Abstract
Background. Robot-assisted gait training (RAGT) has been suggested as an intervention to improve walking capacity in patients with multiple sclerosis (MS). Objective. This study aimed to evaluate whether RAGT (Lokomat) is superior to over-ground walking training in terms of quality of life, activity level, and gait. Methods. A total of 67 patients with MS with the Expanded Disability Status Scale (EDSS) 3.0 to 6.5 were randomized to walking or RAGT, in addition to multimodal rehabilitation. Primary outcomes were walking speed, activity level (estimated metabolic equivalent, metabolic equivalents [METs], using an accelerometer), and quality of life (Well-Being Visual Analogue Scale (VAS) and EQ-5D European VAS. Results. In all, 49 patients finished the interventions. Mean age was 56 years (range 36-74 years), mean EDSS was 5.8 (3.0-6.5), and the preferred walking speed at baseline was 0.56 m/s (0.06-1.43 m/s). Before rehabilitation, participants spent on average 68 min/d at an MET ≥3. The walking group improved gait speed nonsignificantly more than the RAGT; the upper bound of the confidence interval (CI) did not exclude a clinically relevant benefit (defined as a difference of 0.05 m/s) in favor of the walking group; the lower bound of the CI did exclude a clinically important benefit in favor of the Lokomat. Quality of life improved in both groups, with a nonsignificant between-group difference in favor of the walking group. Both groups had reduced their activity by 8 weeks after the rehabilitation. Conclusion. It is unlikely that RAGT is better than over-ground walking training in patients with an EDSS between 3.0 and 6.5.
Introduction
Walking capacity is most valued by persons with multiple sclerosis (MS). 1 Some researchers call walking the sixth vital sign. 2 Gait, coordination, and balance are affected even in people with only mild neurological signs. 3 Patients with MS typically walk slower with shorter stride length and longer double support phases than healthy subjects.4,5 Walking is associated with a higher energy expenditure in patients with MS compared with healthy persons. 6 There is an association between gait impairment and fatigue, 7 and fatigue hampers participation. 8
Rehabilitation can reduce fatigue and enhance walking by improving force, movement patterns (efficiency of the gait cycle), dynamic balance, and self-efficacy.9-11 Gait training on a treadmill, with or without body weight support, may offer advantages beyond over-ground training, in terms of patient safety, reduced fear of falling, more steps practiced, and repeatability of a consistent, kinematically correct gait pattern 12 and less fatigue. However, little evidence supports the efficacy for use in MS13,14 as well as spinal cord injury 15 and stroke, 16 compared with over-ground training. Indeed, well-designed trials of robot-assisted gait training (RAGT) with the Lokomat compared with equal intensity of more conventional training for patients with stroke and spinal cord injury have not revealed superiority of RAGT, and they even suggest a negative impact.17-19
Beer et al 14 reported on patents with MS (median EDSS [Expanded Disability Status Scale] score of 6.5, ranging from 6 to 7.5) with 3 weeks of training that included 5 robot-assisted sessions with the Lokomat (14 subjects completed the protocol) or 5 conventional gait training sessions (15 subjects) per week. No statistically significant between-group differences were found for walking speed or distance. However, the effect size for the walking speed was large (0.7) but the statistical power too low. Lo and Triche 13 found no significant differences in outcomes between RAGT and conventional therapy of equal intensity in a cross-over study (3 weeks of each intervention) with 13 MS patients with a mean EDSS of 4.9 (SD = 1.2). We assessed whether Lokomat RAGT would be superior to over-ground walking training in a larger trial, in terms of quality of life, activity level, gait characteristics, balance, and fatigue.
Methods
The trial was approved by the ethics committee of the Canton Valais, Switzerland and registered on http://www.controlled-trials.com/ISRCTN69803702.
Participants
Patients with diagnosis of MS confirmed by a specialist by the means of the McDonald criteria, 20 who were referred to the rehabilitation inpatient program by either a neurologist or family physician, independent of the trial, were consecutively invited to participate in the study. Inclusion criteria were as follows: men and nonpregnant women at least 18 years old with an EDSS 21 score ≥3 and ≤6.5 who were able to walk 14 m with or without assistive devices.
Exclusion criteria were an acute exacerbation of MS in the past 3 months, medical disorders preventing active rehabilitation (eg, cardiovascular disease, unhealed decubitus, orthostatic hypotension), severe osteoporosis, and strong scoliosis or leg length difference. All patients gave written informed consent.
Study Design
This randomized controlled trial was conducted in an in-patient setting at a rehabilitation clinic in Montana, Switzerland. Patients referred to 3 weeks of inpatient rehabilitation were contacted by phone three to two weeks before the start of the rehabilitation by the chief medical officer of the rehabilitation clinic and were invited to participate in the study. The medical doctor asked about walking capacity (“Are you able to walk outside?”) to provide information for stratification in the randomization.
A computer-generated randomization list was generated with stratification in 2 groups: (a) patients who reported to be able to walk outside the house and (b) patients who reported to be mainly restricted to the house because of difficulties walking outside. The allocation list was concealed with opaque envelopes. Patients were randomized to the walking group or the robot-assisted body weight–supported gait training (Lokomat).
Interventions
Both groups received standardized, but individually tailored, rehabilitation with about 2.5 hours of active therapy (individually and in groups) per day. Treatment consisted of strengthening exercises in a gym room, horseback riding, pool exercises (water temperature 20°C), and occupational therapy. During these therapies, walking was not trained. The Lokomat group received 9 sessions of 30 minutes of RAGT on a Lokomat (Lokomat, Hocoma, Volketswil, Switzerland). Training parameters (weight support, etc) were individually adapted. At the first Lokomat session, support was set at 50% of the body weight and was adapted on observation of the gait. The Lokomat motor guidance system 22 was first set at 100%, corresponding to a passive walk. The guidance was reduced as much as possible on observation of the gait pattern. Walking speed was regulated on observation of gait and was changed randomly to simulate normal gait. The walking group received 9 sessions of 30 minutes of walking in a group with a physiotherapist. Patient were trained to walk for 30 minutes in the gym room or sometimes outside on uneven ground with walking aids of their choice.
Measures
The Kurtzke Expanded Disability Status Scale (EDSS) 21 was used as an inclusion criterion and for the description of the sample.
Primary outcomes
Quality of life was measured at baseline and at discharge using the Well-Being Visual Analogue Scale (VAS; question 17 of the General Well-Being Schedule) 23 and the EQ-5D European VAS value set. 24 Activity was measured during 6 days before the rehabilitation, in the middle week of the 3-week inpatient rehabilitation, and 2 and 9 months after discharge from rehabilitation. Activity level was estimated with an ActiGraph single-axis accelerometer (ActiGraph, Pensacola, FL). Data were analyzed as (a) minutes over 3 metabolic equivalents (METs) with the Crouter-2 formula 25 and (b) as total movement counts (not steps) per day. The patients wore the ActiGraph on a hip belt. The first and last day of the measurements were discarded because we thought that patients might pay too much attention to the new device on the first and last day and therefore move more than usual. The median of 4 days was used for the analysis. More counts reflect more physical activity. Intensity of physical activity can be classified by METs as light (<3 METs), moderate (3-5.99 METs), and vigorous (>6 METs).26,27 As the energy expenditure per count is higher in patients with MS compared with that in healthy persons, 28 the time spent over 3 METs is underestimated. Therefore, we report counts per day in addition. Gait parameters: 10-m walking speed and 3-minute walking speed at baseline and at discharge. The 10-m walking speed has good clinimetric properties in patients with neurological problems. 29 Patients walked 10 m within the clinic at their usual (preferred) speed using their walking aid or physical assistance. Patients walked 3 minutes in the clinic along an 80-m hallway as fast as safely possible, using their walking aid or assistance if needed.
Secondary outcomes
Balance was measured with the 57-point (best) Berg Balance Scale. 30 Physical and cognitive fatigue were assessed at baseline and at discharge with the “Würzburger Erschöpfungsinventar bei MS.” The score ranges from 0 to 36 points in the cognitive fatigue subscale and 0 to 32 points in the physical fatigue subscale. Higher values indicate more fatigue. The questionnaire was developed in German language and had good psychometric properties. 31 General mobility was assessed with the Rivermead Mobility Index (RMI). 32 The RMI is reliable and responsive. 33 The RMI scores range from 0 (low mobility) to 15 (good mobility) points. Spasticity was assessed with the modified Ashworth scale (ankle plantar flexion and dorsal extension, knee extension and flexion, hip extension, flexion, and adduction), which has good psychometric properties. 34 Pain was assessed with a numeric rating scale from 0 to 10. 35 During the inpatient rehabilitation, adverse effects were documented by the health professionals who were in contact with the patients.
Statistical Analysis
This was a superiority trial with 3 equally important primary outcomes: quality of life, activity level, and gait. A sample size calculation with the assumption of a clinically important improvement of about 30 m 36 in the 3-minute walking test and a clinically important difference between the 2 intervention groups as 50% (ie, 15 m), a type I error of 5%, a power of 80%, 2-sided tests, and allowing for a dropout rate of 15% resulted in required 100 patients in each intervention group. We corrected for multiple testing with the Benjamini–Hochberg method. 37 In addition to the unadjusted analyses, we used regression analysis to adjust for baseline values of each outcome, age and the 3-minute walking distance. Because we evaluated activity level 2 weeks before the start of the rehabilitation, we had to assess eligibility based on a telephone call by the chief medical doctor. This procedure led to a high number of patients declining or being excluded after randomization, we decided not to impute missing values of these patients. Thus, this is not strictly an intention-to-treat analysis. 38
We performed 2 sensitivity analyses: (a) We excluded 1 patient who fell the day before the end of rehabilitation assessment because of her severe pain in the feet during the assessments and (b) we performed a per protocol analysis, where 2 patients who switched groups were analyzed as treated and not as allocated (results not presented). In addition, we performed a meta-analysis with our results and the results from previous studies evaluating robot-assisted training in patients with MS. The meta-analysis was performed in STATA version 11. We summarized the continuous outcomes with standardized mean differences (SMD, difference of final mean values across treatment groups divided by the pooled standard deviation). An SMD of 0.2 indicates an effect of the amount of 0.2 standard deviations (small difference), an SMD of 0.5 indicates a moderate difference, and an SMD of 0.8 indicates a large difference.
Results
Between April 2007 and January 2009, a total of 299 patients were screened and 67 were randomized (Figure 1). The median completed Lokomat treatments was 9 (minimum 6, maximum 10) and walking group sessions was 8 (minimum 7, maximum 10). Table 1 shows the baseline characteristics of the patients involved in the final “available data” intention-to-treat analyses.
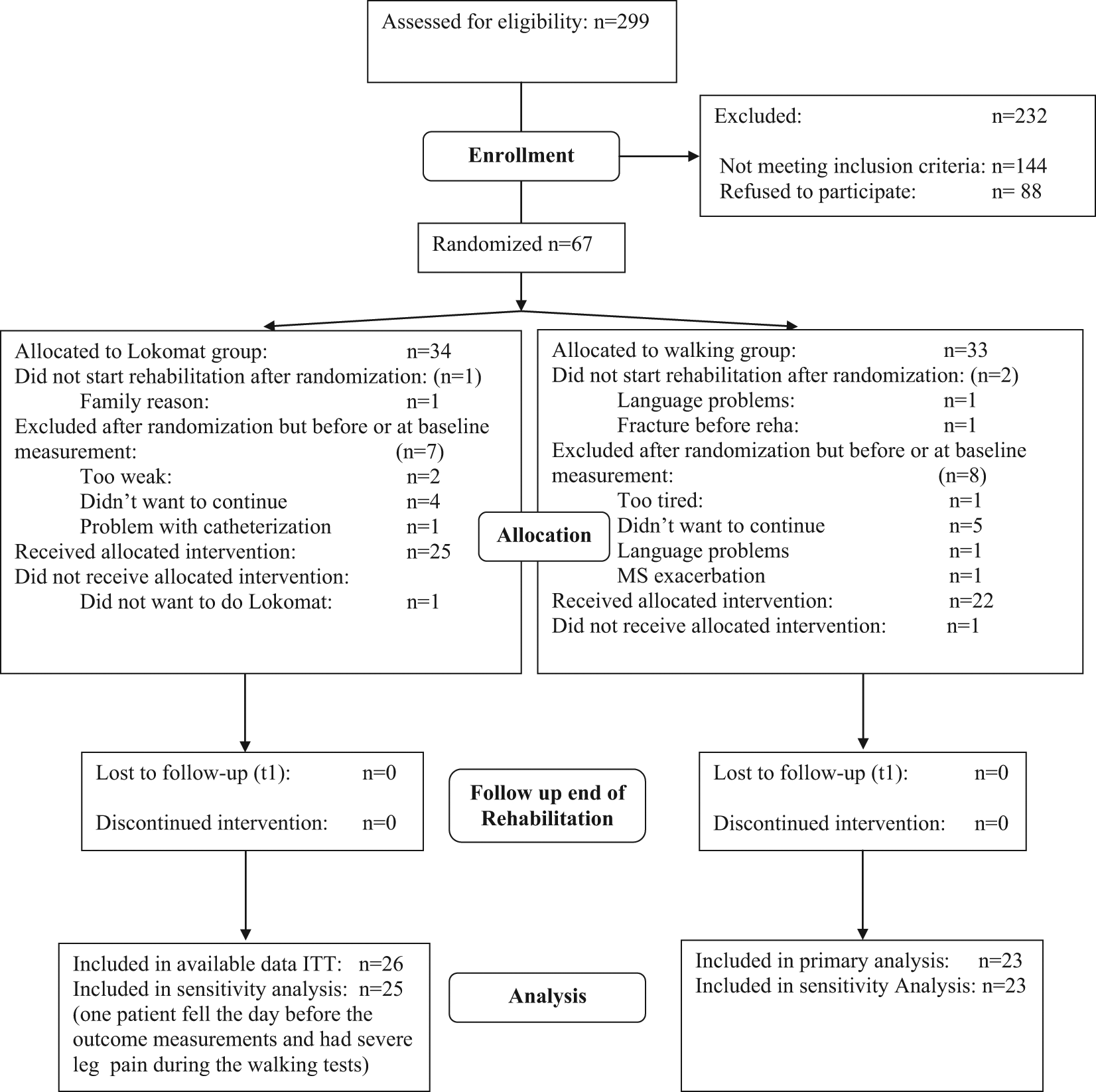
Flow chart with detailed description of recruitment, number of patients lost to intervention and outcome measurements. ITT = Intention to treat analysis (of available data). T1 = Follow-up at end of three week rehabilitation.
Characteristics of Patients at Baseline
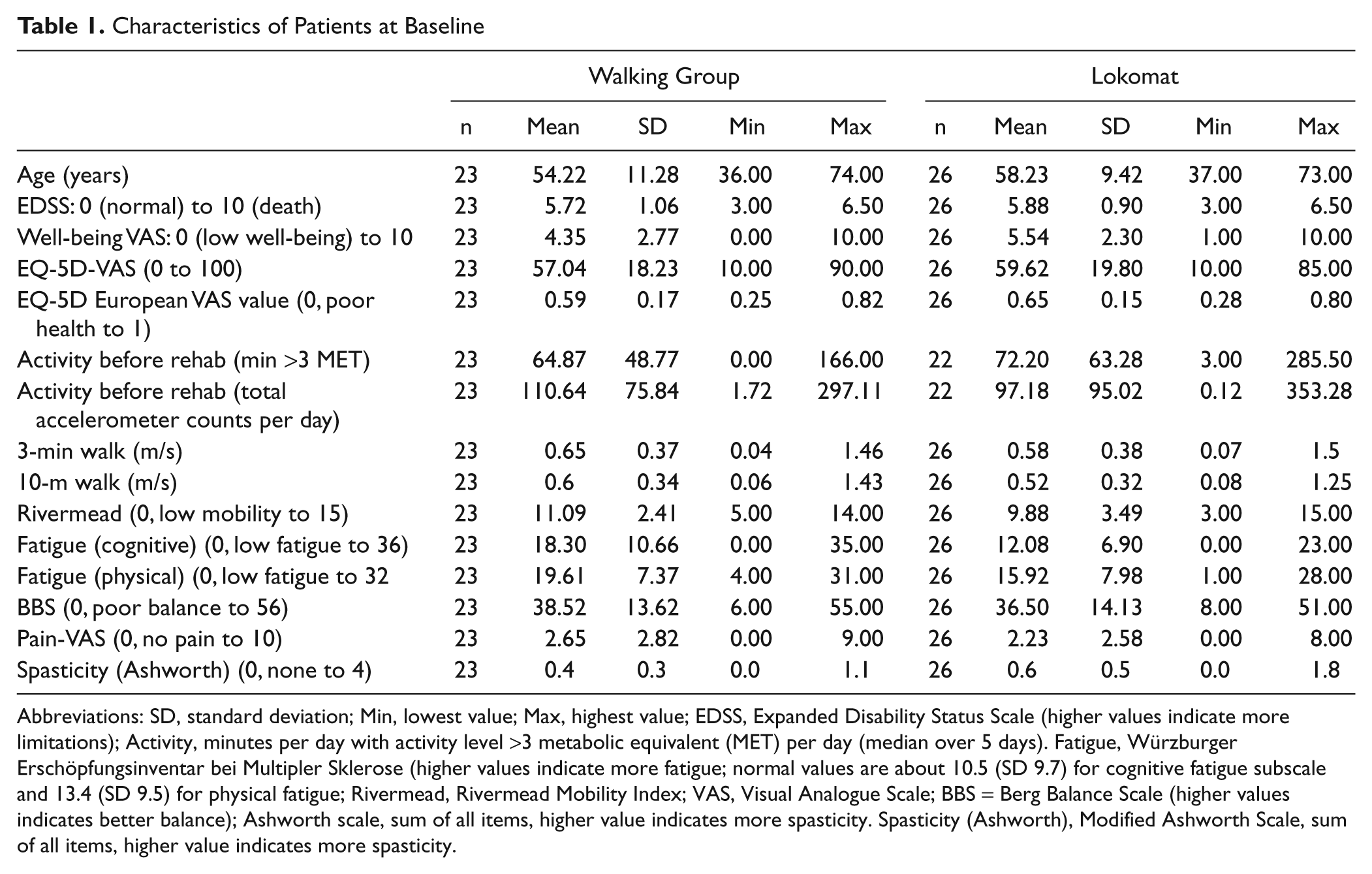
Abbreviations: SD, standard deviation; Min, lowest value; Max, highest value; EDSS, Expanded Disability Status Scale (higher values indicate more limitations); Activity, minutes per day with activity level >3 metabolic equivalent (MET) per day (median over 5 days). Fatigue, Würzburger Erschöpfungsinventar bei Multipler Sklerose (higher values indicate more fatigue; normal values are about 10.5 (SD 9.7) for cognitive fatigue subscale and 13.4 (SD 9.5) for physical fatigue; Rivermead, Rivermead Mobility Index; VAS, Visual Analogue Scale; BBS = Berg Balance Scale (higher values indicates better balance); Ashworth scale, sum of all items, higher value indicates more spasticity. Spasticity (Ashworth), Modified Ashworth Scale, sum of all items, higher value indicates more spasticity.
Quality of Life
Table 2 shows the between-group difference (walking − Lokomat) in the Well-being VAS as 0.79 points in favor of the walking group (95% confidence interval [CI] = −0.56 to 2.15) in the unadjusted analysis and −0.07 (95% CI = −1.17 to 1.04) in the adjusted analysis (adjusted for baseline values of the Well-being VAS, age, and 3-minute walking distance). Results were much the same in the sensitivity analyses, indicating that the change in the well-being did not differ statistically between the two intervention groups. The pre–post change in the rehabilitation period was statistically significant in both groups (walking group mean change of 1.87 points [95% CI = 0.79 to 2.95], RAGT mean change 1.08 points [95% CI = 0.18 to 1.98]). For the EQ-5D European VAS value set, the mean change in the 0 to 1 index in patients treated with RAGT was 0.08 (95% CI = 0.02 to 0.14) and in the walking group 0.06 (95% CI = 0.01 to 0.12). The between-group difference was 0.015 (95% CI = −0.09 to 0.06) and not significant.
Results of All Outcomes a
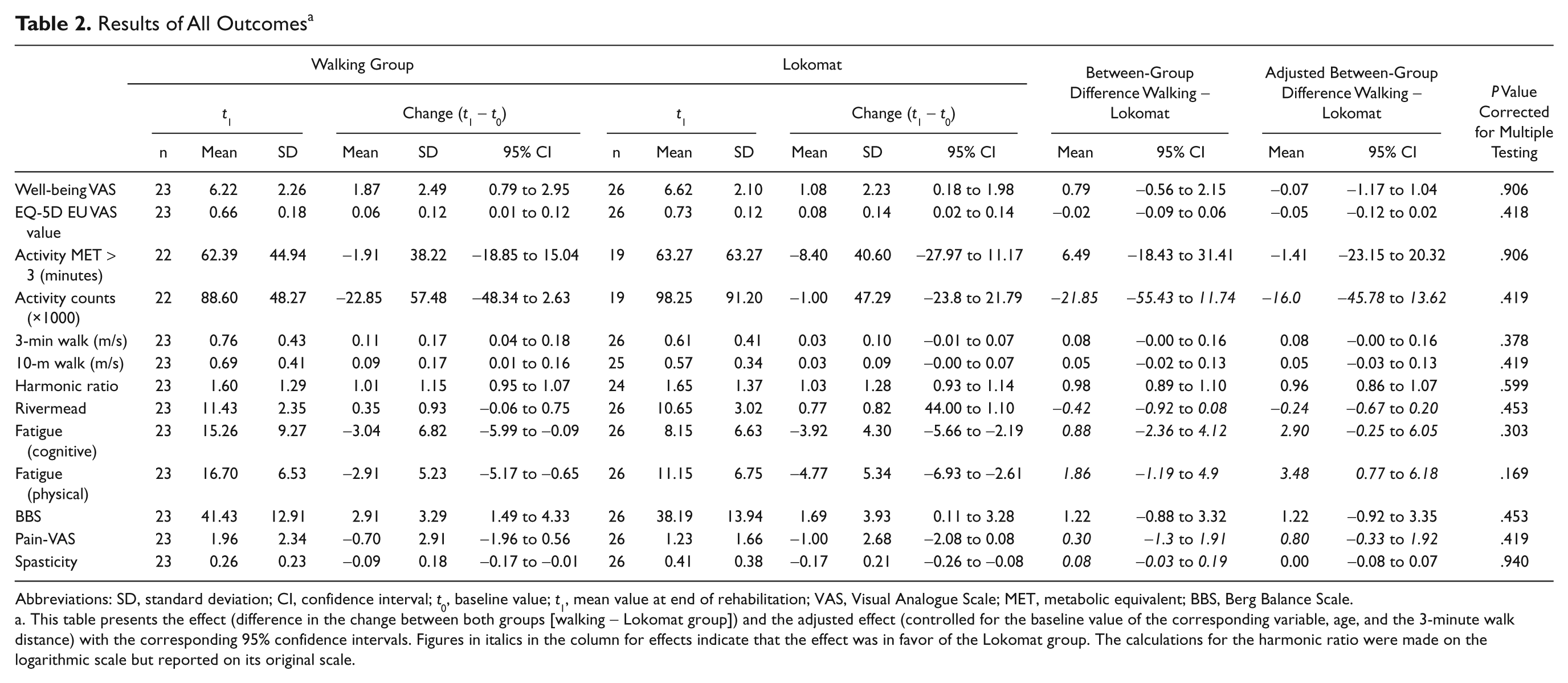
Abbreviations: SD, standard deviation; CI, confidence interval; t0, baseline value; t1, mean value at end of rehabilitation; VAS, Visual Analogue Scale; MET, metabolic equivalent; BBS, Berg Balance Scale.
This table presents the effect (difference in the change between both groups [walking − Lokomat group]) and the adjusted effect (controlled for the baseline value of the corresponding variable, age, and the 3-minute walk distance) with the corresponding 95% confidence intervals. Figures in italics in the column for effects indicate that the effect was in favor of the Lokomat group. The calculations for the harmonic ratio were made on the logarithmic scale but reported on its original scale.
Activity Level
Complete data were available only for 21 patients from the walking group and for 19 patients from the Lokomat group (because of technical problems, lost devices, and problems with data transfer). The between-group difference in the minutes per day that patients were active at the level of at least 3 METs was 6.49 minutes in favor of the walking group (95% CI = −18.43 to 31.41) in the unadjusted analysis and 1.41 minutes (95% CI = −23.15 to 20.32) in the adjusted analyses. There was no significant change in activity (measured 2 weeks before the start of the rehabilitation and 8 weeks after the rehabilitation). The alternative activity level measure, the total counts per day, showed a slightly different result with a small and nonsignificant advantage for the Lokomat group (Figure 3).
Gait
The between-group difference (walking group − Lokomat) in the 3-minute walking speed was 0.08 m/s in favor of the walking group (95% CI = 0.00 to 0.16) in the unadjusted analysis and 0.08 m (95% CI = −0.00 to 0.16) in the adjusted analysis (adjusted for baseline value of age and 3-minute walking distance. Results were much the same in the sensitivity analyses showing a slightly less pronounced advantage for the walking group. Considering all analysis, there is weak evidence that the walking group improved more in the 3-minute walking distance compared with the Lokomat group. The pre–post change in the rehabilitation period was statistically significant in the walking group with a mean change of 0.11 m/s (95% CI = 0.04 to 0.18), the Lokomat group improved by a mean of 0.03 m (95% CI = −0.01 to 0.07). In the 10-m walk, the between-group difference was 0.05 m/s (95% CI = −0.02 to 0.13) in favor of the walking group. The adjusted (for baseline values, age, and 3-minute walking distance) mean difference between the 2 groups was 0.05 (95% CI = −0.03 to 0.13) in favor of the walking group.
Table 2 and Figure 2 show the results of all outcomes. After correction for multiple testing, there were no significant between-group differences. There is only weak evidence for an advantage of either of the interventions.
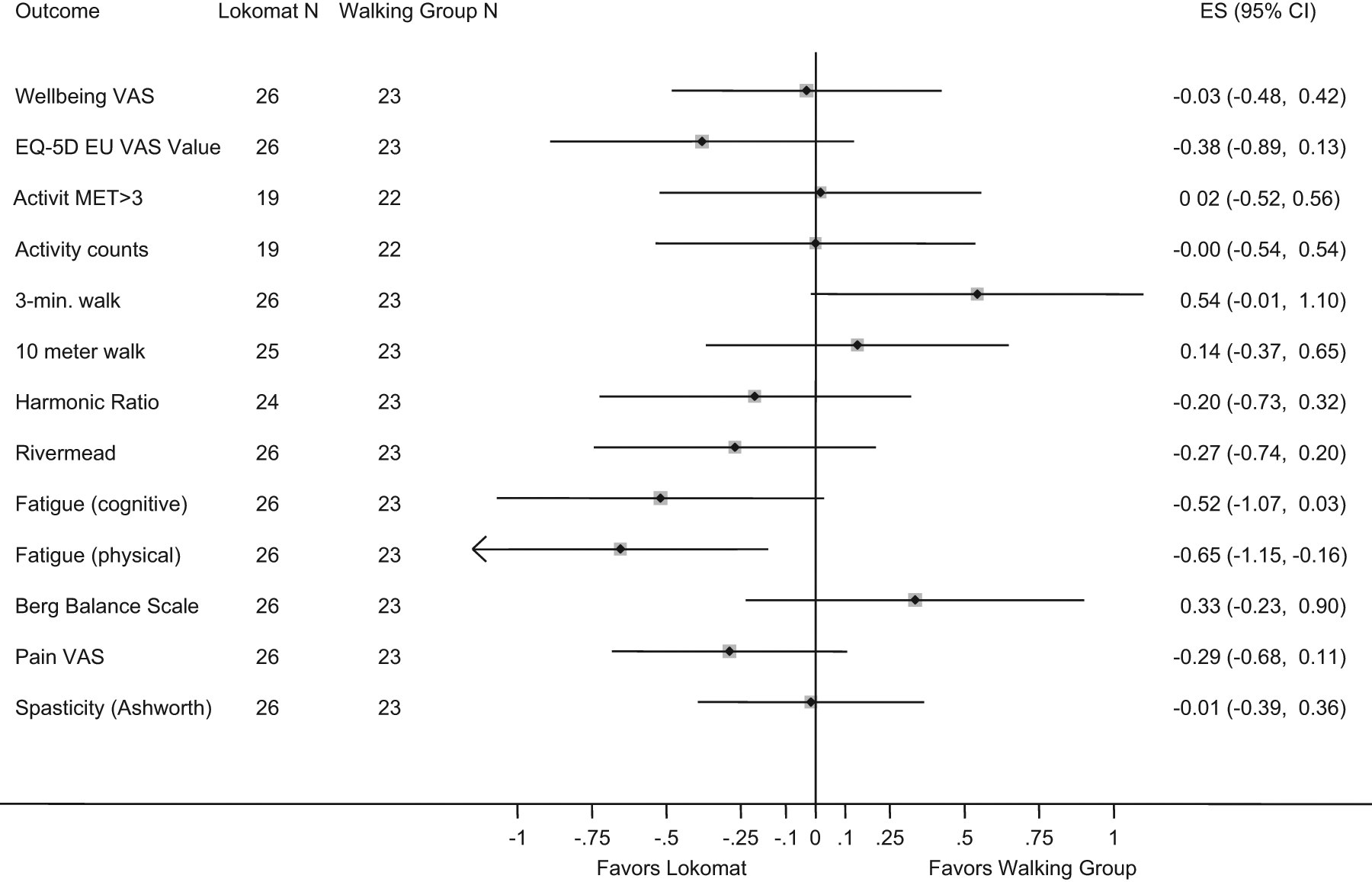
Adjusted between-group-differences expressed as effect sizes (Cohen’s d). The differences are adjusted for the baseline value of the given variable as well as for age and the 3 minute walk distance. The between group differences was divided by the pooled standard deviation of the group difference. Effect Sizes of 0.2 are classified as small effects, 0.5 as moderate effects and 0.8 as large effects. Note that the effect sizes on this figure are the effects between groups and not the pre-post effects.
Adverse events
Between the randomization and the start of rehabilitation, 1 patient broke his leg (unrelated to the study) and 1 patient had a fall the day before the final assessment unrelated to an intervention. During rehabilitation, 1 patient had an ankle sprain, 1 day before the final assessment. The ankle sprain was not related to any of the interventions. Apart some minor bruising from the straps, there were no adverse events for the Lokomat group.
Meta-analysis
Pooling the results from Beer et al 14 (35 patients, 19 RAGT and 16 conventional walking training), Lo and Triche 13 (13 patients, cross-over trial) and our study (49 patients, 26 RAGT and 23 walking group) resulting in a total number of 58 patients in the RAGT and 52 in the walking group, we found no evidence for an advantage of RAGT for the timed walk (25 feet, 10 or 20 m) with a pooled SMD of 0.01 (95% CI = −0.50 to 0.53) and for walking distance (3 or 6 minutes) with a SMD of −0.14 (95% CI = −0.50 to 0.50); Figure 4. However, heterogeneity was substantial (I2: 53.0% and 67.6%).
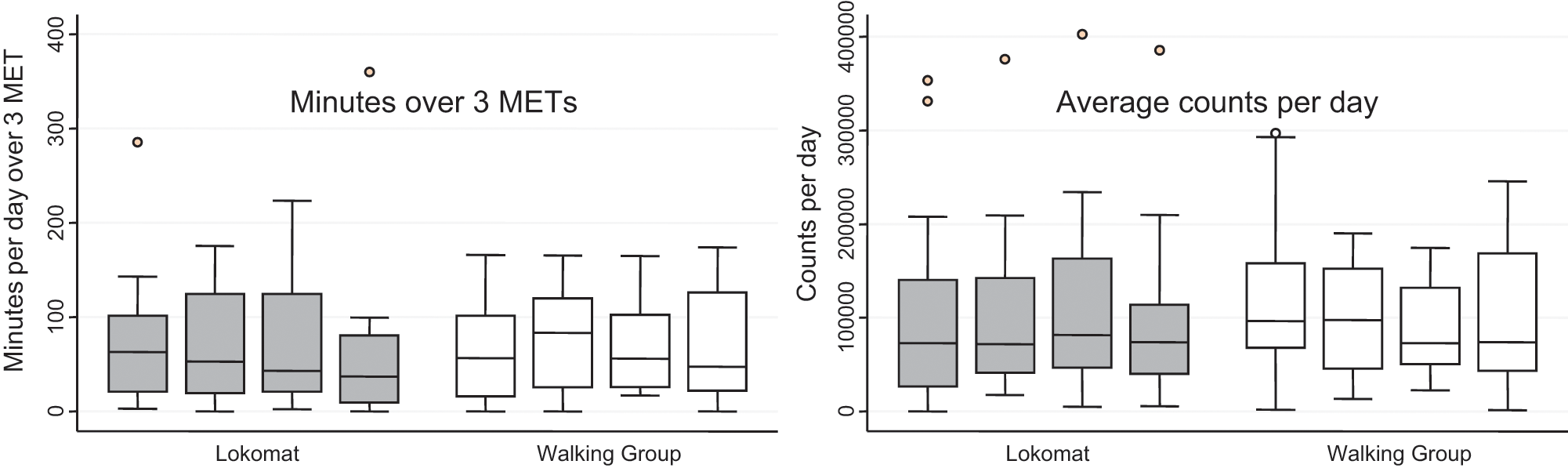
Boxplots of the number of minutes over 3 METS per day (left side) and the average counts per day (right side). The bold line in the box indicates the median (50th percentile), the lower boundary of the box indicates the 25th percentile and the upper limit of the box the 75th percentile. The box indicates the interquartile range (IQR). The whiskers indicate 1.5 times the IQR, i.e. the lowest or highest value still within 1.5 IQR. The points are extreme values, i.e. between 1.5 and 3 IQR from the boxes away. Complete data over the four time points (before the inpatient rehabilitation, during inpatient rehabilitation, 8 weeks follow-up and 9 months follow-up).
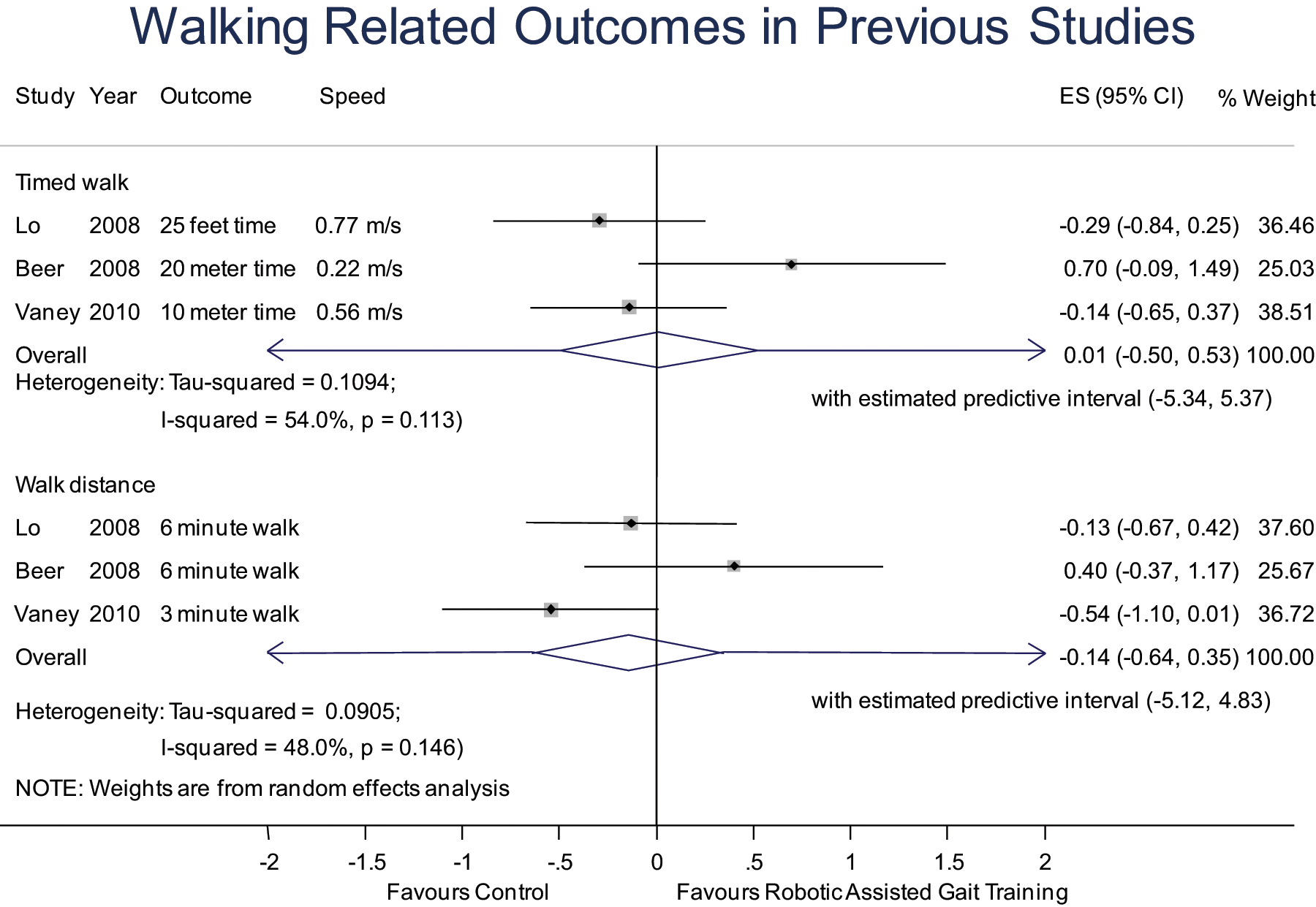
Pooled analysis of this study (Vaney 2010) with previous studies on robot-assisted gait training in patients with Multiple Sclerosis. All data are change values, expressed as standardized mean differences (cohend’s d). Predictive intervals can be interpreted as follows: 95% of the future study-effects are predicted to fall within the predictive interval. N =Total sample size; SMD = Standardized Mean Difference.
Discussion
We found no statistically significant difference in the effect of the RAGT compared with the over-ground walking group. For the improvement in walking speed, the between-group difference was in favor of the over-ground walking group: the difference in the means was 0.08 m/s, which is more than the minimal clinically important difference of 0.05 defined by Perera et al. 39 We can exclude any clinically important benefit for RAGT but we cannot exclude a substantial benefit for the over-ground walking group: The upper boundary of the 95% confidence interval, which delineates the most beneficial effect of the over-ground walking, corresponded to a difference in the walking speed improvement of 0.16 in favour of the over-ground walking group and thus did not exclude a substantial meaningful difference of 0.1 m/s, as defined by Perera et al. 39 The lower bound of the 95% CI, which delineates the most beneficial effect of RAGT compatible with the results of this study, corresponded to a difference in the improvement of −0.0000661 m/s in favor of the RAGT, thus (a) only marginally not significant in favor of the over-ground walking and (b) excluding a clinically important difference of 0.05 m/s in favor of the RAGT, as defined by Perera et al. 39
There was no significant between-group difference in the other primary outcome quality of life, but there was a small advantage for the over-ground walking group. However, the confidence interval did not exclude an advantage for the RAGT. For the activity level, there is only little evidence for a between group difference as the confidence intervals are very wide.
We found statistically significant within-group improvements in quality of life, 3-minute walking speed, fatigue, balance, and spasticity. There was a nonsignificant decrease in the activity level and no significant changes in the 10-meter walking speed, the Rivermead and pain level.
Although our study was underpowered based on prior literature to detect clinical important between-group differences (0.05 m/s), the results in Table 2 suggest that even if we entered 100 subjects in each group, we would not have found a clinically meaningful difference in favor of RAGT.
Our hypotheses that the additional robot-assisted gait training would improve gait more compared with the over-ground walking training could not be corroborated. Our data suggest that for patients with an EDSS score between 3 and 6.5, the over-ground walking is better than the robot-assisted gait training. We hypothesized a better effect of the robot-assisted training on quality of life than the over-ground walking, but our data reveal equality of these modestly provided interventions. Furthermore, our patients were less active after the rehabilitation compared with before.
It is possible that patients with a higher EDSS, that is, more walking disorder, will benefit more from RAGT; this is supported by the meta-analysis, where the large effect size in favor of the RAGT was seen in the study with the slowest walking speed (Beer et al 14 with 0.22 m/s, compared with 0.77 m/s (Lo and Triche 13 ) and the 0.55 m/s of our study. However, this is not strong evidence for a better effect in patients with lower walking speed, because the difference in the walking speed was not the only difference between the studies, as the volume of RAGT was different too (6 sessions in Lo and Triche and 9 sessions in our study versus 15 sessions in the study by Beer et al).
The activity level of our sample was high compared with patients with musculoskeletal problems 40 but was lower than the activity level reported in patients with MS with slightly lower EDSS score (ie, less walking impairment). 41 The fact that we found an improvement in mobility (Rivermead) after the rehabilitation period, but that the activity level 8 weeks after discharge was identical to the level before rehabilitation, can be explained as a drop in performance in the light of a chronic progressive disease or by clinic testing that does not necessarily reflect the reality of every day mobility.
We aimed to include 200 patients but did not receive enough eligible referrals. Our trial, then, was only slightly larger than prior ones. This again led to low power. One limitation of this study was the baseline differences between the 2 intervention groups in walking speed, which shows that our stratification strategy with the self-reported ability to walk outside the house, assessed with a phone call, was not sufficient. But as we wanted to assess the activity level before patients came to the rehabilitation clinic we had to randomize before we could measure walking speed. Other limitations were the low number of walking sessions over 3 weeks, and the lack of specific sessions where practice on the Lokomat was translated back and forth with over-ground walking.
The study supports the assumption that this form of RAGT, in which a “normal” symmetrical gait pattern is imposed, does not allow patients adaptive motor control in which they lean to deal with existing synergistic-dependent motor control and perhaps optimal motor learning, in contrast to usual physical therapy care.
Conclusion
Our study suggests that patients who can walk 10 m in less than 16 seconds do not need to practice on the Lokomat but should practice over ground. Patients with more severe walking impairments will have to be studied in an appropriately designed trial to determine whether they may benefit from RAGT.
Footnotes
The funders had no role in study design, data collection, analysis, and interpretation of the data or the decision to publish and preparation of the article. The corresponding author had full access to all data of the trial and had final responsibility for the decision to submit for publication.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was supported by a grant from the “fonds stratégique HES-SO”/RéSaR to Roger Hilfiker, Véronique Lugon-Moulin, and André Meichtry, and by intramural funds from the Berner Klinik Montana to Dr Claude Vaney, Brigitte Gattlen, and Rita Hausammann, and from University of Applied Sciences Western Switzerland, Institut Santé Social, to Roger Hilfiker.
