Abstract
Background. Freezing of gait (FOG) in Parkinson disease (PD) may involve executive dysfunction. This study examined whether executive functioning and attention are more affected in patients with FOG compared with those without and determined whether these processes are influenced by anti-Parkinson medication. Methods. A total of 11 PD patients with FOG, 11 without FOG, and 10 healthy control subjects, matched for age, gender, and education, participated. General motor, mental and cognitive screening tests, as well as specific neuropsychological assessment of executive functions and the Attention Network Test (ANT) were administered. The ANT was conducted in both ON and OFF phases in a counterbalanced design to determine medication-specific effects. Results. FOG showed a clear association with impairment in the executive control network for conflict resolution (inhibition of unwanted responses and impaired response selection), compared with nonfreezers and healthy controls, F(2, 28) = 5.41, P = .01. Orienting and alerting function did not differ between groups, F < 1. Other executive functions, such as abstract problem solving and mental flexibility were not associated with FOG (P > .10). Anti-Parkinson medication did not ameliorate conflict resolution (P > .10), although orienting attention improved with medication, F(1, 17) = 9.81, P < .01. Conclusions. This study shows an association between impaired conflict resolution and FOG, important in understanding the interplay between cognitive and motor problems, which can lead to specific rehabilitation strategies.
Introduction
Freezing of gait (FOG) is a disabling gait disturbance in Parkinson disease (PD), distinct from the main symptoms, such as postural instability, rigidity, bradykinesia, and tremor.1,2 FOG can be defined as a sudden and episodic inability to generate effective stepping 3 and contributes to the development of major disability and frequent falls.4,5
Recently, a pathophysiological model of FOG has been proposed by Lewis and Barker, 6 suggesting that FOG is linked to motor, limbic, and cognitive brain circuits. When dopaminergic reserve is insufficient, increased demands (stress, anxiety, or a cognitive challenging task) can lead to an inability to shift from one response set to another, triggering FOG. Frontal–basal ganglia circuits, vital to gait, are hypothesized to play an important role in the executive control of action, including the capability to inhibit unwanted response tendencies. 7 The disruption of frontostriatal pathways presumably plays a key role in FOG as it is related to disease progression and cognitive impairment.8,9 Therefore, FOG may be attributed to an insufficient frontostriatal drive, hampering the intended gait cycle. 10 Reduced frontostriatal drive could lead to impaired response selection under conflict, presumably present in both freezers and nonfreezers, but possibly worse in freezers.
Although an association between FOG and disease severity, depression, stress, anxiety, and cognitive function is proposed by Giladi and Hausdorff, 11 an underlying mechanism currently remains unclear. Recent studies have demonstrated impairment in task switching (or set shifting) under temporal pressure in patients with FOG 12 and showed correlations with generalized executive dysfunction and Stroop performance. 13 In this study, we focused on the assessment of executive functioning, and we investigated whether executive dysfunction is associated with attention-related impairment in FOG. Therefore, we examined attention networks in patients with and without FOG using the Attention Network Test (ANT). 14 The ANT is a choice reaction time (RT) task developed to reliably test the efficiency of 3 attentional networks. 15 The alerting network is hypothesized to achieve and maintain an alert state, the orienting network reflects the ability to select information from sensory input, and the executive control network represents cognitive control processes. The ANT is an appropriate tool to test whether one of the proposed attention networks is associated with gait disturbances in PD. We hypothesize that PD patients with FOG are characterized by a specific deficit in executive control, while orienting and alerting function are preserved. Executive control or conflict resolution can be seen as a major component of executive functioning depending on basal ganglia–prefrontal interactions. 16 Interference control in PD patients can be intact 17 or impaired 18 ; however, none of these studies checked for the occurrence of FOG as a contributing factor to conflict resolution deficits. Combining ANT data with neuropsychological assessment, comprising specific domains of executive functioning, will give us the opportunity to construct an enhanced cognitive profile for patients with FOG.
A final research question is whether executive functioning in PD is affected by medication. Hausdorff et al 19 found that stride-to-stride variability was markedly increased in PD patients with FOG in both ON and OFF phases. However, for a great majority of PD patients, freezing episodes are more frequent and severe in OFF phase,20,21 but FOG in ON phase still occurred. Dopamine therapy ameliorates motor symptoms in PD. However, with respect to cognition, both neutral and worsening of performance has been reported. 22 Several studies have yielded mixed results, probably because varying doses of dopaminergic medication are needed to reach optimal dopamine level in different brain regions relevant for cognitive functioning. 23 Knowing that D1 receptors in the prefrontal cortex operate under an inverted U-shaped dose-dependent response curve, 24 it is possible that either insufficient or excessive dopamine levels lead to impairment in cognitive functioning. According to optimal dopamine levels reached with prescribed anti-Parkinson medication, we predict that PD patients will perform better on the executive control network of the ANT in the ON phase of the medication cycle compared with the OFF phase. Moreover, we expect that presumed ANT differences between freezers and nonfreezers arise in both ON and OFF phases of the disease, since medication is known to have variable and limited effects on FOG. 1
Method
Participants
A total of 22 PD patients (11 with FOG, FR; 11 without FOG, nFR) and 10 healthy controls (HC) participated in the study. All PD patients were diagnosed by a specialist neurologist in movement disorders and underwent both neurological and neuropsychological examinations. Patients scoring above zero on the revised Freezing of Gait Questionnaire (NFOGQ), 25 meaning they experienced FOG at least once during the past month, were assigned to the FR group. A video, showing several examples of FOG, was shown as a part of the NFOGQ. Participants had no orthopedic or additional neurological disorders. Participation in the experiment was voluntary with informed consent in accordance with the Ethics Committee of the Vrije Universiteit Brussel (VUB).
Both PD patient groups were compared with the HC group, matched for age, gender, and education. All participants scored above the standard cutoff score of 24 on the Mini-Mental State Examination (MMSE). 26 In addition, the scales for outcomes in Parkinson disease cognition (SCOPA-COG) 27 were used to assess cognitive functions in PD. A measure of affective disturbance was obtained by administering the Hospital Anxiety Depression Scales (HADS). 28 Scores higher than 7 points on HADS subscales indicated increased complaints associated with anxiety and depression. Detailed neuropsychological testing of executive functioning was assessed using abstract problem solving (Matrix Reasoning subtest, Wechsler Adult Intelligence Scale–III), 29 mental flexibility (Brixton Spatial Anticipation Test), 30 and verbal fluency (Controlled Oral Word Association Test) 31 tasks, all conducted during the ON phase of the disease. Low scores on these neuropsychological tests are indicative of a deficit (the reverse is true for the Brixton Spatial Anticipation Test).
Attention Network Test
The ANT gives us the opportunity to dissociate three attention networks, namely alerting, orienting, and executive control on the basis of RT differences. A larger difference between 2 conditions suggests more impairment of the corresponding network.
The ANT was conducted in the patients’ home setting, and the experiment was run on an Intel Core 2 Duo portable computer with 15.6-inch screen (Intel, Santa Clara, California), using E-Prime Version 1.1 software. 32 ANT testing took place under the supervision of the experimenter.
Participants were seated approximately 65 cm from the computer screen. They were instructed to respond as quickly and accurately as possible to the direction (left or right) of a central arrow (target) by pressing the “c” key for the left direction and the “n” key for the right direction. The central arrow was presented above or below the fixation point, with 2 identical distracters on each side, referred to as flankers. Flankers pointed in the same (congruent condition) or opposite (incongruent condition) direction as the central target arrow. In the neutral condition the flankers were 4 lines without arrowheads.
A trial lasted 4000 ms and consisted of 5 events (see Figure 1). First, a fixation cross was presented in the center of the screen for a random variable fixation period between 400 and 1600 ms. Then a warning cue (asterisk) appeared for 100 ms. There were 4 types of warning cues (see Figure 1): no cue, center cue (participants were shown an asterisk at the location of the fixation cross), double cue (2 asterisks appeared; one above and one below fixation cross), or a spatial cue (an asterisk was presented at the location where the target would appear). After the warning cue, there was a short fixation period of 400 ms followed by the stimulus, consisting of the 5 arrows, presented above or below the fixation cross. The stimulus was presented until the participant responded, but no longer than 1700 ms. After a variable posttarget fixation period, the next trial began.
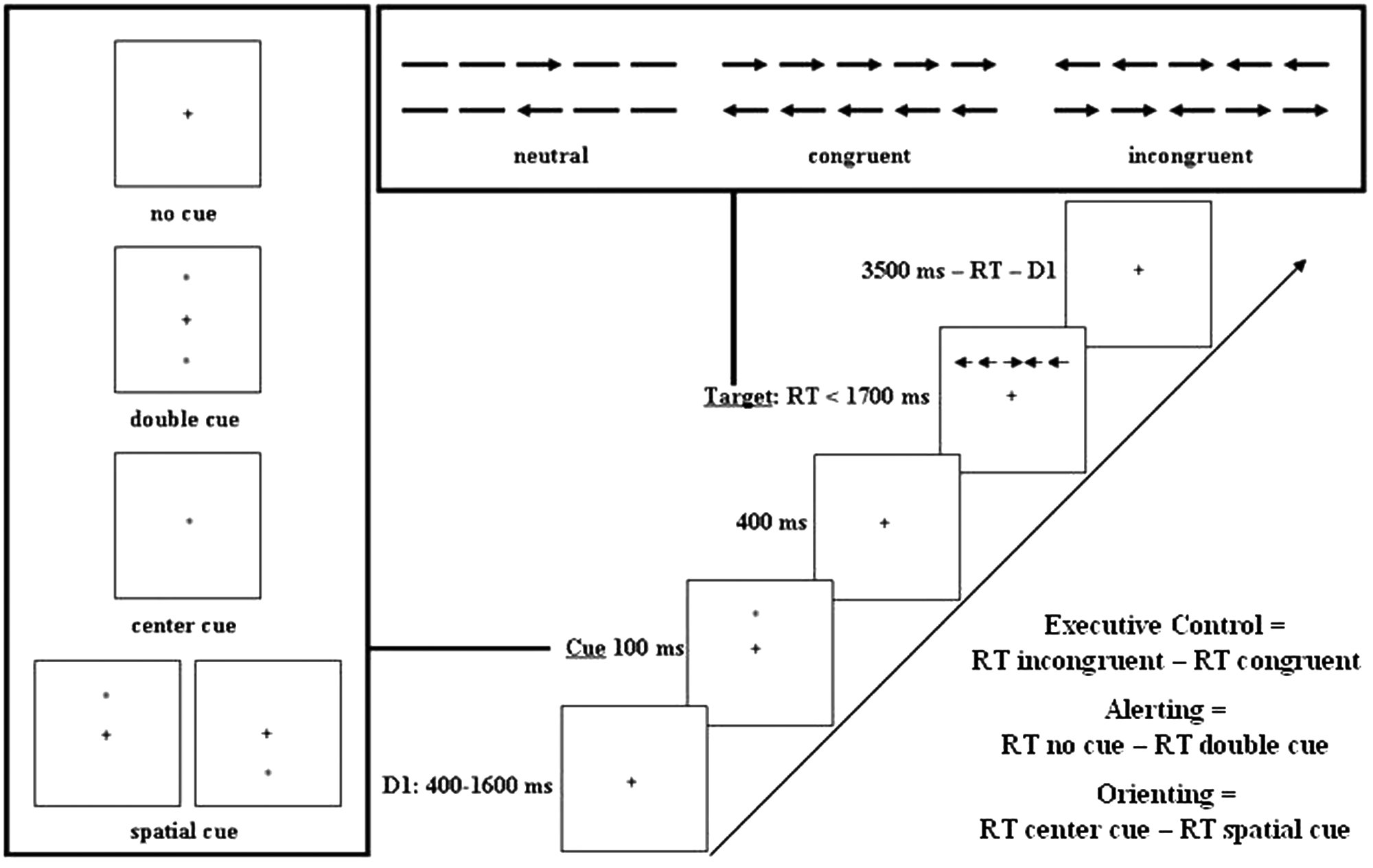
The Attention Network Test (ANT), adapted from Fan et al. 14 The experimental procedure is explained with an example of an incongruent trial, the 4 possible cues, and the 3 flanker conditions. The calculation of the 3 ANT networks is shown in the bottom right corner of the figure.
Median RTs in each condition (no cue, double cue, center cue, spatial cue, neutral flanker, incongruent flanker, and congruent flanker) were used to assess the 3 attentional networks (alerting, orienting, and executive control). The executive control network represents a measure for conflict resolution, since participants need to resolve conflict by inhibiting an irrelevant response. Alerting and orienting networks represent vigilance and the ability to focus attention, respectively.
Participants completed a practice block of 24 trials. After practice, participants executed 3 experimental blocks, each consisting of 96 trials (4 cue conditions × 2 target locations × 2 target directions × 3 flanker conditions × 2 repetitions). The presentation of trials was in random order. The practice block took about 2 minutes and the experimental blocks about 18 minutes to accomplish. Feedback on accuracy was given during the practice block only. Participants could rest between blocks.
All patients were asked to complete the ANT in both ON (about 60 minutes after their morning dose of anti-Parkinson medication) and OFF phase (early in the morning, between 9 and 12 hours after their last medication intake), with 1-week interval between both sessions. ANT administration was counterbalanced: In both groups, half of the patients started with the ANT in ON phase, the other half started with the ANT in OFF phase.
Statistical Analysis
Independent t tests and a correlation analysis, using the Spearman ρ statistic to explore the relationship between severity of freezing and clinical, motor, and neuropsychological measures, were used to analyze group differences. Mixed factorial analyses of variance (ANOVAs; with Huyhn–Feldt corrections for violations of sphericity) were implemented to analyze ANT performance. For the alerting network, we performed a 3 × 2 mixed factorial ANOVA with group as between-subjects factor (FR, nFR, and HC) and cue type (no and double cue) as within-subjects factor. For the orienting network, we used a 3 × 2 mixed factorial ANOVA with group as between-subjects factor (FR, nFR, and HC) and cue type (center and spatial cue) as within-subjects factor. For the executive control network, we applied a 3×2 mixed factor ANOVA with group as between-subjects factor (FR, nFR, and HC), and flanker type (congruent and incongruent flankers) as within-subjects factor. Bonferroni post hoc tests were performed in case of significant group differences. Data analysis was performed using SPSS Version 17.0 (IBM, Somers, New York), and all analyses were 2-tailed, using a significance level of .05.
Results
Clinical, Motor, and Neuropsychological Differences and Correlations
PD patients were rated by the Hoehn and Yahr (H&Y) 33 scale and assessed with section III of the Unified Parkinson’s Disease Rating Scale (UPDRS) 34 in the OFF phase. Medications used by patients consisted mostly of levodopa products and dopamine agonists. One nFR and 2 FRs were on anticholinergic treatment, and 2 patients (one in each group) were taking a selective serotonin reuptake inhibitor.
Neuropsychological assessment of executive functioning showed that FRs and nFRs scored worse than HCs on matrix reasoning, and nFRs scored worse on the Brixton Spatial Anticipation Test compared with HCs. However, differences between FRs and nFRs on these executive-based tests failed to reach significance. General cognitive, affective, and motor measures revealed no differences between PD groups, with the exception of the MMSE score, which was lower for FRs than both nFR and HCs, and the SCOPA-COG, for which FRs scored lower than HCs. Means and standard errors for these tests and group differences are reported in Table 1.
Clinical, Motor, and Neuropsychological Measurements Across PD Groups (FR and nFR) and HC
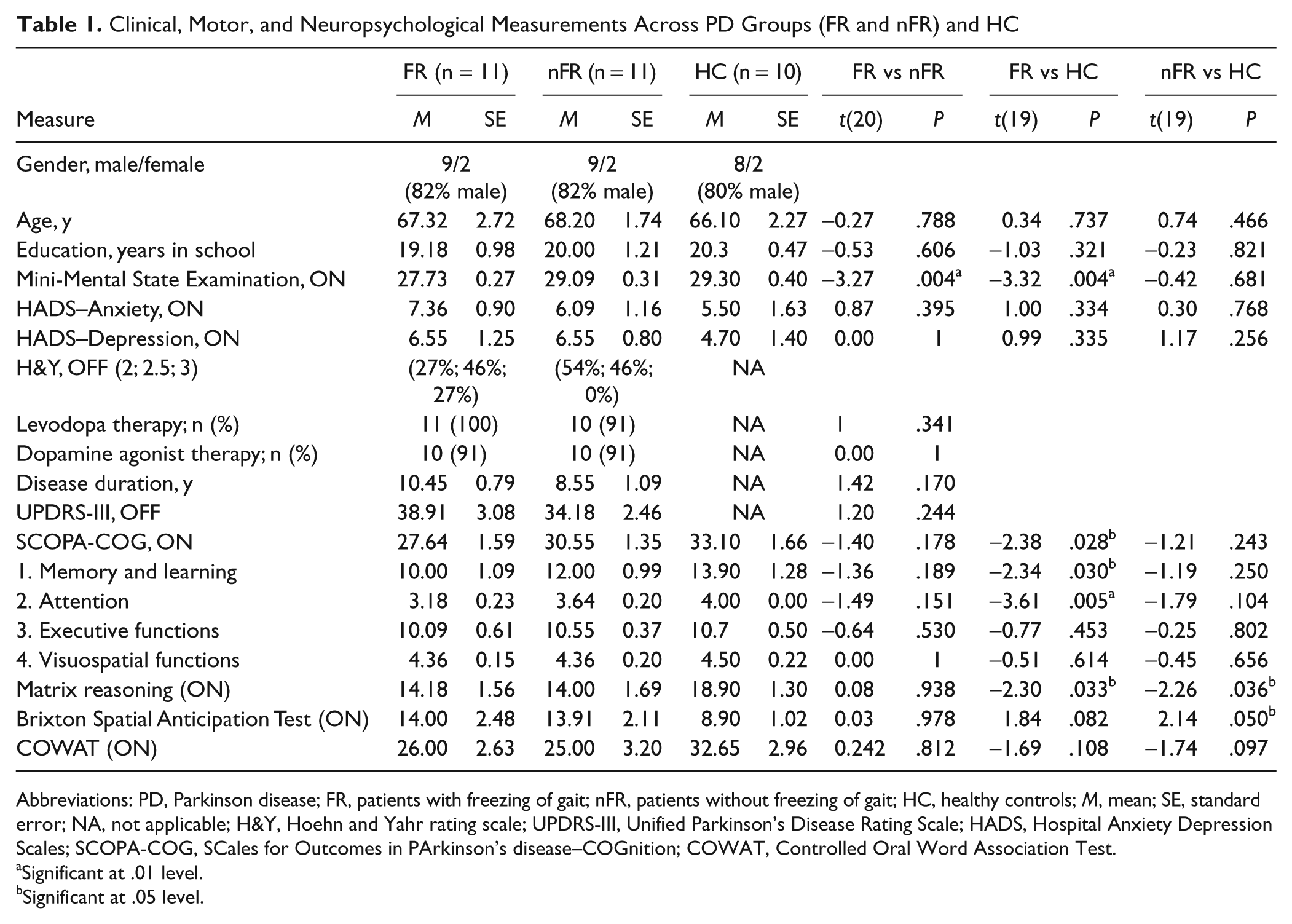
Abbreviations: PD, Parkinson disease; FR, patients with freezing of gait; nFR, patients without freezing of gait; HC, healthy controls; M, mean; SE, standard error; NA, not applicable; H&Y, Hoehn and Yahr rating scale; UPDRS-III, Unified Parkinson’s Disease Rating Scale; HADS, Hospital Anxiety Depression Scales; SCOPA-COG, SCales for Outcomes in PArkinson’s disease–COGnition; COWAT, Controlled Oral Word Association Test.
Significant at .01 level.
Significant at .05 level.
Table 2 shows that severity of freezing (FR: M = 12.7, SE = 2.44) as measured by the NFOGQ, correlated positively with disease duration, stage of the disease (H&Y), motor symptom severity (UPDRS-III), increased depressive (HADS–Depression) and anxiety (HADS–Anxiety) complaints, P < .05. None of the neuropsychological tasks measuring executive functioning related significantly to FOG severity.
Correlations Between Clinical and Neuropsychological Measures and the Total Score on NFOGQ
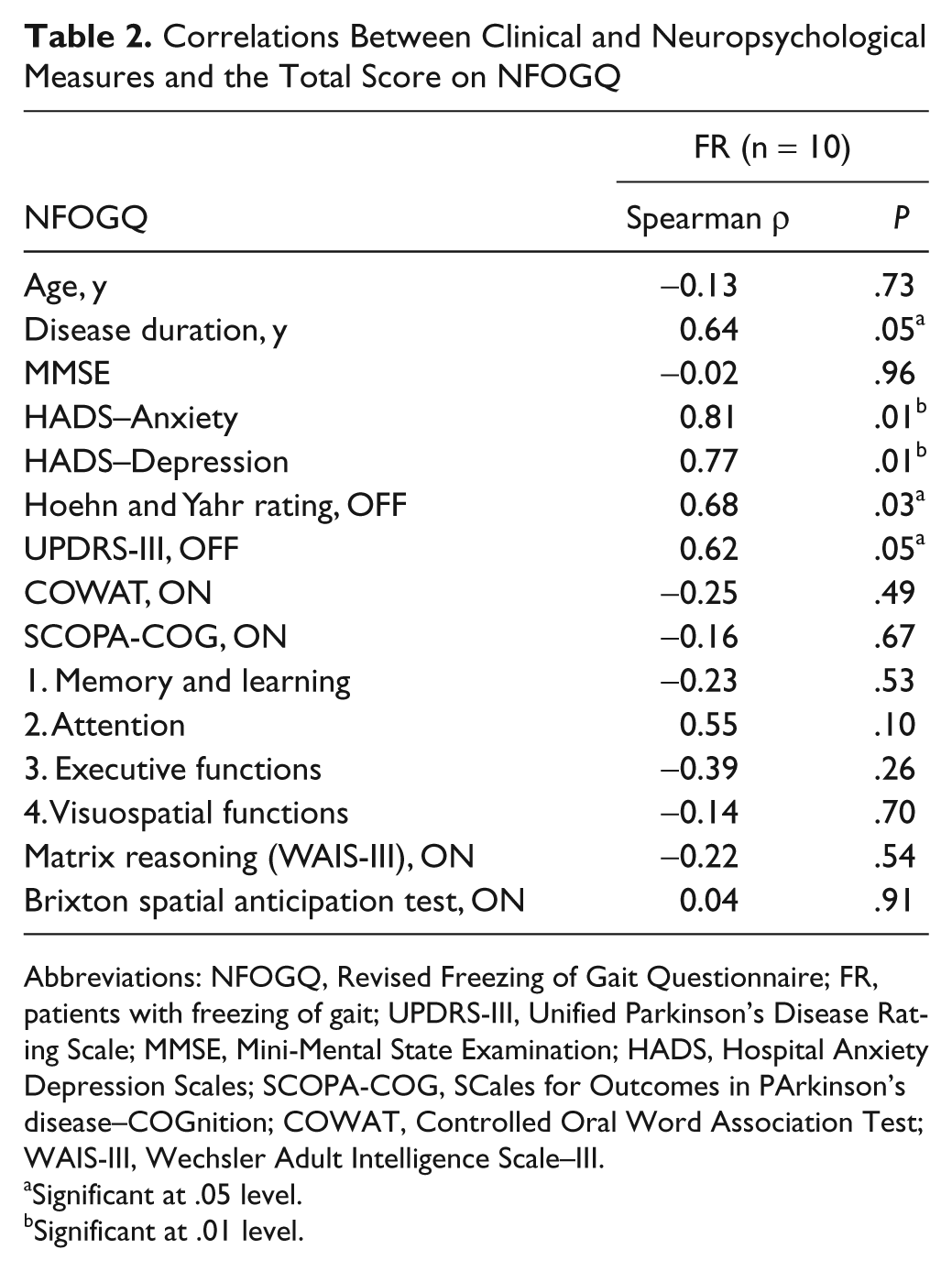
Abbreviations: NFOGQ, Revised Freezing of Gait Questionnaire; FR, patients with freezing of gait; UPDRS-III, Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; HADS, Hospital Anxiety Depression Scales; SCOPA-COG, SCales for Outcomes in PArkinson’s disease–COGnition; COWAT, Controlled Oral Word Association Test; WAIS-III, Wechsler Adult Intelligence Scale–III.
Significant at .05 level.
Significant at .01 level.
Attention Network Test
In the ANT, the 3 attention networks were assessed as follows: alerting, no cue versus double cue trials; orienting, center cue versus spatial cue trials; and executive control, incongruent versus congruent trials. RT analysis was performed on participants’ median RTs of correct trials. Erroneous responses and responses following an error were discarded from the analysis. In ON phase, one patient in the FR group was left out of the analysis because of an extreme low overall accuracy rate (68%). Surprisingly, the same patient performed adequately (accuracy rate >85%) in the OFF phase. However, 2 other FR patients were unable to perform the ANT in the OFF phase because of motor limitations.
Median RTs are displayed in Table 3 for all conditions and groups in both ON and OFF phase. Accuracy for each ANT condition (see Table 3) and overall accuracy rates (FR, M = 96.77%, SE = 0.91; nFR, M = 96.75%, SE = 1.28; HC, M = 98.47%, SE = 0.28) did not differ significantly between groups. Therefore, network effects were determined on the basis of the RT analysis only.
Median Reaction Times in Milliseconds (SE) and Accuracy Rates in Percentage (SE) for All Conditions (Congruent, Incongruent, No Cue, Center Cue, Double Cue, Spatial Cue) and Groups (FR, nFR, HC) in Both ON and OFF Phases
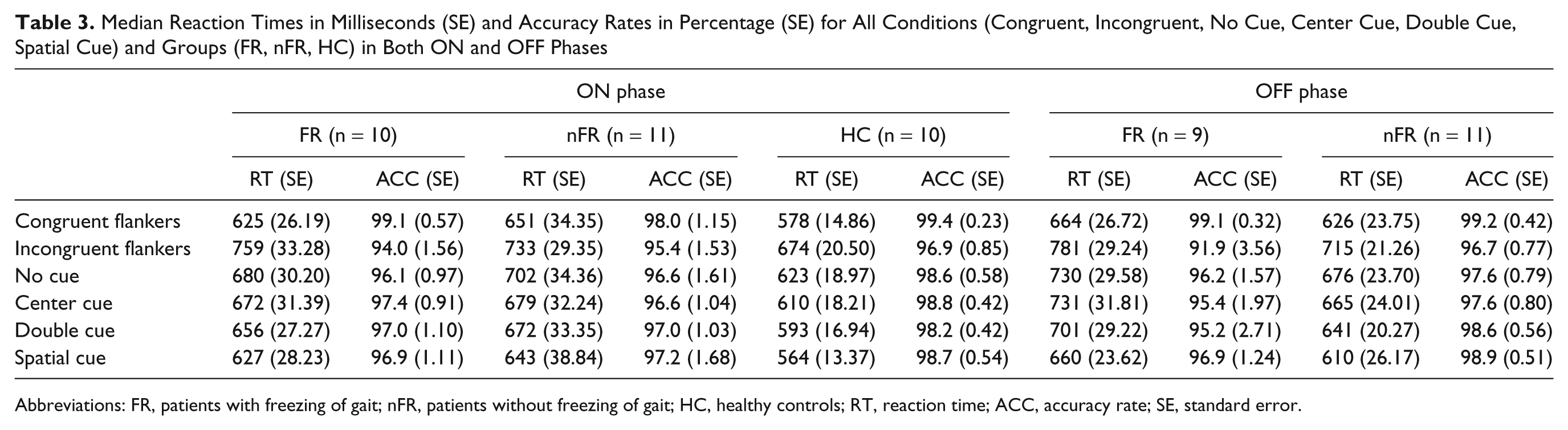
Abbreviations: FR, patients with freezing of gait; nFR, patients without freezing of gait; HC, healthy controls; RT, reaction time; ACC, accuracy rate; SE, standard error.
A mixed factorial ANOVA was carried out for each of the 3 attention networks to selectively examine the association with FOG. Main network and group effects, and group × network interactions for the 3 ANOVAs, will be reported together. To compare patient groups with HCs, first the performance in ON phase was analyzed. OFF phase performance and medication effects are reported later.
Statistically significant differences were found for each of the 2 main network effects, namely responses were faster in (1) double than no cue trials [alerting; F(1, 28) = 64.38, P < .001]; (2) spatial than center cue trials [orienting; F(1, 28) = 54.03, P < .001]; and (3) congruent than incongruent flanker trials [executive control; F(1, 28) = 245.07, P < .001]. There was no significant main group effect in any of the 3 analyses, indicating that RT level did not differ significantly between groups [for alerting, F(2, 28) = 2.17, P = .13; for orienting, F(2, 28) = 1.91, P = .17; and for executive control, F(2, 28) = 1.96, P = .16]. Importantly, we found a significant group × executive control network interaction [F(2, 28) = 5.41, P = .01], meaning that there were congruency differences between groups (see Figure 2). A Bonferroni post hoc test confirmed that FR patients showed a greater RT difference between congruent and incongruent trials (M = 134 ms, SE = 13 ms) compared with nFR patients (M = 82 ms, SE = 8 ms; P = .01, d = 1.51). Surprisingly, congruency differences were not significant between FRs and HCs (M = 96 ms, SE = 14 ms; P = .09, d = 0.90). Subsequent power analyses (G*Power 3) 35 showed that for a statistical F test, type repeated measures within-between interaction, with α = .05, 2 groups (FR and HC; n = 21) and 2 measurements (incongruent vs congruent), the power (1 − β) to detect a small (f = 0.10), medium (f= 0.25), and large effect (f = 0.40) amounted to 0.46, 0.99, and 1, respectively. This demonstrates that our study had insufficient power to detect small effects, thereby possibly masking the significance of a difference in conflict resolution between FR and HC groups.
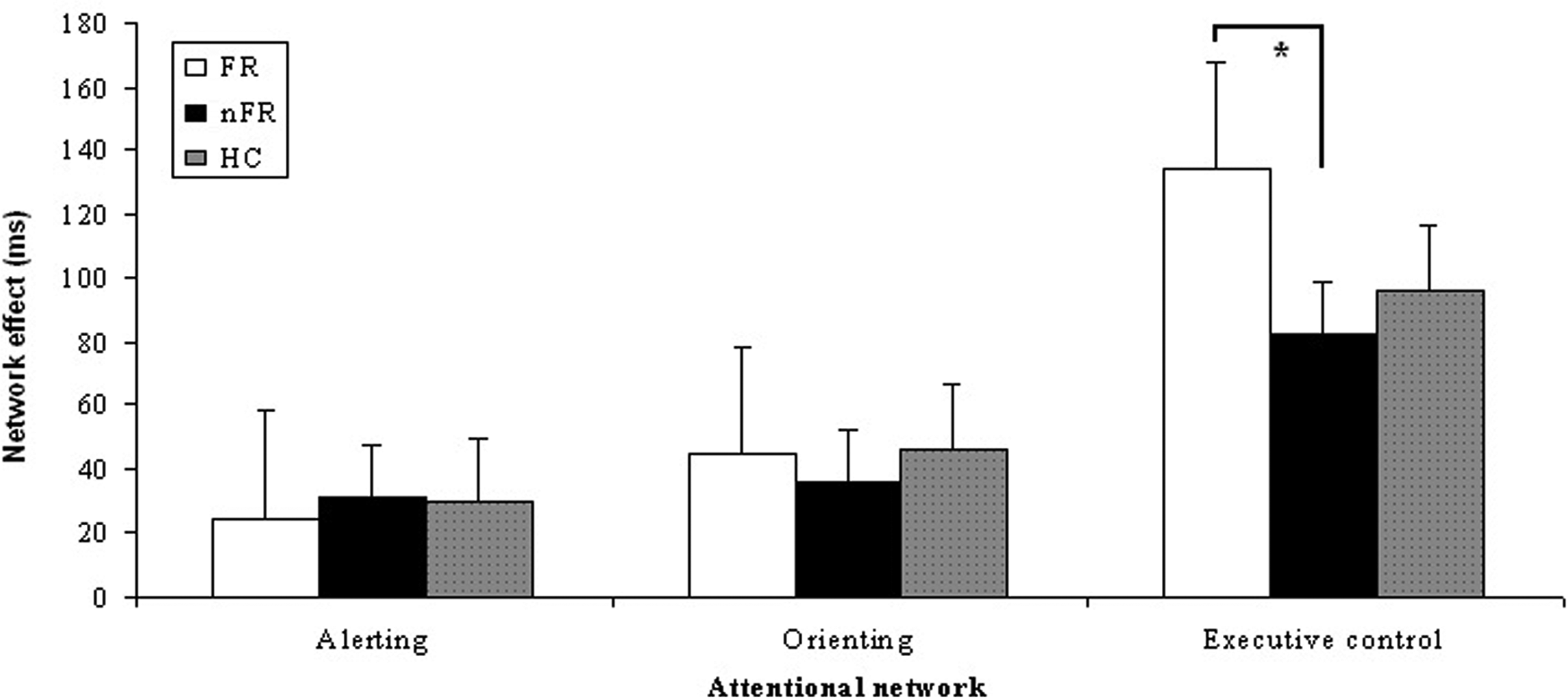
Mean network effects in milliseconds per attentional network (alerting, orienting, and executive control) for the freezing (FR) and nonfreezing (nFR) Parkinsons disease patients and healthy control subjects (HC) in ON phase (*significant at .05 level).
Differences between nFRs and HCs were not significant, P = 1. Group × alerting and group × orienting interactions were nonsignificant, F(2, 28) = 0.32, P = .73 and F(2,28) = 0.31, P = .74, respectively.
RT analysis in OFF phase of the ANT performance showed the same results. Network main effects were significant, main group differences were absent and a group × executive control network interaction was found. To determine differences between the ON and OFF phases of the ANT performance, we conducted a 2 × 2 mixed factorial ANOVA on each of the 3 attention networks with group as between-subjects factor (FR, nFR), and medication (ON and OFF phase) as within-subjects factor. We only observed a main effect of medication for the orienting network, indicating that the orienting function improved with medication [F(1, 17) = 9.81, P < .01]. Moreover, no main group effect [F(1, 17) = 0.75, P = .40] or group × medication interaction [F(1,17) = 0.24, P = .63] could be found for the orienting network, suggesting that medication effects were similar for FRs and nFRs. For the alerting and executive control network, no medication effect was observed (see Figure 3).
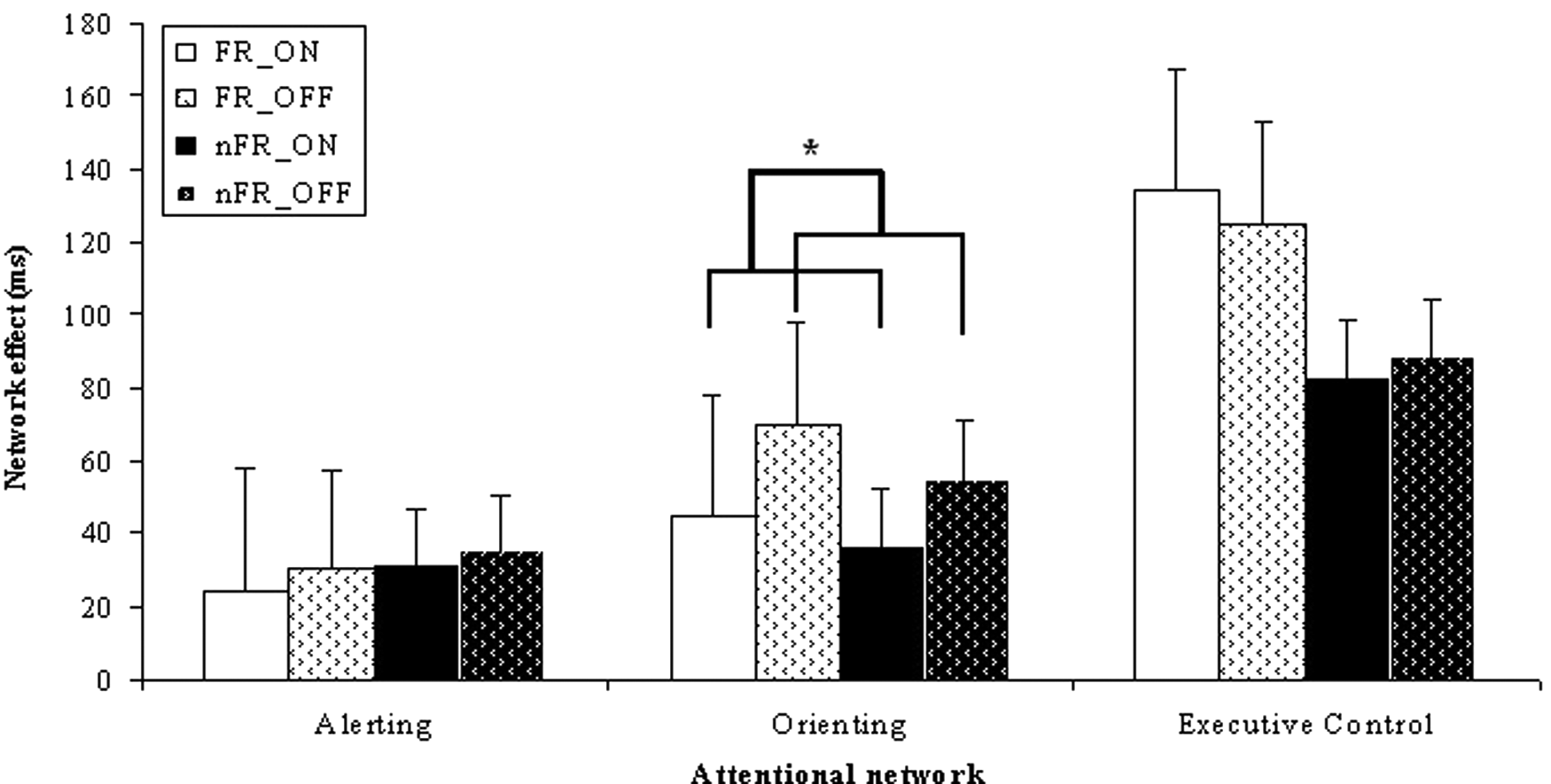
Mean network effects in milliseconds per attentional network (alerting, orienting, and executive control) and medication state (ON and OFF phases) for freezing (FR) and nonfreezing (nFR) Parkinsons disease patients (*significant at .05 level).
Discussion
In the present study, we demonstrated that a specific executive impairment, that is, conflict resolution, can be associated with FOG in PD. Resolving conflict has been shown to rely on frontostriatal and mesocorticolimbic pathways, 36 presumed to be important in FOG. 6 Other attentional networks, such as those for orienting and alerting remained preserved. Neuropsychological assessment, namely abstract problem solving, mental flexibility, and verbal fluency, showed no significant differences between FR and nFR.
If we take a look at the general screening tests, results showed that SCOPA-COG scores were significantly lower for FRs compared with scores for HCs, revealing specific impairments in memory and attention subscales. Although nFRs scored in between FRs and HCs, they did not differ statistically from either group. Surprisingly, the MMSE was better able to make a distinction between FR and nFR patients. Nonetheless, both groups obtained scores above the used cutoff scores of 24, indicating that none of the patients were suffering from a frank dementia. HADS scores did not reveal any group differences, although average scores for the PD groups were relatively high. Furthermore, correlations between FOG severity and clinical and cognitive measures revealed associations with disease duration, motor examination (H&Y and UPDRS-III), and affective state (anxiety and depression) within the FRs. This fits well with the conceptual framework proposed by Giladi and Hausdorff, 11 linking motor, cognitive, and affective aspects to FOG.
The ANT was used to assess executive and attentional networks in FRs and nFRs who were matched on age, gender, and education. ANT performance showed that the executive control network of attention was impaired in PD patients with FOG. Slower RTs in resolving conflict when confronted with irrelevant information and faster responses for automatic responses indicated a specific deficit of conflict resolution. The FR group was unable to control conflict to the same extent as nFRs and HCs. This is in agreement with the results of Amboni et al 13 who found a preliminary indication for a conflict resolution deficit in FOG, namely a correlation between FOG and their performance on a Stroop task. However, conflict resolution involves several subprocesses such as target detection, response selection, and response inhibition. Further studies should focus on these processes to provide further insight in this specific impairment.
In contrast to our hypothesis, impairment in conflict resolution was present in both ON and OFF phases, suggesting that commonly used anti-Parkinson medication exerted no alleviating effect. Although FOG is considered a relatively dopamine-resistant phenomenon in gait, 37 it can also be significantly alleviated by dopamine. Conflict resolution deficits may therefore relate to components of FOG that are nondopaminergic in origin. Orienting and alerting networks, on the other hand, did not reveal deficits related to FOG. Moreover, comparing patients in the ON and OFF phases showed a comparable improvement in the orienting function in both PD groups because of medication. This is rather surprising since the orienting network is assumed to rely on acetylcholine instead of dopamine transmission. 14
Interestingly, the remaining neuropsychological tests were unable to pinpoint executive impairments associated with FOG. Abstract problem solving, verbal fluency, and mental flexibility showed a tendency to differ between PDs and HCs, but these executive measures were apparently not sensitive enough to make the distinction between FRs and nFRs. Recent studies already showed the importance of task switching (or set shifting) in FOG, whereas other executive domains such as verbal fluency and working memory had weaker associations.12,13 Nevertheless, more work on larger groups is needed to capture subtle differences between PD subgroups.
Our findings show similarities with the observations of Naismith et al, 12 who demonstrated the specific role of set shifting under temporal pressure. Conflict resolution and set shifting seem to be key abilities in constructing the cognitive profile of patients with FOG. Our results also confirm clinical observations that FOG is triggered in situations that require response selection, such as during dual tasking and functional transitions.
For therapists, we can recommend 2 approaches based on our findings: First, patients should be made consciously aware of their difficulties with switching in the face of conflicting stimuli and taught to voluntarily stop, rather than freeze and restart walking using a cognitive weight-shifting strategy. 38 Frequent practice of this strategy in complex circumstances and environments may make this compensatory approach easy to access for patients in daily life. Second, patients can use auditory cueing during gait activities when exposed to challenging stimuli. Cues during dual tasking may reduce rather than increase dual task interference,39,40 perhaps because of more efficient allocation of attention or to the fact that external cues may maintain the gait pattern, hence making gait performance less susceptible to rival responses. More knowledge of the underlying cognitive impairments in FOG may produce other efficient therapies adjusted to the abilities of patients.
Footnotes
Acknowledgements
We thank Dr Wim Vandenberghe for his assistance in recruiting patients with Parkinson’s disease.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This research was funded by the Flanders Research Funds (G.0691.08) and the Research Council of the Vrije Universiteit Brussel (grant OZR-1933 BOF).
