Abstract
Introduction
Despite optimal medical and surgical treatment, patients with Parkinson disease (PD) develop progressive disability.1,2 Gait hypokinesia is one of the most important determinants of disability and reduced quality of life, especially in those with mild to moderate PD.3,4
The current management of gait disturbances in PD relies mainly on pharmacological treatment and functional neurosurgery. In addition to these treatments, several nonpharmacological therapies, such as task-oriented exercises, have been shown to be effective.5-8 The aim of rehabilitative treatments in PD is to enable patients to improve or maintain their maximum level of independence and mobility. However, there are still many gray areas regarding PD rehabilitation. On one hand, the results of exercise training are very task specific—that is, the tasks that are trained tend not to generalize to activities not directly exercised. 8 On the other hand, contrary to drug treatment, there is still a need for new concepts and organizational models in the rehabilitation of problems associated with PD. 9
Current evidence suggests that PD patients with gait hypokinesia may improve walking speed using treadmill training more than by conventional physiotherapy. 10 In recent years, the interest in automated robotic devices for gait training for patients with various neurological disorders has grown. By imposing consistent, symmetrical lower-limb trajectories, robotic devices provide many of the proprioceptive inputs that may increase cortical activation and improve motor function11,12 while minimizing the demands on therapists. Moreover, proprioceptive inputs probably share mechanisms that are common to auditory and visual cues, thus stimulating the cerebello-premotor pathway to improve gait. 13
Given the rapid adoption in clinical practice of commercially available robot-assisted gait trainers, there is a pressing need to compare the effectiveness of these interventions with that of less complex and less expensive therapies. A recent randomized controlled trial that compared a comprehensive rehabilitative program with robot-assisted gait training showed some benefit from the latter. 14 A pilot, noncontrolled study explored the effect of robot-assisted gait training on freezing of gait. 15 In general, however, robot-assisted training has not had a clinically important effect on walking across neurological diseases. 16
The aim of the present study was therefore to evaluate if robotic gait training with the Lokomat can improve walking more than conventional gait training with a treadmill in patients with PD.
Materials and Methods
Study Design and Patients
The study is a prospective single-center, single-blind randomized controlled trial. We screened all patients who were referred to the outpatient clinic of the Neurology Unit of the University Hospital of Novara, Italy, between October 2010 and April 2011. We enrolled adults aged <75 years with a diagnosis of PD according to the UK Brain Bank Criteria, 17 disease stage <III according to the classification of Hoehn and Yahr, 18 without motor fluctuations, who were able to ambulate independently. Patients had to have had no treadmill training or other form of specific gait training for at least 6 months before the study. We excluded patients if they had current levodopa therapy that was started more than 6 months before enrollment; medical or neurological conditions that contributed significantly to gait dysfunction, such as musculoskeletal disease, severe osteoarthritis, peripheral neuropathy, or previous lower-limb joint replacement; cardiovascular disease (recent [<4 weeks] myocardial infarction or uncontrolled hypertension with blood pressure >180/110 mm Hg at rest), heart failure (NYHA ≥3); orthostatic hypotension; body weight more than 100 kg; respiratory disease; other neurological diseases; dementia; depression; or uncorrected visual disturbances. Patients who had undergone or planned to undergo deep brain stimulation in the following 6 months were excluded. All patients gave their written informed consent before entering the study, and the study was approved by the local ethics committee.
Randomization and Masking
We randomly assigned eligible patients in a 1-to-1 ratio to 2 arms: an experimental group that received robotic gait training with a Lokomat and a control group that received gait training on a treadmill for the same amount of practice time.
We allocated patients to 1 of the 2 treatment arms using a randomization scheme with blocks of 2 (1 robotic and 1 treadmill training) generated by software. 19 One of the investigators (AB) who was not involved in the evaluations checked correct patient allocation according to the randomization list. After unmasking at the end of the study, we checked that no errors had been made in allocation. A physical therapist who was not involved in the treatment of the enrolled patients and who was blinded to treatment allocation performed all outcome assessments. At enrollment, patients were instructed not to reveal the allocation arm to the outcome assessor. Patients were treated on 2 separate floors of the department, and they were treated at different hours of the day by 2 separate groups of physical therapists. However, at enrollment, patients were not blinded to treatment because they knew that the study had 2 arms.
Procedures
Patients allocated to robotic gait training were trained at 50% body weight support (BWS) for 15 minutes and 30% BWS for 15 minutes on the Lokomat (Hocoma AG, Volketswil [ZH], Switzerland). The treadmill speed was 1.5 km/h initially and increased to 3.0 km/h by increments of 0.5 km/h as tolerated. A physical therapist who was not involved in the evaluations supervised the treatment, adjusting treadmill speed and BWS.
Patients allocated to the treadmill group had 30 minutes of treadmill gait training. After we had calculated the speed during the 6-minute walk test (6MWT), the patient was trained on the treadmill (RHC770CE—RAM Medical srl, Camin, PD, Italy). Patients were trained using 80% of the maximum speed they had reached during the test for the first week, 90% for the second week, and 100% for the third and fourth weeks. A physical therapist provided auditory cueing and direct continuous feedback to the patient.
Treatment was performed 3 d/wk for 4 weeks in both the treadmill and Lokomat groups. All treatments and evaluations were carried out at the Physical and Rehabilitative Medicine Unit of the University Hospital of Novara, Italy. We recorded age, gender, height, weight, Mini-Mental State Examination scores (corrected for age and educational level), 20 6MWT, 21 10-m Walk test (10mWT), 22 and the Timed Up-and-Go test (TUG). 23 The motor subscore of the Unified Parkinson’s Disease Rating Scale (UPDRS III) 24 was used to quantify disease severity and extrapyramidal signs in the patients. Global health status was assessed using the SF-12 questionnaire, 25 and then the physical and mental health composite scores were calculated. Comorbidities were recorded and quantified using the Charlson Index. 26 All drugs prescribed for PD were also recorded.
The primary outcome measure was 6MWT. An experienced neurologist (C. Comi) was responsible for enrolling patients and obtaining informed consent. Outcome assessment was performed before treatment (after randomization), at the end of treatment, and at 3 and 6 months of follow-up. All evaluations were done in the morning between 11
At the end of the study, patients were asked if they thought that having been allocated to one arm or the other of the study had influenced their outcome.
Statistical Analysis
We calculated that a sample size of 28 patients was needed for an 80% probability to detect a difference of 20% between the 2 groups for the primary outcome with a type I error of 0.05, based on previously published data. 27 We assessed all patients who received at least 1 session of treatment (intention-to-treat principle). Intergroup comparisons were done with the Mann-Whitney test, and intragroup comparisons with the Wilcoxon signed-rank test. Statistical analysis was performed using GraphPad Prism 1.4 and GraphPad StatMate 2.0 for Macintosh OS 10.6. The study was registered with Clinicaltrials.gov as “Robotic Locomotor Experience Applied to Parkinson’s Disease (ROLEP),” study ID NCT01302184.
Results
From October 2010 to April 2011, a total of 334 patients were screened, and 304 were excluded. The most common reasons for exclusion were the presence of advanced disease and age ≥75 years. In all, 77 patients refused to participate, mainly because of a lack of public transport. Of the excluded patients, 220 had been using levodopa for more than 6 months. Of the 30 enrolled and randomized patients, 15 were allocated to the experimental arm and 15 to the control arm (CONSORT flow diagram, Figure 1). All received at least 1 treatment and were thus included in the intention-to-treat analysis.
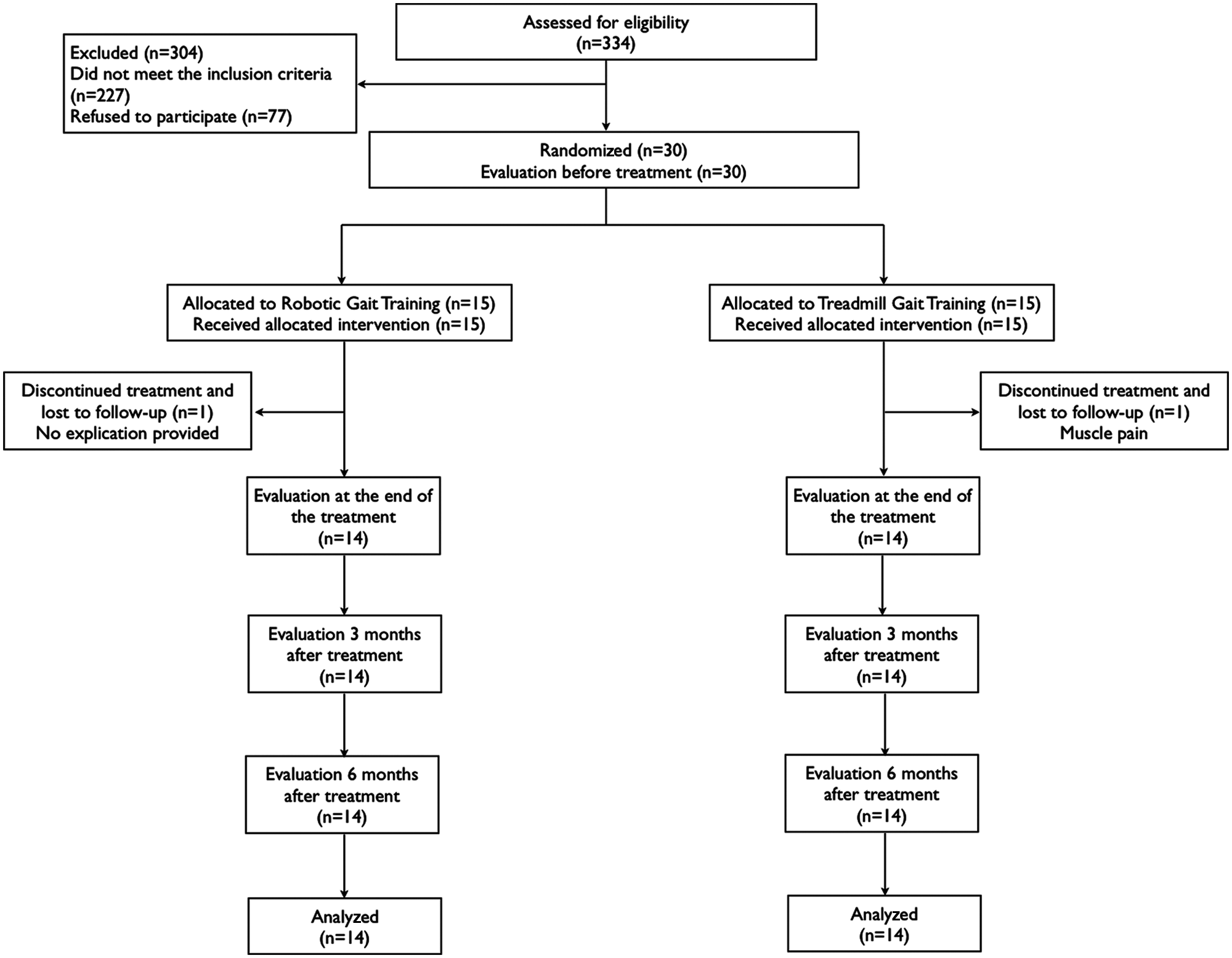
CONSORT diagram.
The mean age of the participants was 67.89 years (standard deviation, 5.4 years); 17 (60.7%) were men. The treatment groups were well matched (Table 1). None of the patients modified his or her drug treatment during the study; 2 patients, 1 of each group, did not end the treatment and refused to be evaluated at follow-up. A patient of the treadmill group stopped after 4 sessions because of muscle pain and fatigue. A patient of the Lokomat group refused to continue with the study after 2 sessions and did not provide an explanation. No relevant adverse events were recorded during the study, with the exception of mild discomfort caused by the harness in 3 male patients of the Lokomat group.
Sample Characteristics
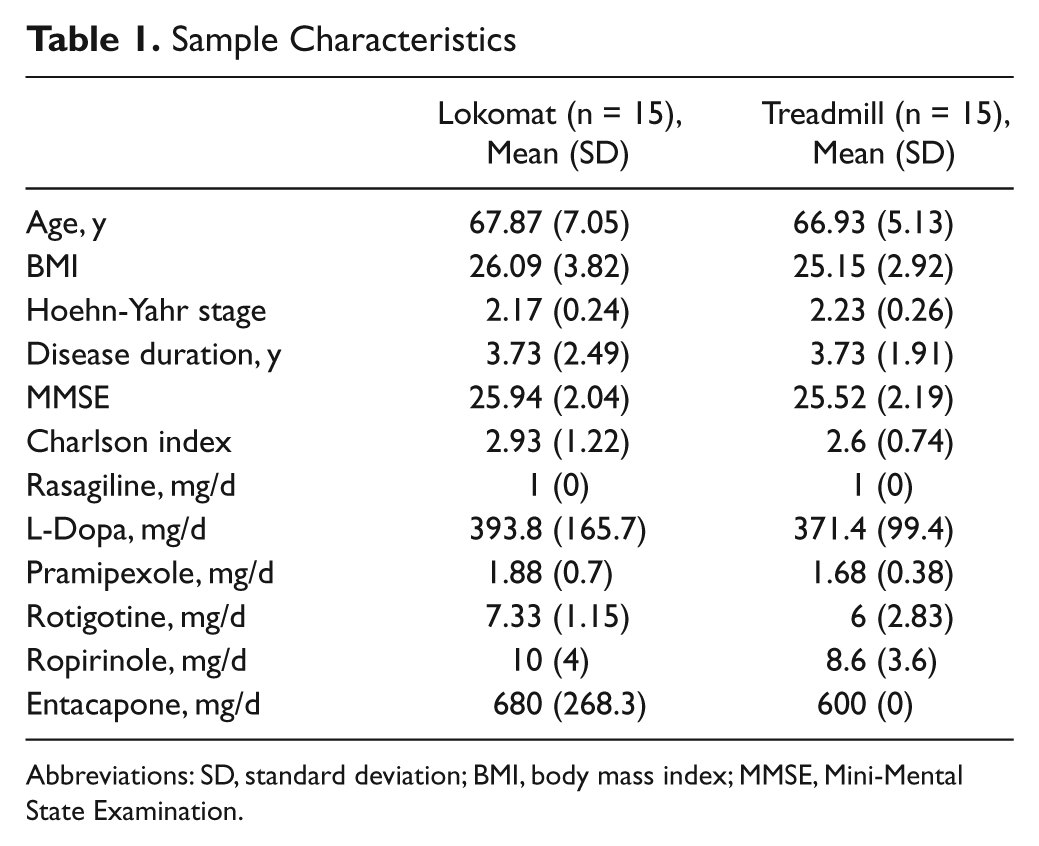
Abbreviations: SD, standard deviation; BMI, body mass index; MMSE, Mini-Mental State Examination.
The patients of both groups showed significant improvement in the distance covered during the 6MWT at the end of treatment and at the 3- and 6-month follow-up. However, the between-group differences were not significant (Table 2). Whereas patients who underwent robotic gait training improved until the third month after treatment and then slightly deteriorated at 6 months, those in the treadmill gait training group maintained their gains at the final follow-up, but these differences were not statistically significant (Table 2 and Figure 2).
Outcome Measures a
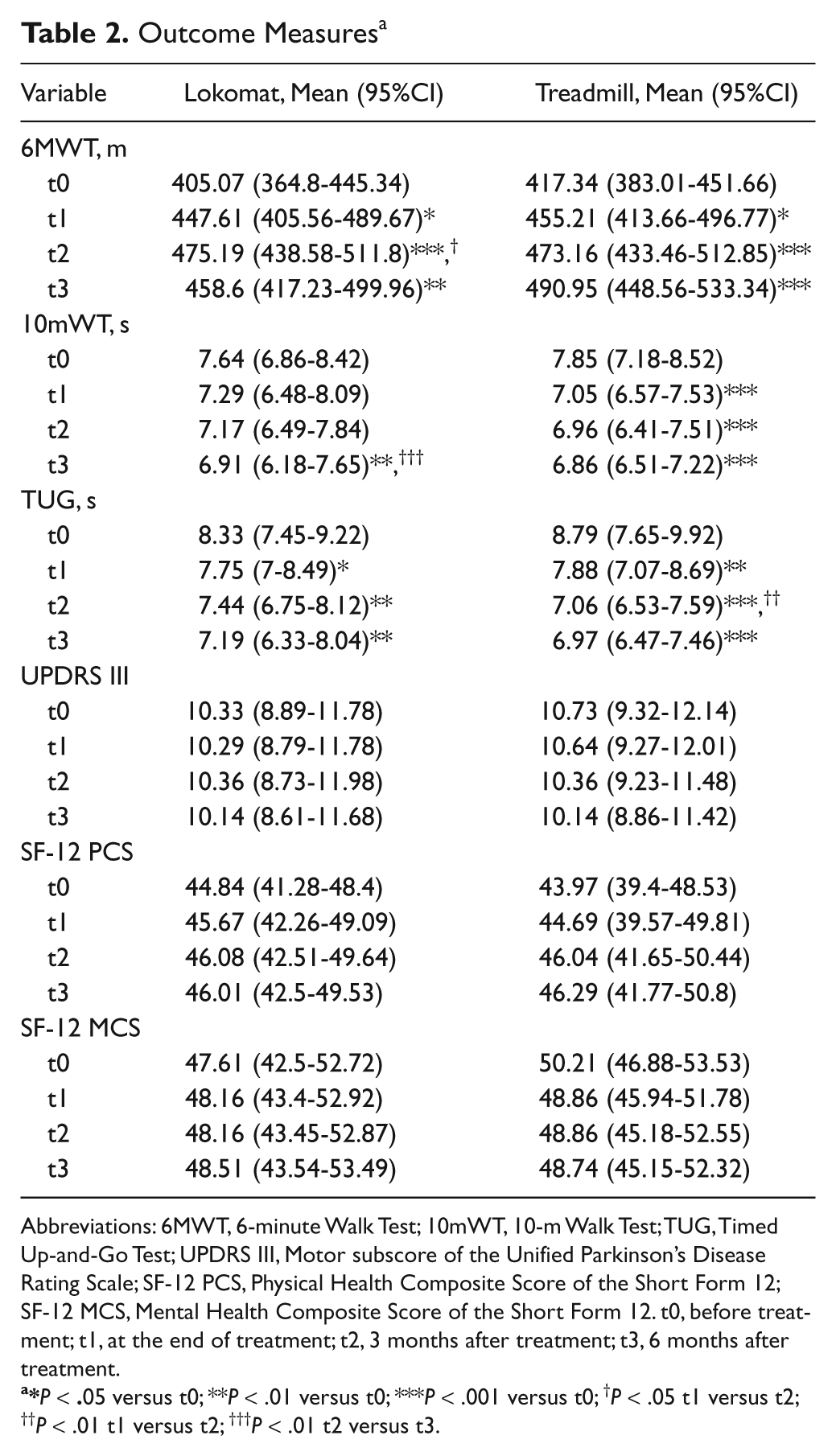
Abbreviations: 6MWT, 6-minute Walk Test; 10mWT, 10-m Walk Test; TUG, Timed Up-and-Go Test; UPDRS III, Motor subscore of the Unified Parkinson’s Disease Rating Scale; SF-12 PCS, Physical Health Composite Score of the Short Form 12; SF-12 MCS, Mental Health Composite Score of the Short Form 12. t0, before treatment; t1, at the end of treatment; t2, 3 months after treatment; t3, 6 months after treatment.
*
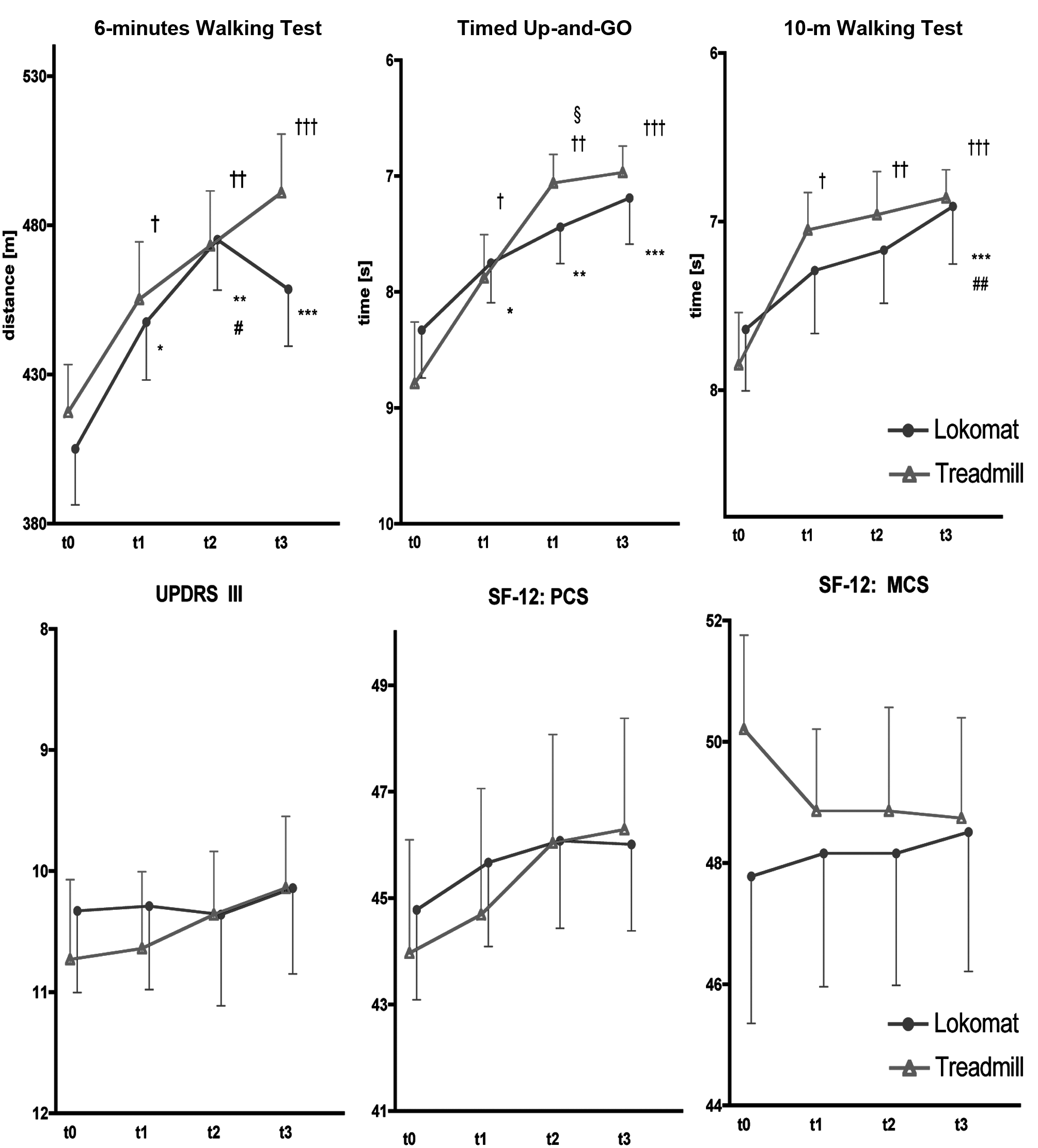
Outcome measures. aAbbreviations: UPDRS III, Motor subscore of the Unified Parkinson Disease Rating Scale; PCS, Physical Health Composite Score; MCS, Mental Health Composite Score; t0, before treatment; t1, at the end of treatment; t2, 3 months after treatment; t3, 6 months after treatment.
Patients receiving either robotic gait training or treadmill training showed significant improvements in both the 10mWT and TUG, which were higher, but not significantly, in the treadmill group (Table 2 and Figure 2). For the 10mWT, significant improvements were observed earlier in the treadmill training group. We also observed a trend for gains in each group in the motor part of the UPDRS III and the physical and mental composite scores of the SF-12 but not between groups.
Among the patients treated with robotic gait training, 2 believed that they would have achieved better results with conventional treadmill training, 4 thought that the results would have been the same, and 8 believed that Lokomat training was better than conventional treadmill training. Among the patients treated with conventional treadmill training, 1 patient believed that with Lokomat, he would have had a better result, 5 that they would have had the same result, and 8 that treadmill training was better than robotic training. It is interesting to note that the patients’ convictions did not correspond to the result obtained.
Discussion
Our data do not support the hypothesis that robotic gait training is superior to treadmill gait training in patients with early-stage PD. Even as both treatments improved the walking performance of PD patients, we failed to observe significant differences between the 2 groups in both primary and secondary outcome measures. Instead, we noticed some advantage for treadmill training with regard to the effect duration and timing of improvement. In fact, patients treated with a treadmill showed an improvement in 6MWT throughout follow-up, which was not the case for robotic gait training; moreover, improvements in the 10mWT and TUG appeared earlier in the treadmill-treated group.
On the other hand, no significant changes were observed in the UPDRS III and in the physical and mental composite scores of the SF-12 within the 2 groups. A possible explanation is that UPDRS III includes many aspects of motor examination that are unlikely to be modified by training, which is directed only at a specific function like gait. However, we cannot exclude that a larger sample could have shown a significant change in UPDRS III too. We should also notice that both treatments were safe and well accepted by patients.
Theoretically, robotic gait training might have been the better alternative compared with conventional treadmill gait training in PD patients because it forcibly matches steps to a rhythmic kinematic stimulation.11,28 However, our study did not show any superiority of this treatment over treadmill training alone. A possible explanation for this finding is that with the Lokomat, patients can be trained at velocities below 3.2 km/h (in our case ranging from 1.5 up to 3 km/h) because of technical limitations associated with the robot, whereas on the treadmill, they can reach higher speeds. As a result, less severely disabled PD patients might have been trained submaximally, thus hiding possible therapeutic effects of the Lokomat. However, gait speed was adapted to patient’s tolerance in the robotic treatment group, and we should consider that this training implies not only the increase of belt speed but also of repetitive tight and shank accelerations produced by the robot, so tolerance may differ from that for a conventional treadmill. Longer or more intense robotic gait training could have produced better results because the optimal amount of treatment is still debated. Nevertheless, participants treated with robotic gait training showed a significant improvement compared with baseline in the primary and some of the secondary outcome measures, so the exercise had an effect.
Another possible explanation for our results is that during robotic gait training, the attentional load needed is lower than that needed during treadmill gait training. 16 The attentional load is probably a key constraint to motor performance in PD,29,30 and it is possible that the use of augmented feedback techniques (visual feedback and virtual reality) could improve the patient’s motivation and concentration on the walking task. 31
Recently, Picelli and colleagues published a randomized controlled trial showing that for PD patients, robotic gait training was more effective compared with a comprehensive physiotherapy program that also included conventional gait training. 14 They found a statistical but not clinically significant effect and a small to medium effect size for one of the primary outcome measures. However, there are several differences between their study and ours. First, the speed of gait in the conventional physiotherapy group was not controlled; second, the follow-up is noticeably shorter. Third, even if probably less important, the robotic device was not the same. In fact, although they used an end-effector system, Lokomat is an exoskeleton-based device. But because a direct comparison between the 2 systems has never been published, any potential difference remains theoretical.
A strong point of our study is the length of follow-up. Previous studies provided data over shorter periods, usually until the end of treatment.27,32-35 The only study published on gait training of PD patients with a comparable follow-up was that of Miyai and colleagues. 36 However, these authors showed that patients treated with treadmill and BWS walked faster than before treatment at 1 month only, and the effect disappeared afterward.
Our data are interesting in this respect. The decline in the effect of a treatment after it has ended suggests the need for continued treatment of patients with PD; in fact, some authors 8 have suggested chronic treatment for this chronic disease. Our patients showed a progression of the improvements throughout follow-up, except for the 6MWT in the robotic gait training group. This observation may suggest that continuous treatment for patients with PD at an early stage is not strictly necessary, at least if the aim is to improve walking endurance and speed. A recent study by Frazzitta and colleagues 37 showed that the results of intensive rehabilitative treatment of patients with PD can counteract the evolution of its symptoms for up to 1 year. They suggested that the natural progression of this disease can be slowed by intensive training. In our study, during treatment, intensity was slightly greater than that recommended by the World Health Organization for older adults, for both groups. After treatment, we did not control physical activity, but according to the above-mentioned recommendations for individuals with motor disorders, all patients were advised to be as active as they could. As previously suggested by some authors, 38 the question of whether this effect is simply a result of a physical reconditioning or a neuroprotective effect is still open.
Our study has some limitations. It was done in 1 center only, and the number of patients was modest. Even though its power was calculated based on previous work, a post hoc analysis revealed that a sample size of at least 164 patients would have been necessary to detect a difference between the 2 treatments even if in favor of treadmill training. Another possible limitation is that we recruited only patients at an early stage of PD. However, we decided to treat less-disabled patients because the current literature suggests that gait training at this stage can produce better and longer lasting results. 39 Another possible limitation is that we did not evaluate balance before and after trials, thus missing one of the possible differences between the 2 treatments.
Conclusions
Our trial shows that robotic gait training is not superior to gait training on a treadmill in patients with early-stage PD, but positive training effects may persist up to 6 months. Because robotic treatments are becoming more popular in neurorehabilitation, their possible advantages and limits, especially as new devices arrive on the market, must be assessed compared with more traditional techniques. The strengths and limitations of robotic devices such as the Lokomat should also be evaluated in terms of costs in future larger multicenter trials.
Footnotes
Acknowledgements
The authors wish to thank Anna Airoldi, PT, Andrea Bottero, PT, Sofia De Grandi, MD, Elisa della Beffa, PT, Marco d’Innocenzio, PT, Roberto Paggio, PT, and Anna Tosco, PT. A special thanks to Marije de Jager for linguistic revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: We certify that no party having a direct interest in the results of the research supporting this article has or will confer a benefit on us or on any organization with which we are associated.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
