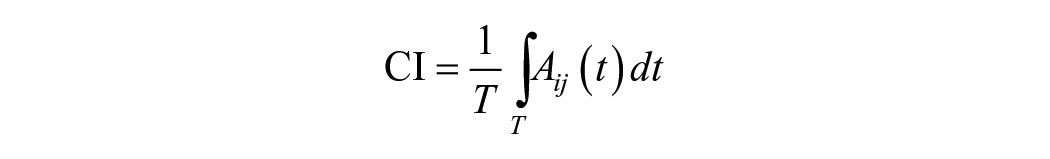Abstract
Background
Intensive task-oriented training has shown promise in enhancing distal motor function among patients with chronic stroke. A personalized electromyography (EMG)-driven soft robotic hand was developed to assist task-oriented object-manipulation training effectively. Objective. To compare the effectiveness of task-oriented training using the EMG-driven soft robotic hand.
Methods
A single-blinded, randomized controlled trial was conducted with 34 chronic stroke survivors. The subjects were randomly assigned to the Hand Task (HT) group (n = 17) or the control (CON) group (n = 17). The HT group received 45 minutes of task-oriented training by manipulating small objects with the robotic hand for 20 sessions, while the CON group received 45 minutes of hand-functional exercises without objects using the same robot. Fugl-Meyer assessment (FMA-UE), Action Research Arm Test (ARAT), Modified Ashworth Score (MAS), Box and Block test (BBT), Maximum Grip Strength, and active range of motion (AROM) of fingers were assessed at baseline, after intervention, and 3 months follow-up. The muscle co-contraction index (CI) was analyzed to evaluate the session-by-session variation of upper limb EMG patterns.
Results
The HT group showed more significant improvement in FMA-UE (wrist/hand, shoulder/elbow) compared to the CON group (P < .05). At 3-month follow-up, the HT group demonstrated significant improvements in FMA-UE, ARAT, BBT, MAS (finger), and AROMs (P < .05). The HT group exhibited a more significant decrease in muscle co-contractions compared to the CON group (P < .05).
Conclusions
EMG-driven task-oriented training with the personalized soft robotic hand was a practical approach to improving motor function and muscle coordination.
Clinical Trial Registry Name:
Soft Robotic Hand System for Stroke Rehabilitation.
Clinical Trial Registration-URL:
Unique Identifier:
NCT03286309.
Keywords
Introduction
Motor deficits, particularly hemiparesis, are a common symptom among stroke survivors. 1 These motor impairments significantly impact a stroke patient’s ability to perform activities of daily living (ADL), ultimately affecting their overall quality of life. 2 Upper extremity (UE) motor performance deficits are prevalent among stroke survivors, with approximately 78% experiencing post-stroke deficits and around 50% specifically facing challenges with hand function for ADLs.3,4 Compensatory movements are commonly observed during UE movements, manifesting as proximal compensation for distal joint movements. 5 Significant improvements in UE motor function typically occur within the first 6 months following stroke onset. However, approximately 65% of stroke survivors report difficulty using their affected hand in the chronic stage. 3 Therefore, chronic stroke survivors urgently need to regain UE functions, mainly hand function, for independence in daily life.
Previous studies have demonstrated that task-oriented training can significantly benefit motor functions of UE, even in chronic stroke survivors. 2 However, due to resource constraints and limited training duration, therapists often prioritize spontaneous motor recovery after stroke and start with proximal joints, while distal joints, especially the fingers, are given less attention. 6 Constraint-induced movement therapy (CIMT) was widely used to address this issue and promote coordinated movement of proximal and distal joints in the UE of stroke survivors. However, only 10% of stroke patients can benefit from this therapy 7 because it is difficult for the patient to maintain sufficient hand mobility, especially extension of the fingers. Stroke survivors with limited movement often exhibit compensation patterns and movement dysfunctions, as the distal joints have a slower recovery rate and are initially weaker. 8 These can lead to the phenomenon of “learned non-use” of the hand in daily activities, and the muscles corresponding to the distal part are particularly challenging to rehabilitate, resulting in limited overall motor function recovery post-stroke. 9
In recent years, wearable robotic hands have gained significant attention as rehabilitation modalities in hospitals and rehabilitation centers. Those robotic hands can assist stroke patients in repetitive hand functional exercises, that is, hand opening and grasping.10,11 Studies have shown that rehabilitation using wearable robotic hands or gloves actively triggered by surface electromyography signals can increase motor performance and a synergistic pattern of abnormal muscle tone.12-14 However, these devices were usually made of metal, and their heavy weight often makes it difficult for patients to lift their paralyzed arms to perform more functional tasks. In previous studies, the paretic arm of the patients was fixed on a table, which allowed the patients to focus on the repetitive exercise of hand opening and grasping. A significant improvement in motor function of the UE could be observed after the 20-session intervention. However, the distal joints have not shown as great a response to the training as the proximal joints in those studies. One limitation of these robotic hands is that they do not fit the finger lengths of each stroke survivor well. For example, female patients may have much shorter fingers than male patients, and their palms may be narrower.
Soft rehabilitation robots were developed to overcome these challenges due to their high compliance and personalization, which offer biomimetic properties and are well-suited for treating ADL. 15 The design of soft robots encourages voluntary coordination of finger joints, allowing stroke patients to perform a wide range of gestures naturally. This design approach has shown promise in improving motor functions in rehabilitation.2,16 Previously, we have developed an electromyography (EMG)-driven soft robotic hand for upper limb rehabilitation, building upon our previous work on bi-directional 3D-printed soft pneumatic actuators.17,18 The soft robotic hand based on this actuator has the potential to be personalized for each patient to fit their hand during rehabilitation.
An accessor-blinded randomized controlled study using the EMG-driven personalized soft robotic hand was conducted to investigate the effects of task-oriented object manipulation training on UE motor function. The repetitive hand opening and grasping exercises were compared in patients with moderate to severe hemiparesis.
M ethod
Study Design
This study was an assessor-blinded randomized controlled trial, and it was registered on ClnicalTrials.gov (ID: NCT03286309). All study protocols adhered to the principles of the Declaration of Helsinki. The Joint Chinese University of Hong Kong-New Territories East Cluster granted ethical approval for the study.
Randomization
Randomization was conducted using a computer-generated randomization sequence. A dedicated researcher, independent of participant recruitment and assessment, performed the randomization process. Each participant was assigned a unique identification number, and a random number generator was used to allocate them (1:1) to either the Hand Task (HT) group or the control (CON) group.
Participants and Sample Size
Participants were recruited for the study from local rehabilitation centers and communities in Hong Kong. The following inclusion criteria were applied: (a) in chronic stage (6 months after the onset of stroke) with a pure unilateral motor paresis after a stroke; (b) sufficient cognition to follow simple instructions and understand the content and purpose of the study; (c) able to sit up for at least 45 minutes; (d) detectable residual EMG signals from the affected side’s flexor digitorum (FD) and extensor digitorum (ED) muscles; and (e) a Modified Ashworth Scale (MAS) score of finger extensors less than or equal to 3.
Exclusion criteria comprised: (a) Patients with severe dysphasia with inadequate communication; (b) any additional medical or psychological condition affecting their ability to comply with the study protocol; (c) history of other neurological disease psychiatric disorders, including alcoholism and substance abuse. Before the start of the experiments, written informed consent was obtained from each participant, following the protocol approved by the local institutional review board.
The sample size for each group was determined based on the anticipated change in the total Fugl-Meyer motor assessment for UE (FMA-UE) after the training intervention. FMA-UE was selected as a reliable and widely utilized measure for monitoring voluntary motor improvement in post-stroke rehabilitation. 19 Our previous research on EMG-driven basic functional training in patients with chronic stroke demonstrated an expected improvement of 3.31 ± 3.79 points in FMA-UE post-intervention (N = 16). 20 The improvement in FMA-UE scores, specifically in task-based training, was estimated to be approximately 7 points, with a similar sample size per group (N = 17). 21 Thus, if the clinically significant difference in improvement between groups in this study is about 3.69 points, similar results should be obtained. A power calculation with a (P = .05 and a power of .8 indicated a minimum of 17 subjects per group (34 subjects in total).
The Personalized Soft Robotic Hand
The personalized EMG-driven soft robotic hand used in this study was initially designed by our group,20,22 with further enhancement to meet the demand of this study by personalized hand size for each stroke survivor. Each soft finger actuator has 2 segments, and the thumb actuator has only 1, as shown in Figure 1B. Before the intervention, the finger length, joint position, and palm width were measured as the reference for fabricating the robotic hand. Participants were asked to place their paretic hands on the table and to measure the hand size. Rectangles for the actuator to attach to the finger segments were created (Figure 1A) with the metacarpophalangeal joint (MCP) and the proximal interphalangeal joint (PIP) of the fingers as the center, and it served as the design dimension for the corresponding joint actuators. For the thumb, only a rectangle covering the MCP joint was considered. Since the distal interphalangeal joint (DIP) contributes only 15% to functional grip, 23 the actuator did not cover the DIP joint and facilitate sensory feedback of the fingertip to correct motor movement during rehabilitation. Then, the finger actuators (ACEO Silicone GP Shore A 30, WACKER Chemie AG, Germany) and palm brace (ABS Plastic) were 3D printed based on the measured parameters. A thin elastic plate was inserted into each actuator to provide enough extension torque for stroke patients to provide extension torque to open their spastic hands. The thickness of this plate was adjusted to provide sufficient torque to fully extend their finger.18,20 Participants can easily wear the robotic hand using the adjustable-length Velcro straps. All electronic components were integrated into a CON box with a touchscreen for user-friendly operations (Figure 1D).
Personalized EMG-Driven soft robotic hand and training setup. (A) Hand measurements for participants, the length of each segment (d), palm width (l), and Velcro position were obtained for customization. (B) Prototype of the 3D-printed soft robotic hand. (C) Flexion and extension of the index finger (MCP and PIP joints) on pneumatic actuation. (D) Overview of the task-oriented training setup. Four electrodes were used to measure EMG signals from FD, ED, BIC, and TRI muscles. The participant was lifting a ball with the assistance mode of the spherical grasp. (E) The participant was lifting a small cube with the assistance mode of tip pinch. (F) The participant was lifting a woodblock with the assistance mode of the tripod pinch.
For this system, a probabilistic model-based learning CON approach was used to detect the intentions of stroke patients when performing different tasks. 18 The soft robotic hand has been programmed to manipulate 3 types of real-life small objects in training, as shown in Figure 1: (a) spherical grasp involving all 5 fingers (Cricket ball, 109 g, 75 mm diameter); (b) tripod pinch utilizing index finger, middle finger, and thumb (Woodblock, 74 g, 50 mm edge length); and (c) tip pinch involving index finger and thumb (Small cube, 8 g, 25 mm edge length). Surface electromyography (EMG) signals were collected from the paretic FD and ED muscles. Before each intervention session, participants underwent an EMG evaluation trial to measure their maximum voluntary contractions. The baseline EMG threshold was determined by setting it 3 times the standard deviation (SD) above the mean value, as suggested by Hu et al. 12 Different grasping movements were activated when the EMG signal from the FD muscle exceeded the default threshold of 10%, while hand opening was triggered when the EMG signal from the ED muscle crossed the same threshold. In addition to the FD and ED muscles, EMG activities from the biceps brachii (BIC) and triceps brachii (TRI) muscles were also recorded during the EMG evaluation trial.
Intervention
In this study, 20 training sessions were provided to all participants in the HT and the CON groups. The training was completed in 7 weeks. Sessions ranged from 2 to 4 days per week, depending on the subjects’ schedules. We tried to maintain an average of around 3 days per week. Each session lasted approximately 60 minutes. The session started with 5 minutes of preparation and warm-up exercises. Subsequently, there were three 15-minute blocks for robot-assisted EMG-driven training. Two 5-minute rests separated each training block. For the HT group, real-life objects were used, and specific functional task goals were set for each trial (Figure 1D-F). The participants were instructed to perform the following tasks during each trial: (a) grasp objects with specific gestures, (b) lift objects (participants were allowed to use their unaffected hand for assistance if necessary), (c) hold and move objects to a specified location based on instructions from the therapists, and (d) open the hand and release the objects. The soft robotic hand assisted the participants in performing the tasks using specific hand gesture modes in each task. Each task was trained with around 30 to 40 repetitions in 15 minutes. Throughout the training, the operator provided verbal instructions emphasizing the importance of using the correct fingers and maintaining a firm grip on the objects. Participants in the CON group were engaged in repetitive hand opening and grasping exercises with the assistance of the same robotic hand as the HT group.
Outcome Measures
Clinical Assessments
Motor function of UE, performance of the paretic hand, and spasticity were assessed at baseline, post-intervention, and 3-month follow-up after intervention. The post-intervention assessments were conducted within 3 days after the subjects finished the last training session. FMA-UE (score range: 0-66) was measured, and the 3 months follow-up was the primary outcome. The 2 subparts of FMA-UE, shoulder/elbow (S/E, score range: 0-36) and wrist/hand (W/H, score range: 0-24) were also evaluated separately. Performance of the paretic hand was also assessed by the Action Research Arm Test (ARAT, score range: 0-57), 24 Box and Block Test (BBT), 25 Maximum Grip Strength (MGS), and Active Range of Motion (AROM) of index finger and thumb. Spasticity was measured with the MAS of the finger, wrist, and elbow. 26 Two assessors who were blinded to the group allocation or any treatment-related information conducted the outcome assessments. Before the assessment phase, the assessors were provided with detailed instructions and training on the standardized administration of the outcome measures to ensure consistent data collection.
Electromyography Parameters
EMG signals were collected from 4 muscles: flexor digitorum (FD) and ED, BIC, and triceps brachii (TRI) muscles. The co-contraction index (CI) was used to assess the coordination pattern of muscle contractions during the designed UE joint movements. This study evaluated 4 actions to determine muscle contraction coordination: (1) finger/wrist flexion, (2) finger/wrist extension, (3) elbow flexion, and (4) elbow extension. CI was defined as quantifying the overlap period of the linear envelope of the EMG for each pair of agonist-antagonist muscles. The equation for the CI calculation was derived from the work by Frost et al. 27 :
where
Statistical Analysis
Statistical analysis of clinical evaluated s was performed with IBM SPSS 26 (IBM Corp., Armonk, NY, USA). Demographic characteristics of the HT and CON groups were examined to assess the quality of randomization. The Shapiro–Wilk test was used to evaluate the normality of each measure. For measures that conformed with normality, an independent t-test was performed. For non-normal measures, the Mann–Whitney U test was applied. χ2 tests were used for dichotomous measures.
To analyze the result, 2-way repeat measures ANOVA (mixed design, within-subject factor: time [3 levels]; between-subject factor: group [2 levels]) was used to compare the effect between HT and CON treatments. For outcomes with significant Group × Time interaction effect identified (ie, P < .05), post-hoc analysis was performed by comparing outcomes in different time levels within groups by paired t-tests. Changes relative to baseline were then compared across groups using an independent t-test. All statistics are Bonferroni-corrected for multiple comparisons with a significance level of P < .05. According to the method suggested by Kaji et al, 28 a rating of 1+ on the MAS score was assigned a value of 1.5. The mean changes in FMA-UE, ARAT, BBT, and MGS were compared with their estimated minimal clinically important difference (MCID) values.
A CI and its post-hoc analysis were performed from statistical packages lme4 29 and emmean 30 using R (version 4.3.2). Specifically, the CI was modeled as the response variable and fitted with a linear mixed model consisting of a full factorial of 3 fixed-effect variables: Group [2 levels], Muscle pair [6 levels], and Time [20 sessions]. Each subject was modeled as a random effect. Analysis of the fixed effects variance was performed to identify the main and interaction effects, with the degree of freedom estimated by Satterthwaite’s method. In the post-hoc analysis, the estimated marginal regression slope (β), which represents the change of CI along training sessions, was compared across groups within each muscle pair. Slopes β were compared across the group in each muscle pair by independent t-test. P-values were Bonferroni corrected for multiple comparisons.
Results
The first participant was enrolled in October 2020. A total of 34 participants met the inclusion criteria and were randomly assigned to the HT group (n = 17) or the CON group (n = 17). The enrolled participants completed all outcome assessments. The last participants completed the outcome assessments in March 2022. Four subjects with high spasticity of finger joints (MAS = 3) were evenly distributed into 2 groups. None of the participants reported any adverse incidents related to the intervention in either the HT or CON groups. Additionally, no discomfort was reported by the recruited subjects.
Table 1 describes the demographic and clinical characteristics of the participants after randomization. The main FMA-UE score of the total participant sample was 25.8, indicating a moderate to severe impairment in motor function of UE. 31 The average score of ARAT (17.7) and BBT (2.6) also revealed that the participants suffered from a motor deficit of UE and lost hand dexterity. There were no significant between-group differences in demographic characteristics and clinical outcomes.
Demographic and Clinical Characteristics of Participants.
Abbreviations: FMA-UE, upper-extremity subscale of Fugl-Meyer Assessment; W/H, wrist and hand; S/E, shoulder and elbow; ARAT, Action Research Arm Test; BBT, Box and Block Test; MAS, Modified Ashworth Scale; MGS, maximum grip strength; AROM, active range of motion.
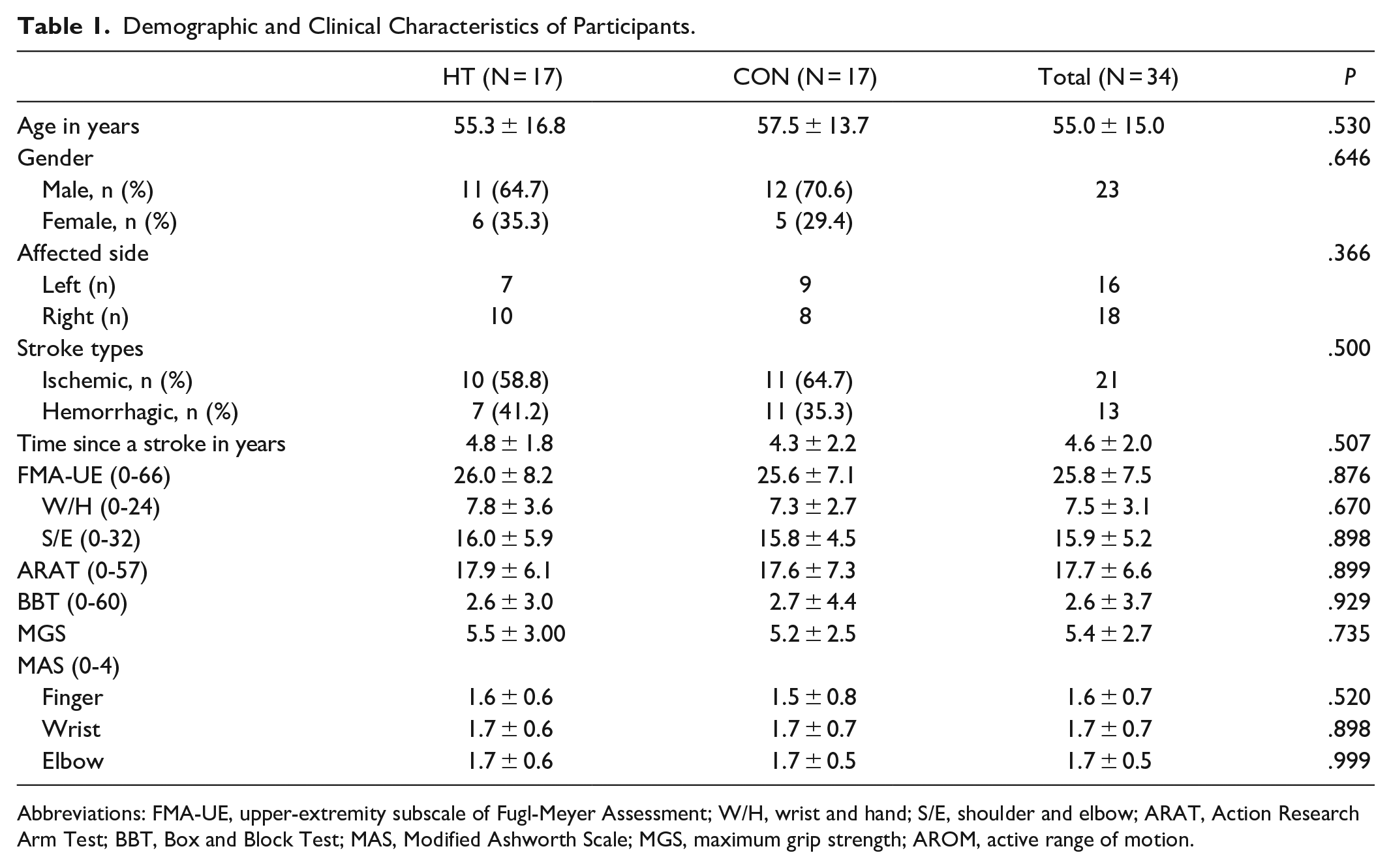
A summary of the analysis for all the clinical outcomes is provided in Table 2. Significant Time effect and Group ×Time interaction (P < .001) were observed for the FMA-UE (both W/E and S/E), ARAT (all the sub-scores), BBT, MAS for finger, MGS, AROM of PIP-I, and MCP-I. Post-hoc analysis of these outcomes is summarized in Table 3. Particularly, all outcomes improved significantly immediately after and 3 months post-intervention (P < .001, Bonferroni corrected). Between-group comparisons identified that the improvement of clinical scores within HT group is significantly higher than the CON group (P < .05) in all outcomes immediately after and 3-month post-intervention, except for MAS of wrist and elbow, which did not demonstrate significant improvement after training in both groups. Within the HT group, all outcomes except MAS of wrist and elbow increases significantly post-intervention and persists over 3 months. On the contrary, clinical scores within the CON group, including ARAT-grasp, BBT, MGS, AROM of PIP-I, and MCP-I decreased in 3-month follow-up session and showed no significant change compared with the baseline. Additionally, ARAT sub-scores (grip and pinch), BBT, and MAS of finger did not improve after intervention in the CON group.
Analysis of Clinical Outcomes at Baseline, Post-Intervention, and Follow-up a : Time Effect and Time × Group Interaction.
Abbreviations: FMA-UE, upper-extremity subscale of Fugl-Meyer Assessment; W/H, wrist and hand; S/E, shoulder and elbow; ARAT, Action Research Arm Test; Grossmt, gross movement subscale of ARAT; BBT, Box and Block Test; MAS, Modified Ashworth Scale; MGS, maximum grip strength; AROM, active range of motion; PIP-I, PIP joint of index finger; MCP-I, MCP joints of index finger; MCP-T, MCP joints of thumb.
Values are presented as mean ±SD. P-values are the results of 2-way repeated measure ANOVA of the outcomes, with a significance level of P < .05.
Outcomes that demonstrated significant change relative to baseline, using paired t-test (P < 0.05, Bonferroni corrected).
Change Scores Compared with Baseline. a
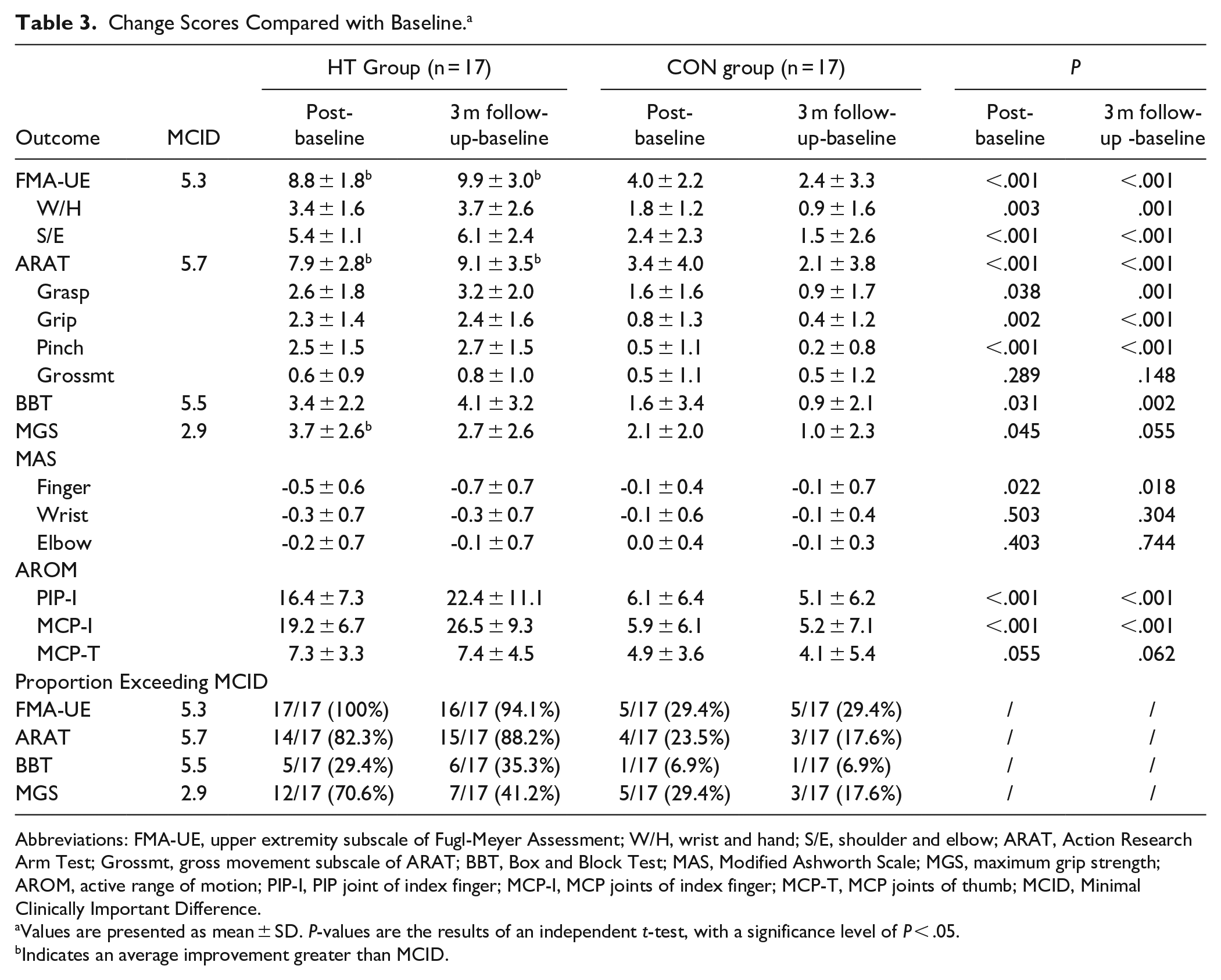
Abbreviations: FMA-UE, upper extremity subscale of Fugl-Meyer Assessment; W/H, wrist and hand; S/E, shoulder and elbow; ARAT, Action Research Arm Test; Grossmt, gross movement subscale of ARAT; BBT, Box and Block Test; MAS, Modified Ashworth Scale; MGS, maximum grip strength; AROM, active range of motion; PIP-I, PIP joint of index finger; MCP-I, MCP joints of index finger; MCP-T, MCP joints of thumb; MCID, Minimal Clinically Important Difference.
Values are presented as mean ± SD. P-values are the results of an independent t-test, with a significance level of P < .05.
Indicates an average improvement greater than MCID.
Table 3 compares the MCID values with the mean score changes in ARAT, FMA-UE, BBT, and MGS for both groups. MCID references were suggested in previous studies.32-34 In the HT group, the average FMAUE, ARAT, and MGS improvements exceeded the MCID values after the intervention. All participants in the HT group exceeded the MCID for FMA-UE scores after the interventions, and this effect persisted at 3-month follow-up (91.4%). None of the CON group’s clinical assessments exceeded the MCID at any given point. Only 29.4% of participants in the CON group exceeded the MCID at 3-month follow-up. Among patients with high finger joint spasticity (MAS = 3), the findings revealed that the HT group exhibited more significant improvement than the CON group. Specifically, 2 subjects with level 3 MAS in the HT group demonstrated a clinically meaningful improvement in MCID, whereas no significant improvements were observed in the CON group. Figure 3 compares CI values between the HT and CON groups. The linear mixed model revealed a significant main effect of time and muscle pair (P < .05) but no group effect. Significant interaction effects were observed between Group and Session, Group and Muscle, and Session and Muscle (P < .05). Marginal 3-way interactions were found between Group, Time, and Session (P = .054). The regression slope of each Group was obtained for each muscle pair to examine the interaction effect. The estimates and 95% CI are presented in Figure 3. Slopes with significant differences between groups (P < .05) were marked by “*” (Figure 2).
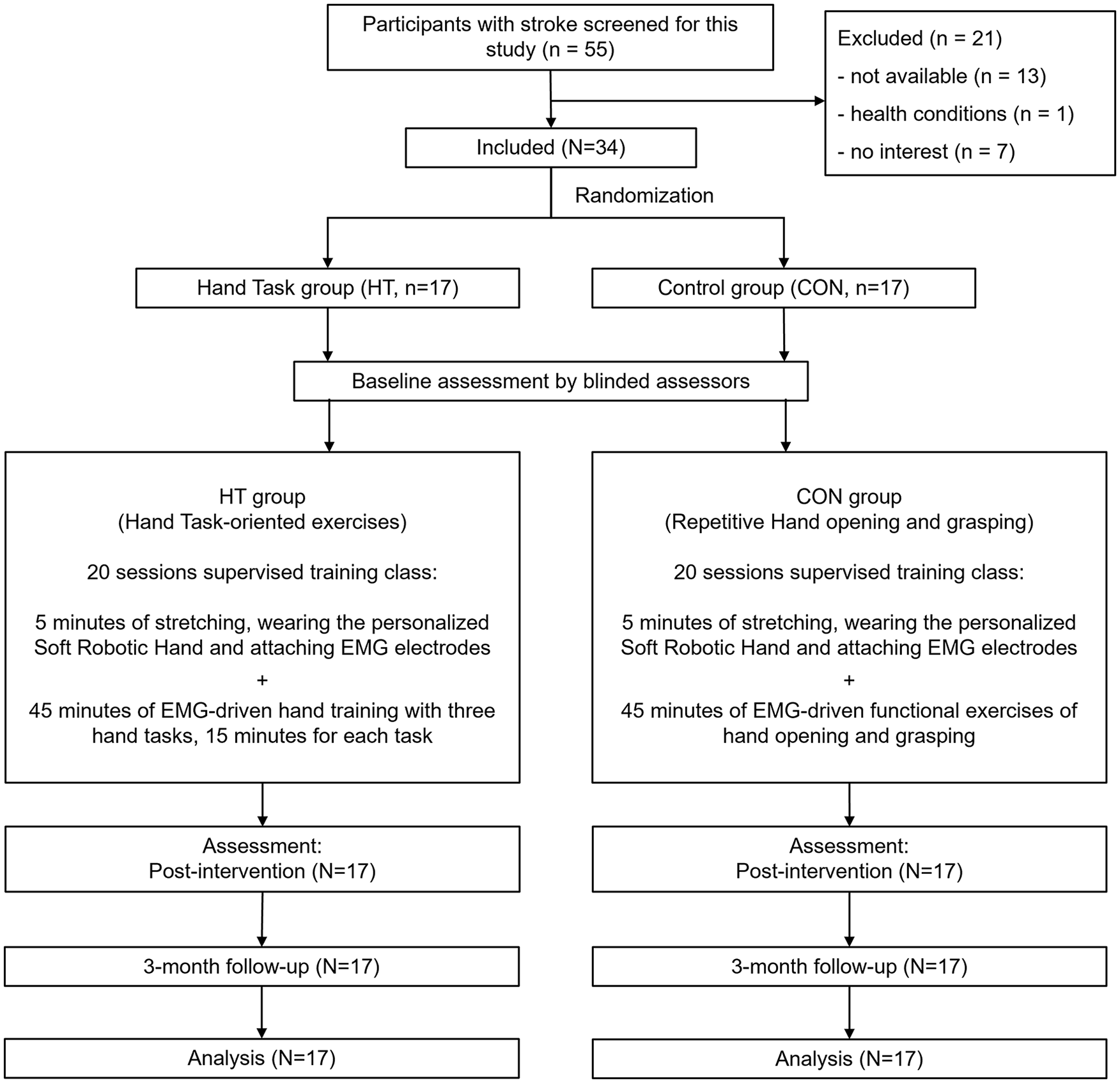
CONSORT study flow diagram.
Discussion
Motor Outcomes Evaluated by Clinical Scores
Significant improvements in FMA-UE were observed after the 20-session intervention and at the 3-month follow-up in the HT group. It is suggested that the functional improvement seen in the HT group can be successfully translated into increased ADL ability, allowing participants to incorporate and use their impaired arm more effectively in their daily lives. Furthermore, the FMA-UE (S/E and W/H) in the HT group was significantly higher than that of the CON group. The ARAT, BBT, finger MAS, and AROMs in the HT group also achieved more significant improvement than the CON group, which further demonstrated the effectiveness of task-oriented training in UE motor function, dexterity, and coordination.
In the HT group, stable mapping between different grasping tasks and objects of various sizes was established through intensive stimulation, which brought better functional progress than the CON group with a single exercise. Timmermans et al 2 highlighted that functional movements, which are actions involved in task execution, may only sometimes be explicitly directed toward distinct ADLs. Combined with the repetitive motion stimulation provided by the personalized robotic hand in his study, these converging stimuli may induce the Hebbian effect in the corticospinal pathway, which could underlie the observed motor relearning and functional improvement of paretic fingers. 35 The highly repetitive and intensive nature of the functional movements made the training goals more “specific.” Thielbar et al 36 conducted a study using a voice and EMG-driven actuated glove for ADL training, showing no significant improvement in FMA-UE scores after the intervention. One possible explanation for this discrepancy is that ADL training often involves modifying tasks based on performance during training, which may need to be more related to learned motor actions. Task-oriented training should focus on the advantages of robot training, establishing specific hand function task training by coordinating different fingers with robot assistance through highly repetitive exercises.
In upper limb rehabilitation approaches like CIMT, recovering distal joints poses more challenges to proximal joints. In this study, the customized design of the robotic hand enables participants to maintain a natural hand movement pattern with the assistance of robots, thereby facilitating practical exercises of the distal joints without the abnormal compensation from the proximal. The continuous stimulation originating from the distal joints can serve as positive reinforcement throughout the training process and makes the patients focus solely on task execution. Real-life daily object tasks maximize patients’ motivation and lead them to maintain a high level of concentration and energy during the training, thereby directly revealing any lack of focus or attempts to slack off. 37
The follow-up assessment after 3 months revealed that the CON group exhibited a decline in motor function, indicating a slightly forgetting effect. 38 This result shows that even with a large amount of repetitive training, more than the improvement brought by a single exercise is needed. However, establishing grasping gestures with real-life small objects can enhance the HT group’s confidence in using their affected hand with daily tasks. After 3 months, the improved motor function ability to grasp objects in daily living could be maintained. The kinesthetic sense is derived from muscle forces used to lift objects. Fingertip load forces used to maintain a stable grip during tasks, the tactile sensation from touch receptors used to generate grip forces suitable for object texture, and visual input about the contours of objects for determining the appropriate hand shape during grasping all contribute to this improvement. These sensory inputs are integrated into the task-oriented training process, allowing patients to effectively sense errors to CON their affected hand again by touching and attempting to lift objects, subsequently relearning their motor behavior.
Motor Outcomes Evaluated by Electromyography Parameters
Regarding the EMG parameter measured by CI values, the HT group displayed a significant downward trend in all muscle pairs (Figure 3), indicating increased independent performance of the selected muscle pairs during different tasks. Moreover, notable group differences were observed in almost all the muscle pairs. During task-oriented training, the CI value decreased at twice the rate of the CON group. Previous studies have indicated that the CI of the FD-ED muscle pair in the paralyzed arm after stroke is higher compared to the healthy side during tasks. 39 Due to the substantial difference in cross-sectional areas between those muscles, significant activation of the ED muscles is required to counteract even a moderate increase in FD muscle activation. 40 The increased grip strength and reduced co-contraction in the FD-ED muscle pair for the HT group suggest a gradual separation of the firing phases of the antagonist muscle pair over the course of training sessions, and this separation might be predictive of the notable UE recovery during therapy. 41 The enhanced improvement in reducing abnormal compensation indicates effective recovery of impaired fingers, particularly in extension ability.
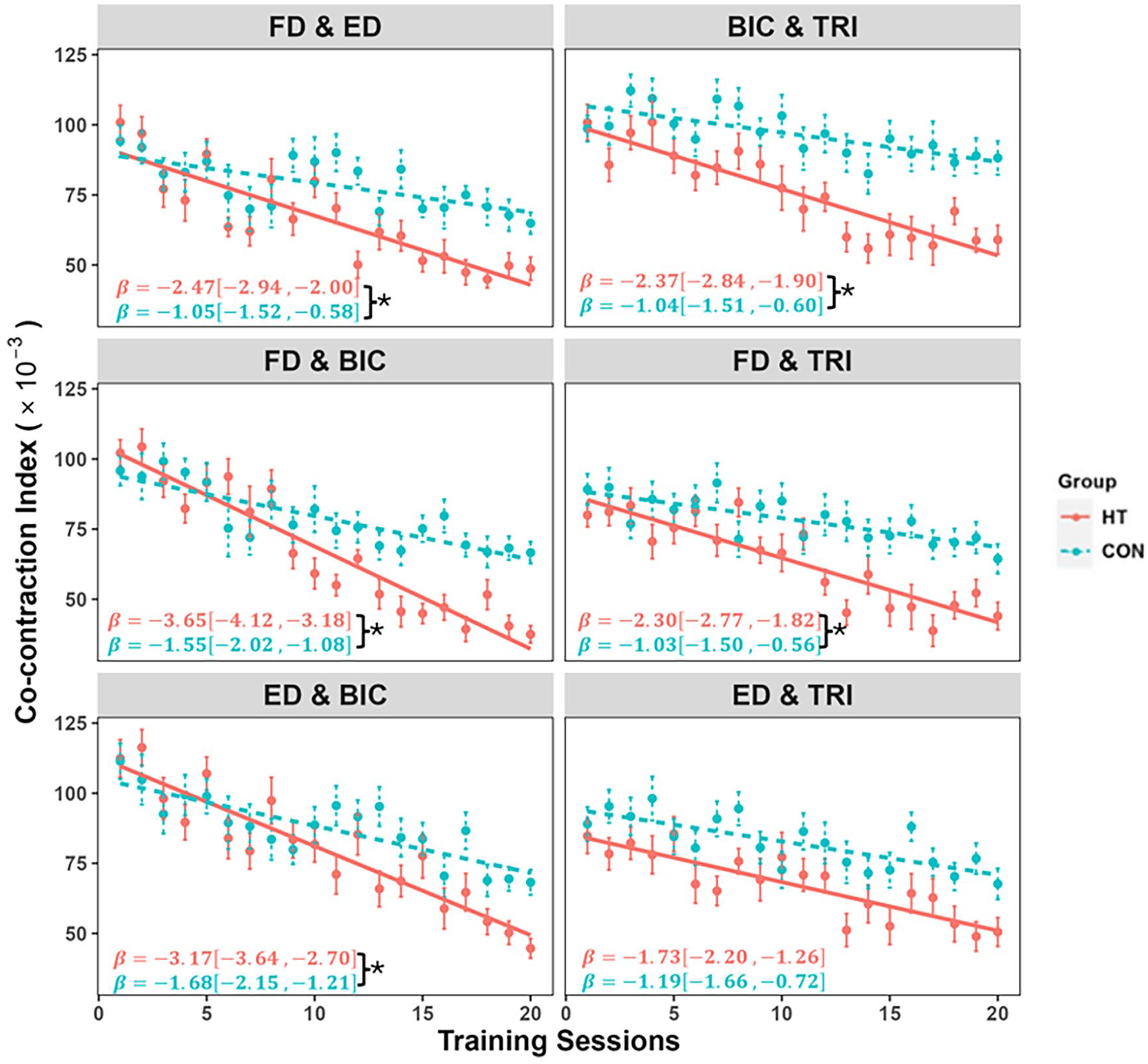
The variation in co-contraction indexes (CI). CI values throughout the session are represented by the mean and standard deviation of the muscle pairs (FD and ED, ED and BIC, FD and BIC, ED and TRI, FD and TRI, and BIC and TRI). The regression slope β was present with a 95% of confidence interval. The significant group difference is indicated by “*” (Independent t-tests, P < .05) on top of the error bar.
Additionally, the accelerated decline of CI between forearm and upper arm muscles in the HT group might indicate a reduced compensatory effect of proximal joints on distal joints. Stroke survivors with distal motor disabilities often exhibit compensatory movements, characterized by a significant involvement of the proximal limb in distal movements. 42 For instance, involuntary elbow flexion was frequently observed in the initial sessions, rendering most stroke participants in both groups unable to perform finger/wrist movements. However, following the complete training, participants in the HT group demonstrated improved arm CON and increased independence in performing the upper limb tasks. The decrease in finger MAS scores and improved AROM in the fingers after the intervention indicated effective activation of the distal limb among HT group participants. The consistent improvement in outcome measures further supports reducing the “learned non-use” phenomenon. Ballester et al 43 presented the theory of a bistable recovery process, wherein subjects’ performance either improves or deteriorates following the prescribed treatment. A virtuous cycle can reinforce arm-hand function if arm-hand use surpasses an adequate threshold. In the HT group, subjects could CON their finger function with robotic assistance.
Conversely, patients who do not utilize their limbs in daily activities may enter a vicious cycle that can reinforce arm-hand function. Task-oriented training utilizing personalized robotic hands offers a promising approach to reaching this effective threshold. Furthermore, it was observed that the forearm with BIC muscle pairs exhibited more pronounced decreases than others. These reductions indicate a decline in excessive muscle activity during evaluations. This may be attributed to the initial flexed arm state commonly observed in most stroke patients, as BIC demonstrates a more substantial synergistic effect with distal movements post-stroke.
Limitations
It is essential to note that wrist movement should have been more emphasized in the current training despite its significance and strong correlation with hand therapy. Future studies could consider integrating a soft wrist/hand robot to enable more comprehensive distal task-oriented training. This would allow for the inclusion of more complex tasks involving both the wrist and hand. Investigating variations in brain activity with fMRI or EEG during different tasks and examining the relationship between movement intention and brain activities would be valuable for future research. We suggest conducting further studies to evaluate the effectiveness of using this soft robotic hand in home-based environments after the stroke survivors have received training from therapists on how to train independently. Future studies should also consider including more participants with higher levels of finger joint spasticity.
Conclusions
This study investigated the effectiveness of task-oriented training with small objects assisted by a personalized EMG-driven soft robotic hand with task-oriented object manipulation for UE rehabilitation. It was demonstrated that task-oriented training significantly improved upper limb motor functions, particularly in the distal part. Task-oriented training with the personalized soft robotic hand can be considered a comfortable and effective form of exercise that can be integrated into stroke rehabilitation programs.
Footnotes
Author Contributions
Xiang-Qian Shi: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Validation; Visualization; Writing—original draft. Chun-Hang Eden Ti: Formal analysis; Software; Visualization; Writing—review & editing. Hsuan-Yu Lu: Investigation; Writing—review & editing. Cheng-Peng Hu: Methodology; Validation; Writing—review & editing. Di-Sheng Xie: Investigation; Writing—review & editing. Kai Yuan: Investigation; Writing—review & editing. Ho-Lam Heung: Conceptualization; Writing—review & editing. Thomas Wai-Hong Leung: Methodology; Writing—review & editing. Zheng Li: Conceptualization; Methodology; Supervision; Writing—review & editing. Raymond Kai-Yu Tong: Conceptualization; Funding acquisition; Project administration; Resources; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The university encourages knowledge transfer activities, and this robotic hand was licensed by the spin-off company (Hopebotics Limited, Hong Kong SAR, China) in 2021. The authors make every effort to ensure the impartiality, integrity, and scientific rigor of this study. The interpretation of the results and reporting of the findings were conducted objectively and without bias.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by an ITF grant (GHP/061/21GD) from the Innovation and Technology Commission of the Hong Kong Special Administrative Region, a GRF grant (CUHK 14216622) from the Research Grants Council, and a grant (Grant No. 2020B1515120064) from the Guangdong Science and Technology Research Council.