Abstract
Background. Although recent evidence has shown a new role of fluoxetine in motor rehabilitation, results are mixed. We conducted a randomized clinical trial to evaluate whether combining repetitive transcranial magnetic stimulation (rTMS) with fluoxetine increases upper limb motor function in stroke. Methods. Twenty-seven hemiparetic patients within 2 years of ischemic stroke were randomized into 3 groups: Combined (active rTMS + fluoxetine), Fluoxetine (sham rTMS + fluoxetine), or Placebo (sham rTMS + placebo fluoxetine). Participants received 18 sessions of 1-Hz rTMS in the unaffected primary motor cortex and 90 days of fluoxetine (20 mg/d). Motor function was assessed using Jebsen-Taylor Hand Function (JTHF) and Fugl-Meyer Assessment (FMA) scales. Corticospinal excitability was assessed with TMS. Results. After adjusting for time since stroke, there was significantly greater improvement in JTHF in the combined rTMS + fluoxetine group (mean improvement: −214.33 seconds) than in the placebo (−177.98 seconds, P = 0.005) and fluoxetine (−50.16 seconds, P < 0.001) groups. The fluoxetine group had less improvement than placebo on both scales (respectively, JTHF: −50.16 vs −117.98 seconds, P = 0.038; and FMA: 6.72 vs 15.55 points, P = 0.039), suggesting that fluoxetine possibly had detrimental effects. The unaffected hemisphere showed decreased intracortical inhibition in the combined and fluoxetine groups, and increased intracortical facilitation in the fluoxetine group. This facilitation was negatively correlated with motor function improvement (FMA, r2 = −0.398, P = 0.0395). Conclusion. Combined fluoxetine and rTMS treatment leads to better motor function in stroke than fluoxetine alone and placebo. Moreover, fluoxetine leads to smaller improvements than placebo, and fluoxetine’s effects on intracortical facilitation suggest a potential diffuse mechanism that may hinder beneficial plasticity on motor recovery.
Keywords
Introduction
Stroke is the main cause of long-term disability worldwide.1,2 Although reperfusion can improve neurological outcomes acutely, 55% to 75% of stroke survivors have functional limitations 3 to 6 months later.3,4 Motor disability after stroke has substantial impact on patient quality of life as well as overall stroke rehabilitation costs.1,5 Innovative rehabilitation approaches such as pharmacological agents, including selective serotonin reuptake inhibitors (SSRIs), and noninvasive brain stimulation (NIBS) techniques are thus being investigated to promote functional recovery in stroke.3,6
The serotonergic system has neuroprotective effects, and SSRIs may play a role in enhancing overall brain excitability,7-9 blood flow regulation/angiogenesis, 10 neuronal sprouting/re-hardwiring and potentially balancing excitation/inhibition.10,11 While previous studies showed that SSRIs, especially fluoxetine, could improve motor function and reduce stroke-related disability, the evidence remains insufficient to modify stroke treatment guidelines or reduce concerns regarding serious adverse effects.11,12 Early prescriptions of fluoxetine (20 mg/d 5-10 days after stroke onset) combined with physiotherapy enhanced motor function recovery (FLAME trial) and decreased stroke-related disability 13 ; a Cochrane review on SSRI effects on poststroke rehabilitation also demonstrated improved dependency and disability. 14 However, this review and a previous meta-analysis 15 concluded that motor deficits did not significantly benefit from SSRIs (including fluoxetine) when comparing motor scales, potentially due to SSRI nonfocality. Moreover, patients with mild depression were included in the FLAME trial and the results might have been mediated by fluoxetine’s antidepressant effects. 14 While the FLAME trial showed better motor results for the fluoxetine versus placebo groups even after adjusting for new-onset depression (diagnosed in the first 90 days of the trial), yet the adjusted mean 90-day change on Montgomery-Åsberg Depression Rating Scale (MADRS) was also higher in the placebo group (by 3.3 points, P = 0.032). Therefore, it is still unclear if antidepressants’ positive effects on stroke motor recovery are mediated by enhanced motor pathway neuroplasticity or via other pathways such as mood. 10
NIBS techniques, such as repetitive transcranial magnetic stimulation (rTMS), enhance poststroke motor performance by influencing cortical excitability in a relatively focal area.16-18 Low-frequency rTMS over the unaffected hemisphere may enhance motor performance, potentially by modulating interhemispheric competition.19-23 Additionally, recent major depression trials have shown that combining a focal NIBS technique with SSRIs or tricyclic antidepressants may increase the efficacy of each treatment, especially in mood modulation.24-26
Given that fluoxetine has shown potential to induce neuroplastic effects in stroke patients and the evidence that rTMS may enhance its neuroplastic effects on motor function recovery, 27 combining these nonfocal and focal mechanisms may improve motor function after stroke. Therefore, this study aimed to investigate whether the combination of rTMS with fluoxetine would benefit poststroke motor function beyond pharmacotherapy alone and placebo after 3 months of treatment. The motor function measures are Jebsen-Taylor Hand Function (JTHF) test and Fugl-Meyer Assessment (FMA) upper extremity motor score.
Aim 1: to test the effects of the combined therapy (fluoxetine plus low-frequency rTMS over the unaffected primary motor cortex [M1]) on motor function compared with fluoxetine and placebo. Aim 2: to evaluate intervention mechanisms by assessing neurophysiologic M1 changes using motor evoked potentials (MEPs) and cortical excitability/inhibition measurements. Our hypothesis is that combined rTMS and fluoxetine would induce larger motor gains than each of fluoxetine alone and placebo after 3 months, and that both the combined treatment and fluoxetine alone groups would be superior to placebo (a hierarchical model). Finally, we assessed if these effects were mood dependent, and measured preliminary safety effects.
Materials and Methods
Trial Design
This exploratory double-blinded randomized clinical trial (NCT02208466) was approved by Partners Institutional Review Board, Spaulding Hospital. All participants consented before trial procedures.
Participants
Inclusion criteria: adults with hemiparesis/hemiplegia due to ischemic stroke within the previous 2 years, upper extremity Fugl-Meyer (FMA) motor score >11 and <56, and prestroke disability score <3 on Modified Rankin Scale (mRS). Exclusion criteria: unable to understand instructions, TMS contraindications,16,28 concurrent medical condition likely to worsen functional status within 6 months, score of 24 or higher on Hamilton Depression Rating Scale (HDRS), joint or paretic extremity pain, pregnancy, recent intake of SSRI or any medication likely to interact with SSRIs (5-week washout period was required before enrollment in either case). All trials procedures were performed at the Spaulding Neuromodulation Center, Boston, Massachusetts. This study was structured as per Figure 1.
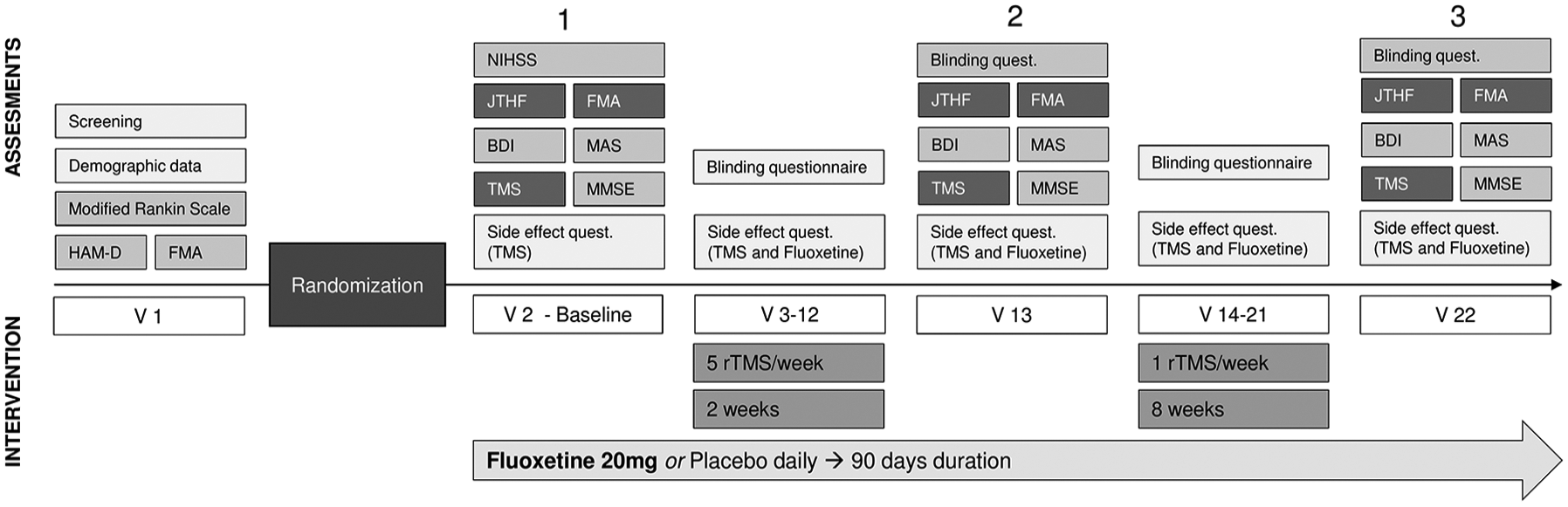
Study visits outline: V1: screening; V2: baseline assessments (day 0), study medication given; V3 (2 weeks after V2): daily rTMS sessions begin; V13: day 30 assessments; V14-21: 8 weekly rTMS sessions; V22: day 90 assessments. HDRS, Hamilton Depression Rating Scale; FMA, Fugl-Meyer Assessment; JTHF, Jebsen-Taylor Hand Function; BDI, Beck’s Depression Inventory; MAS, Modified Ashworth Scale; TMS, Transcranial Magnetic Stimulation; rTMS = repetitive TMS; MMSE, Mini-Mental State Examination.
Interventions
Repetitive Transcranial Magnetic Stimulation
Participants received 18 sessions (10 daily session—Monday to Friday; followed by 8 weekly sessions) of sham or active rTMS over the “hot spot” M1 area of the unaffected hemisphere leading to a response in the contralateral first dorsal interosseous (FDI) muscle. 16 “Hot spot” localization was performed by mapping the hand area: first marking 5 cm below the vertex, then placing the center of the coil at that point, and orienting the coil toward the contralateral supraorbital region at a 45° angle (to guarantee current flow approximately perpendicular to the central sulcus). Then we provided the lowest magnetic pulse stimulus intensity that could induce a motor-evoked potential (MEP) of at least 100 µV peak to peak in 5/10 successive attempts at rest (ie, resting motor threshold [rMT]). 16 Surface electromyography (EMG) was used to measure the muscle contraction response generated by the TMS pulse in a sensitive manner.
Low-frequency rTMS was applied at 100% rMT intensity, 1 Hz, 1200 pulses as a single, continuous train lasting 20 minutes. rMT and “hot spot” were confirmed on each stimulation day, and each day’s stimulation intensity was used for the low-frequency rTMS protocol. We used the same swim cap for each subject to mark the “hot spot” location on each stimulation session. We used MagPro stimulators (MagPro G3 Main, X100+Option Model; serial number: 1099).
For sham-rTMS, parameters were identical except the coil was flipped over to the sham side and an electrode (Ambu Neuroline Surface Electrodes) was placed over the forehead to deliver a small current mimicking stimulation sensations 29 ; this electrode was also used in active rTMS but without a current (Smith JE1, Peterchev AV.MagVenture 2017 Cool-coil system: user manual US 501-1282). The researcher administering rTMS was blinded to drug allocation.
Fluoxetine
Subjects received fluoxetine 20 mg or placebo (matched in color, taste, and appearance) daily for 90 days in an identical protocol. On randomization, a study staff member contacted the compounding pharmacy, which sent the subject’s randomized study drug to the hospital pharmacy. Only the pharmacist and the one uninvolved study staff member above knew the randomization codes. Medication compliance was monitored weekly using a questionnaire.
Participants were scheduled roughly at the same time during the morning or afternoon; however, this was subject to participant availability, as we believed that a few hours’ difference would improve adherence without affecting our results. No physical or occupational therapy was provided in this trial, but patients were allowed to continue their therapy regimens (Table 1), which remained unchanged for most patients (ie, they had been receiving the same therapy at baseline).
Demographic and Baseline Characteristics.
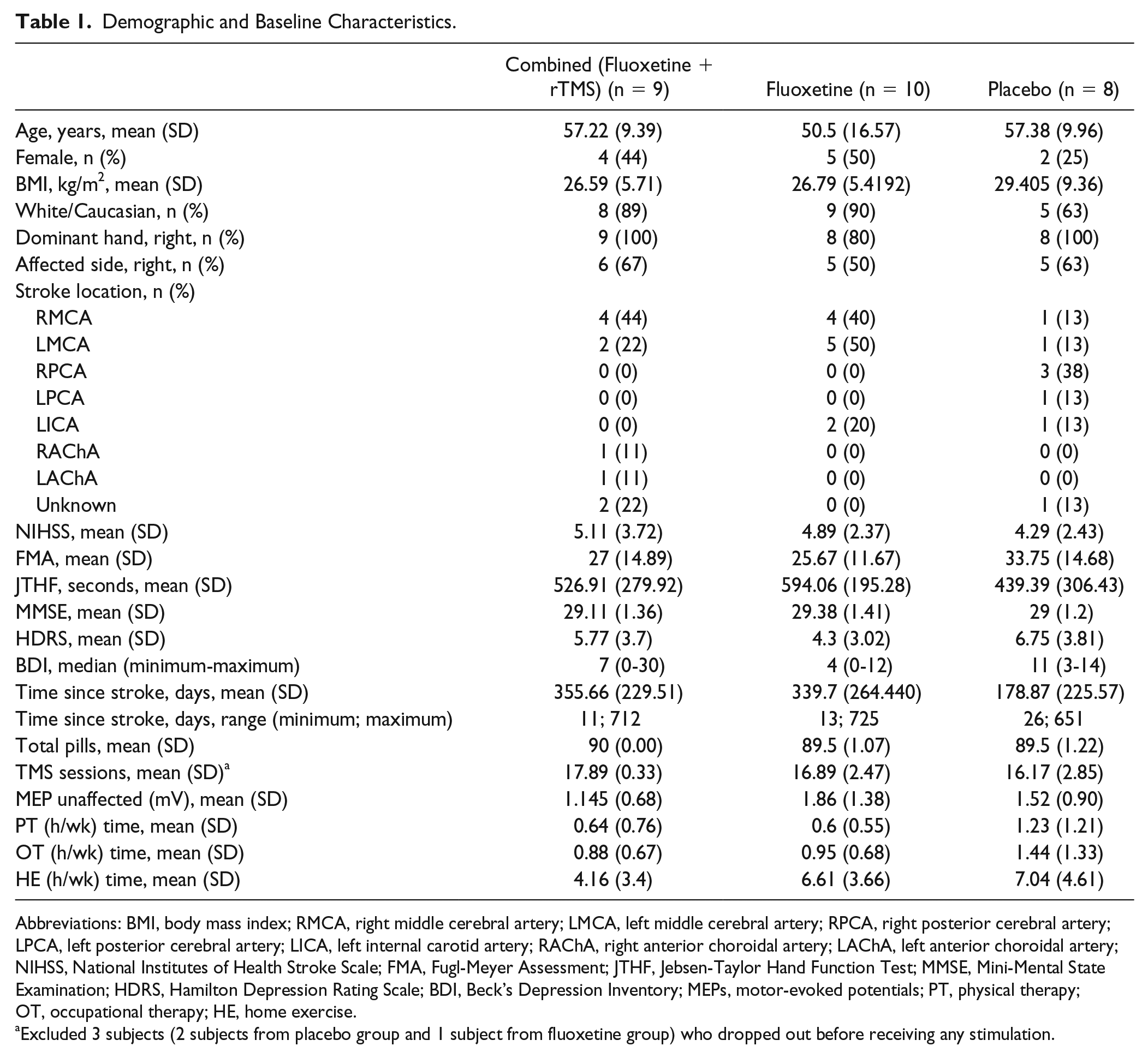
Abbreviations: BMI, body mass index; RMCA, right middle cerebral artery; LMCA, left middle cerebral artery; RPCA, right posterior cerebral artery; LPCA, left posterior cerebral artery; LICA, left internal carotid artery; RAChA, right anterior choroidal artery; LAChA, left anterior choroidal artery; NIHSS, National Institutes of Health Stroke Scale; FMA, Fugl-Meyer Assessment; JTHF, Jebsen-Taylor Hand Function Test; MMSE, Mini-Mental State Examination; HDRS, Hamilton Depression Rating Scale; BDI, Beck’s Depression Inventory; MEPs, motor-evoked potentials; PT, physical therapy; OT, occupational therapy; HE, home exercise.
Excluded 3 subjects (2 subjects from placebo group and 1 subject from fluoxetine group) who dropped out before receiving any stimulation.
Outcome Measures
All outcomes were assessed on day 0 (baseline visit), day 30 (after 10 daily stimulations and 1 month of drug treatment), and day 90 (at treatment end, ie, 90 days of drugs and 18 rTMS sessions). A rater blinded to rTMS and pharmacotherapy performed all assessments. The primary outcomes were motor function scales: JTHF and FMA upper extremity changes from day 0 to day 90. JTHF test was designed as a broad measure of hand function in activities of daily living. It provides information on the time required to turn cards, pick up small objects, simulate feeding by picking up beans with a spoon, stack checkers, and lift cans that are empty and weighing 500 g. JTHF is sensitive to motor changes induced by motor cortex stimulation. The maximum time to execute each task is 120 seconds, 30 which is also the score if unable to complete a task. Handwriting was excluded, so the maximum score was 720 seconds.
Secondary Outcomes
Modified Ashworth Scale (MAS), Beck’s Depression Inventory (BDI), Mini-Mental State Examination (MMSE), medication diary, antidepressant side effect, and rTMS side effects questionnaires. BDI was included to assess for possible mood effects of fluoxetine, and MMSE to rule out cognitive changes resulting from the interventions (safety outcome). A questionnaire assessed blinding at days 0, 30 and 90. All analyses were performed on score differences from day 0 to day 90.
Neurophysiological Assessments
Neurophysiological outcomes were also assessed on day 0 (baseline visit), day 30 (after 10 daily stimulations and 1 month of drug treatment), and day 90 (at treatment end—90 days of drug and 18 rTMS sessions). Cortical excitability changes were investigated using TMS Bistim2 stimulator (Magstim Company Ltd, Whitland, UK) and a 70-mm figure-of-eight coil, with EMG recordings over the contralesional FDI. To record MEPs, a silver/silver chloride electrode was placed over the FDI muscle belly (active electrode); the reference was placed over the index finger proximal interphalangeal joint, and the ground electrode was placed over the wrist. MEPs were amplified and filtered using Powerlab 4/30 with a band pass of 20 to 2000 kHz. Signals were fed to a computer for off-line analyses using data collection software and conditional averaging software.
We investigated cortical excitability changes in our previous study using MEPs and rMT. 28 For the MEP study, we initially adjusted TMS intensity (usually 120% of the rMT) to achieve a baseline MEP of about 1 mV peak-to peak amplitude. Stimulation intensity was constant for each subject throughout the experiment. We recorded 10 MEPs and averaged their peak-to-peak amplitudes. We used paired-pulse to measure intracortical excitability: a conditioning stimulus (subthreshold: 80% of rMT) was first applied, followed by a test stimulus (suprathreshold: 120% of rMT or the intensity used for MEP) at variable interstimulus intervals (ISIs: 2, 3, 6, 10, and 12 ms). The raw average values of intracortical inhibition (ICI: 2 and 3 ms ISI) and intracortical facilitation (ICF: 10 and 12 ms ISI) were divided by the average MEP amplitude (unconditioned stimuli of 1 mV peak to peak—usually 120% of the rMT); therefore, ICI and ICF percentages are normalized by MEP average. Cortical excitability markers (rMT, MEPs, ICI, and ICF) were acquired through M1. Outlier pulse amplitudes were excluded based on the interquartile range method. 31 All analyses were performed considering differences in measurements from day 0 to day 90. When baseline rMT could not be determined—due to corticospinal tract damage (common in the ipsilateral hemisphere) or due to technical issues (ie, noise during EMG)—the cortical excitability assessments were not performed in that hemisphere for the remaining visits.
Randomization
A study staff member who was not involved in the protocol generated a web-based randomization sequence with blocks of 3 and 5 subjects; randomization was kept in sealed sequentially numbered opaque envelopes. Subjects were randomized (allocation ratio 1:1:1) to: Combined group—fluoxetine + active rTMS, Fluoxetine—fluoxetine + sham rTMS, and Placebo—sham rTMS + placebo fluoxetine. The rTMS operator was not blinded to rTMS allocation; however she or he was blinded to fluoxetine allocation and did not collect outcome data.
Statistical Analysis
The primary outcome was JTHF and FMA changes from day 0 to day 90. All outcomes were calculated using the differences between day 90 and day 0; additionally, for motor outcomes (JTHF and FMA) a comparison between day 30 and day 0 was also performed. Histogram and Shapiro-Wilk test assessed data distribution. Baseline and stroke characteristics were reported by use of descriptive statistics (eg, mean and SD for continuous variables, frequency tabulations for categorical variables) for each group and compared using 1-way analysis of variance (ANOVA) or Fisher’s exact test (for continuous and categorical variables, respectively). Differences between groups were investigated using repeated-measures ANOVA (for secondary nonmotor outcomes) or mixed linear regression models (for primary outcomes JTHF and FMA) when data were normal, and Kruskal-Wallis test when not normal. We used Spearman or Pearson correlation analyses to verify correlation between neurophysiological and clinical outcomes.
For the primary motor outcomes (JTHF and FMA) we performed a prespecified hierarchical test: we first used our mixed linear model to test the main hypothesis—that the combined group would have a better outcome on JTHF and FMA than the fluoxetine group; after that, we tested our second hypothesis that the combined and fluoxetine groups would each perform better than placebo. We used hierarchical model testing as it addresses the problem of multiple comparisons in a more efficient manner (due to partial pooling of point estimates) than traditional methods of correction. 32
For JTHF and FMA, this regression model was performed to account for covariate effects, and adjusted means were compared. Results for these variables were adjusted for time since stroke (categorized <180 or >180 days) and the interaction term (time * treatment). We also tested for effects of other covariates—age, dominant hand and stroke side. However, only time since stroke (categorized <180 or >180 days) and interaction term (time * treatment) significantly altered the regression model and were therefore included in the final model.
As the vascular territory data was taken from available medical records information, the MRIs were not available for quantitative analysis, and specific vascular territory information was missing for 4 patients. We were neither able to test for the volume of corticospinal tract infarcted nor clearly account for different levels and locations of infarcts in anterior and posterior circulation-supplied vascular territories on each side (eg, right/left M1, internal capsule, cerebral peduncles). We therefore did not include vascular territory or infarct volume in our model due to the potential inaccuracy, missing data (which cannot be imputed) and as this variable would have 8 different levels (4 locations, plus right or left side), which would have made the analysis biased and underpowered.
All analyses were based on 2-tailed tests. We used a significance level of 0.05 and an intention-to-treat (ITT) analysis for motor scales and neurophysiological data. For missing JTHF and FMA data we estimated the delta differences (day 90 – day 0) and applied the regression imputation method (using age, gender, time since stroke and FMA at enrollment as predictors). We also estimated the delta differences (day 90 – day 0) of missing cortical excitability data (ICI, ICF, and MEP) using multiple imputation regression (with group and time since stroke as predictors) for ITT analysis. For the remaining clinical outcomes only a complete case analysis was performed reporting the uncorrected P values. We also conducted an exploratory regression analysis to understand if time since stroke altered the results of these secondary outcomes, however for neurophysiological analysis the interaction term—time * treatment—was excluded because of lack of statistical power and not altering the final model in the ICI and ICF analysis.
Results
Between June 3, 2015, and March 5, 2018, 27 patients were randomized into 1 of the 3 groups (Figure 2). There were no significant demographic or baseline differences between groups (Table 1). However, average age was slightly lower in the fluoxetine group, and time since stroke was lower in the placebo group (Table 1, both nonsignificant). Baseline FMA and JTHF scores were nonsignificantly higher in the placebo group (Table 1). All covariates with heterogeneous baseline values (nonsignificant) were tested in the initial regression model to assess for potential confounding. We only adjusted the final results for time since stroke as no other variable changed the model. For the adjusted means comparison, we used hierarchical model testing to efficiently address the problem of multiple comparisons, and as our aims were hierarchical (priority being for combined vs placebo group testing, then combined or fluoxetine vs placebo group testing). Treatment compliance for fluoxetine and rTMS did not significantly differ between groups.
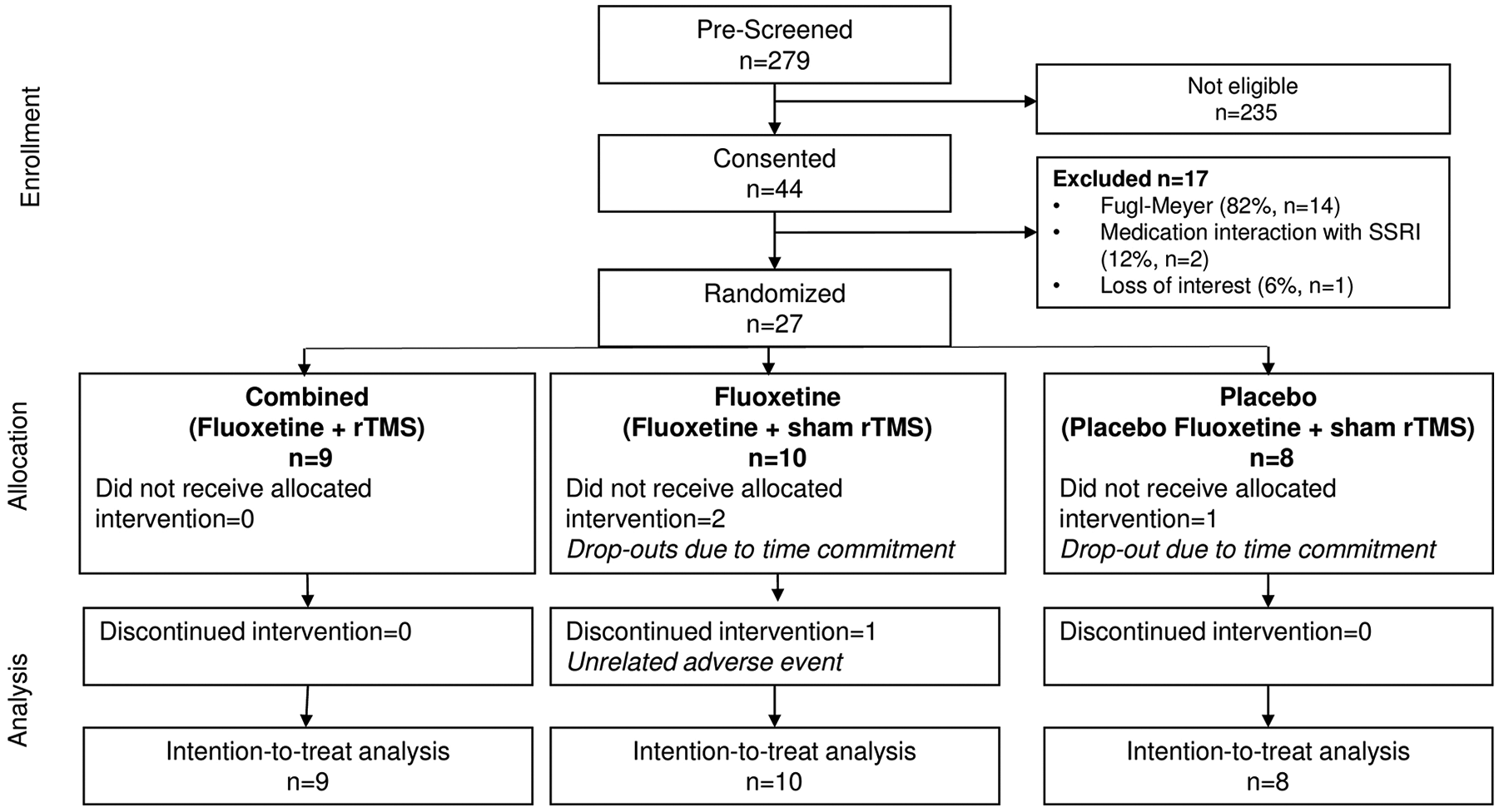
Study flowchart.
All results below are from the same mixed linear model; we first emphasize the combined group and then the fluoxetine group.
Combined Group Motor Function
A linear repeated-measures hierarchical model was performed; the dependent variable was motor function (change in FMA and JTHF scores) and the covariates were group (combined: fluoxetine + rTMS, fluoxetine and placebo), time since stroke (categorized <180 or >180 days) and interaction term (time * treatment). After adjusting for time since stroke, mean JTHF changes (differences between day 90 and day 0) were significantly higher in the combined group (−214.33 seconds) than in the fluoxetine (−50.16 seconds, P < 0.001) and placebo groups (−117.98 seconds, P = 0.005); however, mean FMA change for the combined group did not differ from the other groups. Finally, at 30 days, there were no differences in FMA and JTHF between the 3 groups after adjusting for time since stroke.
Fluoxetine Group Motor Function
After adjusting for time since stroke, the mean JTHF and FMA improvements at the final endpoint (day 90) were both significantly lower in the fluoxetine versus placebo groups (respectively, −50.16 vs −117.98 seconds, P = 0.038; 6.73 vs 15.55 points, P = 0.039). There were no differences between groups at 30 days (Table 2).
Motor Assessments—Day 0 to Day 90 Changes and Day 0 to Day 30 Changes. a

Abbreviations: rTMS, repetitive transcranial magnetic stimulation; FMA, Fugl-Meyer Assessment; JTHF, Jebsen-Taylor Hand Function; C vs F, Combined versus Fluoxetine groups; C vs P, Combined versus Placebo groups; F vs P, Fluoxetine versus Placebo groups.
Means were adjusted for time since stroke (categorized to <180 days or >180 days) with a linear regression model for both FMA and JTHF scores. Boldfaced values indicate significance with P < 0.05.
Corticospinal Excitability Changes
The affected hemisphere MEP analysis was not performed as rMT was observed in only 5 participants. In the unaffected hemisphere, the unadjusted median MEP amplitudes were compared using Kruskal-Wallis test. There was a significant decrease in the MEP amplitude in the combined versus placebo groups (−0.05 vs 0.33, P = 0.037) (Table 3). An exploratory linear repeated-measures hierarchical model was also performed. The dependent variable was MEP amplitude changes (day 90 – day 0) and the covariates were group (combined, fluoxetine, and placebo) and time since stroke (categorized <180 or > 180 days); the interaction term was not used. MEP amplitude changes were significantly smaller in the combined group than in the placebo group (−0.057 vs 0.490 mV, P = 0.018) (Supplementary Table 1). There were no significant correlations between clinical improvements and MEP amplitude changes in both scales.
Cortical Excitability Assessments.
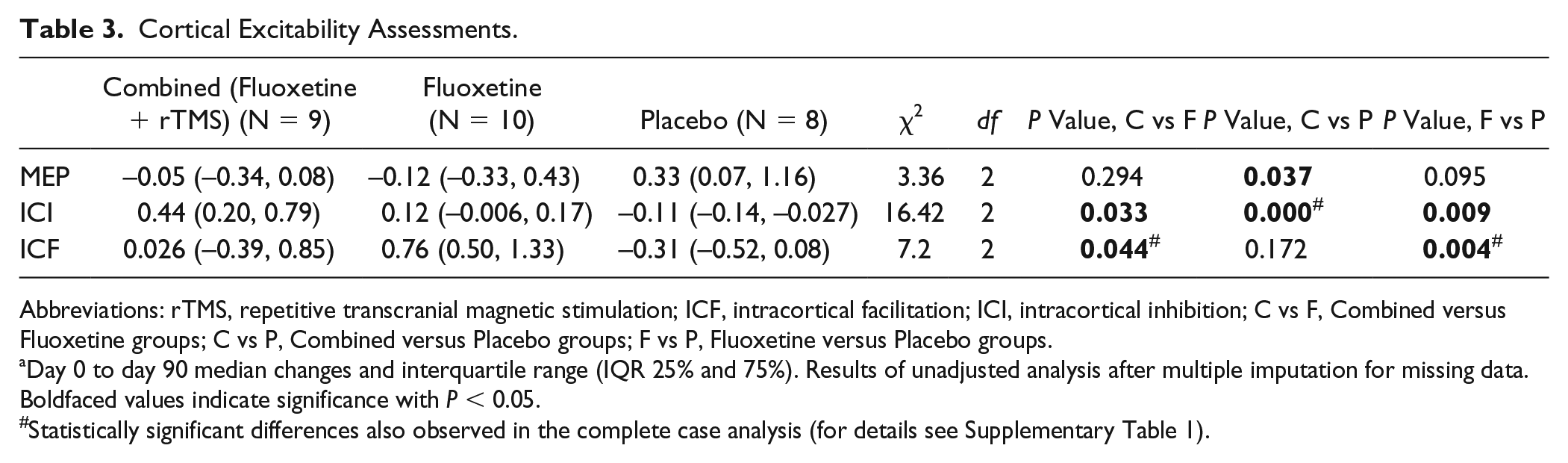
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; ICF, intracortical facilitation; ICI, intracortical inhibition; C vs F, Combined versus Fluoxetine groups; C vs P, Combined versus Placebo groups; F vs P, Fluoxetine versus Placebo groups.
Day 0 to day 90 median changes and interquartile range (IQR 25% and 75%). Results of unadjusted analysis after multiple imputation for missing data. Boldfaced values indicate significance with P < 0.05.
Statistically significant differences also observed in the complete case analysis (for details see Supplementary Table 1).
We used a mean ISI of 2 and 3 ms for ICI determination, and a mean ISI of 10 and 12 ms for ICF determination. The unadjusted analysis using Kruskal-Wallis test showed significantly different median ICI and ICF score changes (differences between day 90 and day 0) between groups (Table 3), with a significantly increased median ICF score in the fluoxetine (0.76) versus combined (0.026, P = 0.044) and placebo groups (−0.31, P = 0.004), as well as a significantly increased median ICI score (less inhibitory response) in the combined (0.44) vs placebo (−0.11, P < 0.001) and fluoxetine groups (0.12, P = 0.033). An exploratory repeated-measures hierarchical linear model was performed. The dependent variable was ICI or ICF changes (day 90 – day 0) and the covariates were group and time since stroke; the interaction term was not used in this analysis. ICI scores were significantly higher in the combined group than in the placebo group (0.848 vs −0.128, P = 0.036); that is, the MEP amplitude increased during the ICI protocol in the combined group after the treatment. There were no significant differences in the ICF score after adjusting for time since stroke. There was a significant negative correlation between motor impairment changes (FMA r2= −0.398, P = 0.0395) and ICF amplitude changes (Supplementary Table 1). Please note that the missing data for neurophysiological parameters (median changes for MEP, ICI, and ICF) were 29%. Despite the high percentage of missing data we kept ITT analysis with regression imputation but added a complete case analysis; we found that the results were similar for the ITT and complete case analyses (statistical differences for complete case analysis are shown as “#” in Table 3, and detailed results are shown in Supplementary Table 1).
Motor Improvement Independent of Mood
BDI scores did not differ significantly between groups at inclusion, at 30 or at 90 days. Median BDI changes at day 90 were not different between groups, χ2(2) = 0.0533, P = 0.974, suggesting that treatment effects were not associated with mood changes (Supplementary Table 2). The regression analysis showed that time since stroke did not significantly alter BDI scores.
Secondary Assessments and Safety Evaluation
Kruskal-Wallis test showed no differences in MAS median changes between groups, χ2(2) = 0.971, P = 0.6154), and repeated-measures ANOVA for MMSE showed no significant differences in the main effect of time, treatment group, or interaction term time * group (F < 1 for the 3 analyses) (Supplementary Table 2). No severe adverse events occurred, and there were no significant differences in all adverse events between groups (Supplementary Tables 3 and 4).
Discussion
While all groups improved in this study, the hemiparetic stroke patients receiving combined rTMS and fluoxetine had superior JTHF improvements after adjusting for time since stroke (subacute <180 days, chronic >180 days) 33 compared with those receiving fluoxetine or placebo. We found that fluoxetine might decrease (worsen) motor function as seen by their lower improvements (JTHF and FMA) compared with placebo. These motor outcomes were assessed using a hierarchical model accounting for multiple comparisons. Additionally, the fluoxetine group showed an increase in the unaffected hemisphere’s intracortical facilitation (which correlated with the reduced motor benefits), while the combined group had decreased intracortical inhibition compared with placebo (no correlation). Moreover, there were no significant changes in MEP amplitudes or secondary outcomes (eg, BDI and MMSE) between groups. Finally, no major adverse events were reported during or after either treatment, which is consistent with previous rTMS stroke studies 28 and with fluoxetine being a well-tolerated drug.
Combination of rTMS and Fluoxetine
In our study, combining rTMS with fluoxetine enhanced motor function; however, these effects did not seem to result from rTMS enhancing fluoxetine’s anticipated motor benefits. Fluoxetine may not have aided motor function in our sample, in line with previous trials in more chronic stroke populations where SSRIs did not improve motor function or reduce disability.20,34 Additionally, fluoxetine-induced motor gains in other studies may have been confounded by improved mood, which was not the case in our study.
Furthermore, in depression studies combining NIBS with antidepressants, 24 an additive effect was only seen when the drugs alone significantly improved depression. In our study, rTMS may have overcome fluoxetine’s unfavorable effects in the combined group; this would explain the modest motor benefits, especially when compared with recent clinical trials and a meta-analysis on low-frequency rTMS in stroke.11,17-20,34-37 Those studies often also had positive rTMS findings on both FMA and JTHF.
Motor Function Outcome Differences (JTHF vs FMA)
Motor function improved significantly in the combined group on JTHF but not FMA. Unlike FMA, JTHF is a continuous scale that measures time to perform tasks and is more sensitive to minor changes. If patients attempt to utilize different body parts to compensate for deficits, that strategy might increase or decrease performance time (eg, increasing movement amplitude can increase performance time); meanwhile, if their motor function improves then they are less likely to try to compensate or improve their compensatory techniques. Therefore, the reduction in time could be due to increased compensatory motion (though this would not explain the difference between groups) or due to motor function improvement, and the JTHF score might be more informative overall.38-41 The FMA rates movement performance as: not correct, mostly correct or equal to the unaffected limb (0, 1, and 2 points); therefore, it may have low sensitivity for specific motor gains that nevertheless improve patient quality of life. Additionally, FMA measurements of finger movements might underestimate distal motor disabilities and any motor function improvement 42 that may be enhanced by rTMS.
Fluoxetine Motor Function Effects
In our study, the fluoxetine group had less motor improvements than the placebo group. While we did not anticipate this, few randomized trials have tested the effects of fluoxetine on motor function, and mainly in acute stroke patients; therefore, there could be several explanations for our results.
One possible explanation is the difference in time since stroke compared with the FLAME trial, 13 which gave patients fluoxetine or placebo 5 to 10 days after stroke, while we included subjects up to 2 years poststroke. However, adjusting for time since stroke only confirmed our results, and other acute stroke studies also showed no significant SSRI effects on motor function and disability.43-46 Furthermore, the recently published FOCUS trial also recruited acute stroke (n = 1543, 2-15 days after stroke), but showed that 6 months of fluoxetine administration after stroke did not improve functional outcomes (mRS). 47 That said, if fluoxetine benefits are confirmed in acute stroke, they may be mediated by fluoxetine’s anti-inflammatory, angiogenesis-inducing, and antioxidant properties that are more relevant for acute than chronic stroke.10,48-51 In that case, combining fluoxetine with rTMS (as in this trial) would not be helpful since these fluoxetine’s anti-inflammatory and angiogenesis-inducing effects would be less relevant in chronic stroke.
Another explanation may be that fluoxetine effects are mediated by mood changes, which we did not observe in our study. Mood and attention can have major negative repercussions on learning and memory, 2 fundamental processes influencing motor recovery. 52 In fact, the FLAME trial showed significant differences in adjusted mean changes (MADRS) and frequency of depression between groups, although the results still favored fluoxetine when controlling for new-onset clinical depression. However, they did not perform an ITT analysis, and they did not report how many patients in each group received open-label fluoxetine for depression during the trial; that is, some of the patients in the fluoxetine and placebo groups may have received 40 and 20 mg of fluoxetine respectively; the correlation between mood changes and motor improvement was unclear as well. 12 In the FOCUS trial, fluoxetine also reduced the occurrence of depression in the first 6 months after stroke; however, no correlations with functional or motor outcomes were presented 47 and the trial results did not support the use of fluoxetine to prevent post-stroke depression or to improve functional outcomes.
Thus far, there is only one randomized clinical trial showing positive effects of fluoxetine on stroke motor function, 13 and the available meta-analysis evaluated dependency and disability scales (such as mRS) rather than sensorimotor function scales relating to impairment such as FMA. Furthermore, most trials using SSRIs (including fluoxetine) evaluated different outcomes, some relating to motor function (ie, FMA, finger tapping), some to functional independency after stroke (mRS) or even stroke severity (NIHSS). Evidence used to justify trials such as FLAME, FOCUS, and our trial considered overall effects of fluoxetine/SSRIS on body function impairment as well as disability scales. Therefore, it is important to understand that the different results between these trials, the FLAME trial and FOCUS could be explained by different factors, such as the different stroke populations (acute vs chronic or hemorrhagic population—in FOCUS), timings and specific measurements of outcomes. However, the main goal of any poststroke rehabilitation trial is to enhance functional recovery after stroke—not only increase the score of a specific scale. Therefore, estimations of minimal clinical differences are important to evaluate if functional recovery is considered to be worthwhile. The estimates for upper limb FMA scales takes into consideration mRS score changes as part of the equation to define how many points of improvement of FMA would be considered as clinical important changes; changes in these scales are therefore associated with meaningful clinical differences. 53 To date, the mechanisms by which fluoxetine may potentially favor motor function remain uncertain 12 and despite the increase in fluoxetine prescriptions poststroke, the accumulated evidence has not been compelling enough to definitively alter stroke management. Our initial hypothesis was that fluoxetine could modulate the excitatory-inhibitory network balance, thus promoting motor recovery in later stroke phases. 11 Although this is a small exploratory study, we showed that fluoxetine may affect excitation-inhibition in subacute/chronic stroke in ways that may differ from acute stroke.
Changes in Intracortical Excitability
There was a decrease in MEP amplitudes in the unaffected hemisphere in the combined group when compared to the placebo group. Even thought our study did not show any significant correlation between motor improvement and changes in MEP amplitudes, other studies showed that improvements in motor performance were associated with a decrease in cortical excitability (MEP amplitudes) in the unaffected hemisphere,54-61 while an increase—as observed in our placebo group—was associated with less motor improvement. However, these changes cannot explain the motor differences observed between the placebo and fluoxetine groups; therefore, these results should be carefully interpreted.
The fluoxetine group developed increased ICF in the unaffected hemisphere by day 90. ICF’s physiological basis is still poorly understood,62,63 but recent studies showed that intra- and interhemispheric competition are correlated with maladaptive plasticity, which can hinder motor function improvements.54,64-67 Following stroke, the reduced inhibitory tonus in both hemispheres contributes to maladaptive plasticity. Therefore, increasing ICF in the unaffected hemisphere could reinforce the excitation-inhibition imbalance 63 ; indeed, in our study, the higher the ICF the less the motor function improvement. However, most stroke trials neither report significant ICF changes (in either hemisphere) nor report any correlation with motor improvement.21,68
The combined and fluoxetine groups had less of an inhibitory response (higher ICI score) than placebo. The association between motor function improvements and ICI is still controversial as motor gains are not always accompanied by ICI changes. 69 Although some studies showed that the greater the ICI response the greater the motor improvement, ICI has not yet been established as a motor function recovery predictor.55,56 Conversely, in our study, the combined group with decreased ICI had a better motor outcome than the placebo and fluoxetine groups—but in the unaffected hemisphere. Most patients had no MEPs in the affected hemisphere, so it was not possible to reliably measure intracortical excitability in the affected M1.
To date, the mechanisms behind motor impairment and recovery after stroke are not fully understood. While MEPs in the affected hemisphere do predict motor responses, multiple trials have been unable to consistently model stroke recovery and treatment response based on other excitability/inhibition measures (eg, MEPs of the unaffected hemisphere, ICF, ICI, and interhemispheric transcallosal inhibition). Moreover, the poststroke interhemispheric inhibition-disinhibition balance depends on a complex system affected by lesion location, infarct size, and degree of dysfunction. 54
Limitations
This mechanistic study has some limitations, such as the small and heterogeneous sample size that reduces the likelihood of detecting treatment effects, as well as nonsignificant differences in time since stroke and vascular territories between the placebo and other groups. However, we adjusted our analyses for potential confounding variables and presented the final results of the adjusted means for time since stroke—the only variable that had a significant confounding effect.
The placebo group had a preponderance of posterior cerebral artery (PCA) strokes, though there were no significant differences between groups with regards to reported vascular territories infarcted on each side. Nevertheless, we cannot rule out the possibility that this nonsignificant difference in vascular territory might have influenced the results, especially regarding placebo group improvements. We described in the Materials and Methods section why we could not quantitatively evaluate for infarcted areas; nor did we consider it a reliable to do an exploratory analysis based on vascular territories as the data was incomplete, the analysis would have been underpowered and would not have accounted for degree of effect on corticospinal tracts. That said, the combined and fluoxetine groups had roughly similarly infarcted motor areas (per available reports), and when each of those groups was compared to placebo the direction of effect was different (combined improved significantly more than placebo while fluoxetine group did significantly worse than placebo). Therefore, while this is a limitation, it did not seem to affect the results of this trial. However, vascular territory is an important—though challenging—consideration for future trials. 70
As we did not have an rTMS-only group (rTMS + placebo fluoxetine) to compare with the combined group, we cannot discern whether the combined intervention’s motor benefits resulted from combining rTMS with fluoxetine per se or whether it resulted from rTMS overcoming any potentially unfavorable fluoxetine effects. Additionally, a long-term follow-up after the end of the treatment would be required to assess for duration of effects. Another potential limitation is that the sham coil has been known to induce low-strength electric fields that can reportedly reach up to 7.2% of the intensity of the active coil. 71 However, our sham method was effective in blinding partly due to the recurrent auditory clicks and the electrode used.
Conclusions
Combined fluoxetine and low-frequency rTMS treatment of the unaffected hemisphere improved motor function in stroke beyond the effects of fluoxetine alone and placebo. Fluoxetine may have had a relatively detrimental effect, leading to increased intracortical facilitation and decreased motor function improvements compared with placebo. Further mechanistic stroke trials are needed to clarify the effects of both treatments.
Supplemental Material
Supplementary_Tables – Supplemental material for Combining Fluoxetine and rTMS in Poststroke Motor Recovery: A Placebo-Controlled Double-Blind Randomized Phase 2 Clinical Trial
Supplemental material, Supplementary_Tables for Combining Fluoxetine and rTMS in Poststroke Motor Recovery: A Placebo-Controlled Double-Blind Randomized Phase 2 Clinical Trial by Camila Bonin Pinto, Leon Morales-Quezada, Polyana Vulcano de Toledo Piza, Dian Zeng, Faddi Ghassan Saleh Vélez, Isadora Santos Ferreira, Pedro Henrique Lucena, Dante Duarte, Fernanda Lopes, Mirret M. El-Hagrassy, Luiz Vicente Rizzo, Erica C. Camargo, David J. Lin, Nicole Mazwi, Qing Mei Wang, Randie Black-Schaffer and Felipe Fregni in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We are grateful to this trial’s participants and study staff that participated in data collection.
Authors’ Note
David J. Lin served as a consultant for Boehringer Ingelheim from 2017 to 2018.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest withrespect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by an NIH R21 grant (R21HD079048-01A1). Clinical Trial Registration Unique identifier: NCT02208466.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
