Abstract
Background
In stroke patients, preserved cognitive function plays a role in motor recovery, but there is insufficient evidence on the involved mechanisms. These mechanisms require investigation in the human brain, which is composed of large-scale functionally specialized networks.
Objective
In this study, we investigated the role of cognition-related networks on upper extremity motor recovery using neuroimaging data of subacute stroke patients.
Methods
This study retrospectively analyzed cohort data of 108 subacute ischemic stroke patients. All patients underwent resting-state functional MRI and motor function assessments using the Fugl-Meyer assessment (FMA) at 2 weeks after stroke onset. The FMA upper extremity (FMA-UE) score was obtained again at three months after stroke onset to assess motor recovery. To construct a resting-state network, cortical surface parcellation was performed using the Gordon atlas, which included 333 regions of interest, and 12 resting-state networks were extracted. Linear regression was used to identify the relationships between the FMA-UE recovery score and resting-state networks.
Results
Cognition-related networks were correlated with the FMA-UE recovery score, as were motor-related networks. Interaction effects between motor- and cognition-related network states existed in motor recovery. Specifically, cognition-related networks were associated with motor recovery in patients with a lower strength of motor-related networks.
Conclusions
These results suggested that the greater the damage to the motor network caused by stroke is, the more important the cognition-related networks are in motor recovery.
Introduction
Motor and cognitive function and recovery have been mainly studied separately in stroke patients. Specifically, studies related to motor function and recovery have dealt with important motor-specific biomarkers such as initial motor impairment, motor evoked potential (MEP) response, corticospinal tract (CST) lesion load, and preservation of motor-related tracts and networks.1 -3 However, the human brain is composed of complex and large-scale functionally specialized networks, including sensorimotor, visual, auditory, attention, and executive networks, which are reciprocally connected to each other.4,5 Cooperation between functions is involved even in simple actions. 6 The interrelationships between functions, particularly between motor and cognitive functions, have been reported for healthy individuals and stroke patients.7 -11 The interplay between cognitive and motor functioning in healthy older adults was found in dual-task studies, and a positive relationship between cognitive and motor skills was reported in developing children.7,8 In stroke patients, motor functions, including upper limb function and gait and balance functions, were consistently correlated with cognitive function, and preserved cognitive function is known to play a role in motor recovery.9 -11 Therefore, it is necessary to consider other functional states when analyzing the recovery of a specific function in stroke patients.
Despite the evidence regarding the relationship between motor and cognitive functions, their functional roles in the injured brain were not sufficiently examined. According to previous neuroimaging and MEP studies in healthy individuals, the primary motor cortex is activated during motor imagery and movement observation in the absence of movement, while corticospinal excitability was altered at an initial intention to perform a motor action. 12 On the other hand, it is difficult to find studies dealing with the relationship between motor and cognition-related measures in terms of recovery in the brain after a stroke, although such studies in stroke patients may provide important insights into functional recovery.
In this study, we investigated the role of cognitive networks on motor recovery using neuroimaging data of subacute stroke patients. Although upper and lower limb recovery patterns are similar for acute and subacute phases, previous studies have shown different underlying mechanisms of upper and lower limb motor recovery after stroke.11,13 Lower limb recovery seems to be more complicated than that of the upper limb due to greater involvement of additional extrapyramidal tracts and the absence of definite neuroimaging biomarkers in the lower limb.14,15 Therefore, we focused on the role of cognitive networks on motor recovery of the upper limbs. Neuroimaging techniques can explore large-scale functionally specialized networks in the brain. Through extraction of large-scale networks from resting-state functional MRI (rs-fMRI) data, structurally segregated and functionally specialized networks, including motor and cognition-related networks, can be characterized. 13 We hypothesized that motor- and cognition-related network states are associated with each other in subacute stroke patients, and the cognition-related network is involved in motor recovery as in is the motor-related network. Additionally, we identified the effects of the cognition-related network state depending on the motor-related network state for motor recovery.
Methods
Participants
One-hundred twenty-four ischemic stroke patients from the stroke database of the Department of Physical and Rehabilitation Medicine, Samsung Medical Center were screened retrospectively. The database includes MRI data of stroke patients with a motor impairment who received comprehensive inpatient rehabilitation therapy for 3 weeks during the subacute phase. All patients received at least 3 hours of rehabilitation therapy including 2 hours of physical and 1 hour of occupational therapies per day, 5 days per week during their admission. In addition, speech-language and swallowing therapies were provided as needed. The inclusion criteria were: (1) age 19 years or older at the time of stroke onset, (2) first-ever ischemic stroke, (3) T1-weighted MRI and rs-fMRI data acquisition at 2 weeks after stroke onset, and (4) Fugl-Meyer Assessment upper extremity (FMA-UE) score at 2 weeks and 3 months. The exclusion criteria were: (1) clinically significant neuropsychiatric comorbidities in addition to stroke, (2) metallic implants in the brain, (3) hemorrhagic stroke, (4) bilateral lesions, and (5) recurrent stroke. One-hundred and fifteen stroke patients were included in this study. Of these, there was a failure of spatial registration during data processing in 6 participants, and the FMA-UE score at 3 months was not precisely assessed by a severe tremor in 1 participant. Thus, data analyses were finally performed on 108 ischemic stroke patients, and participants’ demographic and clinical information is summarized in Table 1.
Demographic and Clinical Characteristics of Participants.

Abbreviations: SD, standard deviation; NIHSS, National Institutes of Health Stroke Scale; MMSE, Mini Mental State Examination; FMA, Fugl-Meyer assessment; UE, upper extremity; LE, lower extremity.
Indicates data of some participants (n = 64).
Each patient lesion was segmented on a T1-weighted structural image with reference to the apparent hyper-intensities on the diffusion-weighted image, which was acquired at the patient’s first neurology appointment. One medical doctor manually drew the lesion maps of all patients in native space using MRIcron software (University of South Carolina, Columbia, SC, USA, http://www.cabiatl.com/mricro/mricron/index.html). The individual lesion was normalized in standard Montreal Neurological Institute space using a forward warp field generated during T1-weighted structural image processing based on SPM (Wellcome Trust Center for Neuroimaging, University College London, London, UK, https://www.fil.ion.ucl.ac.uk/spm/software/spm12). The lesion distribution was visualized using MRIcroGL (McCausland Center for Brain Imaging, University of South Carolina, USA, http://www.cabiatl.com/mricrogl; Figure 1). The lesion side of the map was flipped to the right side for visualization.
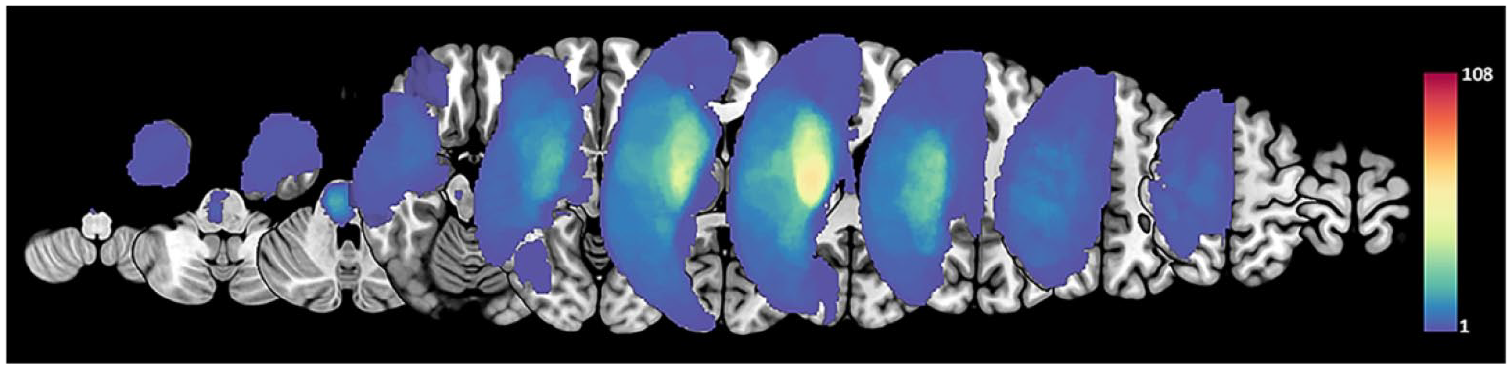
Lesion maps. The stroke lesions were flipped for patients with lesions on the left side. All lesions were overlaid on the right side. The colored bar indicates the number of participants.
This study was approved by the Institutional Review Board (IRB) of Samsung Medical Center, Seoul, Republic of Korea. This study uses only previously collected anonymous data, and an exemption for informed consent was received from the IRB.
Data Acquisition
Resting-state fMRI and T1-weighted structural MRI data were sequentially acquired using a Philips ACHIEVA® MR scanner (Philips Medical Systems, Best, The Netherlands) operating at 3 T. Participants were instructed to keep their eyes closed and to remain motionless during the rs-fMRI scan. One-hundred whole-brain rs-fMRI images were acquired using a T2*-weighted gradient echo-planar imaging (EPI) sequence with the following metrics: 35 axial slices, slice thickness = 4 mm, no gap, matrix size = 128 × 128, voxel size = 1.72 mm × 1.72 mm, repetition time = 3000 ms, echo time = 35 ms, flip angle = 90°, and field of view = 220 × 220 mm. T1-weighted structural images were acquired with the following settings: 124 axial slices, slice thickness = 1.6 mm, no gap, matrix size = 512 × 512, voxel size = 0.47 mm × 0.47 mm, repetition time = 13.9 ms, echo time = 6.89 ms, flip angle = 8°, and field of view = 240 mm × 240 mm. Diffusion-weighted images were acquired at the patient’s first neurology appointment using the following image sequence and metrics:
Data Processing for Extracting Resting-State Networks
To extract resting-state networks from rs-fMRI data, CONN toolbox (McGovern Institute for Brain Research, Massachusetts Institute of Technology, Cambridge, MA, USA, http://www.nitrc.org/projects/conn) and CAT12 toolbox (Structural Brain Mapping Group, Jena University Hospital, Jena, Germany, http://www.neuro.uni-jena.de/cat/), based on SPM12 (Wellcome Trust Center for Neuroimaging, University College London, London, UK, https://www.fil.ion.ucl.ac.uk/spm/software/spm12), implemented in MATLAB® were employed.
fMRI data pre-processing contained head motion and slice timing correction, outlier detection for scrubbing using ART-based identification, registration of structural images, and segmentation. Removal of nuisance sources, including motion-related artifacts and other confounding signals (6 head motion parameters, 6 first-order temporal derivatives of the motion parameters, each of the 5 parameters obtained from a principal component analysis of the temporal components of white matter and ventricle signals, and ART-based scrubbing parameters for outlier volumes), was performed using linear regression. Band-pass filtering between 0.008 and 0.09 Hz and linear detrending were performed.
Morphological parameter computation using structural images was performed in the subject native space using the projection method of CAT12. These processes included surface-based spherical registration using the previously generated surfaces of the individual T1 images. fMRI data for each subject and cortical surface parcellation atlas were mapped to the individual surface using a weighted-mean method provided by the CAT12. The parcellation atlas was generated by Gordon et al 14 and included 333 cortical regions for 12 resting-state networks (VIS, visual network; PO, parieto-occipital network; SMD, dorsal somato-motor network; SMV, ventral somato-motor network; AUD, auditory network; CO, cingulo-opercular network; VAN, ventral attention/language network; SA, salience network; CP, cingulo-parietal network; DAN, dorsal attention network; FP, fronto-parietal network; DEF, default network; NON, none). In the study, motor-related networks were the SMD and SMV, and cognition-related networks were the PO, CO, VAN, SA, CP, DAN, FP, and DEF.
The connectivity strength of the resting-state network was measured by calculating Pearson’s correlation coefficients between the mean time courses across regions in the parcellation atlas. Within-networks consisting of connections within each resting-state network and between-networks consisting of connections between 12 resting-state networks were extracted from the brain network.
Statistical Analysis
The relationship between the FMA-UE recovery score (FMA-UE3m–FMA-UE2w) and strength of within- or between-networks obtained from resting-state networks at 2 weeks was investigated using multiple linear regression adjusting for age, sex, lesion side, and education level. The strength of the network quantified the average values of connectivity strengths in within-networks or between-networks. The relationship between the strengths of motor- and cognition-related networks were investigated, and the interaction effect between the strengths of motor- and cognition-related networks for motor recovery (FMA-UE recovery score − motor-related network × cognition-related network) was investigated using multiple linear regression. In cases of statistically significant interaction between networks for motor recovery, subgroup analysis was additionally conducted to investigate the effects of cognition-related networks according to the state of the motor-related network, which is our main interest. Patients were divided into 54 (50%) with higher strength of the motor-related network and 54 (50%) with lower strength of the motor-related network, and the relationship between the FMA-UE recovery score and the strength of cognition-related network was investigated in each subgroup. These results were derived using the statistics toolbox of MATLAB R2022a, where
Results
To investigate resting-state networks related to motor recovery, brain networks consisting of 333 regions for 12 resting-state networks were extracted in 108 stroke patients and are displayed in Figure 2A. Figure 2B shows the relationship between the strength of within- and between-networks and motor recovery. Node sizes are proportional to the contribution of within-network connections for motor recovery. Edge thicknesses are proportional to the weighting of between-network connections for motor recovery. Grayed nodes indicate non-significant within-network relationships. Strength of within-network associations, such as those of the SMD, SMV, AUD, CO, SA, CP, DAN, and DEF, showed a significant positive association with motor function recovery for 3 months. Also, strength of between-networks associations of SMD and SMV, SMD and DEF, SMV and FP, AUD and VIS, AUD and CP, AUD and CO, CO and DEF, and DAN and DEF demonstrated significant positive association with motor recovery for 3 months. Specifically, non-motor networks as well as motor networks were involved in motor recovery.
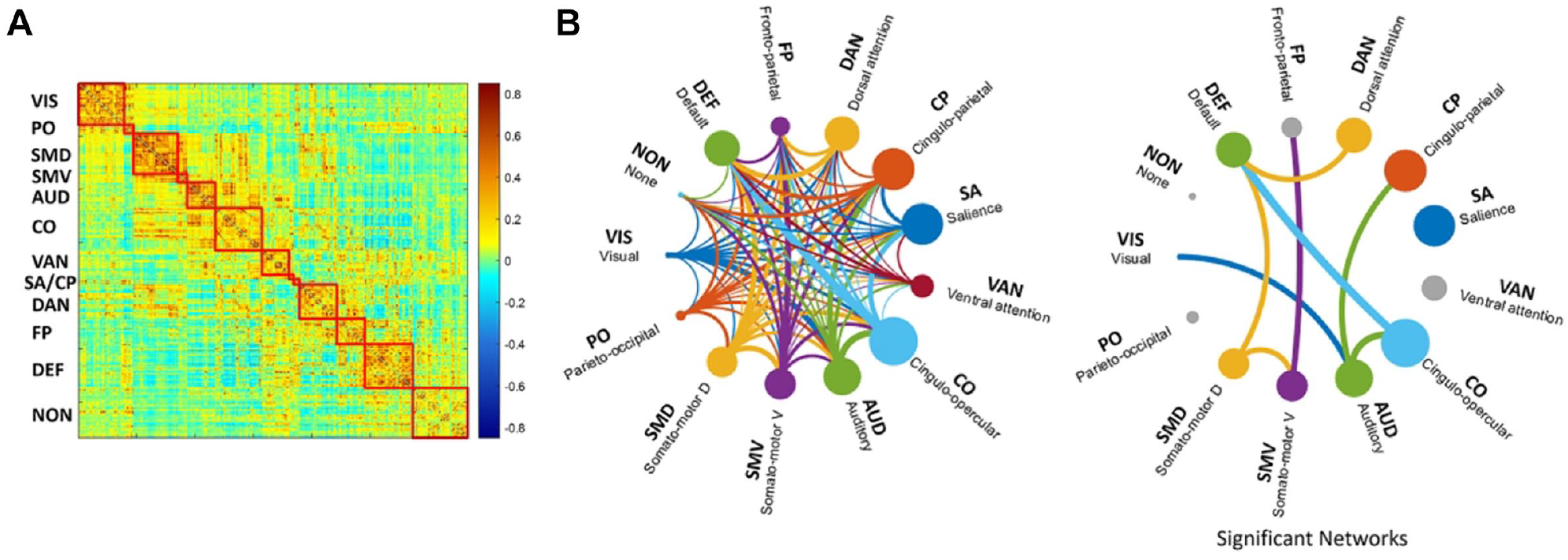
(A) Resting-state network with 333 cortical regions for 12 networks. (B) Brain networks associated with upper motor function recovery. Node sizes are proportional to the contribution of within-network connections, and edge thicknesses are proportional to the contribution of between-network connections for motor recovery. Gray nodes indicate not significant within-networks.
The association relationship between motor- and cognition-related within-networks is shown in Figure 3A. Motor-related networks, including SMD and SMA, and cognition-related networks, including VAN, SA, DAN, FP, and DEF, were associated. The strength of motor-related networks and strength of cognition-related networks were positively correlated at 2 weeks post-stroke.
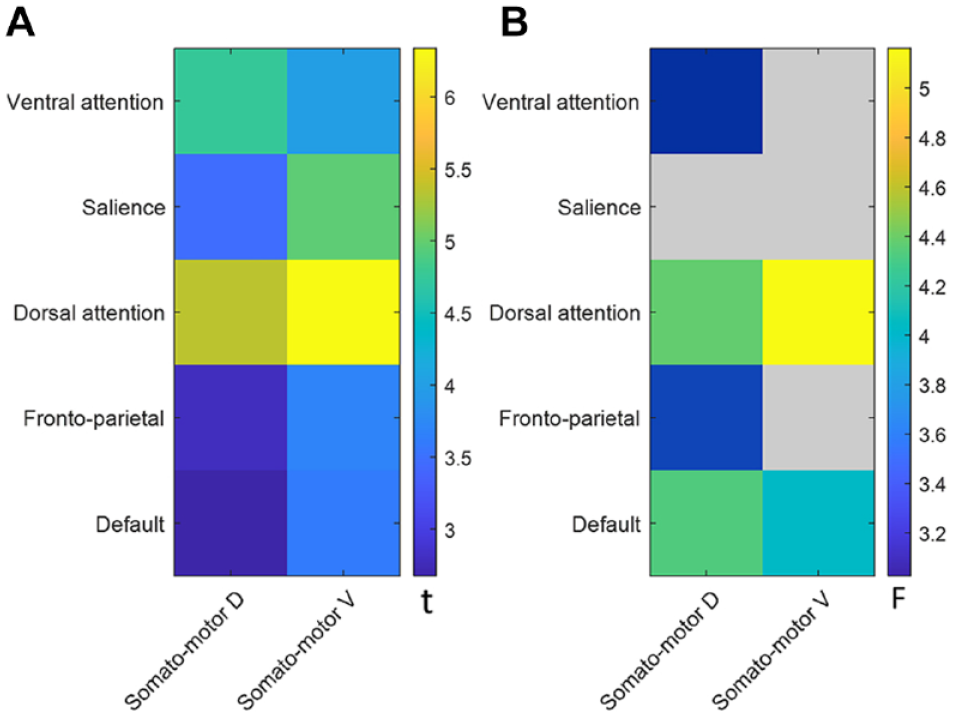
(A) Association relationships between strengths of motor- and cognition-related within-networks. (B) Interaction effect between strengths of motor- and cognition-related within-networks for upper motor function recovery. The colored bar indicates the statistical value, and gray color indicates lack of statistical significance.
To identify the effect of a cognition-related network state depending on motor-related network state for motor recovery, the interaction effect between motor and cognition-related within-networks for motor recovery were investigated in Figure 3B. Many pairs of motor- and cognition-related networks were significant. VAN, DAN, FP, and DEF depended on SMD state for motor recovery. DAN and DEF also depended on SMV state for motor recovery. When adjusting age, sex, lesion side, and education levels, all pairs were still significant except for DEF and SMV. All probability values are listed in Supplemental Table 1.
Based on the interaction effect, subgroup analysis was conducted by dividing all participants into 2 equal groups according to the strength of SMD (cut-off value of 0.361) or SMV (cut-off value of 0.420). Figure 4A and B show the relationship between the cognition-related networks and motor recovery in 54 patients with a lower strength of SMD and 54 patients with a higher strength of SMD. DAN, and DEF were significantly associated with motor recovery in patients with a lower strength of SMD (DAN,
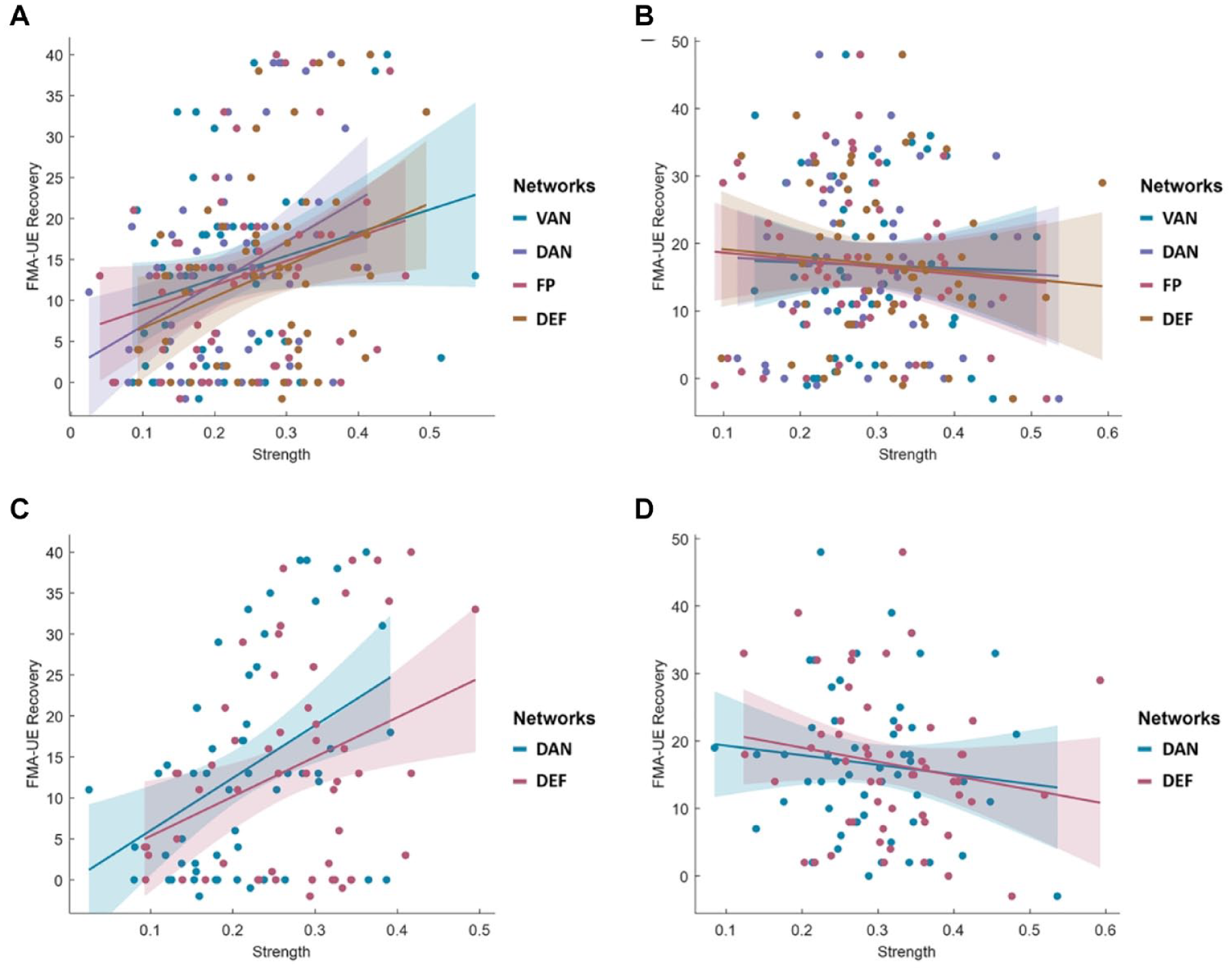
The relationship between cognition-related networks and upper limb motor function recovery depending on motor-related network states. (A) Relationship between cognition-related networks and motor recovery in 54 patients with lower strength of SMD. (B) The relationship in 54 patients with higher strength of SMD. (C) The relationships between cognition-related networks and motor recovery in 54 patients with lower strength of SMV. (D) The relationship in 54 patients with higher strength of SMV. DAN and DEF were significantly associated with motor recovery in patients with lower strength of SMD and SMV.
Discussion
In this study, we investigated large-scale networks associated with motor recovery, the relationship between motor- and cognition-related network states, and the effects of cognition-related network states depending on motor-related network states for motor recovery using neuroimaging data of subacute stroke patients.
The strength of motor- and cognition-related within-networks was associated with motor recovery. The strength of between-networks, for example, between motor- and cognition-related networks, was also associated with motor recovery. These findings show that not only motor network states, but also cognition-related network states contribute to motor recovery. Several stroke studies have reported that higher cognitive function at admission predicted better motor outcome.10,15,16 In a motor task-based fMRI study of stroke patients, neural correlates of motor recovery were found in several areas of the prefrontal cortex (PFC) as well as motor-related regions such as the primary motor cortex, supplementary motor area, and premotor cortex. 17 The dorsolateral prefrontal cortex is one of the essential regions in motor learning. 18 Cognition-related regions particularly play an important role in the early phase of motor learning. Patients with prefrontal lesions showed a significant reduction of motor learning according to previous studies.19,20 In diffusion tensor imaging study of stroke patients, the integrity of some tracts correlated with both motor and cognitive states. 21 Considering the functional relationship through previous studies mentioned above, motor function cannot be separated from cognitive function. In this respect, it is reasonable for cognition-related networks to be involved in motor recovery in our large-network study.
The strengths of motor-related networks and cognition-related networks were positively correlated each other in subacute patients. There are strong connections between cognition and motor-related regions. PFC is connected with secondary motor areas such as the premotor cortex and supplementary motor area.22,23 Also, PFC is interlinked with the posterior parietal cortex, anterior basal ganglia, and cerebellum, which are involved in motor function, through corticostriatal and cortocerebellar loops.18,24,25 From the connection structure perspective, the functional deficit can influence remote areas through the connections between motor- and cognition-related regions, even when the damage of brain is a focal lesion.26,27 Therefore, motor- and cognition-related network states may be influential to each other after stroke onset.
To identify the effect of a cognition-related network state depending on motor-related network state for motor recovery, the interaction effect between motor- and cognition-related networks for motor recovery was investigated. The characteristics of the interaction effect revealed that the strength of the cognition-related network was strongly associated with motor recovery in patients with lower strength of the motor-related network but not in patients with higher strength of the motor-related network. These results support that use of cognition-related neural resources for motor recovery occurred in patients with a severe deficit of motor-related neural resources after stroke onset.
According to meta-analyses on 54 experimental contrasts for movement of the paretic upper limb in stroke patients, 28 the inferior frontal cortex, which is an important region in the attention system,29 -32 was additionally involved in the movement of the upper limb in stroke patients but not in healthy individuals. Also, supplementary regions and pathways played an important role in motor recovery in patients with severe motor impairment and CST injury.33,34 The human brain seems to compensate for its deficiencies using other resources when the primary resources of a specific function are significantly lacking. In our large-scale network study, cognition-related networks supported the motor recovery in patients with more disrupted motor-related networks.
This study has several limitations in terms of study design. In our study, the effect of the cognitive network depending on motor network state was investigated for motor recovery. Another possible approach to examine the relationship between motor and cognitive networks for functional recovery is to verify the 2-way hypotheses (the effect of the cognitive network depending on motor network state for motor recovery and the effect of the motor network depending on cognitive network state for cognitive recovery) using extended function data. Also, our results may have been affected by the more robust general organization of the whole brain. To compensate, further multicenter prospective studies for acquisition of additional data are needed, and a study using causality analysis based on brain signals between motor- and cognition-related networks needs to be conducted.
In conclusion, our large cohort of subacute stroke patients showed that cognitive networks were involved in motor recovery, especially in patients with more highly disrupted motor networks. These results can be interpreted as the greater the damage to the motor network caused by stroke is, the more important the cognitive network is in motor recovery. This study is meaningful to confirm neural network correlations related to motor recovery by neuroimaging study beyond the existing functional score-oriented studies. Furthermore, this study provides important insights into developing novel treatment strategy and recovery prediction in neurorehabilitation of subacute stroke patients.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231177604 – Supplemental material for Does a Cognitive Network Contribute to Motor Recovery After Ischemic Stroke?
Supplemental material, sj-docx-1-nnr-10.1177_15459683231177604 for Does a Cognitive Network Contribute to Motor Recovery After Ischemic Stroke? by Jungsoo Lee and Yun-Hee Kim in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank the Neuroplasticity Neurorehabilitation and Imaging laboratory members for their assistance. We also thank the staff of the Department of Radiology at Samsung Medical Center.
Author Contributions
J.L. contributed to the design and conceptualization of the study, data acquisition, analysis and interpretation of data, and drafting the manuscript. Y-H. K. contributed to the design and conceptualization of the study, analysis and interpretation of data, critical revision of the manuscript for important intellectual content, and final approval of the version to be submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT; No. RS-2023-00208884) and the Korea Medical Device Development Fund grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, the Ministry of Health & Welfare, the Ministry of Food and Drug Safety; KMDF-RS-2022-00140478).
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
