Abstract
Background. Up to 85% of people with chronic stroke experience somatosensory impairment, which contributes to poor sensorimotor control and non-use of the affected limb. Neurophysiological mechanisms suggest motor rehabilitation may improve tactile sense post-stroke, however, somatosensory recovery has rarely been reported in controlled trials. Objective. To compare the effect of four upper limb motor rehabilitation programs on the recovery of tactile sensation in adults with chronic stroke. Methods. Adults with chronic stroke and mild or moderate upper extremity hemiparesis (n = 167) were enrolled in a multi-site randomized controlled trial. Participants completed three weeks of gaming therapy, gaming therapy with additional telerehabilition, Constraint-Induced Movement therapy, or traditional rehabilitation. Here, we report the results of a secondary outcome, tactile sensation, measured with monofilaments, before and after treatment, and 6 months later. Results. A mixed-effects general linear model revealed similar positive change in tactile sensitivity regardless of the type of training. On average, participants were able to detect a stimulus that was 32% and 33% less after training and at 6-month follow-up, respectively. One-third of participants experienced recategorization of their level of somatosensory impairment (e.g., regained protective sensation) following training. Poorer tactile sensation at baseline was associated with greater change. Conclusions. About one-third of individuals with mild/moderate chronic hemiparesis experience sustained improvements in tactile sensation following motor rehabilitation, regardless of the extent of tactile input in the rehabilitation program. Potential for sensory improvement is an additional motivator for those post-stroke. Characteristics of those who improve and mechanisms of improvement are important future questions. Clinicaltrials.gov NCT02631850
Introduction
Impaired somatosensation is common following stroke and is associated with reduced function, 1,2 participation in activities, 3 and quality of life. 4 Early after stroke, spontaneous neurobiological recovery drives somatosensory recovery. 5 The determinants of somatosensory recovery in the chronic phase post-stroke are less certain. Reviews of post-stroke somatosensory retraining studies indicate most are limited by low power, heterogeneity and coarseness of measures, and poor-quality research design. 6 -8 The effect of motor rehabilitation on upper limb somatosensory recovery is rarely reported in clinical trials.
Motor learning research in humans and other animals suggests that it is possible for motor rehabilitation to drive post-stroke somatosensory recovery. Areas of the brain that process somatosensory information, including the posterior parietal cortex, are highly interconnected with frontal lobe motor planning areas. 9 These sensorimotor networks undergo lasting functional and connectivity changes following motor learning. 10 While the mechanisms underlying these changes remain uncertain, 10 one possibility is positive neural adaptation caused by increased somatosensory inflow from movement. During movement, afferent input increases from muscles, joint receptors, and even cutaneous mechanoreceptors. 11 Another consideration is that upper limb motor rehabilitation typically involves the manipulation of objects, which stimulates cutaneous receptors directly. Via parallel pathways, these submodalities of touch and proprioceptive somatosensory information converge in the cortex and are processed in distributed networks. 12 The feedback loop present under typical conditions between somatosensation and movement supports the bidirectional process of sensorimotor learning 13 that is associated with motor and somatosensory system network changes. 14
Despite physiological evidence in support of bidirectional sensory and motor learning and their documented co-occurrence under typical conditions, 10,15,16 stroke rehabilitation research has largely siloed the two. Post-stroke motor recovery has been studied following motor rehabilitation paradigms, 17 -19 while somatosensory recovery has been examined following somatosensory training paradigms, such as somatosensory discrimination training. 20 It remains largely unknown whether motor focused rehabilitation concurrently improves somatosensation in the chronic post-stroke phase.
To our knowledge, only three small trials have reported the effect of motor rehabilitation on somatosensory function in people with stroke. A small RCT found that inpatient stroke motor rehabilitation produced similar somatosensory recovery to integrated sensorimotor rehabilitation. 21 This trial occurred during the early recovery period, however, in which spontaneous neurobiological recovery co-occurs with rehabilitation-induced recovery. 21 Two small case series in chronic stroke concluded that motor rehabilitation improved tactile sensation for some participants, 22,23 but larger controlled trials were needed.
Given that most somatosensory rehabilitation research has involved a somatosensory (versus motor) emphasis to the training, 6 -8 it also remains unclear whether the extent of tactile input during training influences the extent of sensory improvement. Accordingly, tactile sense was included as a secondary outcome measure during the Video Game Rehabilitation for Outpatient Stroke (VIGoROUS) multi-site clinical trial in people with chronic stroke. 24,25 This trial compared video-game-based upper limb rehabilitation (which involved minimal object manipulation during training) to two different interventions that involved manipulation of real objects: high-dose Constraint-Induced Movement (CI) therapy and lower-dose traditional rehabilitation. Based on results from a feasibility study, 23 we hypothesized that training would result in improved tactile sense in all treatment groups. We further hypothesized that greater tactile input during training (i.e., within the CI therapy and traditional rehabilitation groups that manipulate actual objects) would yield greater tactile improvements compared to game-based rehabilitation.
Methods
Design
The VIGoROUS clinical trial was a pragmatic, randomized, assessor-masked, four parallel arm trial that compared the effectiveness of three weeks of in-home, gamified, upper limb rehabilitation in people with chronic stroke to in-clinic CI therapy 17 and traditional rehabilitation. 24 The primary outcomes; arm use (Motor Activity Log Quality of Movement) and motor function (Wolf Motor Function Test) have been reported elsewhere. 25 In this paper, we report the results of a secondary outcome, tactile sensation, measured with monofilaments before and after treatment and 6 months later. Stratified randomization based on baseline motor ability was used to balance the groups. Allocation was masked by having participants draw sealed opaque envelopes at random from a larger envelope.
Inclusion/Exclusion Criteria
Adults with chronic stroke (>6 months) were recruited at five sociogeographically diverse sites between February 2016 through May 2019. Eligible participants were community-dwelling adults who had experienced a stroke of any etiology with resulting mild to moderate upper limb hemiparesis. Participants exhibited some voluntary control of the thumb, fingers, wrist, elbow, and shoulder, but limited arm use during daily life (Motor Activity Log Quality of Movement scale ≤2.5) at baseline. 26 They agreed to be randomized to one of four treatment groups and to forego other upper extremity interventions during the three weeks of study intervention. Exclusion criteria included inability to provide informed consent, having previously received either CI therapy or the gaming intervention, and having received upper extremity Botox within 3 months of beginning treatment.
Interventions
Comparison of treatment arms.
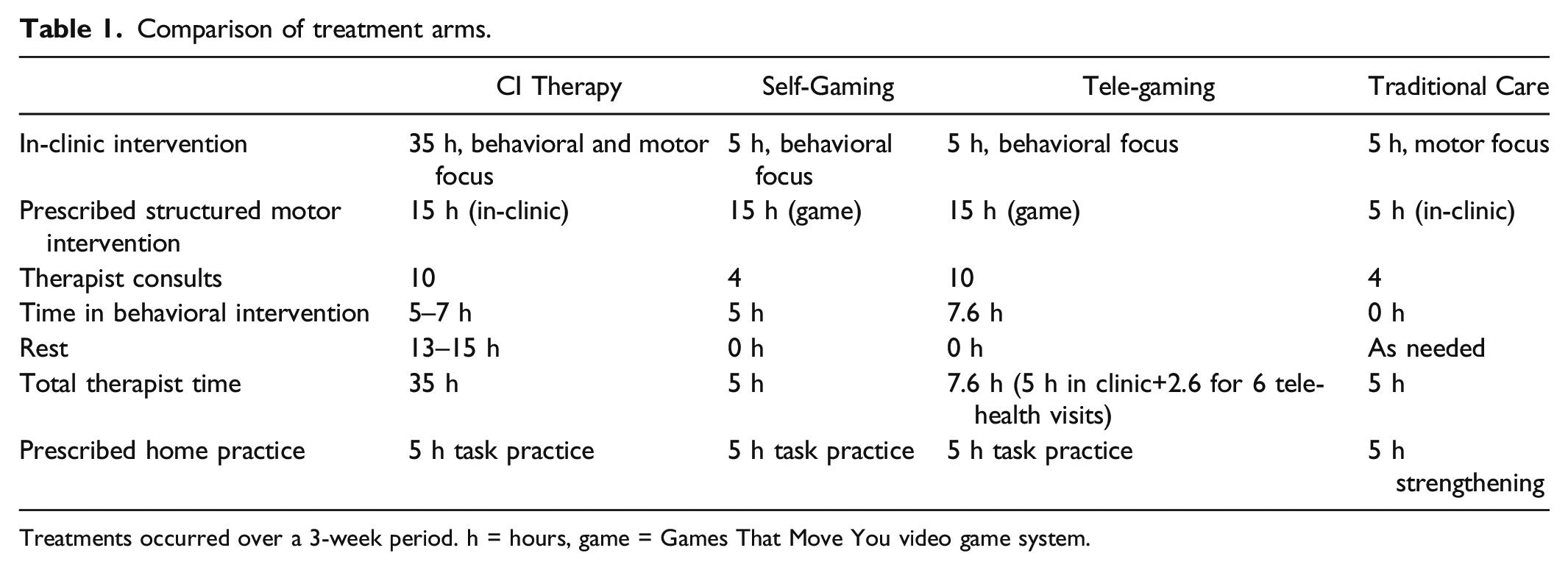
Treatments occurred over a 3-week period. h = hours, game = Games That Move You video game system.
Gaming
Participants in the two gaming groups were prescribed in-home, unsupervised upper limb movement training via the commercially available Games That Move You video game system. 27 Movements of the proximal and distal paretic arm/hand were captured by a camera sensor to drive gameplay (e.g., opening and closing the hand caused an Avatar to “grasp” a virtual object, left shoulder abduction moved an Avatar to the left). As the gaming therapy involved only virtual object manipulation, there was presumably less tactile input during gameplay than would occur during traditional motor rehabilitation of the upper limb. In-game movements were specifically designed to be therapeutic for the post-stroke population. The difficulty of each task progressed as the participant’s range of active movement improved.
To aid participants in translating in-game movements to everyday activities, participants in both groups agreed to complete a total of 5 hours of unsupervised practice (30 minutes on at least 10 days) performing everyday in-home activities with their paretic arm. Four in-clinic face-to-face treatment sessions with a therapist focused on goal setting, self-assessment, problem-solving, and other behavioral techniques to reflect on the success of the participants’ daily activities practice and to counteract non-use of the paretic arm. 26
One gaming group received no therapist contact between clinic visits (Self-Gaming). The other received 6 additional brief video teleconsultations focused on the behavioral techniques (Tele-Gaming).
Constraint-Induced Movement Therapy
Participants received 10 sessions of face-to-face treatment that included the same behavioral techniques plus 15 hours of in-clinic active movement practice involving object manipulation. As with the gaming therapy, difficulty progressed as the participant showed greater mastery over a task. 26 Participants similarly agreed to complete a total of 5 hours of in-home unsupervised practice performing everyday activities with the paretic arm.
Traditional Therapy
Participants completed four in-clinic treatment sessions (total duration = 5 hours) that included neuromuscular reeducation, functional training, and progressive strengthening. In-home unsupervised strengthening exercises totaling 5 hours were prescribed between in-clinic visits. The reader is referred to the VIGoROUS protocol and primary outcome paper for additional methodological detail. 24,25
Quantitative Assessment of Tactile Sensation
Test instrument
Semmes-Weinstein monofilaments were used to determine tactile sensitivity at pre-treatment, post-treatment, and 5–7 months after treatment by an assessor naïve to group assignment. Kits of 20 monofilaments that ranged from .008 to 300 grams (calibrated to ±5%) were used. Monofilaments are the most established objective measure of tactile sensitivity used in both clinical and research settings. 28 Tactile sensitivity is defined as the threshold (in grams) detected consistently. 29 In patients with stroke, monofilaments were found to have acceptable inter-and intra-rater agreement and have convergent validity with the Stroke Impairment Assessment Set. 30 Monofilament scores are correlated among the digits, 30 responsive to change, and immune to practice effects in those with stroke. 31
Adaptive Staircase Method
The tactile sensitivity threshold was defined as the lowest force monofilament detected using an adaptive staircase approach. 32 In this approach, monofilaments are applied in series from coarse to fine until the participant no longer detects a stimulus. The examiner then reverses the order and presents in order from finer to coarser, then back to finer until the finest (lowest force) monofilament the participant detects in three consecutive applications of that monofilament is determined. This method yields accurate results while minimizing participant and experimenter time. 32 Participants were seated at a table across from the examiner in a quiet area. Their hand and arm were supported in supination on a tabletop with their vision of their hand occluded by a curtain. Monofilaments were applied at a 90° angle to the center of the pad of the index finger until they bowed and were held there for approximately 1 second. Participants were instructed to say “yes” when they felt a monofilament on their index fingertip and “no” if they did not. 31 Participants were also told that trials in which the monofilament was not pressed to the skin would be interspersed throughout testing. These trials, termed catch trials, are used to reduce the likelihood of false-positive responses from guessing and response bias. No feedback was provided regarding performance. Detection of ≤.07 grams indicates normal tactile sensitivity, 33 .16 or .4 grams is classified as diminished tactile sensitivity. Detection in the range of .6 to 2 grams indicates diminished protective sensation. Failure to detect monofilaments ≥4.0 grams indicates the loss of protective sensation. 29
Data were log-transformed as recommended for analysis. 34 Note that estimates derived from log-transformed variables approximate, but slightly overestimate, percent improvement. For example, an estimate of −.39 log g reflects a 33% decrease in pressure required to detect the stimulus, thus improved tactile sensitivity. For ease of interpretation, the change in grams and percent change are reported with the effect size estimates. The minimally clinically important difference has yet to be established for Semmes-Weinstein monofilaments; a 20% decrease in force was estimated as a potential conservative reporting benchmark for clinically meaningful improvement given that decreases of 17%–19% result in reclassification of a person’s sensory performance (e.g., “loss of protective sensation” to “diminished protective sensation”). 29
Analysis of Baseline Relationships
Between-group differences in tactile sense at baseline were examined using ANOVA. To determine whether poorer tactile sense at baseline co-occurred with poorer motor function or quality of everyday arm use, simple regressions examined Monofilament log grams of force as a function of log-transformed Wolf Motor Function test performance time (WMFT) and mean Motor Activity Log (MAL) scores, respectively.
Intent-To-Treat Analysis of Comparative Treatment Effects
All 167 participants who began treatment were included in modified intent-to-treat analyses. Random Forest Multiple Imputation was used to estimate missing data. 35 This method used group assignment, Monofilament scores and their changes, summary scores on the WMFT and Motor Activity Log MAL and their changes, individual item responses on the WMFT and MAL, Montreal Cognitive Assessment (MoCA) total score, and select demographic information (affected side, sex, handedness) to estimate the missing data. 25
Change in tactile sense was examined via mixed-effects linear models using Matlab 2020b. The initial model included treatment group, time (treatment effect), and their interaction (comparative treatment effect) as fixed effects, and study site and participant as random slopes and intercepts. Several participant factors were examined as covariate candidates: baseline tactile sense in grams (log-transformed), baseline motor ability (log-transformed WMFT performance time), baseline arm use (MAL), change in WMFT performance over time, change in MAL performance over time, cognition (MoCA), age, sex, chronicity (years since the stroke), and whether or not the dominant hand was more affected. Before adding a new covariate, multicollinearity was assessed via cross-correlation plots, examining the influence of the additional predictor on the standard errors of the estimates, examining whether adding the predictor dramatically changed existing estimates in unexpected ways, and ensuring that the Variance Inflation Factor did not exceed 5. Covariates exhibiting evidence of multicollinearity with the treatment effect or comparative treatment effect were excluded. A covariate was added to the statistical model if it significantly influenced treatment response or comparative treatment response (P < .05) without adversely influencing the Bayesian Information Criterion of the model. Covariates and interactions were removed from the model when P > .1.
Residuals from the mixed-effect model were not normally distributed (leptokurtic with heavy tails) and heteroscedastic. To account for this, permutation analysis was employed to determine the P-values of the estimates. Permutation analysis involved randomly shuffling the Monofilaments data without replacement to generate a null distribution for the mixed-effect model. Upon each of 5000 iterations, Monofilaments scores for each participant were randomly assigned to the baseline, post-treatment, and follow-up time points to generate a null distribution for the treatment effect. They were also randomly shuffled between participants to generate null distributions for the covariates. The P-value for each fixed effect variable was reported as the proportion of the null distribution with an effect size of greater magnitude than the observed effect.
Power
Power was calculated via permutation analysis in Matlab 2020b. Despite Monofilaments’ status as a secondary outcome measure in the VIGoROUS study, there was adequate power to detect between-group differences on this measure. Power was >99% to detect a change in Monofilaments sensory threshold over time given effect size (−.34, 29% decrease in force) and variance from a prior pilot study, 23 167 participants, and an alpha of .05. The power to detect a comparative treatment effect of the same magnitude between the two gaming and the two in-clinic groups was >95%.
Regulatory Information
This trial was prospectively registered on Clinicaltrials.gov: NCT02631850. The Institutional Review Boards of The Ohio State University, OhioHealth, University of Alabama, and Missouri University approved the study. Participants provided written informed consent. Data from this study are available from the corresponding author upon request.
Results
Data Completeness
Baseline demographic and clinical characteristics (N = 167).
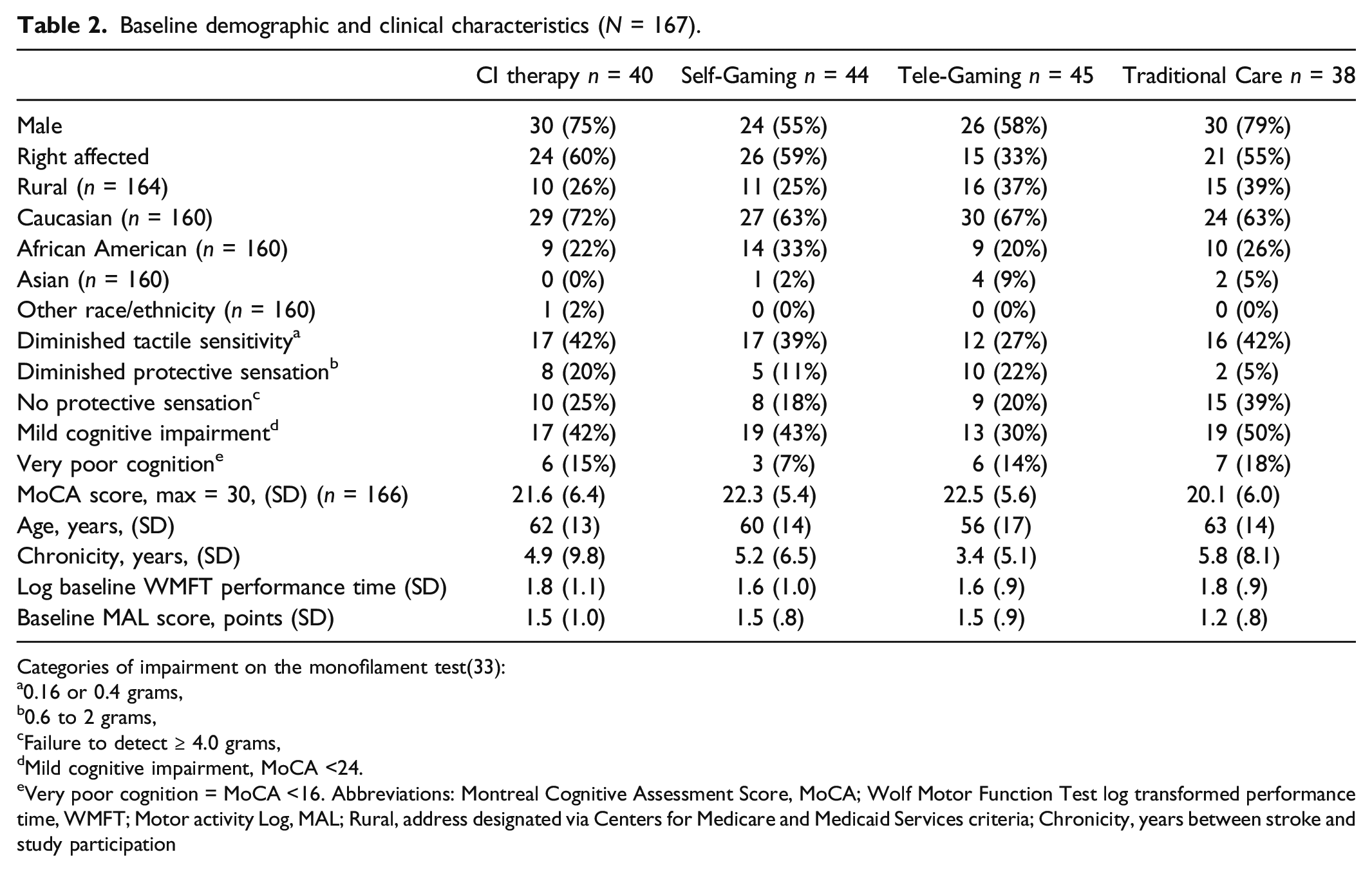
Categories of impairment on the monofilament test(33):
a0.16 or 0.4 grams,
b0.6 to 2 grams,
cFailure to detect ≥ 4.0 grams,
dMild cognitive impairment, MoCA <24.
eVery poor cognition = MoCA <16. Abbreviations: Montreal Cognitive Assessment Score, MoCA; Wolf Motor Function Test log transformed performance time, WMFT; Motor activity Log, MAL; Rural, address designated via Centers for Medicare and Medicaid Services criteria; Chronicity, years between stroke and study participation
Baseline Relationships
Poorer tactile sense at baseline co-occurred with poorer movement capacity (log Wolf Motor Function Test performance time, beta = .93, r = .25, P<.001) and poorer quality of everyday arm use (Motor Activity Log Quality of Movement scale, beta = −.08, r = .26, P = <.001).
Intent-To-Treat Analysis of Comparative Treatment Effects
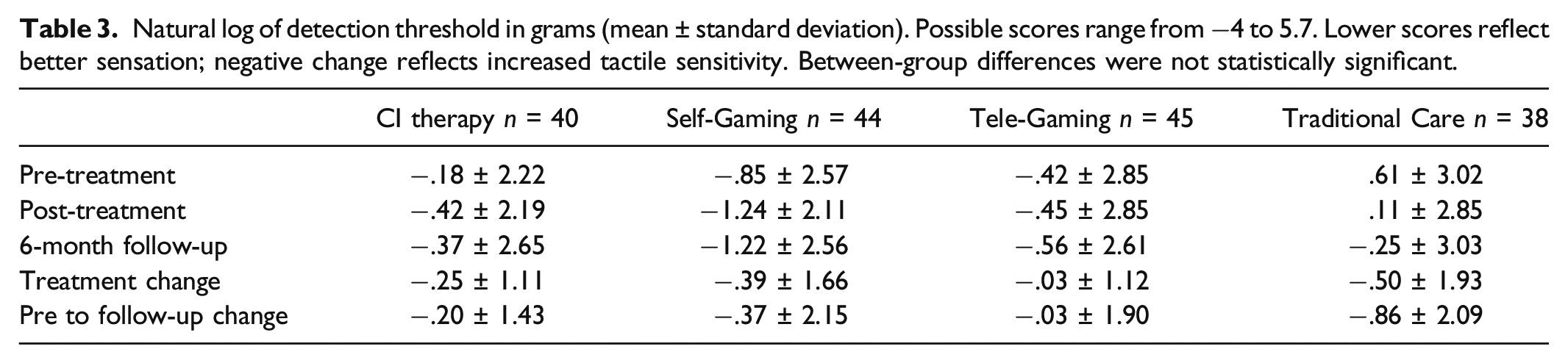
Natural log of detection threshold in grams (mean ± standard deviation). Possible scores range from −4 to 5.7. Lower scores reflect better sensation; negative change reflects increased tactile sensitivity. Between-group differences were not statistically significant.
Effect sizes from the mixed-effect linear model (N = 167). Estimates derived from log-transformed variables approximate, but slightly overestimate, percent improvement. An estimate of −.39 log g reflects a 33% decrease in force required to detect the stimulus. For ease of interpretation, the decrease in grams of force and percent decrease in force required to detect a stimulus are reported alongside the estimates of the treatment and follow-up effects. Negative values reflect improvement.

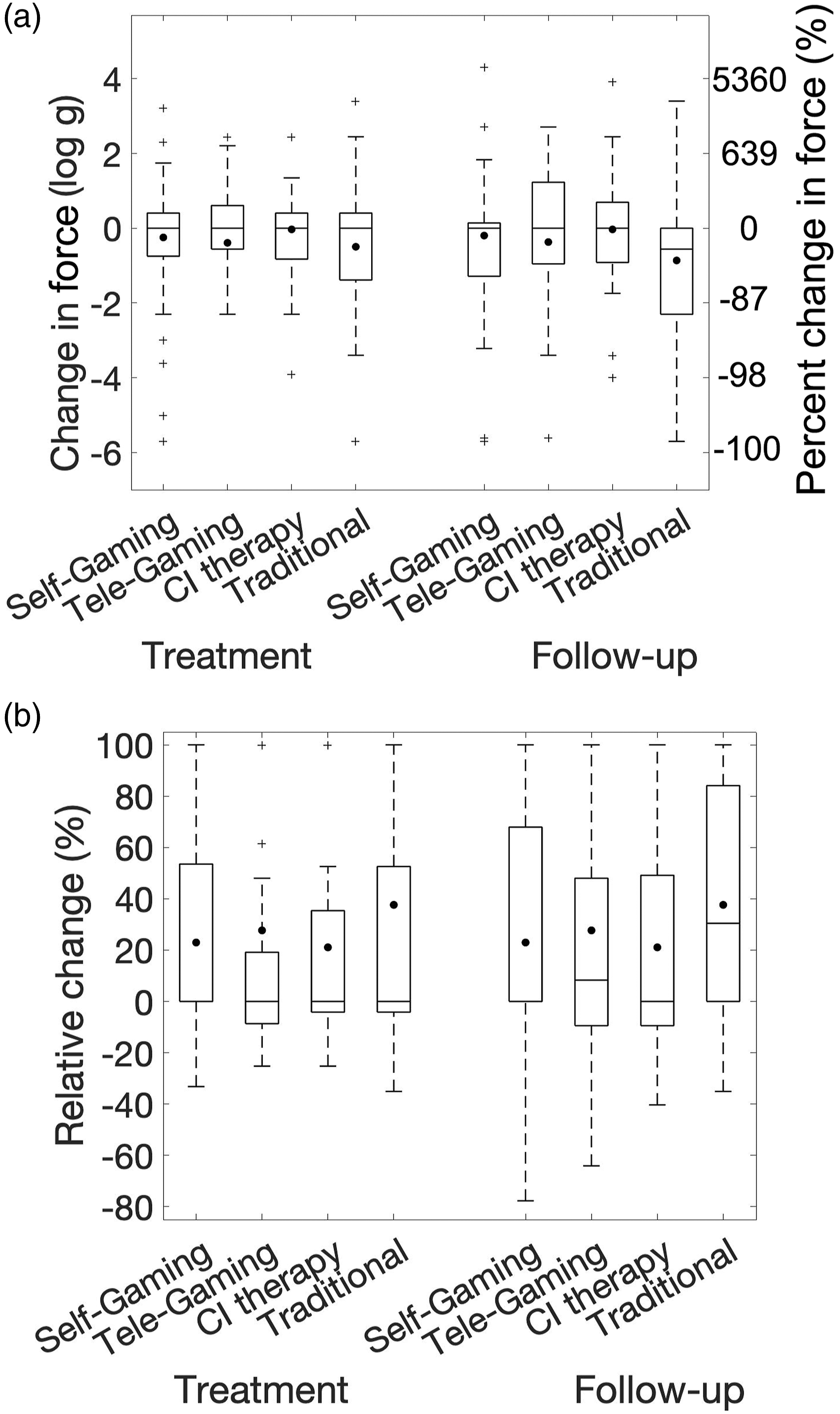
Boxplots illustrating the within-group distributions of the change in tactile force required to detect a stimulus, assessed via Monofilaments. The left y-axis of Figure 1A represents the difference in log g, while the right y-axis expresses this difference as percent change in force. Figure 1B visualizes the data as relative change (change in log force divided by the decrease in log force that would be required to attain normal-range sensation). Negative values in Figure 1A and positive values in Figure 1B reflect improvement. The left 4 boxplots of each figure reflect the treatment change, whereas the right 4 boxplots reflect the change from baseline to 6-month follow-up. The center lines within each box represent the median change, the borders of each box denote the middle 50% of values, the whiskers reflect the most extreme points whose distance from the median is less than 1.5 times the range of the middle 50% of values, and “+” represent more extreme individual data points. The mean change within each group is represented with a small circle. Between-group differences were not statistically significant.
Poorer sensation at baseline was associated with significantly greater change in sensation (P < .001, Table 4). Younger individuals and those with better cognition tended to have better tactile sensation overall, but neither age nor cognition influenced training-induced changes in sensation. Improvements in tactile sense occurred independently of baseline motor performance or motor gains (the mixed-effects model revealed no interaction effects between time and baseline or change WMFT or MAL scores).
Descriptive Results by Baseline Sensory Classification
Of the 129 participants who had impaired tactile sensation at baseline, 33% showed positive change in tactile sensitivity of at least 20% (relative to their maximum possible change); 12% regained normal tactile functioning. Of the subset of participants with severe impairment at baseline (complete loss of protective sensation, detection threshold >4 g,
29
n = 42), 2 (5%) were able to fully regain protective sensation, and 10 (24%) were able to regain some protective sensation. Of the subset of participants with moderate impairment at baseline (diminished protective sensation, detection threshold = .6–2 g,
29
n = 25), one (4%) was able to fully recover the ability to detect light touch (<.07 grams of force) and eight (32%) were able to fully regain protective sensation. Of the subset of participants with mildly impaired sensation at baseline (diminished capacity to detect light touch, detection threshold = .16 - .4 g,
29
n = 62), 15 (24%) were able to recover the ability to detect light touch. Figure 2 displays relative changes separately for individuals with initially severe, moderate, and mild tactile impairments. Histograms of relative change in tactile sense post-treatment for the 129 participants with sensory impairment at baseline (top panel), subdivided by severity of baseline tactile impairment (bottom 3 panels). Severe tactile impairment (2nd panel) denotes individuals who lacked protective sensation. Moderate tactile impairment (3rd panel) denotes individuals who had diminished protective sensation and were unable to sense light touch. Mild impairment (4th panel) refers to individuals for whom protective sensation was preserved, but who had diminished capacity to detect light touch. The x axis reflects an individual’s change as a percentage of their maximal possible change (decrease in force divided by the decrease in force that would be required to attain normal-range sensation). The y-axis reflects the proportion of participants that exhibited relative changes within each 10% bin.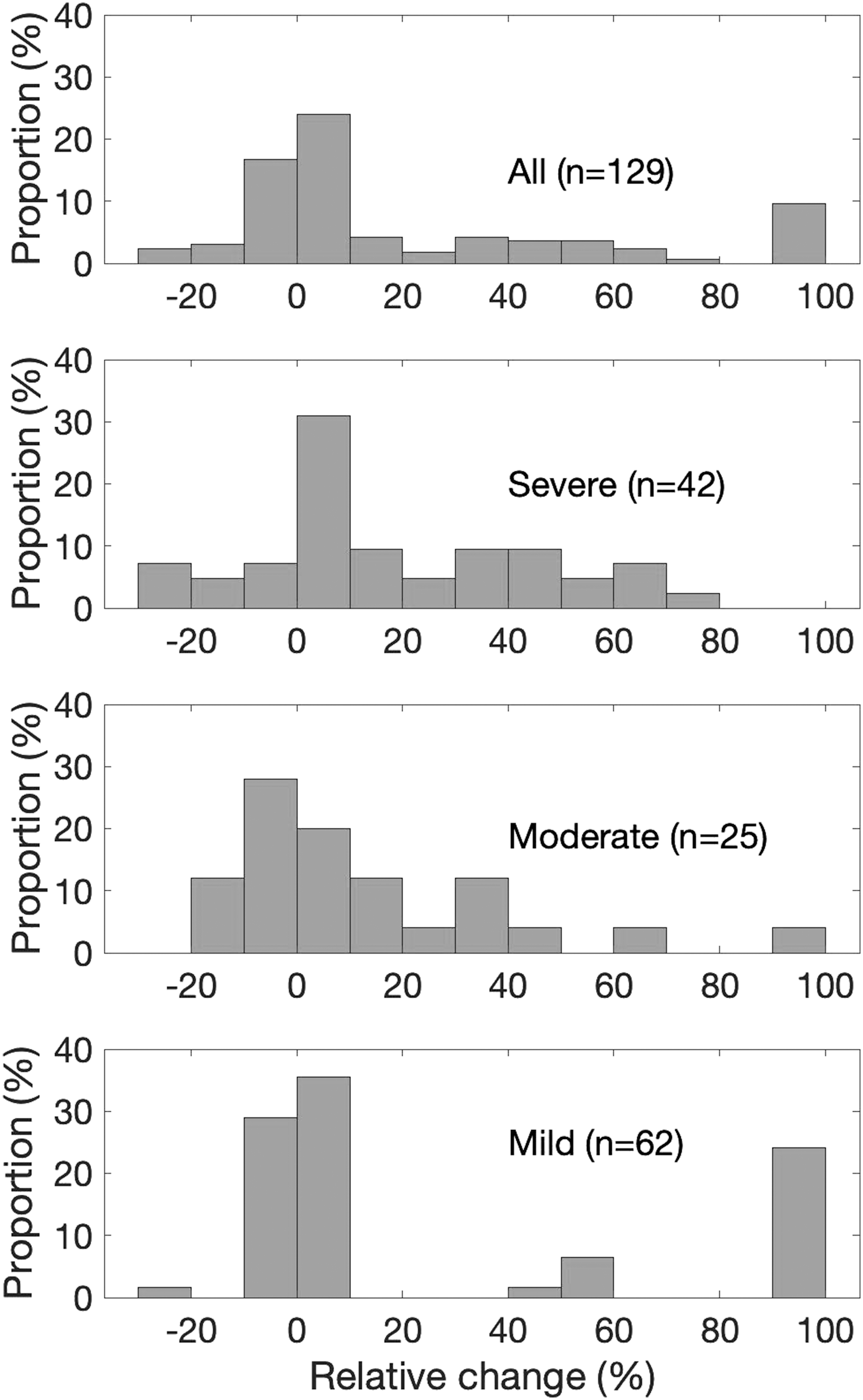
Discussion
Tactile sensitivity of the hand has high functional relevance, 36 so not surprisingly, recovery of tactile sensation is important to people with stroke. 6,37 However, evidence regarding the effects of motor rehabilitation on somatosensory recovery in people with chronic stroke is limited. 7,8,38 To our knowledge, this is the first large RCT of upper limb motor rehabilitation to report improvements in tactile sense. Notably, tactile sense improved for one-third of the participants, and improvements were sustained for at least 6 months. These findings are consistent with prior findings from smaller motor 21,23 and somatosensory 39,40 rehabilitation studies, but extend them in three important ways. First, participants in the current study were on average 4.8 years post-stroke, so spontaneous neurobiological recovery is unlikely to account for the improvements. Second, the large sample size allowed us to characterize the probability of full and partial recovery based on patient characteristics, such as the severity of the initial impairment. Finally, the different treatment groups received varying amounts of tactile input during training (i.e., engaged with real versus virtual objects) and different doses of motor practice (i.e., CI therapy versus traditional rehabilitation), yet experienced similar tactile recovery. This suggests that somatosensory-based rehabilitation, such as sensory discrimination training, may not be required to improve tactile sensitivity in some adults with mild or moderate hemiparesis due to chronic stroke. This interpretation is consistent with that of Tyson et al., following a pooled analysis of lower limb training after stroke; they concluded “…interventions that treat the motor system during functional tasks may be as effective at treating the sensory system as sensory retraining alone.” 41
Even relatively modest doses of motor rehabilitation (i.e., 5 hours of in-clinic therapy plus 5 hours of prescribed home practice) may improve somatosensation. Consistent with this, in a recent study of people with chronic stroke, participants experienced improved wrist position sense accuracy following just two 24-minute sessions of robotic wrist training. 42
An increase in afferent input may drive improved tactile sense following motor rehabilitation. Regardless of whether motor practice involved object manipulation, study participants experienced increased afferent input from the skin, muscle, and joint receptors during motor practice. 43 These sensory inputs have been linked to remodeling of the somatosensory cortex throughout life. 44 -46 Improved functional connectivity in the parietal operculum, 47 thalamic reorganization, 48 improved afferent input processing, 49 and cortical reorganization 50 are all potential neural mechanisms proposed to underlie somatosensory recovery in people with chronic stroke. We suggest that the changes in tactile sensitivity identified here are one aspect of improved sensorimotor performance that supports retention of use and upper limb function among these participants as in previous studies with similar training. 17,26,51
The parallel processing model describes one mechanism by which improved afferent input processing may result in improved somatosensation. During parallel processing, stimuli from cutaneous mechanoreceptors are represented in the brain for their location on the skin surface (e.g. I feel touch on the tip of my index finger) as well as for their location in external space (e.g. the tip of my index finger was touched when it was ∼15 cm anterior to my abdomen, in midline). 52 It was recently determined that the processing of cutaneous stimuli, in these two different ways, happens in parallel and that information from tactile receptors regarding external space feeds back to influence judgments about localization of touch on the surface of the skin. 52 In this study, the repeated opportunities to make judgments about limb position during motor practice or rehabilitation gaming may have resulted in improved afferent input processing. Improved afferent input processing could result in improved tactile sensitivity, as measured here, similar to previous reports of improved proprioception. 41
Limitations of the current study preclude a conclusive determination that the interventions caused sensory improvement. The RCT design resulted in varied tactile input during training, yet sensory input inevitably occurred outside of formal training (e.g., during everyday use of the paretic arm). As sensory experience during daily life was not measured or controlled for, it is thus possible that all groups experienced increased tactile input during the course of the trial that could be sufficient to induce tactile recovery in the absence of any effect of the training itself. Finally, this study design could not rule out natural recovery. Two studies of recovery of upper limb tactile sensation in chronic stroke found it plateaus by 6 months. 53,54 At 4.8 years post-stroke on average, spontaneous recovery is unlikely to account for the improvements. Higher than expected attrition during the follow-up period additionally poses a threat to the study’s internal validity. The follow-up data should thus be interpreted cautiously.
In future research, it will be important to assess additional characteristics that may differentiate between those who improve and those who do not. Also of interest is whether other senses, such as upper limb kinesthesia, improve long-term following motor training. We suggest it may be useful to compare matched doses of motor verses sensory plus motor rehabilitation on somatosensory outcomes. Finally, understanding neural mechanisms associated with this improvement is important as we strive for a solution for the 2/3 of participants who showed no change in tactile sensitivity.
Conclusion
About one-third of people with mild or moderate chronic post-stroke hemiparesis who underwent motor rehabilitation improved their tactile sense, irrespective of the specific treatment modality or prescribed dose of practice. Improvements were fully maintained at the 6-month follow-up. Somatosensory improvements can thus be achieved for a subset of the stroke population through rehabilitation that lacks an explicit somatosensory focus. This information may serve as an additional, important motivator for people living with post-stroke paresis to engage in motor practice.
Footnotes
Acknowledgments
A Stroke Advisory Board collaborated in formulating the study design, determining the recruitment strategy, and interpreting the results. Dr. Ryan J. McPherson, PhD provided technical support. Dr. Mohammed H. Rafiei provided data analysis consultation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Gauthier and Borstad are among the co-founders of Games That Move You, a Public Benefits Corporation that commercialized the gaming technology utilized in this research. To ensure research integrity, conflict management plans were put in place through The Ohio State University prior to conducting this trial.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Patient-Centered Outcomes Research Institute [grant number AD-1409-20772 to The Ohio State University].
