDRS, Unified Parkinson Disease Rating Scale; MMSE, Mini-Mental State Examination; FSST, Four Step Square Test; 2MWT, 2-Minute Walk Test; NA, not applicable.
Entries are the mean ± standard error or ratio, as indicated.
Score of a 100 representing the age and education normed value.
Changes in Frontal Lobe Activation
Significant differences in HbO2 levels between walking conditions were found (
Oxygenated Hemoglobin (HbO2) Concentrations During the Different Walking Tasks. a
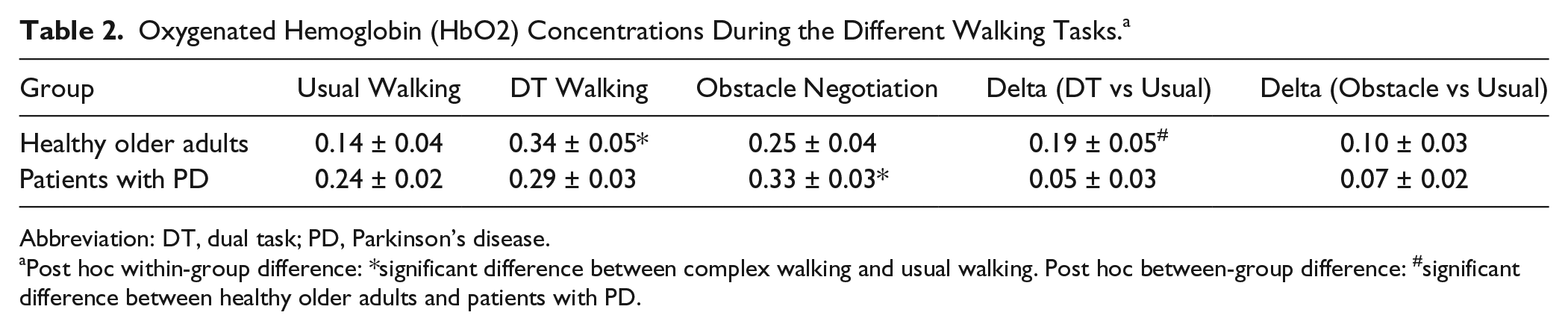
Abbreviation: DT, dual task; PD, Parkinson’s disease.
Post hoc within-group difference: *significant difference between complex walking and usual walking. Post hoc between-group difference: #significant difference between healthy older adults and patients with PD.
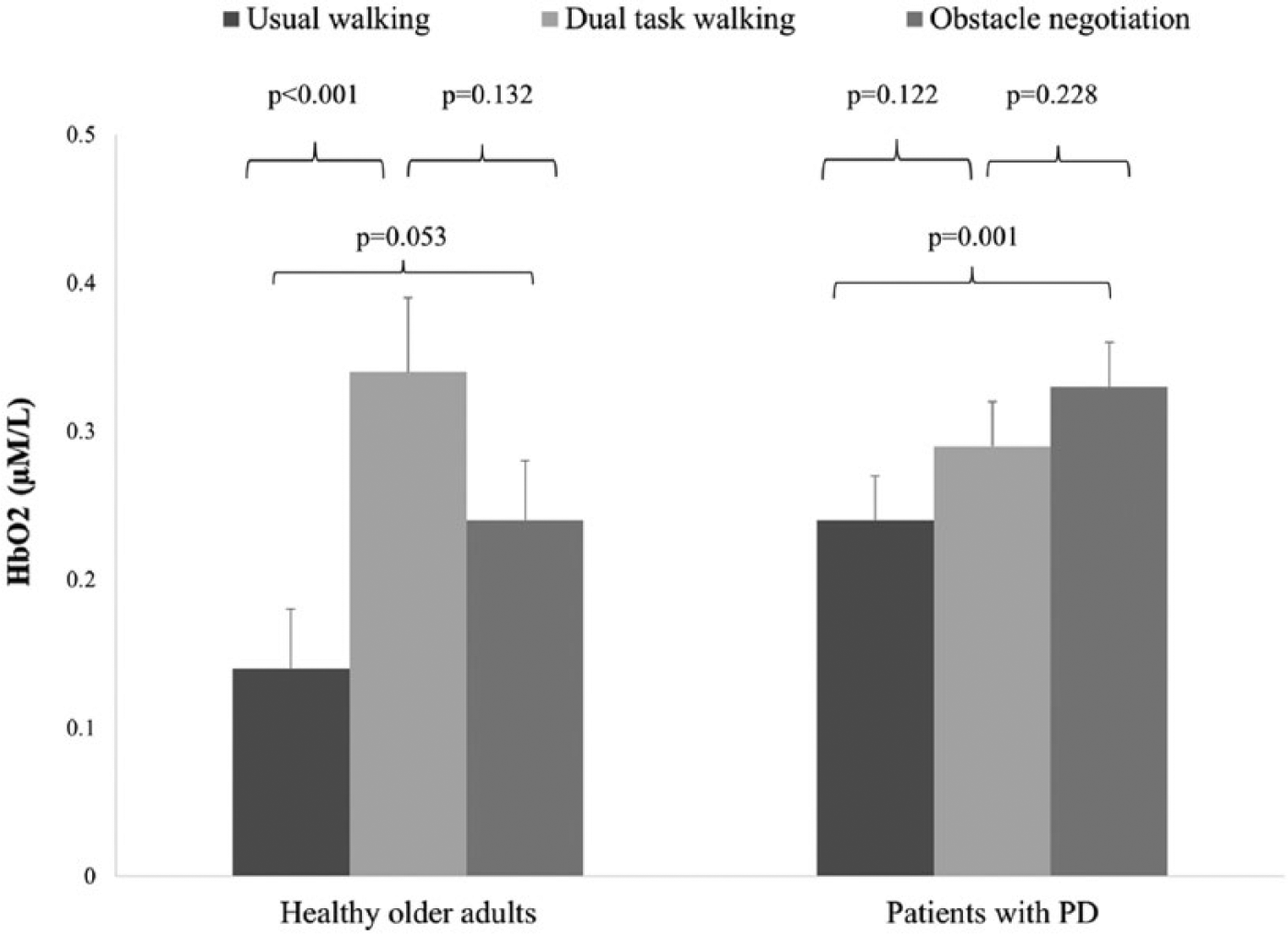
Oxygenated hemoglobin (HbO2) levels during the 3 walking conditions in each group. A significant difference between usual walking and dual task walking was observed in the healthy older adults. In contrast, in the patients with Parkinson’s disease (PD), the HbO2 level was significantly higher during obstacle negotiation walking, compared with usual walking.
Between-group comparisons for each condition separately revealed a significantly larger increase in HbO2 level during usual walking in the patients with PD, compared with the older adults (
Gender significantly influenced HbO2 levels, with men having higher HbO2 levels than women in all tasks (
Behavioral Results
Table 3 shows between-group comparisons of functional performance on the 3 walking conditions. As expected, patients with PD performed worse than the healthy older adults in all 3 walking conditions. The stride length was shorter, gait speed was lower, and percentage of correct subtractions was lower in the patients with PD. Dual task costs for gait were significantly higher in the patients with PD than in the older adults. Similarly, the effect of obstacle negotiation on gait speed was higher in the patients with PD than in the older adults.
Differences in Performance of Walking Conditions Between Groups.
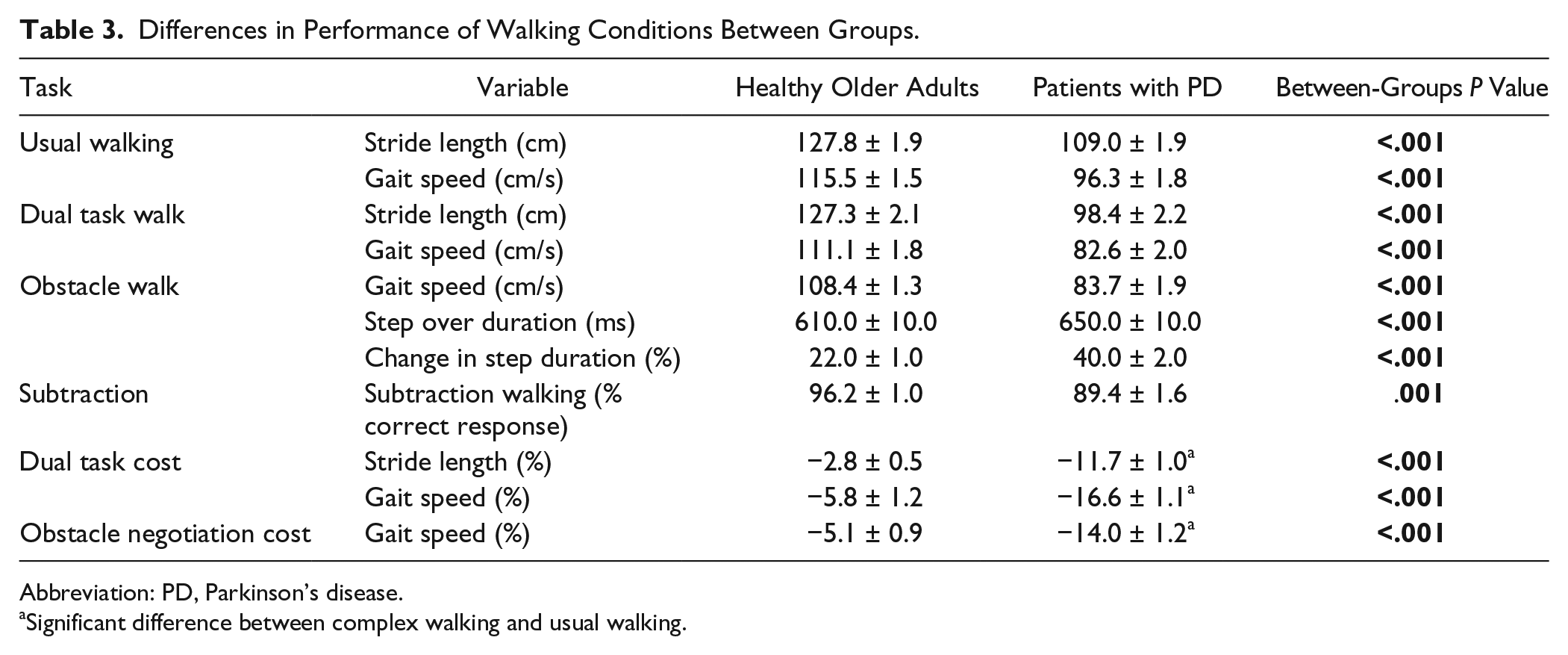
Abbreviation: PD, Parkinson’s disease.
Significant difference between complex walking and usual walking.
Associations Between HbO2 Levels and Behavioral Measures
No significant correlations between HbO2 levels during walking conditions and gait speed were found in the healthy older adults. In contrast, in patients with PD, a positive correlation was found between level of HbO2 and gait speed during obstacle negotiation walking (
Comparison between delta HbO2 of DT and usual walking (change in frontal activation) and dual task cost for gait speed (change in behavioral measure) revealed different patterns in healthy older adults and patients with PD. In the healthy older adults, a large increase in HbO2 during DT, compared with usual walking coincided with low dual task cost for gait speed. In the patients with PD, small increase in HbO2 of DT, compared with usual walking, coincided with high DT cost for gait speed (Figure 2A). On the other hand, comparison between delta HbO2 of obstacle negotiation and usual walking (change in frontal activation) and obstacle negotiation cost for gait speed (change in behavioral measure) revealed similar change in delta HbO2 in both groups but differences in obstacle negotiation costs between groups. While healthy older adults had low obstacle negotiation cost for gait speed, patients with PD had a high obstacle negotiation cost for gait speed (Figure 2B). No significant correlations between HbO2 levels and mobility tests were found.
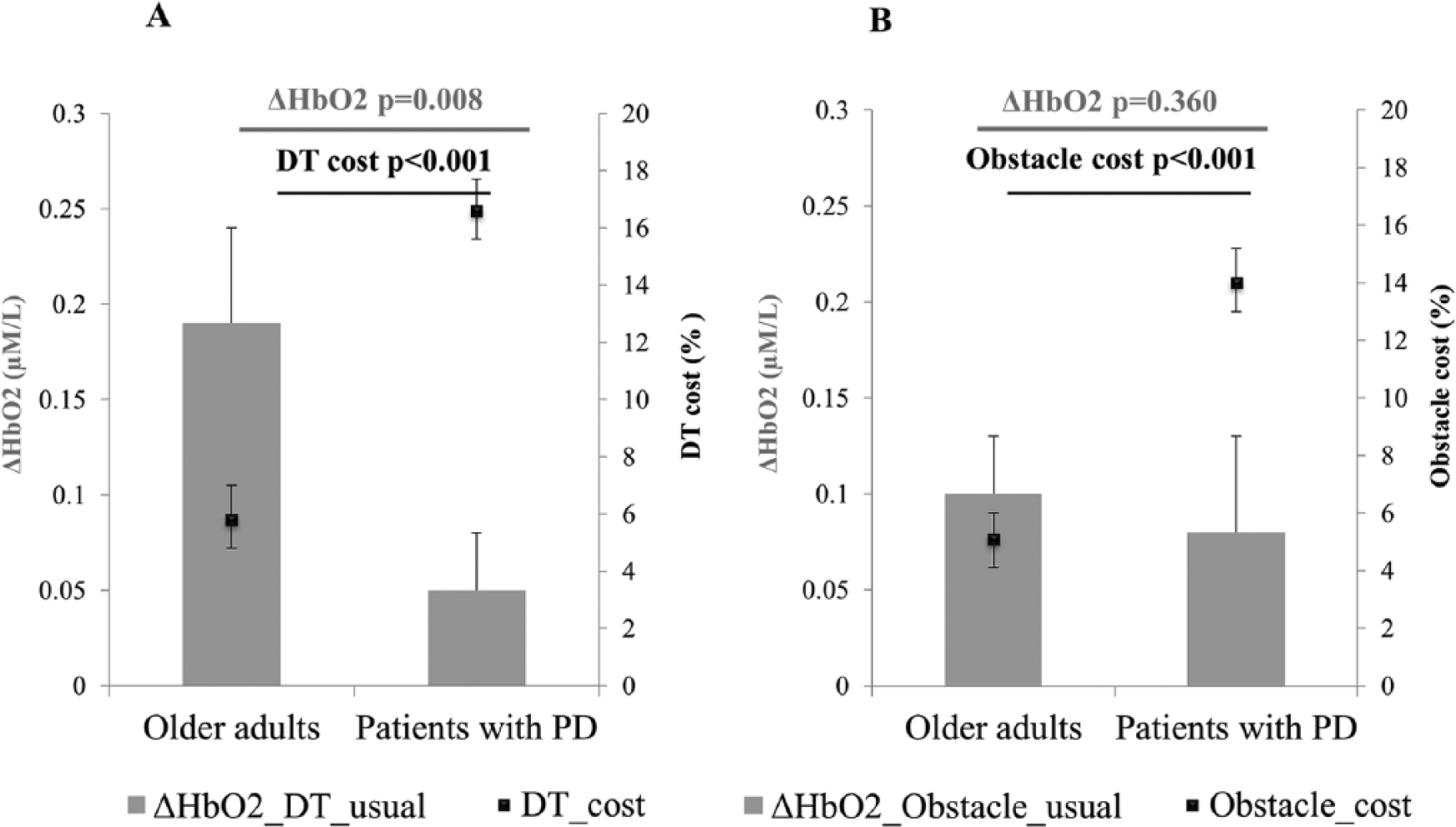
Comparison between relative increase (delta) in oxygenated hemoglobin (HbO2) during walking conditions (gray bars) and behavioral measures of the absolute % cost (black squares) in healthy older adults and patients with Parkinson’s disease (PD). (A) Delta HbO2 of dual task (DT) and usual walking and dual task cost for gait speed. (B) Delta HbO2 of obstacle negotiation and usual walking and obstacle negotiation cost for gait speed.
Discussion
Our study is, to our knowledge, the first to report that prefrontal activation during usual walking and complex walking change in patients with PD. In contrast to healthy older adults, patients with PD showed a significant increase in prefrontal activation during obstacle negotiation and only a slight increase in activation during dual task walking, as compared with usual walking. However, during usual walking, higher HbO2 levels were observed in patients with PD as compared with healthy older adults.
The higher level of HbO2 in Brodmann area (BA) 10, a region that reflects the dorsolateral prefrontal cortex (DLPFC), during usual walking in patients with PD may reflect the need for utilization of cognitive resources even in this relatively “simple” task. The association between cognitive abilities and BA 10 activation is supported by the finding that after adjusting for the executive function score the group difference in the change in HbO2 values is no longer significant. The use of cognitive resources to compensate for the deficits associated with PD stands in line with a recently suggested model.32-34 This model proposes that during walking, patients with PD may attempt to compensate for deficits in automaticity by switching to increased cognitive control, in particular executive control. 34 However, deficits in executive control limit this ability. Therefore, during more challenging walking conditions, the system may not be able to adequately respond, producing marked behavioral changes and in some cases, falls. The present results in the patients with PD are, to some degree, consistent with this model.
The high activation during usual walking observed in patients with PD suggests 2 possible scenarios during complex walking tasks. One is a further increase in frontal activation as observed in healthy older adults. The other is inability to further increase activation due to saturation (a ceiling effect), which results in lower performance. Our results show that patients with PD only slightly increased activation during dual task walking. This may provide a possible explanation to the reduced performance of the cognitive task, and the higher dual task cost for gait speed observed in patients with PD. Indeed, we found correlations that indicate that patients with higher prefrontal activation during usual walking and obstacle negotiation had lower disease severity and better gait performance. In addition, in healthy older adults the higher frontal activation was associated with lower dual task cost. This suggests that sufficient frontal activation is required to maintain stable gait that is not affected by the cognitive competition in the presence of the secondary task.
The further increase in HbO2 concentrations during obstacle negotiation in patients with PD reveals that the attenuated response is specific to DT and is actually not a result of HbO2 saturation. Perhaps during walking while negotiating obstacles, the obstacles in the path forced the patients to allocate cognitive resources to motor planning. During DT, low priority could be given to the cognitive task, indicating they prioritize gait and minimized attention to the cognitive task, which may also explain the low performance. However, the high obstacle negotiation cost for gait speed found in patients with PD indicates that this increase in prefrontal activation is not sufficient to compensate for the motor deficits associated with PD.27,29,51 The ability of healthy older adults to maintain gait speed in tasks that pose higher motor and cognitive load as dual tasking and obstacle negotiation can be explained, at least in part, by the alterations in level of frontal activation.
The differences in levels of prefrontal activation between DT and obstacle negotiation can be further explained by the nature of the tasks. Whereas obstacle negotiation can be viewed as an externally driven process, dual tasking is considered an internally driven process. Externally and internally driven processes have been associated with distinct patterns of brain activation. Previous work has shown that externally driven processes are associated with activation of caudal supplementary motor area and posterior occipital-temporal areas while internally driven processes involve greater activation of the rostral supplementary motor area and its adjacent cingulate cortex, as well as the DLPFC.52,53 This might explain the lower prefrontal activation during obstacle negotiation as compared to dual tasking in healthy older adults.
This study has a number of limitations. For example, the fNIRS system that was used included only 2 probes placed on the forehead. This limited our ability to assess activation in other regions of the brain and the involvement of other neural networks. It also limits our capacity to interpret the results in the context other than the DLPFC. Future studies should investigate a more generalized approach, perhaps with more fNIRS probes or other imaging techniques. In addition, fNIRS has good temporal but limited spatial resolution compared to traditional neuroimaging methods as fMRI. We also did not control for superficial hemodynamics54,55 or systemic changes like blood flow or heart rate. Thus, we cannot rule out the influence of these factors, which might have changed during task performance. 56 However, these factors were likely similar between walking tasks and thus cannot explain differences in HbO2 between tasks, or between healthy older adults and patients with PD. In addition, it will be interesting to investigate changes in frontal activation during the OFF state and to explore the associations between changes in HbO2 and other gait features. One more point that should be considered in future studies is randomize the order of the walking conditions to ensure that this did not play a role in the observed results.
Conclusions
The present findings suggest that the specific nature of complex walking plays an important role in dictating the involvement of the frontal lobe in gait. In healthy older adults, a cognitive task apparently leads to increased frontal lobe activation, while obstacle negotiation does to a much lesser extent. The opposite is true in patients with PD. These findings may have important implications for rehabilitation of gait in patients with PD. It suggests 2 complementary approaches for improving gait in more complex conditions in patients with PD: (
Footnotes
Acknowledgements
We would like to thank all the participants and partners in the V-TIME project including the Institute for Aging and Health, University of Newcastle (UNEW), Department of Neurosciences Universita Degli Studi Di Genova (UNIGE); Department of Rehabilitation Sciences, Katholieke Universiteit Leuven (KULeuven), University of Sassari in Sardinia (UNISS), Inition 3D technologies (INITION), Advanced Drug Development Services (ADDS) and Beacon Tech Limited (BTL) for their contribution.
Authors’ Note
Inbal Maidan and Freek Nieuwhof share first authorship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded in part by the European Commission (FP7 project V-TIME- 278169).
