Abstract
Background. The Arm Motor Ability Test (AMAT) is used to assess and quantify upper-extremity (UE) functional limitation in stroke and other conditions. However, the AMAT score change indicative of important and clinically meaningful change has not been determined. Objective. To determine the clinically important difference (CID) for the AMAT for individuals with stroke exhibiting mild to moderate hemiparesis. Methods. A total of 146 chronic stroke survivors exhibiting stable, mild to moderate UE hemiparesis were administered the AMAT before and after interventions targeting their affected UEs. Patients and treating therapists rated perceived amount of UE motor recovery for each participant on a global rating of change (GROC) scale evaluating several facets of UE movement (grasp, release, move the affected UE, perform 5 important functional tasks, overall UE function). Estimated CID of the Functional Ability Scale of the AMAT was calculated using the receiver operating characteristics curve with the GROC scale as the anchor. Distribution-based methods were also used to estimate the CID. Results. Mean baseline, postintervention, and change in AMAT values for all participants were 3.0 (0.68), 3.3 (0.73), and 0.33 (0.43) respectively. The CID was estimated as an improvement of 0.32 to 0.42 when anchored by the therapist’s perception of improvement and 0.29 to 0.40 when anchored by the patient’s perception of improvement. The CID using distribution-based methods ranged from 0.40 to 0.44. Conclusions. A change of 0.44 or greater on the AMAT indicates a clinically meaningful improvement in UE functional movements. Clinicians should use this value to determine goals and interpret change scores.
Introduction
Upper-extremity (UE) hemiparesis remains a common motor impairment exhibited following a variety of neurological and musculoskeletal disorders. When these debilitating conditions occur, measurement of clients’ UE functional limitation is critical to designing cost-effective, appropriate interventions and in determining whether clients are increasing their ability to perform activities.
The Arm Motor Ability Test (AMAT) 1 is often used to measure UE functional limitation in rehabilitative trials enrolling individuals with stroke2-4 and is applicable to other neurological and musculoskeletal conditions in which UE weakness occurs (eg, spinal cord injury, amyotrophic lateral sclerosis, peripheral nerve injury). The measure requires clients to perform 13 common unilateral and bilateral UE tasks. Performance on each task is timed by the evaluator and rated by the evaluator using two 6-point ordinal scales: the Functional Ability and Quality of Movement scale.
Anchor-based computation of the clinically important difference (CID) determines the relationship between an outcome measure (eg, the AMAT) and an “anchor” to determine if the change in the outcome measure was clinically meaningful. Anchors are often a self-reported opinion of change by patients, clinicians, family members, or other individuals of interest.5,6 Ultimately, the value of CIDs is their utility for clinicians and researchers to objectively interpret the importance of score changes that their clients exhibit and, in the case of the AMAT, the likelihood that reductions in UE functional limitation will translate to a client being more capable of performing common tasks integrating the paretic UE. Although the AMAT’s reliability and validity have been established poststroke 1 and the importance of CIDs in clinical decision making is widely appreciated, 7 the AMAT’s ability to detect important changes after an intervention has not yet been determined. Previous studies have cited a change of 0.21 on the functional ability scale of the AMAT as being clinically meaningful.3,8,9 However, none of these studies or the ones they cite1,2,10 actually determined the CID of the AMAT using either anchor- or distribution-based methods. When comparing the dosage of UE task-specific training paired with electrical stimulation, Page et al 2 found that the group with the highest dosage had a mean change on the AMAT of 0.21, which was significantly different from that of the 2 other groups with a lower dosage. Although this change was statistically different compared with the other 2 groups, it does not reflect clinically important change.
The purpose of the current study was to estimate the CID of the AMAT in patients with chronic stroke exhibiting minimal to moderate UE hemiparesis using global rating of change (GROC) of the evaluating physical and occupational therapists and participants as anchors of clinically important change. Although the AMAT can be used with a number of groups, the stroke population was chosen because of the frequency of their involvement in UE rehabilitative programs.
Methods
Study Design
This study was a secondary analysis of data from the Everest randomized controlled trial of implanted cortical stimulation for UE function in chronic stroke. 3 Following administration of the AMAT as well as several other UE measures, participants were randomized to either (1) a control group, in which motor learning–based, repetitive task-specific practice targeting the affected UE was administered for approximately 2.5 h/weekday over a 6-week period, or (2) a treatment group, in which the above repetitive task-specific practice regimen was coadministered with electrical cortical stimulation by the Northstar Stroke Recovery System, as described elsewhere. 3 This study reports only on the CID of the AMAT obtained from change in the AMAT between preintervention and postintervention using anchor-based methods described below, whereas changes from preintervention to postintervention periods between the control and treatment groups are described elsewhere. 3
Participant Recruitment and Study Criteria
Participants were recruited from across the United States using print advertisements placed in clinics near enrolling sites and in national periodicals, radio advertisements in the markets of enrolling sites, and in-person visits to stroke support groups and clinicians working with stroke survivors. As volunteers came forward, the following screening criteria were applied: inclusion criteria were ischemic stroke >4 months, medically and neurologically stable, mild to moderate hemiparesis of the affected UE, and age ≥21 years; exclusion criteria were hemorrhagic stroke, more than 1 stroke, other neurological condition that resulted in impaired affected UE, moderate to severe neglect or anosognosia, severe sensory impairment, inability to understand and cooperate with the study, and severe spasticity of the affected UE. Appendix A contains a full list of specific inclusion/exclusion criteria.
Instruments
The AMAT 1 contains 13 unimanual and bimanual activities of daily living (ADL) compound tasks such as cutting meat, opening a jar, and combing hair. Each of the 13 compound tasks is subdivided into task components of the main compound task. For example, the ADL compound task “open jar” is subdivided into “grasp jar top” and “screw jar top open” task components. This results in a total of 28 task components that are rated according to a Functional Ability Scale that examines paretic limb use (0 = does not perform with paretic UE; 5 = does use arm at a level comparable to less affected side) and a Quality of Movement Scale (0 = no movement initiated; 5 = normal movement). As part of the Everest trial, 3 the AMAT was administered by a blinded rater at each participating center at which the Everest study was being conducted. All raters were certified and recertified on the AMAT and the other outcome measures every 3 months using standardized, live and video-based interrater reliability checks at the main study center.
Immediately after the 6-week intervention period, each treating therapist rated the amount of improvement exhibited by each participant in 5 different aspects of affected UE function: (1) ability to grasp objects, (2) ability to release objects, (3) ability to move the affected UE, (4) ability to perform the 5 most important activities identified by the participant in the Canadian Occupational Performance Measure (COPM), 11 and (5) overall arm and hand function. This was accomplished using a 5-point, observation-based, GROC scale (Table 1). This GROC has been used previously to determine the CID of the UE motor component of the Fugl-Meyer. 12 GROC scales are commonly used as an anchor to determine clinically important change on outcome measures.6,7,12-14 Using this method, patients, health care providers, or another party of interest rate the amount of change they perceive that they (or their patient) have experienced over the course of the intervention using a GROC scale.5-7,13,15 In addition to the treating therapist, participants rated their perceived change in their affected UE on the same 5 aspects of UE function/movement as the treating therapists did: ability to grasp objects, ability to release objects, ability to move the affected UE, ability to perform the 5 most important activities identified by the participant in the COPM, and overall arm and hand function on the GROC. Having participants rate their change on a GROC provides a patient-centered approach to estimating the CID of an outcome measure and the interpretation of change on that outcome measure. 13 Because of the complex nature of CID, it is important to triangulate estimates of CID using multiple anchors. 7 In this study, we used the treating therapists’ perception of participant change and participants’ perception of change on 5 different aspects of UE function/movement measured on the GROC as anchors to estimate important change on the AMAT.
Global Rating of Change Scale.

Data Analysis
Because there were no significant differences between the intervention and control groups, 3 AMAT data from both groups were combined to calculate the CID of the AMAT. AMAT baseline, postintervention, and change data were analyzed using descriptive statistics. Participants were dichotomized into 2 groups based on scores from the treating therapists and participants’ ratings of improvement using the previously described 5-point ordinal GROC scale for each of the 5 different aspects of UE function (grasp, release, move the UE, ability to perform identified COPM tasks, and overall arm and hand function). This resulted in 10 separate anchors and estimated CID values of the AMAT. For each anchor, those who received a score of 5 were considered to have experienced clinically important improvement, and those who scored below 5 were considered to not have experienced clinically important improvements. A cutoff score of 5 to identify clinically important change was selected because other researchers have indicated a need to identify changes in outcome measures that are of high clinical importance and are more than minimally clinically important.7,12,13
Independent t-tests were performed comparing scores on the AMAT between the dichotomized groups of important improvement and not an important improvement to further validate the GROC. There should be a statistically significant difference in AMAT scores between those with perceived clinically important improvement on the GROC (score of 5) and those without (score <5 on the GROC).
Receiver operating characteristic (ROC) curves were next constructed by plotting sensitivity values (true positive rate) on the y-axis and specificity values (false positive rate) on the x-axis for different changes in AMAT scores for distinguishing participants who demonstrated highly important improvement (5 on the GROC) from those who did not have highly important improvement (<5 on the GROC). In all, 10 separate ROC curves were constructed for therapists’ and participants’ perception of improvement related to the different aspects of UE function (grasp, release, move UE, COPM score, and overall). The area under the curve (AUC) and 95% CIs were obtained as a method for describing the ability of the AMAT to distinguish participants who improved from those who did not improve. The CID of the AMAT was selected by identifying the point on the ROC curve in the uppermost left corner, which is considered the best estimate for distinguishing between those with and without clinically important improvement.
The sensitivity and specificity of the CID was calculated as well. This was done for each ROC curve, so that a CID score and the corresponding sensitivity and specificity for the AMAT were estimated for each of the 2 different anchors (treating therapists’ and participants’ GROC) and the 5 different aspects of UE function (grasp, release, move UE, ability to perform COPM tasks, and overall arm and hand function).
In addition to the anchor-based methods described above to determine the CID of the AMAT, we used distribution-based methods to determine the minimal detectable change (MDC) of the AMAT.5,13,15-17 The MDC was calculated using the standard error of measure (SEM) for the AMAT from the groups that were categorized as not having clinically important improvement (GROC < 5) on the 5 different aspects of UE movement/function. This was done because these groups were considered stable. 18 The SEM was calculated using the formula (SD × [1 − r]1/2) where r is the test-retest reliability coefficient.5,17 We used a test-retest reliability of 0.93 as reported by Kopp et al, 1 and SD is the square root of the total variance. The SEM was multiplied by 1.65 to determine the 90% CI.5,17 This value was multiplied by the square root of 2 to take into account multiple measurements.5,17
Results
Participant Demographics
Using the previously mentioned study criteria, 146 participants were included in the current analysis (87 male; mean age of all participants = 57.1 ± 10.96 [SD] years; range of ages of participants = 29-83 years; mean time since stroke onset for participants in sample = 59.37 ± 63.22 [SD] months; 88 participants with hemiparesis affecting their right UEs; 82 participants with hemiparesis affecting their dominant UEs).
Validity of the GROC
Mean (SD) baseline, postintervention, and change in AMAT values for all participants were 3.0 (0.68), 3.3 (0.73), and 0.33 (0.43), respectively. For the therapists’ rating of improvement groups, there was a significant difference in change in AMAT scores between those with and without clinically important improvement for grasp, release, ability to perform COPM activities, and overall arm and hand function but not for ability to move the affected UE (Table 2). For the participant-perceived rating of improvement groups, there was a significant difference in change in AMAT scores between those with and without CID for grasp, release, ability to perform COPM activities, and overall arm and hand function but not for ability to move the affected UE (Table 2).
Mean Change in AMAT Scores of Participants With Clinically Important Change (GROC = 5) and Participants Without Clinically Important Change (GROC < 5).
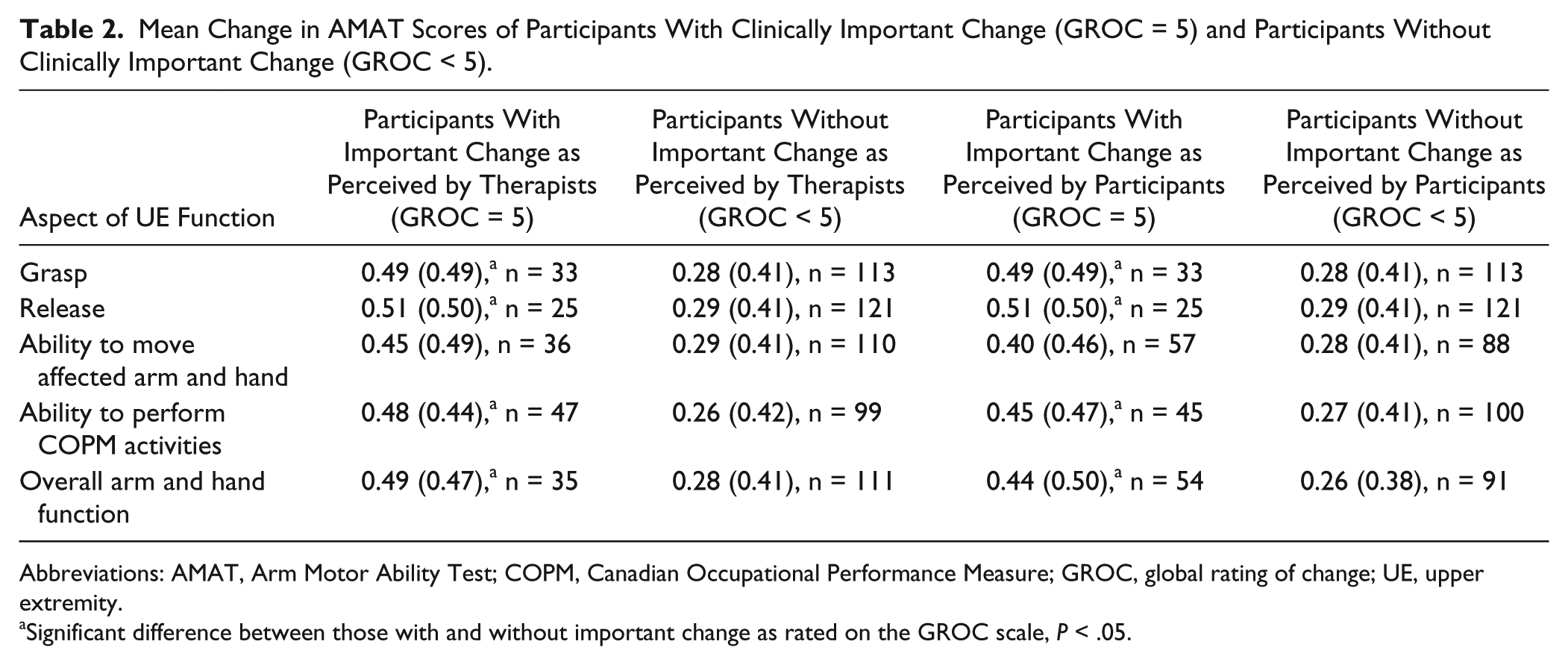
Abbreviations: AMAT, Arm Motor Ability Test; COPM, Canadian Occupational Performance Measure; GROC, global rating of change; UE, upper extremity.
Significant difference between those with and without important change as rated on the GROC scale, P < .05.
Anchor-Based CID Computations
The ROC curve analysis revealed that change in AMAT score was able to distinguish between participants who experienced clinically important improvement (GROC score = 5) and those who did not (GROC score < 5) based on the treating therapists’ perception of improvement in grasp ability, release ability, ability to move the affected UE, ability to perform COPM activities, and overall arm and hand function. The area under the curve ranged from 0.62 to 0.66, the sensitivity ranged from 0.64 to 0.58, and the specificity ranged from 0.70 to 0.62. The estimated CID for the AMAT with the therapists’ perception of improvement for the 5 different aspects of affected UE function ranged from 0.42 to 0.34 (Table 3 and Figure 1).
Clinically Important Change in AMAT Based on Therapists’ Perception of Change (GROC = 5).
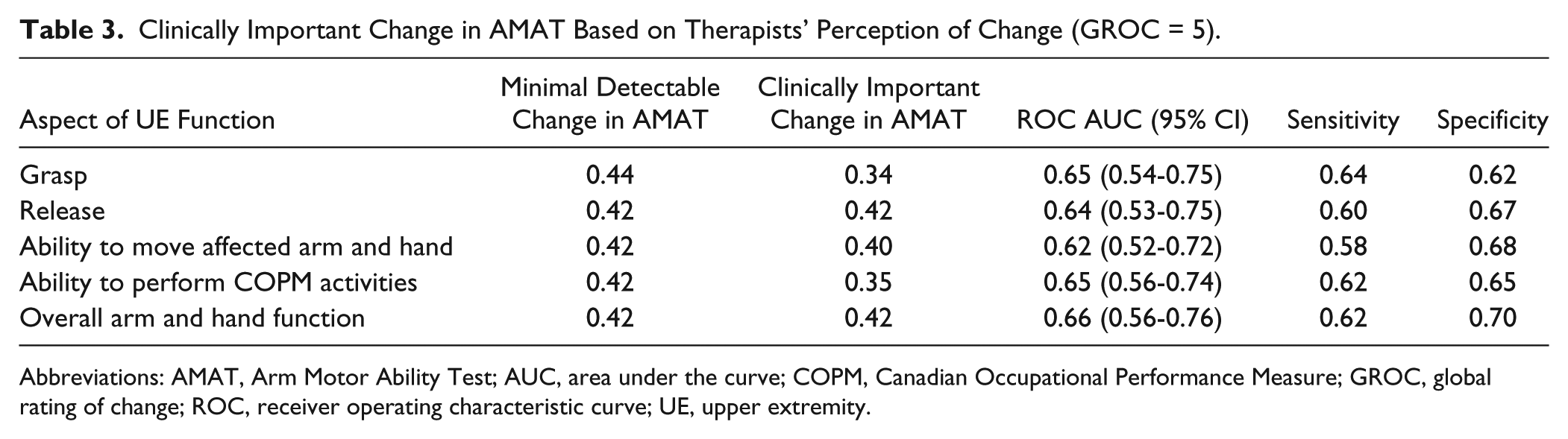
Abbreviations: AMAT, Arm Motor Ability Test; AUC, area under the curve; COPM, Canadian Occupational Performance Measure; GROC, global rating of change; ROC, receiver operating characteristic curve; UE, upper extremity.
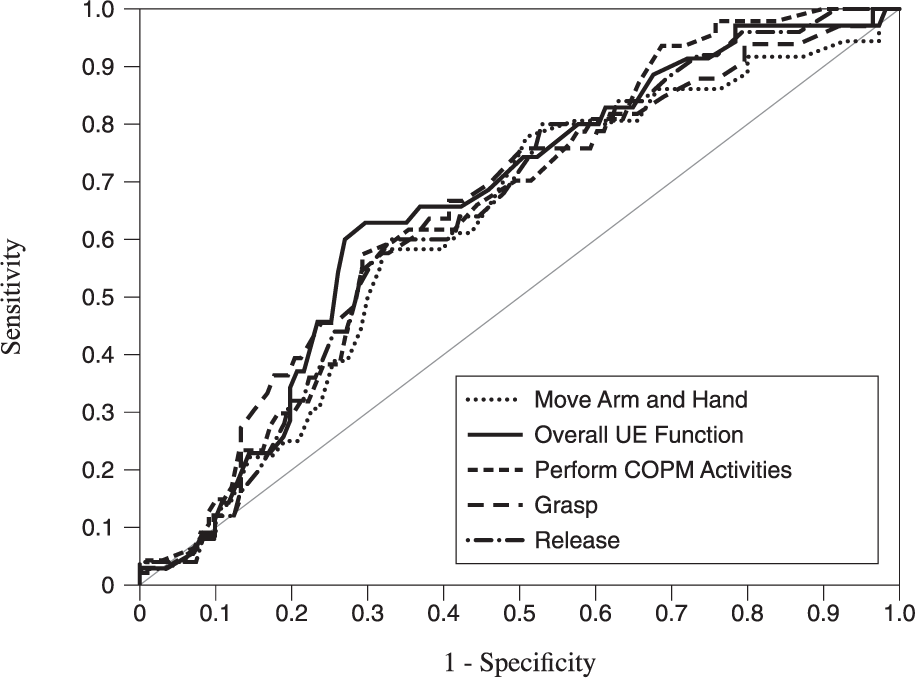
Receiver operating characteristic curves for therapists global ratings of change scores on upper extremity function and movement.
The ROC curve analysis revealed that change in AMAT score was able to distinguish between participants who experienced clinically important improvement (GROC score = 5) and those who did not (GROC score < 5) based on the participants’ perception of improvement in grasp ability, release ability, ability to perform COPM activities, and overall arm and hand function. The area under the curve ranged from 0.61 to 0.66, the sensitivity ranged from 0.67 to 0.50, and the specificity ranged from 0.68 to 0.58. The estimated CID for the AMAT with the participants’ perception of improvement for the 4 different aspects of affected UE function ranged from 0.40 to 0.29. Change in AMAT scores were not able to distinguish between participants who experienced clinically important improvement and those who did not based on the participants’ perception of improvement in ability to move the affected UE; AUC 95% CI <0.50 (Table 4 and Figure 2).
Clinically Important Change in AMAT Based on Participants’ Perception of Change (GROC = 5).
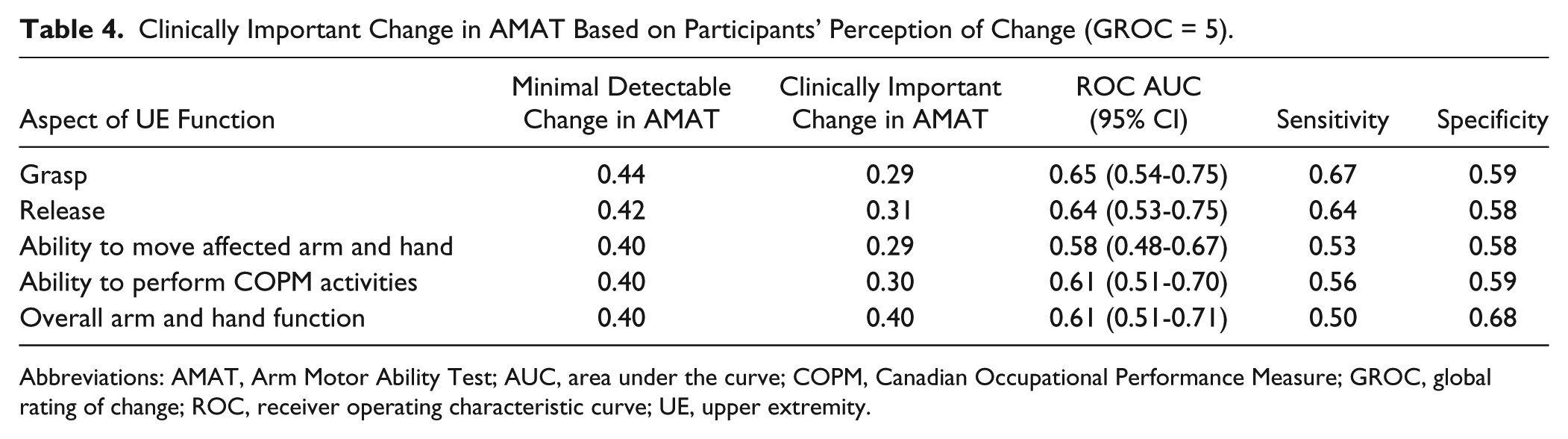
Abbreviations: AMAT, Arm Motor Ability Test; AUC, area under the curve; COPM, Canadian Occupational Performance Measure; GROC, global rating of change; ROC, receiver operating characteristic curve; UE, upper extremity.
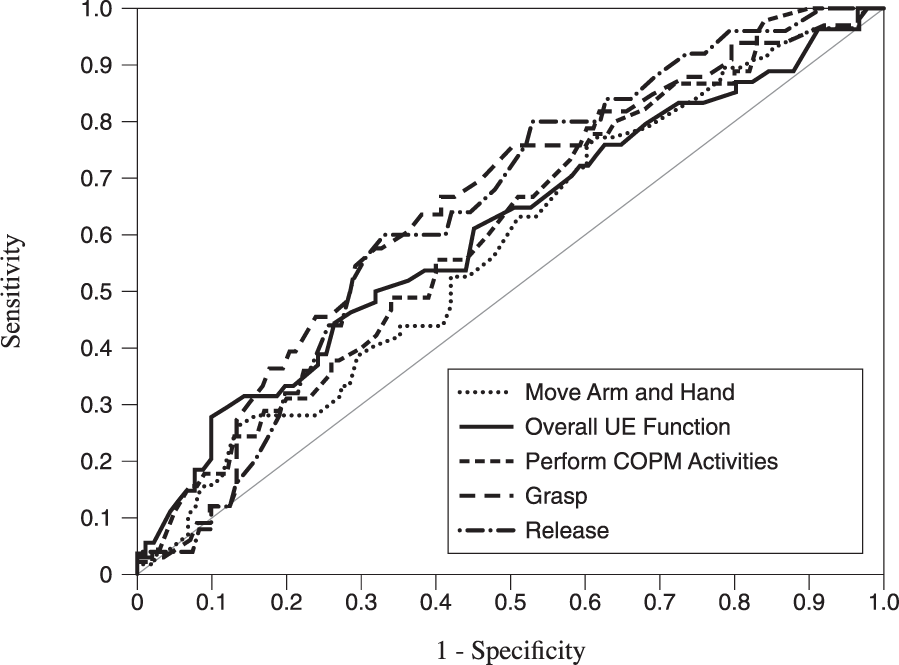
Receiver operating characteristic curves for subjects global ratings of change scores on upper extremity function and movement.
Distribution-Based Computation of MDC
The MDC for the AMAT based on the groups without clinically important change based on therapists’ perception (GROC < 5) for the 5 different aspects of affected UE function ranged from 0.42 to 0.44 (Table 3). The MDC for the AMAT based on the groups without clinically important change based on participants’ perceptions’ (GROC < 5) for the 5 different aspects of affected UE function ranged from 0.40 to 0.44 (Table 4).
Discussion
To the best of our knowledge, this is the first study to estimate CID values for the AMAT. Our findings using multiple anchors of importance indicate that a change between 0.29 and 0.42 on the AMAT for people with a mild to moderate degree of UE hemiparesis who are undergoing an intensive, task-oriented UE intervention can be considered a clinically important amount of change. Using distribution-based methods, the estimated MDC of the AMAT is between 0.40 and 0.44. Clinicians and researchers can use these estimated clinically important change values to assist with interpreting change scores from baseline in the AMAT in people with stroke with similar characteristics to those in this study.
Functional use of the UE requires a complex interaction among sensory, cognitive, and motor systems. People poststroke often have impairments in all these areas, and functional recovery of the UE is limited in many individuals. It is estimated that between 33% and 66% of people poststroke do not regain any functional ability after stroke19,20 and that only 5% to 20% will exhibit full functional recovery of the paretic UE. 21 Because of this complexity of UE function and limited recovery, a strength of our study is the use of multiple anchors that took into account both therapists’ and participants’ perception of important change related to different aspects of UE movement and function (ability to grasp and release, move the arm and hand, ability to perform 5 important ADL tasks, and overall arm and hand function). CID values for an outcome measure are context specific. 7 The use of different anchors improves the clinical applicability of the AMAT for clinicians and researchers seeking to utilize it in different or multiple contexts.
Clinicians and researchers can use the estimated CID values of the AMAT from this study when interpreting the impact of rehabilitation interventions targeted at the UE for individuals who are poststroke with mild to moderate hemiparesis. Because there were different estimated CID values (0.29-0.42 for anchor-based methods and 0.40-0.44 for distribution-based methods) across the different aspects of UE function/movement for both therapists’ and participants’ perception, conservatively, 22 a change of 0.44 or greater in the AMAT is likely clinically meaningful to both patients and clinicians and indicates clinically meaningful improvement in a variety of important UE functions and movements (ability to grasp and release, move the arm and hand, perform important ADLs, and overall function). For an individual patient, an improvement on the AMAT ≥0.42 can be an indication to the clinician that the patient experienced important change in UE function. A change of <0.42 in the AMAT may indicate that the selected intervention strategy needs to be changed or possibly that the patient’s UE function has plateaued. Researchers can use the estimated CID as a criterion for efficacy of the experimental intervention. The proportion of participants in the experimental group who exceeded the estimated CID value can be compared with the proportion of participants in the comparison group to determine the number needed to treat, 23 which is a clinically relevant method to compare the effectiveness of rehabilitation interventions.
A 10% change in the Fugl-Meyer and the Stroke Impact Scale has been suggested as a criterion for clinically important improvement.24,25 The CID values estimated in this study range from 5.8% to 8.8%. These percentages are within the range for estimated CIDs for other tests and measures that assess UE movement and function after stroke. A 6.4% to 15.1% change in the UE motor section of the Fugl-Meyer has been estimated to be clinically important.12,26 Lang et al 14 estimated that a 20% to 31% change in the Wolf Motor Function Test, Action Research Arm Test, and the Motor Activity Log is clinically meaningful in the early stages of recovery after stroke. The difference between studies is likely a result of differences in anchors, chronicity, and severity of participants, the outcome measures assessed, and methods used to calculate the CID. For example, the Fugl-Meyer assesses the ability to move the affected UE out of abnormal synergistic patterns that are common after stroke, whereas the AMAT assesses the ability to complete functional tasks. In the study by Lang et al, participants were 26 days poststroke and still in the hospital. In this early stage of recovery after stroke, perhaps a greater change is necessary for it to be perceived as important by people with stroke. Additionally, the methods used by Lang et al to determine the CID of the various outcome measures they examined do not allow for calculations of the accuracy of the different cutoff values they found.
The strength of the estimated CID values can be assessed based on the AUC and the resulting sensitivity and specificity associated with the specific CID values. If the low end of the 95% CI of the AUC is ≤0.50, then it indicates that the identified change in AMAT cutoff score has no ability beyond chance to distinguish between those with clinically important improvement and those without. All the CID values for the AMAT associated with the various anchors, except for the CID associated with participants’ perception of change in ability to move the affected arm and hand, are able to distinguish between those with clinically meaningful improvement and those who did not experience clinically important change (AUC 95% CI > 0.50). The resulting sensitivity and specificity values ranged from 0.50 to 0.70. These are likely relatively low because of the small number of participants who had important improvement (GROC score = 5) on the various anchors (ranged from 25 to 57). Although our overall sample size was large (146), to have sensitivity and specificity values in the mid-0.70s, a total of approximately 78 participants reporting important change would be necessary. 27
Similar to intervention studies, estimated CID values are context specific. 7 The estimated CID values for the AMAT from this study should be used only with patients and participants who have characteristics (>4 months poststroke with mild/moderate hemiparesis) similar to those in this study and who are undergoing repetitive task practice.
A strength of this study is the use of multiple anchors (both therapists’ and participants’ perception of improvement on 5 different aspects of UE function/movement) and distribution-based methods to estimate the CID of the AMAT. The resulting CIDs are very similar across these different anchors and methods. They only vary by 3% of the total score of the AMAT (0.29-0.44).
Conclusions/Implications
Although UE functional limitation is an important construct for measuring status and discerning response to intervention, the amount of functional limitation change that one must exhibit to be clinically significant has heretofore not been measured. Results from this study indicated that a change ≥0.44 on the AMAT is indicative of clinically meaningful improvement in important, common UE functional movements. With this information, clinicians will be able to use this value to determine appropriate goals and interpret change scores of the AMAT.
Footnotes
Appendix
Study Inclusion and Exclusion Criteria
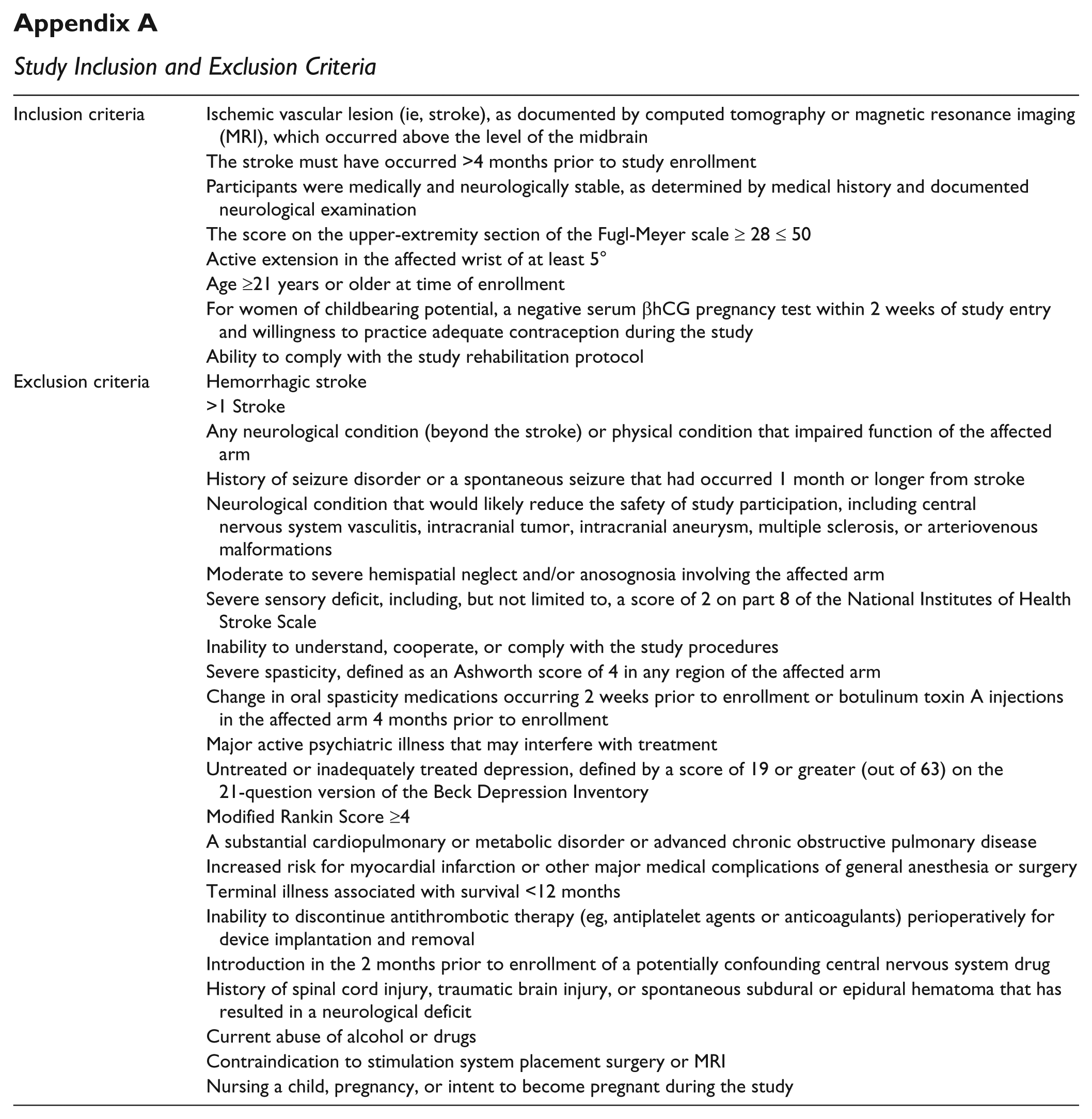
| Inclusion criteria | Ischemic vascular lesion (ie, stroke), as documented by computed tomography or magnetic resonance imaging (MRI), which occurred above the level of the midbrain |
| The stroke must have occurred >4 months prior to study enrollment | |
| Participants were medically and neurologically stable, as determined by medical history and documented neurological examination | |
| The score on the upper-extremity section of the Fugl-Meyer scale ≥ 28 ≤ 50 | |
| Active extension in the affected wrist of at least 5° | |
| Age ≥21 years or older at time of enrollment | |
| For women of childbearing potential, a negative serum βhCG pregnancy test within 2 weeks of study entry and willingness to practice adequate contraception during the study | |
| Ability to comply with the study rehabilitation protocol | |
| Exclusion criteria | Hemorrhagic stroke |
| >1 Stroke | |
| Any neurological condition (beyond the stroke) or physical condition that impaired function of the affected arm | |
| History of seizure disorder or a spontaneous seizure that had occurred 1 month or longer from stroke | |
| Neurological condition that would likely reduce the safety of study participation, including central nervous system vasculitis, intracranial tumor, intracranial aneurysm, multiple sclerosis, or arteriovenous malformations | |
| Moderate to severe hemispatial neglect and/or anosognosia involving the affected arm | |
| Severe sensory deficit, including, but not limited to, a score of 2 on part 8 of the National Institutes of Health Stroke Scale | |
| Inability to understand, cooperate, or comply with the study procedures | |
| Severe spasticity, defined as an Ashworth score of 4 in any region of the affected arm | |
| Change in oral spasticity medications occurring 2 weeks prior to enrollment or botulinum toxin A injections in the affected arm 4 months prior to enrollment | |
| Major active psychiatric illness that may interfere with treatment | |
| Untreated or inadequately treated depression, defined by a score of 19 or greater (out of 63) on the 21-question version of the Beck Depression Inventory | |
| Modified Rankin Score ≥4 | |
| A substantial cardiopulmonary or metabolic disorder or advanced chronic obstructive pulmonary disease | |
| Increased risk for myocardial infarction or other major medical complications of general anesthesia or surgery | |
| Terminal illness associated with survival <12 months | |
| Inability to discontinue antithrombotic therapy (eg, antiplatelet agents or anticoagulants) perioperatively for device implantation and removal | |
| Introduction in the 2 months prior to enrollment of a potentially confounding central nervous system drug | |
| History of spinal cord injury, traumatic brain injury, or spontaneous subdural or epidural hematoma that has resulted in a neurological deficit | |
| Current abuse of alcohol or drugs | |
| Contraindication to stimulation system placement surgery or MRI | |
| Nursing a child, pregnancy, or intent to become pregnant during the study |
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The data from this study were originally supported by a grant from Northstar Neuroscience.
