Abstract
Background
Several studies have shown that robotic devices can effectively improve motor function in stroke patients through limb activation. However, the effects of robot-assisted therapy on perceptual deficits after stroke is unclear.
Objective
This review aimed to evaluate the effectiveness of robotic limb activation in patients with unilateral spatial neglect (USN) after stroke.
Methods
In this systematic review, a literature search was performed using MEDLINE, EMBASE, CENTRAL, CINAHL, and LILACS databases without language restrictions. Randomized controlled trials (RCTs) and quasi-RCTs of robot-assisted therapy for USN after stroke were selected. Two reviewers independently assessed the risk of bias and certainty of the evidence of the included studies.
Results
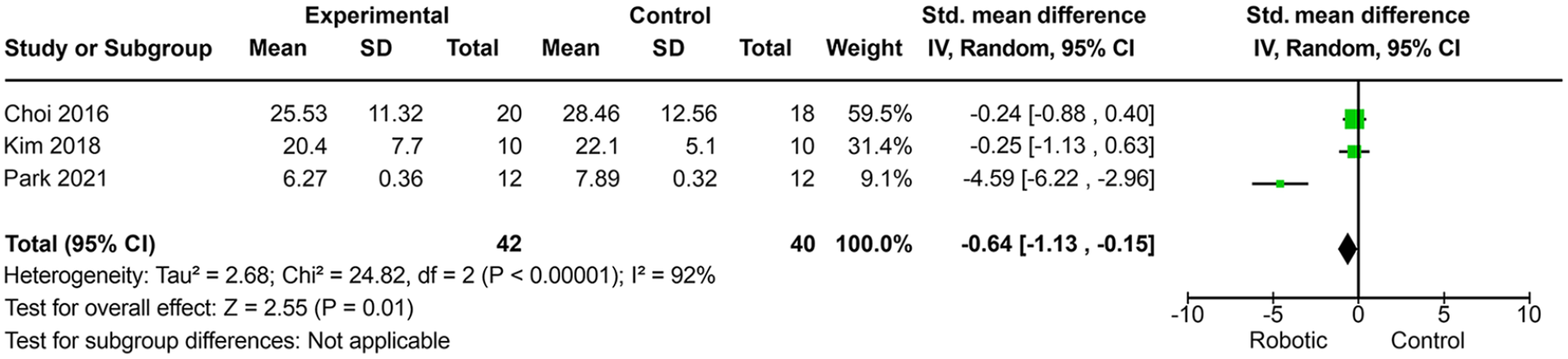
A total of 630 studies were identified, including five studies for qualitative synthesis and four meta-analyses. The results of RCTs comparing robotic limb activation with a control group suggested an improvement in the degree of USN measured by the line bisection test (standardized mean difference [SMD], −0.64; 95% confidence interval [CI], −1.13 to −0.15; P = .01). There were no differences between the groups in the motor-free visual perception test 3rd edition (SMD, 0.27; 95% CI, −0.25–0.79; P = .31), star cancellation test (SMD, 0.26; 95% CI, −0.42−0.94; P = .54), Albert’s test (SMD, −0.67; 95% CI, −2.01−0.66; P = .32), and Catherine Bergego Scale (SMD, −0.81; 95% CI, −2.07−0.45; P = .21).
Conclusion
The study demonstrated that limb activation through robotic therapy can improve midline perception. However, there was no impact on tasks assessing visual scanning, functionality, or activities of daily living.
Introduction
The nonrecognition of space, usually on one side of the body, or any stimulus applied to it can be called hemineglect syndrome or unilateral spatial neglect (USN).1,2 USN is characterized by the inability to report or respond to people or objects presenting contralaterally to the injured side of the brain, which cannot be explained by primary motor or sensory deficits. 3 This syndrome occurs in approximately 50% of individuals after the stroke of the right hemisphere, and up to 75% may persist with some symptoms in the chronic phase. 4 Moreover, USN has important clinical implications, as it leads to prolonged periods of hospitalization, impaired functional recovery, and worsening quality of life.5,6
A tendency to explore and accentuate orientation to the ipsilesional side of the patients and nonrecognition of items on the contralesional side is provoked by USN.2,7-14 Main treatments for patients with USN after stroke are the “top-down” approaches, 15 which aim to implant voluntary attention, and “bottom-up” techniques, which aim to automatically capture attention to stimuli or events in the environment.16-22 There is evidence of the effectiveness of several “bottom-up” approaches to improve USN performance, among which limb activation has been highlighted as a technique to reduce visuospatial deficits in patients with USN after stroke.23-27
Given the high incidence of upper limb paresis associated with USN after stroke, the positive effect of passive or active movement of the left upper or lower limb can be an excellent strategy in the field of USN rehabilitation. 27 Furthermore, Frassinetti et al. observed a reduction in USN during passive and active movements of the left hand. 28 Some authors have reported that somatosensory activation on the contralateral side of limb activation facilitates the neural circuits underlying the representation of space, which could explain the clinical efficacy of activation of the left hand in improving USN.29,30
With the development of robotic devices, there is a growing interest in using them in post-stroke rehabilitation, as they can provide a consistent, intensive, and accurate rehabilitation process for the paretic limb. 31 In fact, several robotic devices have been developed in the last decade and have been shown to be effective in improving motor function in stroke patients using passive, assisted, and active training modes. 32 However, unlike motor function in the upper limb, few studies have reported the effects of robot-assisted therapy in patients with USN after stroke. In the last decade, there have been some clinical trials using limb activation through robotic therapy for patients with USN; however, no systematic review has assessed the effectiveness of this training in guiding clinical practice. Therefore, this review aimed to identify the effects of robot-assisted training on patients with USN after stroke.
Materials and Methods
This review was based on the methods described in the Cochrane Handbook for Intervention Reviews 33 and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 34 Randomized clinical trials (RCTs) and quasi-RCTs conducted up to February 2022 were included in this evaluation. This review was registered in the International Prospective Register of Systematic Reviews (CRD42022303587).
Eligibility Criteria
Study Design
RCTs and quasi-RCTs.
Participants
Adults with an objective diagnosis of USN after stroke (ischemic or hemorrhagic), over 18 years of age, regardless of sex, duration of illness, or severity of initial impairment.
Interventions
Therapies that use robot-assisted therapy including an exoskeleton (based on torque actuators that control each joint of the affected limb) or end-effector (guides only the distal part of the affected limb).
Comparators
The interventions were compared with any conventional rehabilitation for USN after stroke (e.g., pharmacological or non-pharmacological therapies, such as “top-down” techniques, which include noninvasive brain stimulation and mental practice, and “bottom-up” techniques, which include prismatic adaptation, visual scanning training, optokinetic stimulation, virtual reality, combined neck vibration, voluntary trunk rotation, vestibular rehabilitation, and paretic limb activation).15-24
Outcomes
(1) USN degree measured by any paper and pencil test, 35 such as the line cancellation task, line bisection test (LBT), and star cancellation test, and any validated specific instrument, such as the Catherine Bergego Scale 36 or the behavioral inattention test (BIT) 37 after treatment and in the long-term; (2) functional disability measured by the modified Rankin scale 38 or the Fugl–Meyer scale 39 after treatment and in the long-term; (3) autonomy in activities of daily living measured by any scale validated in this population, such as the Barthel index 38 after treatment and in the long-term; (4) balance measured using the Berg Balance scale 40 or mini-best test 41 after treatment and in the long-term; (5) depression or anxiety measured using the Beck Depression Inventory, 42 hospital anxiety and depression scale, 43 and Hamilton Depression Rating Scale 44 after treatment and in the long-term; and (6) quality of life, measured by the stroke impact scale, 45 EUROQOL, 46 and SF-36 47 after treatment and in the long-term.
Search Strategy and Databases
The Cochrane Central Register of Controlled Trials (CENTRAL) (1966 to February 2022), EMBASE (OvidSP) (1980 to February 2022), Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library, 2022), CINAHL (1961 to February 2022), and Latin American and Caribbean Center on Health Sciences Information (LILACS) (1982 to February 2022) were used without language restrictions. The most recent search date was February 26, 2022. All searches were performed by a librarian trained in systematic reviews. The reference lists of relevant studies and conference proceedings were also searched, and the authors of the included studies were contacted. The review search strategy for each database is shown in Supplemental File 1.
Selection of Studies
Two pairs of reviewers independently examined all titles and abstracts identified in the literature search, obtained full articles from all potentially eligible studies, and assessed them for eligibility. Disagreements were resolved by discussion or, if necessary, with the help of a third reviewer. Studies that were presented at conferences were also included.
Data Extraction
The reviewers performed calibration exercises and worked in pairs to independently extract data from the included studies. The following data were extracted using a pre-tested data extraction form: study design, participants, interventions, comparators, assessed outcomes, and relevant statistical data.
Risk of Bias Assessment
Reviewers working in pairs independently assessed the risk of bias in the included studies using the modified version of the Cochrane Collaboration instrument (http://distillercer.com/resources/).48,49 For incomplete outcome data in individual studies, we set a low risk of bias for loss of follow-up at <10% and a difference of less than 5% in missing data between the intervention and control groups. When information on the risk of bias or other aspects of the methods or results was not available, we attempted to contact the authors for additional information.
The Physiotherapy Evidence Database (PEDRO) scale was also used to assess the risk of bias. It is based on the Delphi list and consists of verifying eligibility criteria, randomization and allocation concealment, the similarity between groups and subjects, verifying whether therapists and evaluators were blinded, 85% of subjects remaining to initiate the study, intent-to-treat outcome analysis, intergroup comparison, and measurement of precision and variability in at least one outcome. The total score is 10 points; the higher the score, the lower the risk of bias. 50
Certainty of Evidence
The GRADE (Recommendations Assessment, Development, and Evaluation) methodology was used to classify the certainty of the evidence for each outcome as high, moderate, low, or very low. 51 In the GRADE approach, RCTs start with high certainty and non-randomized trials with moderate certainty. We planned to assess publication bias by visually inspecting funnel plots for each outcome in which we identified 10 or more eligible studies; however, this was not possible because there was an insufficient number of studies to allow this assessment.
Data Synthesis and Statistical Analysis
A meta-analysis was performed by using similar studies with homogeneous outcomes. Standardized mean differences in combined inverse variance (SMD) and 95% confidence intervals (CIs) between groups were calculated using random-effects models. The variability in the results between the studies was analyzed using the I 2 statistic and P-value obtained from the Cochran chi-square test. To determine the mean difference between the groups (experimental—physical exercise and control – any comparison), the Z statistic was used, with P < .05 considered statistically significant. Review Manager (RevMan) (version 5.3; Nordic Cochrane Center, Cochrane) 52 was used for analysis.
Subgroup and Sensitivity Analysis
Possible subgroup analyses have been planned according to the following characteristics:
(i) Participants (stroke type: ischemic stroke vs. intracranial hemorrhage)
(ii) Interventions (type of robotic assistance: exoskeleton versus end-effector; frequency: high-frequency versus low-frequency training)
(iii) Comparator (type of control intervention: pharmacological therapy versus non-pharmacological therapy; associated use of noninvasive brain stimulation versus sham group).
Subgroup analyses were performed only when five or more studies were available, with at least two in each subgroup.
Results
A total of 630 studies were identified, and after reading the titles and abstracts, 23 studies were selected for eligibility analysis, 18 of which were excluded because they were prospective studies (n = 2), case series (n = 3), proof of concept (n = 1), did not use any type of robot (n = 4), did not include USN patients (n = 5), used robot without limb activation (n = 1), and were clinical trial protocols (n = 2). In total, two RCTs,53,54 two pilot RCTs,55,56 and one quasi-RCT 57 were included in the qualitative analysis. A flowchart of the review steps is shown in Figures 1 and 2.
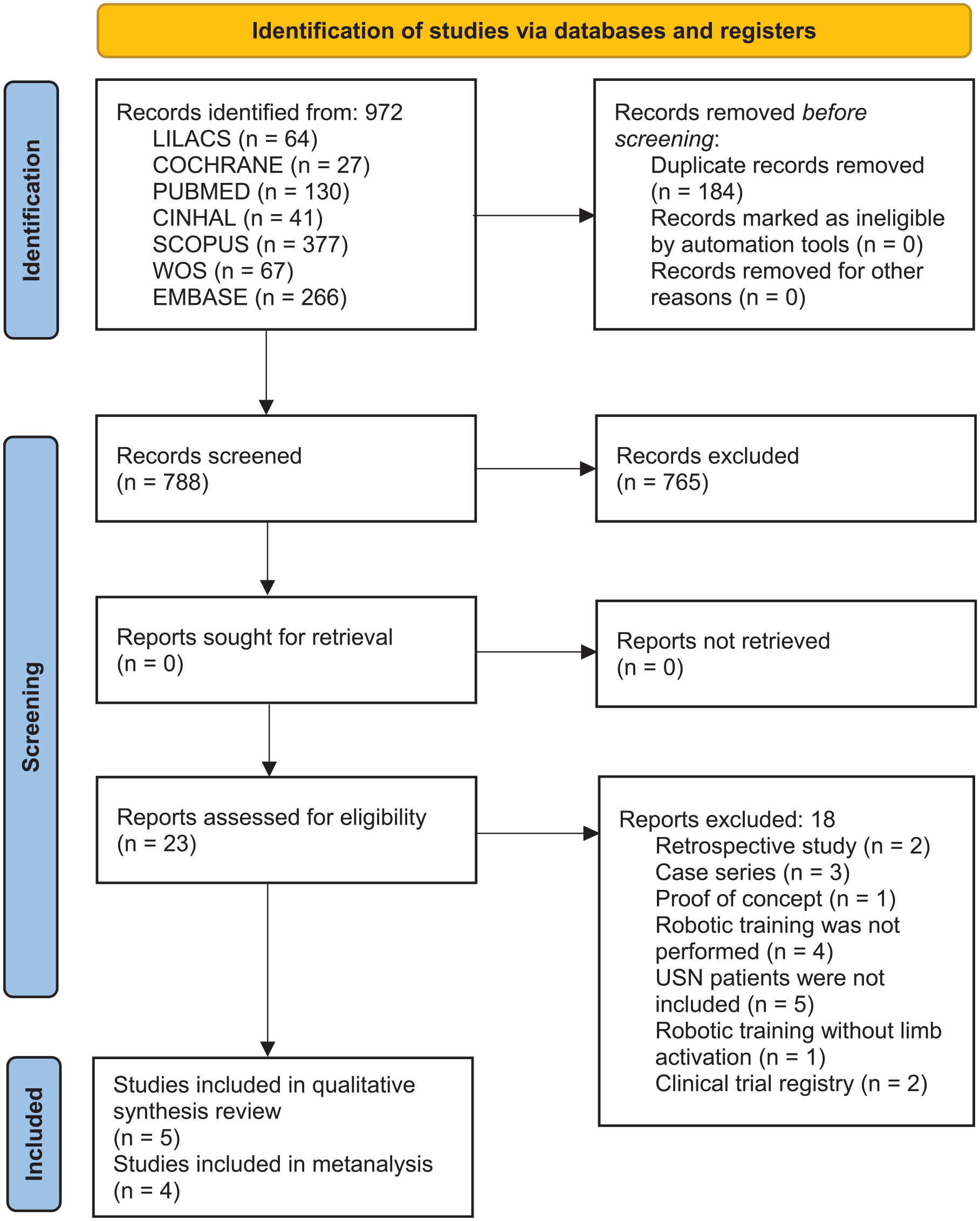
Flowchart of the review steps.
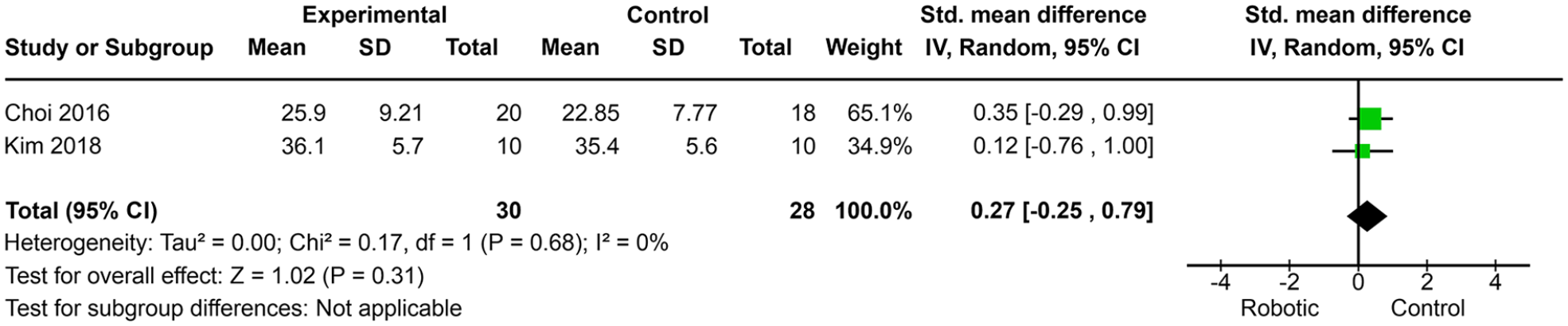
Meta-analysis of the unilateral spatial neglect degree measured by the motor-free visual perception test third edition.
The relevant characteristics of each study included in the qualitative analysis are presented in Tables 1 and 2. There were 73 and 69 participants in the intervention and control groups, respectively. The mean age of the individuals in the experimental and control groups was 60.65 (±8.88) years and 63.41 (±9.13) years, respectively. Three studies included patients in the subacute phase,53,54,56 one in the chronic phase, 55 and one in the acute phase 57 of stroke. In all studies, the individuals had ischemic and/or hemorrhagic strokes. The type of robotic therapy was the end-effector type in four studies53-55,57 and the exoskeleton type in one study. 56 In all studies, the outcome assessment was performed after the intervention protocol. No long-term follow-up was performed for any of the included studies.
Characteristics of Included Studies.
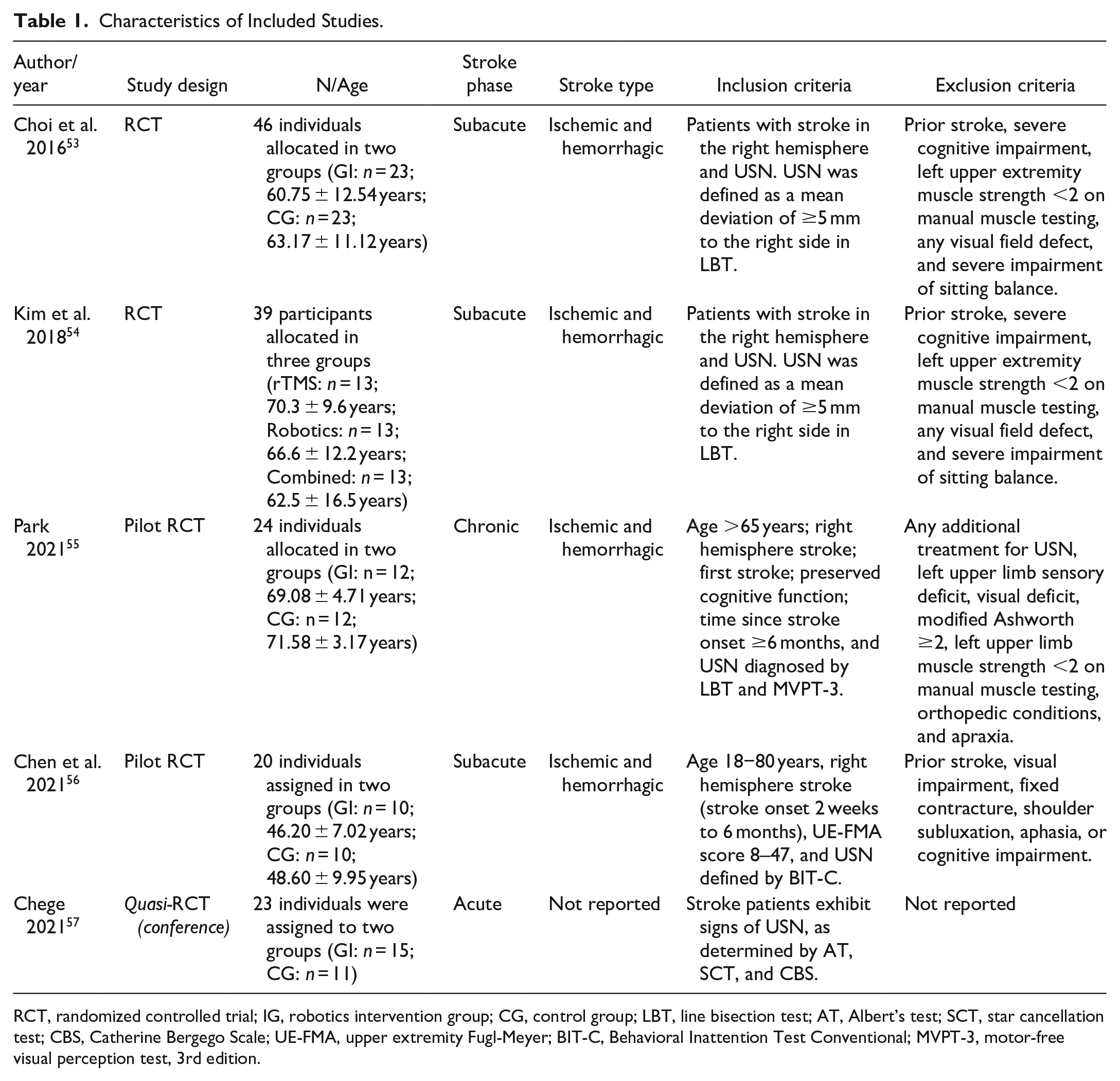
RCT, randomized controlled trial; IG, robotics intervention group; CG, control group; LBT, line bisection test; AT, Albert’s test; SCT, star cancellation test; CBS, Catherine Bergego Scale; UE-FMA, upper extremity Fugl-Meyer; BIT-C, Behavioral Inattention Test Conventional; MVPT-3, motor-free visual perception test, 3rd edition.
Outcomes and Treatment of Included Studies.
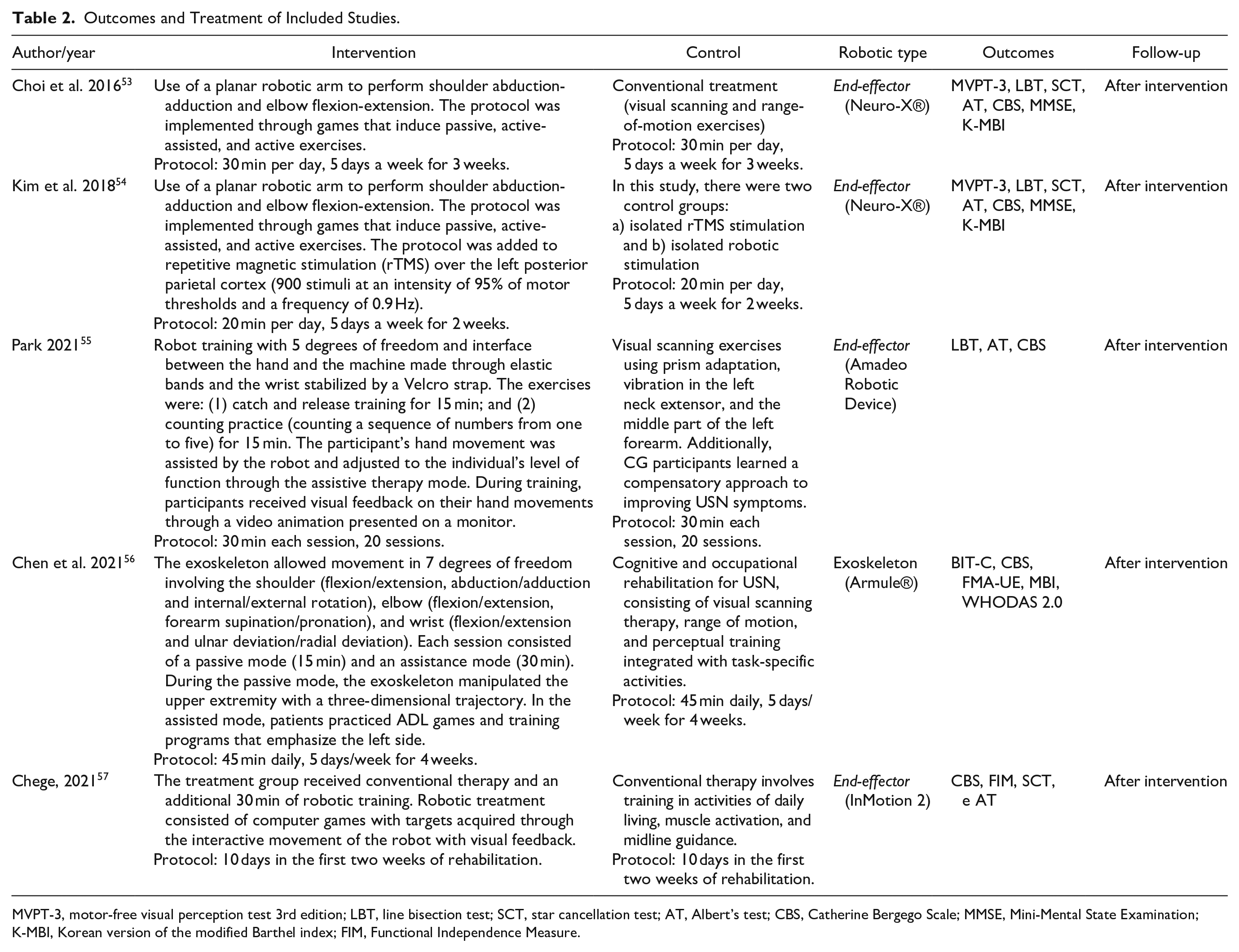
MVPT-3, motor-free visual perception test 3rd edition; LBT, line bisection test; SCT, star cancellation test; AT, Albert’s test; CBS, Catherine Bergego Scale; MMSE, Mini-Mental State Examination; K-MBI, Korean version of the modified Barthel index; FIM, Functional Independence Measure.
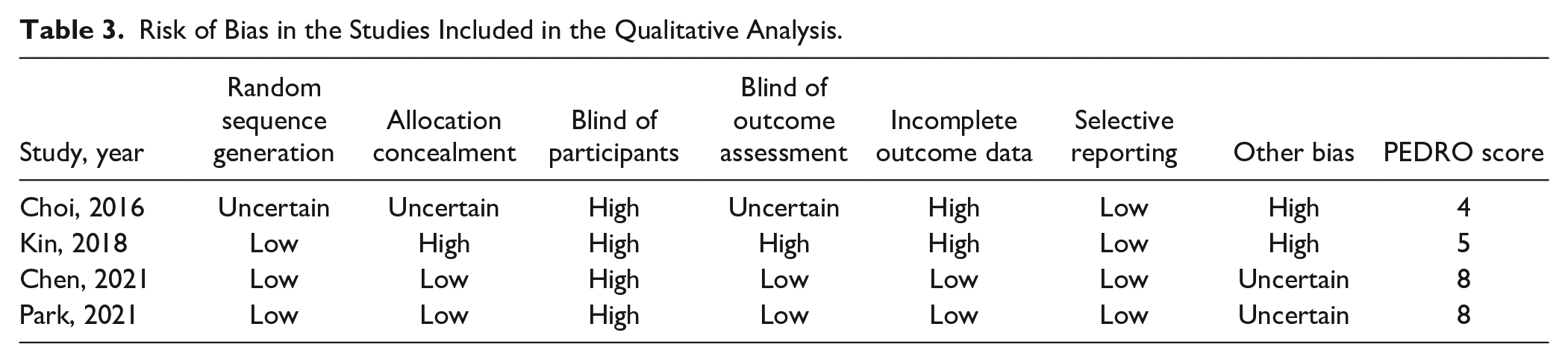
The risk of bias in the included studies is presented in Table 3. The PEDRO score was eight in two studies,55,56 five in one study, 54 and four in one study. 53 The detailed guidance of GRADE in the evidence profile is presented in Table 4.
Risk of Bias in the Studies Included in the Qualitative Analysis.
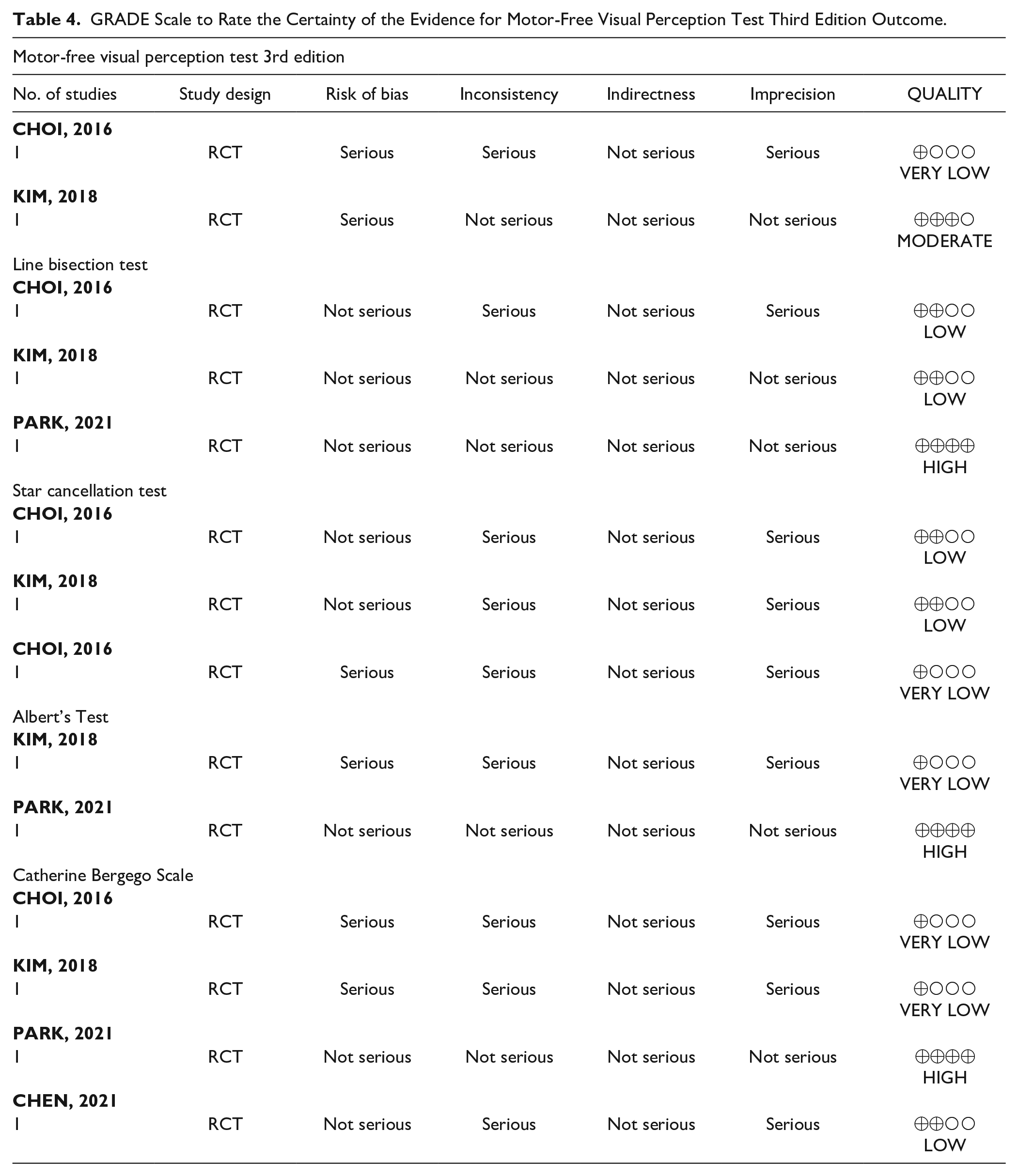
GRADE Scale to Rate the Certainty of the Evidence for Motor-Free Visual Perception Test Third Edition Outcome.
Four studies53-56 were included in the meta-analysis because of the presence of homogeneous outcomes. The study by Chege 57 was not reported in the meta-analysis because of a lack of data.
Motor-Free Visual Perception Test Third Edition (MVPT-3)
The results of the two RCTs (Choi et al. 53 and Kim et al. 54 ), comparing robotic limb activation with a control group, suggested that there was no improvement in the degree of USN measured by MVPT-3 (SMD, 0.27; 95% CI, −0.25−0.79; P = .31; I 2 = 0%) (Figure 3). The quality of this outcome was rated very low by one study 53 because of the high risk of bias in sample blinding and incomplete data, while the other study 54 was rated moderate quality due to the low risk of bias in the randomization of the allocation sequence, consistency, and accuracy in the data.
Meta-analysis of the unilateral spatial neglect degree measured by the line bisection test.
LBT
The results of the three RCTs (Choi et al 53 Kim et al, 54 and Park 55 ), comparing robotic limb activation with a control group, suggested an improvement in the degree of USN measured by the LBT (SMD, −0.64; 95% CI, −1.13 to −0.15; P = .01; I 2 = 92%) (Figure 3). The quality of this outcome was rated as low in one study 53 because of the high risk of bias in sample blinding and incomplete data. However, one study 54 was rated moderate quality because of the low risk of bias in sequence randomization, allocation, consistency, precision, and direct data, while another study 55 was rated high quality evidence because of the low risk of bias in blinding and randomization of the allocation sequence, blinding of evaluators, and incomplete data.
Star cancellation test (SCT)
The results of the two RCTs (Choi et al 53 and Kim et al 54 ), comparing robotic limb activation with a control group, suggested that there was no improvement in the degree of USN measured by SCT (SMD, 0.26; 95% CI, −0.42−0.94; P = .54; I 2 = 38%) (Figure 4). The quality of this outcome was rated as low in two studies53,54 because of the high risk of bias in sample and rater blinding, incomplete data, and allocation concealment.
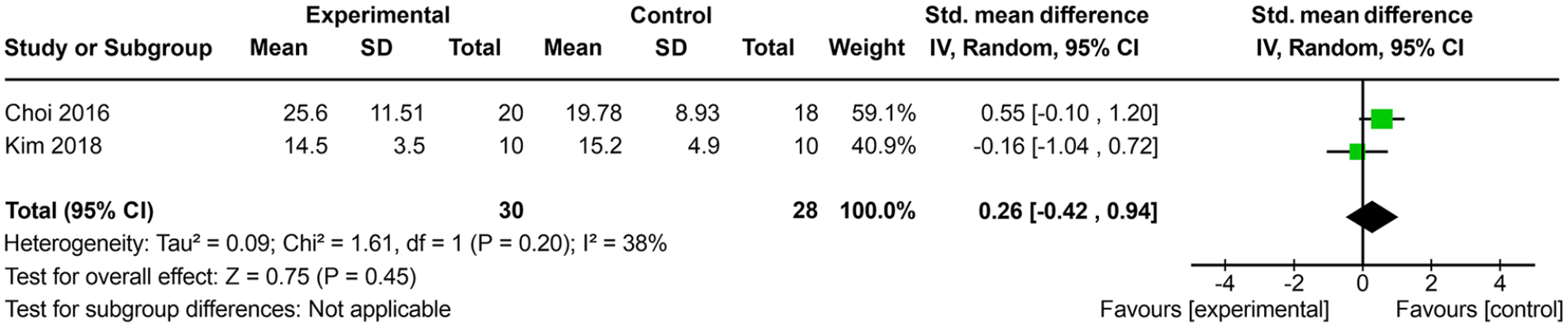
Meta-analysis of the unilateral spatial neglect degree measured by the star cancellation test.
Albert’s Test (AT)
The results of the three RCTs (Choi et al, 53 Kim et al, 54 and Park 55 ), comparing robotic limb activation with a control group, suggested that there was no improvement in the USN degree measured by AT (SMD, −0.67; 95% CI, −2.01−0.66; P = .32; I 2 = 87%) (Figure 5). The quality of evidence for this outcome was rated very low in two studies53,54 because of imprecision, inconsistency, and high risk of bias for patient blinding and incomplete data. However, one study 55 was rated high quality evidence because of a low risk of bias for allocation concealment and allocation sequence, incomplete data, and blinding of the rater.
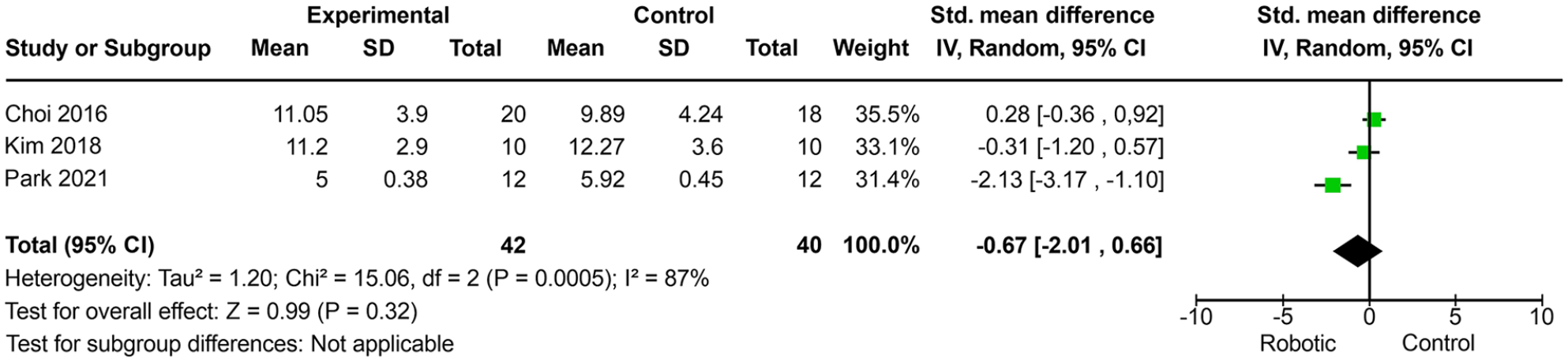
Meta-analysis of the unilateral spatial neglect degree measured by Albert’s test.
Catherine Bergego Scale (CBS)
The results of the four RCTs (Choi et al, 53 Kim et al, 54 Park, 55 and Chen et al. 56 ), comparing robotic limb activation with a control group, suggested that there was no improvement in the functional impact of USN as measured by CBS (SMD, −0.81; 95% CI, −2.07−0.45; P = .21; I 2 = 87%) (Figure 6). The quality of evidence for this outcome was rated high in one study 55 ; very low in two studies53,54 because of the high risk of bias in sequence blinding and allocation, sample and rater blinding, and incomplete data; and low in one study 56 because of the high risk of bias in sequence blinding and allocation and sample and rater blinding.
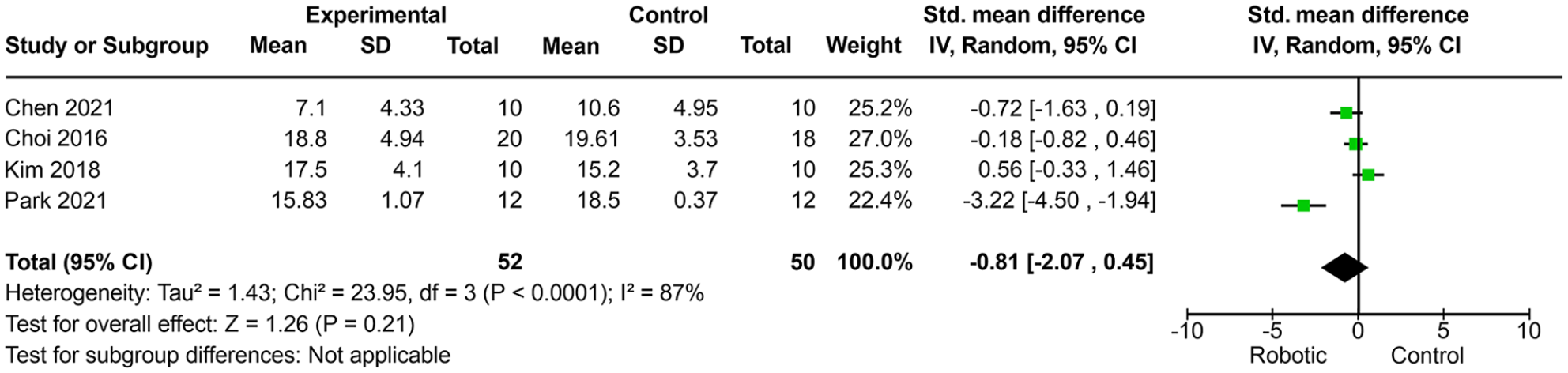
Meta-analysis of the functional impairment degree measured by the Catherine Bergego Scale.
Discussion
The main objective of this review is to investigate the effects of limb activation using a robotic device in individuals with USN following stroke. The results of this review suggest that robot-assisted training of the left hand may improve LBT-midline perception, indicating that robot-assisted limb activation training may be useful in improving USN symptoms in stroke patients. However, no impact was observed in the other visual-perceptual neglect tests (star cancellation test, motor-free visual perception test, and Albert’s test), showing that robotic therapy did not affect tasks assessing visual scanning. In addition, we did not observe positive effects on functionality and activities of daily living.
Previous studies have reported that passive and active movements in the left limb can reduce USN in stroke patients.23,30,55 The positive effects of robot-assisted training can be attributed to increased activation of the right hemisphere motor circuits and recruitment of the associated attention mechanism during the task, improving attention to the left side of space. 7 Takahashi et al observed that robot-assisted manual training increased the size of the representational map of the primary sensorimotor cortex. 26 In addition, continuous visual feedback during robotic training can activate participants’ extra-personal space when viewing the animation of their hand movements on the monitor.
USN is an attentional neuropsychological disorder characterized by the inability to explore, orient, or respond to the contralesional side of space. Another possible explanation for these findings is that robotic training focused on personal and peripersonal space, increasing performance on pencil and paper tests. 58 Furthermore, in all robot-assisted therapy intervention protocols, the therapist provided continuous feedback to participants to increase attention and explore the space contralateral to the brain injury, which can serve as a visual scanning training for visual search and exploring objects. 22 Another theory that can be added to explain the effects of robotic therapy in USN is that increased activation of the right hemisphere by activation of the left upper limb increases inhibition of the ipsilesional hemisphere and reduces hemispheric inhibition via the corpus callosum to the affected hemisphere. 59 In addition, passive movements for multiple joints with the exoskeleton may play a role in limb activation therapy, arousing contralateral attention. 60
In this review, we observed that only one study (Park et al) 55 had a significant effect on line bisection improvement. In contrast to Choi et al 53 and Kim et al, 54 Park et al 55 investigated the transfer effects of robot-assisted limb activation on a participant’s daily function. The study by Park et al. used a different robot training system than other studies, and the exercises were focused on grasping and release training with visual feedback of their hand movements via video animation. 55 Choi et al and Kim et al used a robot treatment program through games that induced passive and active assistive exercises of the upper limb in general.53,54 In addition, the study by Park et al 55 had a higher frequency and number of total sessions than other studies.53,54 The higher frequency of stimulation and type of exercise may have been favorable for improving the outcome associated with midline perception.
The lack of a significant effect of robotic therapy in the other visual-perceptual neglect tests (star cancellation test, motor-free visual perception test, Albert’s test) showed that robotic therapy did not affect tasks assessing visual scanning. Line bisection and cancellation tasks have different components of neglect syndrome. Line bisection necessitates the correct perception of the size of a single stimulus (“midline perception”), and cancellation tasks depend on the ocular exploration of a display containing many visual stimuli that have to be scanned systematically from left to right. 61
The line bisection test is a screening test that is valid and reliable 35 ; however, the minimal clinically important difference (MCID) of this test as a tool to measure responsiveness to intervention is not known. As only improvement in midline perception by LBT was observed in this review, we should be cautious in stating that robotic therapy can improve the neglect syndrome, and the use of the gold standard test with known responsiveness should be encouraged in other clinical trials.
In all studies, there was another potential stimulus in addition to robotic assistance, directing attention to the affected hand. Choi et al 53 used game programs to prompt participants’ attention through sound. Kim et al 54 added repetitive magnetic stimulation (rTMS) to the left posterior parietal cortex during robotic training and auditory feedback. Park 55 performed training with visual feedback on hand movements via video animation. Chen et al 56 used three-dimensional animation and voice prompt from the exoskeleton. Potentially, these may be critical active ingredients as the passive-active movements induced by the robot because visual and auditory feedback can improve perception during upper limb exercises.
Robot-assisted therapy can improve the degree of USN at the level of structure and function (midline perception); however, in the analyzed studies, we did not observe any impact on functionality at the level of activity and participation. Chen 56 was the first to investigate the effects of exoskeleton-driven robotic training according to the International Classification of Functioning, Disability, and Health (ICF) framework. The authors noted that robotic training using an exoskeleton was feasible and safe for patients with USN after stroke. The results demonstrated that RAT could be effective for USN symptoms, arm motor impairment, and social participation, except for the performance of daily living, compared with the control group.
Although this study provided evidence to support the therapeutic concept of robot-assisted therapy in USN after stroke, there are still limitations to this review. First, the relatively small sample size of the included studies prevented the generalization of the training effects and reduced the effect size. Second, the long-term effects of robot-assisted limb activation were not confirmed by the included studies, as none of them performed follow-ups. Furthermore, the inclusion criteria for some studies were not multidimensional and did not use the gold standard test (BIT-conventional) for objective diagnosis of USN after stroke.
Therefore, future studies should provide robotic limb activation interventions in the acute and subacute stroke phases, with larger sample size, using exercises oriented to specific tasks of the patient’s daily life, and using more sensitive tests to include patients in future clinical trials.
Conclusion
An improvement in degree of USN (“midline perception”) was observed after robot-assisted limb activation therapy in stroke patients; however, there was no impact on tasks assessing visual scanning, functionality, and activities of daily living. The quality of the evidence from the available RCTs for statistically significant outcomes was high in one study and low in two studies. The applicability of these findings may be compromised, as most of the results described in this review have been obtained from studies with very small sample sizes. However, this review provides preliminary support for the use of upper limb activation by robotics in future clinical trials for USN.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221110894 – Supplemental material for Effect of Robot-Assisted Training on Unilateral Spatial Neglect After Stroke: Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-nnr-10.1177_15459683221110894 for Effect of Robot-Assisted Training on Unilateral Spatial Neglect After Stroke: Systematic Review and Meta-Analysis of Randomized Controlled Trials by Rodrigo Bazan, Bruno Henrique de Souza Fonseca, Jessica Mariana de Aquino Miranda, Hélio Rubens de Carvalho Nunes, Silméia Garcia Zanati Bazan and Gustavo José Luvizutto in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
