Abstract
Background
After stroke, increases in contralesional primary motor cortex (M1CL) activity and excitability have been reported. In pre-clinical studies, M1CL reorganization is related to the extent of ipsilesional M1 (M1IL) injury, but this has yet to be tested clinically.
Objectives
We tested the hypothesis that the extent of damage to the ipsilesional M1 and/or its corticospinal tract (CST) determines the magnitude of M1CL reorganization and its relationship to affected hand function in humans recovering from stroke.
Methods
Thirty-five participants with a single subacute ischemic stroke affecting M1 or CST and hand paresis underwent MRI scans of the brain to measure lesion volume and CST lesion load. Transcranial magnetic stimulation (TMS) of M1IL was used to determine the presence of an electromyographic response (motor evoked potential (MEP+ and MEP−)). M1CL reorganization was determined by TMS applied to M1CL at increasing intensities. Hand function was quantified with the Jebsen Taylor Hand Function Test.
Results
The extent of M1CL reorganization was related to greater lesion volume in the MEP− group, but not in the MEP+ group. Greater M1CL reorganization was associated with more impaired hand function in MEP− but not MEP+ participants. Absence of an MEP (MEP−), larger lesion volumes and higher lesion loads in CST, particularly in CST fibers originating in M1 were associated with greater impairment of hand function.
Conclusions
In the subacute post-stroke period, stroke volume and M1IL output determine the extent of M1CL reorganization and its relationship to affected hand function, consistent with pre-clinical evidence.
ClinicalTrials.gov Identifier: NCT02544503
Introduction
Compromised skilled hand function is one of the most common long-term deficits after stroke 1 due to the dependence of skilled hand function on the primary motor cortex (M1) and its corticospinal projections (CST) 2 which are commonly affected by ischemic stroke. 3 In non-human primates, lesions to M1 and/or lesions to the CST detrimentally affect hand function contralateral to the lesion4,5 and the ability to move the fingers independently does not recover.6,7 The neuronal substrate supporting affected hand function post-stroke recovery likely involves M1 and CST of the lesioned hemisphere, referred to as ipsilesional M1 and CST (M1IL and CSTIL).8,9 In patients with chronic stroke involving CST or M1, impaired skilled hand function is related to abnormally low output from M1IL 10 and extent of CST and M1 damage. 11 Interventions targeting M1IL hand area result in improved hand function. 12
While these data provide evidence that M1IL and CSTIL serve as the primary neural substrates for supporting hand function, reorganization of M1 in the hemisphere contralateral to the stroke (M1CL) may, however, serve as an additional substrate to support hand function13,14 for review see Refs.9,15 The extent and outcome of such reorganization depends on many factors, including lesion size and time since stroke. 16 In the chronic phase post-stroke, M1CL seems to interfere with motor function of the paretic hand in a subset of patients, possibly through abnormally increased inhibition of lesioned M1 by M1CL. In such patients, decreasing M1CL excitability results in improved performance of the paretic hand (for review see Ref. 9 ). However, emerging evidence, suggests a potentially supportive role of M1CL because abnormally increased excitatory neural activity and activation in M1CL was also seen in patients with good hand function.9,17-23 Decreasing M1CL excitability in these patients may result in deterioration of dexterity of hand performance. 22 Furthermore, in non-human primates and rats the extent of M1 injury is related to motor area reorganization in the contralesional hemisphere, which includes long-term changes in neurotransmitter systems, dendritic growth, and synapse formation (for review see Ref. 24 ). Inhibiting the contralesional hemisphere in rats that recovered from large ischemic infarcts generates behavioral deficits of the impaired forelimb 25 and excitatory stimulation can promote reorganization and recovery. 26
Taken together, these findings support the view that M1CL reorganization is involved in supporting hand function in a subgroup of patients with stroke but the factors that determine its extent are not well understood. Here, we determined key factors that are associated with the reorganization of M1CL during the subacute phase of stroke. Our working hypothesis is that the extent of M1IL and/or corticospinal tract (CSTIL) damage, as measured by the electromyographic response to transcranial magnetic stimulation (TMS) of M1IL (MEP presence or absence) and structural MRI (stroke lesion volume, lesion load of the entire CSTIL, and lesion load of the CSTIL originating in M1 (CSTIL-M1) 27 ), will determine the extent of M1CL reorganization as measured by the area under the TMS stimulus response curve (M1CL SRCAUC), impairment of hand function as measured by the Jebsen Taylor Hand Function Test (JTT), 28 and the relationship between M1CL reorganization and affected hand function (Figure 1).
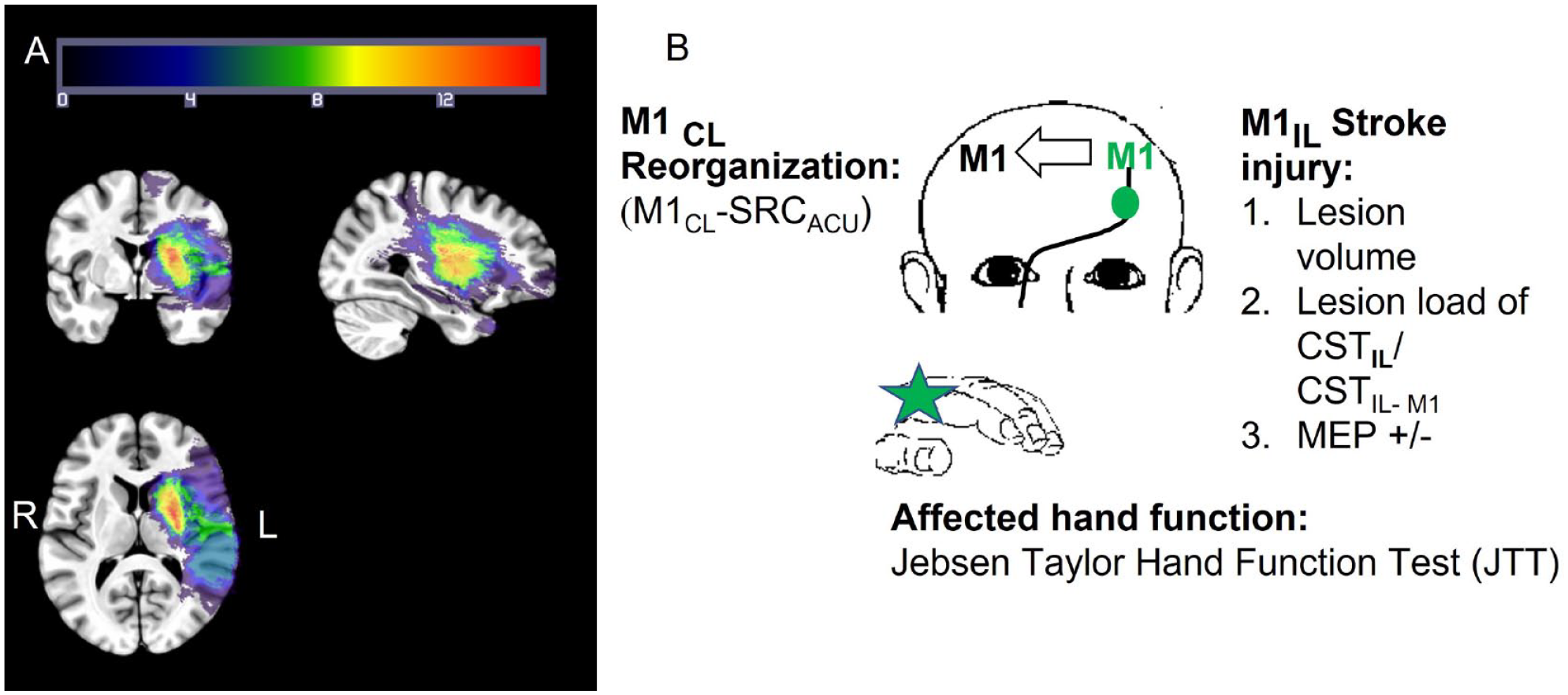
Relationship between injury of stroke and contralesional M1 reorganization: (A) lesion masks from the 35 participants with stroke projected into standard space. Lesions from the right hemisphere were flipped across the sagittal axis for illustration purposes only. Hot colors indicate a voxel is lesioned in more participants’ lesion masks and (B) our working hypothesis is that the extent of M1IL and/or corticospinal tract (CSTIL) damage, as measured by the electromyographic response to TMS of M1IL (MEP presence or absence) and structural MRI (stroke lesion volume, lesion load of CSTIL or CSTIL originating in M1(CSTIL-M1) 27 will determine the extent of M1CL reorganization (M1CL-SRCAUC), impairment of hand function (JTT) and how M1CL reorganization is related to affected hand function (JTT). Primary outcome measures: M1CL reorganization (M1CL-SRCACU), lesion volume, lesion load of CSTIL and CSTIL-M1, presence of MEP in response to M1IL stimulation (MEP +/−), and JTT score.
Methods
Study Design and Overview of Experiments
We conducted a prospective longitudinal study of patients with stroke who were included into the study based on our inclusion criteria (see below) to determine the role of M1CL reorganization in the subacute (1 month) and chronic (6 months) phase of stroke. In the present paper we report the results of the structural MRI, TMS, and behavioral measures obtained during the subacute phase of stroke only. Primary outcome measures were M1CL area under the SRC (M1CL SRCACU), presence of MEP in response to M1IL stimulation (MEP+/−), CSTIL lesion load, CSTIL lesion load originating from M1 (CSTIL-M1), stroke volume, and JTT score (Figure 1). All patients with stroke admitted to medical centers of the Georgia-NIH StrokeNet of all sexes, races, and ethnicities, age 40 to 80 years were considered for inclusion into study. The study was approved by the Emory University Institutional Review Board. Imaging data analysis was performed by KPR and MWH who were blinded to the results of the TMS and behavioral measures. TMS data analysis was performed by persons blinded to the recording side, the time of recording, the results of the imaging, and the behavioral measures.
Inclusion Criteria
Single lesion cerebral ischemic infarction <1 month affecting the M1 output system as defined by MRI of the brain, paresis of the UE (as assessed in their NIH stroke scale and chart review), no other neurological disorders, no aphasia that prevented participants from communicating effectively with the study team, no or only mild cognitive impairment based on RBANS 29 and clinical assessment, no or only mild depression (Hamilton Depression score of <19 30 ) no contraindication to TMS or MRI, no intake of CNS active drugs that block plasticity, ability to provide informed consent.
Measurement of Hand Function
Hand function was assessed with the 7 time-based subtests of the JTT. 28 Two subtests (writing and simulated feeding) were subsequently omitted due to low test-retest reliability. 31 The raw score was normalized to age- and sex-matched standard scores that accounted for hand dominance.28,32 A normalized score >0 indicated impaired hand function.
Brain Imaging
MRI Data Acquisition
High-resolution structural scans of all participants were collected on a Siemens Prisma 3T MRI system with a 64-channel head coil. T1-MPRAGE and T2-weighted structural images were collected according to the HCP Lifespan Project protocol. 33 T1_MPRAGE: TR = 2400 ms, TE = 2.24 ms, flip angle = 8°, FOV = 256 mm × 240 mm × 208 mm, voxel size 0.8 mm3. T2w_SPC: TR = 3200 ms, TE = 564 ms, FOV = 256 mm × 240 mm × 208 mm, voxel size 0.8 mm3.
M1CL Reorganization Post-Stroke
TMS was used to measure M1 excitability. The experimental design, as detailed below, is similar to the approach used successfully in previous studies.10,17,19 Briefly, TMS was applied through a figure of 8-coil (7 cm wing diameter) using 2 Magstim 2002 stimulators connected via a Bistim module (Magstim Company, UK). Electromyographic (EMG; bandpass 1 Hz-1 kHz) activity was recorded from the extensor carpi ulnaris muscle (ECU), using surface electrodes (11 mm diameter) in a belly-tendon montage and a data acquisition system (LabVIEW, NI, CA, USA). The optimal site (hot spot) for each participant was identified and marked on the structural MRI of their brain using a neuronavigation system (BrainSight, Rogue Research, Montreal, Canada). Raw EMG was digitized at 5 kHz and stored on a PC for off-line analysis. The following measures were obtained in a random order with respect to the hemisphere.
Motor Threshold (MT)
The coil was placed tangentially to the scalp and rotated 45° away from the midline. The MT was determined to the nearest 1% of the maximum stimulator output (MSO) using adaptive parameter estimation by sequential testing (PEST) 34 where the probabilistic relationship between TMS intensity and evoked MEP amplitude is modeled. This threshold- hunting method is faster when compared to the relative-frequency approach without sacrificing accuracy. 35 In participants with no measurable response to TMS of the lesioned M1 at 100% MSO, MT was defined as >100% MSO, and MEP response was noted as absent (MEP−).
Stimulus Response Curve
Single TMS pulses were applied to the ECU hotspot at increasing intensities in increments of 5% of MSO up to 80% of MSO.10,19 At each intensity, 10 stimuli were given.
Data Analysis
Analysis of Brain Imaging Data
Stroke Lesion Volume
Lesion masks were hand-drawn on each participant’s T1 image using MRIcron. 36 Participants’ structural images and lesion masks were then transformed to standard space. Transformation between an individual’s structural image and a template in atlas space can be challenging when lesions are present as standard affine or non-linear normalization techniques may result in poor template matches or overnormalization. 37 Enantiomorphic normalization 38 was used to reduce the effects of varying lesion size that may occur when normalization is restricted in certain brain areas by lesion masks. “Healed” T1 images were nonlinearly warped to the MNI152 2009 template in MNI space, with the resulting warp applied to the (lesioned) original image and the corresponding lesion mask to bring each participant’s brain and lesion mask into MNI space. Normalized lesion mask volumes (mm3) were extracted from MRIcron.
CST Lesion Load and CST Subpathway Originating in M1
CST lesion load was quantified using the SMATT template 39 because individual CST tract construction from DTI data can be challenging when large lesions are present. Subjects’ normalized lesion masks were projected onto the SMATT template. The number of overlapping voxels was divided by the total number of voxels in the template tract to quantify the percent overlap for each participant’s lesion (CSTIL). In addition to the entire CST, we also determined lesion load for the component of the SMATT template probabilistically beginning in M1 (CSTIL-M1).
Analysis of TMS Data
Trials were excluded from further data analysis because of coil mispositioning, participant’s eye closure, or increased EMG background activity defined as mean amplitude exceeding 0.05 mV in the 50 ms preceding the TMS pulse. This resulted in the exclusion of 3.18% of the data. In all other trials, the peak-to-peak MEP amplitudes were measured off-line in LabVIEW. A minimum of 5 trials per intensity were required for the data to reach criterion for mean calculation.10,40 Intensities with fewer than 5 trials were considered “missing data.” For each SRC the area under the curve (SRCAUC) was calculated in the range from 25% to 80% MSO. In the current study, M1CL-SRCAUC serves as a measure of M1CL reorganization, while M1IL-SRCAUC serves as an estimate of the stroke injury although reorganizational processes are likely also ongoing. 41 To account for these different processes, we will refer to M1IL-SRCAUC as M1 output measure.
Statistical Analysis
The primary statistical analysis was done to address our pre-stated hypothesis. Lesion volume, CSTIL lesion load, and CSTIL-M1 lesion load are continuous predictors, while MEP presence is categorical and binary. Prior to implementation of linear regression analysis, all assumptions were assessed. The appropriateness of the assumptions of normality and homogeneity of variance were examined through graphical analysis of the residuals (the difference between the observed value and fitted value). Log10 transforms of lesion volume and M1CL-SRCAUC were performed. Linear regression analyses were performed to analyze how each continuous predictor, MEP presence, and the interaction between the continuous predictor and MEP presence affected the 2 primary study outcomes: hand function (JTT score) and M1CL-SRCAUC. When the interaction term was not significant, a common slope model or simple linear regression model was used to determine the relationship between a predictor and the outcome measures. Because of the small sample size and concern with model overfitting, multiple linear regression analyses were limited to the covariates age,42,43 race,44,45 and sex.42-44 Covariate selection was driven by available knowledge and biological plausibility of potential confounders taking into consideration the research hypothesis.
Linear regression analyses were implemented with SAS PROC REG (version 9.4, SAS Institute Inc). Statistical significance was defined as 2-sided
In a secondary analysis, we explored the relationship between affected hand function and M1CL-SRCAUC and M1IL-SRCAUC. Potential multicollinearity between M1CL-SRCAUC and M1IL-SRCAUC was evaluated by the variance inflation factor (VIF).46,47 The VIF identifies correlation between independent variables and the strength of that correlation. VIFs between 1 and 5 suggest that there is a moderate correlation, but it is not severe enough to warrant corrective measures.
Results
Study Population
Sixty-six participants with reported ischemic stroke were consented and enrolled into the study. Thirty participants were subsequently excluded because of screening failure (n = 25) or because they declined to participate in all aspects of the experiments (n = 5). Thirty-six participants met all inclusion criteria indicated above and participated in all experiments. Data from 1 participant was subsequently excluded because of excessive head motion artifacts in the structural MRI. Data from the remaining 35 participants (19 female, age 59.49 ± 9.63 years) were included in the group analysis (see Supplemental Table 1 and Figure 1 for a detailed description of study population).
Covariates That Could Potentially Modify the Tested Relationship Between Our Predictors and Outcome Measures
Univariate analyses did not reveal any relationships between age, sex or race, and the measure of M1CL reorganization (M1CL-SRCAUC, Supplemental Table 2). Similarly, there were no significant relationships between age, sex or race, and hand function (Table 1).
Simple linear Regression of JTT Score With Baseline Predictors and Covariates (n = 35).
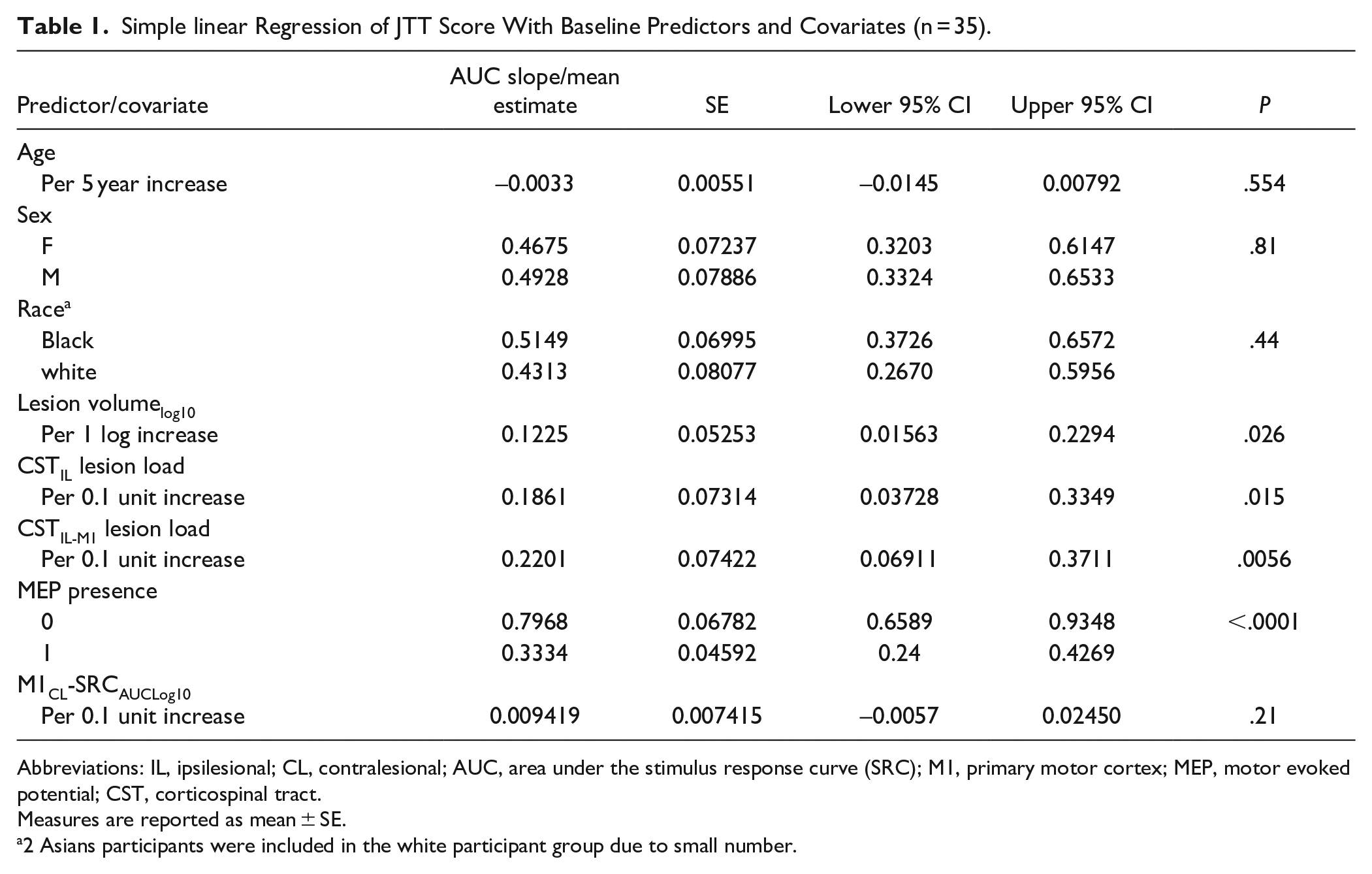
Abbreviations: IL, ipsilesional; CL, contralesional; AUC, area under the stimulus response curve (SRC); M1, primary motor cortex; MEP, motor evoked potential; CST, corticospinal tract.
Measures are reported as mean ± SE.
2 Asians participants were included in the white participant group due to small number.
Relationship Between Stoke Volume, CST Lesion Load, and M1CL Reorganization
No single measure of CSTIL functional or structural damage (MEP presence, lesion volume, CST lesion load) was significantly related to M1CL reorganization in univariate analyses (Figure 2, Supplemental Table 2). Adding MEP status to the regression analysis (Table 2) showed that the relationship between stroke lesion volume (Lesion Vol Log10) and M1CL reorganization (M1CL-SRCAUCLog10) changed depending on MEP status (
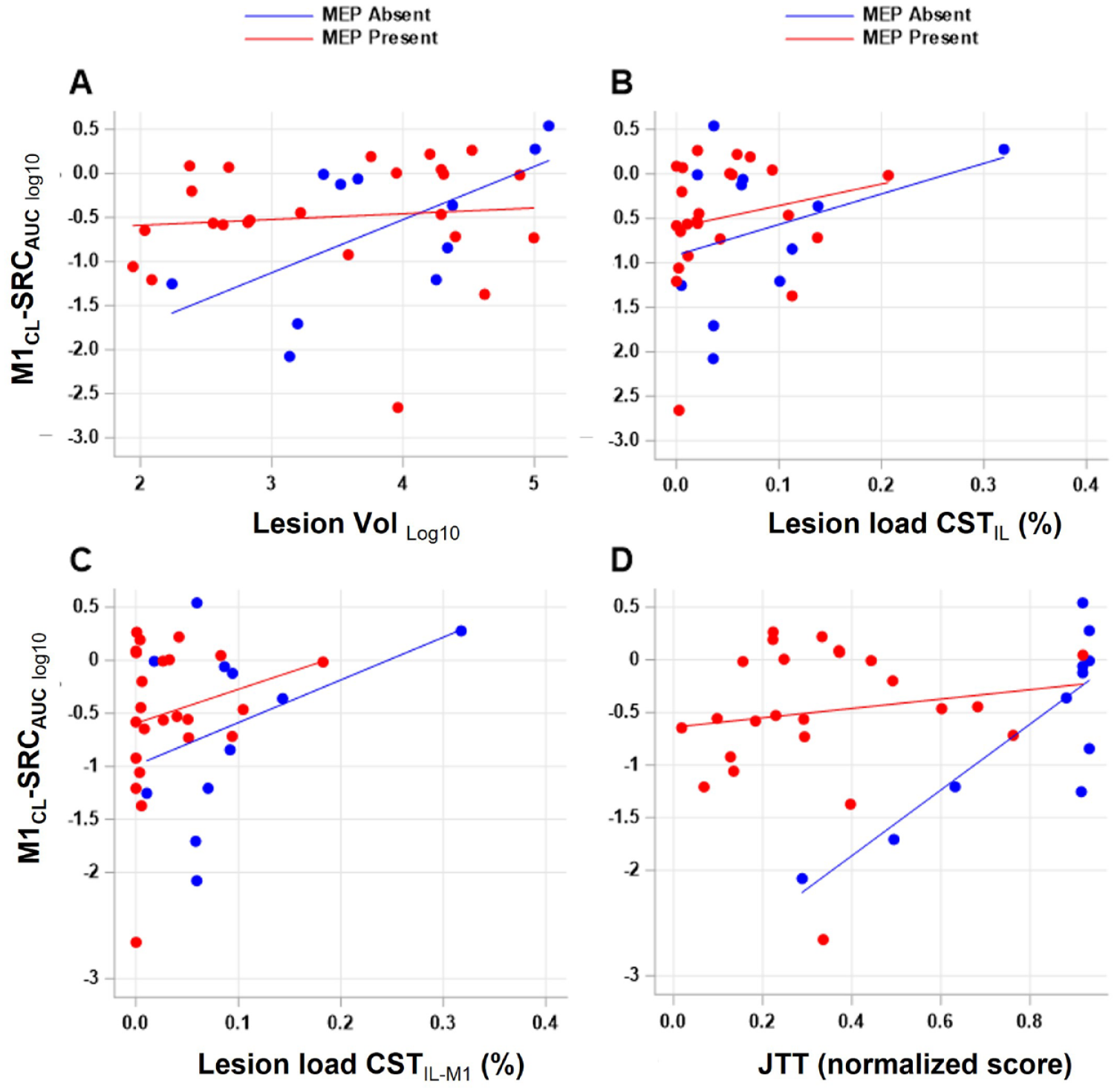
Relationship between M1CL reorganization and stroke lesion volume and/or lesion load of CST and hand function in MEP− and MEP+ participants with stroke. (A) Lesion volume (Lesion Vollog10), (B, C) lesion load of CSTIL and CSTIL-M1, and (D) affected hand function (JTT normalized score) is plotted against the extent of M1CL reorganization (M1CL-SRCAUClog10) depending on presence or absence of an MEP (MEP+/−). There is a significant relationship between extent of M1CL reorganization and lesion volume in the MEP− participants with stroke. In this group of MEP− participants with stroke, greater impairment of hand function was associated with greater M1CL reorganization.
Linear Regression for the Outcome Measure M1CL-SRCAUClog10 on Each Continuous Predictor Plus the Presence or Absence of MEP and the Interaction Between M1CL-SRCAUClog10 and MEP.
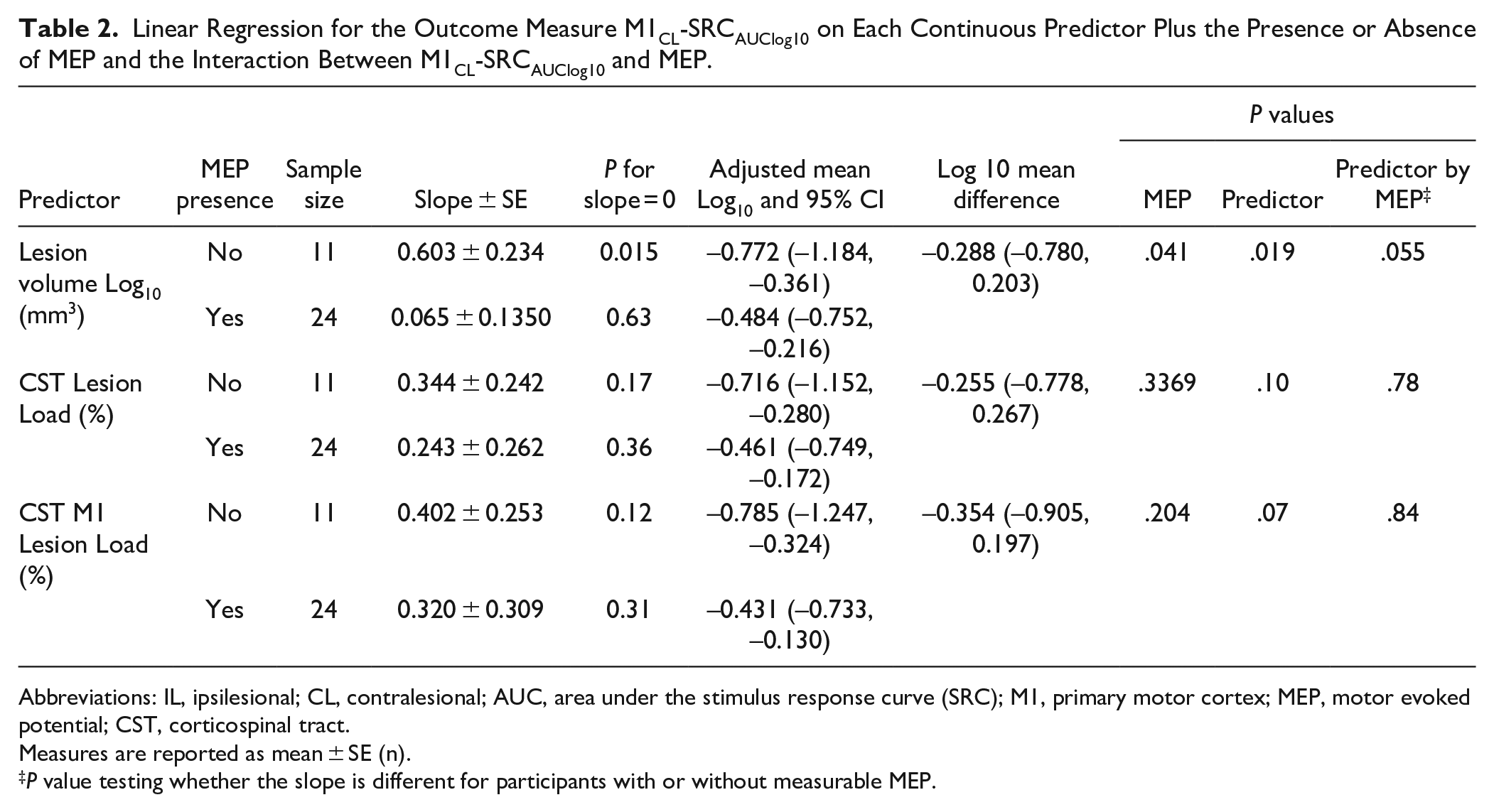
Abbreviations: IL, ipsilesional; CL, contralesional; AUC, area under the stimulus response curve (SRC); M1, primary motor cortex; MEP, motor evoked potential; CST, corticospinal tract.
Measures are reported as mean ± SE (n).
MEP status was not related to stroke volume (MEP+ = 16.0 ± 5.2 cm3, MEP− = 27.9 ± 13.4 cm3,
Extent of Damage to M1 and/or CST Relates to Hand Function
All structural (stroke volume, CST lesion load (CSTIL and CSTIL-M1)) and functional (MEP+/MEP−) measures of stroke injury were significantly related to hand function in univariate analyses (Table 1). Greater lesion volume (Figure 3A, Lesion volumelog10 slope: 0.123 ± 0.05,
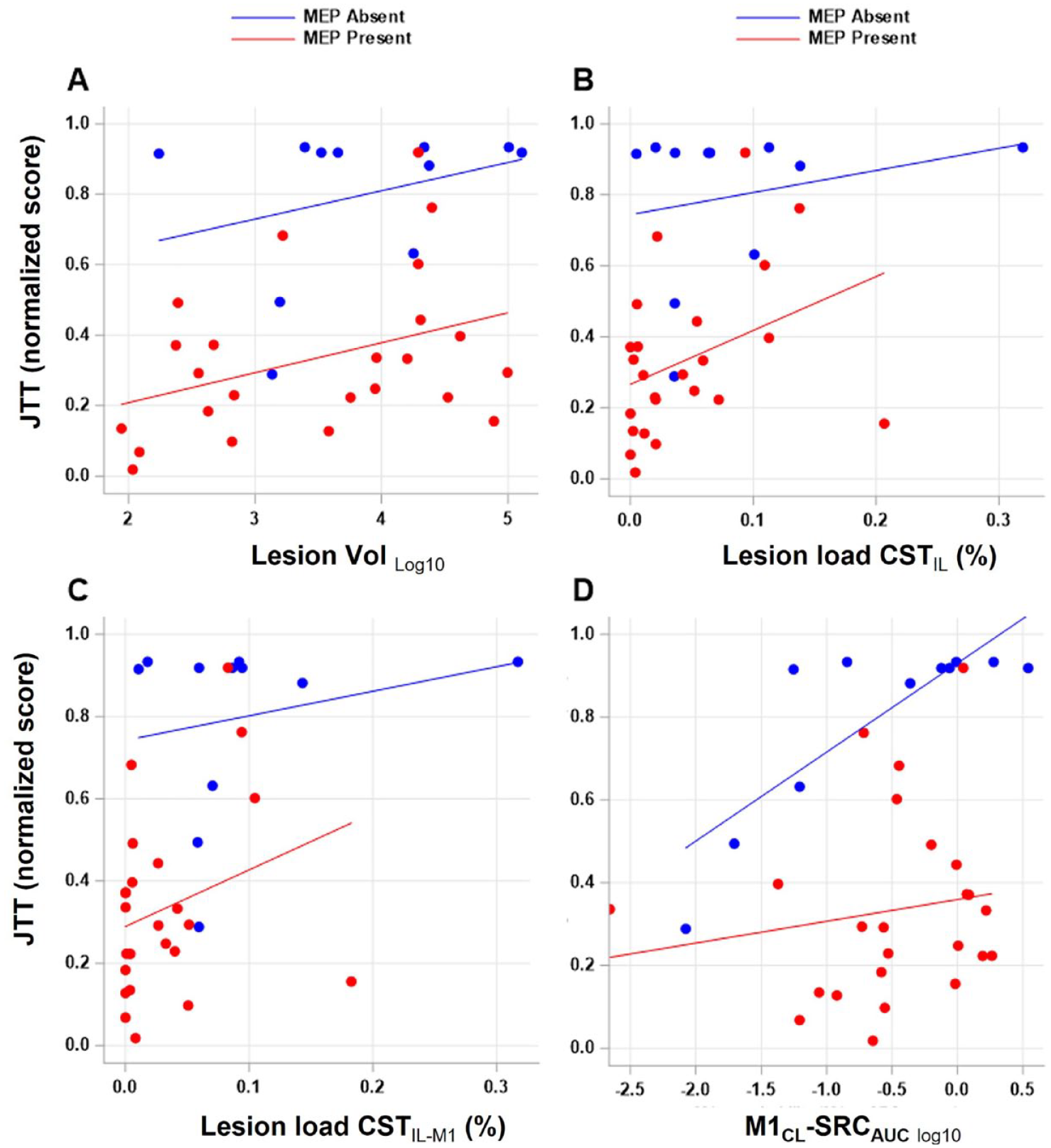
Extent of damage to M1 (M1IL) and/or CST (CSTIL) and M1CL reorganization relates to hand function. The extent of stroke injury as measured by (A) lesion volume (Lesion Vollog10), (B, C) lesion load of CSTIL and CSTIL-M1, and (D) M1CL reorganization (M1CL-SRCAUClog10) is plotted against hand function (JTT, normalized score) depending on the presence or absence of an MEP (MEP+/−). There was a positive relationship between greater lesion volume, lesion load of CSTIL and CSTIL-M1 and greater impairment of hand function.
Linear Regression for the Outcome Measure Hand Function (JTT Score) on Each Continuous Predictor Plus the Presence or Absence of MEP and the Interaction Between JTT Score and MEP (n = 35).
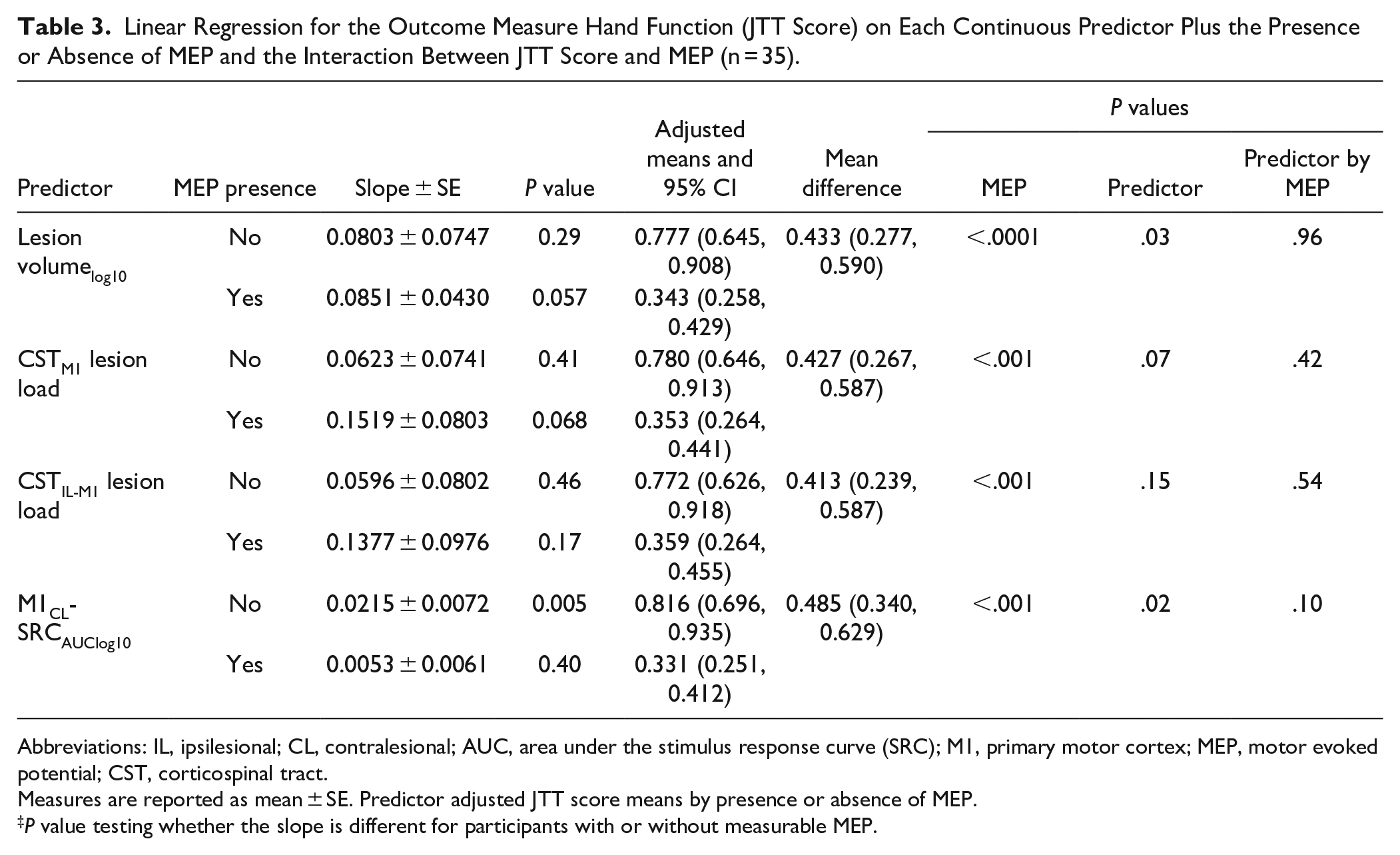
Abbreviations: IL, ipsilesional; CL, contralesional; AUC, area under the stimulus response curve (SRC); M1, primary motor cortex; MEP, motor evoked potential; CST, corticospinal tract.
Measures are reported as mean ± SE. Predictor adjusted JTT score means by presence or absence of MEP.
Relationship Between M1CL Reorganization and M1IL Output
In a secondary analysis, we explored the relationship between M1CL reorganization and M1IL output in more detail. In 12 participants, TMS of the M1IL did not elicit a measurable MEP. In these MEP− individuals the SRC areas were 0. Linear regression analysis showed a significant positive correlation between ipsilesional M1 output (M1IL-SRCAUC) and contralesional M1 reorganization (M1CL-SRCAUClog10; slope: 0.678 ± 0.24,
Relationship Between M1CL Reorganization, M1IL Functional Injury, and Affected Hand Function
We also tested the relationship between M1CL reorganization (M1CL-SRCAUClog10), M1IL injury as measured by MEP status, and hand function (Figure 1B). A model including M1CL reorganization, MEP status, and the interaction did not show a significant interaction between M1IL injury and M1CL reorganization (Table 3,
We also explored the relationship between hand function and the measures of M1CL reorganization and M1IL output as measured with M1-SRCAUC. Specifically, we asked whether the magnitude of M1IL output was related to affected hand function and whether variability in affected hand function was better explained by including measures of both M1CL reorganization and M1IL output. Univariate analyses did not show a statistically significant relationship between hand function and either M1CL reorganization or M1IL output alone (M1CL-SRCAUClog10 slope: 0.094 ± 0.07,
Finally, we explored whether stroke lesion volume explains variability in both M1CL reorganization and impaired hand function. We compared models where stroke lesion volume and M1CL reorganization are independent variables and hand function is the dependent variable. As described above, M1CL reorganization was not significantly related to hand function. Adding lesion volume to the model significantly improved the fit of the model (χ2(1) = 4.35,
Discussion
In this study of participants with subacute stroke, we tested the hypothesis that the extent of M1IL and/or CSTIL damage determines the extent of M1CL reorganization, the degree of impairment of hand function, and how M1CL reorganization is related to hand function. While M1CL reorganization was not significantly related to stroke volume or measures of CST lesion load across our entire sample, we found that larger stroke volume was significantly related to greater M1CL reorganization in the subset of participants with no measurable (MEP−). We also found that participants’ hand function declined as extent of M1IL and/or CST damage increased. The variability in hand function was best explained by a model that included both M1IL output and M1CL reorganization.
We used structural MRI measures to determine the extent of damage to the hemisphere (lesion volume) and CST. At the subacute time point of about 4 weeks the stroke lesion approaches final infarct volume. 48 While structural reorganizational changes are likely ongoing 49 and may affect these measures, the small magnitude of these changes and the relatively short time since stroke would suggest that the selected measures are indeed a good estimate of the lesion extent. We did not separately calculate injury to anatomically defined M1 as only 3 participants had purely cortical lesions. Both lesion load of the entire CST and CST fractional anisotropy measures suffer from a lack of specificity for the tract anatomy, 10 where 2 similar sized lesions can impact different pathways and therefore may impact the motor output of the hand very differently. 50 In contrast, lesion load of CSTIL-M1 more specifically measures injury to the fibers mediating output from M1.
We also used a functional measure to estimate the extent of functional impact of the stroke on the M1IL and its corticospinal projections. Specifically, MEP status (MEP+/−) and SRC were determined. Both measures depend on excitability of multiple levels including the spinal cord.9,17,19 The close relationship between CSTIL-M1 lesion load and MEP status point to a supraspinal level of functional disruption, specifically, M1 cortiocospinal projections located in the corona radiata to the level of the cerebral peduncle. Extensive functional reorganization of M1IL has been described in humans and animal stroke models.9,24 MEP status is therefore a binary measure that reflects both the injury of the stroke (i.e disruption of projections due to the lesion) but also functional reorganizational processes. 41 The close relationship between the structural measure of CSTIL-M1 lesion load and MEP presence in our study would support the notion that this measure captures functional injury to the CSTIL-M1.
In the current study, M1CL reorganization is determined from SRC. A greater area under the SRC indicates either greater excitability of the stimulated neurons or greater number of neurons being stimulated. 51 Increased M1CL excitability following stroke has been described in studies of both humans (for detailed review see Ref. 9 ) and animals where the balance between excitatory and inhibitory activity was shifted toward an increase of excitatory activity(for detailed review24,41). These remote changes are considered part of the neuronal network undergoing plastic changes in response to stroke injury and subsequent behavioral modification. Our results are in line with results from non-human primate and rodent experiments, where a relationship between M1 lesion size and the reorganization of motor representations in M1CL has been reported (for review see Ref. 24 ). However, a direct comparison between the results of these pre-clinical data and our participants with stroke is hampered by the fact that, in our participants, stroke injury was not restricted to M1 or even to cortex, which is a shortcoming of these animal stroke models (for review see Ref. 52 ). The findings of a relationship between greater stroke volume and greater M1CL reorganization in MEP− participants indicates that the extent of the lesion is important but that there are other factors such as the functional damage to M1IL/CSTIL (MEP−) that determine the extent of the M1CL reorganization. This is topic of future research but it is conceivable that greater stroke volume is more likely to affect critical structures such as premotor areas that have abundant interhemispheric connections to homotopic areas and project to M1 that impact M1CL reorganization 24 and that these processes are facilitated in the setting of severely impaired M1IL/CSTIL function.
We found a positive association between measures of M1CL reorganization (M1CL-SRCAUC) and M1IL output (M1IL-SRCAUC). M1IL output depends on the structural integrity of M1 and CST but also on the neuronal excitability at the cortical, subcortical, and spinal levels.9,17,19 In the current study, the origin of M1IL output variability likely localizes to the supraspinal level because of the occurrence of greater MEP amplitude with smaller lesion load. The positive relationship between M1IL output and M1CL reorganization would argue against excessive inhibition from M1CL on M1IL (for review see Ref. 9 ). Instead, our results are in line with earlier cross-sectional studies of participants with subacute stroke17,19 and longitudinal studies of stroke recovery53,54 who did not find any evidence for M1CL related inhibition of M1IL output.
We found that hand function is related to structural injury of CSTIL, particularly the lesion load of CSTIL-M1, which is consistent with the established relationship between CST anatomy and hand function in non-human primates. CSTIL functional damage, as measured by MEP status, is similarly related to hand function where MEP+ participants had significantly better hand function than MEP− participants. In non-human primates, anatomical and electrophysiological evidence supports the direct projections of pyramidal tract neurons located in the caudal subdivision of M1projecting to the alpha motor neurons innervating hand- and finger muscles (for review see Ref. 24 ). Because the MEP evoked by TMS of M1 is mediated by these monosynaptic connections, 9 our results support the notion that dexterity of the fingers and hand depends on this pathway. There are few reports of a close relationship between MEP amplitude and hand function in the subacute55,56 and chronic phase of stroke.10,56 Whether gross hand functions such as power grasp demonstrate a similarly strong relationship with M1IL output needs to be further investigated. A different M1IL output for distal and proximal arm muscles in relation to recovery was reported 56 and would support a differential approach to different aspects of hand and upper extremity function. Results from non-human primate studies demonstrate that muscles of the upper extremity are innervated by multiple motor tracts such as the CST, 4 the reticulospinal tract, 57 and the vestibulospinal tract. 58 Loss of more gross aspects of motor control was more transient after CST transection compared the dexterity of hand function. 6 In contrast to other studies, 59 we report a correlation between stroke volume and hand function which could be explained by our inclusion criterion requiring injuries to M1 or CST.
In the current study, hand function was better explained by the combination of M1CL reorganization and M1IL functional output than by either measure alone which is in line with evidence from Lotze et al 22 where transient disruption of M1CL had a detrimental effect on affected hand function in patients with stroke. While the evidence from non-human primates and humans supports a causal relationship between compromised M1IL output and impaired hand function,8,10,12 the nature of the relationship between M1CL reorganization and hand function is less clear. In the present study, there is shared variability between stroke volume and impaired hand function and M1CL reorganization. Whether there is a causal relationship is not evident from this analysis. While adding M1CL reorganization to the model of stroke volume and hand function does not explain additional variability in hand function, adding M1CL reorganization to the model of M1IL output and hand function does. This lack of relationship in 1 case but presence in another would support the notion of a weak relationship between M1CL reorganization and hand function. While comparison is limited, our findings differ from results by Stinear et al 53 where improvement of UE function was unrelated to M1CLexcitability. Our results would suggest that the extent of stroke injury should be considered when studying functional measures of M1CL reorganization, M1 M1IL output and hand function (for review see Ref. 60 ).
In conclusion, we provide evidence for stroke volume and M1IL output determining the extent of M1CL reorganization and its relationship to affected hand function in the subacute post-stroke period which confirms our stated hypothesis. Consistent with the pre-clinical data we also found that participants’ hand function declined as stroke volume or lesion load to CST, particularly CSTM1, increased. In extending previous findings, the variability in hand function was best explained by a model that included both M1IL output and M1CL reorganization.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231152816 – Supplemental material for Stroke Lesion Volume and Injury to Motor Cortex Output Determines Extent of Contralesional Motor Cortex Reorganization
Supplemental material, sj-docx-1-nnr-10.1177_15459683231152816 for Stroke Lesion Volume and Injury to Motor Cortex Output Determines Extent of Contralesional Motor Cortex Reorganization by Cathrin M. Buetefisch, Marc W. Haut, Kate P. Revill, Scott Shaeffer, Lauren Edwards, Deborah A. Barany, Samir R. Belagaje, Fadi Nahab, Neeta Shenvi and Kirk Easley in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683231152816 – Supplemental material for Stroke Lesion Volume and Injury to Motor Cortex Output Determines Extent of Contralesional Motor Cortex Reorganization
Supplemental material, sj-docx-2-nnr-10.1177_15459683231152816 for Stroke Lesion Volume and Injury to Motor Cortex Output Determines Extent of Contralesional Motor Cortex Reorganization by Cathrin M. Buetefisch, Marc W. Haut, Kate P. Revill, Scott Shaeffer, Lauren Edwards, Deborah A. Barany, Samir R. Belagaje, Fadi Nahab, Neeta Shenvi and Kirk Easley in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the participants for their contribution to this study, S. Buetefisch and J. Hudson for technical support, E. Sperin for assistance with regulatory work, and G. Kowalski, A. Caliban, A. Mangin, I. Vernon, and I. Chung for assistance with the data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurological Diseases and Stroke, National Institute of Child Development and Health, National Institutes of Health (grant numbers R56NS070879, R01NS090677, 3R01NS090677-04S1). DAB received support from the Emory University NIH/NINDS T32 training and translational research in neurology program (T32NS007480), Georgia StrokeNet (1U10NS086607), and the American Heart Association (18POST34060183). DAB is currently at the Department of Kinesiology, University of Georgia, and Augusta University/University of Georgia Medical Partnership, Athens, Georgia. LE received support from the Emory University NIH/NINDST32 training and translational research in neurology program (T32NS007480), and the American Heart Association (18POST34060183).
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
