Abstract
Background
In recent meta-analyses, robot-assisted gait training for patients with multiple sclerosis (MS) have yielded limited clinical benefits compared with conventional overground gait training.
Objective
To investigate the effect of robot-assisted gait training for patients with MS on clinical outcomes through a systematic review and meta-analysis.
Methods
We searched for relevant studies in the PubMed, EMBASE, Cochrane Library, and Physiotherapy Evidence Database databases from their inception to April 7, 2022. We selected studies that (1) included participants with MS, (2) used robot-assisted gait training as the intervention, (3) included conventional overground gait training or another gait training protocol as control treatment, and (4) reported clinical outcomes. Continuous variables are expressed as standardized mean differences with 95% confidence intervals. Statistical analyses were performed using RevMan 5.4 software.
Results
We included 16 studies enrolling 536 participants. Significant improvement was observed in the intervention group, with low heterogeneity at the end of the intervention with regard to walking velocity (standardized mean difference [SMD]: 0.38, 95% confidence interval [CI]: [0.15, 0.60]), walking endurance (SMD: 0.26, 95% CI [0.04, 0.48]), mobility (SMD: −0.37, 95% CI [−0.60, −0.14]), balance (SMD: 0.26, 95% CI [0.04, 0.48]), and fatigue (SMD: −0.27, 95% CI [−0.49, −0.04]). The results of subgroup analyses revealed improvements in these outcomes for the intervention group using grounded exoskeletons. No significant differences were noted in all the outcomes between the groups at follow-up.
Conclusions
Robot-assisted gait training with grounded exoskeletons exerts a positive short-term effect and is an adequate treatment option for patients with MS.
Introduction
Multiple sclerosis (MS) is among the most common causes of progressive, nontraumatic disability. 1 MS is an autoimmune-mediated neurodegenerative disease of the central nervous system and is characterized by inflammatory demyelination with axonal transection; previously, MS was classified as an organ-specific T-cell-mediated autoimmune disease.1,2 The pooled incidence of MS across 75 countries is 2.1 per 100 000 person-years. 3 The classic presentation of MS includes afferent pupillary defects, impaired sensation, motor weakness, ataxia, and gait impairment. 1 Despite advancements in treatment strategies, current therapies only partially protect against the neurodegenerative component of MS. 1 An increasing number of studies have reported the clinical efficacy and disease-modifying potential of physical rehabilitation and have recommended physical rehabilitation to enhance the physical performance of patients with MS. 4
Robotics is an emerging technology that can be effectively used by the affected population to improve their physical activity and social participation. 5 Various types of robotic devices are available currently. The types of robotic devices are categorized by the motion they apply. For example, exoskeletons move joints, such as the hips, knees, and ankles, in coordination with the phases of gait, whereas end-effector robots move only the feet, which are often placed on a support (footplate). 6 Robot-assisted gait training is applied in combination with conservative physiotherapy or a treadmill system. 5 In robotic devices, a robotic orthosis provides a guidance force during ambulation, thus allowing patients to engage in the repeated practice of complex gait patterns at near-normal speed over a long period. 7
Robot-assisted gait training is applied for different categories of rehabilitation and provides promising improvements. 8 Sattelmayer et al 9 conducted a meta-analysis of 9 randomized controlled trials (RCTs) and concluded that robot-assisted gait training is not significantly more effective than conventional overground walking. Yeh et al 10 conducted a systematic review and meta-analysis of 10 RCTs and reported the limited improved efficacy of robot-assisted gait training compared with conventional overground gait training. Calabrò et al 8 conducted a systematic review of 17 studies and, on the basis of the 13 included RCTs, concluded that robot-assisted gait training may provide better nonmotor outcomes than conventional treatment. Robot-assisted gait training has attracted greater attention in these 2 decades with its greater availability in rehabilitation centers. 11 It may provide guidance for lower-limb movement during walking training. 12 Participants have been observed to perform exercises over longer durations with partial body-weight support. 7 Because numerous RCTs have been published in these 2 years, we determined that a study using recent data from electronic databases is warranted. Thus, we conducted a systematic review and meta-analysis of RCTs to provide comprehensive and updated data on the effectiveness of robot-assisted gait training. This study investigated the effect of robot-assisted gait training on not only gait-associated assessment but also the fatigue, quality of life (QoL), and activities of daily living of patients with MS.
Methods
This review was performed in accordance with the recommendations of Cochrane Handbook for Systematic Reviews of Interventions 13 and is reported following the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses. 14 This systematic review was registered in the International Prospective Register of Systematic Reviews database under the number CRD42022326110 on May 10, 2022.
We included RCTs with pilot or crossover study designs. The patient (P), intervention (I), comparison (C), and outcome (O) model was used to identify eligible studies as follows:
(P) involved participants with MS of any severity level;
(I) involved robot-assisted gait training as the intervention;
(C) involved either conventional overground gait training or other gait training protocol as the control treatment;
(O) involved clinical outcomes, namely, walking velocity, walking endurance, fatigue, balance, mobility, QoL, activities of daily living, spasticity, pain, and disease progression.
We excluded articles that were protocols, non–peer-reviewed articles, conference papers, and letters to the editor. Moreover, we excluded crossover studies that did not include a washout period. No language restriction was applied in our search strategy.
We searched electronic databases, namely PubMed, EMBASE, Cochrane Library, and Physiotherapy Evidence Database (PEDro). In our search strategy, we included terms related to both MS and robot-assisted gait training and equivalent terms (the search strategies are presented in the Supplemental Appendix). If available, RCTs were identified using the refined search function of the databases. Additional articles were identified by manually searching the reference lists of relevant articles. The databases were searched from their inception to April 7, 2022. Two reviewers independently evaluated the eligibility of all titles and abstracts, and disagreements were resolved through discussion. If necessary, a third reviewer was involved. Subsequently, the full texts of remaining articles were screened to determine the eligibility of the articles. Only RCTs that investigated the effects of robot-assisted gait training for patients with MS in terms of the aforementioned outcomes were included.
Two authors extracted data from each study by using a structured form, and the characteristics of all eligible studies are summarized in a table. The following data were extracted: (1) basic information of the qualifying studies (first author and publication date); (2) inclusion criteria of each study; (3) demographic, clinical, and treatment characteristics (eg, number and mean age of patients in the control and treatment groups); (4) treatment protocol, duration, and follow-up period; and (5) outcome measures. The means and standard deviations of outcome measures before and after the treatment for the experimental and control groups were extracted. If crucial data could not be extracted from an article, we sent an email to the corresponding author requesting the data.
The primary outcomes of this study are direct walking–related measures, including walking velocity and endurance. The secondary outcomes are fatigue, balance, mobility, QoL, activities of daily living, spasticity, pain, and disease progression. The data were extracted to determine the effect of robot-assisted gait training on patients with MS. Walking velocity was examined using the 10-m walk test, 15 20-m walk test, 16 timed 25-foot walk test 17 or gait speed 18 (measured using temporal and spatial parameters on a walkway equipped with a camera motion capture system). Walking endurance was measured using the 6-minute walk test, 19 3-minute walk test, 20 or 2-minute walk test. 21 Mobility was assessed using the timed up and go test 22 or the Rivermead Mobility Index. 23 Balance was examined using the Berg Balance Scale 24 or Tinetti Test. 25 Fatigue was evaluated using the Fatigue Severity Scale, 26 Modified Fatigue Impact Scale, 27 or Würzburger Erschöpfungsinventar bei Multipler Sklerose scale. 28 QoL was measured using Multiple Sclerosis Quality of Life-54, 29 36-item short-form health survey, 30 or RAND-36. 31 Each assessment included physical and mental health measurements. Spasticity was determined using the Modified Ashworth Scale. 32 The pain score was calculated according to either the Visual Analog Scale or the Medical Outcomes Study Pain Effects Scale. Disease progression was assessed using the Expanded Disability Status Scale (EDSS). 33 Finally, the activities of daily living was measured using the Functional Independence Measure 34 or modified Barthel Index. 35
The quality of the included studies was assessed using the PEDro scale, which is widely used for evaluating the risk of bias in RCTs. 36 PEDro scale scores provided by 2 assessors were compared, and differences were resolved through discussion with a third researcher. The ratings of PEDro scale items 2–11 are summed to obtain a total PEDro scale score ranging between 0 and 10. Scores of <4 are considered “poor,” 4–5 are considered “fair,” 6–8 are considered “good,” and 9–10 are considered “excellent.” 36 All articles were included in this review irrespective of the PEDro score.
Statistical analyses were performed using RevMan 5.4 software, which is provided by the Cochrane Collaboration (https://training.cochrane.org/online-learning/core-software-cochrane-reviews/revman/revman-5-download). Continuous data were determined as changes from measures at baseline. For studies not reporting standard deviations, the authors were contacted for raw data, or, if unavailable, the data were estimated by calculating correlation coefficients in accordance with instructions provided in Cochrane Handbook for Systematic Reviews of Interventions. 13 The results with P < .05 were considered statistically significant. We used the I2 test to objectively measure statistical heterogeneity, with I2 ≥ 50% indicating significant heterogeneity. 37 A random-effects model was used in this meta-analysis because of the various study methods used in the included articles. Continuous variables are presented as standardized mean differences (SMDs) with 95% confidence intervals (CIs). SMDs calculated using Cohen’s d were employed to measure the probable clinical meaningfulness of the relationships between variables in a population; an SMD of <0.2 indicated a clinically meaningless effect; an SMD of 0.2 to 0.5 indicated a small effect; an SMD of 0.5 to 0.8 indicated a moderate effect; and an SMD of >0.8 indicated a large effect. 38
We analyzed the outcomes at the end of the intervention and at follow-up. Grounded exoskeletons and wearable exoskeletons are different robot-assisted gait training devices that are utilized on treadmills and in real-world settings, respectively. Subgroup analyses were conducted using these 2 devices. A funnel plot was constructed to examine publication bias if the number of studies included in each analysis was more than 10.
The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach was used to measure the quality of evidence as confidence in effect estimates. 39 This method examines the quality of the publication based on the study design (randomized trials vs. nonrandomized design), risk of bias, inconsistency, imprecision, indirectness, and publication bias; size and trend in the effect are also considered. 39
Results
By the using search terms reported in the Supplemental Appendix, we initially retrieved 373 studies (72 studies from PubMed, 74 studies from Embase, 203 from Cochrane Library, and 24 studies from PEDro). Among these, 123 duplicates were excluded using EndNote 20. 40 Furthermore, 206 studies that did not meet the inclusion criteria were excluded after their titles and abstracts were screened. We screened the full text of the remaining 44 articles and determined that 28 studies did not fulfill our inclusion and exclusion criteria. Finally, 16 studies were included in our study.41 -56 One study was not included in our quantitative analysis because it did not provide information on standard deviation and we could not contact the author with the e-mail address they provided. 54 Supplementary Figure 1 presents the flowchart of article selection.
The selected studies included 281 and 255 patients in the intervention and control groups, respectively. All of the selected studies were RCTs.41 -56 A total of 13 studies were parallel RCTs,42 -53,56 and 3 were crossover studies.41,54,55 Two studies used a wearable exoskeleton,43,44 and the other 14 studies applied grounded exoskeletons.41,42,45 -56 Regarding primary outcomes, 10 studies reported walking velocity as an outcome,41,43,45,49,51 -53,55 -57 and 12 reported walking endurance.41,42,44 -46,48,49,51 -53,55,56 Regarding secondary outcomes, 8 studies reported mobility,43 -45,47,49,51 -53 8 reported balance,41,42,45,47,49,52,53,57 8 reported fatigue,42,43,45,48,49,52,54,57 7 reported QoL,41,42,45,49,53,54,57 7 reported disease progression,41,42,47,48,53 -55 3 reported pain,48,52,54 4 reported activities of daily living,41,47,48,53 and 2 reported spasticity.41,52 The training duration was less than or equal to 4 weeks in 7 studies,42,44,45,48,52,53,56 1 to 3 months in 8 studies,41,43,46,49,51,54,55,57 and more than 3 months in one study. 47 Training frequency was twice a week in 7 studies,43,44,46,49,51,55,57 twice to 3 times a week in one study, 53 3 times a week in 6 studies,42,45,47,48,52,54 and 5 times a week in 2 studies.41,56 All the participants in the control group were assigned to conventional overground gait training or other gait training protocols.41 -56 In the 3 crossover trials, participants were allowed to engage in individual activity without additional training protocols during the washout period.41,54,55 All the studies measured the outcomes at the end of the intervention.41 -56 Two,47,57 four,42,45,49,51 and one 53 study followed up the patients 1, 3, and 6 months after the intervention, respectively. Supplemental Table 1 lists the characteristics of the included studies.
Two reviewers independently evaluated the quality of the included RCTs by using a PEDro scale. 36 All PEDro scale scores for the included studies were between 4 and 8. On the basis of the scores, 6 studies were considered “fair,” and 10 studies were considered “good.” Supplemental Table 2 summarizes the results of the risk of bias assessment.
The results of our meta-analysis revealed low heterogeneity (I2 < 50%), with significant differences between the groups. At the end of the intervention, the experimental group exhibited better results for some primary outcomes, specifically walking velocity (SMD = 0.38, 95% CI: 0.15-0.60) and walking endurance (SMD = 0.26, 95% CI: 0.04-0.48), and for some secondary outcomes, specifically mobility (SMD = −0.37, 95% CI: −0.60 to −0.14), balance (SMD = 0.26, 95% CI: 0.04-0.48), and fatigue (SMD = −0.27, 95% CI: −0.49 to −0.04; Figures 1–5). The results of the subgroup analysis at the end of the intervention revealed significant differences in specific primary outcomes, specifically walking velocity (SMD = 0.42, 95% CI: 0.18 to 0.65) and walking endurance (SMD = 0.27, 95% CI: 0.03 to 0.50), and in some secondary outcomes, specifically mobility (SMD = −0.40, 95% CI: −0.64 to −0.15), balance (SMD = 0.26, 95% CI: 0.04 to 0.48), and fatigue (SMD = −0.30, 95% CI: −0.54 to −0.06) between the control group and the experimental group receiving training with grounded exoskeletons (Figures 1-5). The funnel plots of walking velocity and walking endurance appeared to be symmetric (Supplemental Figures 2 and 3). No significant differences were observed at the end of the intervention in the other secondary outcomes (physical or mental health–related QoL, disease progression, pain score, activities of daily living, and spasticity) between the groups (Supplemental Figures 4–-9). At the follow-up period, no significant differences were observed in all the outcomes between the groups (Supplemental Figures 10–18). Table 1 summarizes the results of all the analyses.
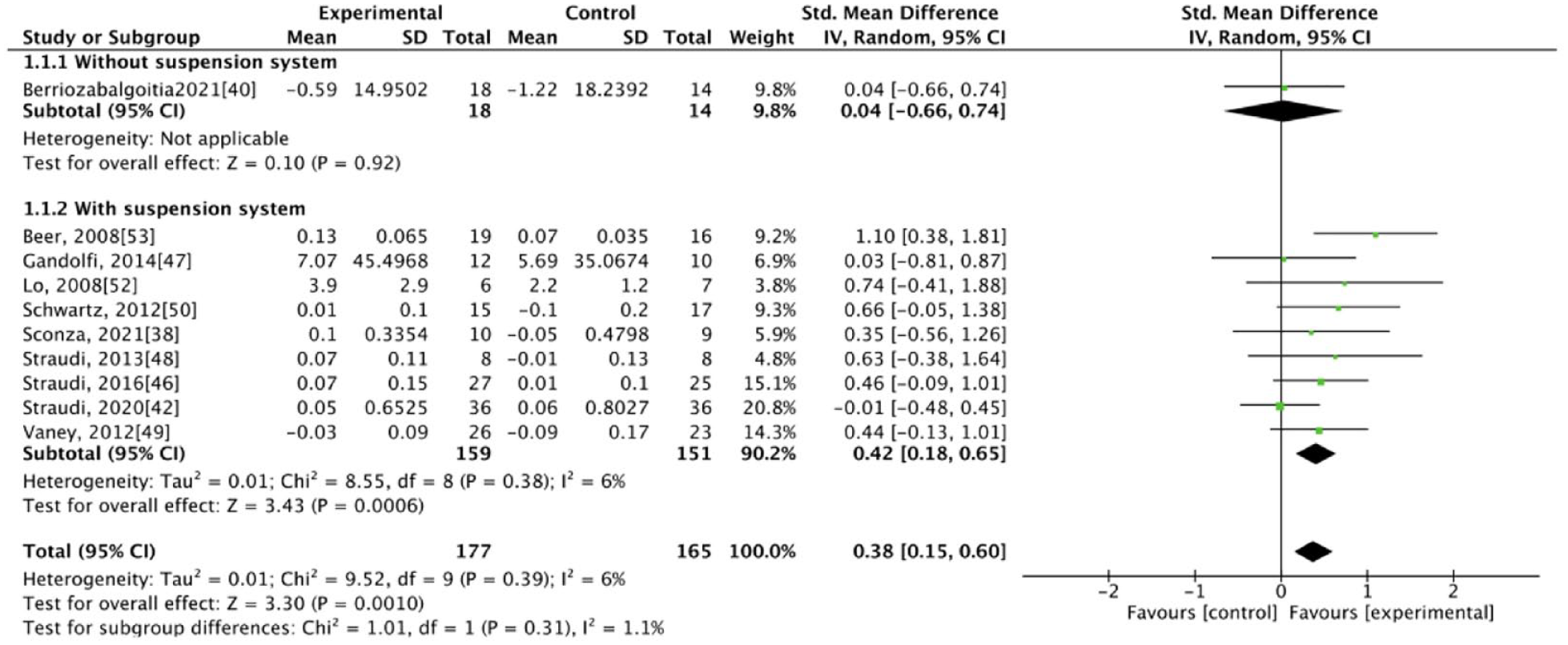
Forest plot for walking velocity at the end of intervention.
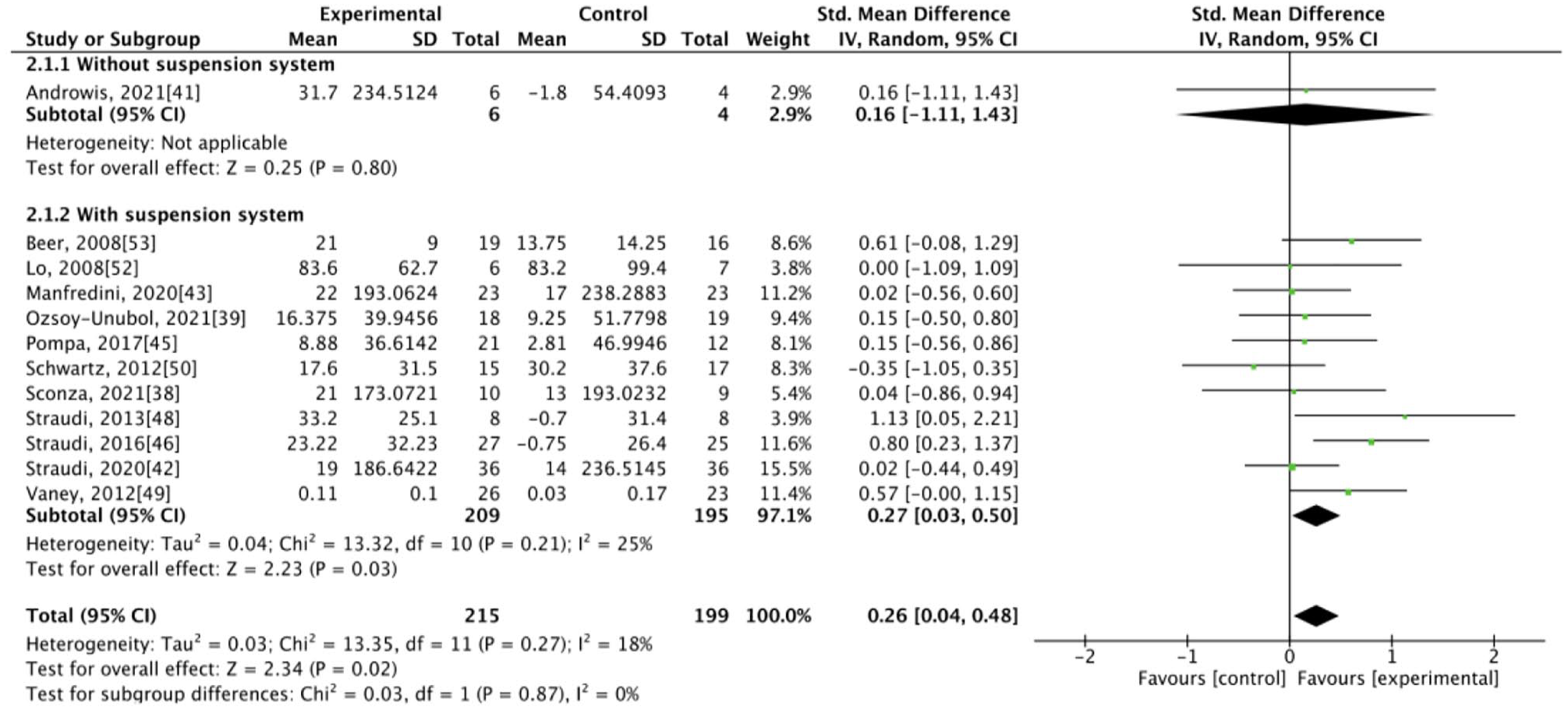
Forest plot for walking endurance at the end of intervention.
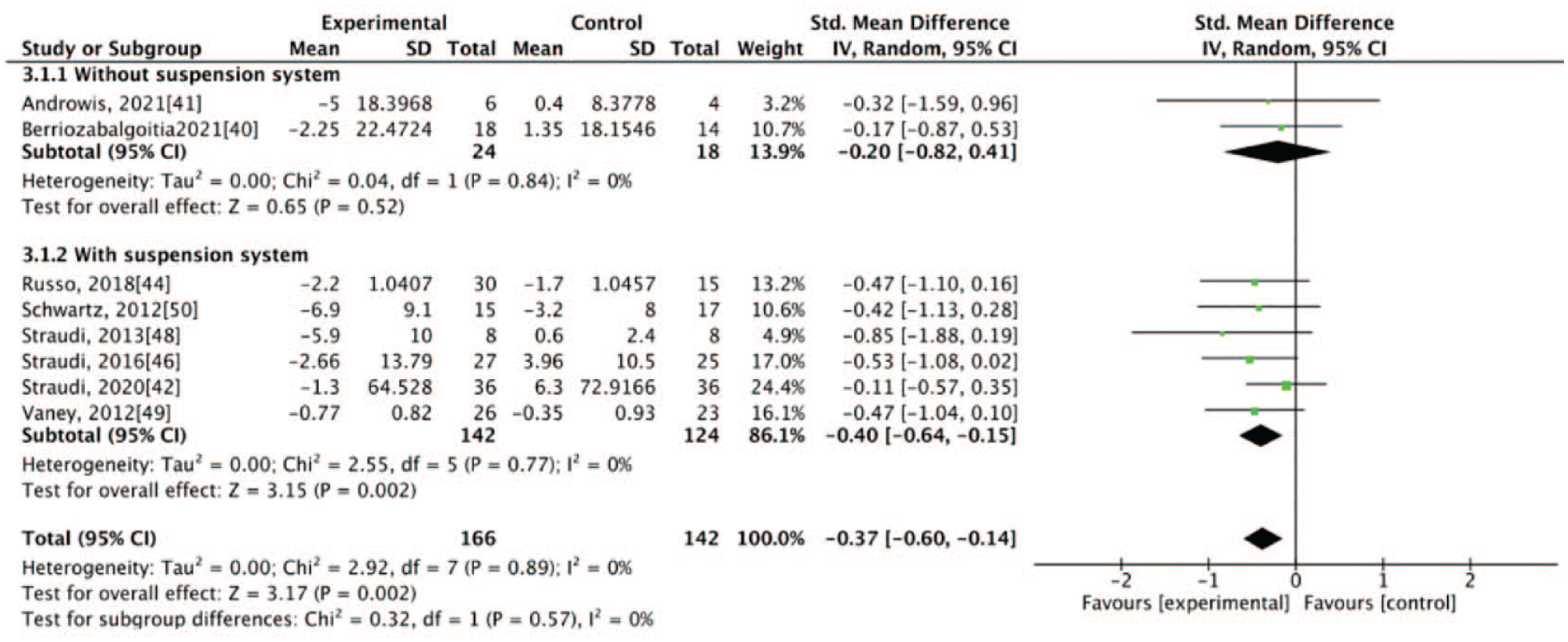
Forest plot for mobility at the end of intervention.
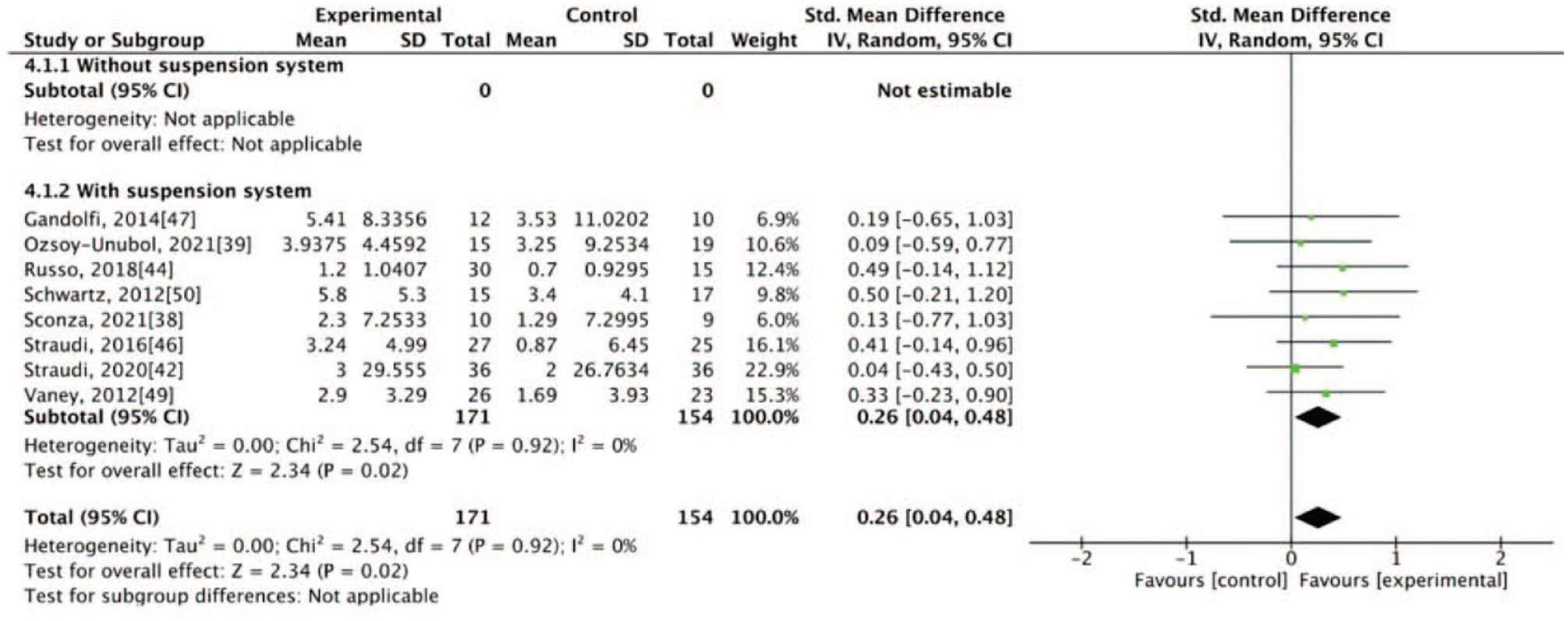
Forest plot for balance at the end of intervention.
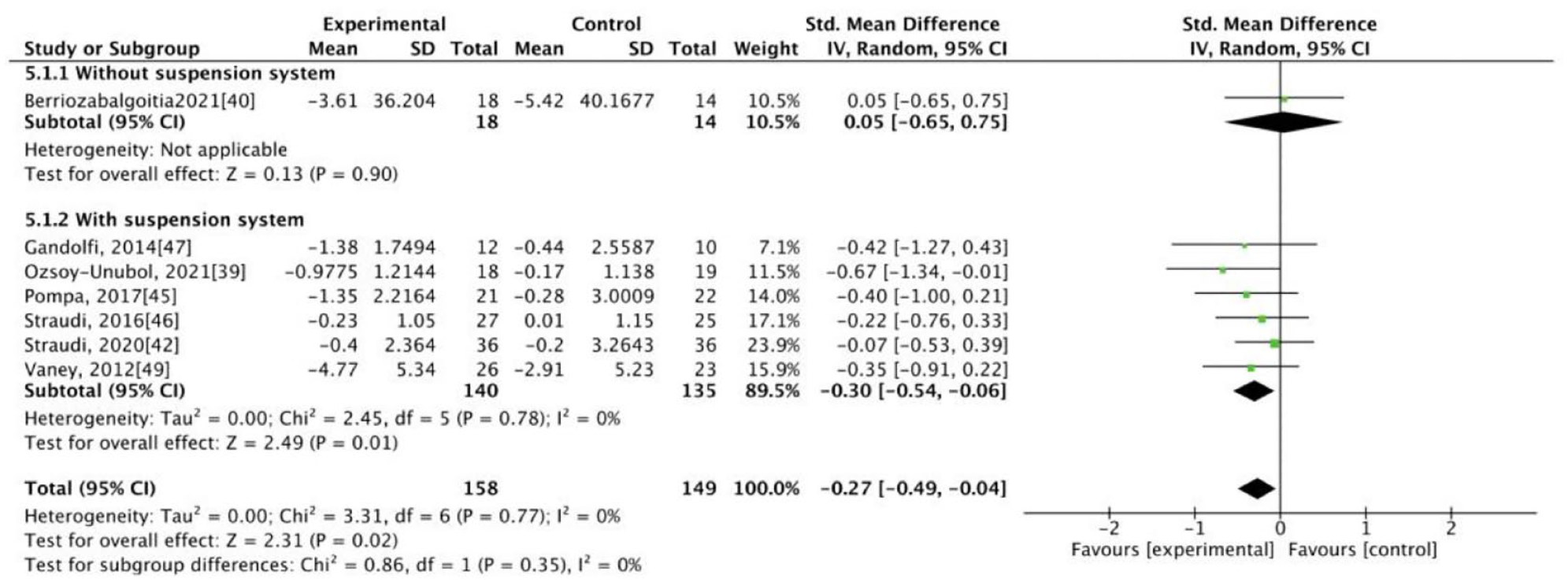
Forest plot for fatigue at the end of intervention.
Summary of the Results of Analyses.
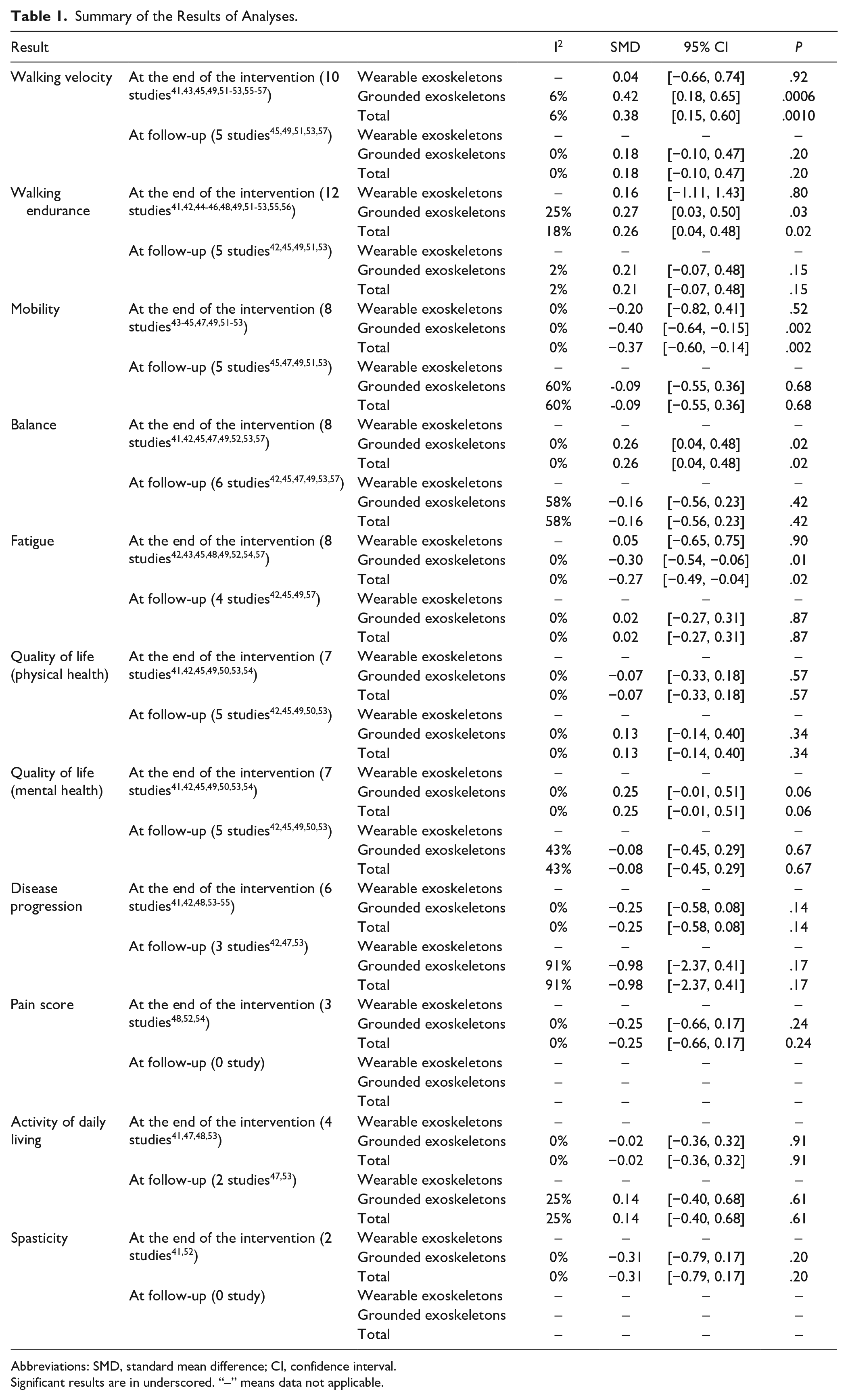
Abbreviations: SMD, standard mean difference; CI, confidence interval.
Significant results are in underscored. “–” means data not applicable.
Quality of evidence was determined using the GRADE approach, and we determined moderate quality of evidence for all the outcomes, except for pain and spasticity with low quality of evidence. Supplemental Table 3 presents details regarding the quality of evidence.
Discussion
MS is among the most common progressive nontraumatic disabling diseases affecting the activities of daily living of adults. 1 An increasing number of RCTs have evaluated the effectiveness of robot-assisted gait training in patients with MS. Thus, we conducted this systematic review and meta-analysis to provide comprehensive and updated data on the effect of robot-assisted gait training on not only gait-associated assessments but also the fatigue, QoL, and activities of daily living of patients with MS.
We presented the results with their statistical significance and clinical importance indicators. Compared with the control group, the intervention group exhibited significant improvement with low heterogeneity with respect to primary outcomes (ie, walking velocity and walking endurance) and some secondary outcomes (ie, mobility, balance, and fatigue). We measured probable clinical meaningfulness based on the effect size. All significant results were also clinically meaningful (SMD > 0.2). 38
The minimal clinically important difference (MCID) is the smallest change in a treatment outcome that an individual patient would identify as important and that would indicate a change in the patient’s management. 58 In addition to Cohen’s d, which was used to evaluate clinical meaningfulness, we also considered the anchor-based method. This method compares changes in either patient-reported or performance-based outcomes with a reference “anchor,” or an external measure of change. 59 Reference anchor values are only available for the 6-minute walk test (MD = 53.35), the Rivermead Mobility Index (MD = 2), and the Berg Balance Scale (MD = 6).23,60,61 Although all outcomes reached the MCID based on Cohen’s d, the analyses of outcomes from the included studies did not reach the MCID (MD = 15.26, 0.77, and 4.14, respectively) when the anchor-based method was used. This study did not employ the anchor-based method to measure the MCID for multiple reasons. First, the outcome measures of this study varied by assessment. Second, most of the outcomes focused on objective assessments. Third, the reference range of the MCID for each assessment is limited and not well-established. Furthermore, some published MCIDs in MS were not discussed in our article. Therefore, future studies can assess the MCID of the intervention based on anchor-based methods when complete reference values are available and leverage the advantages of both methods to make comparisons of clinical importance.
Robot-assisted gait training has been developed recently and has several advantages. First, this type of training provides guidance for lower-limb movements during walking training, enabling prolonged walking training with the afferent input of a normal gait pattern. 12 Second, extensive exposure within task-specific repetitive training promotes the reorganization of the primary motor cortex, thus improving clinical outcomes in patients with neurological conditions. 62 Furthermore, it enhances training-induced neuroplasticity by increasing input stimulation. 44 Third, robot-assisted gait training facilitates the repetition of specific and stereotyped movements to yield a correct and reproducible gait pattern in the conditions of balance and symmetry. 53 Walking can be regarded as a repeated sequence of balancing challenges that may be affected by postural control, complex dynamic interactions, and proprioceptive information. 50 With the aforementioned advantages, robot-assisted gait training could improve participants’ postural control and central integration of sensory input through repetitive sensorimotor balance training. 50 Furthermore, with improved balance, the participants required less effort to maintain a normal gait pattern; they exhibited improved tolerance and, thus, exhibited improvements with regard to fatigue.43,56
Numerous studies have investigated the effect of robot-assisted gait training on patients with MS. Calabrò et al 8 conducted a comprehensive systematic review of clinical outcomes and provided clinical recommendations. They included 17 studies, including both randomized and nonrandomized trials, in their review. They demonstrated the potential beneficial effects of lower-limb robotic devices on the clinical outcomes of patients with MS. 8 In particular, the beneficial effects of robotic training were similar to the matched dose of standard therapy, with greater benefits noted in patients with more severe disability. 8 Sattelmayer et al 9 investigated the effect of robot-assisted gait training on patients with disease-related disabilities using the EDSS. The results revealed that robot-assisted gait training was not significantly more effective than conventional overground walking training in patients with MS. Yeh et al 10 conducted a systematic review and meta-analysis of 10 RCTs and reported limited improved efficacy of robot-assisted gait training compared with conventional overground gait training. Our analysis result differs from that of previous studies in several respects. First, this analysis included only RCTs. Second, of the 16 included RCTs, 7 have not been analyzed by previous studies. Third, we conducted subgroup analysis with wearable exoskeletons and grounded exoskeletons, and our analyses indicated greater improvements with grounded exoskeletons. Although our results did not demonstrate the superiority of wearable exoskeletons, it improved the understanding of this novel intervention. Fourth, our analysis revealed significant improvement with low heterogeneity regarding walking velocity, walking endurance, mobility, balance, and fatigue at the end of the intervention; previous studies were unable to demonstrate the superiority of this intervention. Thus, our study provides a more updated and comprehensive review of the literature.
Grounded exoskeletons consist of a powered gait orthosis with integrated computer-controlled linear actuators, a body-weight support system, and a treadmill. 47 When combined with a suspension system, this type of training can partially support the body weight of a patient during training, thus reducing the burden on the lower limbs, safeguarding patients by reducing their fear of falling, and allowing for the practice of more steps, repeatability of a consistent kinematically-correct gait pattern, and less fatigue.12,52 Wearable exoskeletons allow individuals to walk on hard and flat surfaces in the real-world setting. 44 They provide gait assistance through computer-controlled actuators at lower-limb joints. 44 However, most wearable exoskeletons require user active trunk displacement to control an externally powered gait. 44 Wearable exoskeletons provide greater motor control stimulation and rich multisensory information (visual, proprioceptive, tactile, and vestibular). 63 Only 2 studies investigating the effect of a wearable exoskeleton were included in our analysis.43,44 In our subgroup analysis, no significant differences in all outcome measures were noted between the intervention group using wearable exoskeletons and the control group. This result may be due to the low number of studies using this intervention. Therefore, research focusing on wearable exoskeletons is warranted.
This study has numerous strengths. First, this systematic review and meta-analysis of RCTs is the most updated study to determine the effectiveness of robot-assisted gait training in patients with MS and to report positive results with clinical meaningfulness. Second, we analyzed the effect of robot-assisted gait training on patients with MS by focusing on not only gait-associated assessments but also fatigue, QoL, and activities of daily living. Third, the heterogeneity of our analyses was low for all the outcomes. Fourth, we searched for relevant RCTs according to our wide inclusion criteria using multiple major databases without language restrictions.
The literature we reviewed had some limitations. First, limited data on long-term follow-up was available. Second, because of the nature of the intervention, blinding participants and therapists is difficult. Third, the disease severity and the training protocol (training frequency and duration) differed among the studies; thus, we could not conduct an analysis because of these differences. Fourth, the number of studies focusing on wearable exoskeletons was very few for us to make conclusions. Furthermore, the 2 studies focused on wearable exoskeletons limited this intervention to overground walking in clinical settings. Future studies must evaluate the results with consideration of these limitations. Therefore, larger-scale, better-designed RCTs with long-term follow-up are warranted to overcome these limitations.
Conclusion
This systematic review and meta-analysis of current RCTs investigated the effect of robot-assisted gait training on not only gait-associated assessments but also the fatigue, QoL, and activities of daily living of patients with MS. The results revealed that robot-assisted gait training, particularly with grounded exoskeletons, induced short-term therapeutic effects in these patients. Furthermore, these results were clinically meaningful. These findings indicate that robot-assisted gait training with grounded exoskeletons exerts a positive short-term effect and is an adequate treatment option for patients with MS. Additional RCTs with a longer follow-up period are required to determine the long-term effects.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231167850 – Supplemental material for Effect of Robot-Assisted Gait Training on Multiple Sclerosis: A Systematic Review and Meta-analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-nnr-10.1177_15459683231167850 for Effect of Robot-Assisted Gait Training on Multiple Sclerosis: A Systematic Review and Meta-analysis of Randomized Controlled Trials by Fu-An Yang, Chien-Lin Lin, Wan-Chien Huang, Hsun-Yi Wang, Chih-Wei Peng P. T. and Hung-Chou Chen in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
This manuscript was edited by Wallace Academic Editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
