Abstract
Background
The cerebellum plays a crucial role in functional movement by influencing sensorimotor coordination and learning. However, the effects of cortico-cerebellar connectivity on the recovery of upper extremity motor function after stroke have not been investigated. We hypothesized that the integrity of the cortico-cerebellar connections would be reduced in patients with a subacute middle cerebral artery (MCA) stroke, and that this reduction may help to predict chronic upper extremity motor function.
Methods
We retrospectively analyzed the diffusion-tensor imaging of 25 patients with a subacute MCA stroke (mean age: 62.2 ± 2.7 years; 14 females) and 25 age- and sex-matched healthy controls. We evaluated the microstructural integrity of the corticospinal tract (CST), dentatothalamocortical tract (DTCT), and corticopontocerebellar tract (CPCT). Furthermore, we created linear regression models to predict chronic upper extremity motor function based on the structural integrity of each tract.
Results
In stroke patients, the affected DTCT and CST showed significantly impaired structural integrity compared to unaffected tracts and the tracts in controls. When all models were compared, the model that used the fractional anisotropy (FA) asymmetry indices of CST and DTCT as independent variables best predicted chronic upper extremity motor function (R2 = .506, P = .001). The extent of structural integrity of the CPCT did not significantly differ between hemispheres or groups and was not predictive of motor function.
Conclusions
We found evidence that microstructural integrity of the DTCT in the subacute phase of an MCA stroke helped to predict chronic upper extremity motor function, independent of CST status.
Introduction
The recovery of upper extremity motor function after a stroke is a major concern for stroke survivors and clinicians.1,2 The ability to plan and execute coordinated upper extremity movements determines an individual’s independence in activities of daily living. During the subacute phase of a stroke, when most patients begin active rehabilitation treatment, the prediction of upper extremity motor function is important for developing patient-specific treatment strategies and efficiently allocating treatment resources. 3
The corticospinal tract (CST) is a major neural tract of the human brain that mediates voluntary movements, particularly those of the upper extremities.4,5 Previous studies have shown that the lesion load or microstructural integrity of the CST on diffusion tensor imaging (DTI) in the acute to subacute phase of a stroke predicts the motor recovery and motor function of the upper extremities after a stroke.6 -10 Recently, researchers exploring biomarkers of motor recovery after stroke have focused on regions beyond the CST: cortico-cortical connections with secondary motor areas, 11 the transcallosal motor projection, 12 and cortico-cerebellar connections. 13
Recovery from hemiparesis requires a process of motor learning: either true recovery that recruits undamaged brain regions or a form of compensation that uses alternative muscles to perform the task. 14 The relearning of motor function after stroke is similar to motor learning in healthy individuals, 15 and several current neurorehabilitation methods are attempting to achieve effective motor learning. 16 Based on this principle, the role of the cerebellum, which is involved in error-based sensorimotor coordination and learning, must be considered during post-stroke recovery.17,18 The cerebellum integrates and processes information from multiple brain regions to coordinate body movements and participates in motor learning using its cortical and brainstem connections. 17 The cortico-cerebellar connections consist mainly of the corticopontocerebellar tract (CPCT; cerebellar afferent pathway) and the dentatothalamocortical tract (DTCT; cerebellar efferent pathway). The CPCT descends from the motor and non-motor areas of the cerebral cortex, synapses on the pontine nuclei, and crosses through the contralateral middle cerebellar peduncle (MCP) to transmit information to the cerebellar cortex. Extensive cerebral damage after stroke can cause cerebellar deafferentation, followed by hypometabolism of the contralateral cerebellum; this phenomenon is known as crossed cerebellar diaschisis. 19 The DTCT originates from the dentate nucleus, ascends via the superior cerebellar peduncle (SCP), decussates in the midbrain, and projects to the contralateral thalamus to transmit motor-planning information to the cerebral cortex. 20 Recent studies showed a correlation between cortico-cerebellar functional connectivity and motor function after stroke, as measured by functional magnetic resonance imaging (fMRI), or motor-evoked potentials.21 -23 However, to our knowledge, few studies have investigated whether the structural connectivity of the cortico-cerebellar tracts predicts motor recovery or chronic motor function after a stroke.
We compared the cortico-cerebellar microstructural integrity of patients with a subacute middle cerebral artery (MCA) stroke and healthy controls, then evaluated the correlation between the microstructural integrity of these white matter tracts and chronic upper extremity motor function in stroke patients. We hypothesized that the cortico-cerebellar structural connection may be disrupted in patients with a subacute MCA stroke compared to healthy controls, and that this reduction may predict chronic upper extremity motor function independent of CST integrity.
Methods
Study Design and Subjects
Patients were retrospectively selected from a hospital-based stroke cohort registered in the Department of Rehabilitation Medicine, St. Vincent’s Hospital, Suwon, Republic of Korea, between December 2015 and May 2021. These patients were admitted for management of the subacute phase of a stroke in the inpatient rehabilitation facility. Of the 104 patients who underwent brain DTI, 25 met the following inclusion criteria. All subjects were aged ≥18 years, had experienced a first-ever unilateral MCA-territory ischemic stroke, underwent brain DTI within 30 days of stroke onset, and underwent Fugl-Meyer Assessment for Upper Extremity (FMA-UE; score range 0-66) evaluation more than 6 months after the stroke. We excluded subjects with new-onset neurological impairment during the observation period, neurologic disorders other than stroke, or an inability to receive standard rehabilitation treatment due to serious medical complications during their stay in the inpatient rehabilitation facility. This protocol was reviewed and approved by the Institutional Review Board of St. Vincent’s Hospital, Suwon, Republic of Korea (Registry No. VC22RISI0245, approval date 13 October 2022) and was performed in accordance with the tenets of the Declaration of Helsinki. The Board waived the need for informed consent because we used retrospective and anonymized data.
The 25 age- and sex-matched healthy right-handed controls were recruited from individuals who underwent brain MRI as part of an advanced health checkup at our institutional health promotion center between March 2015 and June 2016. The exclusion criteria were a history of neurological disorder, as revealed by the health questionnaire or physical examination; diffuse white matter hyperintensity on T2-weighted images (Fazekas score ≥2 for periventricular and/or deep white matter); and/or any evidence of structural abnormality on the scan. Informed consent was obtained from the subjects concerning the use of their brain MRI data for research purposes. All controls were right-handed; the dominant hemispheres of 15 healthy controls were randomly assigned to “affected” to equalize the distribution ratio of lesions in the dominant and nondominant hemispheres of stroke patients.
DTI Acquisition
DTI was performed using a 3.0-T MRI (MAGNETOM® Verio; Siemens, Erlangen, Germany) equipped with a 6-channel head coil. All data from patients and controls were acquired in the form of single-shot spin-echo echo-planar images, with axial slices covering the entire brain across 76 interleaved 2.0-mm thick slices (no gap; repetition time/echo time = 14 300/84 ms; field of view = 224 × 224 mm2; matrix = 224 × 224; voxel size = 1 × 1 × 2 mm3; number of excitations = 1). Diffusion sensitizing gradients were applied in 64 noncollinear directions with a b-value of 1000 ms/mm2. The b = 0 images were scanned before acquisition of the diffusion-weighted images (65 volumes).
Image Processing and DTI Analysis
The images were processed using the FMRIB Software Library (FSL; version 6.0.5; http://www.fmrib.ox.ac.uk/fsl). Source data were corrected for eddy currents and head motion by registration to the first b = 0 image using affine transformation. Fractional anisotropy (FA) maps were constructed using the DTIFIT program. The regions of interest (ROIs) were manually defined on a voxel-by-voxel basis by 2 investigators (Y.Y. and Y.K.) who had > 5 years of experience in brain anatomy and DTI, and who were blinded to the motor function of the patients.24,25 The ROI design at each location was determined based on the topographical maps in previous studies and color FA maps (Figure 1).26 -29 Two ROIs for the CST were placed in the area represented by the blue fibers in the anterior portion of the color FA map at the level of the mid-pons and pontomedullary junction (PMJ) in the axial plane. One ROI for the CPCT was defined in the MCP and another ROI for the DTCT was defined in the SCP, both in the same coronal image. In the section posterior to the CST in the coronal plane of the FA map, MCPs were delineated in the areas consisting of green fibers to the left and right of the transverse pontine fibers, and SCPs were delineated as cyan fibers rostral to the transverse pontine fibers. ROIs of the affected CPCT and DTCT were defined on the contralateral side of supratentorial stroke lesions, and ROIs of the affected CST were defined on the ipsilateral side of supratentorial stroke lesions. Quantitative values of the FA were obtained by averaging all voxels within the ROI using the FSLUTILS program. The asymmetry index of FA (AI_FA) was calculated using the following formula: (FAaffected – FAunaffected)/(FAaffected + FAunaffected). 30 A low AI_FA indicated that the FA of the affected side was lower than that of the unaffected side, whereas a high AI_FA indicated the opposite. Lesion volume was manually measured.
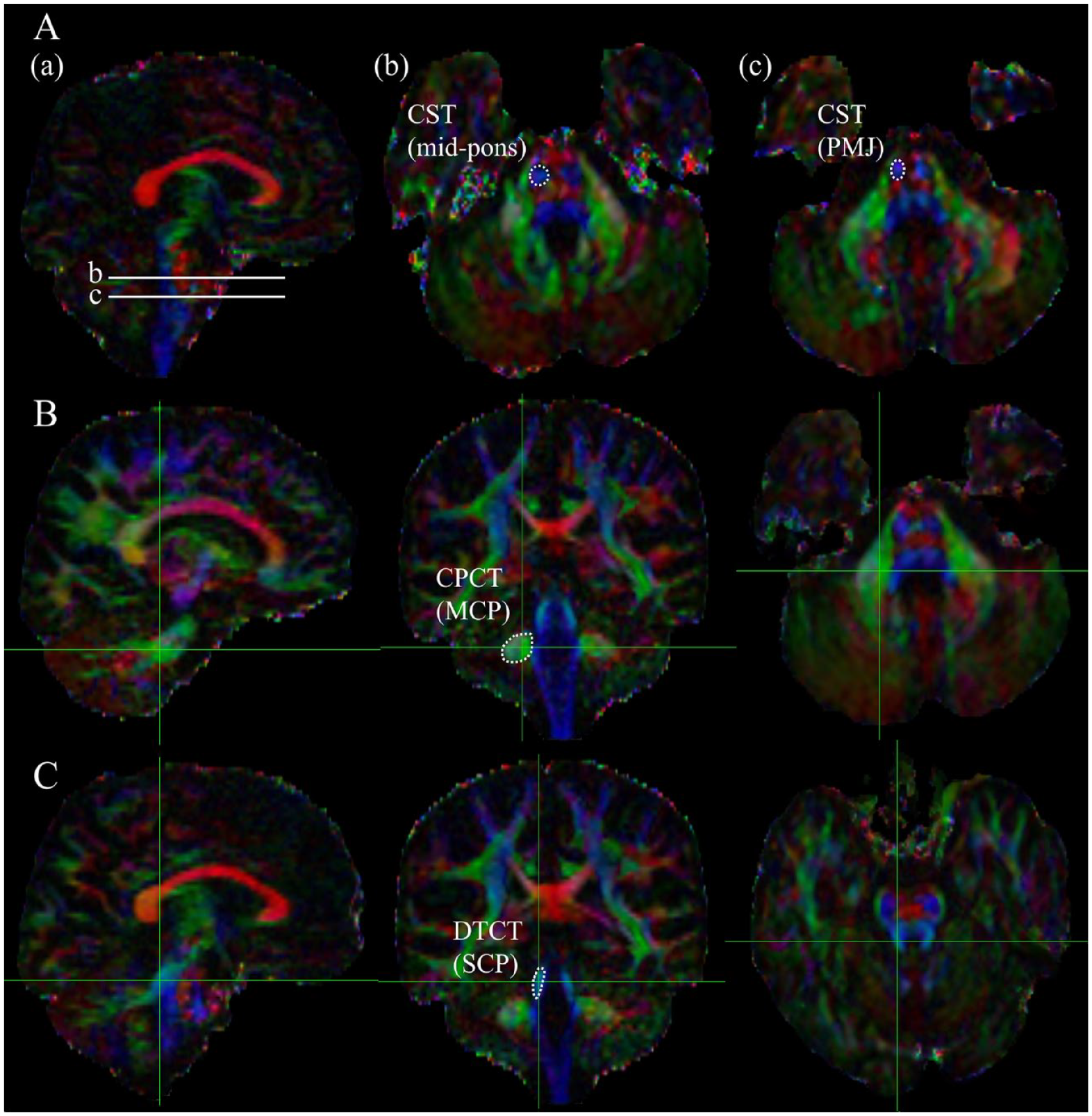
Color fractional anisotropy (FA) maps of diffusion-tensor images delineating the regions of interest (ROIs) for each tract. (A) Sections for the corticospinal tract (CST); (a) 2 levels for placing the ROIs are indicated on sagittal images [b. mid-pons and c. pontomedullary junction (PMJ)]. ROIs of the CST at the (b) mid-pons and (c) PMJ. (B) Sections for the corticopontocerebellar tract (CPCT); sagittal, coronal, and axial sections are shown from left to right. The ROI for the middle cerebellar peduncle (MCP) is indicated by a dotted line in the middle coronal section image. (C) Sections for the dentatothalamocortical tract (DTCT); sagittal, coronal, and axial sections are shown from left to right. The ROI for the superior cerebellar peduncle (SCP) is indicated by a dotted line in the middle coronal section image.
Statistical Analysis
The Kolmogorov–Smirnov test and Shapiro–Wilk test were used to determine whether the functional parameters and DTI-derived indices were normally distributed. The inter-rater reliability of the ROI measurements of Y.Y. and Y.K. was assessed by interclass correlation coefficients (ICCs) estimates with 95% confidence intervals (CI) using a mean-rating (k = 2), absolute-agreement, 2-way mixed-effects model. Paired t-tests were used to compare the FA values of the tracts of affected and unaffected hemispheres; the DTCT of controls was assessed using the Wilcoxon signed-rank test because the data were not normally distributed. Unpaired t-tests were used to compare AI_FA values between patients and controls. Univariate and multivariate linear regression models based on the stepwise method were used to determine the association between the AI_FA and FMA-UE scores. Age, sex, lesion volume, and lesion location in the dominant hemisphere were selected as the confounding variables, whereas the AI_FA obtained at each anatomical location was used as the independent variable. We also investigated the interactions between the FA values of each white matter tract, which were significant in univariate analysis. The stepping method criteria were based on P < .05 for entry and P > .10 for removal. Statistical tests were performed using the stepwiselm functions and the interaction terms (products of pairs of predictors) in the statistics toolbox of MATLAB Release 2022a (MathWorks, Natick, MA, USA). All tests were 2-tailed, and P < .05 was considered statistically significant.
Results
The demographic and clinical characteristics of the patients are presented in Table 1. The inter-rater reliability of ROI measurements (ICC) between the 2 investigators (Y.Y. and Y.K.) was 0.855 (95% CI: 0.733-0.919).
Demographic and Clinical Characteristics of the Patients and Controls.
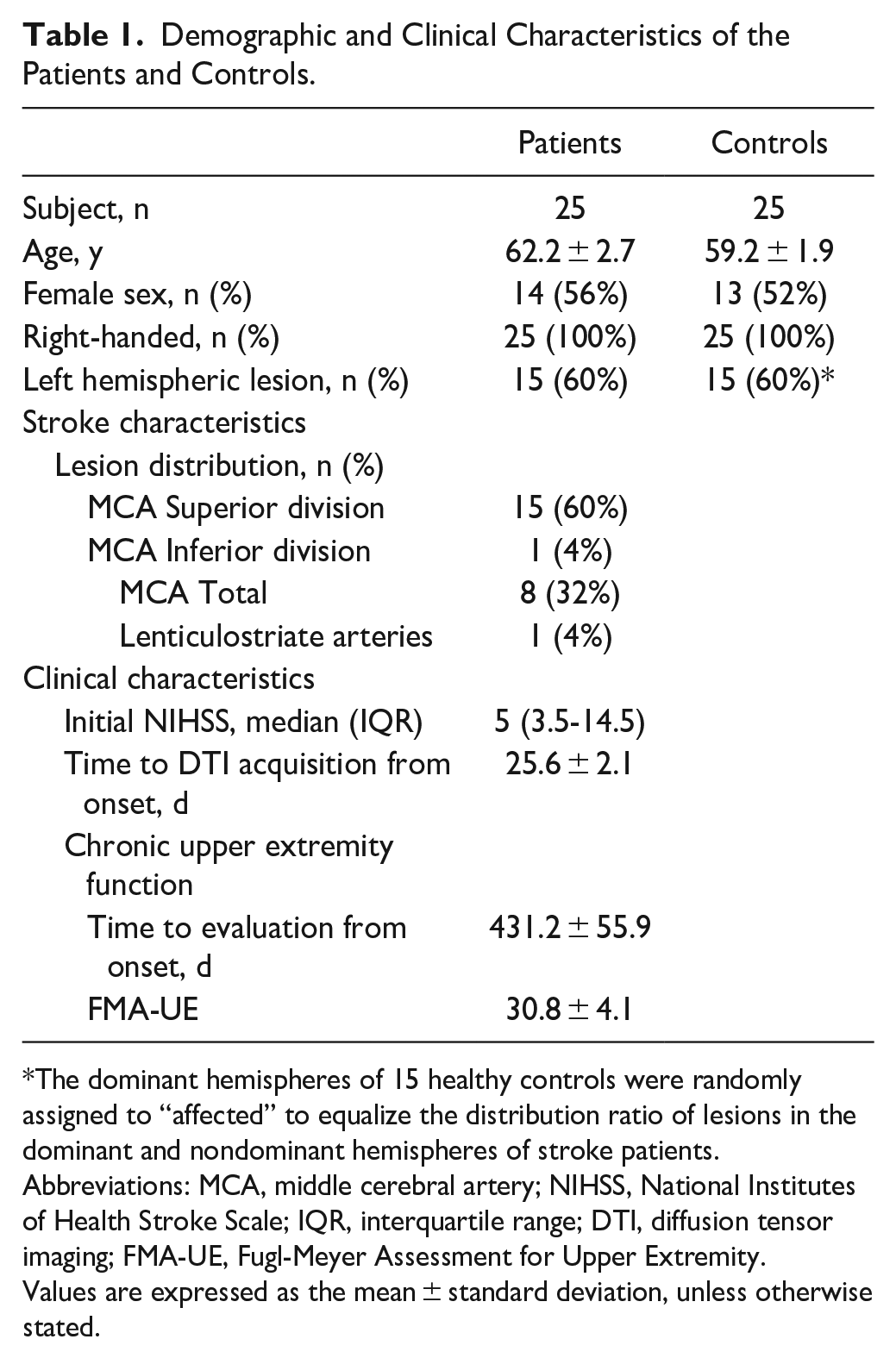
The dominant hemispheres of 15 healthy controls were randomly assigned to “affected” to equalize the distribution ratio of lesions in the dominant and nondominant hemispheres of stroke patients.
Abbreviations: MCA, middle cerebral artery; NIHSS, National Institutes of Health Stroke Scale; IQR, interquartile range; DTI, diffusion tensor imaging; FMA-UE, Fugl-Meyer Assessment for Upper Extremity.
Values are expressed as the mean ± standard deviation, unless otherwise stated.
Comparison of White Matter Integrity Within and Between Subjects
Table 2 summarizes the FA values and AI_FA for each tract in the stroke patients and controls. A pairwise comparison was performed for each tract in the affected and unaffected hemispheres. In the stroke group, the FA was significantly lower for the affected CST at the PMJ and the mid-pons (both P < .001), and for the DTCT (P = .029), compared to the unaffected tracts. However, no significant differences were observed between the sides for the CPCT (P = .448). In the control group, there were no significant differences between the hemispheres, except for the CST at the PMJ (P = .043). Figure 2 shows representative images of the fiber connection differences between stroke patients and healthy controls.
Comparison of Regional FA Between Affected and Unaffected Tracts and Comparison of FA Asymmetry Indices Between Patients and Controls.
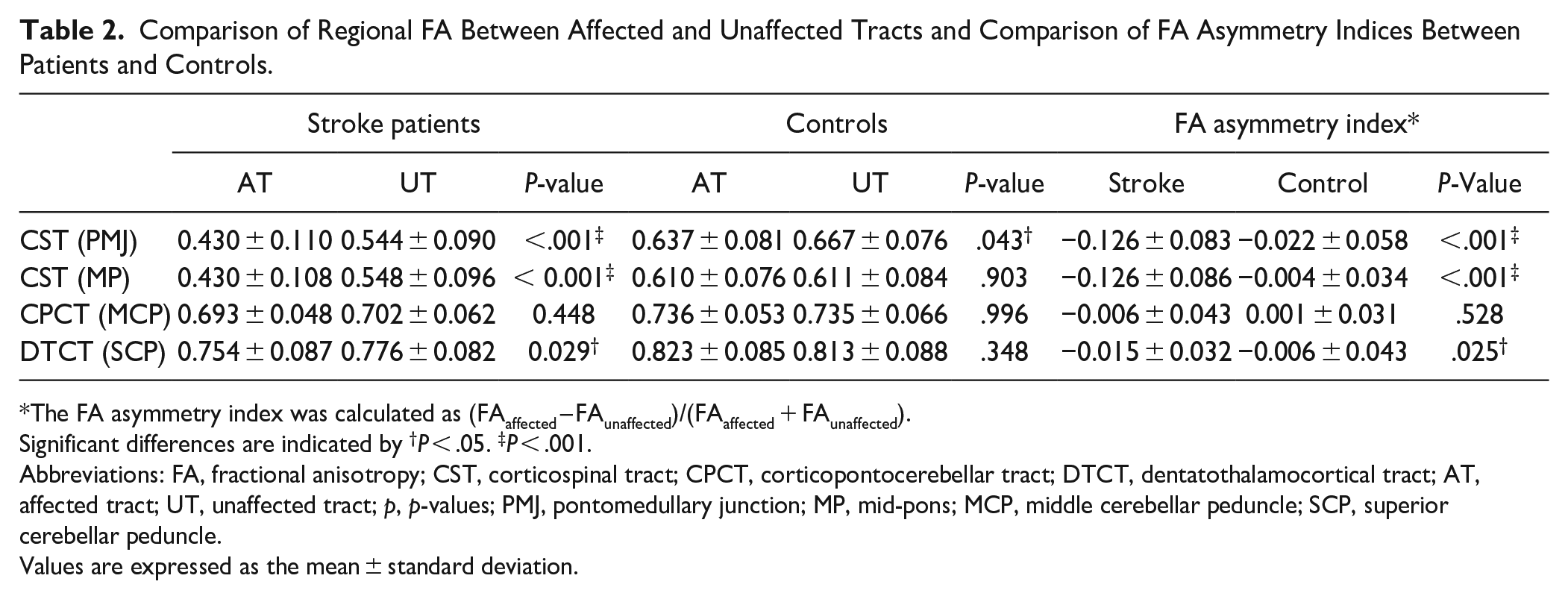
The FA asymmetry index was calculated as (FAaffected – FAunaffected)/(FAaffected + FAunaffected).
Significant differences are indicated by †P < .05. ‡P < .001.
Abbreviations: FA, fractional anisotropy; CST, corticospinal tract; CPCT, corticopontocerebellar tract; DTCT, dentatothalamocortical tract; AT, affected tract; UT, unaffected tract; p, p-values; PMJ, pontomedullary junction; MP, mid-pons; MCP, middle cerebellar peduncle; SCP, superior cerebellar peduncle.
Values are expressed as the mean ± standard deviation.
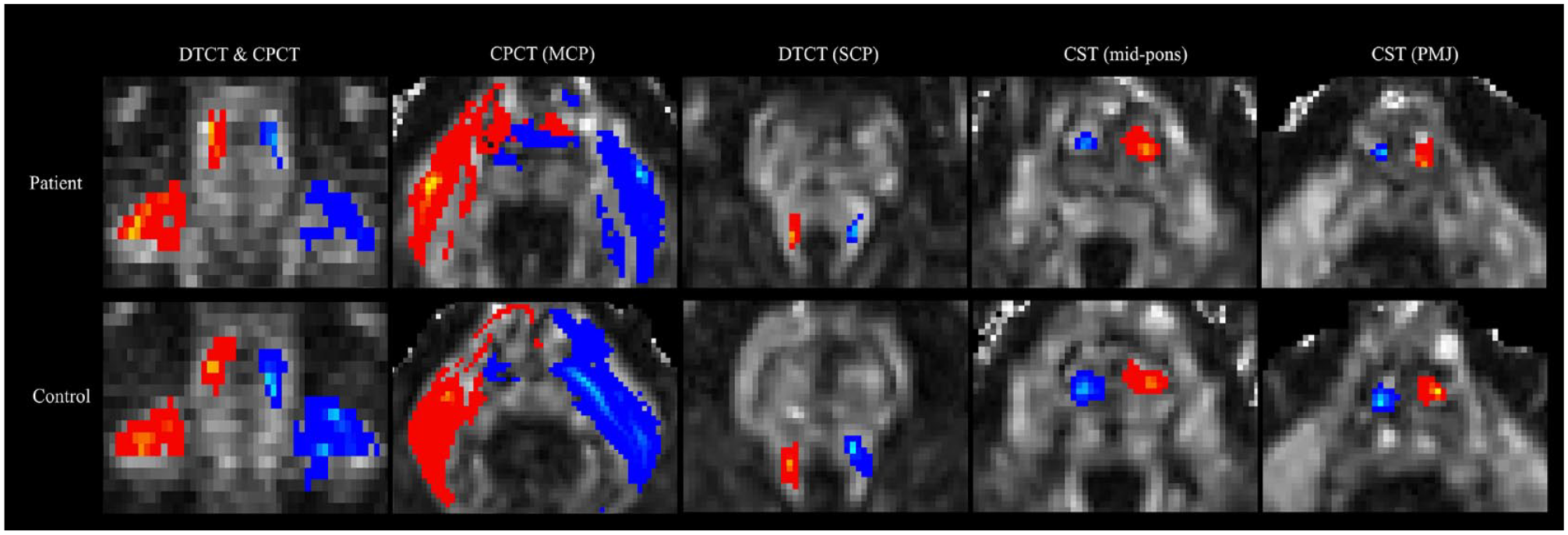
Representative images of the differences in fiber connections between a stroke patient (top) and a healthy control (bottom). The patient had a lesion in the right cerebral hemisphere. Depending on hemisphere orientation, the right tracts are shown in blue-light blue, and the left tracts are shown in red-yellow. The dentatothalamocortical tract (DTCT) at the superior cerebellar peduncle (SCP) and the corticopontocerebellar tract (CPCT) at the middle cerebellar peduncle (MCP) are shown together on the coronal slice, and separately on axial slices, of the FA map. The corticospinal tract (CST) at the mid-pons and pontomedullary junction (PMJ) is shown on axial slices in the FA map.
The AI_FA was significantly lower in the stroke group than in the control group for the CST (at the PMJ and mid-pons, both P < .001). The AI_FA was also significantly lower in stroke patients for the DTCT (P = .025) compared to the controls; however, there was no significant difference in the AI_FA for the CPCT between the groups (P = .528).
Predictive Model for Upper Extremity Motor Function Based on Regional AI_FA
In univariate analysis, the AI_FA of the CST and DTCT significantly predicted the chronic FMA-UE; the explanatory power of the CST at the PMJ (R2 = .354, F = 12.034, P = .002), the CST at the mid-pons (R2 = .284, F = 8.715, P = .007), and the DTCT (R2 = .250, F = 7.340, P = .013). However, the AI_FA of the CPCT did not predict upper extremity motor function (Table 3).
Univariate and multivariate linear regression models for predicting upper limb motor outcomes based on FA asymmetry indices for the CST, CPCT, and DTCT after a stroke.
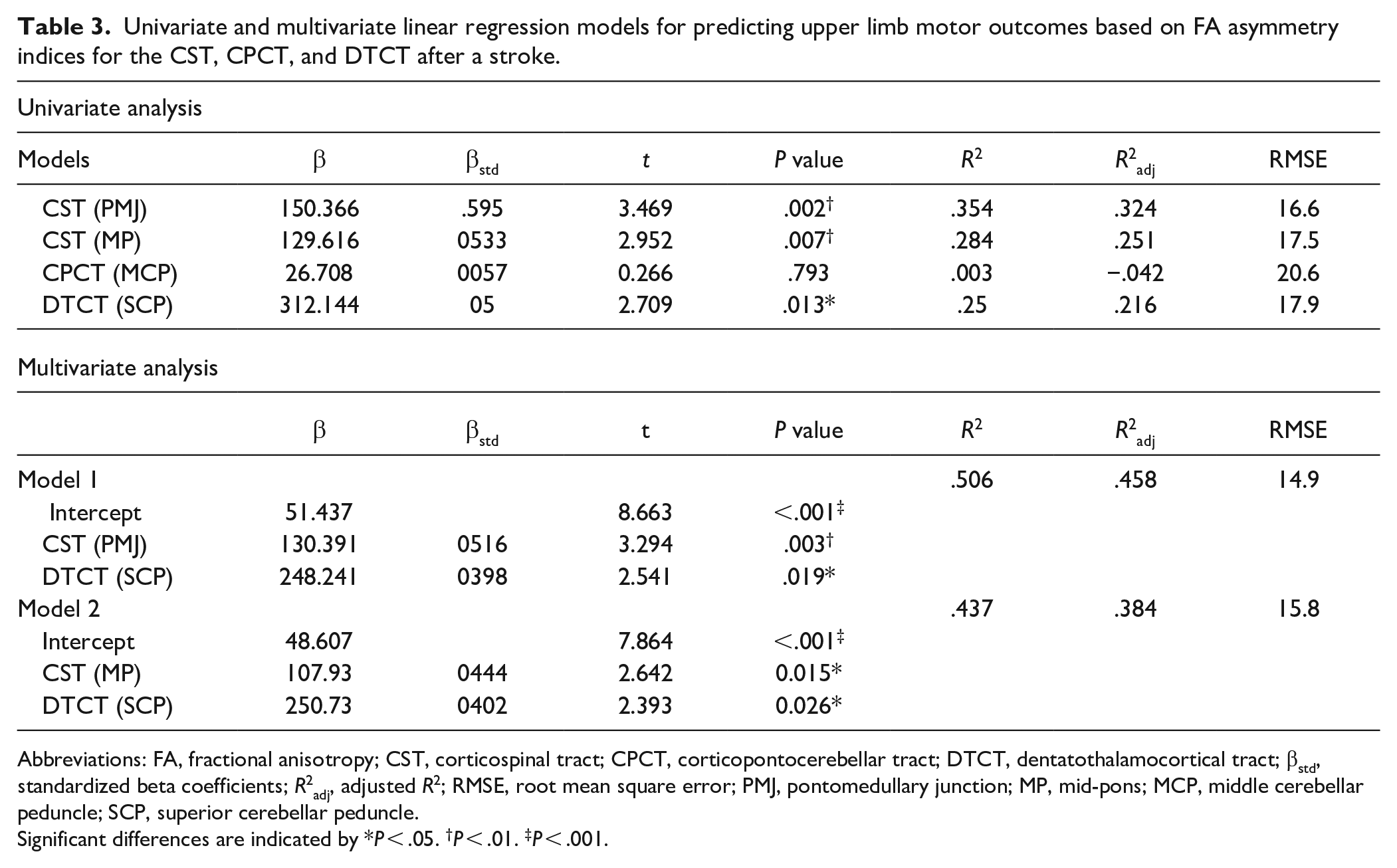
Abbreviations: FA, fractional anisotropy; CST, corticospinal tract; CPCT, corticopontocerebellar tract; DTCT, dentatothalamocortical tract; βstd, standardized beta coefficients; R2adj, adjusted R2; RMSE, root mean square error; PMJ, pontomedullary junction; MP, mid-pons; MCP, middle cerebellar peduncle; SCP, superior cerebellar peduncle.
Significant differences are indicated by *P < .05. †P < .01. ‡P < .001.
Based on the results of univariate analysis, we constructed 2 multivariate linear regression models to predict the FMA-UE: Model 1 used the AI_FA of the CST (at the PMJ) and DTCT, along with their interaction, whereas Model 2 used the AI_FA of the CST (at the mid-pons) and DTCT, along with their interaction. Age, sex, lesion volume, and lesion location (in the dominant hemisphere or not) were regarded as confounding variables, but they were not included in the final models.
In Model 1, the AI_FA of the CST (at the PMJ) and DTCT independently contributed to the FMA-UE (P = .003 and .019, respectively), but no interaction was observed (P = .813). The explanatory power of the Model 1 was R2 = .506 (F = 10.737, P < .001). In Model 2, the AI_FA of the CST (at the mid-pons) and the DTCT independently contributed to the FMA-UE (P = .015 and .026, respectively), but no interaction was observed (P = .686). The explanatory power of the Model 2 was R2 = .437 (F = 8.157, P = .002).
Table 3 summarizes the statistics of the models used in univariate and multivariate linear regression analyses. When all models were compared, the model that best predicted chronic upper extremity motor function was Model 1, which used the AI_FA of the CST (at the PMJ) and the DTCT as independent variables (R2 = .506 and root-mean-square error of 14.9). Multivariate linear regression plots of Model 1 and Model 2 are shown in Figure 3.
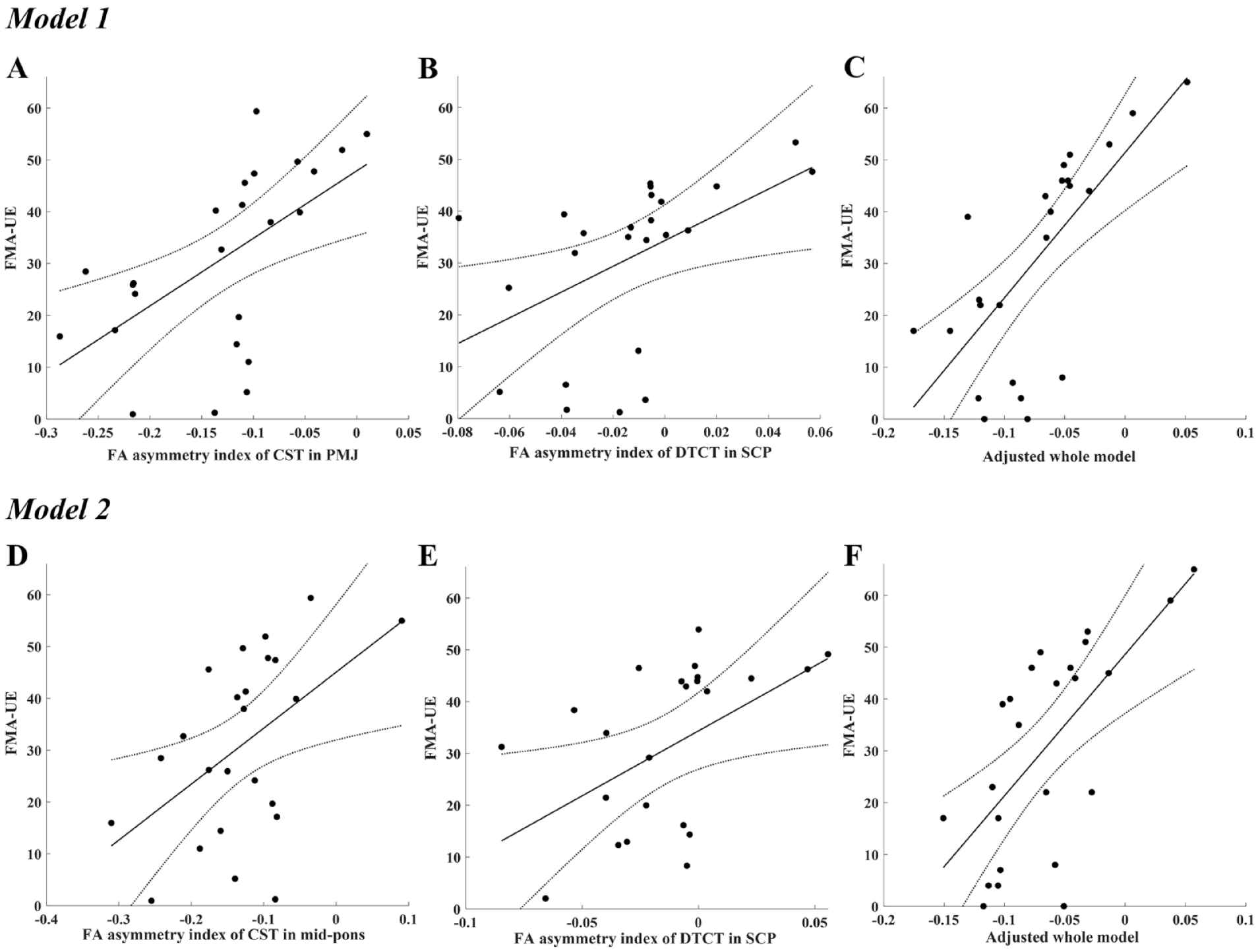
Multivariate linear regression models used to predict the Fugl-Meyer Assessment for Upper Extremity (FMA-UE) based on the fractional anisotropy (FA) asymmetry indices of each tract. Fitted lines and 95% confidence intervals for the FMA-UE are plotted for each FA asymmetry index. Model 1: plots A and B show the relationships between the FMA-UE and the FA asymmetry index of each tract [A, the corticospinal tract (CST) measured at the pontomedullary junction (PMJ); B, the dentatothalamocortical tract (DTCT) measured at the superior cerebellar peduncle (SCP)] after controlling for the effects of other tract. Plot C shows the final adjusted model, which considers the effects of both tracts. Model 2: plots D and E show the relationship between the FMA-UE and the FA asymmetry index of each tract (D, CST measured at the mid-pons; E, DTCT measured at the SCP) after controlling for the effects of other tract. Plot F shows the final adjusted model, which considers the effects of both tracts.
Discussion
In this study, we hypothesized that the microstructural integrity of cortico-cerebellar structural connections would be reduced in patients with a subacute MCA stroke compared to healthy controls, and that this reduction in white matter integrity would be a useful independent predictor of chronic upper extremity motor function. We evaluated the CPCT, DTCT, and CST using DTI in patients at the subacute phase of an MCA stroke and healthy controls. In the stroke group, the affected CST and DTCT had lower FA values compared to those of the unaffected side and the control group. The AI_FA of the DTCT improved the explanatory power of the multivariate model predicting chronic upper extremity motor function to R2 = .506, compared with a univariate model using the CST (PMJ) only (R2 = .354). No differences were observed in the microstructural integrity of the CPCT between hemispheres or groups. Furthermore, there was no correlation between the AI_FA of the CPCT and motor function. In summary, the microstructural integrity of the cerebellar outflow tract (the DTCT) measured within 1 month of stroke onset, but not the CPCT, helped to predict chronic upper extremity motor function. These findings imply that the effect of the cerebellum on the excitability of the lesioned motor cortex plays an important role in the recovery of motor function.
Microstructural Integrity of the CST and Motor Function
Previous neuroimaging studies have shown an effect of CST injury on motor recovery and functional outcome. 31 FA asymmetry measured at the posterior limb of the internal capsule in the acute phase of stroke predicted motor function 3 months after the stroke, and FA asymmetry measured at the pons 30 days after stroke onset predicted motor function at 2 years.32,33 In the present study, FA asymmetry measured at the PMJ was a better predictor of chronic motor function than that measured at the mid-pons. FA asymmetry at the mid-pons and PMJ reflects Wallerian degeneration of the CST in the subacute phase of supratentorial strokes.34,35 However, the CST is intertwined with the transverse pontine fibers in the ventral pons, whereas the PMJ consists of the relatively pure CST. Accordingly, in the subacute phase of an MCA stroke, FA asymmetry in the PMJ better reflects the structural reserve of the CST than that of the mid-pons.
Microstructural Integrity of Cortico-Cerebellar Pathways and Motor Function
The DTCT, the major cerebellar outflow tract projecting into the neocortex, is involved in the accurate timing of movements and the acquisition of novel movement skills. 36 The DTCT originates from the dentate nucleus and targets the ventral premotor area, dorsal premotor cortex, supplementary motor area, and primary motor cortex. 37 Cerebellar outputs to the primary motor and premotor areas are presumed to participate in cortical excitability. In patients with unilateral cerebellar strokes, decreased neuronal excitability of the contralesional motor cortex has been observed.38,39 A previous study suggested that transcranial magnetic stimulation (TMS) modulated connectivity between the cerebellum and primary motor cortex. When TMS of the contralateral cerebellar hemisphere preceded TMS of the motor cortex by 5 to 7 ms, the motor evoked potential (MEP) amplitude decreased through inhibition of the dentatothalamic excitatory projection; the phenomenon known as “cerebellum-brain inhibition (CBI)”. 40 Lesions involving this cerebello-thalamo-cortical pathway, such as in the posterior limb of the internal capsule or the thalamus, can reduce the CBI. 41 Novel treatment strategies might target the cerebellar hemisphere. Such an approach would allow non-impaired entry prior to stimulation of the cortico-cerebellar connections, thus potentiating cerebellar-mediated motor learning after supratentorial stroke. 42 Recent studies revealed that intermittent theta-burst stimulation of the contralesional cerebellar hemisphere improved the visuo-motor learning and spasticity of the affected upper extremity in patients with subacute to chronic supratentorial strokes.43,44 However, further research is required prior to clinical application.
Patients with chronic supratentorial strokes develop changes in the microstructural integrity of the DTCT, which may be due to retrograde axonal degeneration after cortical lesions.13,22 In a study of patients with chronic supratentorial strokes, cortical excitability was associated with cortico-cerebellar connectivity, as measured by the DTCT, but not with the CPCT. 22 Based on our findings, we speculate that the decreased microstructural integrity of the DTCT reduces the excitability of cortico-cortical connections, thereby disrupting the neuroplasticity in cortical regions and inhibiting motor re-learning after a stroke. Conversely, the CPCT does not target the motor cortex and has limited effects on cortical excitability.
The CPCT, a major input pathway to the cerebellum (via the MCP), originates in several cerebral cortical areas, including the sensorimotor cortex; it conveys information regarding the coordination of voluntary motor activity to the dentate nucleus. 20 Previous studies have revealed that regional FA values for the CPCT in patients during the subacute and chronic phases of a supratentorial stroke were comparable to those of the unaffected side or controls.13,24 In these studies, there was no association between FA asymmetry of the CPCT and the strength or general motor function of the upper extremities. However, other studies have demonstrated significant correlations between FA asymmetry of the CPCT and fine motor skills, as measured by the 9-hole peg test or Jepson’s hand-function test.13,45 These findings imply that the microstructural integrity of CPCT is poorly correlated with the general motor output of the upper extremities, although it may be correlated with fine motor skills in the hands.
Limitations
There were several limitations in this study. First, we obtained DTI-derived indices using a region-based ROI analysis method that involved manually drawing anatomical regions rather than a tract-based ROI analysis method. Both methods have technical limitations. Tract-based ROI analysis has the potential to underestimate DTI-derived indices, as degeneration of the target tract results in ROIs that are not included in the analysis. Therefore, we selected the region-based ROI analysis method. However, the accuracy of this method depends on the spatial resolution of the data. Second, the cortico-cerebellar connection has a complex white matter architecture that includes crossing fibers. For example, while the SCP is the primary efferent connection from the cerebellum to the thalamus, it also includes afferent and efferent pathways that target other areas such as the cerebellorubral and anterior spinocerebellar tracts, which may also affect outcomes. 46 Although the MCP and SCP include only a small proportion of other fibers and we selected these as representative regions to obtain DTI-derived indices, the results may vary when the analysis includes the entire tract or other parts of the tract. Third, the number of subjects was small because of the strict inclusion criteria; this small number may undermine the clinical significance of the findings. Fourth, the median (interquartile range) of the initial National Institutes of Health Stroke Scale (NIHSS) of patients was 5 (3.5-14.5), indicating mild-to-moderate impairment. Accordingly, our predictive model may not be generalizable. External validation is required to determine model reproducibility and generalizability using more patients with various severities of stroke. Fifth, the extent of rehabilitation therapy was not considered; this factor may influence motor recovery. 47 All patients admitted to the inpatient rehabilitation facility in the subacute phase of stroke underwent comprehensive rehabilitation therapy including physical, occupational, and speech-language therapy, as needed, for 1 hour twice per day, 5 days per week, for 3 to 4 weeks. However, this approach was not strictly controlled, and no rehabilitation therapy received after discharge from our hospital was investigated.
Conclusions
We found evidence that the microstructural integrity of the DTCT in the subacute phase of an MCA stroke helped to predict the chronic upper extremity motor function, independent of CST status. We speculate that this relationship can be explained by an effect of the cerebellar outflow tract on the excitability of the cortico-cortical connection, which contributes to the recovery of upper extremity motor function. Based on our results, the utility of DTI as a prognostic tool can be enhanced using an accurate recovery prediction model that considers cortico-cerebellar connectivity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosures
No commercial party with financial interests in this study has provided or will provide benefits to the authors or any organization with which the authors are associated.
