Abstract
Background
Most individuals with Parkinson’s disease (PD) develop balance dysfunction. Previous studies showed that individuals with PD have abnormal corticomotor changes related to severity of motor symptoms and disease progression. Cortical disinhibition was observed in PD and this alteration can be an early sign of PD. Balance training seems to be an effective intervention to improve balance in individuals with PD. However, it is not much known about the effect of balance training on cortical neuroplasticity in PD population.
Objective
To investigate the effects of balance training on corticomotor excitability in individuals with PD.
Methods
Twenty-eight PD participants were recruited and randomly assigned to either the balance training (BT) or the control (CON) group. Both groups underwent 16 training sessions over 8 weeks. Outcome measures for corticomotor inhibition included the cortical silent period (CSP) and short-interval intracortical inhibition (SICI) on transcranial magnetic stimulation. Balance performance was measured using the Mini-Balance Evaluation Systems Test (Mini-BEST) and the Timed Up and Go (TUG) test.
Results
Participants in the BT group showed a significant increase in corticomotor inhibition (CSP: P = .028, SICI: P = .04) and a significant improvement in balance performance (Mini-BEST: P = .001, TUG: P = .04) after training. Compared to the CON group, the BT group showed a greater increase in corticomotor inhibition (CSP: P = .017, SICI: P = .046) and better improvement in balance (Mini-BEST: P = .046).
Conclusion
Balance training could modulate corticomotor inhibition in the primary motor cortex and improve balance performance in individuals with PD.
Keywords
Introduction
Parkinson’s disease (PD) is one of the most common neurodegenerative disorders in which progressive loss of dopaminergic neurons in the basal ganglia give rise to multiple motor and non-motor symptoms. 1 Most of the individuals with PD develop balance impairment, which worsens with disease progression. 1
Transcranial magnetic stimulation (TMS) provides useful information about cortical function and the pathophysiology of underlying motor disturbances in PD. Previous TMS studies have found abnormalities in the excitability and plasticity of the cortical motor areas in PD.2,3 Electrophysiological characterization of individuals with PD has shown increased corticospinal excitability, decreased corticospinal inhibition, and decreased intracortical inhibition when compared with healthy subjects.4 -6 According to the TMS evidence, the most congruous findings in PD include a shortened cortical silent period (CSP) and a decreased short-interval intracortical inhibition (SICI) at rest. Abnormal inhibitory activity may cause excessive neuronal firing and activity. Moreover, defective intracortical inhibition would result in a lack of focusing activity on appropriate groups of neurons for each movement. 6 Possible mechanisms of the changes may involve secondary effects of the abnormal basal ganglia output on the primary motor cortex (M1), adaptations within the M1 to disease symptoms, or subtle changes in dopaminergic innervation of the M1 as the disease progress in PD. 7 The abnormal changes in TMS findings may be related to the severity of motor symptoms and clinical progression in PD.8,9 Although these abnormalities may be reversed by levodopa or dopamine agonists, the results are still inconsistent. 10
Increasing evidence supports the involvement of the M1 in postural control.11,12 Corticomotor excitability can flexibly change according to the posture for modulating balance and stability.13,14 According to TMS studies, the older adults had poor postural stability and more reduction of cortical inhibition, compared to young adults. 15 The modulation of corticomotor excitability and inhibition in the M1 seems to significantly contribute to balance performance, yet there is still insufficient evidence in the PD population. It needs to be further confirmed possible underlying neural mechanisms that involved in balance improvement after training.
The aim of this study was to investigate the effects of balance training on corticomotor excitability and balance performance in people with PD. We hypothesized that balance training can induce changes in corticomotor inhibition and improvement in balance performance in PD.
Methods
Study Design and Participants
In this single-blinded randomized controlled trial, participants were randomly assigned to either the experimental group that received balance training (BT) or the control (CON) group that received regular physical therapy including strength and coordination training. A researcher who was not involved in this study used simple randomization by generating a random digit to assign participants, and worked as an evaluator to perform all evaluations. This research didn’t know what the next group allocation would be and didn’t engage in defining the eligibility of subjects. All participants were evaluated at baseline and at the end of the intervention. The duration of the intervention was 8 weeks in both the groups. The study procedures were in accordance with the principles of the Declaration of Helsinki. This study was approved by the Institutional Review Board. Written informed consent was obtained from all the participants before participation. Data were collected at the National Yang Ming Chiao Tung University. The trial was registered in the Thai Clinical Trials Registry (identifier TCTR 20180111003).
Twenty-eight participants with PD from a medical center were recruited between March 2018 and August 2020. Inclusion criteria were clinical diagnosis of idiopathic PD according to the United Kingdom Parkinson’s Disease Society Brain Bank Diagnostic Criteria, 16 Hoehn and Yahr stage (H&Y) 1-3, age between 50 and 85 years, stable anti-PD medications for at least 2 weeks, and the ability to walk 10 m with or without any assistive device. Exclusion criteria were contraindications to TMS, severe musculoskeletal or cardiopulmonary disorders, neurologic conditions other than PD, Mini-Mental State Examination (MMSE) score <24, and participants who attended another exercise program in the previous 3 months.
Interventions
Participants in the BT group followed a specific training program called square-stepping exercise (SSE). Participants were instructed to perform a sequence of different steps on a thin mat (100 × 250 cm) with 40 squares and to complete the stepping pattern smoothly. The exercise session enabled subjects to practice weight-shifting in different directions using different step distances and challenged their postural stability. The principles and the patterns of the SSE program were based on a previous study. 17 If someone was stuck in a certain stepping pattern, it would be skipped. In the CON group, participants received strengthening and coordination training. The training protocol consisted of upper-limb and lower-limb resistance training with sand bags, postural re-education, and hand-eye coordination exercises (e.g., upper ball throwing and catching). The program of strengthening focused on the muscles of the upper and lower limbs, which were related to functional movements such as shoulder flexors, abductors, external rotators, elbow flexors, extensors, hip flexors, extensors, abductors, knee flexors, and extensors.
Subjects from both the groups underwent training for 60 minutes per session twice a week for 8 weeks. Each training session began with a 10-minute warm-up followed by a main training program for 40 minutes and ended with a 10-minute cool-down. The warm-up and the cool-down protocols included the active range of joint movements and stretching exercises of the upper and lower extremities.
Outcome Measures
Participant characteristics and clinical data including age, sex, disease duration, the modified H&Y scale score, the more affected side, motor subtype, freezing of gait, part III of the Unified Parkinson’s Disease Rating Scale (UPDRS-III), levodopa equivalent daily dosages, and the MMSE score were recorded at baseline. All participants were evaluated while they were on dopaminergic medications and underwent assessments involving corticomotor inhibition and balance performance.
In TMS experimental set-up, surface electromyographic (EMG) recordings were obtained from the tibialis anterior (TA) muscle on the more affected side. The active electrode was placed over the muscle belly and the reference electrode was placed on the medial aspect of the tibia. The ground was placed on the patella. The EMG signals were amplified with filters set at 20 Hz to 3 kHz and recorded on a computer (Neuropack MEB-9100; Nihon Kohden Corp., Tokyo, Japan). All participants were seated in a chair with the knee in 90° flexion and the ankle in a neutral position. TMS was delivered to the cranium over the more affected M1 using a Magstim 200 stimulator (The Magstim Company Limited, Spring Gardens, Whitland, Carmarthenshire, UK) with a double cone coil. The hotspot was defined as the location of M1, where the maximal motor-evoked potential (MEP) was obtained from the TA. To determine the stimulation intensity of corticomotor outcomes, the resting motor threshold (RMT) was set as the minimum intensity producing MEP > 50 μV in the resting TA muscle in five out of ten trials. Participants with an RMT of > 83% of the maximum stimulator output were considered inexcitable, as stimulation at 120% of this value would be higher than 100% of the maximum stimulator output.
The measures of corticomotor inhibition included the CSP and SICI. The CSP was a period of suppressed EMG activity following the MEP induced by a single-pulse stimulation. It was assessed during isometric voluntary contraction and was used to evaluate corticospinal inhibition. The stimulation intensity of CSP was set to 120% of the RMT. To ensure a consistent force of the TA muscle, an upside-down U-shaped plastic board was set as a target, and participants were asked to touch the target by maintaining ankle dorsiflexion (around 30°) of the more affected side with the heel on the ground. 18 SICI was assessed at rest to evaluate intracortical inhibition. SICI was a paired-pulse stimulation elicited by a subthreshold conditioning stimulus (CS) followed by a suprathreshold test stimulus (TS) at an interstimulus interval of 3 ms. The stimulation intensities for the CS and TS were 80% and 120% of the RMT, respectively. Compared with the MEP induced by 100% of the RMT, an inhibited MEP would be observed after a paired-pulse stimulation of SICI. The amount of reduction represented SICI. Thus, the greater reduction of the MEP indicated more intracortical inhibition.
Balance was evaluated using the Mini-Balance Evaluation Systems Test (Mini-BEST) and the Timed Up and Go (TUG) test. The Mini-BEST is a useful and easy tool to evaluate functional balance. It consists of 14 items included under four subscales: anticipatory postural adjustments, postural responses, sensory orientation, and stability in gait. Each item is rated from 0 to 2 and the total score ranges from 0 to 28, with higher scores indicating better balance performance. The TUG test measures functional mobility and balance. In this test, subjects were instructed to stand up from a chair, walk 3 m, turn around, walk back to the chair, and sit down. They were instructed to use a comfortable walking speed to finish the task and the completion time was recorded.
Data Analysis
For the background EMG activity (bEMG), the root mean square of the bEMG signal was calculated for a time interval of 30 ms before TMS stimulation. CSP duration was defined as the interval between TMS stimulus onset and CSP offset and was quantified visually. 19 To determine SICI, the amplitude of the elicited MEPs was measured according to the peak-to-peak voltage (mV) of the EMG response. For both CSP and SICI, 10 trials were collected and averaged for data analysis. The mean amplitudes of SICI were expressed as percentages of inhibition using the following formula: (conditioned MEPs/test MEPs) × 100%. All values were presented as the mean ± standard deviation.
Statistical Analysis
The sample size was 28 calculated by using the G*Power 3.1.9 (power = 0.7; effect size = 0.85; α = 0.05). 20 All analyses were performed using IBM SPSS Statistics, version 25.0 (IBM Corp., Armonk, NY, USA). All data were checked for normal distribution using the Shapiro-Wilk test before the analysis. The independent t-test or the Mann-Whitney U test and the chi-squared test were used to analyze continuous and categorical variables, respectively, in between-group comparisons of baseline demographic data and pretraining outcomes. The paired t-test or the Wilcoxon signed-rank test were used to examine the effects of training on corticomotor inhibition (CSP and SICI) and balance performance (Mini-BEST and TUG) in each group. For between-group comparisons, the pre-post changes were compared using the analysis of covariance (ANCOVA) and the Quade test for normally (SICI and Mini-BEST) and non-normally (CSP and TUG) distributed data, respectively. The H&Y stage, UPDRS-III, and age were included as covariates. Effect sizes were evaluated using Cohen’s d. The significance level was set at P < .05.
Results
Participants and Baseline Characteristics
Twenty-eight participants were randomly assigned to the BT (n = 14) or the CON group (n = 14; Figure 1). One subject in the CON group dropped out during the intervention period. We used intention-to-treat analysis by substituting missing values with baseline data to manage the dropout. The baseline demographics and clinical characteristics are presented in Table 1. There were no significant differences in these characteristics between the groups except the H&Y stage. All participants were right-handed and right-footed.
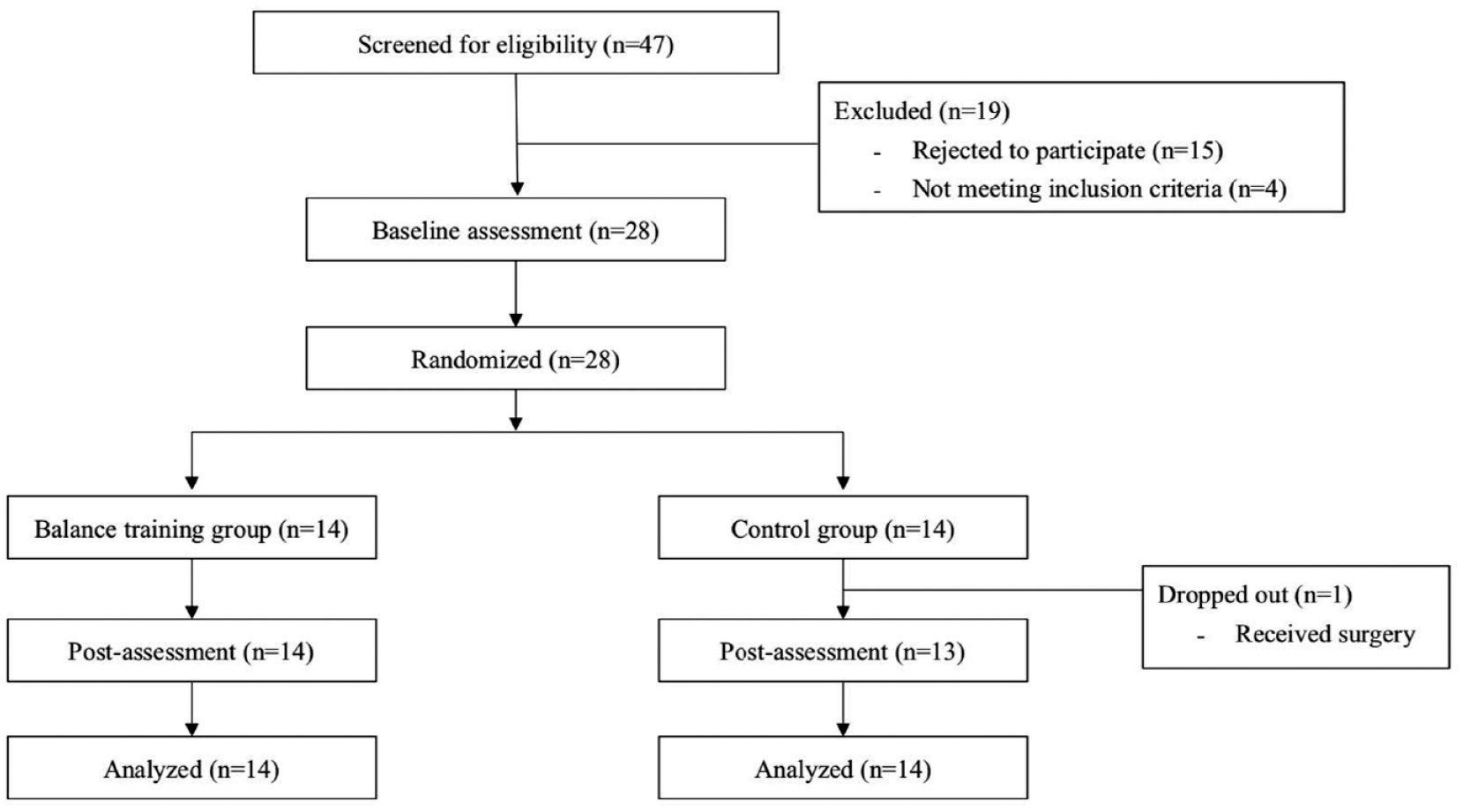
Flow Chart of the Experimental Design.
Baseline Characteristics and Clinical Features of Participants.
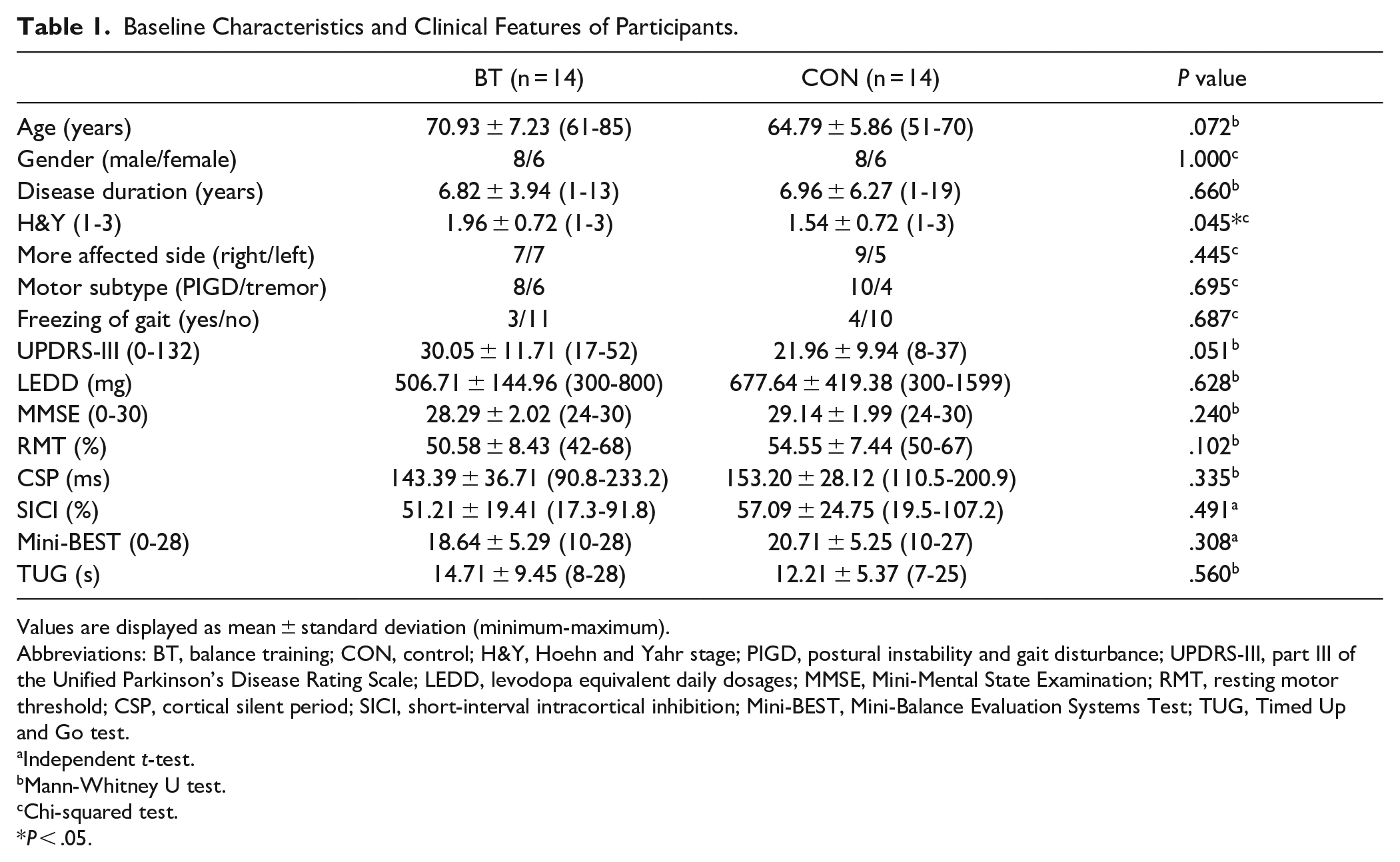
Values are displayed as mean ± standard deviation (minimum-maximum).
Abbreviations: BT, balance training; CON, control; H&Y, Hoehn and Yahr stage; PIGD, postural instability and gait disturbance; UPDRS-III, part III of the Unified Parkinson’s Disease Rating Scale; LEDD, levodopa equivalent daily dosages; MMSE, Mini-Mental State Examination; RMT, resting motor threshold; CSP, cortical silent period; SICI, short-interval intracortical inhibition; Mini-BEST, Mini-Balance Evaluation Systems Test; TUG, Timed Up and Go test.
Independent t-test.
Mann-Whitney U test.
Chi-squared test.
P < .05.
Corticomotor Inhibition
The baseline RMT was compared to ensure that the stimulation intensity between the groups was similar (Table 1). Five subjects were inexcitable in the CSP (two in the BT group, three in the CON group) and six subjects were inexcitable in the SICI (two in the BT group, four in the CON group). We managed and replaced the missing values using multiple imputations. 21
At baseline, between-group comparisons of CSP showed no significant differences. There was a significant increase in the CSP after balance training (P = .028), whereas no significant change was observed in the CON group (P = .196; Table 2). CSP showed a greater increase in the BT group than in the CON group (BT vs. CON: 19.89 ms vs −5.23 ms, P = .017), and a large effect size was observed (d = 0.828; Table 3).
Within-group Comparisons of Corticomotor Inhibition and Balance Performance Outcomes.
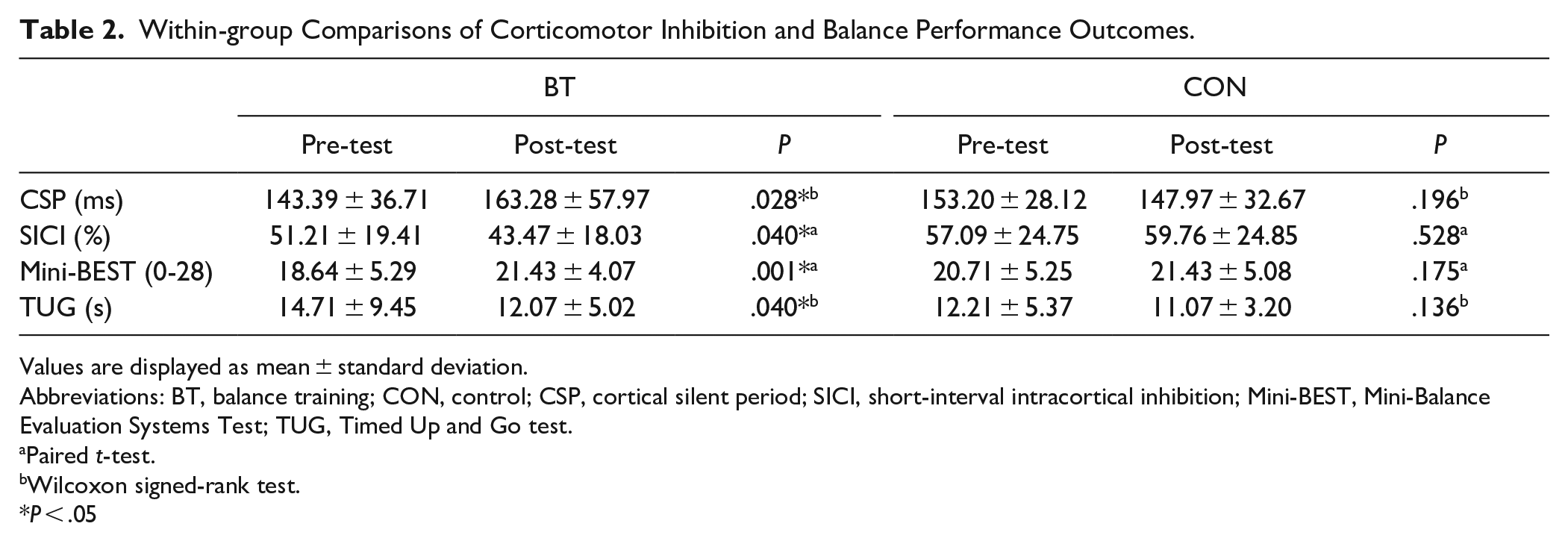
Values are displayed as mean ± standard deviation.
Abbreviations: BT, balance training; CON, control; CSP, cortical silent period; SICI, short-interval intracortical inhibition; Mini-BEST, Mini-Balance Evaluation Systems Test; TUG, Timed Up and Go test.
Paired t-test.
Wilcoxon signed-rank test.
P < .05
Between-group Comparisons of Corticomotor Inhibition and Balance Performance Outcomes.
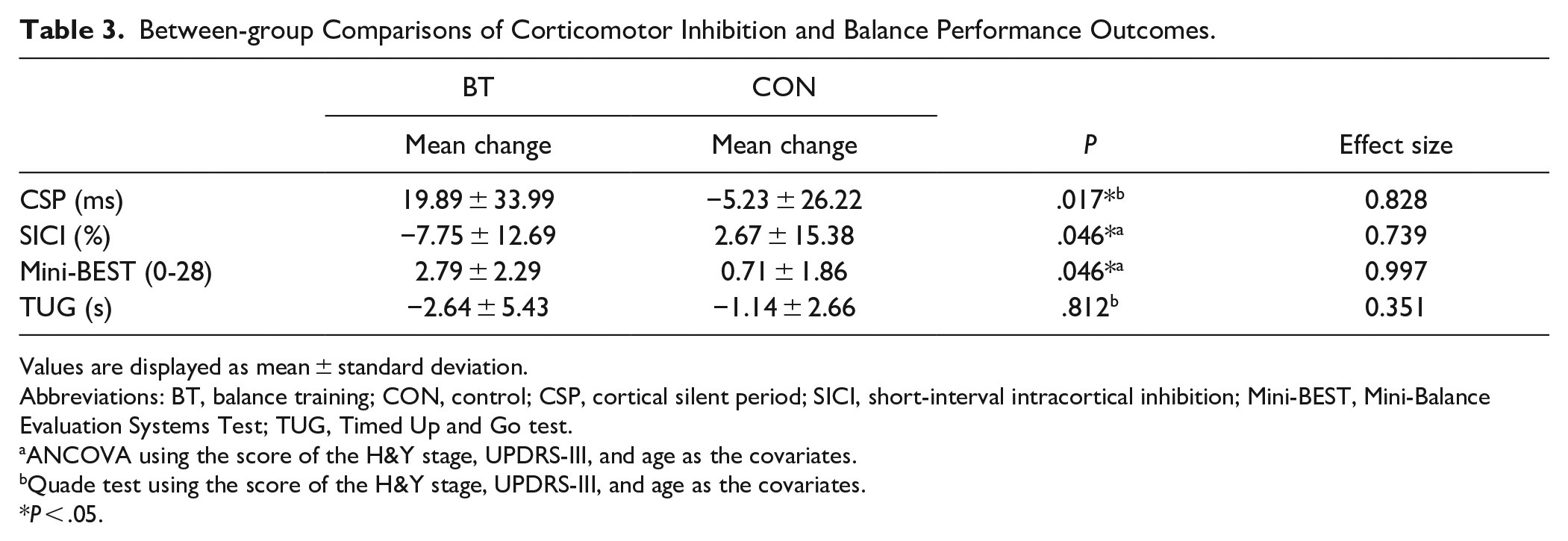
Values are displayed as mean ± standard deviation.
Abbreviations: BT, balance training; CON, control; CSP, cortical silent period; SICI, short-interval intracortical inhibition; Mini-BEST, Mini-Balance Evaluation Systems Test; TUG, Timed Up and Go test.
ANCOVA using the score of the H&Y stage, UPDRS-III, and age as the covariates.
Quade test using the score of the H&Y stage, UPDRS-III, and age as the covariates.
P < .05.
No significant difference was observed in the SICI between the groups at baseline. After 8-week training, percentage of the SICI decreased significantly in the BT group (P = .04), but no apparent change was observed in the CON group (P = .528) (Table 2). The BT group exhibited a greater decrease in the SICI (BT vs. CON: −7.75% vs. 2.67%, P = .046) than the CON group and the effect size was 0.739 (Table 3). Lower SICI values indicated higher intracortical inhibition.
Balance Performance
The baseline comparison between the groups revealed no significant difference in the Mini-BEST scores and the TUG test. At the end of the intervention, the mean Mini-BEST score in the BT group had significantly increased from 18.64 to 21.43 (P = .001), while it did not show a significant change (P = .175) in the CON group (Table 2). In the between-group comparison, the BT group showed a greater improvement in the Mini-BEST score than the CON group (BT vs. CON: 2.79 vs. 0.71, P = .046). The results demonstrated a large effect size (d = 0.997) (Table 3). There was a significant reduction in the TUG test scores after training in the BT group (P = .04) (Table 2), but no significant difference was observed in improvement between the groups (P = .812).
Discussion
To the best of our knowledge, this is the first randomized controlled trial to examine the effects of balance training on corticomotor excitability in individuals with PD. The results of the present study showed that balance training led to a significant increase in corticomotor inhibition in terms of both CSP and SICI. In addition, balance performance improved after 8 weeks of balance training. Thus, we observed that balance training could induce neuroplasticity of the M1 by modulating corticomotor inhibition and improving balance performance in people with PD.
In PD, striatal dopamine depletion causes alterations in the neural activity of the cortico-basal ganglia motor loop, leading to dysfunction of motor output. 22 Previous TMS studies showed abnormalities of excitability in the cortical motor areas of PD.2,8 A recent study indicated that cortical disinhibition with a reduction in the SICI was observed in PD and this alteration could be an early prodromal feature of PD. 23 Corticomotor inhibitory modulation is believed to play a crucial role in intact motor control, motor learning, and even in postural control. 15 Previous research has demonstrated that corticomotor excitability in the M1 significantly contributes to postural control in humans.11,12 Reduced corticomotor inhibition may affect balance performance and stability. 15 Furthermore, it was found that increased cortical inhibition was related to balance improvement in healthy adults. 24 Pelosin et al. also found that training-induced change in short-latency afferent inhibition was correlated with training-induced improvement in obstacle negotiation performance in participants at fall risk. 25 A recent study showed that reduced recruitment of the frontal gyrus correlated with improved balance performance after combining action observation and dual-task training in individuals with PD. 26 According to our results, cortical inhibition increased in terms of CSP and SICI after balance training. In the present study, the improvement of balance performance seems to be attributed to training-induced effects on corticomotor inhibition in PD. Studies are needed in the future to investigate the relationship between balance performance and corticomotor inhibition in PD.
Exercise has been proven to alleviate disease-related symptoms and facilitate neuroplasticity in PD. 27 It was also suggested that exercise has a neuroprotective effect in PD. 28 Noteworthily, different types of exercise may lead to various changes in corticomotor excitability and neuroplasticity. 28 A reduction of MEPs was observed after postural training, while an enhancement of MEPs was found after strength training. 29 These findings indicate the training-specific changes in cortical excitability. The mechanisms would be different between strength training and balance training. It could be assumed that strength tasks may rely on reducing inhibition to increase MEP recruitment and activate the muscles, whereas postural or balance tasks need to avoid unnecessary activations and movement by raising inhibition to focus on the proper muscles. Interestingly, aerobic training showed a similar result in CSP with balance training.30,31 The explication and the mechanism for the coincident effects on corticomotor inhibition remain unclear and need further evidence.
Based on previous TMS studies, a shortened CSP and a decreased SICI at rest compared to those in normal individuals are considered undoubted and congruous findings in PD.3,23 In addition, these abnormalities of cortical inhibition may correlate with the severity of motor symptoms.8,32 SICI is mainly mediated by GABA-A receptors representing the intracortical activity, while CSP is produced through activation of both spinal and cortical circuits mediated by GABA-B receptors. 3 The loss of corticomotor inhibition in PD (as shown by the CSP and SICI findings) may add to the difficulties in focusing neuronal activity onto appropriate pathways, enhancing unspecific motor program transmission and further motor disturbance. 32 We found that the participants in the BT group exhibited increased CSP and SICI in the more affected hemisphere. Therefore, we assume that balance training could enhance appropriate and specific neuronal activity in the motor areas by improving cortical inhibitory modulation in PD.
Balance dysfunction is a common and serious issue in PD. Pharmacological management is the mainstay of treatment in PD. However, balance dysfunction shows minimal improvement by treatment with levodopa. 33 Notably, the performance in the Mini-BEST and TUG tests showed obvious improvements with large effect sizes after balance training in the present results. SSE has been proven to have positive effects on improving balance performance and on reducing the risk of falls in older adults.17,34 Based on the positive response to this protocol in the present study, we believe that SSE is a safe and feasible intervention for improving balance performance and functional mobility in people with PD.
We did not find any significant improvements after regular physical therapy. There were some possible reasons for these findings. The lack of training effect on balance outcomes in the CON group might be due to the design of the training program. A review article reported that resistance training emphasizing the lower limb and trunk muscles could promote improvements in balance in PD. 35 However, the training program in the CON group mainly focused on the upper limbs with coordination and resistance training, and very few lower-limb muscles were challenged. Hence, the effects of this training program need to be confirmed in future studies. The lack of training effect on corticomotor inhibition might be due to the target muscle. It is believed that cortical excitability is probably not systemic, but specific to the involved muscles. 36 Recently, a TMS study indicated that the assessment of corticomotor excitability should carefully consider the task and the target muscle used for interventions to maximize the generalizability and sensitivity. 37 Moreover, TA was more remarkable and sensitive to postural responses. 14 Despite no significant changes in the CSP and SICI after training in the CON group, this finding needs to be interpreted cautiously and further studies are needed to elucidate it.
The present study has some limitations. The study had a small sample size and included 28 participants with PD in various stages of the disease. A significant difference was observed in the H&Y stages between the groups. There was a marginally significant difference in the UPDRS-III and age. However, the possible confounding factors had been included as covariates. After adjusting for confounders, results still showed greater improvements in corticomotor inhibition and balance performance in the BT group than in the CON group. Future research with more participants and a more even distribution of disability may be needed to validate our findings. In addition, a matched group of healthy subjects may provide more information to understand how individuals with PD showed altered cortical excitability and to interpret changes after training.
Conclusion
The present study demonstrated that balance training could induce inhibitory modulation with an increase in corticospinal and intracortical inhibition and improve balance performance in people with PD. In addition, the SSE training program seems to be a feasible and effective intervention for individuals with PD. Our findings provide a recommended therapeutic approach in clinical to reduce the impact of PD, in particular balance performance.
Footnotes
Acknowledgements
The authors express their sincere appreciation to the participating therapist and participants for their cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of the Republic of China (grant number MOST107-2314-B-010-041-MY3).
